Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill
Abstract
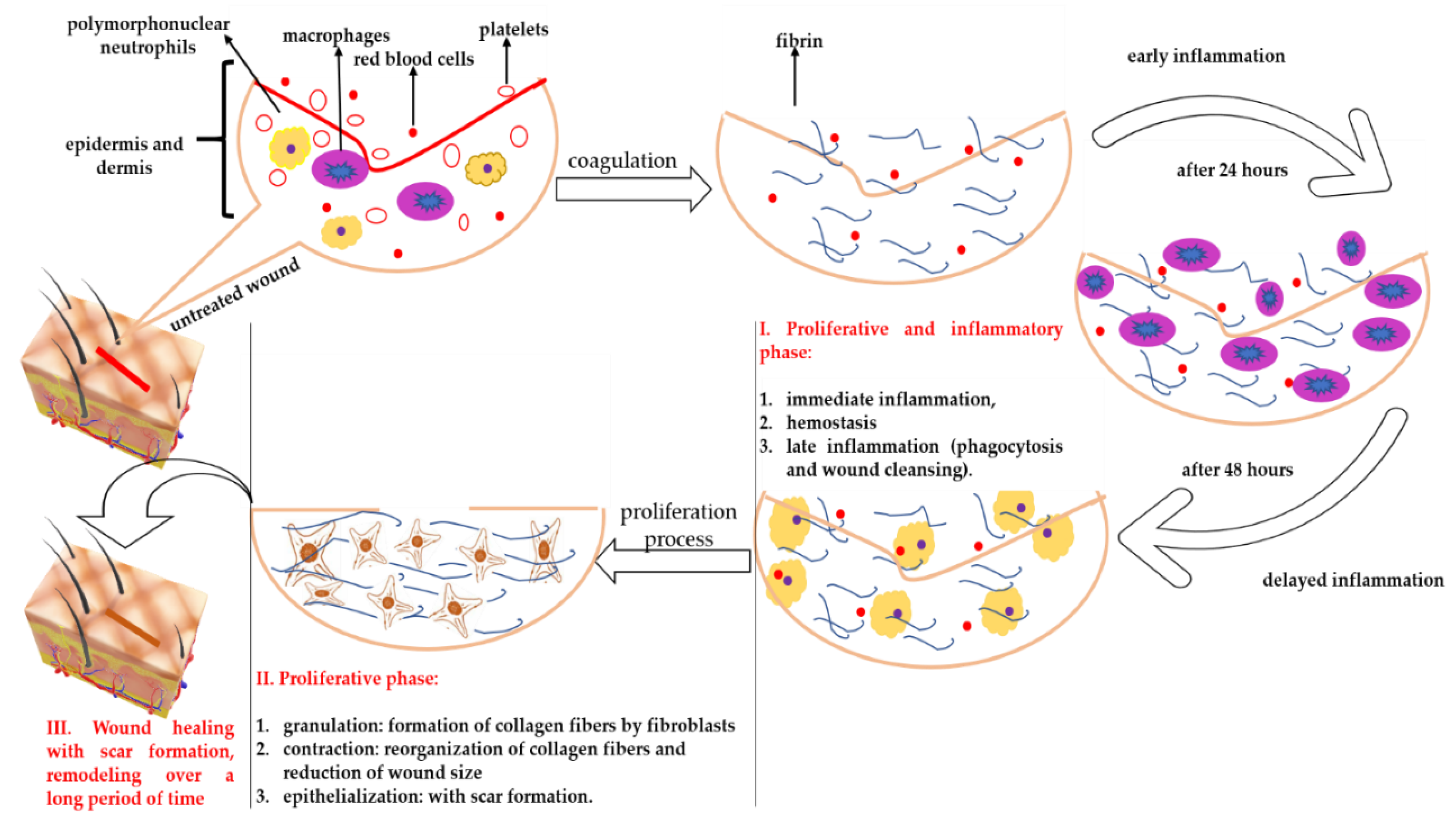
1. Introduction
2. Results
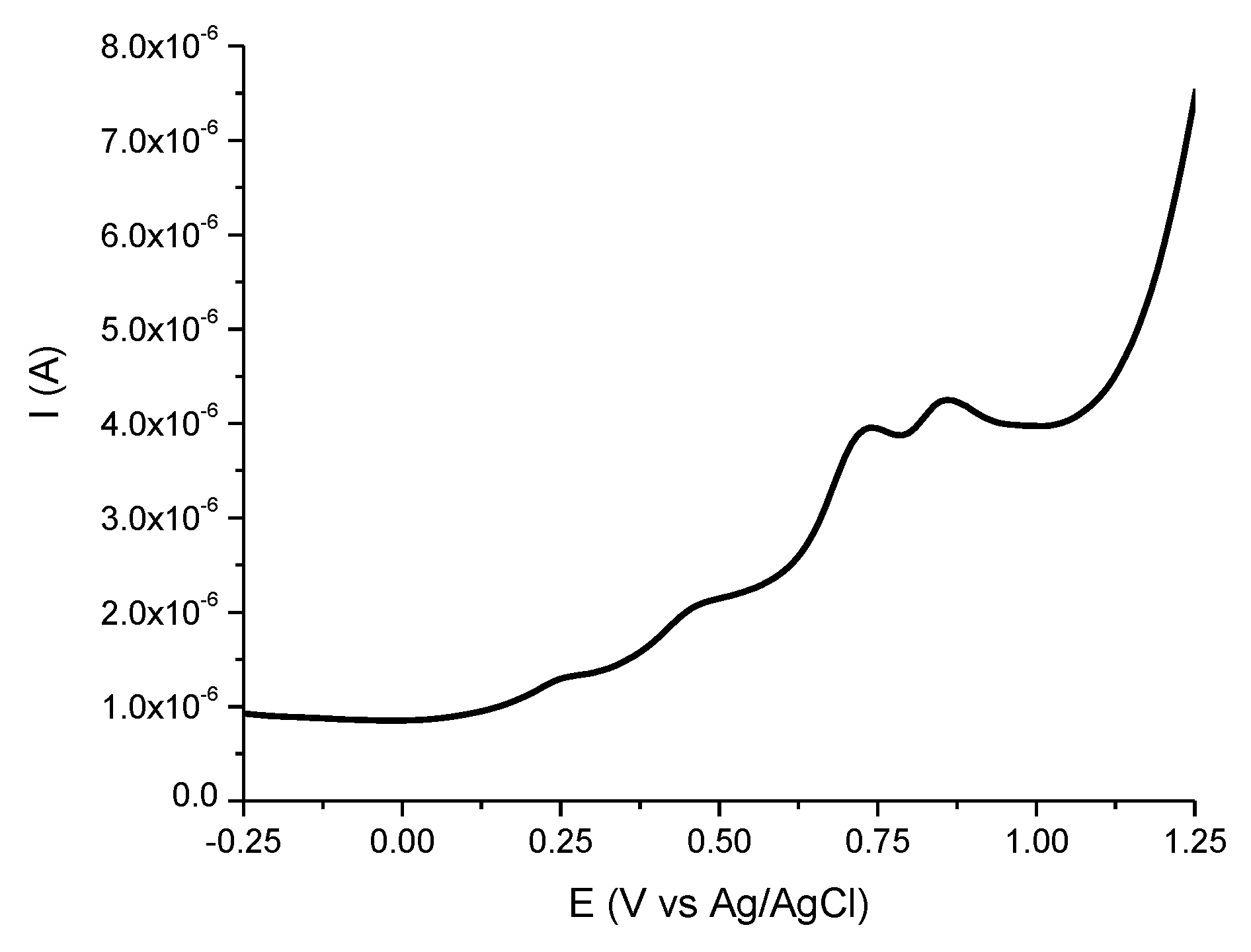
2.1. Total Phenols, Flavonoids Content, and Antioxidant Capacity of SM
2.2. Antimicrobial Activity of the SM Extract
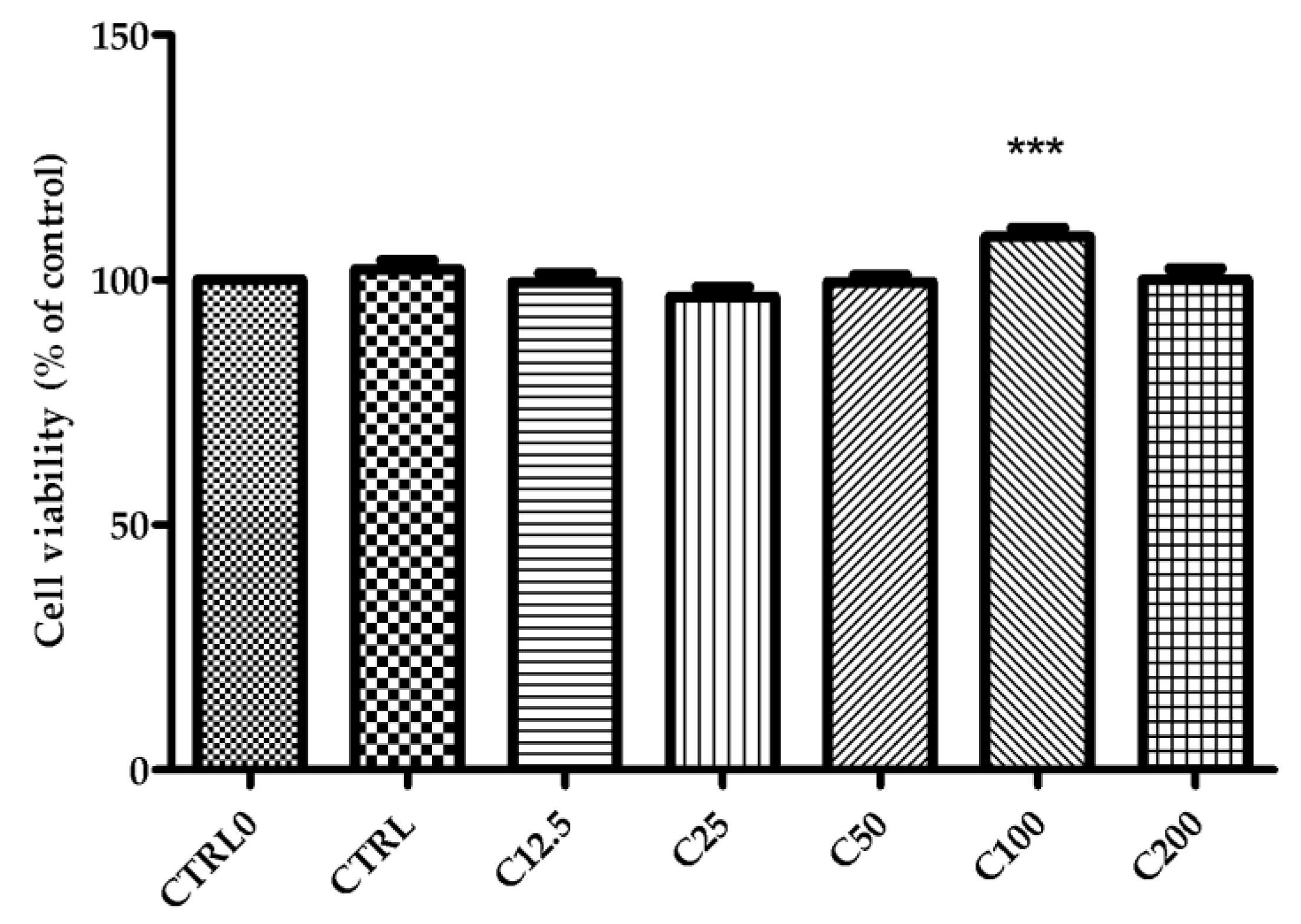
2.3. Cell Viability Assay
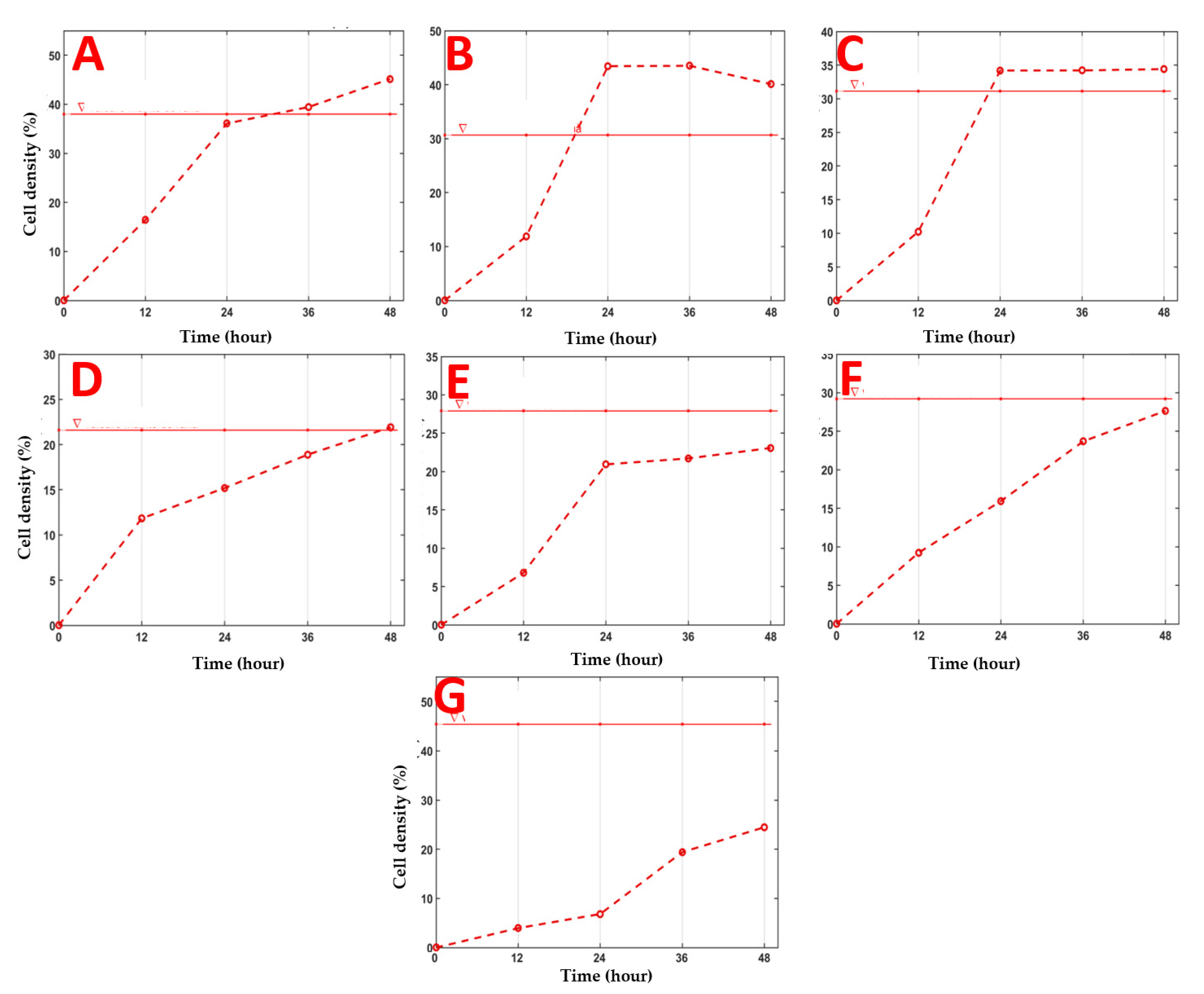
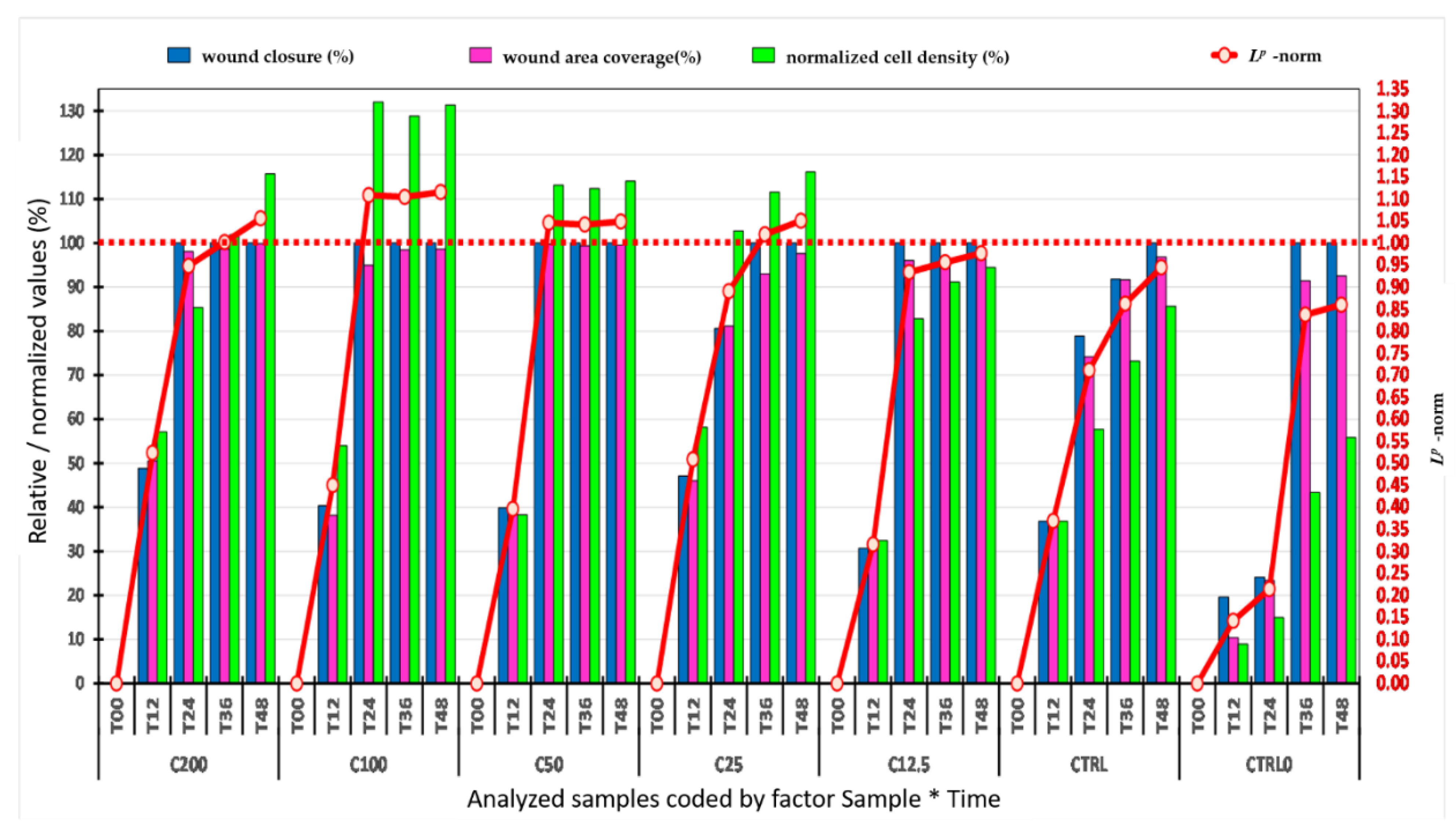
2.4. Evaluation of the Wound-Healing Effect of SM Extract Using In Vitro Scratch Assay
2.4.1. Image Analysis
2.4.2. ANOVA Statistics
3. Discussion
4. Materials and Methods
4.1. The Plant Material and Extract Preparation
4.2. Phytochemical Analysis
4.2.1. Determination of Total Phenols Content
4.2.2. Determination of Total Flavonoid Content
4.2.3. Determination of Antioxidant Capacity by FRAP Assay
4.2.4. Determination of Antioxidant Capacity by Electrochemical Method
4.3. Determination of Antimicrobial Activity
4.4. In Vitro Determination of the Healing and Proliferative Biological Effects of the SM Extract Using the Scratch Method
4.4.1. Cell Culture and Treatments
4.4.2. Cell Viability Assay
4.4.3. Scratch Assay
4.5. Statistical Analysis
4.5.1. Image Analysis
4.5.2. Univariate Statistical Analysis (ANOVA)
4.5.3. Lp Method
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moreno Romero, J.A.; Pérez Muñoz, N.; Campoy Sánchez, A.; Urbano Carrillo, M.; Fernández Figueras, M.T. Derm Dotting: A New Technique That Improves Diagnostic Precision in the Evaluation of Skin Lesions. Actas Dermosifiliogr. 2019, 110, 193–196. [Google Scholar] [CrossRef]
- Tracy, L.E.; Minasian, R.A.; Caterson, E.J. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv. Wound Care 2016, 5, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-J.; Oh, J.W.; Spandau, D.F.; Tholpady, S.; Diaz, J.; Schroeder, L.J.; Offutt, C.D.; Glick, A.B.; Plikus, M.V.; Koyama, S.; et al. Estrogen Modulates Mesenchyme-Epidermis Interactions in the Adult Nipple. Development 2017, 144, 1498–1509. [Google Scholar] [CrossRef]
- Cole, M.A.; Quan, T.; Voorhees, J.J.; Fisher, G.J. Extracellular Matrix Regulation of Fibroblast Function: Redefining Our Perspective on Skin Aging. J. Cell Commun. Signal 2018, 12, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Givol, O.; Kornhaber, R.; Visentin, D.; Cleary, M.; Haik, J.; Harats, M. A Systematic Review of Calendula Officinalis Extract for Wound Healing. Wound Repair Regen. 2019, 27, 548–561. [Google Scholar] [CrossRef]
- Rognoni, E.; Gomez, C.; Pisco, A.O.; Rawlins, E.L.; Simons, B.D.; Watt, F.M.; Driskell, R.R. Inhibition of β-Catenin Signalling in Dermal Fibroblasts Enhances Hair Follicle Regeneration during Wound Healing. Development 2016, 143, 2522–2535. [Google Scholar] [CrossRef]
- Miere (Groza), F.; Fritea, L.; Cavalu, S.; Vicaş, S.I. Formulation, Characterization, and Advantages of Using Liposomes in Multiple Therapies. Pharmacophore 2020, 11, 1–12. [Google Scholar]
- Miere (Groza), F.; Vicas, S.I.; Timar, A.V.; Ganea, M.; Zdrinca, M.; Cavalu, S.; Fritea, L.; Vicas, L.; Muresan, M.; Pallag, A.; et al. Preparation and Characterization of Two Different Liposomal Formulations with Bioactive Natural Extract for Multiple Applications. Processes 2021, 9, 432. [Google Scholar] [CrossRef]
- Vang Mouritzen, M.; Jenssen, H. Optimized Scratch Assay for In Vitro Testing of Cell Migration with an Automated Optical Camera. J. Vis. Exp. 2018, 138, 57691. [Google Scholar] [CrossRef] [PubMed]
- den Dekker, A.; Davis, F.M.; Kunkel, S.L.; Gallagher, K.A. Targeting Epigenetic Mechanisms in Diabetic Wound Healing. Transl. Res. 2019, 204, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Hesketh, M.; Sahin, K.B.; West, Z.E.; Murray, R.Z. Macrophage Phenotypes Regulate Scar Formation and Chronic Wound Healing. Int. J. Mol. Sci. 2017, 18, 1545. [Google Scholar] [CrossRef] [PubMed]
- Antonescu, A.I.; Jurca, T.; Gligor, F.; Craciun, I.; Fritea, L.; Patay, E.B.; Muresan, M.; Udeanu, D.I.; Ionita, C.A.; Antonescu, A.; et al. Comparative Phytochemical and Antioxidative Characterization of Trifolium Pratense L. and Ocimum Basilicum L. Farmacia 2019, 67, 146–153. [Google Scholar] [CrossRef]
- Dobjanschi, L.; Patay, E.B.; Fritea, L.; Tamas, M.; Jurca, A.; Jurca, C.; Zdrinca, M. Morpho-Histological Studies of Three Romanian Solidago Species. Pak. J. Bot. 2021, 53, 161–166. [Google Scholar] [CrossRef]
- Oladeji, O.S.; Oyebamiji, A.K. Stellaria Media (L.) Vill.—A Plant with Immense Therapeutic Potentials: Phytochemistry and Pharmacology. Heliyon 2020, 6, e04150. [Google Scholar] [CrossRef]
- Demján, V.; Kiss, T.; Siska, A.; Szabó, M.R.; Sárközy, M.; Földesi, I.; Csupor, D.; Csont, T. Effect of Stellaria media Tea on Lipid Profile in Rats. Evid.-Based Complement. Altern. Med. 2020, 2020, 5109328. [Google Scholar] [CrossRef]
- Antonescu (Mintas), A.-I.; Miere (Groza), F.; Fritea, L.; Ganea, M.; Zdrinca, M.; Dobjanschi, L.; Antonescu, A.; Vicas, S.I.; Bodog, F.; Sindhu, R.K.; et al. Perspectives on the Combined Effects of Ocimum Basilicum and Trifolium Pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants 2021, 10, 1390. [Google Scholar] [CrossRef]
- Rani, N.; Vasudeva, N.; Sharma, S.K. Quality Assessment and Anti-Obesity Activity of Stellaria media (Linn.) Vill. BMC Complement. Altern. Med. 2012, 12, 145. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.H.; Bellefeuille, S.D.; Miller, H.N.; Gaborski, T.R. Extended Live-Tracking and Quantitative Characterization of Wound Healing and Cell Migration with SiR-Hoechst. Exp. Cell Res. 2018, 373, 198–210. [Google Scholar] [CrossRef]
- Browning, A.P.; Jin, W.; Plank, M.J.; Simpson, M.J. Identifying Density-Dependent Interactions in Collective Cell Behaviour. J. R. Soc. Interface 2020, 17, 20200143. [Google Scholar] [CrossRef]
- Antonescu (Mintaș), I.A.; Antonescu, A.; Miere (Groza), F.; Fritea, L.; Teușdea, A.C.; Vicaș, L.; Vicaș, S.I.; Brihan, I.; Domuța, M.; Zdrinca, M.; et al. Evaluation of Wound Healing Potential of Novel Hydrogel Based on Ocimum Basilicum and Trifolium Pratense Extracts. Processes 2021, 9, 2096. [Google Scholar] [CrossRef]
- Miere (Groza), F.; Teusdea, A.C.; Laslo, V.; Fritea, L.; Moldovan, L.; Costea, T.; Uivarosan, D.; Vicas, S.I.; Pallag, A. Natural Polymeric Beads for Encapsulation of Stellaria media Extract with Antioxidant Properties. Mat. Plast. 2019, 56, 671–679. [Google Scholar] [CrossRef]
- Park, C.-H.; Min, S.-Y.; Yu, H.-W.; Kim, K.; Kim, S.; Lee, H.-J.; Kim, J.-H.; Park, Y.-J. Effects of Apigenin on RBL-2H3, RAW264.7, and HaCaT Cells: Anti-Allergic, Anti-Inflammatory, and Skin-Protective Activities. Int. J. Mol. Sci. 2020, 21, 4620. [Google Scholar] [CrossRef]
- Choi, S.; Youn, J.; Kim, K.; Joo, D.H.; Shin, S.; Lee, J.; Lee, H.K.; An, I.-S.; Kwon, S.; Youn, H.J.; et al. Apigenin Inhibits UVA-Induced Cytotoxicity in Vitro and Prevents Signs of Skin Aging In Vivo. Int. J. Mol. Med. 2016, 38, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Kooti, W.; Daraei, N. A Review of the Antioxidant Activity of Celery (Apium Graveolens L). J. Evid.-Based Complement. Altern. Med. 2017, 22, 1029–1034. [Google Scholar] [CrossRef]
- Lopez-Jornet, P.; Camacho-Alonso, F.; Gómez-Garcia, F.; Molina Miñano, F.; Cañas, X.; Serafín, A.; Castillo, J.; Vicente-Ortega, V. Effects of Potassium Apigenin and Verbena Extract on the Wound Healing Process of SKH-1 Mouse Skin. Int. Wound J. 2014, 11, 489–495. [Google Scholar] [CrossRef]
- Gholampour, F.; Mohammadi, Z.; Karimi, Z.; Owji, S.M. Protective Effect of Genistein in a Rat Model of Ischemic Acute Kidney Injury. Gene 2020, 753, 144789. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Wu, T.; Liu, T.; Yang, H.; Ding, X.; Chen, Y.; Mu, Y. Vicenin-2 Ameliorates Oxidative Damage and Photoaging via Modulation of MAPKs and MMPs Signaling in UVB Radiation Exposed Human Skin Cells. J. Photochem. Photobiol. B 2019, 190, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.S.; Arulselvan, P.; Ng, S.-F.; Taib, C.N.M.; Sarian, M.N.; Fakurazi, S. Healing Effect of Vicenin-2 (VCN-2) on Human Dermal Fibroblast (HDF) and Development VCN-2 Hydrocolloid Film Based on Alginate as Potential Wound Dressing. BioMed Res. Int. 2020, 2020, 4730858. [Google Scholar] [CrossRef]
- Fan, X.; Du, K.; Li, N.; Zheng, Z.; Qin, Y.; Liu, J.; Sun, R.; Su, Y. Evaluation of Anti-Nociceptive and Anti-Inflammatory Effect of Luteolin in Mice. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 351–364. [Google Scholar] [CrossRef]
- Qian, W.; Liu, M.; Fu, Y.; Zhang, J.; Liu, W.; Li, J.; Li, X.; Li, Y.; Wang, T. Antimicrobial Mechanism of Luteolin against Staphylococcus aureus and Listeria monocytogenes and Its Antibiofilm Properties. Microb. Pathog. 2020, 142, 104056. [Google Scholar] [CrossRef]
- De Luca, C.; Mikhal’chik, E.V.; Suprun, M.V.; Papacharalambous, M.; Truhanov, A.I.; Korkina, L.G. Skin Antiageing and Systemic Redox Effects of Supplementation with Marine Collagen Peptides and Plant-Derived Antioxidants: A Single-Blind Case-Control Clinical Study. Oxid. Med. Cell Longev. 2016, 2016, 4389410. [Google Scholar] [CrossRef]
- Stanely Mainzen Prince, P.; Rajakumar, S.; Dhanasekar, K. Protective Effects of Vanillic Acid on Electrocardiogram, Lipid Peroxidation, Antioxidants, Proinflammatory Markers and Histopathology in Isoproterenol Induced Cardiotoxic Rats. Eur. J. Pharmacol. 2011, 668, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Taqvi, S.; Ahmed Bhat, E.; Sajjad, N.; Sabir, J.S.M.; Qureshi, A.; Rather, I.A.; Rehman, S. Protective Effect of Vanillic Acid in Hydrogen Peroxide-Induced Oxidative Stress in D.Mel-2 Cell Line. Saudi J. Biol. Sci. 2021, 28, 1795–1800. [Google Scholar] [CrossRef]
- Wang, C.; Cai, W.-D.; Yao, J.; Wu, L.-X.; Li, L.; Zhu, J.; Yan, J.-K. Conjugation of Ferulic Acid onto Pectin Affected the Physicochemical, Functional and Antioxidant Properties. J. Sci. Food Agric. 2020, 100, 5352–5362. [Google Scholar] [CrossRef] [PubMed]
- Carbone, C.; Caddeo, C.; Grimaudo, M.A.; Manno, D.E.; Serra, A.; Musumeci, T. Ferulic Acid-NLC with Lavandula Essential Oil: A Possible Strategy for Wound-Healing? Nanomaterials 2020, 10, 898. [Google Scholar] [CrossRef]
- Gül, M.; Günay, A.; Tanik, A. An Evaluation of the Effects of Caffeic Acid Phenethyl Ester and Ankaferd Blood Stopper on Secondary Wound Healing of Oral Mucosal Tissue. Turk. J. Med. Sci. 2020, 50, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Romana-Souza, B.; Dos Santos, J.S.; Monte-Alto-Costa, A. Caffeic Acid Phenethyl Ester Promotes Wound Healing of Mice Pressure Ulcers Affecting NF-ΚB, NOS2 and NRF2 Expression. Life Sci. 2018, 207, 158–165. [Google Scholar] [CrossRef]
- Juneja, K.; Mishra, R.; Chauhan, S.; Gupta, S.; Roy, P.; Sircar, D. Metabolite Profiling and Wound-Healing Activity of Boerhavia Diffusa Leaf Extracts Using In Vitro and In Vivo Models. J. Tradit Complement. Med. 2020, 10, 52–59. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Kao, C.-L.; Liu, C.-M. The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. Int. J. Mol. Sci. 2018, 19, 2729. [Google Scholar] [CrossRef]
- Parzonko, A.; Kiss, A.K. Caffeic Acid Derivatives Isolated from Galinsoga Parviflora Herb Protected Human Dermal Fibroblasts from UVA-Radiation. Phytomedicine 2019, 57, 215–222. [Google Scholar] [CrossRef]
- Wu, Y.; Liang, S.; Zhang, M.; Wang, Z.; Wang, Z.; Ren, X. The Effect of Chlorogenic Acid on Bacillus Subtilis Based on Metabolomics. Molecules 2020, 25, 4038. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Daglia, M.; Orlando, V.; D’Urso, E.; Saadat, S.H.; Novellino, E.; Nabavi, S.F.; Nabavi, S.M. Coffee and Depression: A Short Review of Literature. Curr. Pharm. Des. 2015, 21, 5034–5040. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.; Gośliński, M.; Wojtowicz, E.; Przygoński, K. Antioxidant Properties and Phenolic Compounds of Vitamin C-Rich Juices. J. Food Sci. 2018, 83, 2237–2246. [Google Scholar] [CrossRef]
- Peh, P.; Lim, N.S.J.; Blocki, A.; Chee, S.M.L.; Park, H.C.; Liao, S.; Chan, C.; Raghunath, M. Simultaneous Delivery of Highly Diverse Bioactive Compounds from Blend Electrospun Fibers for Skin Wound Healing. Bioconjug. Chem. 2015, 26, 1348–1358. [Google Scholar] [CrossRef] [PubMed]
- Emirdağ-Öztürk, S.; Babahan, İ.; Özmen, A. Synthesis, Characterization and in Vitro Anti-Neoplastic Activity of Gypsogenin Derivatives. Bioorg. Chem. 2014, 53, 15–23. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Fan, Y.; Yang, B.; Huang, C. Radix Stellariae Extract Prevents High-Fat-Diet-Induced Obesity in C57BL/6 Mice by Accelerating Energy Metabolism. PeerJ 2017, 5, e3305. [Google Scholar] [CrossRef]
- Oyebanji; Bukola, O.; Saba; Bernard, A. Phytochemistry and in Vitro Anti-Oxidant Activities of Stellaria media, Cajanus Cajan and Tetracera Potatoria Methanolic Extracts. J. Med. Plants Res. 2011, 5, 6622–6627. [Google Scholar] [CrossRef]
- Hernández, I.; Alegre, L.; Van Breusegem, F.; Munné-Bosch, S. How Relevant Are Flavonoids as Antioxidants in Plants? Trends Plant Sci. 2009, 14, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.A.; Khan, M.R.; Nadhman, A. Antileishmanial, Toxicity, and Phytochemical Evaluation of Medicinal Plants Collected from Pakistan. BioMed Res. Int. 2014, 2014, 384204. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A. Phytochemical and Pharmacological Potential of Genus Stellaria: A Review. J. Pharm. Res. 2012, 5, 3591–3596. [Google Scholar]
- Arora, D.; Sharma, A. Isolation and characterization of the chemical constituents of Stellaria media linn. Int. J. Pharm. Sci. Res. 2014, 5, 3669–3673. [Google Scholar]
- Veloso, D.J.; Abrão, F.; Martins, C.H.G.; Bronzato, J.D.; Gomes, B.P.F.A.; Higino, J.S.; Sampaio, F.C. Potential Antibacterial and Anti-Halitosis Activity of Medicinal Plants against Oral Bacteria. Arch. Oral. Biol. 2020, 110, 104585. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Song, J.; Shi, Y.; Wang, C.; Chen, B.; Xie, D.; Jia, X. Anti-Hepatitis B Virus Activity of Chickweed [Stellaria Media (L.) Vill.] Extracts in HepG2.2.15 Cells. Molecules 2012, 17, 8633–8646. [Google Scholar] [CrossRef] [PubMed]
- Fritea, L.; Pasca, P.M.; Vlase, L.; Gheldiu, A.-M.; Moldovan, L.; Banica, F.; Dobjanschi, L.; Cavalu, S. Electrochemical Methods for Evaluation of Antioxidant Properties of Propolis Extract Incorporated in Chitosan Nanoparticles. Mater. Plast. 2021, 57, 96–108. [Google Scholar] [CrossRef]
- Sarmah, P.; Sarma, A.; Kashyap, D.; Mahanta, M.; Medhi, P. Nutraceutical Properties of Stellaria media (L.) Vill. and Persicaria Chinensis (L.) H. Gross under Brahmaputra Valley Agro-Climatic Condition. Ann. Plant Sci. 2014, 3, 779–782. [Google Scholar]
- Ristivojević, P.; Trifković, J.; Stanković, D.M.; Radoičić, A.; Manojlović, D.; Milojković-Opsenica, D. Cyclic Voltammetry and UV/Vis Spectroscopy in Combination with Multivariate Data Analysis for the Assessment of Authenticity of Poplar Type Propolis. J. Apic. Res. 2017, 56, 559–568. [Google Scholar] [CrossRef]
- Banica, F.; Bungau, S.; Tit, D.M.; Behl, T.; Otrisal, P.; Nechifor, A.C.; Gitea, D.; Pavel, F.-M.; Nemeth, S. Determination of the Total Polyphenols Content and Antioxidant Activity of Echinacea Purpurea Extracts Using Newly Manufactured Glassy Carbon Electrodes Modified with Carbon Nanotubes. Processes 2020, 8, 833. [Google Scholar] [CrossRef]
- Xu, Z.; Hsia, H.C. The Impact of Microbial Communities on Wound Healing: A Review. Ann. Plast Surg. 2018, 81, 113–123. [Google Scholar] [CrossRef]
- Gao, Y.; Song, N.; Liu, W.; Dong, A.; Wang, Y.-J.; Yang, Y.-W. Construction of Antibacterial N-Halamine Polymer Nanomaterials Capable of Bacterial Membrane Disruption for Efficient Anti-Infective Wound Therapy. Macromol. Biosci. 2019, 19, 1970010. [Google Scholar] [CrossRef]
- Bonjar, S. Evaluation of Antibacterial Properties of Some Medicinal Plants Used in Iran. J. Ethnopharmacol. 2004, 94, 301–305. [Google Scholar] [CrossRef]
- Chandra, S.; Rawat, D.S. Medicinal Plants of the Family Caryophyllaceae: A Review of Ethno-Medicinal Uses and Pharmacological Properties. Integr. Med. Res. 2015, 4, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Barakat, A.B.; Shoman, S.A.; Dina, N.; Alfarouk, O.R. Antiviral Activity and Mode of Action of Dianthus caryophyllus L. and Lupinus termes L. Seed Extracts against in Vitro Herpes Simplex and Hepatitis A Viruses Infection. J. Microbiol. Antimicrob. 2010, 2, 23–29. [Google Scholar] [CrossRef]
- Li, P.; Huang, X.; Li, F.; Wang, X.; Zhou, W.; Liu, H.; Ma, T.; Zhang, T.; Guo, D.; Yao, D.; et al. Robust Granger Analysis in Lp Norm Space for Directed EEG Network Analysis. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 1959–1969. [Google Scholar] [CrossRef] [PubMed]
- Ávila-Salas, F.; Marican, A.; Pinochet, S.; Carreño, G.; Valdés, O.; Venegas, B.; Donoso, W.; Cabrera-Barjas, G.; Vijayakumar, S.; Durán-Lara, E.F. Film Dressings Based on Hydrogels: Simultaneous and Sustained-Release of Bioactive Compounds with Wound Healing Properties. Pharmaceutics 2019, 11, 447. [Google Scholar] [CrossRef]
- Valle, K.Z.M.; Saucedo Acuña, R.A.; Ríos Arana, J.V.; Lobo, N.; Rodriguez, C.; Cuevas-Gonzalez, J.C.; Tovar-Carrillo, K.L. Natural Film Based on Pectin and Allantoin for Wound Healing: Obtaining, Characterization, and Rat Model. BioMed Res. Int. 2020, 2020, 6897497. [Google Scholar] [CrossRef]
- Vicas, S.; Prokisch, J.; Rugina, O.D.; Socaciu, C. Hydrophilic and Lipophilic Antioxidant Activities of Mistletoe (Viscum album) as Determined by FRAP Method. Not. Bot. Horti. Agrobot. Cluj-Napoca. 2009, 37, 112–116. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. [14] Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In Methods in Enzymology; Oxidants and Antioxidants Part A; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Tünde, J.; Vicas, L.; Tóth, I.; Braun, M.; Marian, E.; Teuşdea, A.; Vicaş, S.; Mureșan, M. Mineral Elements Profile, Bioactive Compounds and Antioxidant Capacity of Wild Blueberry and of Pharmaceutical Preparations from Blueberry (Vaccinium myrtillus). Farmacia 2016, 64, 581–587. [Google Scholar]
- Kim, D.-O.; Chun, O.K.; Kim, Y.J.; Moon, H.-Y.; Lee, C.Y. Quantification of Polyphenolics and Their Antioxidant Capacity in Fresh Plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.; Cano, A.; Acosta, M. The Hydrophilic and Lipophilic Contribution to Total Antioxidant Activity. Food Chem. 2001, 73, 239–244. [Google Scholar] [CrossRef]
- Cavalu, S.; Damian, G. Rotational Correlation Times of 3-Carbamoyl-2,2,5,5-Tetramethyl-3-Pyrrolin-1-Yloxy Spin Label with Respect to Heme and Nonheme Proteins. Biomacromolecules 2003, 4, 1630–1635. [Google Scholar] [CrossRef] [PubMed]
- Fritea, L.; Banica, F.; Costea, T.; Moldovan, L.; Iovan, C.; Cavalu, S. A Gold Nanoparticles—Graphene Based Electrochemical Sensor for Sensitive Determination of Nitrazepam. J. Electroanal. Chem. 2018, 830–831, 63–71. [Google Scholar] [CrossRef]
- El Moussaoui, A.; Jawhari, F.Z.; Almehdi, A.M.; Elmsellem, H.; Fikri Benbrahim, K.; Bousta, D.; Bari, A. Antibacterial, Antifungal and Antioxidant Activity of Total Polyphenols of Withania Frutescens.L. Bioorg. Chem. 2019, 93, 103337. [Google Scholar] [CrossRef]
- Martinotti, S.; Ranzato, E. Scratch Wound Healing Assay. Methods Mol. Biol. 2020, 2109, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Leith, M.; Tu, R.; Tahim, G.; Sudra, A.; Bhargava, S. Effects of Diluents on Cell Culture Viability Measured by Automated Cell Counter. PLoS ONE 2017, 12, e0173375. [Google Scholar] [CrossRef] [PubMed]
- Eggers, B.; Marciniak, J.; Memmert, S.; Kramer, F.J.; Deschner, J.; Nokhbehsaim, M. The Beneficial Effect of Cold Atmospheric Plasma on Parameters of Molecules and Cell Function Involved in Wound Healing in Human Osteoblast-like Cells In Vitro. Odontology 2020, 108, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, J.; Shin, Y.-K.; Kim, K.-Y. Gentisic Acid Stimulates Keratinocyte Proliferation through ERK1/2 Phosphorylation. Int. J. Med. Sci. 2020, 17, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Muniandy, K.; Gothai, S.; Tan, W.S.; Kumar, S.S.; Mohd Esa, N.; Chandramohan, G.; Al-Numair, K.S.; Arulselvan, P. In Vitro Wound Healing Potential of Stem Extract of Alternanthera Sessilis. Evid.-Based Complement. Altern. Med. 2018, 2018, e3142073. [Google Scholar] [CrossRef]
- Advanced Wound Healing Assay Workflow Using Automated Scratch Wound Creation, High Contrast Brightfield and Fluorescence Kinetic Imaging Live Cell Imaging|Semantic Scholar. Available online: https://www.semanticscholar.org/paper/Advanced-Wound-Healing-Assay-Workflow-using-Scratch/a6fa0e609f87625c4bd70f127cef7ed7a3748d6c (accessed on 29 September 2021).
- Baiazitova, L.; Skopalik, J.; Chmelik, J.; Zumberg, I.; Cmiel, V.; Polakova, K.; Provaznik, I. The Effect of Rhodamine-Derived Superparamagnetic Maghemite Nanoparticles on the Motility of Human Mesenchymal Stem Cells and Mouse Embryonic Fibroblast Cells. Molecules 2019, 24, 1192. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, A.; Xu, J.; Wang, L. Lp-Norm Regularization in Volumetric Imaging of Cardiac Current Sources. Comput. Math. Methods Med. 2013, 2013, 276478. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Feng, G.; Zhu, J. An Algorithm of l 1-Norm and l 0-Norm Regularization Algorithm for CT Image Reconstruction from Limited Projection. Int. J. Biomed. Imaging 2020, 2020, 8873865. [Google Scholar] [CrossRef] [PubMed]

| Total Phenols (mg GAE/g dw) | Flavonoids (mg QE/g dw) | FRAP 1 (µg AAE/mL) | AEAC 2 (µg/mL) |
|---|---|---|---|
| 17.19 ± 1.32 | 7.28 ± 1.180 | 5.54 ± 0.49 | 26.00 ± 0.02 |
| Antibiotics/Extract | Inhibition Zone (mm) | |
|---|---|---|
| E. coli | S. aureus | |
| CAZ | 28.33 ± 1.43 | n.d. |
| FOX | n.d. | 35.66 ± 2.30 |
| P | n.d. | 34.33 ± 1.93 |
| CIP | 30.66 ± 2.12 | 27.33 ± 2.01 |
| GN | 20.66 ± 1.45 | 23.66 ± 1.99 |
| DOX | 20.66 ± 1.62 | n.d. |
| F | 22.66 ± 1.23 | 24.33 ± 2.14 |
| SM extract (5 µg/mL) | 7.66 ± 0.85 | 10.66 ± 0.86 |
| SM extract (10 µg/mL) | 8.33 ± 0.56 | 11.66 ± 0.99 |
| SM extract (15 µg/mL) | 9.66 ± 0.57 | 15.33 ± 1.27 |
| Compounds Identified in SM | Biological Activities | References | ||
|---|---|---|---|---|
| Antioxidant | Antimicrobial | Wound Healing/Skin Protection | ||
| Flavonoids | ||||
| Apigenin | √ | - | √ | [22,23,24,25] |
| Genistein | √ | - | - | [26] |
| Vincenin-2 | √ | - | √ | [27,28] |
| Luteolin | √ | √ | - | [24,29,30,31] |
| Phenolic acid | ||||
| Vanillic acid | √ | √ | - | [32,33] |
| Ferulic acid | √ | - | √ | [34,35] |
| Caffeic acid | √ | √ | √ | [36,37,38,39,40] |
| Chlorogenic acid | √ | √ | - | [41,42] |
| Vitamin | ||||
| Vitamin C | √ | - | √ | [43,44] |
| Triterpenoid saponin | ||||
| Gypsogenin | √ | - | - | [45] |
| Alkaloids | ||||
| Stellarine A, B, C | - | √ | - | [46] |
| Dichotomine B, H, L | - | √ | - | [46] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miere, F.; Teușdea, A.C.; Laslo, V.; Cavalu, S.; Fritea, L.; Dobjanschi, L.; Zdrinca, M.; Zdrinca, M.; Ganea, M.; Pașc, P.; et al. Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill. Appl. Sci. 2021, 11, 11526. https://doi.org/10.3390/app112311526
Miere F, Teușdea AC, Laslo V, Cavalu S, Fritea L, Dobjanschi L, Zdrinca M, Zdrinca M, Ganea M, Pașc P, et al. Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill. Applied Sciences. 2021; 11(23):11526. https://doi.org/10.3390/app112311526
Chicago/Turabian StyleMiere (Groza), Florina, Alin Cristian Teușdea, Vasile Laslo, Simona Cavalu, Luminița Fritea, Luciana Dobjanschi, Mihaela Zdrinca, Marcel Zdrinca, Mariana Ganea, Priscilla Pașc, and et al. 2021. "Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill" Applied Sciences 11, no. 23: 11526. https://doi.org/10.3390/app112311526
APA StyleMiere, F., Teușdea, A. C., Laslo, V., Cavalu, S., Fritea, L., Dobjanschi, L., Zdrinca, M., Zdrinca, M., Ganea, M., Pașc, P., Memete, A. R., Antonescu, A., Vlad, A. M., & Vicas, S. I. (2021). Evaluation of In Vitro Wound-Healing Potential, Antioxidant Capacity, and Antimicrobial Activity of Stellaria media (L.) Vill. Applied Sciences, 11(23), 11526. https://doi.org/10.3390/app112311526







