Effects of Glyphosate and Its Metabolite AMPA on Aquatic Organisms
Abstract
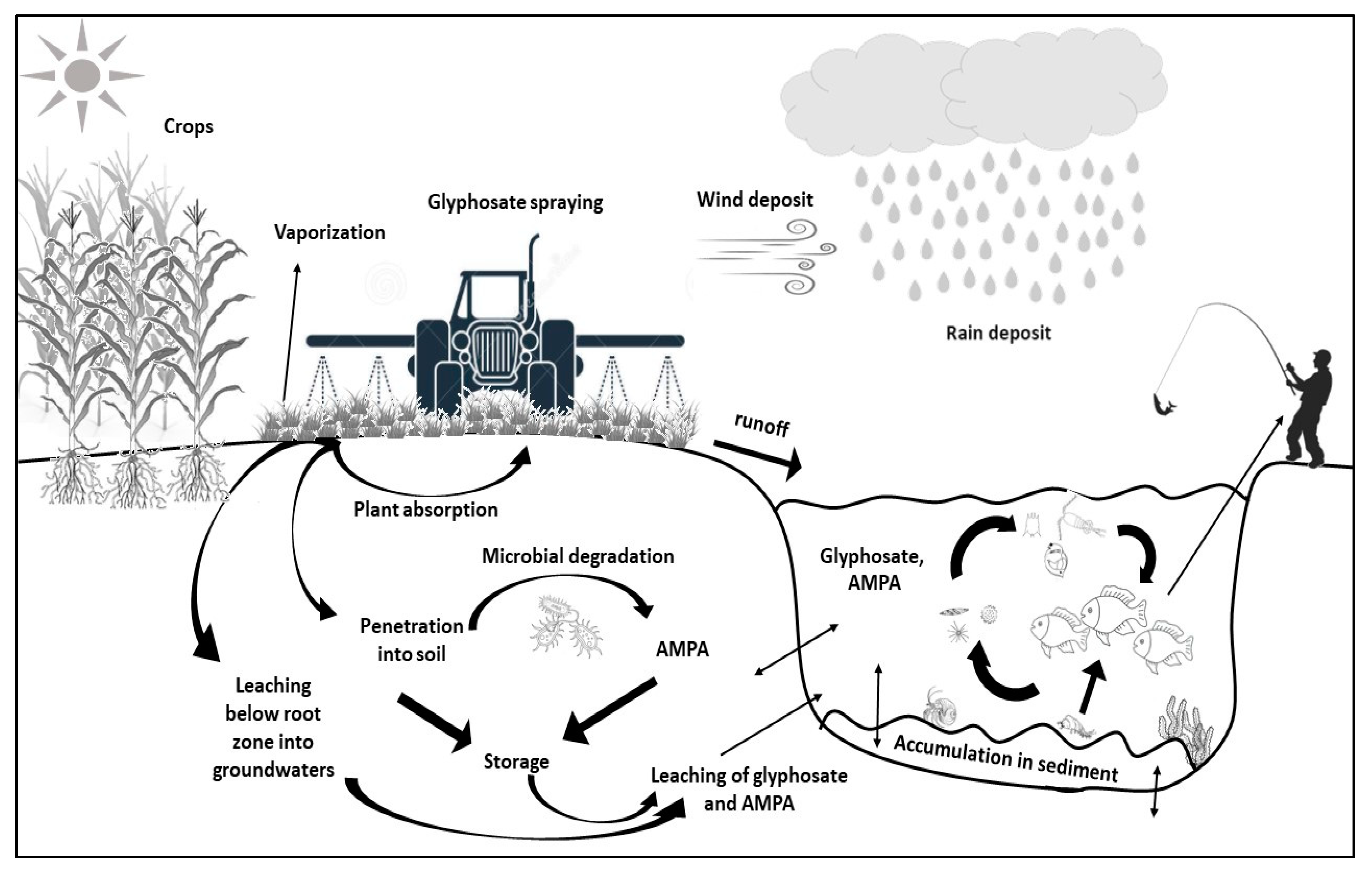
1. Introduction
2. Glyphosate (N-(phosphonomethyl)glycine)
2.1. Environmental Fate
2.2. Acute Toxicity
2.3. Toxic Effects
2.3.1. Fish
2.3.2. Invertebrate Species
3. AMPA (Aminomethylphosphonic Acid)
3.1. Environmental Fate
3.2. Acute Toxicity
3.3. Toxic Effects
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Arias-Estévez, M.; López-Periago, E.; Martínez-Carballo, E.; Simal-Gándara, J.; Mejuto, J.-C.; García-Río, L. The mobility and degradation of pesticides in soils and the pollution of groundwater resources. Agric. Ecosyst. Environ. 2008, 4, 247–260. [Google Scholar] [CrossRef]
- Bilal, M.; Igbal, H.M.N.; Barceló, D. Persistence of pesticides-based contaminants in the environment and their effective degradation using laccase-assisted biocatalytic systems. Sci. Total Environ. 2019, 695, 133896. [Google Scholar] [CrossRef]
- Riahi, B.; Rafatpanah, H.; Mahmoudi, M.; Memar, B.; Brook, A.; Tabasi, N.; Karimia, G. Immunotoxicity of paraquat after subacute exposure to mice. Food Chem. Toxicol. 2010, 48, 1627–1631. [Google Scholar] [CrossRef]
- Fenner, K.; Canonica, S.; Wackett, L.P.; Elsner, M. Evaluating Pesticide Degradation in the Environment: Blind Spots and Emerging Opportunities. Science 2013, 341, 752–758. [Google Scholar] [CrossRef]
- Gill, J.P.K.; Sethi, N.; Mohan, A.; Datta, S.; Girdhar, M. Glyphosate toxicity for animals. Environ. Chem. Lett. 2018, 16, 401–426. [Google Scholar] [CrossRef]
- Grube, A.; Donaldson, D.; Kiely, T.; Wu, L. Pesticides Industry Sales and Usage, 2006 and 2007 Market Estimates; U.S. Environmental Protection Agency: Washington, DC, USA, 2011; p. 41. [Google Scholar]
- Franz, J. N-Phosphonomethyl-Glycine Phytotoxicant Compositions; Monsanto CO, US: St. Louis, MO, USA, 1974; p. 3799758. Available online: https://www.freepatentsonline.com/3799758.html (accessed on 12 February 2021).
- Henderson, A.M.; Gervais, J.A.; Luukinen, B.; Buhl, K.; Stone, D.; Strid, A.; Cross, A.; Jenkins, J. Glyphosate Technical Fact Sheet; National Pesticide Information Center, Oregon State University Extension Services. 2010. Available online: http://npic.orst.edu/factsheets/archive/glyphotech.html (accessed on 10 February 2021).
- Coupe, R.H.; Kalkhoff, S.J.; Capel, P.D.; Gregoire, C. Fate and transport of glyphosate and Aminomethylphosphonic acid in surface waters of agricultural basins. Pest. Manag. Sci. 2012, 68, 16–30. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Myer, M.T.; Kuivila, K.M.; Dietze, J.E. Glyphosate and its gedradation product AMPA occur frequently and widely in U.S. soils, surface water, groundwater, and precipitation. J. Am. Water Resour. Assoc. 2014, 50, 275–290. [Google Scholar] [CrossRef]
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R.; et al. Concerns over use of glyphosate-based herbicides and risks associated with exposures: A consensus statement. Environ. Health 2016, 15, 19. [Google Scholar] [CrossRef]
- Guyton, K.Z.; Loomis, D.; Grosse, Y.; El Ghissassi, F.; Benbrahim-Tallaa, L.; Guha, N.; Scoccianti, C.; Mattock, H.; Straif, K. International Agency for Research on Cancer Monograph Working Group, IARC, Lyon, France. Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet Oncol. 2015, 5, 490–491. [Google Scholar] [CrossRef]
- IARC (International Agency for Research on Cancer). IARC Rejecets False Claims in Reuters Article (“In Glyphosate Review, WHO Cancer Agency Edited Out “Non-Carcinogenic” Findings”). 2017. Available online: https://www.iarc.who.int/wp-content/uploads/2018/07/IARC_Response_Reuters_October2017.pdf (accessed on 14 February 2021).
- IARC (International Agency for Research on Cancer). IRAC Response to Critisms of the Monographs and the Glyphosate Evaluation. 2018. Available online: https://www.iarc.who.int/wp-content/uploads/2018/07/IARC_response_to_criticisms_of_the_Monographs_and_the_glyphosate_evaluation.pdf (accessed on 14 February 2021).
- U.S. EPA (United States Environmental Protection Agency). Preliminary Ecological Risk Assessment in Support of the Registration Review of Glyphosate and Its Salts; U.S. EPA: Washington, DC, USA, 2015; p. 318. [Google Scholar]
- Zhan, H.; Feng, Y.; Fan, X.; Chen, S. Recent advances in glyphosate biodegradation. Appl. Microbiol. Biotechnol. 2018, 102, 5033–5043. [Google Scholar] [CrossRef]
- Sun, M.; Li, H.; Jaisi, D.P. Degradation of glyphosate and bioavailability of phosphorus derived from glyphosate in a soil-water system. Water Res. 2019, 163, 114840. [Google Scholar] [CrossRef]
- Wang, S.; Seiwert, B.; Kästner, M.; Miltner, A.; Schäffer, A.; Reemtsma, T.; Yang, Q.; Nowak, K.M. (Bio)degradation of glyphosate in water-sediment microcosms–A stable isotope co-labeling approach. Water Res. 2016, 99, 91–100. [Google Scholar] [CrossRef]
- Al-Mamun, A. Pesticide Degradations Resides and Environmental Concerns. In Pesticide Residue in Foods: Sources, Management, and Control; Khan, M.S., Rahman, M.S., Eds.; Springer International Publishing AG: Cham, Switzerland, 2017; pp. 87–102. [Google Scholar] [CrossRef]
- Larson, S.J.; Capel, P.D.; Majewski, M.S. Pesticide in surface waters—Distribution, trends, and governing factors. In Series of Pesticides in Hydrologic System; Gilliom, R.J., Ed.; Ann Arbor Press: Chelsea, MI, USA, 1997; Volume 3, p. 392. [Google Scholar]
- Commission Implementing Regulation (EU) no 1372/2011. Concerning the Non-Approval of the Active Substance Acetochlor, in Accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council Concerning the Placing of Plant Protection Products on the Market, and Amending Commission Decision 2008/934/EC. OJEU, L 341/45. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32011R1372 (accessed on 25 November 2020).
- Moulisová, A.; Bendakovská, L.; Kožíšek, F.; Vavrouš, A.; Jeligová, H.; Kotal, F. Pesticidy a jejich metabolity v pitné vodě. Jaký je současný stav v České Republice? Vodní Hospodářství 2017, 68, 4–10. [Google Scholar]
- CHMI (Czech Hydrometeorological Institute). On-Line Water Quality Database. 2020. Available online: http://hydro.chmi.cz/ (accessed on 10 December 2020).
- Kolpin, D.W.; Thurman, E.M.; Linhart, S.M. The environmental occurrence of herbicides: The importance of degradates in ground water. Arch. Environ. Contam. Toxicol. 1998, 35, 385–390. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstetter, T.B.; Johnson, C.A.; Von Gunten, U.; Wehrli, B. The challenge of micropollutants in aquatic systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef]
- Ceyhun, S.B.; Şentürk, M.; Ekinci, D.; Erdoğan, O.; Çiltaş, A.; Kocaman, E.M. Deltamethrin attenuates antioxidant defense system and induces the expression of heat shock protein 70 in rainbow trout. Comp. Biochem. Physiol. C 2010, 152, 215–222. [Google Scholar] [CrossRef]
- Abrantes, N.; Pereira, R.; Gonçalves, F. Occurrence of pesticides in water, sediments, and fish tissues in a Lake Surrounded by agricultural lands: Concerning risks to humans and ecological receptors. Water Air Pollut. 2010, 212, 77–88. [Google Scholar] [CrossRef]
- De Moura, F.R.; da Silva Lima, R.R.; da Cunha, A.P.S.; da Costa Marisco, P.; Aguiar, D.H.; Sugui, M.M.; Sinhorin, A.P.; Sinhorin, V.D.G. Effects of glyphosate-based herbicide on pintado da Amazônia: Hematology, histological aspects, metabolic parameters and genotoxic potential. Environ. Toxicol. Pharmacol. 2017, 56, 241–248. [Google Scholar] [CrossRef]
- Geist, J. Integrative freshwater ecology and biodiversity conservation. Ecol. Indic. 2011, 11, 1507–1516. [Google Scholar] [CrossRef]
- Malaj, E.; Peter, C.; Grote, M.; Kühne, R.; Mondy, C.P.; Usseglio-Polatera, P.; Schäfer, R.B. Organic chemicals jeopardize the health of freshwater ecosystems on the continental scale. Proc. Nat. Acad. Sci. USA 2014, 111, 9549–9554. [Google Scholar] [CrossRef]
- Qui, Y.W.; Zeng, E.Y.; Qiu, H.; Yu, K.; Cai, S. Bioconcentration of polybrominated diphenyl ethers and organochlorine pesticides in algae is an important contaminant route to higher trophic levels. Sci. Total. Environ. 2017, 579, 1885–1893. [Google Scholar] [CrossRef]
- Eddleston, M. Poisoning by pesticides. Medicine 2020, 48, 214–217. [Google Scholar] [CrossRef]
- Ippolito, A.; Kattwinkel, M.; Rasmussen, J.J.; Schäfer, R.B.; Fornaroli, R.; Liess, M. Modeling global distribution of agricultural insecticides in surface waters. Environ. Pollut. 2015, 198, 54–60. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Glyphosate/Published under the Joint Sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. World Health Organization & International Programme on Chemical Safety. 1994. Available online: https://apps.who.int/iris/handle/10665/40044 (accessed on 3 December 2020).
- Franz, J.E.; Mao, M.K.; Sikorski, J.A. Glyphosate: A Unique Global Herbicide; American Chemical Society: Washington, DC, USA, 1997; p. 615. [Google Scholar]
- Wallace, J.; Lingenfelter, D. Glyphosate (Roundup): Understanding Risks to Human Health. PennState Extension: College of Agricultural Sciences. 1–3. 2019. Available online: https://extension.psu.edu/glyphosate-roundup-understanding-risks-to-human-health#:~:text=Glyphosate%20fate%20in%20environment.%20The%20chemical%20properties%20of,water.%20Glyphosate%20does%20not%20degrade%20quickly%20in%20plants (accessed on 10 July 2020).
- Tsui, M.T.K.; Chu, L.M. Aquatic toxicity of glyphosate-based formulations: Comparison between different organisms and the effects of environmental factors. Chemosphere 2003, 52, 1189–1197. [Google Scholar] [CrossRef]
- Modesto, K.A.; Martinez, C.B.R. Roundup causes oxidative stress in liver and inhibits acetylcholinesterase in muscle and brain of the fish Prochilodus lineatus. Chemosphere 2010, 78, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Roundup.cz. RoundupBioaktiv: Okolo Vodních Toků a Nádrží, Fakta. Monsanto ČR s.r.o. Available online: https://www.roundup.cz/roundup-biaktiv/fakta (accessed on 12 December 2020).
- Blake, R.J.; Pallet, K. The environmental fate and ecotoxicity of glyphosate. Outlooks Pest. Manag. 2018, 29, 266–269. [Google Scholar] [CrossRef]
- Folmar, L.C.; Sanders, H.O.; Julin, A.M. Toxicity of the herbicide glyphosate and several of its formulations to fish and aquatic invertebrates. Arch. Environ. Contam. Toxicol. 1979, 8, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, S.; Gaivao, I.; Santos, M.A.; Pacheco, M. European eel (Anguilla Anguilla genotoxic and prooxidant responses following short-term exposure to Roundup®—A glyphosate-based herbicide. Mutagenesis 2010, 25, 523–530. [Google Scholar] [CrossRef]
- Wagner, N.; Reichenbecher, W.; Teichmann, H.; Tappeser, B.; Lotters, S. Questions concerning the potential impact of glyphosate-base herbicides on amphibians. Environ. Toxicol. Chem. 2013, 32, 1688–1700. [Google Scholar] [CrossRef]
- Rodrigues, N.R.; de Souza, A.P.F. Occurrence of glyphosate and AMPA residue in soy-based infant formula sold in Brazil. Food Addit. Contam. A 2018, 35, 724–731. [Google Scholar] [CrossRef]
- Matozzo, V.; Fabrello, J.; Marin, M.G. The effects of glyphosate and its commercial formulations to marine invertebrates: A review. J. Mar. Sci. Eng. 2020, 8, 399. [Google Scholar] [CrossRef]
- Hildebrand, L.D.; Sullivan, D.S.; Sullivan, T.P. Experimental studies of rainbow trout populations exposed to field applications of Roundup® herbicide. Arch. Environ. Contam. Toxicol. 1982, 11, 93–98. [Google Scholar] [CrossRef]
- Neskovic, N.K.; Poleksic, V.; Elezovic, I.; Karan, V.; Budimir, M. Biochemical and histopathological effects of glyphosate on carp, Cyprinus carpio L. Bull. Environ. Contam. Toxicol. 1996, 56, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Gholami-Seyedkolaei, S.; Mirvaghefi, A.; Farahmand, H.; Kosari, A.A. Effect of a glyphosate-based herbicide in Cyprinus carpio: Assessment of acetylcholinesterase activity, hematologigal responses and serum biochemical parameters. Ecotoxicol. Environ. Saf. 2013, 98, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Bu, Y.; Li, X. Immunological and histopathological responses of the kidney of common carp (Cyprinus carpio L.) sublethally exposed to glyphosate. Environ. Toxicol. Pharmacol. 2015, 39, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Antunes, A.M.; Rocha, T.L.; Pires, F.S.; de Freitas, M.A.; Leite, V.R.M.C.; Arana, S.; Moreira, P.C.; Sabóia-Morais, S.M.T. Gender-specific histopathological response in guppies Poecilia reticulata exposed to glyphosate or its metabolite Aminomethylphosphonic acid. J. Appl. Toxicol. 2017, 37, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Kreutz, L.C.; Barcellos, L.J.G.; Silva, T.O.; Anziliero, D.; Martins, D.; Lorenson, M.; Martheninghe, A.; da Silva, L.B. Acute toxicity test of agricultural pesticides on silver catfish (Rhamdia quelen). Cienc. Rural 2008, 38, 1050–1055. [Google Scholar] [CrossRef]
- Ayoola, S.O. Histopathological effects of glyphosate on juvenile African catfish (Clarius gariepinus). Am-Euras. J. Agric. Environ. Sci. 2008, 4, 362–367. [Google Scholar]
- de Brito Rodrigues, L.; Costa, G.G.; Thá, E.L.; da Silva, L.R.; de Oliveira, R.; Leme, D.M.; Cestari, M.M.; Grisolia, C.K.; Valadares, M.C.; de Oliveira, G.A.R. Impact of the glyphosate-based commercial herbicide, its components and its metabolite AMPA on non-target aquatic organisms. Mut. Res.-Genet. Toxicol. Environ. Mut. 2019, 842, 94–101. [Google Scholar] [CrossRef]
- Brodeur, J.C.; Malpel, S.; Anglesio, A.B.; Cristos, D.; D’Andrea, M.F.; Poliserpi, M.B. Toxicities of glyphosate- and cypermethrin-based pesticides are antagonic in the tenspotted livebearer fish (Cnesterodon decemmaculatus). Chemosphere 2016, 155, 429–435. [Google Scholar] [CrossRef]
- Daam, M.A.; Moutinho, M.F.; Espíndola, E.L.G.; Schiesari, L. Lethal toxicity of the herbicides acetochlor, ametryn, glyphosate and metribuzin to tropical frog larvae. Ecology 2019, 28, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Howe, C.M.; Berrill, M.; Pauli, B.D.; Helbing, C.C.; Werry, K.; Veldhoen, N. Toxicity of Glyphosate-based pesticides to four north American frog species. Environ. Toxicol. Chem. 2004, 23, 1928–1938. [Google Scholar] [CrossRef] [PubMed]
- Lajmanovich, R.C.; Attademo, A.M.; Peltzer, P.M.; Junges, C.M.; Cabagna, M.C. Toxicity of four herbicide formulations with glyphosate on Rhinella arenarum (Anura: Bufonidae) tadpoles: B-esterases and gluthation S-transferase inhibitors. Arch. Environ. Contam. Toxicol. 2011, 60, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Yang, X.; Yan, G.; Huang, Y.; Zuo, F.; Shen, Y.; Ding, Y.; Cheng, Y. Effects of glyphosate on immune responses and haemocyte DNA damage of Chinese mitten crab, Eriocheir sinensis. Fish Shellfish Immunol. 2017, 71, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, S.; Santos, M.; Barroso, C.; Gaivão, I.; Mário, P. Differential genotoxicity of Roundup® formulation and its constituents in blood cells of fish (Anguilla anguilla): Considerations on chemical interactions and DNA damaging mechanisms. Ecotoxicology 2012, 21, 1381–1390. [Google Scholar] [CrossRef]
- Ma, J.; Zhu, J.; Wang, W.; Ruan, P.; Rajeshkumar, S.; Li, X. Biochemical and molecular impacts of glyphosate-based herbicide on the gills of common carp. Environm. Pollut. 2019, 252, 1288–1300. [Google Scholar] [CrossRef]
- Gaur, H.; Bhargava, A. Glyphosate induces toxicity and modulates calcium and NO signaling in zebrafish embryos. Biochem. Bioph. Res. Comm. 2019, 513, 1070–1075. [Google Scholar] [CrossRef]
- Nwani, C.D.; Nagpure, N.S.; Kumar, R.; Kushwaha, B.; Lakra, W.S. DNA damage and oxidative stress modulatory effects of glyphosate-based herbicide in freshwater fish, Channa punctatus. Environ. Toxicol. Pharmacol. 2013, 36, 539–547. [Google Scholar] [CrossRef]
- Menéndez-Helman, R.; Ferreyroa, G.V.; dos Santos Afonso, M.; Salibán, A. Glyphosate as an Acetylcholinesterase inhibitor in Cnesterodon decemmaculatus. Bull. Environ. Contam. Toxicol. 2012, 88, 6–9. [Google Scholar] [CrossRef]
- Glusczak, L.; dos Santos Miron, D.; Crestani, M.; da Fonseca, M.B.; de Araújo Pedron, F.; Duarte, M.F.; Pimentel Vieira, V.L. Effect of glyphosate herbicide on acetylcholinesterase activity and metabolic and hematological parameters in piava (Leporinus obtusidens). Ecotoxicol. Environ. Saf. 2006, 65, 237–241. [Google Scholar] [CrossRef]
- Salbego, J.; Pretto, A.; Gioda, C.R.; de Menezes, C.C.; Lazzari, R.; Neto, J.R.; Baldisserotto, B.; Loro, V.L. Herbicide formulation with glyphosate affects growth, acetylcholinesterase activity, and metabolic and hematological parameters in Piava (Leporinus obtusidens). Arch. Environ. Contam. Toxicol. 2010, 58, 740–745. [Google Scholar] [CrossRef]
- Glusczak, L.; dos Santos Miron, D.; Moares, B.S.; Simoes, R.R.; Chitolina Schetinger, M.R.; Morsch, V.M.; Loro, V.L. Acute effects of glyphosate herbicide on metabolic and enzymatic parameters of silver catfish. Comp. Biochem. Physiol. C 2007, 146, 519–524. [Google Scholar] [CrossRef]
- Kreutz, L.C.; Barcellos, L.J.G.; de Faria Valle, S.; de Oliveira Silva, T.; Anziliero, D.; dos Santos, E.D.; Pivato, M.; Zanatta, R. Altered hematological and immunological parameters in silver catfish fish (Rhamdia quelen) following short term exposure to sublethal concentration of glyphosate. Fish Shellfish Immunol. 2011, 30, 51–57. [Google Scholar] [CrossRef]
- Persch, T.S.P.; Weimer, R.N.; Freitas, B.S.; Oliveira, G.T. Metabolic parameters and oxidative balance in juvenile (Rhamdia quelen) exposed to rice paddy herbicides: Roundup®, Primoleo®, and Facet®. Chemosphere 2017, 174, 98–109. [Google Scholar] [CrossRef]
- Lushchak, O.V.; Kubrak, O.I.; Storey, J.M.; Storey, K.B.; Lushchak, V.I. Low toxic herbicide Roundup induces mild oxidative stress in goldfish tissues. Chemosphere 2009, 76, 932–937. [Google Scholar] [CrossRef] [PubMed]
- Li, M.H.; Xu, L.D.; Liu, Y.; Chen, T.; Jiang, L.; Fu, Y.H.; Wang, J.S. Multi-tissue metabolic responses of goldfish (Carassius auratus) exposed to glyphosate-based herbicide. Toxicol. Res. 2016, 5, 1039–1052. [Google Scholar] [CrossRef]
- Li, M.H.; Ruan, L.Y.; Zhou, J.W.; Fu, Y.H.; Jinag, L.; Zhao, H.; Wang, J.S. Metabolic profiling of goldfish (Carassius auratis) after long-term glyphosate-base herbicide exposure. Aquatic. Toxicol. 2017, 188, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.M.; Carneiro, B.; Ochs, J. Glyphosate induces neurotoxicity in zebrafish. Environ. Toxicol. Pharmacol. 2016, 42, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Goulart, T.L.S.; Boyle, R.T.; Souza, M.M. Cytotoxicity of the association of pesticides Roundup Transorb® and Furadan 350 SC® on the zebrafish cell line, ZF-L. Toxicol. Vitro. 2015, 29, 1377–1384. [Google Scholar] [CrossRef][Green Version]
- Samanta, P.; Pal, S.; Mukherjee, A.K.; Glosh, A.R. Biochemical effects of glyphosate based herbicide, Excel Mera 71 on enzyme activities of acetylcholinesterase (AChE), lipid peroxidation (LPO), catalase (CAT), gluthation-S-transferase (GST) and protein content on teleostean fishes. Ecotoxicol. Environ. Saf. 2014, 107, 120–125. [Google Scholar] [CrossRef]
- Le Du-Carrée, J.; Morin, T.; Danion, M. Impact of chronic exposure of rainbow trout, Oncorhynchus mykiss, to low doses of glyphosate or glyphosate-based herbicides. Aquat. Toxicol. 2021, 230, 105687. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Jia, R.; Cao, L.; Du, J.; Gu, Z.; He, Q.; Xu, P.; Yin, G. Effects of chronic glyphosate exposure on antioxdative status, metabolism and immune response in tilapia (GIFT, Oreochromis niloticus). Com. Biochem. Physiol. C 2021, 239, 108878. [Google Scholar] [CrossRef] [PubMed]
- Matozzo, V.; Munari, M.; Maseiro, L.; Finos, L.; Marin, M.G. Ecotoxicological hazard of a mixture of glyphosate and aminomethylphosphonic acid to the mussel Mytilus galloprovincialis (Lamarck 1819). Sci. Rep. 2019, 9, 14302. [Google Scholar] [CrossRef]
- Sandrini, J.Z.; Rola, R.C.; Lopes, F.M.; Buffon, H.F.; Freitas, M.M.; Martins, C.M.; da Rosa, C.E. Effects of glyphosate on cholinesterase activity of the mussel Perna perna and the fish Danio rerio and Jenynsia multidentata: In vitro studies. Aquat. Toxicol. 2013, 130–131, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Matozzo, V.; Fabrello, J.; Masiero, L.; Ferraccioli, F.; Finos, L.; Pastore, P.; Di Gangi, I.M.; Bogialli, S. Ecotoxicological risk assessment for the herbicide glyphosate to non-target aquatic species: A case study with the mussel Mytilus galloprovincialis. Environ. Pollut. 2018, 233, 623–632. [Google Scholar] [CrossRef]
- Iummato, M.M.; Di Fiori, E.; Sabatini, S.E.; Cacciatore, L.C.; Cochón, A.C.; del Carmen Ríos de Molina, M.; Juárez, Á.B. Evaluation of biochemical markers in the golden mussel Limnoperna fortune exposed to glyphosate acid in outdoor microcosms. Ecotoxicol. Environ. Saf. 2013, 95, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Di Fiori, E.; Pizarro, H.; dos Santos Afonso, M.; Cataldo, D. Impact of the invasive mussels Limnoperna fortunei on glyphosate concentration in water. Ecotoxicol. Environ. Saf. 2012, 81, 106–113. [Google Scholar] [CrossRef]
- Séguin, A.; Mottier, A.; Perron, C.; Lebel, J.M.; Serpentini, A.; Costil, K. Sub-lethal effects of glyphosate-based commercial formulation and adjuvants on juvenile oysters (Crassostrea gigas) exposed for 35 days. Mar. Pollut. Bull. 2017, 117, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Contardo-Jara, V.; Klingelmann, E.; Wiegand, C. Bioaccumulation of glyphosate and its formulations Roundup Ultra in Lumbriculus variegatus and its effects on biotransformation and antioxidant enzymes. Environ. Pollut. 2009, 157, 57–63. [Google Scholar] [CrossRef]
- Costa, M.J.; Monteiro, D.A.; Oliveira-Neto, A.L.; Rantin, T.F.; Kalinin, A.L. Oxidative stress biomarkers and heart function in bullfrog tadpoles exposed to Roundup Original. Ecotoxicology 2008, 17, 153–163. [Google Scholar] [CrossRef]
- Barky, F.A.; Abdelsam, H.A.; Mahmoud, M.B.; Hamdi, S.A.H. Influence of Atrazine and Roundup pesticides on biochemical and molecular aspects of Biomphalaria alexandrina snails. Pest. Biochem. Physiol. 2012, 104, 9–18. [Google Scholar] [CrossRef]
- Mohamed, A.H. Sublethal toxicity of Roundup to immunological and molecular aspects of Biomphalaria alexandrina to Schistosoma mansoni infection. Ecotoxicol. Environ. Saf. 2011, 74, 754–760. [Google Scholar] [CrossRef]
- Levine, S.L.; von Mérey, G.; Minderhout, T.; Manson, P.; Sutton, P. Aminomethylphosphonic acid has low chronic toxicity to Daphnia magna and Pimephales promelas. Environ. Toxicol. Chem. 2015, 34, 1382–1389. [Google Scholar] [CrossRef] [PubMed]
- Poppov, K.; Ronkkomaki, H.; Lajunens, L.H.J. Critical evaluation of stability constants of phosphonic acids. Pure Appl. Chem. 2001, 73, 1641–1677. [Google Scholar] [CrossRef]
- Bonansea, R.I.; Filippi, I.; Wunderlin, D.A.; Marino, D.J.G.; Amé, M.V. The Fate of Glyphosate and AMPA in a Freshwater Endorheic Basin: An Ecotoxicological Risk Assessment. Toxics 2018, 6, 3. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Koplin, D.W.; Scribner, E.A.; Kuivila, K.M.; Sandstorm, M.W. Glyphosate, other herbicides, and transformation products in midwestern streams, 2002. J. Am. Water Resour. Assoc. 2005, 41, 323–332. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Rice, K.C.; Focazio, M.J.; Salmons, S.; Barry, R.X. The occurrence of glyphosate, atrazine, and other pesticides in vernal pool and adjacent streams in Washington, DC, Maryland, Iowa, and Wyoming, 2005–2006. Environ. Monitor. Assess. 2009, 155, 281–307. [Google Scholar] [CrossRef]
- Directive 2000/60/EC of the European Parliament and of the Council. 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy. Available online: https://eur-lex.europa.eu/legal-content/CS/ALL/?uri=CELEX%3A32000L0060 (accessed on 22 December 2020).
- Aparicio, V.C.; De Gerónimo, E.; Marino, D.; Primost, J.; Carriquiriborde, P.; Costa, J.L. Environmental fate of glyphosate and aminomethylphosphonic acid in surface waters and soil of agricultural basins. Chemosphere 2013, 93, 1866–1873. [Google Scholar] [CrossRef]
- Di Poi, C.; Costil, K.; Bouchart, V.; Harm-Lemeille, M.P. Toxicity assessment of five emerging pollutants. Alone and in binary or ternary mixtures, towards three aquatic organisms. Environ. Sci. Pollut. 2018, 25, 6122–6134. [Google Scholar] [CrossRef]
- Tajnaiová, L.; Vurm, R.; Kholomyeva, M.; Kobera, M.; Koci, V. Determination of the ecotoxicity of herbicides Roundup® Classis Pro and Garlon New in aquatic and terrestrial environments. Plants 2020, 9, 1203. [Google Scholar] [CrossRef]
- EFSA (European Food and Safety Authority). Conclusion on the peer review of the pesticide risk assessment of the active substance glyphosate. EFSA J. 2015, 13, 4302. [Google Scholar] [CrossRef]
- Reddy, K.N.; Rimando, A.M.; Duke, S.O. Aminomethylphosphonic acid, a metabolite of glyphosate, cases injury in glyphosate-treated, glyphosate-resistant soybean. J. Agric. Food Chem. 2004, 52, 5139–5143. [Google Scholar] [CrossRef] [PubMed]
- Gomez, M.P.; Smedbol, E.; Chalifour, A.; Hénault-Ethier, L.; Labrecque, M.; Lepage, L.; Lucotte, M.; Juneau, P. Alteration of plant physiology by glyphosate and it by-product Aminomethylphosphonic acid: An overview. J. Exp. Bot. 2014, 65, 4691–4703. [Google Scholar] [CrossRef] [PubMed]
- Serra, A.A.; Nuttens, A.; Larvor, V.; Renault, D.; Couée, I.; Sulmon, C.; Gouesbet, G. Low environmentally relevant levels of bioactive xenobiotics and associated degradation products cause cryptic perturbations of metabolism and molecular stress responses in Arabidopsis thaliana. J. Exp. Bot. 2013, 64, 2753–2766. [Google Scholar] [CrossRef]
- Guilherme, S.; Santos, M.A.; Gaivao, I.; Pacheco, M. DNA and chromosomal damage induced in fish (Anguilla anguilla L.) by aminomethylphosphonic acid (AMPA)—The major environmental breakdown product of glyphosate. Environ. Sci. Pollut. Res. 2014, 21, 8730–8739. [Google Scholar] [CrossRef] [PubMed]
- Matozzo, V.; Marin, M.G.; Maseiro, L.; Tremonti, M.; Biamonte, S.; Viale, S.; Finos, L.; Lovato, G.; Pastore, P.; Bogialli, S. Effects of aminomethylphosphonic acid, the main breakdown product of glyphosate, on cellular and biochemical parameters of the mussel Mytilus galloprovincialis. Fish Shellfish Immunol. 2018, 83, 321–329. [Google Scholar] [CrossRef]
- Cheron, M.; Brischoux, F. Aminomethylphosphonic acid alters amphibian embryonic development at environmental concentrations. Environ. Res. 2020, 190, 109944. [Google Scholar] [CrossRef]
- Commission Implementing Regulation (EU) 2017/2324. Renewing the Approval of the Active Substance Glyphosate in Accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council Concerning the Placing of Plant Protection Products on the Market, and Amending the Annex to Commission Implementing Regulation (EU) No 540/2011. OJEU, L333/10. 2017. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32017R2324 (accessed on 19 September 2021).
| Species | Formulation | Exposure (Hours) | Concentration (mg/L) | References |
|---|---|---|---|---|
| Rainbow trout (Oncorhynchus mykiss) | GLY | 96 | 140 | [41] |
| Roundup 1 | 96 | 52–55 | [46] | |
| Common carp (Cyprinus carpio) | GLY | 48 | 645 | [47] |
| 96 | 620 | |||
| Roundup 1 | 96 | 22.19 | [48] | |
| GLY | 48 | 602.61 | [49] | |
| 96 | 520.77 | |||
| Blackhead minnow (Pimephales promelas) | GLY | 96 | 97 | [41] |
| Channel catfish (Ictalurus punctatus) | 96 | 130 | ||
| Bluegills (Lepomis macrochirus) | GLY | 24 | 150 | [41] |
| 96 | 140 | |||
| Guppy (Poecilia reticulata) | GLY | 96 | 69.83 | [50] |
| Rhamdia quelen | GLY | 96 | 7.30 | [51] |
| North African catfish (Clarias gariepinus) | GLY | 96 | 0.295 | [52] |
| Zebrafish (Danio rerio) | Atnor 48 2 | 96 | 76.50 | [53] |
| Ten spotted live-bearer (Cnesterodon decemmaculatus) | Glyfoglex 3 | 96 | 41.40 | [54] |
| Species | Formulation | Exposure (Hours) | Concentration (mg/L) | References |
|---|---|---|---|---|
| Boana pardalis | GLY | 96 | 106 | [55] |
| Physalaemus cuvieri | 96 | 115 | ||
| Green frog (Lithobates clamitans) | Roundup 1 | 24 | 6.6 | [56] |
| 96 | 6.5 | |||
| Northern leopard frog (Lithobates pipiens) | 24 | 11.9 | ||
| 96 | 9.2 | |||
| Wood frog (Lithobates sylvaticus) | 24 | 18.1 | ||
| 96 | 16.5 | |||
| Dwarf American toad (Anaxyrus americanus) | 24 | 13.5 | ||
| 96 | <12.9 | |||
| Rhinella arenarum | Roundup Ultra-Max 2 | 48 | 2.42 | [57] |
| 77.52 |
| Species | Formulation | Exposure (Hours) | Concentration (mg/L) | References |
|---|---|---|---|---|
| Midge larvae (Chironomus plumosus) | GLY | 48 | 55 | [41] |
| Ceriodaphnia dubia | Roundup 1 | 48 | 147 | [37] |
| Acartia tonsa | 48 | 35 | ||
| Chinese mitten crab (Eriocheir sinensis) | GLY | 24 | 461.54 | [58] |
| 96 | 97.89 |
| Species | Concentration | Exposure | Effects | References |
|---|---|---|---|---|
| Common carp (Cyprinus carpio) | 2.5, 5, 10 mg/L (GLY) | 96 h | ↑ ALP in liver, heart, GOT in liver and kidney, GPT in kidney;Subepithelial edema and epithelial hyperplasia in gills, focal fibrosis in liver | [47] |
| 3.5, 7, 14 mg/L (Roundup 1) | 16 days | ↑ MCV, MCH; ↓ AChE in muscle, brain and liver, Hb, HCT, RBC, WBC, AST, ALT, LDH | [48] | |
| 52.08, 104.15 mg/L (GLY) | 7 days | Vacuolization of the renal parenchyma and intumescence of the renal tubule in kidney, immunotoxicity | [49] | |
| ↑ AST, ALT, MDA, PC; ↓ GSH, inhibition of NA+/K+ -ATPase, SOD, CAT, GPx, GR, T-AOC, induce inflammatory response in gills | [60] | |||
| European eel (Anguilla Anguilla) | 58, 116 μg/L (Roundup 1) | 1, 3 days | ↑ TBARS, LPO, GDI, ENA | [42] |
| ↑ GDI, damaged nucleoids, EndoIII | [59] | |||
| Curimbata (Prochilodus lineatus) | 10 mg/L (Roundup 1) | 24 h | ↑ GSH, GST, LPO; ↓ SOD, GPx, inhibition of AChE in muscle | [38] |
| 96 h | ↑ GST, LPO;inhibition of AChE in muscle in brain and muscle | |||
| Spotted snakehead (Channa punctatus) | 32.54 mg/L (Roundup 1) | 1, 7, 14, 21, 28, 35 days | ↑ TBARS, DNA damage, LPO, ROS; ↓ CAT, SOD, GR in gill and blood | [62] |
| Ten spotted live-bearer (Cnesterodon decemmaculatus) | 1, 1.75, 35 mg/L (GLY) | 96 h | ↓ AChE | [63] |
| Megaleporinus obtusidens | 3, 6, 10, 20 mg/L (Roundup 1) | 96 h | ↑ hepatic GL, GLU, NH3 in liver and muscle, PCV, Hb, RBC, WBC, P; ↓ AChE in brain, LACT, P in liver, muscle GL, GLU | [64] |
| 5 mg/L (Roundup 1) | 90 days | ↑ LACT in liver and muscle, P in liver; ↓ AChE, GL in liver, P in muscle, PCV, Hb, RBC, WBC | [65] | |
| Rhamdia quelen | 0.2, 0.4 mg/L (Roundup 1) | 96 h | ↑ hepatic GL, LACT in liver and muscle, P in liver and muscle, NH3 in liver and muscle, TBARS in muscle; ↓ muscle GL, GLU in liver and muscle, AChE in brain | [66] |
| 0.730 mg/L (GLY) | 24, 96 h, 10 days | ↑ immature circulating cells; ↓ RBC, THR, WBC, phagocytic activity, agglutination activity, lysozyme activity | [67] | |
| Rhamdia quelen | 18, 36, 72 μg/L (Roundup 1) | 7 days | ↑ TP in liver, ↑ GL in muscle; ↓ TP, GL, TL in gills, liver, and kidney | [68] |
| Goldfish (Carassius auratus) | 2.5–20 mg/L (Roundup 1) | 2 months | ↑ CAT in liver and kidney; ↓ GR in kidney, liver, and brain, G6PDH in kidney, liver and brain, SOD in kidney, liver and brain | [69] |
| 0.22, 0.44, 0.88 mmol/L (GLY) | 96 h | Behaviour abnormalities (observed depression, erratic swimming, partial loss of equilibrium), liver tissue damage (cellular swelling, inflammatory cell infiltration, hydropic degeneration, loose cytoplasm, ↑ brown particles), kidney tissue damage (edema in the epithelial cells of renal tubules, ↑ cell volume, loose cytoplasm, slight staining), changes in plasma (↑ CK, UN, ↓ LDH) | [70] | |
| 0.2 mmol/L (Nongteshi 2) | 90 days | Hyaline cast in kidney, ↑ CRE, BUN, ALT, AST, LDH, MDA, ↑ 3-hydroxybutyrate, LACT, alanine, acetamide, glutamate, glycine, histidine, inosine, GLU;↓ SOD, GSH-Px, GR, lysine, NAA, citrate, choline, phosphocholine, myo-inosine, nicotinamide, | [71] | |
| North African catfish (Clarias gariepinus) | 0, 19, 42, 94, 207, 455mg/L (GLY) | 96 h | Cellular infiltration in gills; fatty degeneration, fat vacuolation, diffuse hepatic necrosis, infiltration of leukocytes in liver; hematopoietic necrosis, pyknotic nuclei in kidney; mononuclear infiltration, neuronal degeneration, spongiosis in brain; respiratory stress, erratic swimming | [52] |
| Hybrid fish jundiara (Leiarius marmoratus × Psedoplatystoma reticulatum) | 1.357 mg/L (Roundup 1) | 6, 24, 48,96 h | ↑ LACT in liver, P level in liver, ALT, AST, CHOL, TAG in plasma; ↓ GL in liver and muscle, plasma GLU, Hb, PCV, RBC, WBC | [28] |
| Zebrafish (Danio rerio) | 50 μg/mL (GLY) | 24 h | ↓ gene expression in eye, fore, and midbrain delineated brain ventricles and cephalic regions | [72] |
| 32.5, 65, 130 μg/L (Transorb 3) | 48 h | ↓ integrity of plasma membrane of hepatocytes, viability of cells, mitochondrial activity in the cell, lysosomal integrity, inhibition in ABC transporter activity | [73] | |
| 10, 50, 100, 200, 400 μg/L (GLY) | 48 h | ↓ heartbeat, NO generation, downregulation of Cacana1C and ryr2a genes, upregulation of hspb11 | [61] | |
| Climbing bass (Anabas testudineus) | 17.20 mg/L (Excel Mera 71 4) | 30 days | ↑ AChE, LPO, CAT; ↓ TP, GST | [74] |
| Heteropneustes fossilis |
| Species | Concentration | Exposure | Effects | References |
|---|---|---|---|---|
| Mediterranean mussel (Mytilus galloprovincialis) | 100 μg/L (GLY) | 7 days | ↑ THC, haemocyte proliferation; ↓ Haemocyte diameter, AChE in gills | [77] |
| 14 days | ↑ AChE in gills, CAT in digestive gland; ↓ CAT in gills | |||
| 21 days | ↑ CAT in gills; ↓ THC, haemocyte diameter, haemocyte volume, HL, AChE in gills | |||
| 10, 100, 1000 μg/L (GLY) | 7, 14, 21 days | ↑ cell volume of haemocyte, haemolymph pH; ↓ HL, haemolymph acid phosphatase activity; AChE in gills; SOD in digestive gland, THC, | [79] | |
| Limnoperna fortunei | 1, 3, 6 mg/L (GLY) | 26 days | ↑ TBARS, GST, ALP; ↓ CES, SOD | [80] |
| 10, 20, 40 mg/L (GLY) | 3 weeks | ↓ presence of large mussel by 40%, presence empty shell by 25% | [81] | |
| Pacific oyster (Crassostrea gigas) | 0.1, 1, 100 μg/L (Roundup Expres 1) | 35 days | ↑ GST; ↓ growth; LPO, MDA | [82] |
| California blackworm (Lumbriculus variegatus) | 0.05–5 mg/L(GLY) | 4 days | ↑ SOD; ↓ GST, membrane bound GST | [83] |
| Chinese mitten crab (Eriocheir sinensis) | 4.4, 9.8, 44, 98 mg/L(GLY) | 96 h | ↑ % DNA in tail, SOD, POD, β-GD;↓ THC, granulocytes, phagocytic activity, ACP, AKP | [58] |
| American bullfrog (Lithobates catesbeianus) | 1 mg/L (Roundup 2) | 48 h | ↑ swimming activity, CPM; SOD, CAT and LPO in liver; LPO in muscle; ↓ SOD, CAT in muscle, TtHR | [84] |
| Rhinella arenarum | 1.85, 3.75, 7.5, 15, 30, 60, 120, 240 mg/L (Roundup Ultra-Max 3) | 48 h | ↓ AChE, BChE, CbE, GST | [57] |
| Northern leopard frog (Rana pipiens) | 0.6, 1.8 mg/L (Roundup 2) | 166 days | ↑ TRβ mRNA; Late metamorphic climax, developmental delay, abnormal gonads, necrosis of the tail tip, fin damage, abnormal growth on the tail tip, blistering on the tail fin | [56] |
| Snail(Biomphalaria alexandrina) | 3.15 mg/L (Roundup 2) | 6 weeks | ↑ mortality, stopped egg lying, abnormal laid eggs, ↑ GLU, LACT, FAC; ↓ egg hatchability, GL, TP, pyruvate, nucleic acids levels | [85] |
| 10 mg/L (Roundup 2) | 7 days | ↑ in vitro phagocytic activity, DNA damage in haemocytes | [86] |
| Species | Value | Concentration (mg/L) | References |
|---|---|---|---|
| Fish | |||
| Guppy (Poecilia reticulata) | 96hLC50 | 180 for male | [50] |
| 164.32 for female | |||
| Invertebrate | |||
| Pacific oyster (Crassostrea gigas) | 36hEC10 | 38.55 | [94] |
| 36hEC20 | 42.68 | ||
| 36hEC50 | 50.78 | ||
| 24hEC10 | 27.08 | ||
| 24hEC20 | 39.80 | ||
| 24hEC50 | 76.90 | ||
| 48hEC10 | >1005 | ||
| Daphnia magna | 48hEC20 | ||
| 48hEC50 | |||
| Algae | |||
| Pseudokirchneriella subcapitata | 72hEC10 | 85.05 | [94] |
| 72hEC20 | >100 | ||
| 72hEC50 | |||
| Desmodesmus subspicatus | 72hIC50 | 117.8 | [95] |
| 72hEC50 | 89.8 1 | [96] | |
| 452 2 |
| Species | Concentration | Exposure | Effects | References |
|---|---|---|---|---|
| European eel (Anguilla Anguilla) | 11.8, 23.6 μg/L | 1, 3 days | ↑ GDI, FPG, EndoIII | [100] |
| Zebrafish (Danio rerio) | 1.7, 5, 10, 23, 50, 100 mg/L | 24, 48, 72, 96 h | Genotoxicity with LOEC 1.7 mg/L, induce primary DNA lesions, | [53] |
| Guppy (Poecilia reticulata) | 82 mg/L | 96 h | Proliferation of the interlamellar epithelium, fusion of secondary lamellae in gill, steatosis, pyknotic nuclei in liver, degeneration of hepatocytes | [50] |
| Mediterranean mussel (Mytilus galloprovincialis) | 100 μg/L | 7 days | ↑ haemocyte diameter, haemocyte volume, haemocyte proliferation, LDH in haemolymph, HL; ↓ THC, AChE in gills | [77] |
| Mediterranean mussel (Mytilus galloprovincialis) | 100 μg/L | 14 days | ↑ THC, haemocyte diameter, haemocyte volume, haemocyte proliferation, AChE in gills, CAT in digestive gland; ↓ HL | |
| 21 days | ↑ haemocyte volume, LDH in haemolymph; ↓ THC, haemocyte proliferation, HL, AChE in gills | |||
| 1, 10, 100 μg/L | 7 days | ↓ THC | [101] | |
| 14 days | ↑ THC, haemocyte diameter and volume, lysosome activity, acid phosphatase; ↓ haemocyte proliferation, SOD in gill and digestive gland | |||
| 21 days | ↑ haemocyte proliferation, lysosome activity, acid phosphatase, LDH; ↓ THC, haemocyte diameter and volume | |||
| Bufo spinosus | 0.07, 0.32, 3.57 μg/L | 16 days | ↓ embryonic survival, development delay, short tail length | [102] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tresnakova, N.; Stara, A.; Velisek, J. Effects of Glyphosate and Its Metabolite AMPA on Aquatic Organisms. Appl. Sci. 2021, 11, 9004. https://doi.org/10.3390/app11199004
Tresnakova N, Stara A, Velisek J. Effects of Glyphosate and Its Metabolite AMPA on Aquatic Organisms. Applied Sciences. 2021; 11(19):9004. https://doi.org/10.3390/app11199004
Chicago/Turabian StyleTresnakova, Nikola, Alzbeta Stara, and Josef Velisek. 2021. "Effects of Glyphosate and Its Metabolite AMPA on Aquatic Organisms" Applied Sciences 11, no. 19: 9004. https://doi.org/10.3390/app11199004
APA StyleTresnakova, N., Stara, A., & Velisek, J. (2021). Effects of Glyphosate and Its Metabolite AMPA on Aquatic Organisms. Applied Sciences, 11(19), 9004. https://doi.org/10.3390/app11199004







