Effect of Bioaugmentation with Anaerobic Fungi Isolated from Ruminants on the Hydrolysis of Corn Silage and Phragmites australis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Feedstock
2.2. Anaerobic Fungal Strains, Media, and Culturing Conditions
2.3. Hydrolysis Experiment
2.4. Analytical Methods
2.4.1. Gas Analysis
2.4.2. Liquid Analysis
2.4.3. Fiber Analysis and C:N
2.5. Statistical Analysis
3. Results and Discussion
3.1. Feedstock Characteristics
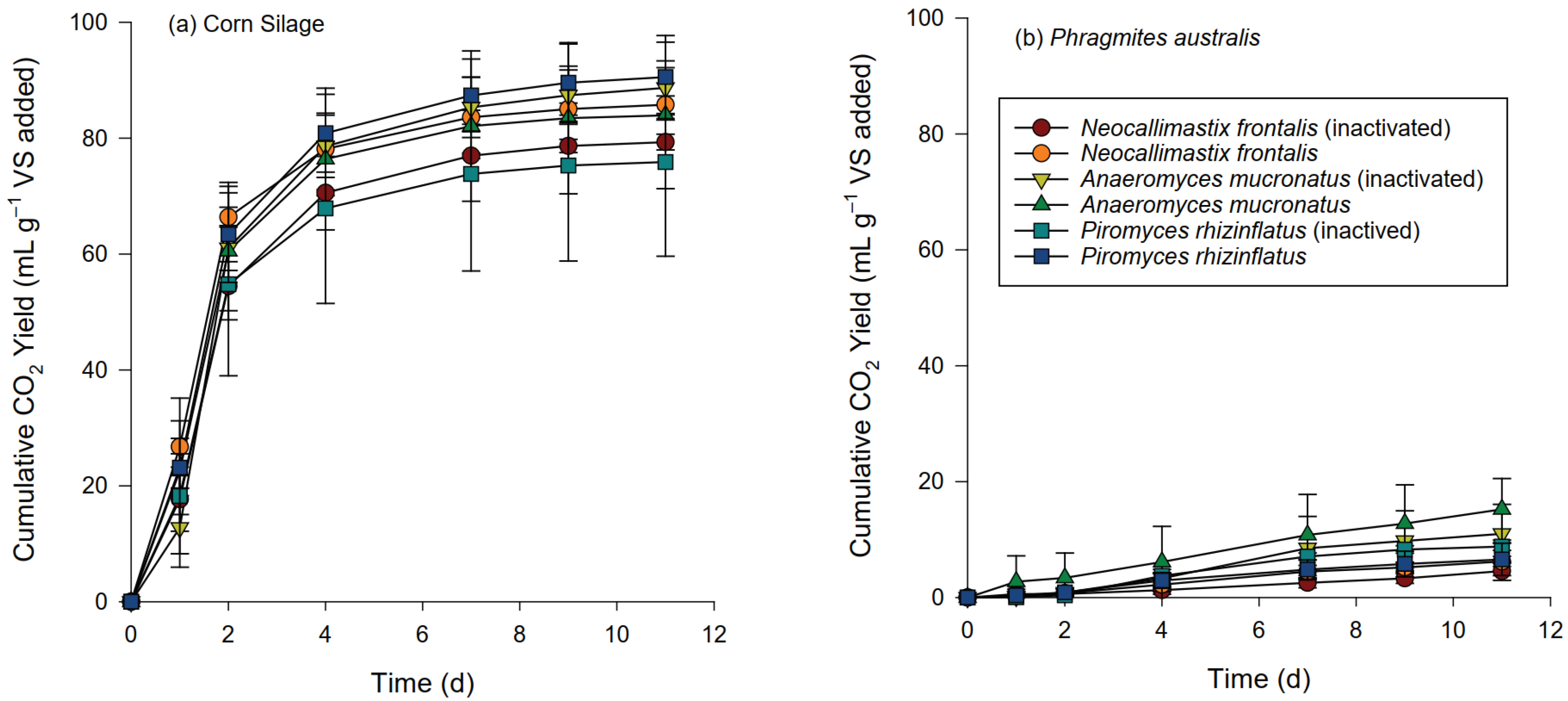
3.2. Gas Production and Composition
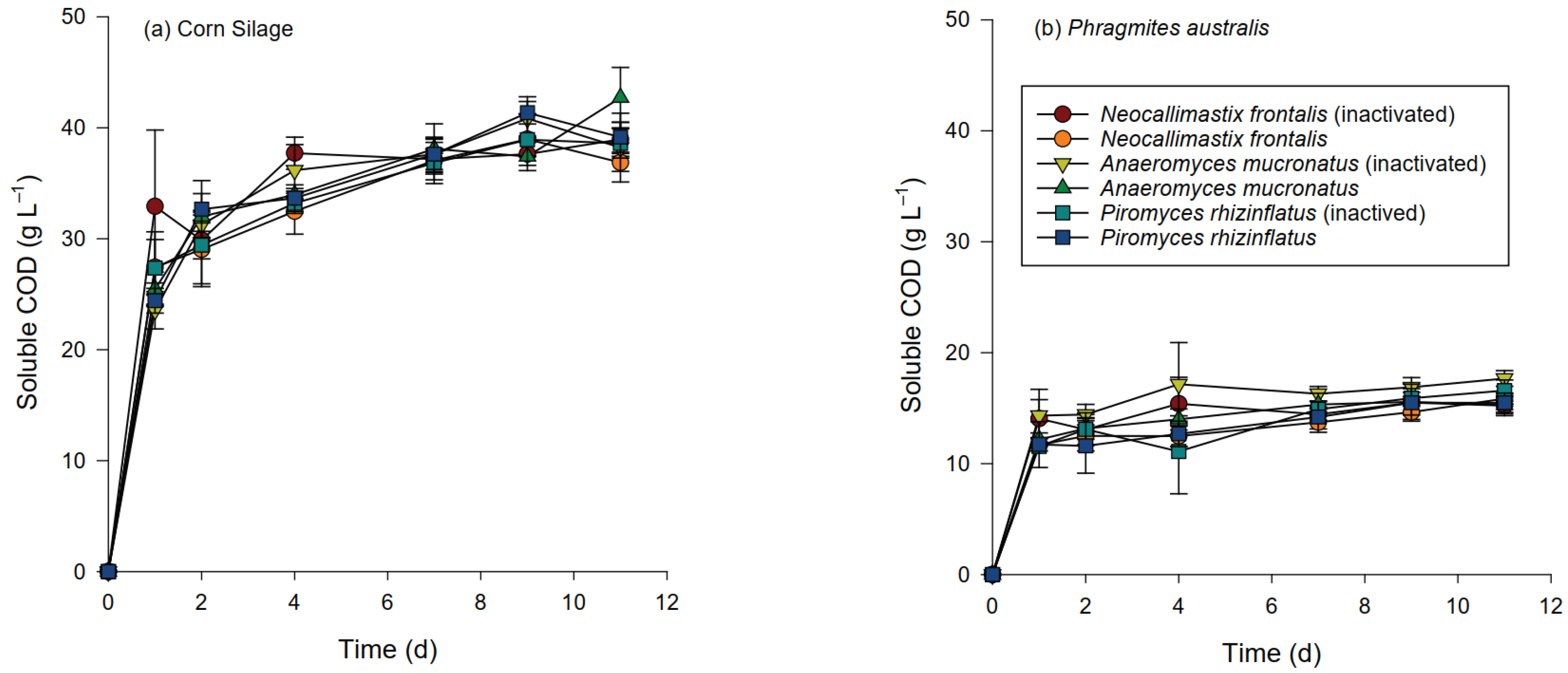
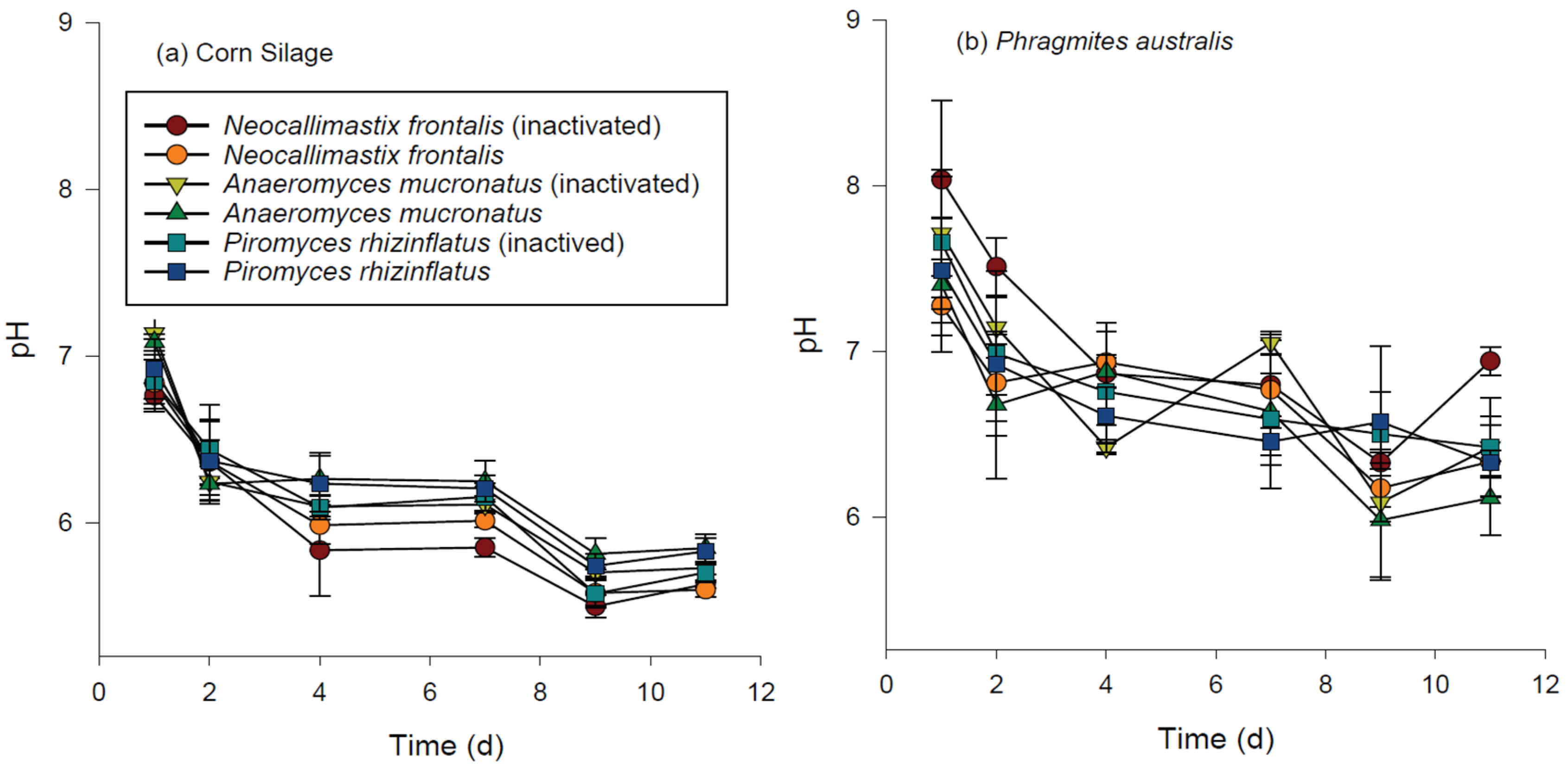
3.3. Chemical Changes during Hydrolysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EBA. EBA Stastical Report 2020; European Biogas Association: Brussels, Belgium, 2020. [Google Scholar]
- Hutňan, M. Maize Silage as Substrate for Biogas Production. In Advances in Silage Production and Utilization; Silva, T.D., Ed.; IntechOpen: Rijeka, Croatia, 2016. [Google Scholar]
- Lask, J.; Guajardo, A.M.; Weik, J.; von Cossel, M.; Lewandowski, I.; Wagner, M. Comparative environmental and economic life cycle assessment of biogas production from perennial wild plant mixtures and maize (Zea mays L.) in southwest Germany. GCB Bioenergy 2020, 12, 571–585. [Google Scholar] [CrossRef]
- Baute, K.; Van Eerd, L.L.; Robinson, D.E.; Sikkema, P.H.; Mushtaq, M.; Gilroyed, B.H. Comparing the Biomass Yield and Biogas Potential of Phragmites australis with Miscanthus x giganteus and Panicum virgatum Grown in Canada. Energies 2018, 11, 2198. [Google Scholar] [CrossRef] [Green Version]
- Vaičekonytė, R.; Kiviat, E.; Nsenga, F.; Ostfeld, A. An exploration of common reed (Phragmites australis) bioenergy potential in North America. Mires Peat 2014, 13, 1–9. [Google Scholar]
- Akula, V.R. Wetland Biomass-Suitable for Biogas Production? Master’s Thesis, Halmsted University, Halmstad, Sweden, 2013. [Google Scholar]
- Wang, C.; Kong, Y.; Hu, R.; Zhou, G. Miscanthus: A fast-growing crop for environmental remediation and biofuel production. GCB Bioenergy 2021, 13, 58–69. [Google Scholar] [CrossRef]
- e Silva, C.F.L.; Schirmer, M.A.; Maeda, R.N.; Barcelos, C.A.; Pereira, N. Potential of giant reed (Arundo donax L.) for second generation ethanol production. Electron. J. Biotechnol. 2015, 18, 10–15. [Google Scholar] [CrossRef] [Green Version]
- Bayané, A.; Guiot, S. Animal digestive strategies versus anaerobic digestion bioprocesses for biogas production from lignocellulosic biomass. Rev. Environ. Sci. Bio/Technol. 2011, 10, 43–62. [Google Scholar] [CrossRef] [Green Version]
- Dollhofer, V.; Podmirseg, S.M.; Callaghan, T.M.; Griffith, G.W.; Fliegerová, K. Anaerobic Fungi and Their Potential for Biogas Production. Adv. Biochem. Eng. Biotechnol. 2015, 151, 41–61. [Google Scholar]
- Wei, Y.Q.; Yang, H.J.; Luan, Y.; Long, R.J.; Wu, Y.J.; Wang, Z.Y. Isolation, identification and fibrolytic characteristics of rumen fungi grown with indigenous methanogen from yaks (Bos grunniens) grazing on the Qinghai-Tibetan Plateau. J. Appl. Microbiol. 2016, 120, 571–587. [Google Scholar] [CrossRef]
- Dollhofer, V.; Podmirseg, S.M.; Callaghan, T.M.; Griffith, G.W.; Fliegerová, K. Anaerobic Fungi and Their Potential for Biogas Production. In Biogas Science and Technology; Guebitz, G.M., Bauer, A., Bochmann, G., Gronauer, A., Weiss, S., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 41–61. [Google Scholar]
- Janusz, G.; Pawlik, A.; Sulej, J.; Swiderska-Burek, U.; Jarosz-Wilkolazka, A.; Paszczynski, A. Lignin degradation: Microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol. Rev. 2017, 41, 941–962. [Google Scholar] [CrossRef] [Green Version]
- Vogel, T.M. Bioaugmentation as a soil bioremediation approach. Curr. Opin. Biotechnol. 1996, 7, 311–316. [Google Scholar] [CrossRef]
- Miah, M.S.; Tada, C.; Sawayama, S. Enhancement of Biogas Production from Sewage Sludge with the Addition of Geobacillus sp. Strain AT1 Culture. Jpn. J. Water Treat. Biol. 2004, 40, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Müller, H.; Trösch, W. Screening of white-rot fungi for biological pretreatment of wheat straw for biogas production. Appl. Microbiol. Biotechnol. 1986, 24, 180–185. [Google Scholar] [CrossRef]
- Saye, L.M.; Navaratna, T.A.; Chong, J.P.; O’Malley, M.A.; Theodorou, M.K.; Reilly, M. The anaerobic fungi: Challenges and opportunities for industrial lignocellulosic biofuel production. Microorganisms 2021, 9, 694. [Google Scholar] [CrossRef] [PubMed]
- Nkemka, V.N.; Gilroyed, B.H.; Yanke, J.; Gruninger, R.; Vedres, D.; McAllister, T.; Hao, X. Bioaugmentation with an anaerobic fungus in a two-stage process for biohydrogen and biogas production using corn silage and cattail. Bioresour. Technol. 2015, 185, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zhang, H.; Zhang, P.; Zhang, G.; Cai, Y.; Wang, Q.; Zhou, Z.; Ding, Y.; Zubair, M. Effect of substrate load on anaerobic fermentation of rice straw with rumen liquid as inoculum: Hydrolysis and acidogenesis efficiency, enzymatic activities and rumen bacterial community structure. Waste Manag. 2021, 124, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Dollhofer, V.; Callaghan, T.M.; Griffith, G.W.; Lebuhn, M.; Bauer, J. Presence and transcriptional activity of anaerobic fungi in agricultural biogas plants. Bioresour. Technol. 2017, 235, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Lowe, S.E.; Theodorou, M.K.; Trinci, A.P.J.; Hespell, R.B. Growth of anaerobic rumen fungi on defined and semi-difined media lacking rumen fluid. J. Gen. Microbiol. 1985, 131, 2225–2229. [Google Scholar]
- Hungate, R.E. The anaerobic mesophilic cellulolytic bacteria. Bacteriol. Rev. 1950, 14, 1–49. [Google Scholar] [CrossRef]
- Nkemka, V.N.; Murto, M. Two-stage anaerobic dry digestion of blue mussel and reed. Renew. Energy 2013, 50, 359–364. [Google Scholar] [CrossRef]
- Gilroyed, B.H.; Li, C.; Hao, X.; Chu, A.; McAllister, T.A. Biohydrogen production from specified risk materials co-digested with cattle manure. Int. J. Hydrogen Energy 2010, 35, 1099–1105. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; APHA-AWWA-WEF: Washington, DC, USA, 2005. [Google Scholar]
- Gilroyed, B.H.; Li, C.; Reuter, T.; Beauchemin, K.A.; Hao, X.; McAllister, T. Influence of distiller’s grains and condensed tannins in the diet of feedlot cattle on biohydrogen production from cattle manure. Int. J. Hydrogen Energy 2015, 40, 6050–6058. [Google Scholar] [CrossRef] [Green Version]
- Gilroyed, B.H.; Reuter, T.; Chu, A.; Hao, X.; Xu, W.; McAllister, T.A. Anaerobic digestion of specified risk materials with cattle manure for biogas production. Bioresour. Technol. 2010, 101, 5780–5785. [Google Scholar] [CrossRef] [PubMed]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Mertens, D.R.; Allen, M.; Carmany, J.; Clegg, J.; Davidowicz, A.; Drouches, M.; Frank, K.; Gambin, D.; Garkie, M.; Gildemeister, B.; et al. Gravimetric determination of amylase-treated neutral detergent fiber in feeds with refluxing in beakers or crucibles: Collaborative study. J. AOAC Int. 2002, 85, 1217–1240. [Google Scholar]
- Rabii, A.; Aldin, S.; Dahman, Y.; Elbeshbishy, E. A review on anaerobic co-digestion with a focus on the microbial populations and the effect of multi-stage digester configuration. Energies 2019, 12, 1106. [Google Scholar] [CrossRef] [Green Version]
- Gruninger, R.J.; Puniya, A.K.; Callaghan, T.M.; Edwards, J.E.; Youssef, N.; Dagar, S.S.; Fliegerova, K.; Griffith, G.W.; Forster, R.; Tsang, A.; et al. Anaerobic fungi (phylum Neocallimastigomycota): Advances in understanding their taxonomy, life cycle, ecology, role and biotechnological potential. Fems. Microbiol. Ecol. 2014, 90, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Borreani, G.I.; Tabacco, E.R.; Schmidt, R.J.; Holmes, B.J.; Muck, R.E. Silage review: Factors affecting dry matter and quality losses in silages. J. Dairy Sci. 2018, 101, 3952–3979. [Google Scholar] [CrossRef] [Green Version]
- Uwineza, C.; Mahboubi, A.; Atmowidjojo, A.C.; Ramadhani, A.N.; Wainaina, S.; Millati, R.; Wikandari, R.; Niklasson, C.; Taherzadeh, M.J. Cultivation of edible filamentous fungus Aspergillus oryzae on volatile fatty acids derived from anaerobic digestion of food waste and cow manure. Bioresour. Technol. 2021, 337, 125410. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Romero-Güiza, M.S.; Mata-Alvarez, J.; Chimenos, J.M.; Astals, S. The effect of magnesium as activator and inhibitor of anaerobic digestion. Waste Manag. 2016, 56, 137–142. [Google Scholar] [CrossRef]
- Veluchamy, C.; Kalamdhad, A.S. Influence of pretreatment techniques on anaerobic digestion of pulp and paper mill sludge: A review. Bioresour. Technol. 2017, 245, 1206–1219. [Google Scholar] [CrossRef] [PubMed]
- Ivarsson, M.; Schnürer, A.; Bengtson, S.; Neubeck, A. Anaerobic fungi: A potential source of biological H2 in the oceanic crust. Front. Microbiol. 2016, 7, 674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hess, M.; Paul, S.S.; Puniya, A.K.; van der Giezen, M.; Shaw, C.; Edwards, J.E.; Fliegerová, K. Anaerobic Fungi: Past, Present, and Future. Front. Microbiol. 2020, 11, 584893. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Shinkai, T.; Koike, S. Ecological and physiological characterization shows that Fibrobacter succinogenes is important in rumen fiber digestion. Folia Microbiol. 2008, 53, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Joblin, K.N.; Matsui, H.; Naylor, G.E.; Ushida, K. Degradation of Fresh Ryegrass by Methanogenic Co-Cultures of Ruminal Fungi Grown in the Presence or Absence of Fibrobacter succinogenes. Curr. Microbiol. 2002, 45, 46–53. [Google Scholar] [CrossRef]
- Dehority, B.A.; Tirabasso, P.A. Antibiosis between Ruminal Bacteria and Ruminal Fungi. Appl. Environ. Microb. 2000, 66, 2921–2927. [Google Scholar] [CrossRef] [Green Version]
- Yıldırım, E.; Ince, O.; Aydin, S.; Ince, B. Improvement of biogas potential of anaerobic digesters using rumen fungi. Renew. Energy 2017, 109, 346–353. [Google Scholar] [CrossRef]
- Aydin, S.; Yıldırım, E.; Ince, O.; Ince, B. Rumen anaerobic fungi create new opportunities for enhanced methane production from microalgae biomass. Algal Res. 2017, 23, 150–160. [Google Scholar] [CrossRef]


| Parameters | Corn Silage | Common Reed |
|---|---|---|
| TS 1 (%) | 33.5 ± 0.6 | 54.7 ± 0.8 |
| VS 2 (% TS) | 96.3 ± 0.6 | 94.9 ± 0.4 |
| VS added (g) | 30 | 30 |
| Moisture content (%) | 66 ± 0.6 | 44.3 ± 0.8 |
| Total carbon: Total nitrogen ratio | 31.2 ± 0.1 | 26.3 ± 0.7 |
| Hemicellulose (% of TS) | 12 ± 4.3 | 28.7 ± 0.4 |
| Cellulose (% of TS) | 17.2 ± 1.8 | 38.7 ± 0.4 |
| ADL 3 (% TS) | 2.7 ± 0.0 | 7.9 ± 0.3 |
| Substrate | Fungal Species | Active/Inactivated | NH3 mg L−1 | Na+ mg L−1 | K+ mg L−1 | Mg2+ mg L−1 | Ca2+ mg L−1 |
|---|---|---|---|---|---|---|---|
| Corn Silage | Neocallimastix frontalis | Inactivated | N/A | 0.87 ± 0.18 | 2.14 ± 0.13 | 0.14 ± 0.00 | 0.17 ± 0.00 |
| Active | N/A | 0.80 ± 0.08 | 2.20 ± 0.08 | 0.17 ± 0.02 | 0.23 ± 0.02 | ||
| Anaeromyces mucronatus | Inactivated | N/A | 0.79 ± 0.08 | 2.19 ± 0.17 | 0.15 ± 0.02 | 0.21 ± 0.03 | |
| Active | N/A | 0.75 ± 0.02 | 2.07 ± 0.02 | 0.14 ± 0.01 | 0.20 ± 0.02 | ||
| Piromyces rhizinflata | Inactivated | N/A | 0.72 ± 0.03 | 2.00 ± 0.11 | 0.12 ± 0.00 | 0.18 ± 0.01 | |
| Active | N/A | 0.72 ± 0.03 | 2.08 ± 0.09 | 0.13 ± 0.01 | 0.20 ± 0.01 | ||
| Phragmites australis | Neocallimastix frontalis | Inactivated | 78.45 ± 5.62 | 0.57 ± 0.03 | 1.63 ± 0.02 | 0.12 ± 0.01 | 0.19 ± 0.02 |
| Active | 14.71 ± 0.09 | 0.61 ± 0.02 | 1.64 ± 0.03 | 0.12 ± 0.00 | 0.22 ± 0.01 | ||
| Anaeromyces mucronatus | Inactivated | 41.10 ± 0.42 | 0.53 ± 0.04 | 1.59 ± 0.02 | 0.10 ± 0.00 | 0.21 ± 0.00 | |
| Active | 37.66 ± 14.56 | 0.64 ± 0.02 | 1.58 ± 0.03 | 0.08 ± 0.00 | 0.16 ± 0.01 | ||
| Piromyces rhizinflata | Inactivated | 44.11 ± 2.05 | 0.59 ± 0.03 | 1.63 ± 0.07 | 0.08 ± 0.00 | 0.16 ± 0.00 | |
| Active | 25.81 ± 0.36 | 0.58 ± 0.03 | 1.58 ± 0.08 | 0.10 ± 0.00 | 0.21 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravi, B.; Nkongndem Nkemka, V.; Hao, X.; Yanke, J.; McAllister, T.A.; Lee, H.; Veluchamy, C.; Gilroyed, B.H. Effect of Bioaugmentation with Anaerobic Fungi Isolated from Ruminants on the Hydrolysis of Corn Silage and Phragmites australis. Appl. Sci. 2021, 11, 9123. https://doi.org/10.3390/app11199123
Ravi B, Nkongndem Nkemka V, Hao X, Yanke J, McAllister TA, Lee H, Veluchamy C, Gilroyed BH. Effect of Bioaugmentation with Anaerobic Fungi Isolated from Ruminants on the Hydrolysis of Corn Silage and Phragmites australis. Applied Sciences. 2021; 11(19):9123. https://doi.org/10.3390/app11199123
Chicago/Turabian StyleRavi, Bhargavi, Valentine Nkongndem Nkemka, Xiying Hao, Jay Yanke, Tim A. McAllister, Hung Lee, Chitraichamy Veluchamy, and Brandon H. Gilroyed. 2021. "Effect of Bioaugmentation with Anaerobic Fungi Isolated from Ruminants on the Hydrolysis of Corn Silage and Phragmites australis" Applied Sciences 11, no. 19: 9123. https://doi.org/10.3390/app11199123
APA StyleRavi, B., Nkongndem Nkemka, V., Hao, X., Yanke, J., McAllister, T. A., Lee, H., Veluchamy, C., & Gilroyed, B. H. (2021). Effect of Bioaugmentation with Anaerobic Fungi Isolated from Ruminants on the Hydrolysis of Corn Silage and Phragmites australis. Applied Sciences, 11(19), 9123. https://doi.org/10.3390/app11199123










