A Review of Experimental and Numerical Studies of Lithium Ion Battery Fires
Abstract
1. Introduction
2. Method
3. LIB Chemistry
3.1. Cathode and Anode
3.2. Electrolyte
3.3. Separator
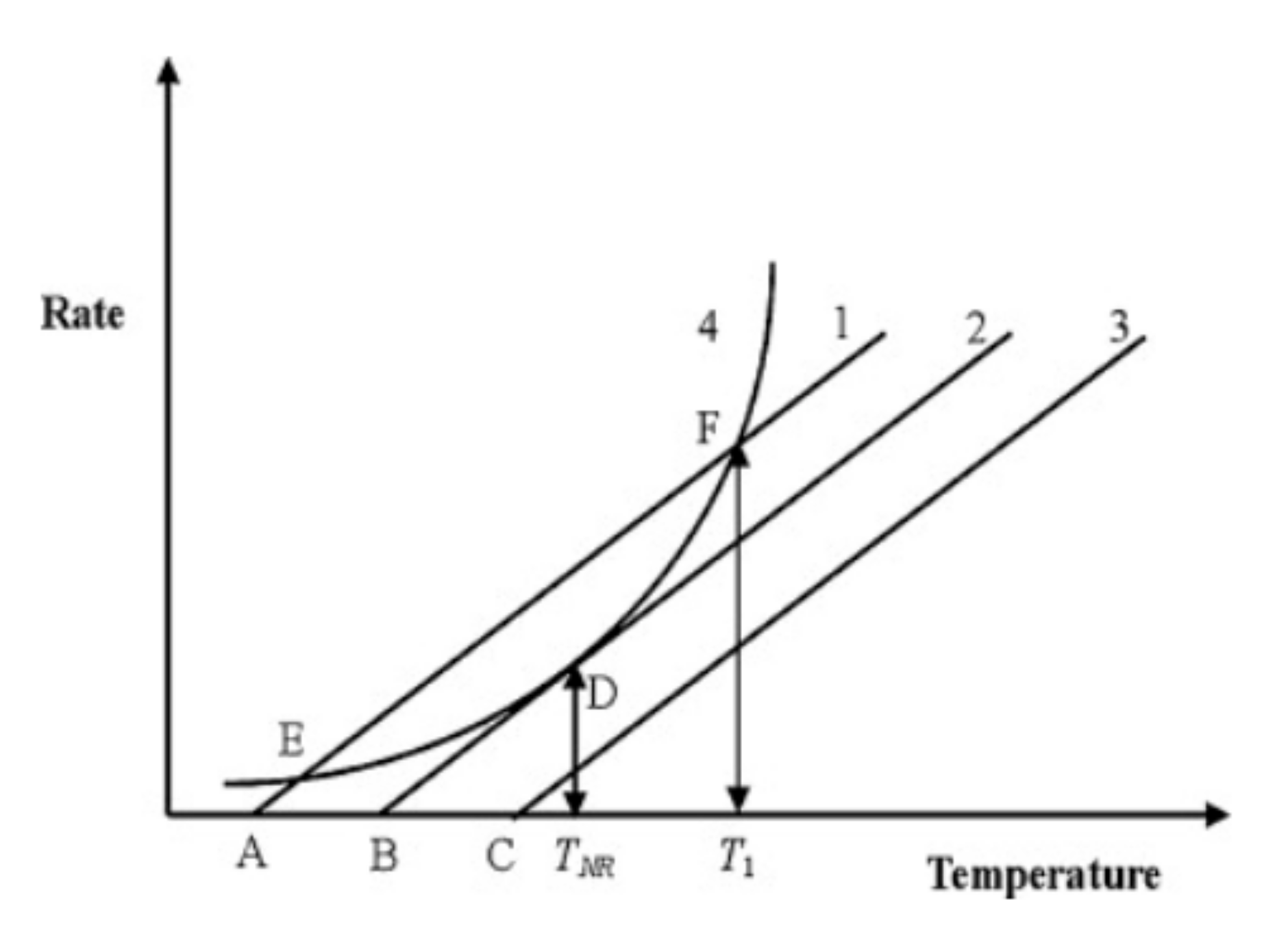
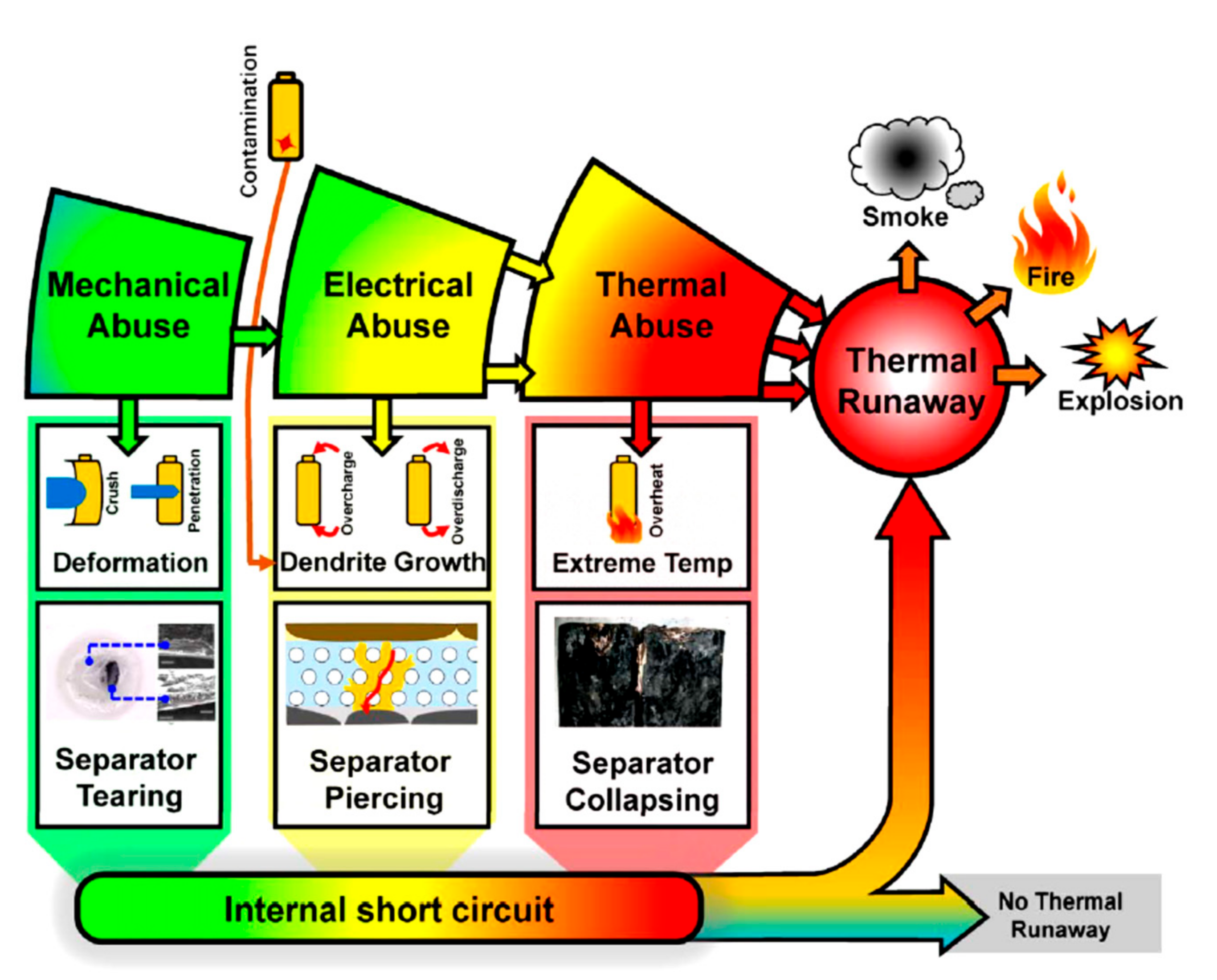
4. Thermal Runaway
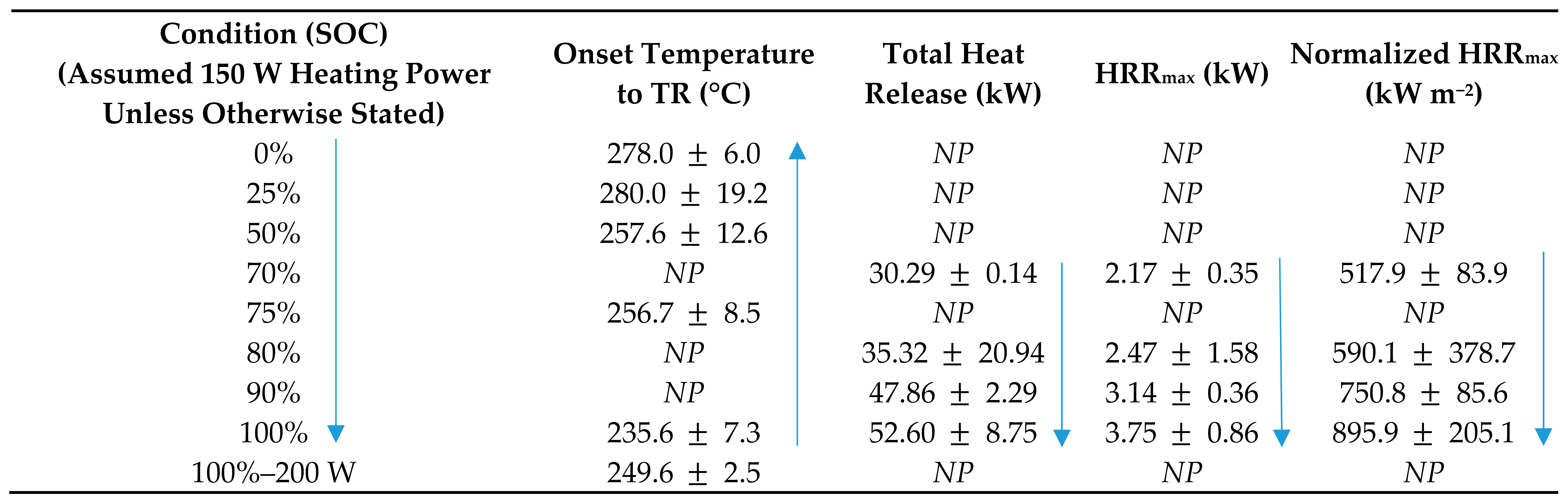
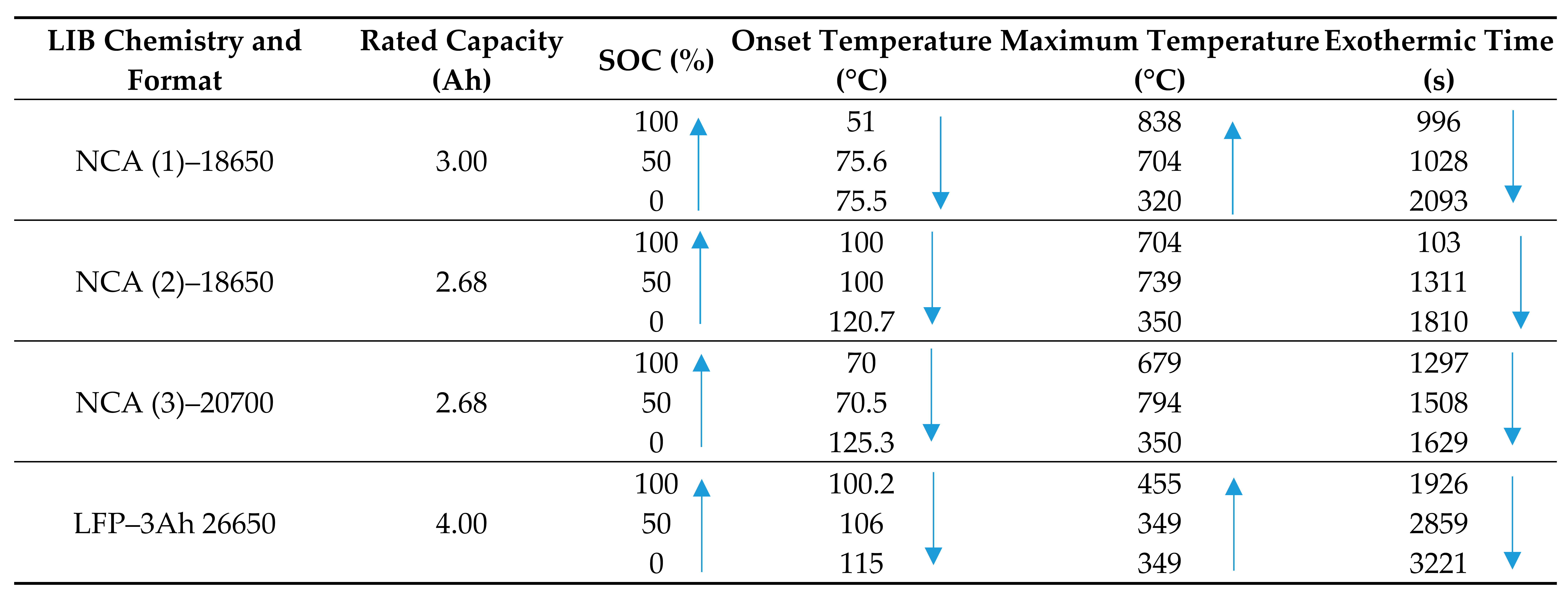
4.1. State of Charge and Its Effect on Thermal Runaway
- Batteries at higher SOC have larger amounts of stored energy leading to more explosive and faster reactions thereby limiting the amount of oxygen consumption, and subsequently incomplete combustion of the LIB;
- Onset of TR occurs earlier for batteries at a higher SOC, resulting in insufficient time to allow chemical reactions to release thermal energy;
- The oxygen (or part of) that is released from the internal electrochemical reactions is consumed in the combustion process, leading to HRR prediction errors based on the oxygen consumption method.
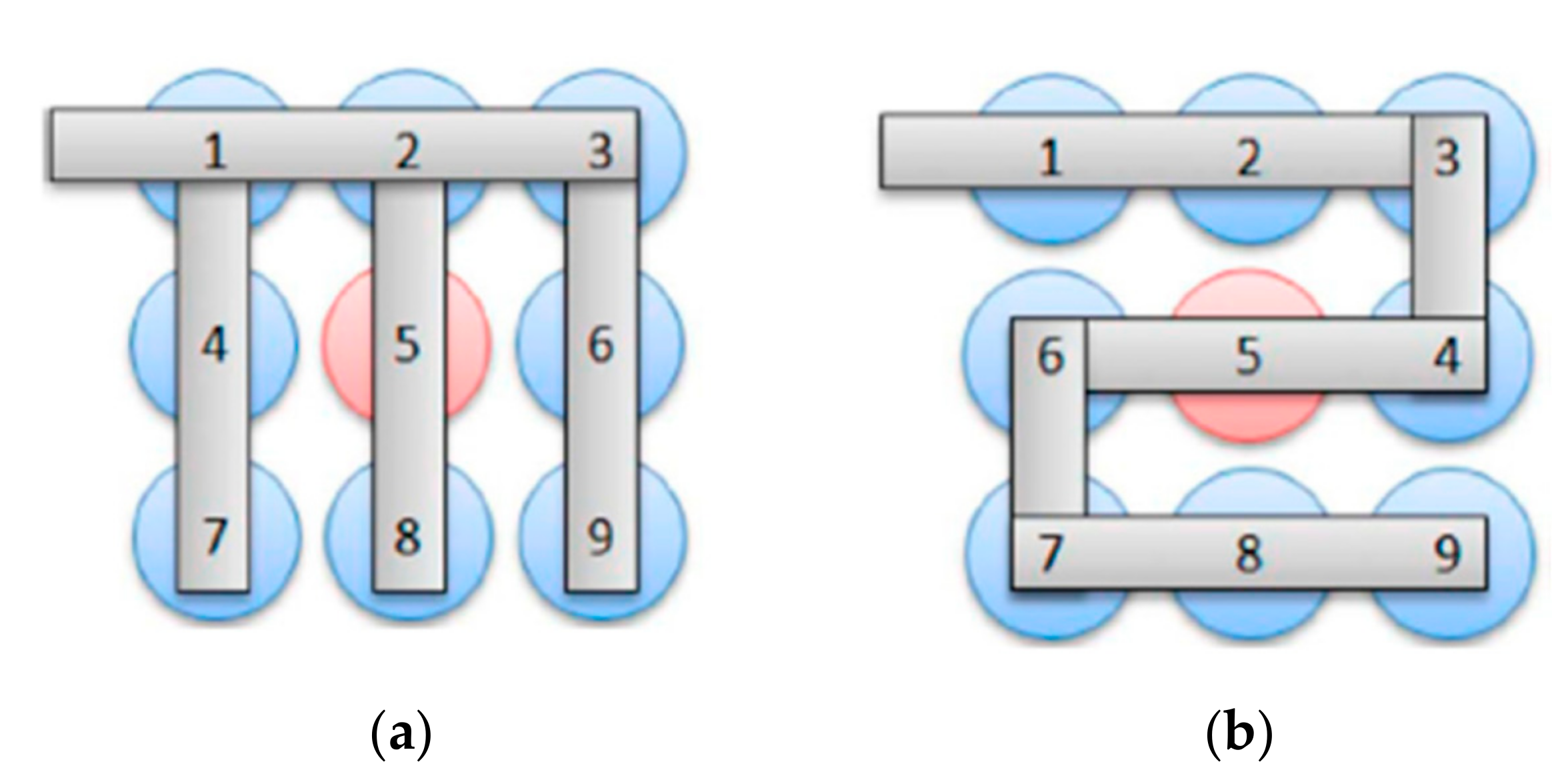
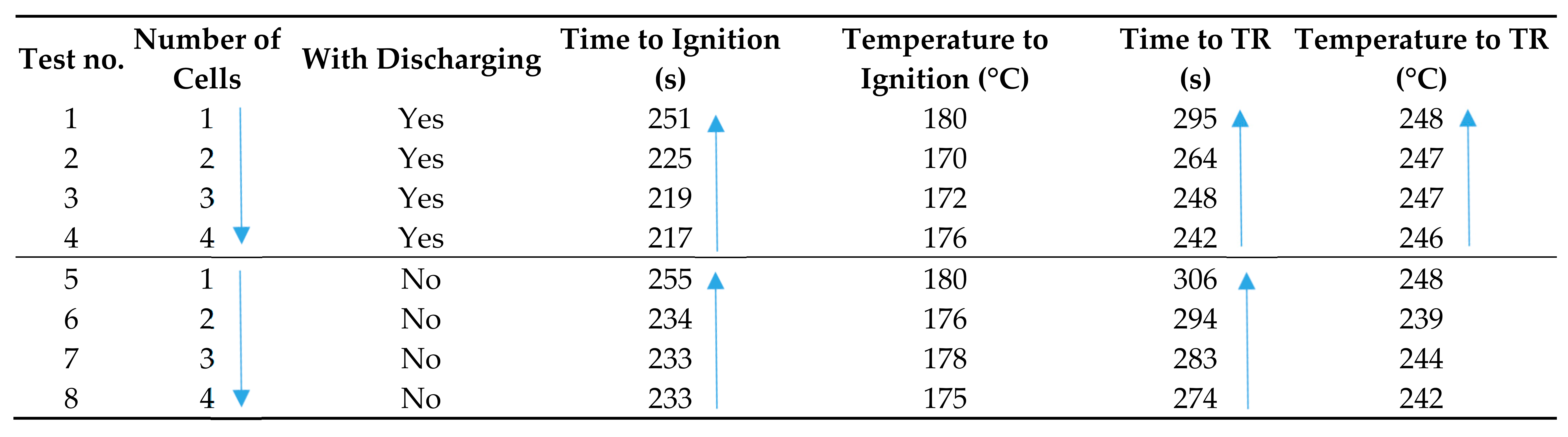
4.2. Thermal Runaway in Multiple Cells
4.3. Mitigating the Risk of Thermal Runaway and Fire
4.3.1. Inherent Safety
- Good chemical stability, no chemical reaction with battery components;
- Electrochemical inertia, no adverse electrochemical reactions within the normal operating voltage range of the LIB;
- Suitable physical properties including conductivity, viscosity, boiling point, density, solubility, etc.;
- Low toxicity, good machinability, and appropriate cost.
- Brominated compound additives [106];
- Fire resistance;
- Mechanical properties;
- Thermomechanical stability;
- Ion-transport resistance.
4.3.2. Safety Devices
4.3.3. Battery Management System
- Internal management system (IMS)—direct current (DC)→ alternating current (AC) method and component optimization;
- External management system (EMS)—air cooling system (ACS), liquid cooling system (LCS), and phase-change material-based cooling system (PCM-CS).
4.3.4. Active Fire Protection Systems
5. Heat Release Rate
5.1. HRR Measurement Methodologies
5.2. HRR and TTI Measured Data for 18650 Batteries
5.3. HRR Data Other than Cone Calorimeter
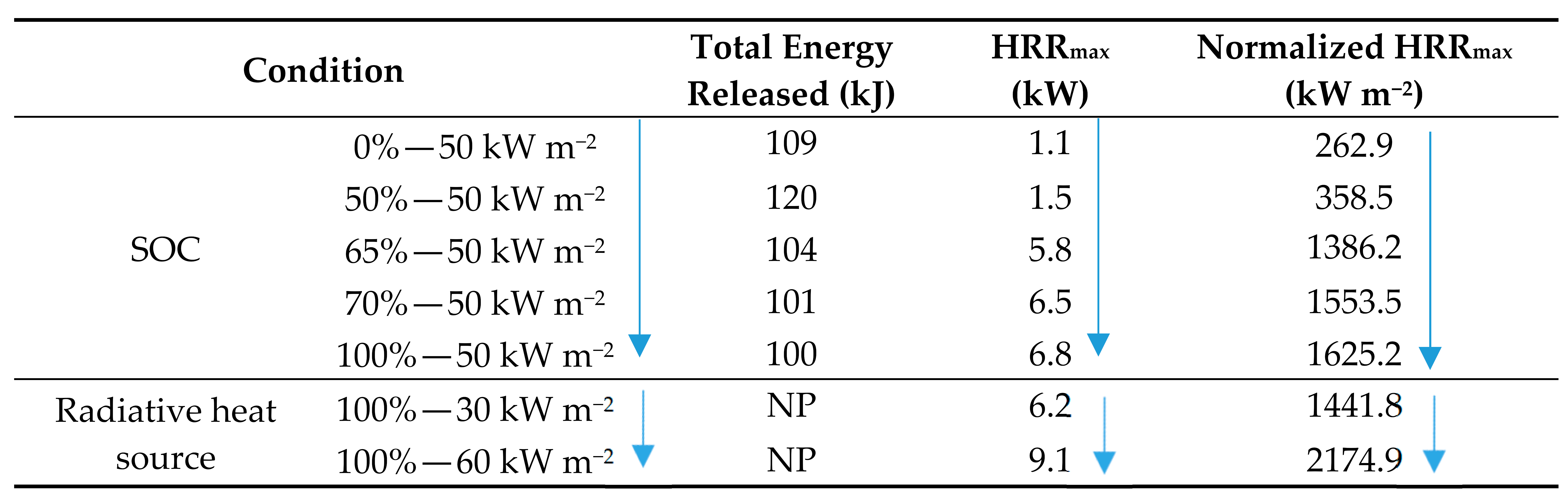
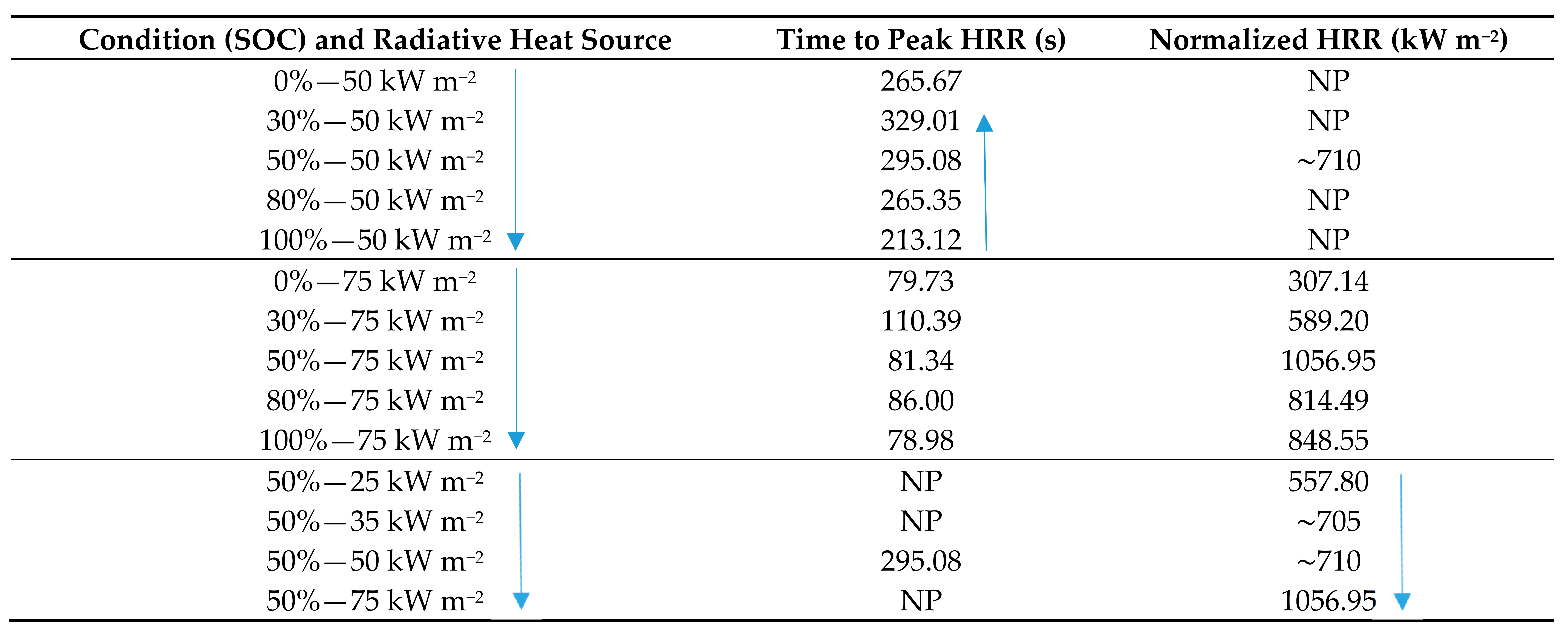
5.4. Comparative Review of Peak and Normalized HRR
- Method of testing;
- Size of external heat sources;
- LIB chemical composition;
- Materials of the LIB, i.e., housing/casing.
6. Modeling of LIBs
- The TR triggering temperature is increased by modifying the separator;
- Total electric energy released during TR can be reduced by discharging the battery;
- By increasing the convection coefficient, the heat dissipation can be enhanced;
- TR propagation can be prevented by adding additional thermal layers between adjacent batteries (validated by experiment).
7. Conclusions
- The chemical composition of the battery changes both the likelihood of a LIB going into TR and the consequence (energy magnitude of the resultant HRR) for the resultant fire/explosion.
- The higher SOC leads to shorter time to TR and ignition, and larger magnitude of the rate of energy released (HRR).
- HRR is considered a major factor after the LIB has gone into TR and the magnitude is directly proportional to the LIB chemistry, the SOC, and incident heat flux.
- Three-dimensional thermal modeling is challenging. Accurate data for thermal property variables such as thermal conductivity, density, and specific heat are critical to LIB fire modeling. Determining how a test object reacts/changes with time in a fire scenario is critical to the boundary conditions required to complete accurate LIB fire modeling.
- Three-dimensional thermal modeling has the potential to identify methods to be incorporated in systems to delay or inhibit TR, such as (a) modifying the separator, (b) varying the charging of the battery, (c) changing the convective heat transfer coefficient, and (d) adding additional thermal layers between adjacent batteries.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shahbazi, A.; Nasab, B.R. Carbon capture and storage (CCS) and its impacts on climate change and global warming. J. Pet. Environ. Biotechnol. 2016, 7, 4. [Google Scholar] [CrossRef]
- Le Quéré, C.; Peters, G.P.; Andres, R.J.; Andrew, R.M.; Boden, T.A.; Ciais, P.; Friedlingstein, P.; Houghton, R.A.; Marland, G.; Moriarty, R.; et al. Global carbon budget 2013. Earth Syst. Sci. Data 2014, 6, 235–263. [Google Scholar] [CrossRef]
- Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210–224. [Google Scholar] [CrossRef]
- Balakrishnan, P.G.; Ramesh, R.; Prem Kumar, T. Safety mechanisms in lithium-ion batteries. J. Power Sources 2006, 155, 401–414. [Google Scholar] [CrossRef]
- Guo, L.S.; Wang, Z.R.; Wang, J.H.; Luo, Q.K.; Liu, J.J. Effects of the environmental temperature and heat dissipation condition on the thermal runaway of lithium ion batteries during the charge-discharge process. J. Loss Prev. Process Ind. 2017, 49, 953–960. [Google Scholar] [CrossRef]
- Fu, Y.; Lu, S.; Li, K.; Liu, C.; Cheng, X.; Zhang, H. An experimental study on burning behaviors of 18650 lithium ion batteries using a cone calorimeter. J. Power Sources 2015, 273, 216–222. [Google Scholar] [CrossRef]
- Feng, X.; Ouyang, M.; Liu, X.; Lu, L.; Xia, Y.; He, X. Thermal runaway mechanism of lithium ion battery for electric vehicles: A review. Energy Storage Mater. 2018, 10, 246–267. [Google Scholar] [CrossRef]
- McDowall, J. A guide to lithium-ion battery safety. Battcon 2014, 1, 1–23. [Google Scholar]
- Ye, J.; Chen, H.; Wang, Q.; Huang, P.; Sun, J.; Lo, S. Thermal behavior and failure mechanism of lithium ion cells during overcharge under adiabatic conditions. Appl. Energy 2016, 182, 464–474. [Google Scholar] [CrossRef]
- Yuan, Q.; Zhao, F.; Wang, W.; Zhao, Y.; Liang, Z.; Yan, D. Overcharge failure investigation of lithium-ion batteries. Electrochim. Acta 2015, 178, 682–688. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Kato, K.; Lin, L.; Fukui, M. Athermal management system for lithium-ion battery in mobilesystems. In Proceedings of the 2013 European Conference on Circuit Theory and Designs (ECCTD), Dresden, Germany, 8–12 September 2013. [Google Scholar]
- McDowall, J. Understanding Lithium-Ion Technology. In Proceedings of the Battcon, Marco Island, FL, USA, 5–7 May 2008. [Google Scholar]
- Lisbona, D.; Snee, T. A review of hazards associated with primary lithium and lithium-ion batteries. Process Saf. Environ. Prot. 2011, 89, 434–442. [Google Scholar] [CrossRef]
- Li, Z.; Huang, J.; Yann Liaw, B.; Metzler, V.; Zhang, J. A review of lithium deposition in lithium-ion and lithium metal secondary batteries. J. Power Sources 2014, 254, 168–182. [Google Scholar] [CrossRef]
- Lee, C.W.; Venkatachalapathy, R.; Prakash, J. A Novel Flame-Retardant Additive for Lithium Batteries. Electrochem. Solid-State Lett. 2000, 3, 63–65. [Google Scholar] [CrossRef]
- Joachin, H.; Kaun, T.D.; Zaghib, K.; Prakash, J. Electrochemical and Thermal studies of LiFePO4 cathode in lithium-ion cells. ECS Trans. 2008, 6, 11–16. [Google Scholar] [CrossRef]
- Yang, H.; Bang, H.; Amine, K.; Prakash, J. Investigations of the exothermic reactions of natural graphite anode for Li-ion batteries during thermal runaway. J. Electrochem. Soc. 2005, 152, A73–A79. [Google Scholar] [CrossRef]
- Spotnitz, R.; Franklin, J. Abuse behavior of high-power, lithium-ion cells. J. Power Sources 2003, 113, 81–100. [Google Scholar]
- Ruiz, V.; Pfrang, A.; Kriston, A.; Omar, N.; Van den Bossche, P.; Boon-Brett, L. A review of international abuse testing standards and regulations for lithium ion batteries in electric and hybrid electric vehicles. Renew. Sustain. Energy Rev. 2018, 81, 1427–1452. [Google Scholar] [CrossRef]
- Lamb, J.; Orendorff, C.J. Evaluation of mechanical abuse techniques in lithium ion batteries. J. Power Sources 2014, 247, 189–196. [Google Scholar] [CrossRef]
- Moinuddin, K.A.M.; Thomas, I.R. An experimental study of fire development in deep enclosures and a new HRR-time-position model for a deep enclosure based on ventilation factor. Fire Mater. 2009, 33, 157–185. [Google Scholar] [CrossRef]
- Moinuddin, K.A.M.; Al-Menhali, J.S.; Prasannan, K.; Thomas, I.R. Rise in structural steel temperatures during ISO 9705 room fires. Fire Saf. J. 2011, 46, 480–496. [Google Scholar] [CrossRef]
- Vytenis Babrauskas, R.D.P. Heat release rate: The single most important variable in fire hazard. Fire Saf. J. 1992, 18, 255–272. [Google Scholar] [CrossRef]
- Brereton, P.; Kitchenham, B.A.; Budgen, D.; Turner, M.; Khalil, M. Lessons from applying the systematic literature review process within the software engineering domain. J. Syst. Softw. 2007, 80, 571–583. [Google Scholar] [CrossRef]
- Khan, K.S.; Ter Riet, G.; Glanville, J.; Sowden, A.J.; Kleijnen, J. Undertaking Systematic Reviews of Research on Effectiveness: CRD’s Guidance for Carrying Out or Commissioning Reviews; NHS Centre for Reviews and Dissemination: University of York: York, UK, 2001. [Google Scholar]
- Tan, S.; Moinuddin, K. Systematic review of human and organizational risks for probabilistic risk analysis in high-rise buildings. Reliab. Eng. Syst. Saf. 2019, 188, 233–250. [Google Scholar] [CrossRef]
- Liu, C.; Neale, Z.G.; Cao, G. Understanding electrochemical potentials of cathode materials in rechargeable batteries. Mater. Today 2016, 19, 109–123. [Google Scholar] [CrossRef]
- Pender, J.P.; Jha, G.; Youn, D.H.; Ziegler, J.M.; Andoni, I.; Choi, E.J.; Heller, A.; Dunn, B.S.; Weiss, P.S.; Penner, R.M. Electrode degradation in lithium-ion batteries. ACS Nano 2020, 14, 1243–1295. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczak, C.; Kahn, M.; White, K.; Long, R.T. Lithium-Ion Batteries Hazard and Use Assessment; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Noel, M.; Suryanarayanan, V. Role of carbon host lattices in Li-ion intercalation/de-intercalation processes. J. Power Sources 2002, 111, 193–209. [Google Scholar] [CrossRef]
- Yuan, Y.; Amine, K.; Lu, J.; Shahbazian-Yassar, R. Understanding materials challenges for rechargeable ion batteries with in situ transmission electron microscopy. Nat. Commun. 2017, 8, 15806. [Google Scholar] [CrossRef]
- Li, Q.; Chen, J.; Fan, L.; Kong, X.; Lu, Y. Progress in electrolytes for rechargeable Li-based batteries and beyond. Green Energy Environ. 2016, 1, 18–42. [Google Scholar] [CrossRef]
- Dahbi, M.; Ghamouss, F.; Tran-Van, F.; Lemordant, D.; Anouti, M. Comparative study of EC/DMC LiTFSI and LiPF6 electrolytes for electrochemical storage. J. Power Sources 2011, 196, 9743–9750. [Google Scholar] [CrossRef]
- Li, F.; Gong, Y.; Jia, G.; Wang, Q.; Peng, Z.; Fan, W.; Bai, B. A novel dual-salts of LiTFSI and LiODFB in LiFePO4-based batteries for suppressing aluminum corrosion and improving cycling stability. J. Power Sources 2015, 295, 47–54. [Google Scholar] [CrossRef]
- Feng, J.; Ai, X.; Cao, Y.; Yang, H. Possible use of non-flammable phosphonate ethers as pure electrolyte solvent for lithium batteries. J. Power Sources 2008, 177, 194–198. [Google Scholar] [CrossRef]
- Feng, J.; Sun, X.; Ai, X.; Cao, Y.; Yang, H. Dimethyl methyl phosphate: A new nonflammable electrolyte solvent for lithium-ion batteries. J. Power Sources 2008, 184, 570–573. [Google Scholar] [CrossRef]
- Lewandowski, A.; Świderska-Mocek, A. Ionic liquids as electrolytes for Li-ion batteries—An overview of electrochemical studies. J. Power Sources 2009, 194, 601–609. [Google Scholar] [CrossRef]
- Francis, C.; Louey, R.; Sammut, K.; Best, A.S. Thermal stability of pyrrolidinium-FSI ionic liquid electrolyte and lithium-ion electrodes at elevated temperatures. J. Electrochem. Soc. 2018, 165, A1204. [Google Scholar] [CrossRef]
- Moganty, S.; Wu, Y.; Abbate, L.; Brown, K.; Sinicropi, J.; Torres, G. Modified Ionic Liquids Containing Triazine. U.S. Patent Application 16/037,902, 17 January 2019. [Google Scholar]
- Takechi, K.; Yang, R. Aqueous Electrolyte with Ethers and Batteries Using the Electrolyte. U.S. Patent 10,193,188, 29 January 2019. [Google Scholar]
- Takechi, K.; Yang, R. Aqueous Electrolytes with Protonic Ionic Liquid and Batteries Using the Electrolyte. U.S. Patent Application 15/663,262, 31 January 2019. [Google Scholar]
- Zhu, Y.; Wang, X.; Hou, Y.; Gao, X.; Liu, L.; Wu, Y.; Shimizu, M. A new single-ion polymer electrolyte based on polyvinyl alcohol for lithium ion batteries. Electrochim. Acta 2013, 87, 113–118. [Google Scholar]
- Du, Z.; Su, Y.; Qu, Y.; Zhao, L.; Jia, X.; Mo, Y.; Yu, F.; Du, J.; Chen, Y. A mechanically robust, biodegradable and high performance cellulose gel membrane as gel polymer electrolyte of lithium-ion battery. Electrochim. Acta 2019, 299, 19–26. [Google Scholar] [CrossRef]
- Zhao, L.; Fu, J.; Du, Z.; Jia, X.; Qu, Y.; Yu, F.; Du, J.; Chen, Y. High-strength and flexible cellulose/PEG based gel polymer electrolyte with high performance for lithium ion batteries. J. Membr. Sci. 2020, 593, 117428. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, L.; Yu, Y.; Sun, J. Progress of enhancing the safety of lithium ion battery from the electrolyte aspect. Nano Energy 2019, 55, 93–114. [Google Scholar]
- Wakihara, M.; Kadoma, Y.; Kumagai, N.; Mita, H.; Araki, R.; Ozawa, K.; Ozawa, Y. Development of nonflammable lithium ion battery using a new all-solid polymer electrolyte. J. Solid State Electrochem. 2012, 16, 847–855. [Google Scholar] [CrossRef]
- Youcef, H.B.; Armand, M.; Orayech, B.; Saurel, D.; Shanmukaraj, D. Solid Polymer Electrolyte Based on Modified Cellulose and Its Use in Lithium or Sodium Secondary Batteries. U.S. Patent Application 16/020,568, 3 January 2019. [Google Scholar]
- Yushin, G.; Turcheniuk, K.; Yiran, X.; Song, A.-Y.; Borodin, O.; Nitta, N. Solid Electrolyte Technology with Rearrangeable Bonds for Metal and Metal-Ion Batteries. U.S. Patent Application 16/022,572, 3 January 2019. [Google Scholar]
- Liu, J.; Liu, Y.; Yang, W.; Ren, Q.; Li, F.; Huang, Z. Lithium ion battery separator with high performance and high safety enabled by tri-layered SiO2@PI/m-PE/SiO2@PI nanofiber composite membrane. J. Power Sources 2018, 396, 265–275. [Google Scholar] [CrossRef]
- Herle, S.P.; Gordon, J.G. Ceramic coating on battery separators. U.S. Patent Application 10,193,116, 29 January 2019. [Google Scholar]
- Shi, C.; Zhang, P.; Chen, L.; Yang, P.; Zhao, J. Effect of a thin ceramic-coating layer on thermal and electrochemical properties of polyethylene separator for lithium-ion batteries. J. Power Sources 2014, 270, 547–553. [Google Scholar] [CrossRef]
- Shin, W.-K.; Kim, D.-W. High performance ceramic-coated separators prepared with lithium ion-containing SiO2 particles for lithium-ion batteries. J. Power Sources 2013, 226, 54–60. [Google Scholar] [CrossRef]
- Kim, C.-S.; Yoo, J.-S.; Jeong, K.-M.; Kim, K.; Yi, C.-W. Investigation on internal short circuits of lithium polymer batteries with a ceramic-coated separator during nail penetration. J. Power Sources 2015, 289, 41–49. [Google Scholar] [CrossRef]
- Sun, G.; Dong, G.; Kong, L.; Yan, X.; Tian, G.; Qi, S.; Wu, D. Robust polyimide nanofibrous membrane with porous-layer-coated morphology by in situ self-bonding and micro-crosslinking for lithium-ion battery separator. Nanoscale 2018, 10, 22439–22447. [Google Scholar] [CrossRef] [PubMed]
- Orendorff, C.J. The role of separators in lithium-ion cell safety. Electrochem. Soc. Interface 2012, 21, 61. [Google Scholar] [CrossRef]
- Weber, C.J.; Geiger, S.; Falusi, S.; Roth, M. Material review of Li ion battery separators. AIP Conf. Proc. 2014, 66–81. [Google Scholar] [CrossRef]
- Li, X.; He, J.; Wu, D.; Zhang, M.; Meng, J.; Ni, P. Development of plasma-treated polypropylene nonwoven-based composites for high-performance lithium-ion battery separators. Electrochim. Acta 2015, 167, 396–403. [Google Scholar] [CrossRef]
- Kong, L.; Wang, Y.; Yu, H.; Liu, B.; Qi, S.; Wu, D.; Zhong, W.H.; Tian, G.; Wang, J. In-Situ Armoring: A Robust, High-Wettability and Fire-Resistant Hybrid Separator for Advanced and Safe Batteries. ACS Appl. Mater. Interfaces 2018. [Google Scholar] [CrossRef]
- Semenov, N.N. Some Problems in Chemical Kinetics and Reactivity; Princeton University Press: Princeton, NJ, USA, 1959; Volume 1. [Google Scholar]
- Ye, Y.; Saw, L.H.; Shi, Y.; Tay, A.A. Numerical analyses on optimizing a heat pipe thermal management system for lithium-ion batteries during fast charging. Appl. Therm. Eng. 2015, 86, 281–291. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, Q.; Ping, P.; Zhao, X.; Sun, J.; Lin, Z. Heat transfer in the dynamic cycling of lithium–titanate batteries. Int. J. Heat Mass Transf. 2016, 93, 896–905. [Google Scholar] [CrossRef]
- Bernardi, D.; Pawlikowski, E.; Newman, J. A general energy balance for battery systems. J. Electrochem. Soc. 1985, 132, 5. [Google Scholar] [CrossRef]
- Ghiji, M.; Novozhilov, V.; Moinuddin, K.; Joseph, P.; Burch, I.; Suendermann, B.; Gamble, G. A Review of Lithium-Ion Battery Fire Suppression. Energies 2020, 13, 5117. [Google Scholar] [CrossRef]
- Ouyang, D.; Chen, M.; Huang, Q.; Weng, J.; Wang, Z.; Wang, J. A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures. Appl. Sci. 2019, 9, 2483. [Google Scholar] [CrossRef]
- Zhong, G.; Mao, B.; Wang, C.; Jiang, L.; Xu, K.; Sun, J.; Wang, Q. Thermal runaway and fire behavior investigation of lithium ion batteries using modified cone calorimeter. J. Therm. Anal. Calorim. 2018. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, P.; Ping, P.; Du, Y.; Li, K.; Sun, J. Combustion behavior of lithium iron phosphate battery induced by external heat radiation. J. Loss Prev. Process Ind. 2017, 49, 961–969. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.; Gong, J.; Liu, K.; Wang, H.; Guo, L. Experimental Study of Thermal Runaway Process of 18650 Lithium-Ion Battery. Materials 2017, 10, 230. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ning, X.; Zhu, K.; Hu, J.; Yang, H.; Wang, J. Evaluating the thermal failure risk of large-format lithium-ion batteries using a cone calorimeter. J. Fire Sci. 2019, 37, 81–95. [Google Scholar] [CrossRef]
- Ouyang, D.; Chen, M.; Wei, R.; Wang, Z.; Wang, J. A study on the fire behaviors of 18650 battery and batteries pack under discharge. J. Therm. Anal. Calorim. 2018. [Google Scholar] [CrossRef]
- Hatchard, T.D.; Trussler, S.; Dahn, J.R. Building a “smart nail” for penetration tests on Li-ion cells. J. Power Sources 2014, 247, 821–823. [Google Scholar] [CrossRef]
- Mao, B.; Chen, H.; Cui, Z.; Wu, T.; Wang, Q. Failure mechanism of the lithium ion battery during nail penetration. Int. J. Heat Mass Transf. 2018, 122, 1103–1115. [Google Scholar] [CrossRef]
- Kitoh, K.; Nemoto, H. 100 Wh Large size Li-ion batteries and safety tests. J. Power Sources 1999, 81–82, 887–890. [Google Scholar] [CrossRef]
- Zhao, R.; Liu, J.; Gu, J. A comprehensive study on Li-ion battery nail penetrations and the possible solutions. Energy 2017, 123, 392–401. [Google Scholar] [CrossRef]
- Yokoshima, T.; Mukoyama, D.; Maeda, F.; Osaka, T.; Takazawa, K.; Egusa, S.; Naoi, S.; Ishikura, S.; Yamamoto, K. Direct observation of internal state of thermal runaway in lithium ion battery during nail-penetration test. J. Power Sources 2018, 393, 67–74. [Google Scholar] [CrossRef]
- Chiu, K.-C.; Lin, C.-H.; Yeh, S.-F.; Lin, Y.-H.; Chen, K.-C. An electrochemical modeling of lithium-ion battery nail penetration. J. Power Sources 2014, 251, 254–263. [Google Scholar] [CrossRef]
- Liu, B.; Yin, S.; Xu, J. Integrated computation model of lithium-ion battery subject to nail penetration. Appl. Energy 2016, 183, 278–289. [Google Scholar] [CrossRef]
- Zhao, W.; Luo, G.; Wang, C.-Y. Modeling nail penetration process in large-format Li-ion cells. J. Electrochem. Soc. 2014, 162, A207. [Google Scholar] [CrossRef]
- Liang, G.; Zhang, Y.; Han, Q.; Liu, Z.; Jiang, Z.; Tian, S. A novel 3D-layered electrochemical-thermal coupled model strategy for the nail-penetration process simulation. J. Power Sources 2017, 342, 836–845. [Google Scholar] [CrossRef]
- Ichimura, M. The safety characteristics of lithium-ion batteries for mobile phones and the nail penetration test. In Proceedings of the INTELEC 07-29th International Telecommunications Energy Conference, Rome, Italy, 30 September–4 October 2007; pp. 687–692. [Google Scholar]
- Liu, X.; Stoliarov, S.I.; Denlinger, M.; Masias, A.; Snyder, K. Comprehensive calorimetry of the thermally-induced failure of a lithium ion battery. J. Power Sources 2015, 280, 516–525. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Z.; Stoliarov, S.I.; Denlinger, M.; Masias, A.; Snyder, K. Heat release during thermally-induced failure of a lithium ion battery: Impact of cathode composition. Fire Saf. J. 2016, 85, 10–22. [Google Scholar] [CrossRef]
- Said, A.O.; Lee, C.; Liu, X.; Wu, Z.; Stoliarov, S.I.J.P.o.t.C.I. Simultaneous measurement of multiple thermal hazards associated with a failure of prismatic lithium ion battery. Proc. Combust. Inst. 2019, 37, 4173–4180. [Google Scholar]
- Feng, X.; Fang, M.; He, X.; Ouyang, M.; Lu, L.; Wang, H.; Zhang, M. Thermal runaway features of large format prismatic lithium ion battery using extended volume accelerating rate calorimetry. J. Power Sources 2014, 255, 294–301. [Google Scholar] [CrossRef]
- Perea, A.; Paolella, A.; Dubé, J.; Champagne, D.; Mauger, A.; Zaghib, K. State of charge influence on thermal reactions and abuse tests in commercial lithium-ion cells. J. Power Sources 2018, 399, 392–397. [Google Scholar] [CrossRef]
- Wang, Z.; Ouyang, D.; Chen, M.; Wang, X.; Zhang, Z.; Wang, J. Fire behavior of lithium-ion battery with different states of charge induced by high incident heat fluxes. J. Therm. Anal. Calorim. 2018. [Google Scholar] [CrossRef]
- Larsson, F.; Andersson, P.; Blomqvist, P.; Lorén, A.; Mellander, B.-E. Characteristics of lithium-ion batteries during fire tests. J. Power Sources 2014, 271, 414–420. [Google Scholar] [CrossRef]
- Chen, M.; Zhou, D.; Chen, X.; Zhang, W.; Liu, J.; Yuen, R.; Wang, J. Investigation on the thermal hazards of 18650 lithium ion batteries by fire calorimeter. J. Therm. Anal. Calorim. 2015, 122, 755–763. [Google Scholar] [CrossRef]
- Ping, P.; Wang, Q.; Huang, P.; Li, K.; Sun, J.; Kong, D.; Chen, C. Study of the fire behavior of high-energy lithium-ion batteries with full-scale burning test. J. Power Sources 2015, 285, 80–89. [Google Scholar] [CrossRef]
- Zheng, Y.; He, Y.-B.; Qian, K.; Li, B.; Wang, X.; Li, J.; Miao, C.; Kang, F. Effects of state of charge on the degradation of LiFePO4/graphite batteries during accelerated storage test. J. Alloys Compd. 2015, 639, 406–414. [Google Scholar] [CrossRef]
- Ouyang, D.; Liu, J.; Chen, M.; Weng, J.; Wang, J. An experimental study on the thermal failure propagation in lithium-ion battery pack. J. Electrochem. Soc. 2018, 165, A2184. [Google Scholar]
- Ouyang, D.; Liu, J.; Chen, M.; Weng, J.; Wang, J. Thermal failure propagation in lithium-ion battery modules with various shapes. Appl. Sci. 2018, 8, 1263. [Google Scholar] [CrossRef]
- Lopez, C.F.; Jeevarajan, J.A.; Mukherjee, P.P. Experimental analysis of thermal runaway and propagation in lithium-ion battery modules. J. Electrochem. Soc. 2015, 162, A1905. [Google Scholar] [CrossRef]
- Larsson, F.; Anderson, J.; Andersson, P.; Mellander, B.-E. Thermal modelling of cell-to-cell fire propagation and cascading thermal runaway failure effects for lithium-ion battery cells and modules using fire walls. J. Electrochem. Soc. 2016, 163, A2854. [Google Scholar] [CrossRef]
- Burch, I.; Ghiji, M.; Gamble, G.; Suendermann, B.; Joseph, P.; Moinuddin, K.; Novozhilov, V. Lithium-ion Battery Fire Suppression in Submarine Battery Compartments. In Proceedings of the PACIFIC 2019, Sydney, Australia, 8–10 October 2019. [Google Scholar]
- Feng, X.; He, X.; Ouyang, M.; Lu, L.; Wu, P.; Kulp, C.; Prasser, S. Thermal runaway propagation model for designing a safer battery pack with 25 Ah LiNixCoyMnzO2 large format lithium ion battery. Appl. Energy 2015, 154, 74–91. [Google Scholar] [CrossRef]
- Wilke, S.; Schweitzer, B.; Khateeb, S.; Al-Hallaj, S. Preventing thermal runaway propagation in lithium ion battery packs using a phase change composite material: An experimental study. J. Power Sources 2017, 340, 51–59. [Google Scholar] [CrossRef]
- Spinner, N.S.; Field, C.R.; Hammond, M.H.; Williams, B.A.; Myers, K.M.; Lubrano, A.L.; Rose-Pehrsson, S.L.; Tuttle, S.G. Physical and chemical analysis of lithium-ion battery cell-to-cell failure events inside custom fire chamber. J. Power Sources 2015, 279, 713–721. [Google Scholar] [CrossRef]
- Gao, S.; Feng, X.; Lu, L.; Ouyang, M.; Kamyab, N.; White, R.E.; Coman, P. Thermal runaway propagation assessment of different battery pack designs using the TF5 draft as framework. J. Electrochem. Soc. 2019, 166, A1653. [Google Scholar]
- Lamb, J.; Orendorff, C.J.; Steele, L.A.M.; Spangler, S.W. Failure propagation in multi-cell lithium ion batteries. J. Power Sources 2015, 283, 517–523. [Google Scholar] [CrossRef]
- Murmann, P.; Mönnighoff, X.; von Aspern, N.; Janssen, P.; Kalinovich, N.; Shevchuk, M.; Kazakova, O.; Röschenthaler, G.-V.; Cekic-Laskovic, I.; Winter, M. Influence of the fluorination degree of organophosphates on flammability and electrochemical performance in lithium ion batteries: Studies on fluorinated compounds deriving from triethyl phosphate. J. Electrochem. Soc. 2016, 163, A751. [Google Scholar]
- Laoutid, F.; Bonnaud, L.; Alexandre, M.; Lopez-Cuesta, J.M.; Dubois, P. New prospects in flame retardant polymer materials: From fundamentals to nanocomposites. Mater. Sci. Eng. R Rep. 2009, 63, 100–125. [Google Scholar] [CrossRef]
- Xiang, H.F.; Xu, H.Y.; Wang, Z.Z.; Chen, C.H. Dimethyl methylphosphonate (DMMP) as an efficient flame retardant additive for the lithium-ion battery electrolytes. J. Power Sources 2007, 173, 562–564. [Google Scholar] [CrossRef]
- Hu, J.; Jin, Z.; Zhong, H.; Zhan, H.; Zhou, Y.; Li, Z. A new phosphonamidate as flame retardant additive in electrolytes for lithium ion batteries. J. Power Sources 2012, 197, 297–300. [Google Scholar] [CrossRef]
- Ding, M.S.; Xu, K.; Jow, T.R. Effects of tris (2, 2, 2-trifluoroethyl) phosphate as a flame-retarding cosolvent on physicochemical properties of electrolytes of LiPF6 in EC-PC-EMC of 3: 3: 4 weight ratios. J. Electrochem. Soc. 2002, 149, A1489. [Google Scholar] [CrossRef]
- Zeng, Z.; Jiang, X.; Wu, B.; Xiao, L.; Ai, X.; Yang, H.; Cao, Y. Bis(2,2,2-trifluoroethyl) methylphosphonate: An Novel Flame-retardant Additive for Safe Lithium-ion Battery. Electrochim. Acta 2014, 129, 300–304. [Google Scholar] [CrossRef]
- Belov, D.G.; Shieh, D.T. A study of tetrabromobisphenol A (TBBA) as a flame retardant additive for Li-ion battery electrolytes. J. Power Sources 2014, 247, 865–875. [Google Scholar] [CrossRef]
- Wu, B.; Pei, F.; Wu, Y.; Mao, R.; Ai, X.; Yang, H.; Cao, Y. An electrochemically compatible and flame-retardant electrolyte additive for safe lithium ion batteries. J. Power Sources 2013, 227, 106–110. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Chen, L.; Lu, Y.; Su, Y.; Bao, L.; Wang, J.; Chen, R.; Chen, S.; Wu, F. Ethoxy (pentafluoro) cyclotriphosphazene (PFPN) as a multi-functional flame retardant electrolyte additive for lithium-ion batteries. J. Power Sources 2018, 378, 707–716. [Google Scholar] [CrossRef]
- Moreno, M.; Simonetti, E.; Appetecchi, G.; Carewska, M.; Montanino, M.; Kim, G.-T.; Loeffler, N.; Passerini, S. Ionic liquid electrolytes for safer lithium batteries. J. Electrochem. Soc. 2016, 164, A6026. [Google Scholar] [CrossRef]
- Chawla, N.; Bharti, N.; Singh, S. Recent advances in non-flammable electrolytes for safer lithium-ion batteries. Batteries 2019, 5, 19. [Google Scholar] [CrossRef]
- Shi, P.; Zheng, H.; Liang, X.; Sun, Y.; Cheng, S.; Chen, C.; Xiang, H. A highly concentrated phosphate-based electrolyte for high-safety rechargeable lithium batteries. Chem. Commun. 2018, 54, 4453–4456. [Google Scholar] [CrossRef]
- Safa, M.; Chamaani, A.; Chawla, N.; El-Zahab, B. Polymeric Ionic Liquid Gel Electrolyte for Room Temperature Lithium Battery Applications. Electrochim. Acta 2016, 213, 587–593. [Google Scholar] [CrossRef]
- Guo, Q.; Han, Y.; Wang, H.; Xiong, S.; Sun, W.; Zheng, C.; Xie, K. Flame Retardant and stable Li1.5Al0.5Ge1.5 (PO4)3-supported ionic liquid gel polymer electrolytes for high safety rechargeable solid-state lithium metal batteries. J. Phys. Chem. C 2018, 122, 10334–10342. [Google Scholar]
- Rectenwald, M.F.; Gaffen, J.R.; Rheingold, A.L.; Morgan, A.B.; Protasiewicz, J.D. Phosphoryl-Rich Flame-Retardant Ions (FRIONs): Towards Safer Lithium-Ion Batteries. Angew. Chem. 2014, 126, 4257–4260. [Google Scholar] [CrossRef]
- Dagger, T.; Meier, V.; Hildebrand, S.; Brüggemann, D.; Winter, M.; Schappacher, F.M. Safety Performance of 5 Ah Lithium Ion Battery Cells Containing the Flame Retardant Electrolyte Additive (Phenoxy) Pentafluorocyclotriphosphazene. Energy Technol. 2018, 6, 2001–2010. [Google Scholar] [CrossRef]
- Enotiadis, A.; Fernandes, N.J.; Becerra, N.A.; Zammarano, M.; Giannelis, E.P. Nanocomposite electrolytes for lithium batteries with reduced flammability. Electrochim. Acta 2018, 269, 76–82. [Google Scholar] [CrossRef]
- Wu, H.; Zhuo, D.; Kong, D.; Cui, Y. Improving battery safety by early detection of internal shorting with a bifunctional separator. Nat. Commun. 2014, 5, 5193. [Google Scholar] [CrossRef] [PubMed]
- Ryou, M.H.; Lee, D.J.; Lee, J.N.; Lee, Y.M.; Park, J.K.; Choi, J.W. Excellent cycle life of lithium-metal anodes in lithium-ion batteries with mussel-inspired polydopamine-coated separators. Adv. Energy Mater. 2012, 2, 645–650. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, J.-H.; Choi, K.-H.; Yu, H.K.; Kim, J.H.; Lee, J.S.; Lee, S.-Y. Inverse opal-inspired, nanoscaffold battery separators: A new membrane opportunity for high-performance energy storage systems. Nano Lett. 2014, 14, 4438–4448. [Google Scholar] [CrossRef] [PubMed]
- Yeon, D.; Lee, Y.; Ryou, M.-H.; Lee, Y.M. New flame-retardant composite separators based on metal hydroxides for lithium-ion batteries. Electrochim. Acta 2015, 157, 282–289. [Google Scholar] [CrossRef]
- Costa, C.M.; Kundu, M.; Cardoso, V.F.; Machado, A.V.; Silva, M.M.; Lanceros-Méndez, S. Silica/poly(vinylidene fluoride) porous composite membranes for lithium-ion battery separators. J. Membr. Sci. 2018, 564, 842–851. [Google Scholar] [CrossRef]
- Liu, J.; Yang, K.; Mo, Y.; Wang, S.; Han, D.; Xiao, M.; Meng, Y. Highly safe lithium-ion batteries: High strength separator from polyformaldehyde/cellulose nanofibers blend. J. Power Sources 2018, 400, 502–510. [Google Scholar] [CrossRef]
- Wang, Q.; Mao, B.; Stoliarov, S.I.; Sun, J. A review of lithium ion battery failure mechanisms and fire prevention strategies. Prog. Energy Combust. Sci. 2019, 73, 95–131. [Google Scholar] [CrossRef]
- Kong, L.; Li, C.; Jiang, J.; Pecht, M.G. Li-ion battery fire hazards and safety strategies. Energies 2018, 11, 2191. [Google Scholar] [CrossRef]
- Austin Mier, F.; Hargather, M.J.; Ferreira, S.R. Experimental quantification of vent mechanism flow parameters in 18650 format lithium ion batteries. J. Fluids Eng. 2019, 141, 061403. [Google Scholar] [CrossRef]
- Zhang, H.; Pang, J.; Ai, X.; Cao, Y.; Yang, H.; Lu, S. Poly(3-butylthiophene)-based positive-temperature-coefficient electrodes for safer lithium-ion batteries. Electrochim. Acta 2016, 187, 173–178. [Google Scholar] [CrossRef]
- Meyer, J. Glass transition temperature as a guide to selection of polymers suitable for PTC materials. Polymer Eng. Sci. 1973, 13, 462–468. [Google Scholar] [CrossRef]
- Johnson, B.A.; White, R.E. Characterization of commercially available lithium-ion batteries. J. Power Sources 1998, 70, 48–54. [Google Scholar] [CrossRef]
- Huo, W.; Qu, Y. Effects of Bi1/2Na1/2TiO3 on the Curie temperature and the PTC effects of BaTiO3-based positive temperature coefficient ceramics. Sens. Actuators A Phys. 2006, 128, 265–269. [Google Scholar] [CrossRef]
- Ding, S.W.; Jia, G.; Wang, J.; He, Z.Y. Electrical properties of Y- and Mn-doped BaTiO3-based PTC ceramics. Ceram. Int. 2008, 34, 2007–2010. [Google Scholar] [CrossRef]
- Zhao, S.; Lou, D.; Zhan, P.; Li, G.; Dai, K.; Guo, J.; Zheng, G.; Liu, C.; Shen, C.; Guo, Z. Heating-induced negative temperature coefficient effect in conductive graphene/polymer ternary nanocomposites with a segregated and double-percolated structure. J. Mater. Chem. C 2017, 5, 8233–8242. [Google Scholar]
- Long, R.T.; Blum, A.F.; Bress, T.J.; Cotts, B.R. Best Practices for Emergency Response to Incidents Involving Electric Vehicles Battery Hazards: A Report on Full-Scale Testing Results; National Fire Protection Research Foundation: Quincy, MA, USA, 2013; p. 110. [Google Scholar]
- Maloney, T. Extinguishment of Lithium-Ion and Lithium-Metal Battery Fires; US Federal Aviation Administration: Washington, DC, USA, 2014; pp. 46–51. [Google Scholar]
- Hill, D. Considerations for Energy Storage Systems (ESS) Fire Safety; OAPUS301WIKO(PP151894), Rev. 4; Det Norske Veritas (U.S.A.), Inc. (DNV GL): Dublin, OH, USA, 9 February 2017. [Google Scholar]
- Ribière, P.; Grugeon, S.; Morcrette, M.; Boyanov, S.; Laruelle, S.; Marlair, G. Investigation on the fire-induced hazards of Li-ion battery cells by fire calorimetry. Energy Environ. Sci. 2012, 5, 5271–5280. [Google Scholar] [CrossRef]
- Fu, Y.; Lu, S.; Shi, L.; Cheng, X.; Zhang, H. Combustion characteristics of electrolyte pool fires for lithium ion batteries. J. Electrochem. Soc. 2016, 163, A2022. [Google Scholar] [CrossRef]
- Eshetu, G.G.; Grugeon, S.; Laruelle, S.; Boyanov, S.; Lecocq, A.; Bertrand, J.-P.; Marlair, G. In-depth safety-focused analysis of solvents used in electrolytes for large scale lithium ion batteries. Phys. Chem. Chem. Phys. 2013, 15, 9145–9155. [Google Scholar] [CrossRef] [PubMed]
- Beyler, C.; Croce, P.; Dubay, C.; Johnson, P.; McNamee, M. Oxygen consumption calorimetry, William Parker: 2016 DiNenno Prize. Fire Sci. Rev. 2017, 6. [Google Scholar] [CrossRef]
- Huang, P.; Wang, Q.; Li, K.; Ping, P.; Sun, J. The combustion behavior of large scale lithium titanate battery. Sci. Rep. 2015, 5, 7788. [Google Scholar] [CrossRef] [PubMed]
- Rao, H.; Huang, Z.; Zhang, H.; Xiao, S. Study of fire tests and fire safety measures on lithiumion battery used on ships. In Proceedings of the 2015 International Conference on Transportation Information and Safety (ICTIS), Wuhan, China, 25–28 June 2015; pp. 865–870. [Google Scholar]
- Wang, Q.; Ping, P.; Sun, J. Catastrophe analysis of cylindrical lithium ion battery. Nonlinear Dyn. 2010, 61, 763–772. [Google Scholar] [CrossRef]
- Hatchard, T.; MacNeil, D.; Basu, A.; Dahn, J.R. Thermal model of cylindrical and prismatic lithium-ion cells. J. Electrochem. Soc. 2001, 148, A755. [Google Scholar] [CrossRef]
- Kim, G.-H.; Pesaran, A.; Spotnitz, R. A three-dimensional thermal abuse model for lithium-ion cells. J. Power Sources 2007, 170, 476–489. [Google Scholar] [CrossRef]
- Esho, I.; Shah, K.; Jain, A. Measurements and modeling to determine the critical temperature for preventing thermal runaway in Li-ion cells. Appl. Therm. Eng. 2018, 145, 287–294. [Google Scholar] [CrossRef]
- Anderson, J.; Larsson, F.; Andersson, P.; Mellander, B.-E. Thermal Modeling of Fire Propagation in Lithium-Ion Batteries. Proceedings of The 24th International Technical Conference on the Enhanced Safety of Vehicles (ESV), Gothenburg, Sweden, 8–11 June 2015. [Google Scholar]
- Feng, X.; Lu, L.; Ouyang, M.; Li, J.; He, X. A 3D thermal runaway propagation model for a large format lithium ion battery module. Energy 2016, 115, 194–208. [Google Scholar] [CrossRef]
- Huang, P.; Ping, P.; Li, K.; Chen, H.; Wang, Q.; Wen, J.; Sun, J. Experimental and modeling analysis of thermal runaway propagation over the large format energy storage battery module with Li4Ti5O12 anode. Appl. Energy 2016, 183, 659–673. [Google Scholar] [CrossRef]
- Huang, P.; Chen, H.; Verma, A.; Wang, Q.; Mukherjee, P.; Sun, J. Non-dimensional analysis of the criticality of Li-ion battery thermal runaway behavior. J. Hazard. Mater. 2019, 369, 268–278. [Google Scholar] [CrossRef]
- Harris, S.J.; Timmons, A.; Pitz, W.J. A combustion chemistry analysis of carbonate solvents used in Li-ion batteries. J. Power Sources 2009, 193, 855–858. [Google Scholar] [CrossRef]
- Fernandes, Y.; Bry, A.; de Persis, S. Identification and quantification of gases emitted during abuse tests by overcharge of a commercial Li-ion battery. J. Power Sources 2018, 389, 106–119. [Google Scholar] [CrossRef]
- Johnsplass, J.; Henriksen, M.; Vaagsaether, K.; Lundberg, J.; Bjerketvedt, D. Simulation of Burning Velocities in Gases Vented from Thermal Run-A-Way Lithium Ion Batteries. In Proceedings of the 58th Conference on Simulation and Modelling (SIMS 58), Reykjavik, Iceland, 25–27 September 2017. [Google Scholar]
| Component | Common Composition |
|---|---|
| Anode | Graphitic carbons, hard carbons, synthetic graphite, LTO, tin-based alloys, silicon-based alloys |
| Cathode | LMO, LCO, NCA, NMC, LFP, ECPs |
| Electrolyte | Lithium salts (mostly LiPF6) in organic solvents such as EC, DEC, DMC, PC, GBL, RTILs |
| Separator | Polypropylene, polyethylene, cellulosic paper, nonwoven fabrics, ceramic |
| Current Collector | Copper for anode Aluminum for cathode |
| Abuse Condition | Direct Cause |
|---|---|
| Thermal | Overheat Extreme cold Fire Thermal shock |
| Mechanical | Shock Drop Penetration Immersion Crush Vibration |
| Electrical | External short circuit Internal short circuit Overcharge Overdischarge |
 |
 |
 |
 |
 |
| BMS | Advantages | Disadvantages | |
|---|---|---|---|
| ACS | Natural convection | Low cost; simple structure; easy to integrate; little electricity consumption | Low heat transfer coefficient; dependent on ambient temperature; uneven temperature distribution |
| Forced convection | Low cost; easy to maintain | Low efficiency; dependent on ambient environment; insufficient for extreme conditions | |
| LCS | Liquid cooling | Low cost; easy to maintain | Risk of leakage |
| Vapor cooling | Higher efficiency; low operating cost; better uniformity | Higher cost for structure design; high cost for circulation | |
| PCM-CS | Organic Inorganic Eutectic | High efficiency; uniform temperature distribution; appropriate to extreme conditions | Risk of leakage; volume difference with phase change; risk of supercooling |
| Solvent | Molecular Formula | Flash point (°C) | Boiling Point (°C) | ΔHc (MJ kg−1) | ΔHvap (MJ kg−1) | ΔfG0 (kJ mol−1) |
| EC [136] | C3 H4 O3 | 146 | 248 | 12.66 | 0.62 | −276.19 |
| DMC [136] | C3 H6 O3 | 18 | 91 | 14.48 | 0.43 | −364.54 |
| EMC [136] | C4 H8 O3 | 23 | 110 | 18.41 | 0.34 | −356.12 |
| DEC [137] | C5 H10 O3 | 445 | 126 | 21.63 | 0.34 | −347.70 |
| PC [137] | C4 H6 O3 | 455 | 242 | 16.56 | 0.55 | −275.48 |
| EA [137] | C4 H8 O2 | 427 | 77 | 23.77 | 0.35 | −251.12 |
 |
 |
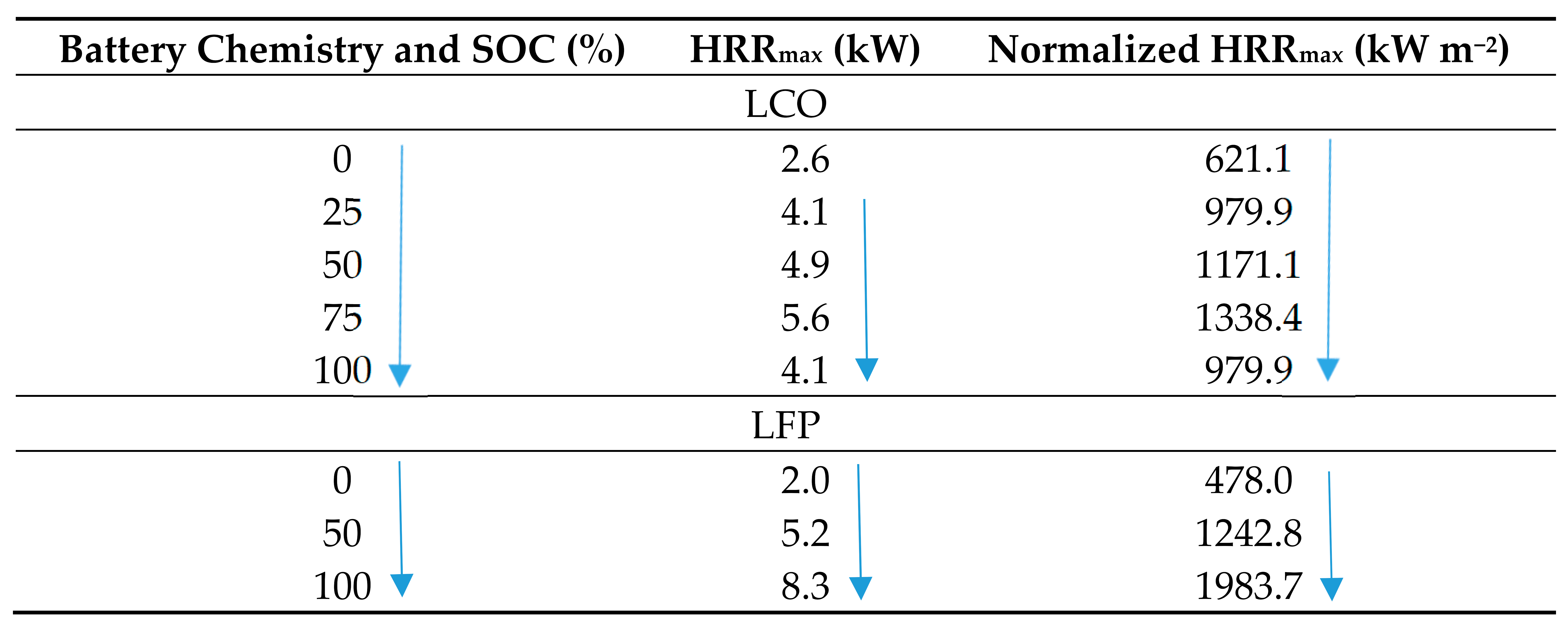 |
| Test no. | Battery Type | No. of Cells | Nominal Capacity (Ah) | Test Condition | Time to the Peak HRR (s) | HRRmax (kW) | HRRmax (kW)/cell | Normalized HRRmax (kW m−2) |
|---|---|---|---|---|---|---|---|---|
| 1 | EiG ePLB-F007A | 5 | 35 | 100% SOC | 90 | 55 | 11 | 110–490 |
| 2 | EiG ePLB-F007A | 5 | 35 | 100% SOC | 51 | 10.2 | ||
| 3 | EiG ePLB-F007A | 5 | 35 | 100% SOC + water mist | 49 | 9.8 | ||
| 4 | EiG ePLB-F007A | 5 | 35 | 0% SOC | 13 | 2.6 | ||
| 5 | EiG ePLB-F007A | 5 | 35 | 50% SOC | 200 | 17 | 3.4 | |
| 6 | K2 LFP26650EV | 9 | 28.8 | 100% SOC | 390 | 29 | 5.8 | 310 |
| 7 | Lenovo laptop battery pack | 12 | 33.6 | 100% SOC | 200 | 57 | 11.4 | 460 |
| SOC (%). | HRRmax (kW) | Time to Peak HRR (s) | Normalized HRRmax (kW m−2) |
|---|---|---|---|
| 0% peak 1 | 7.7 | 1629 | 229.6 |
| 0% peak 2 | 12.9 | 1770 | |
| 50% peak 1 | 15.5 | 1578 | 536.9 |
| 50% peak 2 | 30.1 | 1655 | |
| 50% peak 3 | 20.1 | 1847 | |
| 100% peak 1 | 18.9 | 1532 | 881.7 |
| 100% peak 2 | 48.4 | 1705 | |
| 100% peak3 | 49.4 | 1760 |
| Battery Number | #1 (50 Ah) | #2 (50 Ah) |
|---|---|---|
| SOC % | 50 | 100 |
| Peak HRR (kW) | 55.93 | 64.32 |
| Normalized HRR (kW m−2) | 587.50 | 675.63 |
| Ignition time (s) | 976 | 1108 |
| Surface temperature at ignition (°C) | 124.50 | 128.90 |
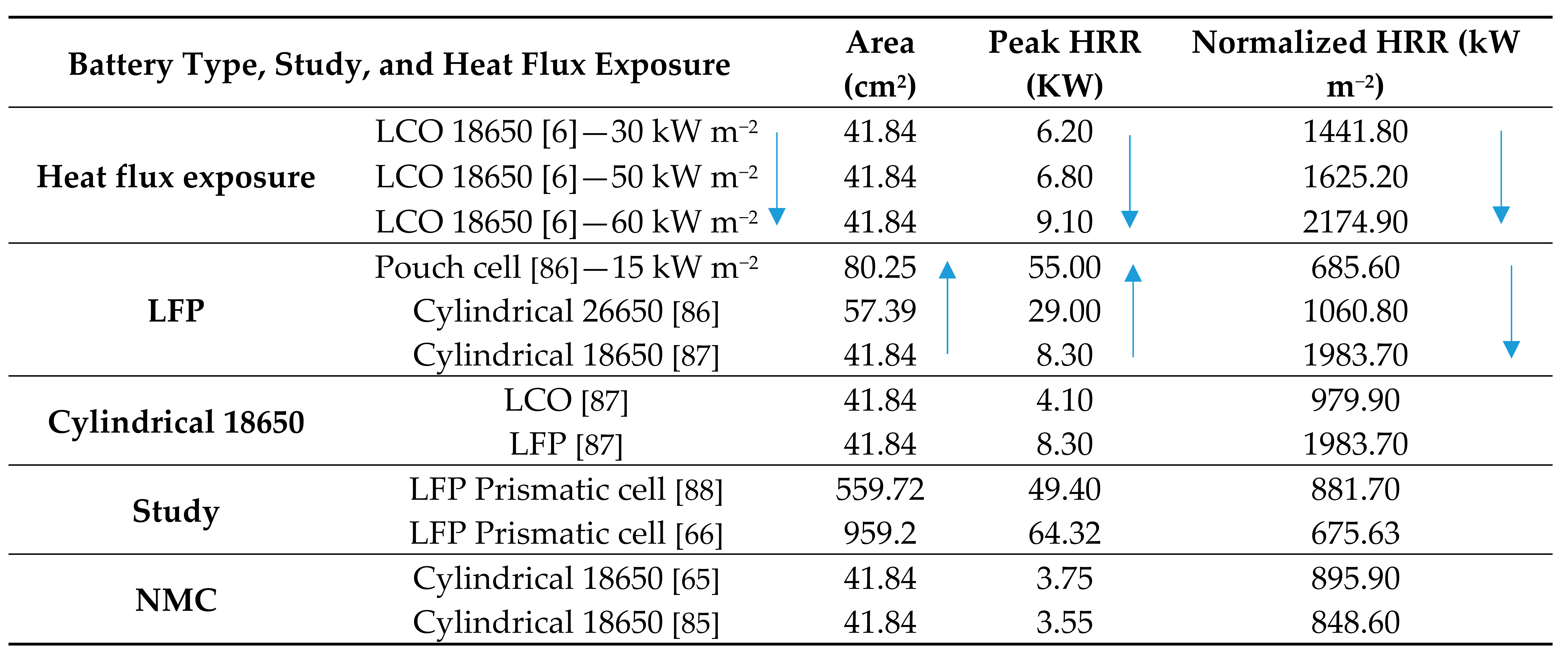 |
| SOC | Peak HRR (kW) | |||||
|---|---|---|---|---|---|---|
| LCO [6] | LCO [87] | NMC [65] | NMC [85] | LFP [87] | Average | |
| 0% | 1.1 | 2 | NP | 1.29 | 1.3 | 1.4 |
| 25% | NP | 4.1 | NP | NP | NP | NP |
| 30% | NP | NP | NP | 2.47 | NP | NP |
| 50% | 1.5 | 4.4 | NP | 4.42 | 5.1 | 3.9 |
| 65% | 5.8 | 4.9 | NP | NP | NP | 5.4 |
| 70% | 6.5 | NP | 2.17 | NP | NP | 4.3 |
| 75% | NP | 5.6 | NP | NP | NP | NP |
| 80% | NP | NP | 2.47 | 3.41 | NP | 1.94 |
| 90% | NP | NP | 3.14 | NP | NP | NP |
| 100% | 6.8 | 4.1 | 3.75 | 3.55 | 6.8 | 5 |
| Duration (s) | D1,2 | D2,3 | D3,4 | D4,5 | D5,6 |
|---|---|---|---|---|---|
| Experiment | 245 | 163 | 186 | 164 | 159 |
| Model | 236 | 158 | 179 | 189 | 170 |
| Temperature (°C) | p_1,2 | p_2,3 | p_3,4 | p_4,5 | p_5,6 |
| Experiment | 144 | 121 | 183 | 146 | 146 |
| Model | 141 | 174 | 168 | 165 | 168 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghiji, M.; Edmonds, S.; Moinuddin, K. A Review of Experimental and Numerical Studies of Lithium Ion Battery Fires. Appl. Sci. 2021, 11, 1247. https://doi.org/10.3390/app11031247
Ghiji M, Edmonds S, Moinuddin K. A Review of Experimental and Numerical Studies of Lithium Ion Battery Fires. Applied Sciences. 2021; 11(3):1247. https://doi.org/10.3390/app11031247
Chicago/Turabian StyleGhiji, Matt, Shane Edmonds, and Khalid Moinuddin. 2021. "A Review of Experimental and Numerical Studies of Lithium Ion Battery Fires" Applied Sciences 11, no. 3: 1247. https://doi.org/10.3390/app11031247
APA StyleGhiji, M., Edmonds, S., & Moinuddin, K. (2021). A Review of Experimental and Numerical Studies of Lithium Ion Battery Fires. Applied Sciences, 11(3), 1247. https://doi.org/10.3390/app11031247







