Korean Panax Ginseng Reduces Orthodontic Tooth Movement in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
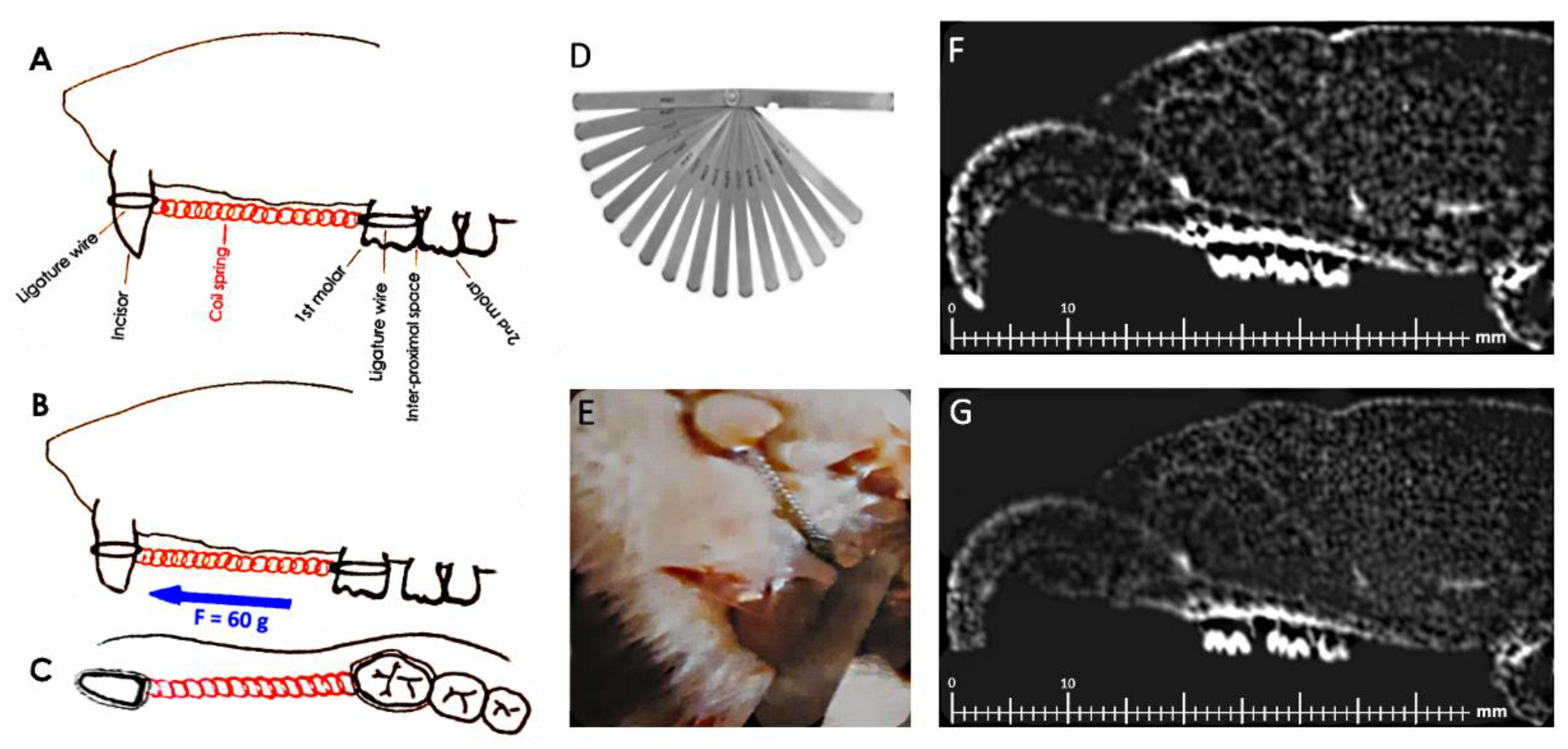
2.2. Orthodontic Treatment
2.3. OTM Measurement
2.4. Statistical Analysis
3. Results
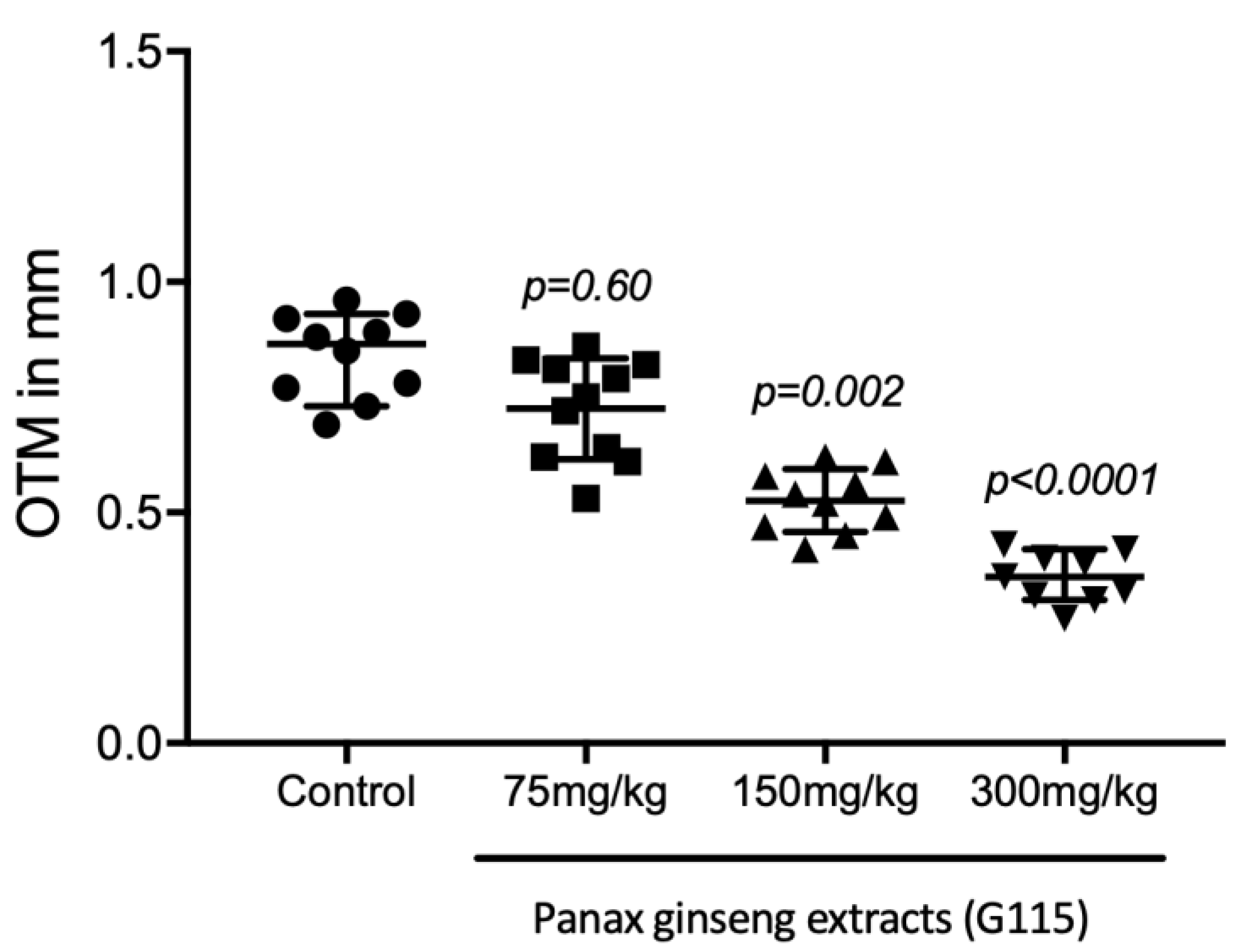
3.1. Orthodontic Tooth Movement
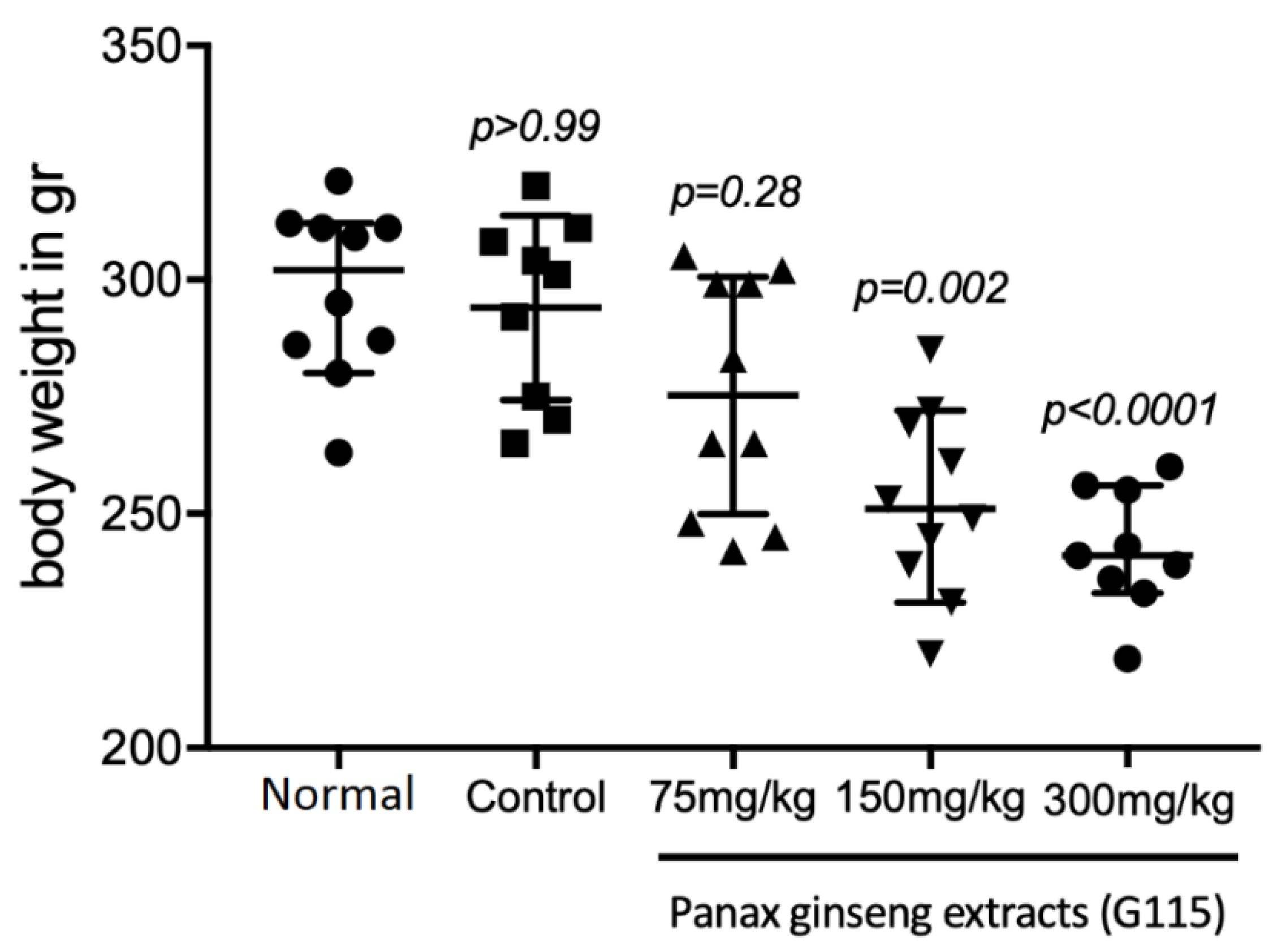
3.2. Body Weight
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jiang, C.; Li, Z.; Quan, H.; Xiao, L.; Zhao, J.; Jiang, C.; Wang, Y.; Liu, J.; Gou, Y.; An, S.; et al. Osteoimmunology in orthodontic tooth movement. Oral. Dis. 2015, 21, 694–704. [Google Scholar] [CrossRef]
- Matsumoto, T.; Iimura, T.; Ogura, K.; Moriyama, K.; Yamaguchi, A. The role of osteocytes in bone resorption during orthodontic tooth movement. J. Dent. Res. 2013, 92, 340–345. [Google Scholar] [CrossRef]
- Chaushu, S.; Klein, Y.; Mandelboim, O.; Barenholz, Y.; Fleissig, O. Immune Changes Induced by Orthodontic Forces: A Critical Review. J. Dent. Res. 2021, 220345211016285. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Fukasawa, S. Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement. Int. J. Mol. Sci. 2021, 22, 2388. [Google Scholar] [CrossRef]
- Kirschneck, C.; Kuchler, E.C.; Wahlmann, U.; Proff, P.; Schroder, A. Effects of the highly COX-2-selective analgesic NSAID etoricoxib on the rate of orthodontic tooth movement and cranial growth. Ann. Anat. 2018, 220, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Sodagar, A.; Etezadi, T.; Motahhary, P.; Dehpour, A.R.; Vaziri, H.; Khojasteh, A. The effect of celecoxib on orthodontic tooth movement and root resorption in rat. J. Dent. 2013, 10, 303–311. [Google Scholar]
- Liu, Y.; Zhang, T.; Zhang, C.; Jin, S.S.; Yang, R.L.; Wang, X.D.; Jiang, N.; Gan, Y.H.; Kou, X.X.; Zhou, Y.H. Aspirin Blocks Orthodontic Relapse via Inhibition of CD4(+) T Lymphocytes. J. Dent. Res. 2017, 96, 586–594. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Kou, X.; Yang, R.; Liu, D.; Wang, X.; Luo, Q.; Song, Y.; Liu, F.; Yan, Y.; Gan, Y.; et al. M1-like Macrophage Polarization Promotes Orthodontic Tooth Movement. J. Dent. Res. 2015, 94, 1286–1294. [Google Scholar] [CrossRef]
- He, D.; Kou, X.; Luo, Q.; Yang, R.; Liu, D.; Wang, X.; Song, Y.; Cao, H.; Zeng, M.; Gan, Y.; et al. Enhanced M1/M2 macrophage ratio promotes orthodontic root resorption. J. Dent. Res. 2015, 94, 129–139. [Google Scholar] [CrossRef]
- Jeon, H.H.; Teixeira, H.; Tsai, A. Mechanistic Insight into Orthodontic Tooth Movement Based on Animal Studies: A Critical Review. J. Clin. Med. 2021, 10, 1733. [Google Scholar] [CrossRef] [PubMed]
- Flagg, A.J. Traditional and Current Use of Ginseng. Nurs. Clin. North. Am. 2021, 56, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Xu, F.R.; Wang, Y.Z. Traditional uses, chemical diversity and biological activities of Panax L. (Araliaceae): A review. J. Ethnopharmacol 2020, 263, 112792. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ju, Z.; Yang, Y.; Zhang, Y.; Yang, L.; Wang, Z. Phytochemical analysis of Panax species: A review. J. Ginseng Res. 2021, 45, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, B.; Zhang, C.; Sun, G.; Sun, X. Effect of Panax notoginseng Saponins and Major Anti-Obesity Components on Weight Loss. Front. Pharmacol. 2020, 11, 601751. [Google Scholar] [CrossRef]
- Xue, Q.; He, N.; Wang, Z.; Fu, X.; Aung, L.H.H.; Liu, Y.; Li, M.; Cho, J.Y.; Yang, Y.; Yu, T. Functional roles and mechanisms of ginsenosides from Panax ginseng in atherosclerosis. J. Ginseng Res. 2021, 45, 22–31. [Google Scholar] [CrossRef]
- Wang, S.; Yue, R.; Huang, X.; Li, L.; Xu, C.; Liu, L. Renshen (Panax ginseng) and Huanglian (Rhizoma Coptidis) For T2DM: A protocol of systematic review and meta-analysis of randomized clinical trials. Medicine 2021, 100, e23743. [Google Scholar] [CrossRef]
- Kang, Z.; Zhonga, Y.; Wu, T.; Huang, J.; Zhao, H.; Liu, D. Ginsenoside from ginseng: A promising treatment for inflammatory bowel disease. Pharmacol. Rep. 2021, 73, 700–711. [Google Scholar] [CrossRef]
- Park, S.K.; Hyun, S.H.; In, G.; Park, C.K.; Kwak, Y.S.; Jang, Y.J.; Kim, B.; Kim, J.H.; Han, C.K. The antioxidant activities of Korean Red Ginseng (Panax ginseng) and ginsenosides: A systemic review through in vivo and clinical trials. J. Ginseng Res. 2021, 45, 41–47. [Google Scholar] [CrossRef]
- Hwang, Y.P.; Jeong, H.G. Ginsenoside Rb1 protects against 6-hydroxydopamine-induced oxidative stress by increasing heme oxygenase-1 expression through an estrogen receptor-related PI3K/Akt/Nrf2-dependent pathway in human dopaminergic cells. Toxicol Appl. Pharmacol. 2010, 242, 18–28. [Google Scholar] [CrossRef]
- Zeng, X.; Li, J.; Li, Z. Ginsenoside Rd mitigates myocardial ischemia-reperfusion injury via Nrf2/HO-1 signaling pathway. Int. J. Clin. Exp. Med. 2015, 8, 14497–14504. [Google Scholar]
- Katsumata, Y.; Kanzaki, H.; Honda, Y.; Tanaka, T.; Yamaguchi, Y.; Itohiya, K.; Fukaya, S.; Miyamoto, Y.; Narimiya, T.; Wada, S.; et al. Single Local Injection of Epigallocatechin Gallate-Modified Gelatin Attenuates Bone Resorption and Orthodontic Tooth Movement in Mice. Polymers 2018, 10, 1384. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Sun, Y. Nrf2 is required for suppressing osteoclast RANKL-induced differentiation in RAW 264.7 cells via inactivating cannabinoid receptor type 2 with AM630. Regen. Ther. 2020, 14, 191–195. [Google Scholar] [CrossRef]
- Florczyk-Soluch, U.; Jozefczuk, E.; Stepniewski, J.; Bukowska-Strakova, K.; Mendel, M.; Viscardi, M.; Nowak, W.N.; Jozkowicz, A.; Dulak, J. Various roles of heme oxygenase-1 in response of bone marrow macrophages to RANKL and in the early stage of osteoclastogenesis. Sci. Rep. 2018, 8, 10797. [Google Scholar] [CrossRef] [Green Version]
- Hess, F.G., Jr.; Parent, R.A.; Cox, G.E.; Stevens, K.R.; Becci, P.J. Reproduction study in rats or ginseng extract G115. Food Chem. Toxicol. 1982, 20, 189–192. [Google Scholar] [CrossRef]
- Bilia, A.R.; Bergonzi, M.C. The G115 standardized ginseng extract: An example for safety, efficacy, and quality of an herbal medicine. J. Ginseng Res. 2020, 44, 179–193. [Google Scholar] [CrossRef]
- Van Kampen, J.; Robertson, H.; Hagg, T.; Drobitch, R. Neuroprotective actions of the ginseng extract G115 in two rodent models of Parkinson’s disease. Exp. Neurol. 2003, 184, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Terstege, D.J.; MacDonald, D.S.; Tasker, R.A. Standardised ginseng extract G115(R) potentiates the antidepressant-like properties of fluoxetine in the forced swim test. Acta Neuropsychiatr. 2021, 33, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Lelovas, P.P.; Xanthos, T.T.; Thoma, S.E.; Lyritis, G.P.; Dontas, I.A. The laboratory rat as an animal model for osteoporosis research. Comp. Med. 2008, 58, 424–430. [Google Scholar] [PubMed]
- Ren, Y.; Maltha, J.C.; Van ’t Hof, M.A.; Kuijpers-Jagtman, A.M. Age effect on orthodontic tooth movement in rats. J. Dent. Res. 2003, 82, 38–42. [Google Scholar] [CrossRef]
- King, G.J.; Fischlschweiger, W. The effect of force magnitude on extractable bone resorptive activity and cemental cratering in orthodontic tooth movement. J. Dent. Res. 1982, 61, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Heller, I.J.; Nanda, R. Effect of metabolic alteration of periodontal fibers on orthodontic tooth movement. An experimental study. Am. J. Orthod. 1979, 75, 239–258. [Google Scholar] [CrossRef]
- Ahmad Akhoundi, M.S.; Shaygan-Mehr, M.; Keshvad, M.A.; Etemad Moghaddam, S.; Alaeddini, M.; Dehpour, A.; Mirhashemi, A.H. Effect of amitriptyline on orthodontic tooth movement in rats: An experimental study. J. Dent. Res. Dent. Clin. Dent. Prospects 2020, 14, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Mirhashemi, A.H.; Akhoundi, M.S.; Ghazanfari, R.; Etemad-Moghadam, S.; Alaeddini, M.; Khorshidian, A.; Dehpour, A.R.; Momeni, N. Assessment of the Role of NO-cGMP Pathway in Orthodontic Tooth Movement Using PDE5 Inhibitors: An Animal Study. J. Dent. 2016, 13, 388–393. [Google Scholar]
- Seifi, M.; Asefi, S.; Hatamifard, G.; Lotfi, A. Effect of local injection of Zolena, zoledronic acid made in Iran, on orthodontic tooth movement and root and bone resorption in rats. J. Dent. Res. Dent. Clin. Dent. Prospects 2017, 11, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Talebian, R.; Jafari, F.; Dehpour, A.R.; Gruber, R. Effects of lithiumchloride and nitric oxide Inhibitor on orthodontic tooth movement in the rat. Appl. Sci. 2021, 11, 3607. [Google Scholar] [CrossRef]
- Shirazi, M.; Vaziri, H.; Salari, B.; Motahhari, P.; Etemad-Moghadam, S.; Dehpour, A.R. The effect of caffeine on orthodontic tooth movement in rats. Iran. J. Basic Med. Sci. 2017, 20, 260–264. [Google Scholar] [CrossRef]
- Shirazi, M.; Nilforoushan, D.; Alghasi, H.; Dehpour, A.R. The role of nitric oxide in orthodontic tooth movement in rats. Angle Orthod. 2002, 72, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Park, E.K.; Shin, Y.W.; Lee, H.U.; Kim, S.S.; Lee, Y.C.; Lee, B.Y.; Kim, D.H. Inhibitory effect of ginsenoside Rb1 and compound K on NO and prostaglandin E2 biosyntheses of RAW264.7 cells induced by lipopolysaccharide. Biol. Pharm. Bull. 2005, 28, 652–656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, Y.B.; An, Y.R.; Kim, S.J.; Park, H.W.; Jung, J.W.; Kyung, J.S.; Hwang, S.Y.; Kim, Y.S. Lipid metabolic effect of Korean red ginseng extract in mice fed on a high-fat diet. J. Sci. Food Agric. 2012, 92, 388–396. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, H.J.; Lee, Y.H.; Lee, B.W.; Cha, B.S.; Kang, E.S.; Ahn, C.W.; Park, J.S.; Kim, H.J.; Lee, E.Y.; et al. Korean red ginseng (Panax ginseng) improves insulin sensitivity in high fat fed Sprague-Dawley rats. Phytother. Res. 2012, 26, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Levy, B.D. Resolvins in inflammation: Emergence of the pro-resolving superfamily of mediators. J. Clin. Invest. 2018, 128, 2657–2669. [Google Scholar] [CrossRef] [PubMed]
- Mirfazaelian, A.; Monzavi, A. Use of a feeler gauge to measure the gap between adjacent teeth. J. Prosthet. Dent. 2003, 90, 613. [Google Scholar] [CrossRef]
- Siegel, R.K. Ginseng abuse syndrome. Problems with the panacea. JAMA 1979, 241, 1614–1615. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Woo, J.Y.; Han, C.K.; Chang, I.M. Safety Analysis of Panax Ginseng in Randomized Clinical Trials: A Systematic Review. Medicines 2015, 2, 106–126. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talebian, R.; Mollabashi, V.; Motaghedifard, A.; Gruber, R. Korean Panax Ginseng Reduces Orthodontic Tooth Movement in Rats. Appl. Sci. 2021, 11, 8856. https://doi.org/10.3390/app11198856
Talebian R, Mollabashi V, Motaghedifard A, Gruber R. Korean Panax Ginseng Reduces Orthodontic Tooth Movement in Rats. Applied Sciences. 2021; 11(19):8856. https://doi.org/10.3390/app11198856
Chicago/Turabian StyleTalebian, Reza, Vahid Mollabashi, Arezoo Motaghedifard, and Reinhard Gruber. 2021. "Korean Panax Ginseng Reduces Orthodontic Tooth Movement in Rats" Applied Sciences 11, no. 19: 8856. https://doi.org/10.3390/app11198856
APA StyleTalebian, R., Mollabashi, V., Motaghedifard, A., & Gruber, R. (2021). Korean Panax Ginseng Reduces Orthodontic Tooth Movement in Rats. Applied Sciences, 11(19), 8856. https://doi.org/10.3390/app11198856





