Photostabilization of Poly(vinyl chloride) Films Blended with Organotin Complexes of Mefenamic Acid for Outdoor Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. General
2.2. Synthesis of Triphenyltin Complex 1
2.3. Synthesis of Complexes 2–4
2.4. Preparation of PVC Films Containing Tin Complexes
3. Results and Discussion
3.1. Synthesis of Complexes 1–4
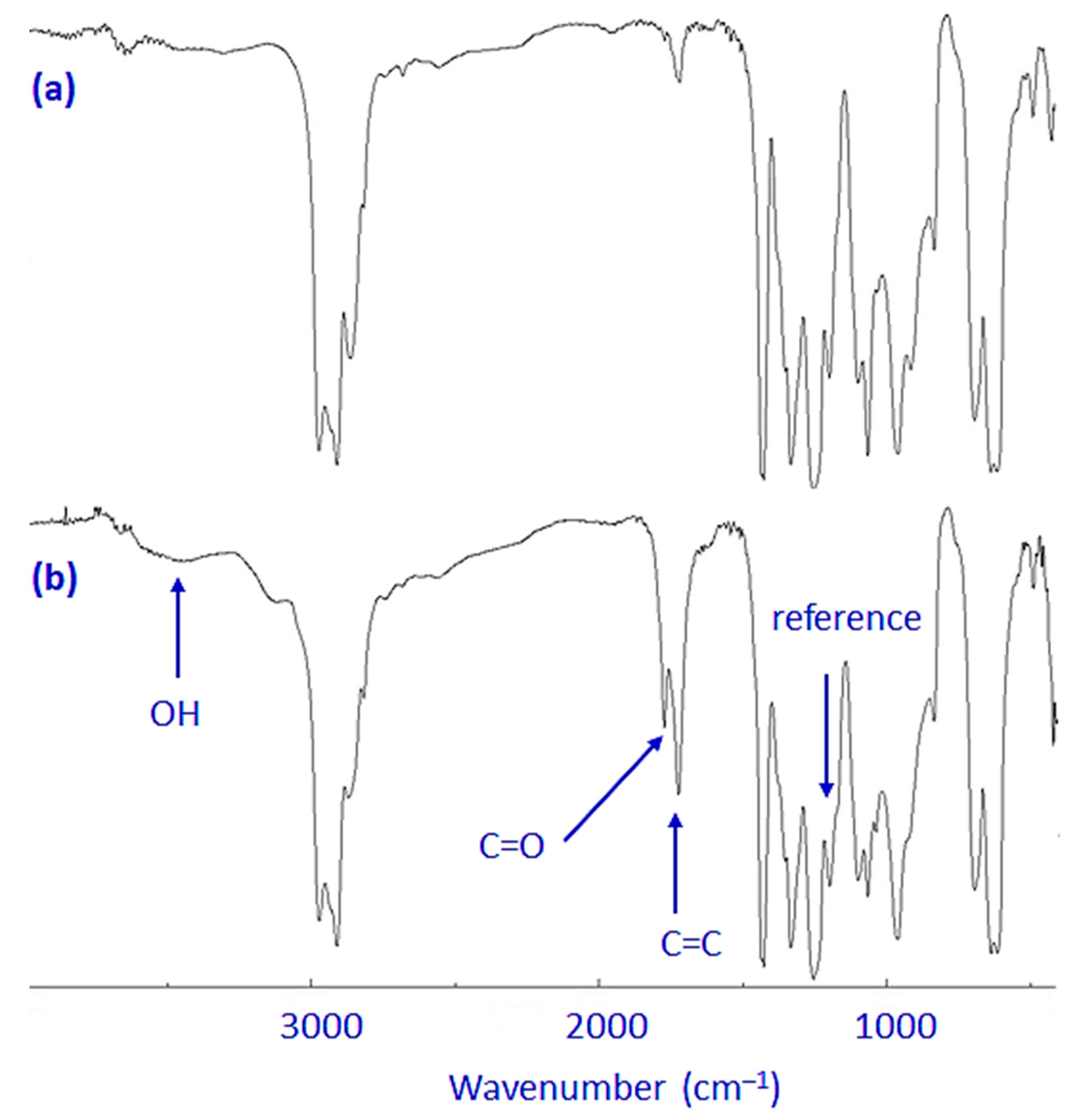
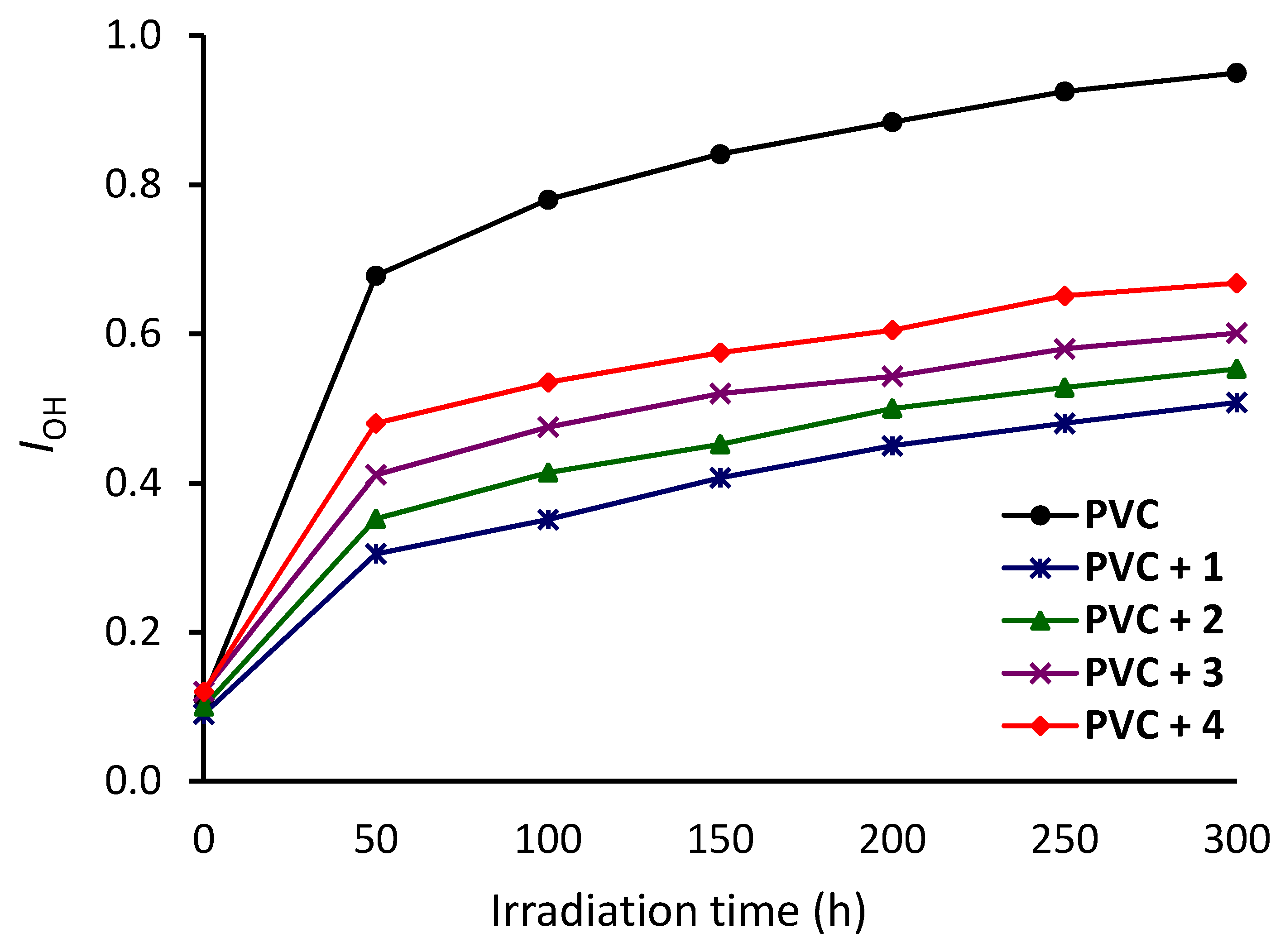
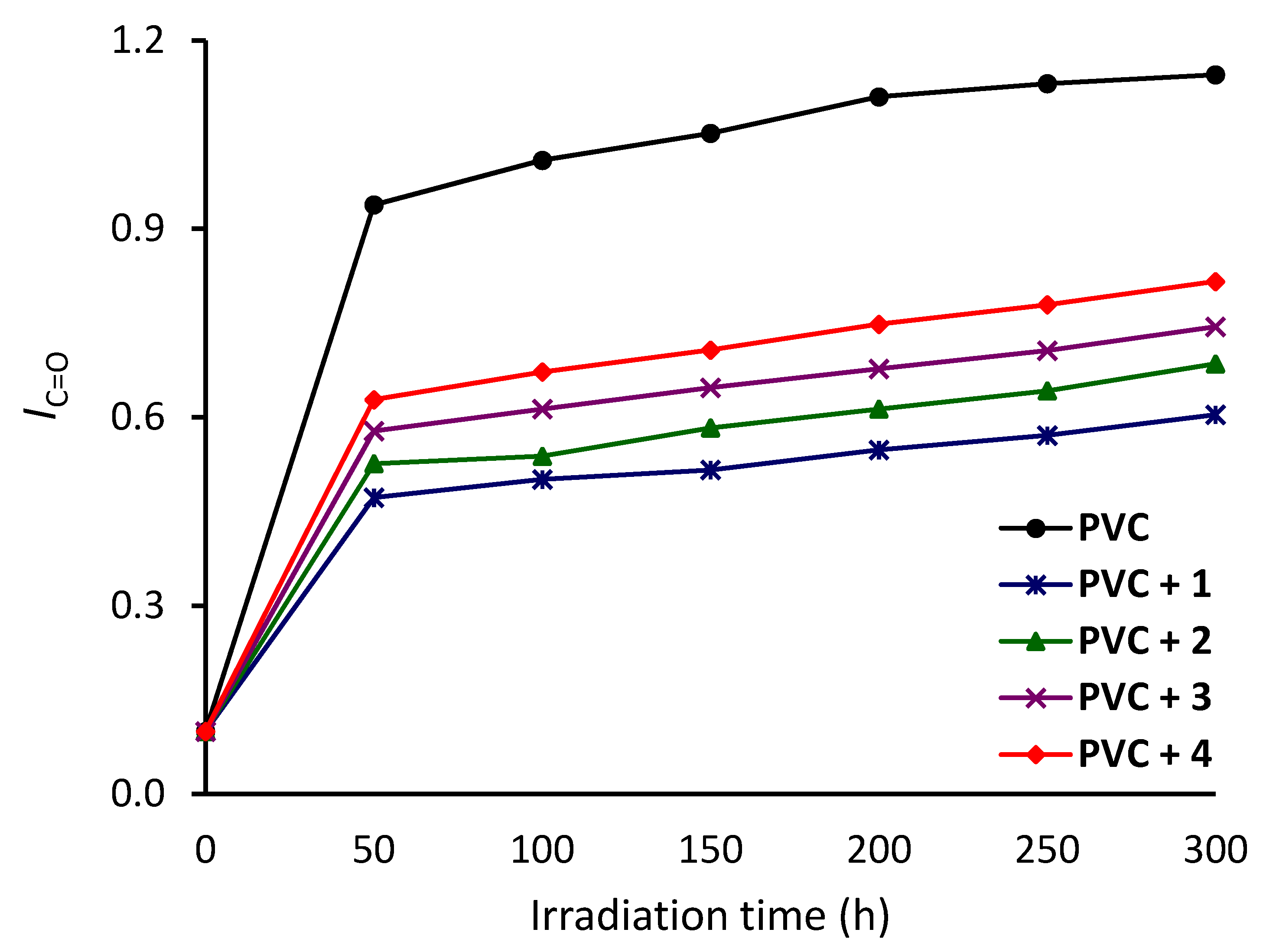
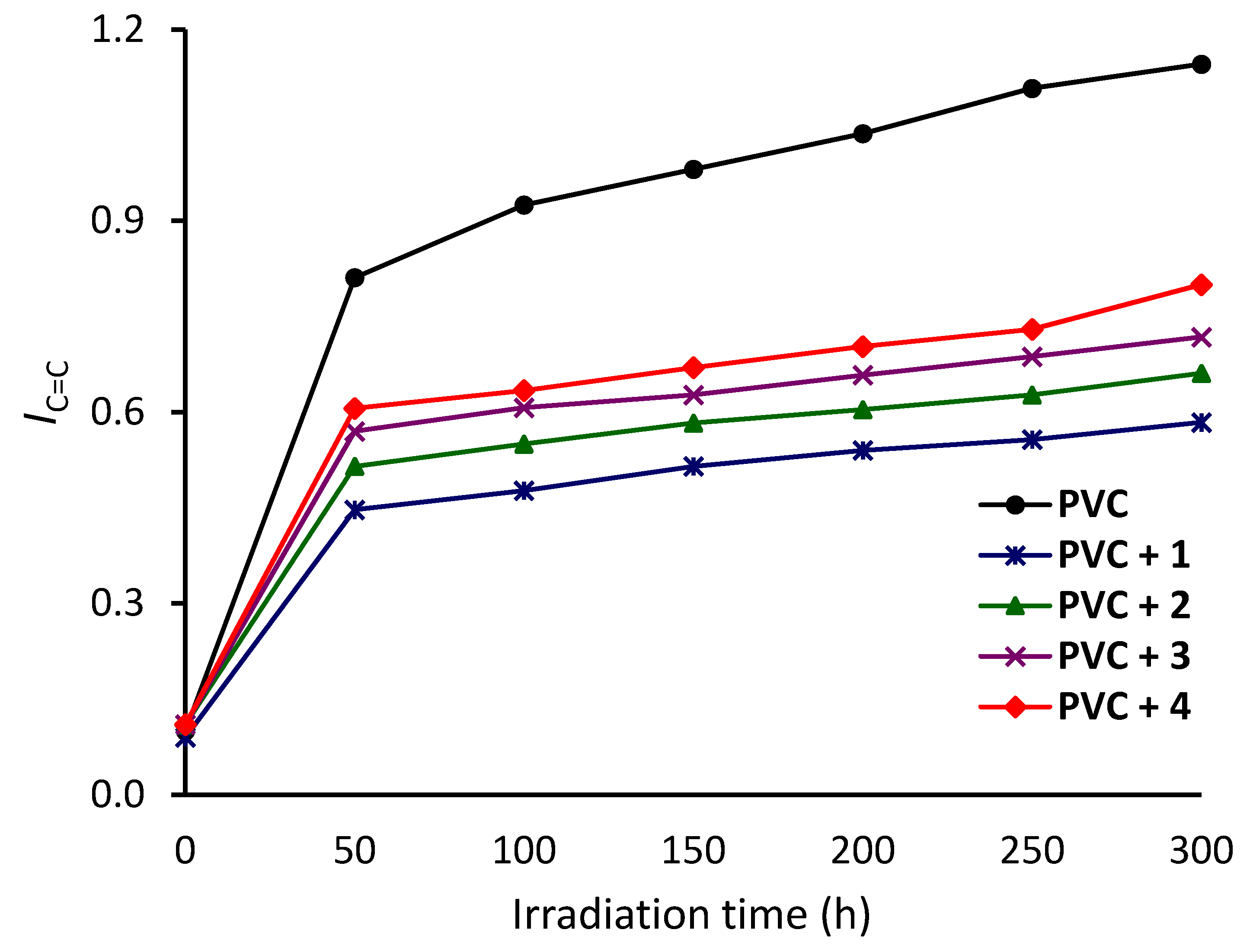
3.2. Investigation of PVC Photodegradation Using FTIR Spectroscopy
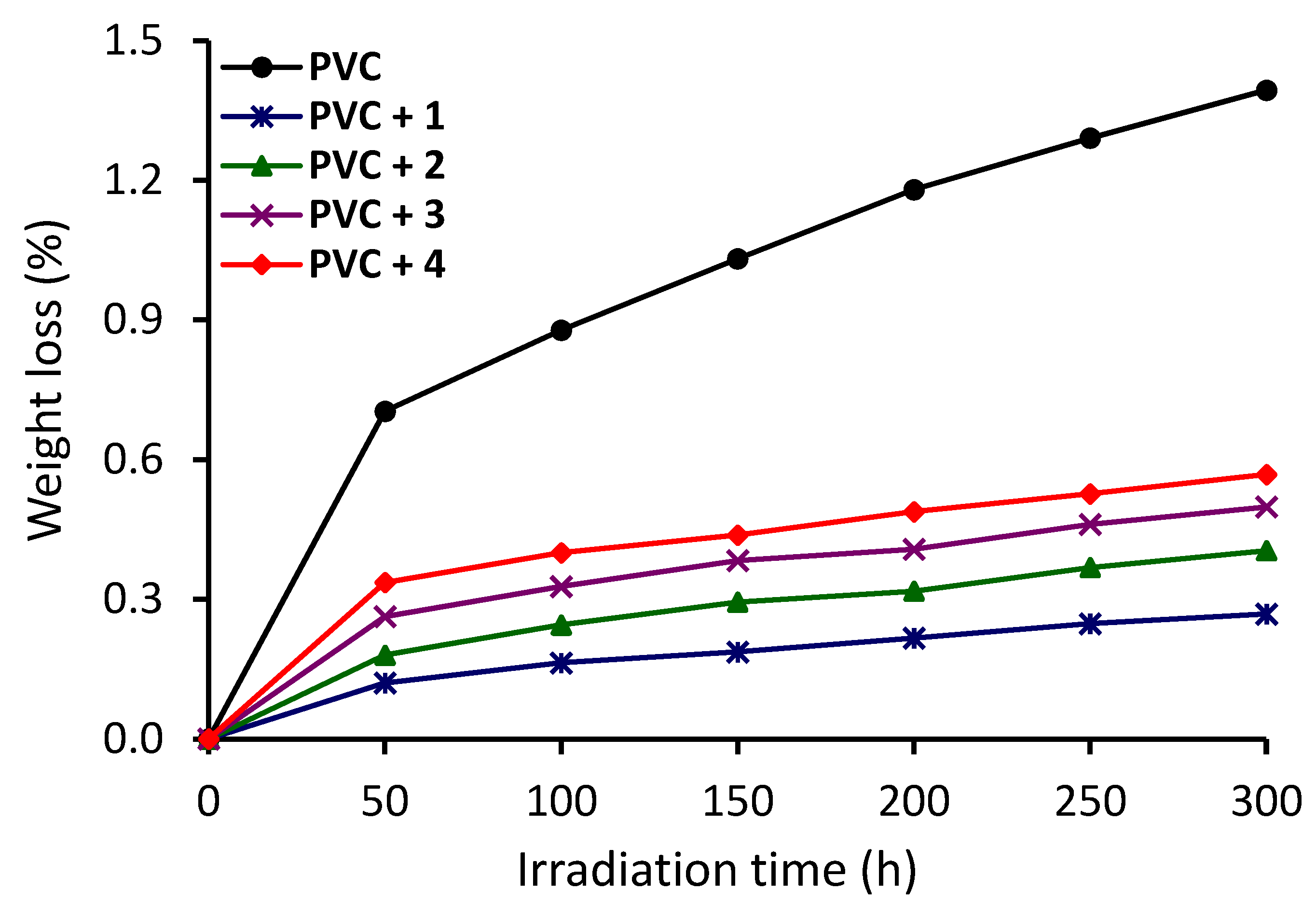
3.3. Investigation of PVC Photodegradation Using Weight Loss
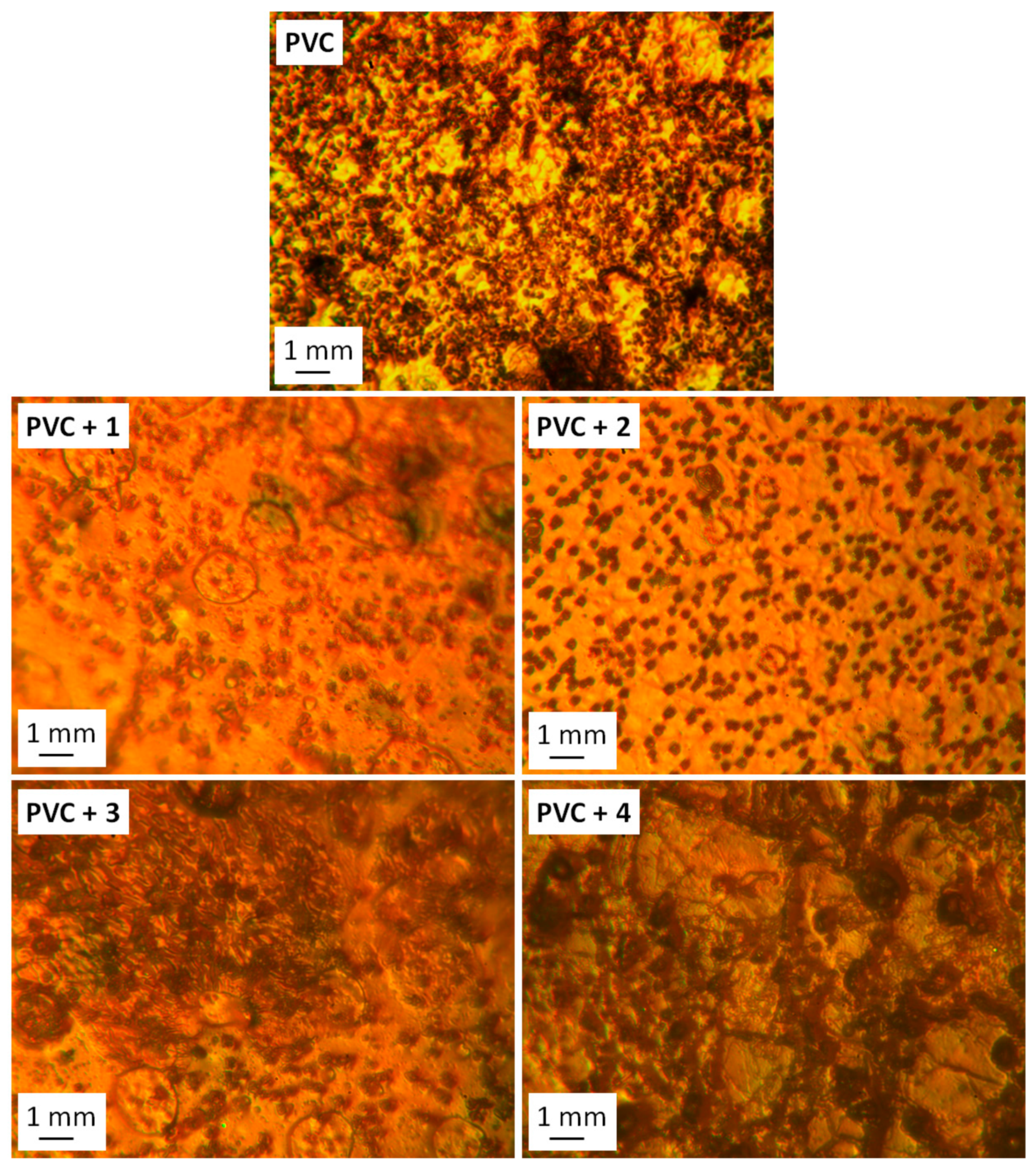
3.4. Investigation of PVC Photodegradation Using Surface Morphology
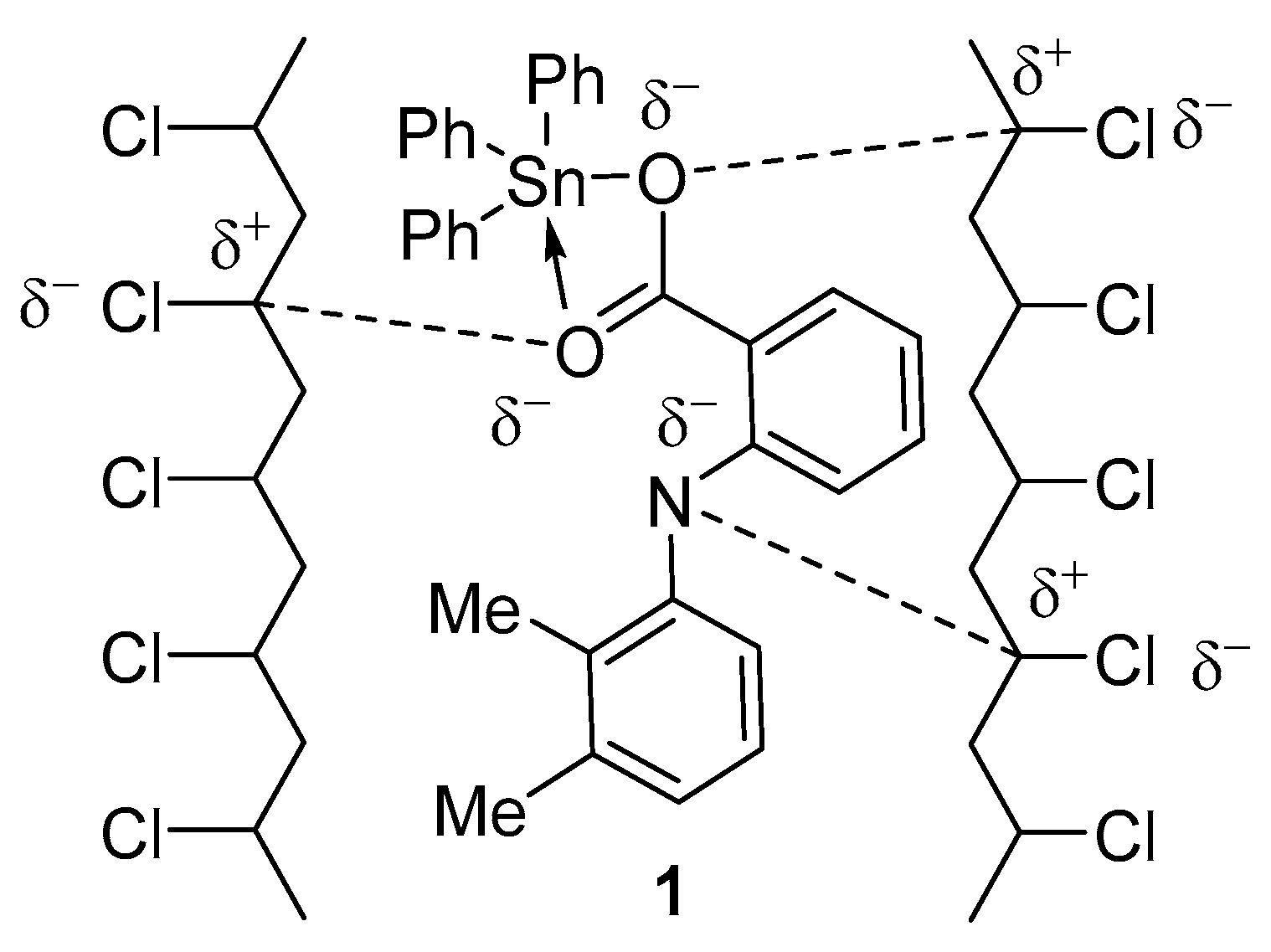
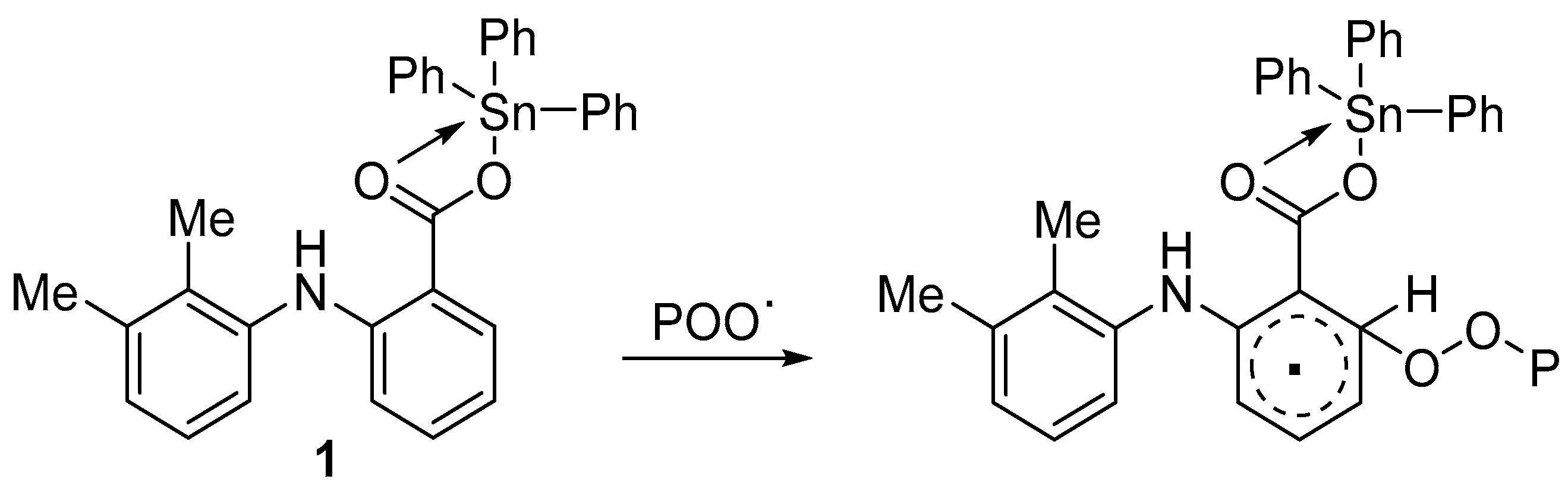
3.5. PVC Photostabilization Suggested Mechanisms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bodaghi, A. An overview on the recent developments in reactive plasticizers in polymers. Polym. Adv. Technol. 2020, 31, 355–367. [Google Scholar] [CrossRef]
- Feldman, D. Polymer history. Des. Monomers Polym. 2008, 11, 1–15. [Google Scholar] [CrossRef]
- Feldman, D. Polymer weathering: Photo-oxidation. J. Polym. Environ. 2002, 10, 163–173. [Google Scholar] [CrossRef]
- Mango, L.A.; Lenz, R.W. Hydrogenation of unsaturated polymers with diimide. Makromol. Chem. 1973, 163, 13–36. [Google Scholar] [CrossRef]
- Starnes, W.H. Structural defects in poly(vinyl chloride). J. Polym. Sci. Pol. Chem. 2005, 43, 2451–2467. [Google Scholar] [CrossRef]
- Cadogan, D.F.; Howick, C.J. Plasticizers. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar] [CrossRef]
- Porta, M.; Zumeta, E. Implementing the Stockholm treaty on persistent organic pollutants. Occup. Environ. Med. 2002, 59, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Grossman, R.F. Mixed metal vinyl stabilizer synergism. II: Reactions with zinc replacing cadmium. J. Vinyl Addit. Technol. 1990, 12, 142–145. [Google Scholar] [CrossRef]
- Tomi, I.H.R.; Ali, G.Q.; Jawad, A.H.; Yousef, E. Synthesis and characterization of gallic acid derivatives and their utilized as organic photo-stabilizers for poly(vinyl chloride). J. Polym. Res. 2017, 24, 119. [Google Scholar] [CrossRef]
- Balakit, A.A.; Ahmed, A.; El-Hiti, G.A.; Smith, K.; Yousif, E. Synthesis of new thiophene derivatives and their use as photostabilizers for rigid poly(vinyl chloride). Int. J. Polym. Sci. 2015, 2015, 510390. [Google Scholar] [CrossRef]
- Zhao, Y.; Dan, Y. Preparation and characterization of a high molecular weight UV-stabilizer based on a derivative of 2,4-dihydroxybenzophenone and its application in polymer materials. J. Appl. Polym. Sci. 2006, 102, 2203–2211. [Google Scholar] [CrossRef]
- Sabaa, M.W.; Oraby, E.H.; Abdel Naby, A.S.; Mohammed, R.R. Anthraquinone derivatives as organic stabilizers for rigid poly(vinyl chloride) against photo-degradation. Eur. Polym. J. 2005, 41, 2530–2543. [Google Scholar] [CrossRef]
- El-Hiti, G.A.; Ahmed, D.S.; Yousif, E.; Alotaibi, M.H.; Star, H.A.; Ahmed, A.A. Influence of polyphosphates on the physicochemical properties of poly(vinyl chloride) after irradiation with ultraviolet light. Polymers 2020, 12, 193. [Google Scholar] [CrossRef]
- Ahmed, D.S.; El-Hiti, G.A.; Yousif, E.; Hameed, A.S. Polyphosphates as inhibitors for poly(vinyl chloride) photodegradation. Molecules 2017, 22, 1849. [Google Scholar] [CrossRef]
- Alotaibi, M.H.; El-Hiti, G.A.; Hashim, H.; Hameed, A.S.; Ahmed, D.S.; Yousif, E. SEM analysis of the tunable honeycomb structure of irradiated poly(vinyl chloride) films doped with polyphosphate. Heliyon 2018, 4, e01013. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Ahmed, D.S.; El-Hiti, G.A.; Alotaibi, M.H.; Hashim, H.; Yousif, E. SEM morphological analysis of irradiated polystyrene film doped by a Schiff base containing a 1,2,4-triazole ring system. Appl. Petrochem. Res. 2019, 9, 169–177. [Google Scholar] [CrossRef]
- El-Hiti, G.A.; Alotaibi, M.H.; Ahmed, A.A.; Hamad, B.A.; Ahmed, D.S.; Ahmed, A.; Hashim, H.; Yousif, E. The morphology and performance of poly(vinyl chloride) containing melamine Schiff bases against ultraviolet light. Molecules 2019, 24, 803. [Google Scholar] [CrossRef]
- Shaalan, N.; Laftah, N.; El-Hiti, G.A.; Alotaibi, M.H.; Muslih, R.; Ahmed, D.S.; Yousif, E. Poly(vinyl chloride) photostabilization in the presence of Schiff bases containing a thiadiazole moiety. Molecules 2018, 23, 913. [Google Scholar] [CrossRef]
- Ahmed, D.S.; El-Hiti, G.A.; Hameed, A.S.; Yousif, E.; Ahmed, A. New tetra-Schiff bases as efficient photostabilizers for poly(vinyl chloride). Molecules 2017, 22, 1506. [Google Scholar] [CrossRef] [PubMed]
- Yousif, E.; Hasan, A.; El-Hiti, G.A. Spectroscopic, physical and topography of photochemical process of PVC films in the presence of Schiff base metal complexes. Polymers 2016, 8, 204. [Google Scholar] [CrossRef]
- Yousif, E.; El-Hiti, G.A.; Hussain, Z.; Altaie, A. Viscoelastic, spectroscopic and microscopic study of the photo irradiation effect on the stability of PVC in the presence of sulfamethoxazole Schiff’s bases. Polymers 2015, 7, 2190–2204. [Google Scholar] [CrossRef]
- Sterzyński, T.; Tomaszewska, J.; Piszczek, K.; Skórczewska, K. The influence of carbon nanotubes on the PVC glass transition temperature. Compos. Sci. Technol. 2010, 70, 966–969. [Google Scholar] [CrossRef]
- Mallakpour, S.; Sadeghzadeh, R. A Benign and simple strategy for surface modification of Al2O3 nanoparticles with citric acid and L(+)-ascorbic acid and its application for the preparation of novel poly(vinyl chloride) nanocomposite films. Adv. Polym. Technol. 2017, 36, 409–417. [Google Scholar] [CrossRef]
- Yang, T.C.T.; Noguchi, T.; Isshiki, M.; Wu, J.H. Effect of titanium dioxide particles on the surface morphology and the mechanical properties of PVC composites during QUV accelerated weathering. Polym. Compos. 2016, 37, 3391–3397. [Google Scholar] [CrossRef]
- Yang, T.C.; Noguchi, T.; Isshiki, M.; Wu, J.H. Effect of titanium dioxide on chemical and molecular changes in PVC sidings during QUV accelerated weathering. Polym. Degrad. Stab. 2014, 104, 33–39. [Google Scholar] [CrossRef]
- Cheng, Q.; Li, C.; Pavlinek, V.; Saha, P.; Wang, H. Surface-modified antibacterial TiO2/Ag+ nanoparticles: Preparation and properties. Appl. Surf. Sci. 2006, 252, 4154–4160. [Google Scholar] [CrossRef]
- Birmingham, J.N. The effect of surface oxidation and titanium dioxide on exterior PVC color retention. J. Vinyl Addit. Technol. 1995, 1, 84–87. [Google Scholar] [CrossRef]
- Mohamed, S.H.; Hameed, A.S.; El-Hiti, G.A.; Ahmed, D.S.; Kadhom, M.; Baashen, M.A.; Bufaroosha, M.; Ahmed, A.A.; Yousif, E. A process for the synthesis and use of highly aromatic organosilanes as additives for poly(vinyl chloride) films. Processes 2021, 9, 91. [Google Scholar] [CrossRef]
- Mohammed, R.; El-Hiti, G.A.; Ahmed, A.; Yousif, E. Poly(vinyl chloride) doped by 2-(4-isobutylphenyl)propanoate metal complexes: Enhanced resistance to UV irradiation. Arab. J. Sci. Eng. 2017, 42, 4307–4315. [Google Scholar] [CrossRef]
- Schiller, M. PVC Additives: Performance, Chemistry, Developments, and Sustainability; Carl Hanser Verlag: Munich, Germany, 2015. [Google Scholar]
- Devi, J.; Jyoti Yadav, J. Recent advancements in organotin(IV) complexes as potential anticancer agents. Anticancer Agents Med. Chem. 2018, 18, 335–353. [Google Scholar] [CrossRef]
- Sirajuddin, M.; Ali, S. Organotin(IV) carboxylates as promising potential drug candidates in the field of cancer chemotherapy. Curr. Pharm. Des. 2016, 22, 6665–6681. [Google Scholar] [CrossRef]
- Shah, S.S.A.; Ashfaq, M.; Waseem, A.; Ahmed, M.M.; Najam, T.; Shaheen, S.; Rivera, G. Synthesis and biological activities of organotin(IV) complexes as antitumoral and antimicrobial agents. A review. Mini Rev. Med. Chem. 2015, 15, 406–426. [Google Scholar] [CrossRef] [PubMed]
- Romero-Chávez, M.M.; Pineda-Urbina, K.; Pérez, D.J.; Obledo-Benicio, F.; Flores-Parra, A.; Gómez-Sandoval, Z.; Ramos-Organillo, Á. Organotin(IV) compounds derived from ibuprofen and cinnamic acids, an alternative into design of anti-inflammatory by the cyclooxygenases (COX-1 and COX-2) pathway. J. Organometal. Chem. 2018, 862, 58–70. [Google Scholar] [CrossRef]
- Iqbal, H.; Ali, S.; Shahzadi, S. Antituberculosis study of organotin(IV) complexes: A review. Cogent Chem. 2015, 1, 1029039. [Google Scholar] [CrossRef]
- Davies, A.G. Organotin Chemistry, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Araújo da Silva, M.; Silva dos Santos, A.S.; Veríssimo dos Santos, T.; Meneghetti, M.R.; Meneghetti, S.M.P. Organotin(IV) compounds with high catalytic activities and selectivities in the glycerolysis of triacylglycerides. Catal. Sci. Technol. 2017, 7, 5750–5757. [Google Scholar] [CrossRef]
- Ghazi, D.; Rasheed, Z.; Yousif, E. A review of organotin compounds: Chemistry and applications. Arch. Org. Inorg. Chem. Sci. 2018, 3, 344–352. [Google Scholar] [CrossRef]
- Mousa, O.G.; El-Hiti, G.A.; Baashen, M.A.; Bufaroosha, M.; Ahmed, A.; Ahmed, A.A.; Ahmed, D.S.; Emad Yousif, E. Synthesis of carvedilol-organotin complexes and their effects on reducing photodegradation of poly(vinyl chloride). Polymers 2021, 13, 500. [Google Scholar] [CrossRef]
- Salam, B.; El-Hiti, G.A.; Bufaroosha, M.; Ahmed, D.S.; Ahmed, A.; Alotaibi, M.H.; Yousif, E. Tin complexes containing an atenolol moiety as photostabilizers for poly(vinyl chloride). Polymers 2020, 12, 2923. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, Z.N.; Yousif, E.; Alias, M.; El-Hiti, G.A.; Ahmed, D.S. Synthesis, characterization, properties, and use of new fusidate organotin complexes as additives to inhibit poly(vinyl chloride) photodegradation. J. Polym. Res. 2020, 27, 267. [Google Scholar] [CrossRef]
- Ahmed, D.S.; El-Hiti, G.A.; Ibraheem, H.; Alotaibi, M.H.; Abdallh, M.; Ahmed, A.A.; Ismael, M.; Yousif, E. Enhancement of photostabilization of poly(vinyl chloride) doped with sulfadiazine tin complexes. J. Vinyl Additive Technol. 2020, 25, 370–379. [Google Scholar] [CrossRef]
- Mohammed, A.; El-Hiti, G.A.; Yousif, E.; Ahmed, A.A.; Ahmed, D.S.; Alotaibi, M.H. Protection of poly(vinyl chloride) films against photodegradation using various valsartan tin complexes. Polymers 2020, 12, 969. [Google Scholar] [CrossRef]
- Hadi, A.G.; Jawad, K.; El-Hiti, G.A.; Alotaibi, M.H.; Ahmed, A.A.; Ahmed, D.S.; Yousif, E. Photostabilization of poly(vinyl chloride) by organotin(IV) compounds against photodegradation. Molecules 2019, 24, 3557. [Google Scholar] [CrossRef] [PubMed]
- Hadi, A.G.; Yousif, E.; El-Hiti, G.A.; Ahmed, D.S.; Jawad, K.; Alotaibi, M.H.; Hashim, H. Long-term effect of ultraviolet irradiation on poly(vinyl chloride) films containing naproxen diorganotin(IV) complexes. Molecules 2019, 24, 2396. [Google Scholar] [CrossRef]
- Ghazi, D.; El-Hiti, G.A.; Yousif, E.; Ahmed, D.S.; Alotaibi, M.H. The effect of ultraviolet irradiation on the physicochemical properties of poly(vinyl chloride) films containing organotin(IV) complexes as photostabilizers. Molecules 2018, 23, 254. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.M.; El-Hiti, G.A.; Yousif, E. Photostabilizing efficiency of poly(vinyl chloride) in the presence of organotin(IV) complexes as photostabilizers. Molecules 2016, 21, 1151. [Google Scholar] [CrossRef]
- Tarafder, M.T.H.; Chew, K.-B.; Crouse, K.A.; Ali, M.A.; Yamin, B.M.; Fun, H.-K. Synthesis and characterization of Cu(II), Ni(II) and Zn(II) metal complexes of bidentate NS isomeric Schiff bases derived from S-methyldithiocarbazate (SMDTC): Bioactivity of the bidentate NS isomeric Schiff bases, some of their Cu(II), Ni(II) and Zn(II) complexes and the X-ray structure of the bis[S-methyl-β-N-(2-furyl-methyl)methylenedithiocarbazato]zinc(II) complex. Polyhedron 2002, 21, 2683–2690. [Google Scholar] [CrossRef]
- Morozov, O.S.; Vyshinskii, N.N.; Rudnevskii, N.K. Investigation of some organotin compounds and their complexes by IR spectroscopy. J. Appl. Spectrosc. 1981, 35, 1019–1023. [Google Scholar] [CrossRef]
- Gacki, M.; Wolf, W.M. Synthesis, spectroscopic, and thermal investigations of metal complexes with mefenamic acid. J. Chem. 2017, 2017, 6172626. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds; John Wiley & Sons, Inc.: New York, NY, USA, 2009. [Google Scholar]
- Alcock, N.W.; Culver, J.; Roe, S.M. Secondary bonding. Part 15. Influence of lone pairs on coordination: Comparison of diphenyl-tin(IV) and –tellurium(IV) carboxylates and dithiocarbamates. J. Chem. Soc. Dalton Trans. 1992, 1477–1484. [Google Scholar] [CrossRef]
- Gardette, J.L.; Gaumet, S.; Lemaire, J. Photooxidation of poly(vinyl chloride). 1. A reexamination of the mechanism. Macromolecules 1989, 22, 2576–2581. [Google Scholar] [CrossRef]
- Gaumet, S.; Gardette, J.-L. Photo-oxidation of poly(vinyl chloride): Part 2–A comparative study of the carbonylated products in photo-chemical and thermal oxidations. Polym. Degrad. Stab. 1991, 33, 17–34. [Google Scholar] [CrossRef]
- Almond, J.; Sugumaar, P.; Wenzel, M.N.; Hill, G.; Wallis, C. Determination of the carbonyl index of polyethylene and polypropylene using specified area under band methodology with ATR-FTIR spectroscopy. e-Polymers 2020, 20, 369–381. [Google Scholar] [CrossRef]
- Nief, O.A. Photostabilization of polyvinyl chloride by some new thiadiazole derivatives. Eur. J. Chem. 2015, 6, 242–247. [Google Scholar] [CrossRef]
- Jafari, A.J.; Donaldson, J.D. Determination of HCl and VOC emission from thermal degradation of PVC in the absence and presence of copper, copper(II) oxide and copper(II) chloride. Eur. J. Chem. 2009, 6, 685–692. [Google Scholar] [CrossRef]
- Valko, L.; Klein, E.; Kovařík, P.; Bleha, T.; Šimon, P. Kinetic study of thermal dehydrochlorination of poly(vinyl chloride) in the presence of oxygen: III. Statistical thermodynamic interpretation of the oxygen catalytic activity. Eur. Polym. J. 2001, 37, 1123–1132. [Google Scholar] [CrossRef]
- Nikafshar, S.; Zabihi, O.; Ahmadi, M.; Mirmohseni, A.; Taseidifar, M.; Naebe, M. The effect of UV light on the chemical and mechanical properties of a transparent epoxy-diamine system in the presence of an organic UV absorber. Materials 2017, 10, 180. [Google Scholar] [CrossRef]
- Kayyarapu, B.; Kumar, M.Y.; Mohommad, H.B.; Neeruganti, G.O.; Chekuri, R. Structural, thermal and optical properties of pure and Mn2+ doped poly(vinyl chloride) films. Mater. Res. 2016, 19, 1167–1175. [Google Scholar] [CrossRef]
- Ma, Q.; Ma, S.; Bai, J.; Wang, H. Influence of lead-free glass frit in the front contact paste on the conversion efficiency of polycrystalline silicon solar cells. RSC Adv. 2017, 7, 47500–47506. [Google Scholar] [CrossRef]
- Hashim, H.; El-Hiti, G.A.; Alotaibi, M.H.; Ahmed, D.S.; Yousif, E. Fabrication of ordered honeycomb porous poly(vinyl chloride) thin film doped with a Schiff base and nickel(II) chloride. Heliyon 2018, 4, e00743. [Google Scholar] [CrossRef] [PubMed]
- Folarin, O.M.; Sadiku, E.R. Thermal stabilizers for poly(vinyl chloride): A review. Int. J. Phys. Sci. 2011, 6, 4323–4330. [Google Scholar] [CrossRef]
- Pospíšil, J.; Klemchuk, P.P. Oxidation Inhibition in Organic Materials; CRC Press: Boca Raton, FL, USA, 1989; Volume 1, pp. 48–49. [Google Scholar]
- Xiang, X.; Chen, S.; Chai, R. Photodegradation of plasticized poly(vinyl chloride) stabilized by different types of thermal stabilizers. Polym. Eng. Sci. 2011, 51, 624–631. [Google Scholar] [CrossRef]

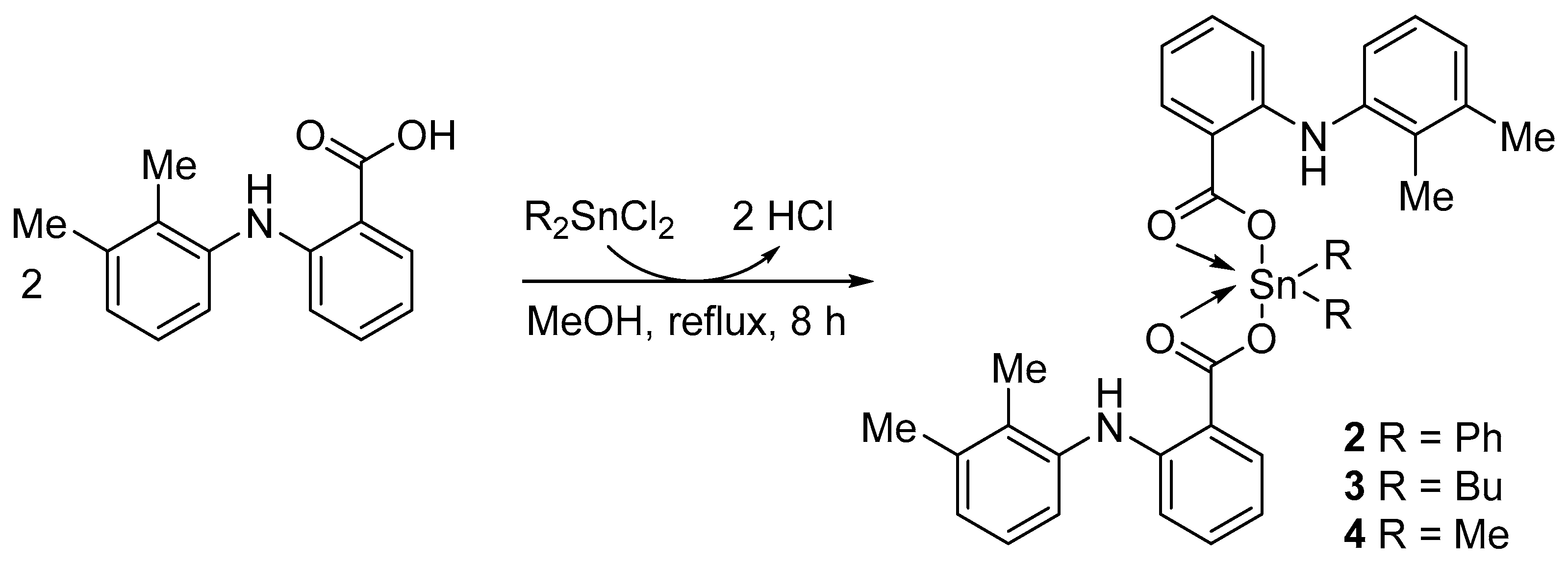



| Complex | R | Yield (%) | MP (°C) | Elemental Analysis (%) Calculated (Found) | ||
|---|---|---|---|---|---|---|
| C | H | N | ||||
| 1 | – | 70 | 123–125 | 67.14 (67.22) | 4.95 (5.05) | 2.37 (2.46) |
| 2 | Ph | 72 | 134–136 | 66.95 (66.98) | 5.08 (5.15) | 3.72 (3.77) |
| 3 | Bu | 71 | 118–120 | 63.97 (64.02) | 6.50 (6.23) | 3.93 (3.95) |
| 4 | Me | 77 | 213–215 | 61.07 (61.14) | 5.45 (5.55) | 4.45 (4.50) |
| Complex | FTIR (υ, cm–1) | ||||||
|---|---|---|---|---|---|---|---|
| NH | C=O | C=C | Sn–C | Sn–O | |||
| asym | sym | ∆v (asym − sym) | |||||
| 1 | 3312 | 1643 | 1508 | 135 | 1452 | 525 | 449 |
| 2 | 3312 | 1641 | 1508 | 133 | 1450 | 520 | 448 |
| 3 | 3333 | 1643 | 1506 | 137 | 1456 | 526 | 447 |
| 4 | 3312 | 1643 | 1506 | 137 | 1454 | 524 | 447 |
| Complex | 1H (δ, ppm, Hz) |
|---|---|
| 1 | 9.46 (s, 1H, NH), 7.90–7.84 (m, 3H, Ar), 7.44–7.41 (m, 6H, Ar), 7.33–7.29 (m, 9H, Ar), 7.13–6.67 (m, 4H, Ar), 2.50 (s, 3H, Me), 2.09 (s, 3H, Me) |
| 2 | 9.45 (s, 2H, 2NH), 7.89–7.80 (m, 6H, Ar), 7.36–7.10 (m, 10H, Ar), 7.03–6.67 (m, 6H, Ar), 2.50 (s, 6H, 2Me), 2.11 (s, 6H, 2Me) |
| 3 | 9.77 (s, 2H, 2NH), 7.89–7.85 (m, 6H, Ar), 7.46–6.96 (m, 8H, Ar), 2.50 (s, 6H, 2Me), 2.04 (s, 6H, 2Me), 1.64–1.29 (m, 12H, 6CH2), 0.81 (t, J 7.6 Hz, 6H, 2Me) |
| 4 | 9.45 (s, 2H, 2NH), 7.89–7.84 (m, 6H, Ar), 7.32–6.98 (m, 8H, Ar), 2.50 (s, 6H, 2Me), 2.08 (s, 6H, 2Me), 0.78 (s, 6H, 2Me) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, A.; El-Hiti, G.A.; Hadi, A.G.; Ahmed, D.S.; Baashen, M.A.; Hashim, H.; Yousif, E. Photostabilization of Poly(vinyl chloride) Films Blended with Organotin Complexes of Mefenamic Acid for Outdoor Applications. Appl. Sci. 2021, 11, 2853. https://doi.org/10.3390/app11062853
Ahmed A, El-Hiti GA, Hadi AG, Ahmed DS, Baashen MA, Hashim H, Yousif E. Photostabilization of Poly(vinyl chloride) Films Blended with Organotin Complexes of Mefenamic Acid for Outdoor Applications. Applied Sciences. 2021; 11(6):2853. https://doi.org/10.3390/app11062853
Chicago/Turabian StyleAhmed, Ahmed, Gamal A. El-Hiti, Angham G. Hadi, Dina S. Ahmed, Mohammed A. Baashen, Hassan Hashim, and Emad Yousif. 2021. "Photostabilization of Poly(vinyl chloride) Films Blended with Organotin Complexes of Mefenamic Acid for Outdoor Applications" Applied Sciences 11, no. 6: 2853. https://doi.org/10.3390/app11062853
APA StyleAhmed, A., El-Hiti, G. A., Hadi, A. G., Ahmed, D. S., Baashen, M. A., Hashim, H., & Yousif, E. (2021). Photostabilization of Poly(vinyl chloride) Films Blended with Organotin Complexes of Mefenamic Acid for Outdoor Applications. Applied Sciences, 11(6), 2853. https://doi.org/10.3390/app11062853









