Non-Surgical Touch-Up with Hyaluronic Acid Fillers Following Facial Reconstructive Surgery
Abstract
1. Introduction
2. Materials and Methods
2.1. Case 1
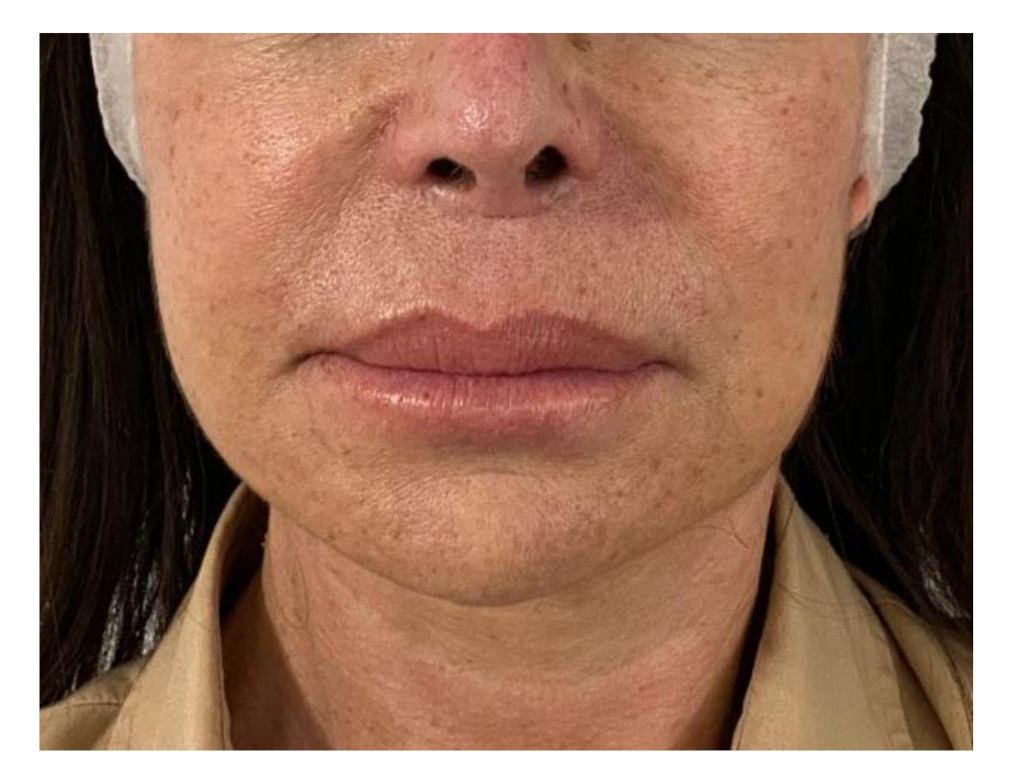
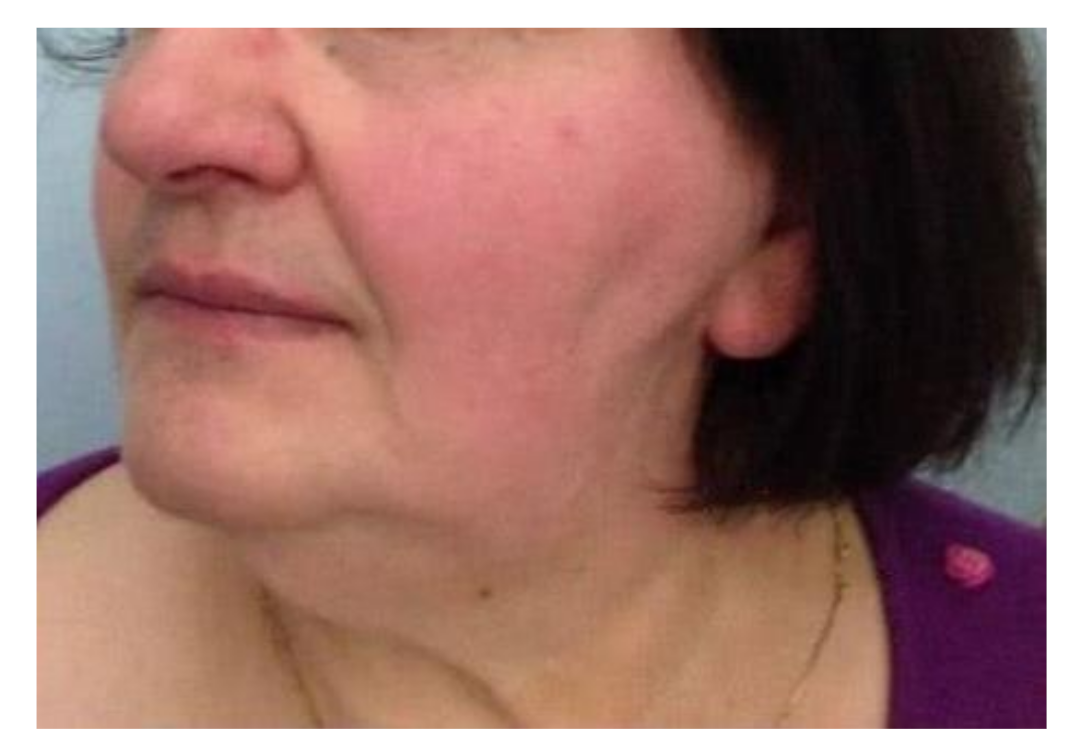
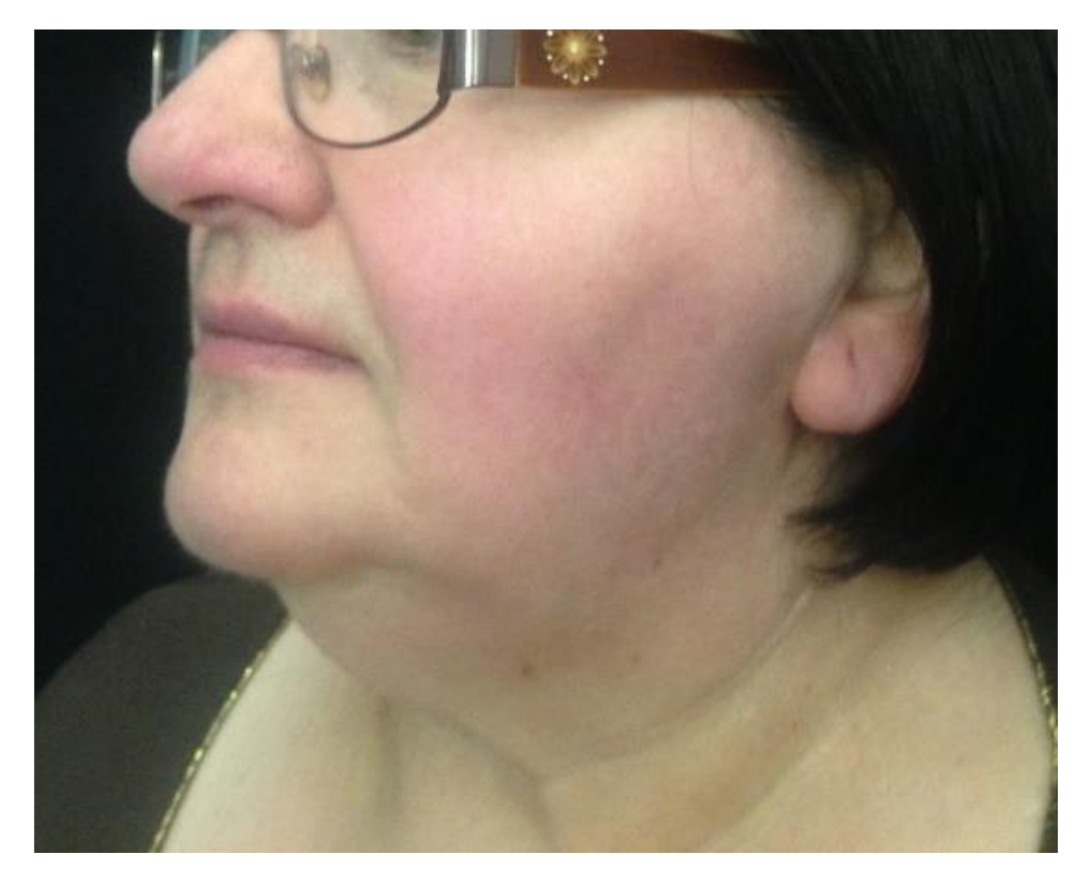
2.2. Case 2
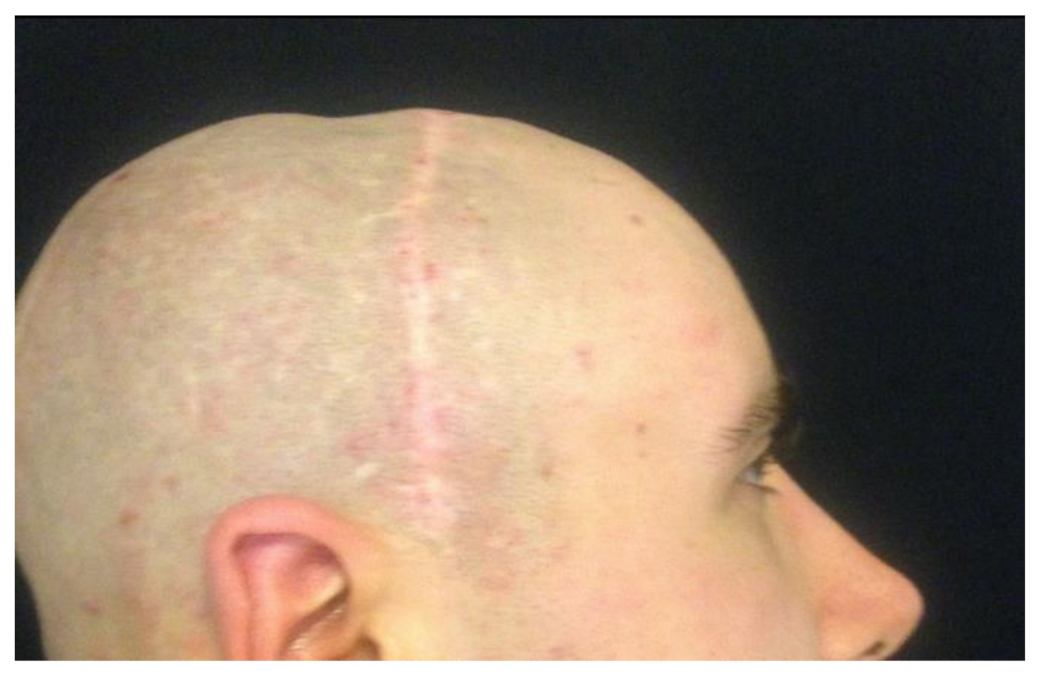
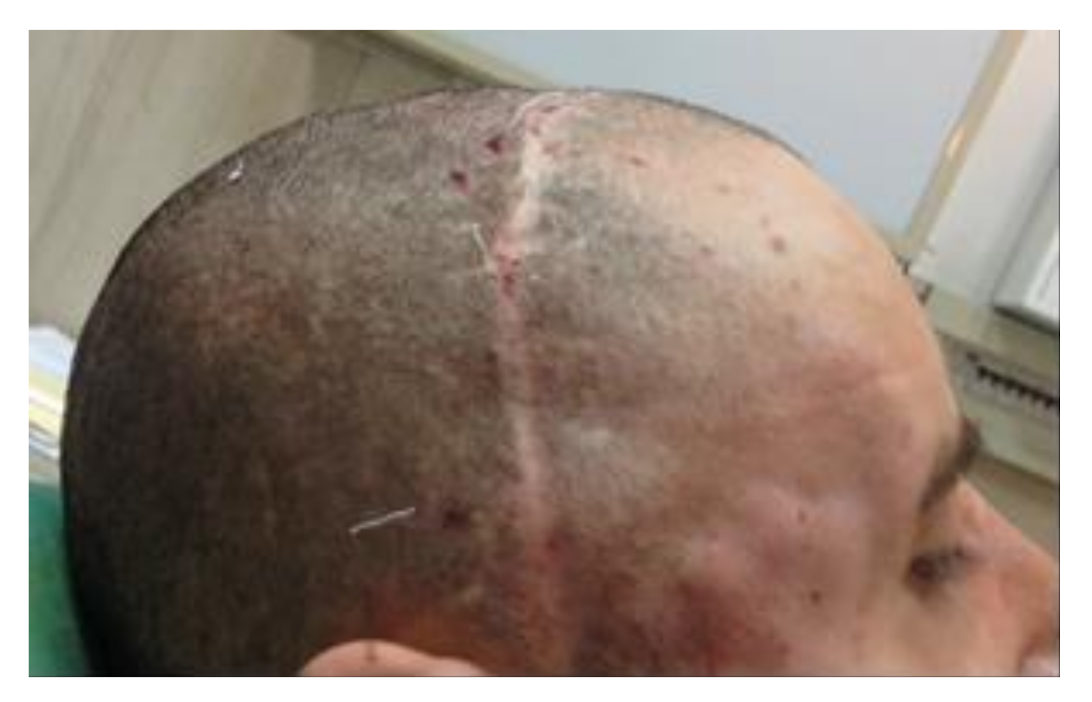
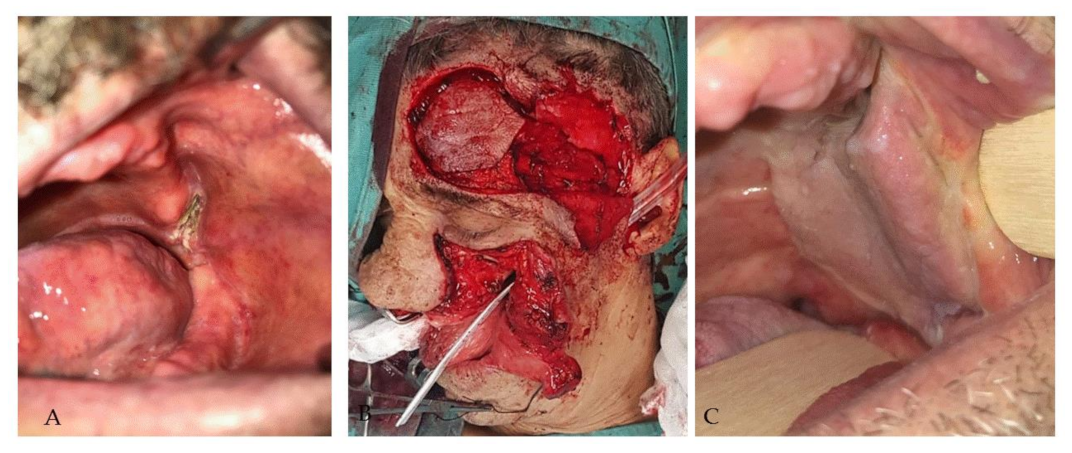
2.3. Case 3
2.4. Case 4
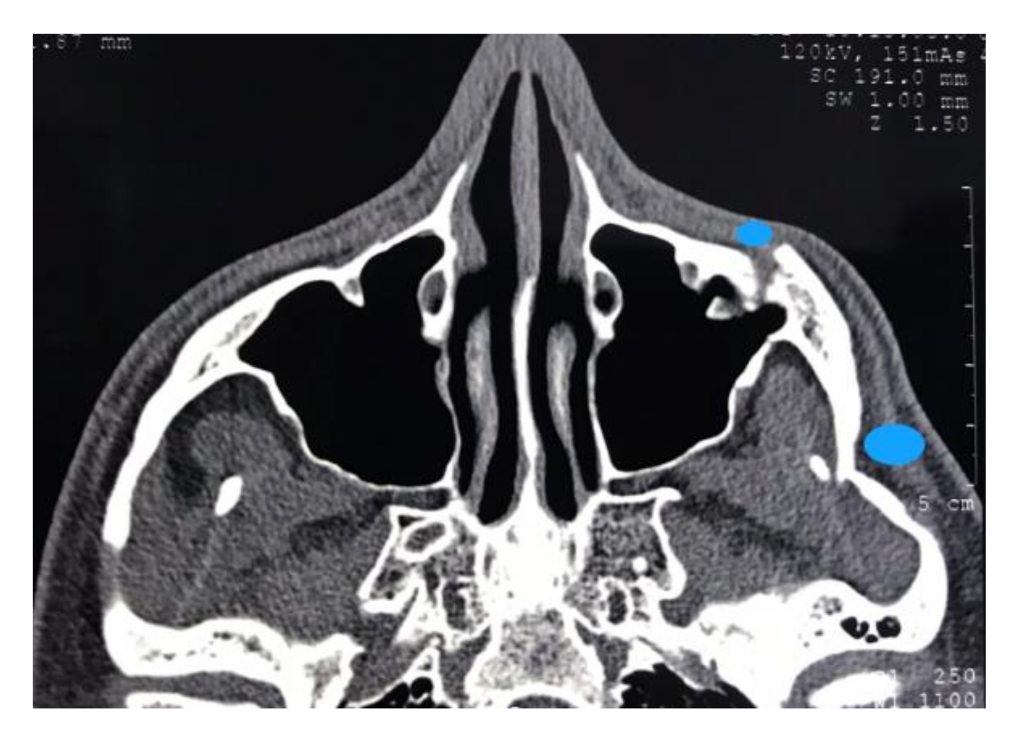
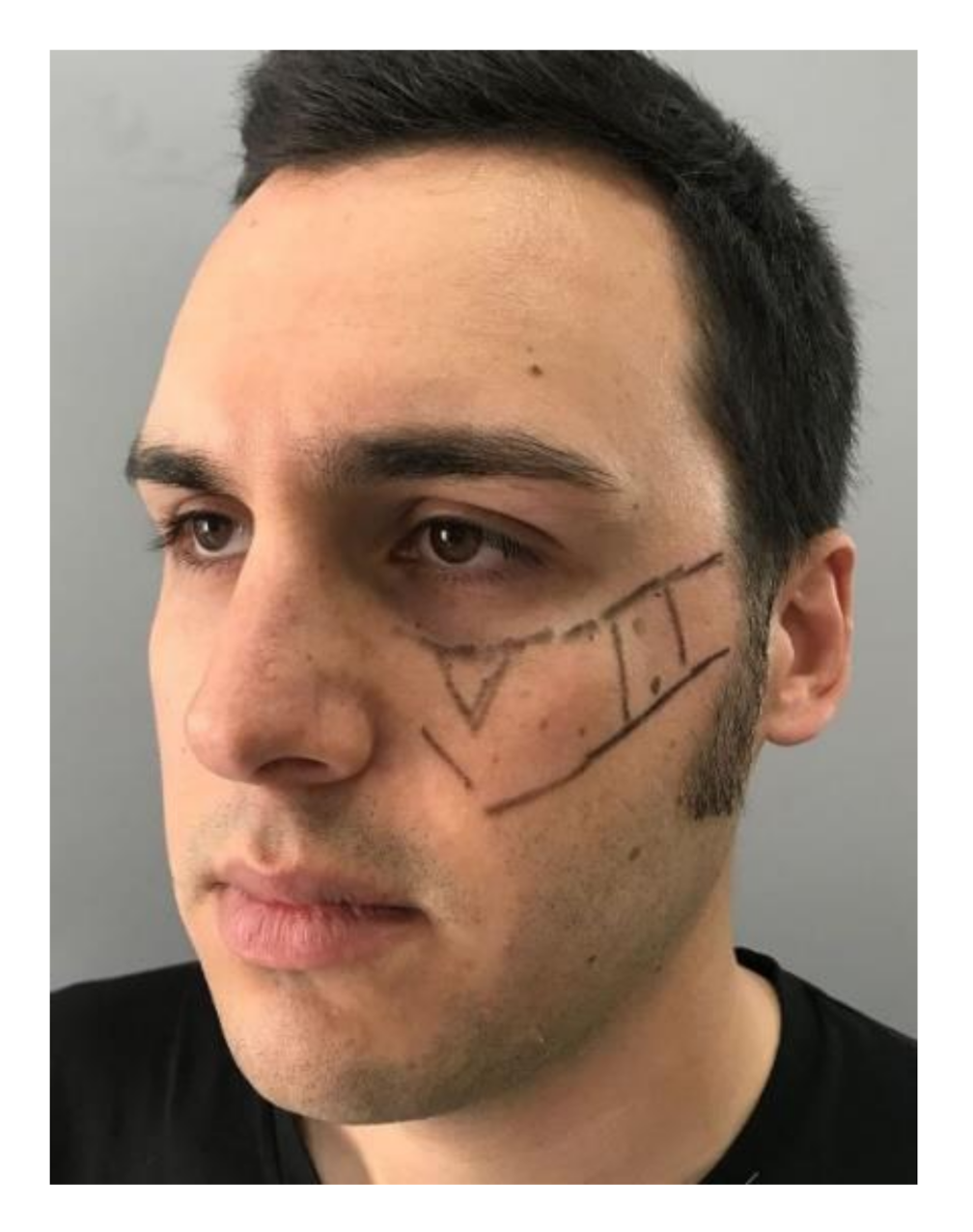
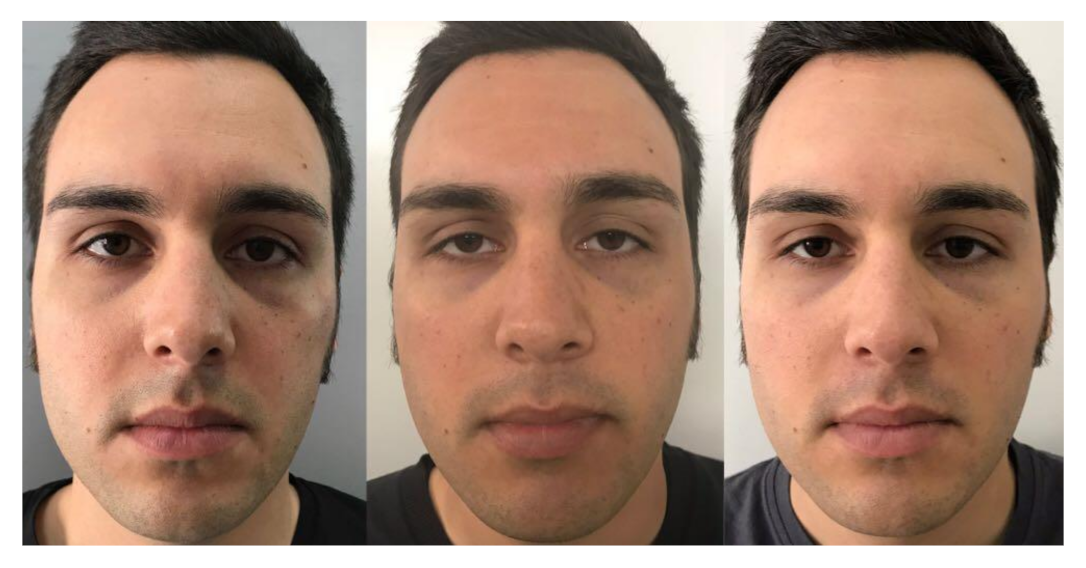
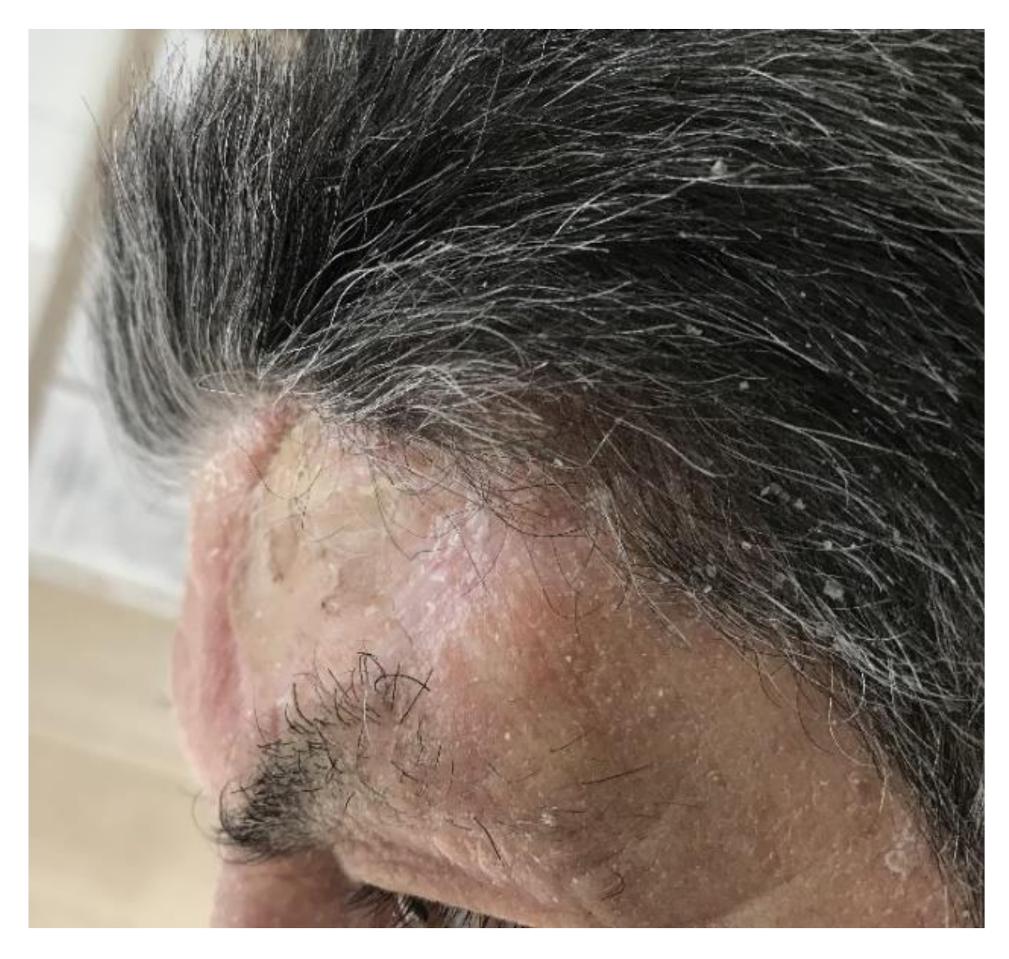
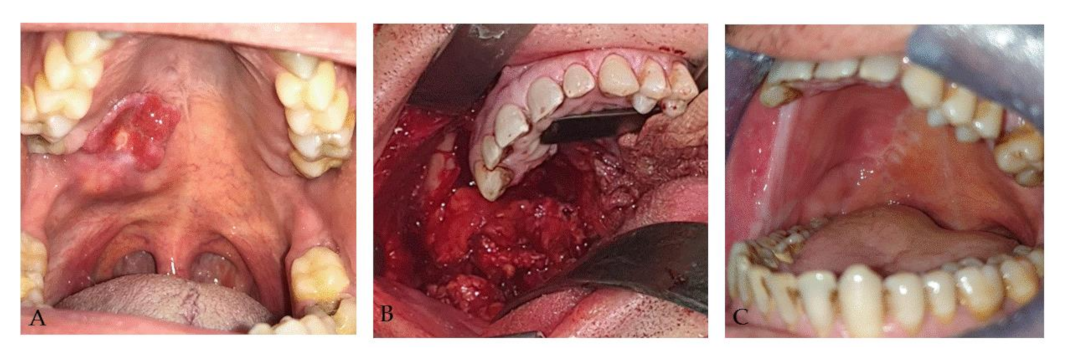
2.5. Case 5
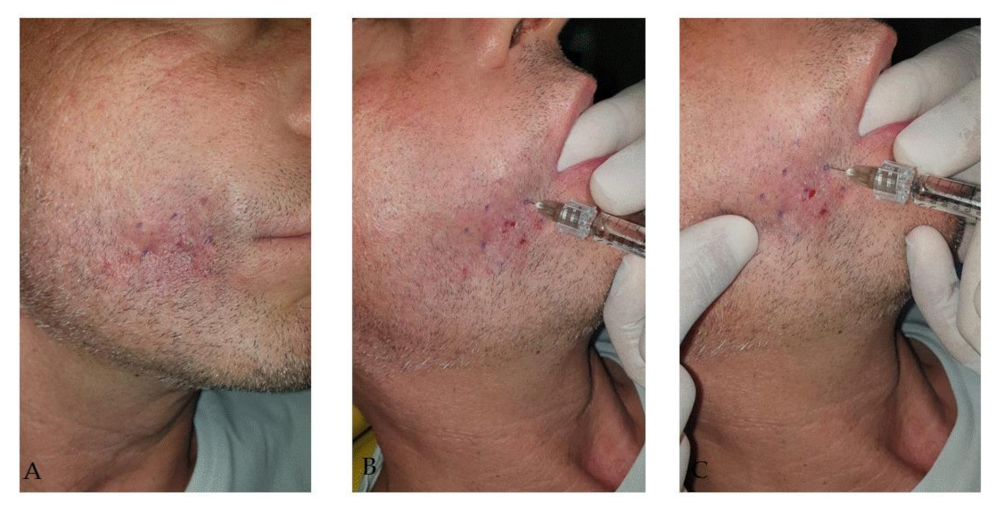
2.6. Case 6
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Neuber, G.A. Fettransplantation. Chir. Kongr. Verhandl. Dsch. Gesellch. Chir. 1893, 22, 66. [Google Scholar]
- Jones, D.H. Semipermanent and Permanent Injectable Fillers. Dermatol. Clin. 2009, 27, 433–444. [Google Scholar] [CrossRef]
- Broder, K.W.; Cohen, S.R. An Overview of Permanent and Semipermanent Fillers. Plast. Reconstr. Surg. 2006, 118, 7S–14S. [Google Scholar] [CrossRef]
- Chirico, F.; Bove, P.; Fragola, R.; Cosenza, A.; De Falco, N.; Giudice, G.L.; Audino, G.; Rauso, G. Biphasic Injection for Masseter Muscle Reduction with Botulinum Toxin. Appl. Sci. 2021, 11, 6478. [Google Scholar] [CrossRef]
- Rauso, R.; Tartaro, G.; Chirico, F.; Zerbinati, N.; Albani, G.; Rugge, L. Rhinofilling with hyaluronic acid thought as a cartilage graft. J. Cranio Maxillofac. Surg. 2020, 48, 223–228. [Google Scholar] [CrossRef]
- Öhrlund, J.Å.; Edsman, K.L. The myth of the “biphasic” hyaluronic acid filler. Dermatol. Surg. 2015, 41, S358–S364. [Google Scholar] [CrossRef] [PubMed]
- Borrell, M.; Leslie, D.B.; Tezel, A. Lift capabilities of hyaluronic acid fillers. J. Cosmet. Laser Ther. 2011, 13, 21–27. [Google Scholar] [CrossRef]
- Rauso, R.; Giudice, G.L.; Zerbinati, N.; Tartaro, G. Hyaluronic Acid Injections as Nonsurgical Alternative in case of Delayed Diagnosis of Malar Arch Fracture: Case Report and Literature Review. Case Rep. Surg. 2019, 1360741. [Google Scholar] [CrossRef]
- González, N.; Goldberg, D.J. Update on the Treatment of Scars. J. Drugs Dermatol. 2019, 18, 550. [Google Scholar]
- Kavoussi, H.; Kavoussi, R. Revision of a depressed scar across a relaxed skin tension line by punch elevation, filler injection, and fractional CO2 laser. Acta Dermatovenerol. Alp. Pannonica Adriat. 2019, 28, 129–130. [Google Scholar] [CrossRef]
- Sahan, A.; Tamer, F. Restoring facial symmetry through non-surgical cosmetic procedures after permanent facial paralysis: A case report. Acta Dermatovenerol. Alp. Pannonica Adriat. 2017, 26, 41–42. [Google Scholar] [CrossRef]
- Rauso, R.; Albani, G.; Salini, C.; Rugge, L.; Chirico, F.; Tartaro, G. Continuum: Late Onset Complications Secondary to Polyacrylamide Hydrogel-Based Filler for Rehabilitation of HIV-Related Facial Lipoatropy. Aesthetic Surg. J. 2019, 39, NP152–NP155. [Google Scholar] [CrossRef]
- Rauso, R.; Califano, L.; Rugge, L.; Chirico, F.; Tartaro, G. Late onset complications secondary to polyacrylamide hydrogel-based filler for rehabilitation of HIV-related facial lipoatropy. Aesthetic Surg. J. 2018, 38, N170–N174. [Google Scholar] [CrossRef]
- Rauso, R. 5-Year Study of a Polyacrylamide Hydrogel-Based Filler for Rehabilitation of HIV-Related Facial Lipoatrophy. Aesthetic Surg. J. 2015, 35, 1021–1029. [Google Scholar] [CrossRef]
- Ho, D.; Jagdeo, J. Safety and Efficacy of a Volumizing Hyaluronic Acid Filler for Treatment of HIV-Associated Facial Lipoatrophy. JAMA Dermatol. 2017, 153, 61–65. [Google Scholar] [CrossRef]
- Jagdeo, J.; Ho, D.; Lo, A.; Carruthers, A. A systematic review of filler agents for aesthetic treatment of HIV facial lipoatrophy (FLA). J. Am. Acad. Dermatol. 2015, 73, 1040–1054.e14. [Google Scholar] [CrossRef] [PubMed]
- Chirico, F.; Rauso, G.; Fragola, R.; Giudice, G.L.; Pinto, L.; De Falco, N.; Cosenza, A.; Rugge, L.; Sgaramella, N.; Tartaro, G. Complications Following Non-Surgical Aesthetic Treatments in HIV+ Patients Receiving Antiretroviral Therapy: A 12-Years Experience. Appl. Sci. 2021, 11, 4059. [Google Scholar] [CrossRef]
- Franchi, G.; Neiva-Vaz, C.; Picard, A.; Vazquez, M.P. Facial Injections of Hyaluronic Acid-Based Fillers for Malformations. Preliminary Study Regarding Scar Tissue Improvement and Cosmetic Betterment. Ann. Chir. Plast. Esthet. 2018, 63, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Inbal, A.; Lemelman, B.T.; Millet, E.; Greensmith, A. Tissue Expansion Using Hyaluronic Acid Filler for Single-Stage Ear Reconstruction: A Novel Concept for Difficult Areas. Aesthetic Surg. J. 2017, 37, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Kajdic, N.; Spazzapan, P.; Velnar, T. Craniosynostosis—Recognition, Clinical Characteristics, and Treatment. Bosn. J. Basic Med. Sci. 2018, 18, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Dempsey, R.F.; Monson, L.A.; Maricevich, R.S.; Truong, T.A.; Olarunnipa, S.; Lam, S.K.; Dauser, R.C.; Hollier, L.H., Jr.; Buchanan, E.P. Nonsyndromic Craniosynostosis. Clin. Plast. Surg. 2019, 46, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Gosain, A.K. Hydroxyapatite Cement Paste Cranioplasty for the Treatment of Temporal Hollowing After Cranial Vault Remodeling in a Growing Child. J. Craniofac. Surg. 1997, 8, 506–511. [Google Scholar] [CrossRef]
- Grant, J.H., 3rd; Roberts, T.S.; Loeser, J.D.; Gruss, J.S. Onlay Bone Graft Augmentation for Refined Correction of Coronal Synostosis. Cleft Palate Craniofac. J. 2002, 39, 546–554. [Google Scholar] [CrossRef]
- Hollier, L.; Stal, S. Calcium Phosphate Cement Cranioplasty: Clinical and Radiographic Follow-up. Int. J. Plast. Surg. 2003, 2, 1–6. [Google Scholar]
- Persing, J.A.; Mayer, P.L.; Spinelli, H.M.; Miller, L.; Criscuolo, G.R. Prevention of “Temporal Hollowing” After Fronto-orbital Advancement for Craniosynostosis. J. Craniofac. Surg. 1994, 5, 271–274. [Google Scholar] [CrossRef]
- Clauser, L.C.; Tieghi, R.; Consorti, G. Parry–Romberg syndrome: Volumetric regeneration by structural fat grafting technique. J. Cranio Maxillofac. Surg. 2010, 38, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Clauser, L.C.; Tieghi, R.; Galiè, M.; Carinci, F. Structural fat grafting: Facial volumetric restoration in complex reconstructive surgery. J. Craniofac. Surg. 2011, 22, 1695–1701. [Google Scholar] [CrossRef]
- Petrone, P.; Trecca, E.; Cassano, M.; Quaranta, N.A.; Fiorella, M.L.; De Santis, V.; Ressa, M.; Dalena, E.; Dalena, P.; Fortunato, F.; et al. Effects of ischemic preconditioning with resveratrol on epigastric rat flap: New perspectives for head and neck reconstruction. J. Biol. Regul. Homeost. Agents 2019, 33, 1603–1607. [Google Scholar]
- Portincasa, A.; Cecchino, L.R.; Trecca, E.; Lembo, F.; Annacontini, L.; Ciancio, F.; Corsi, F.; Cassano, M.; Parisi, D. A rare case of Brooke-Spiegler syndrome: Integrated surgical treatment of multiple giant eccrine spiradenomas of the head and neck in a young girl. Int. J. Surg. Case Rep. 2018, 51, 277–281. [Google Scholar] [CrossRef]
- Trecca, E.M.C.; Adunka, O.F.; Mattingly, J.K.; Hiss, M.M.; Cassano, M.; Malhotra, P.S.; Riggs, W.J. Electrocochleography Observations in a Series of Cochlear Implant Electrode Tip Fold-Overs. Otol. Neurotol. 2020, 42, e433–e437. [Google Scholar] [CrossRef]
- Portincasa, A.; Trecca, E.; Ciancio, F.; Annacontini, L.; Bufo, P.; Fortunato, F.; Cecchino, L.R.; Parisi, D.; Cassano, M. The role of lipofilling in reconstructions with dermal regeneration template: Clinical and histological assessment. J. Biol. Regul. Homeost. Agents 2018, 32, 171–176. [Google Scholar] [PubMed]
- Illouz, Y.G. The fat cell ‘graft’, a new technique to fill depressions. Plast. Reconstr. Surg. 1986, 78, 122–123. [Google Scholar] [CrossRef] [PubMed]
- Illouz, Y.G. Body contouring by lipolysis: A five-year experience with over 3000 cases. Plast. Reconstr. Surg. 1983, 72, 591–597. [Google Scholar] [CrossRef]
- Illouz, Y.G. De l’utilization de la graisse aspiree pour combler les defects cutanes observations. Aesthetic Plast. Surg. Rev. Chir. Esthet. Lang. Fr. 1985, 10, 13–20. [Google Scholar]
- Wetmore, S.J. Injection of Fat for Soft Tissue Augmentation. Laryngoscope 1989, 99, 50–57. [Google Scholar] [CrossRef]
- Teimourian, B. Blindness following fat injections [letter]. Plast. Reconstr. Surg. 1988, 82, 361. [Google Scholar] [CrossRef]
- Illouz, Y.G. Present results of fat injection. Aesthetic Plast. Surg. 1988, 12, 175–181. [Google Scholar] [CrossRef]
- Chia, C.T.; Neinstein, R.M.; Theodorou, S.J. Evidence-Based Medicine: Liposuction. Plast. Reconstr. Surg. 2017, 139, 267e–274e. [Google Scholar] [CrossRef]
- Agris, J. Autologous fat transplantation: A 3-year study [abstract]. Am. J. Cosmet. Surg. 1987, 4, 111. [Google Scholar] [CrossRef]
- Šmahel, J. Adipose Tissue in Plastic Surgery. Ann. Plast. Surg. 1986, 16, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Zocchi, M. Methode de production de collagene autologue par traitement du tissue graisseux. J. Med. Esthet. Chirur. Dermatol. 1990, 66, 105–114. [Google Scholar]
- Klein, J.A. The Tumescent Technique for Lipo-Suction Surgery. Am. J. Cosmet. Surg. 1987, 4, 263–267. [Google Scholar] [CrossRef]
- Billings, E., Jr.; May, J.W., Jr. Historical review and present status of free graft and autotransplantation in plastic and reconstructive surgery. Plast. Reconstr. Surg. 1989, 83, 368–381. [Google Scholar] [CrossRef] [PubMed]
- Roenigk, H., Jr.; Rubenstein, R. Combined scalp reduction and autologous fat implant treatment of localized soft tissue defects. J. Dermatol. Surg. Oncol. 1988, 14, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Moscona, R.; Ullman, Y.; Har-Shai, Y.; Hirshowitz, B. Free-fat injections for the correction of hemifacial atrophy. Plast. Reconstr. Surg. 1989, 84, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Tonnard, P.; Verpaele, A.; Carvas, M. Fat Grafting for Facial Rejuvenation with Nanofat Grafts. Clin. Plast. Surg. 2019, 47, 53–62. [Google Scholar] [CrossRef]
- Shukla, L.; Yuan, Y.; Shayan, R.; Greening, D.; Karnezis, T. Fat Therapeutics: The Clinical Capacity of Adipose-Derived Stem Cells and Exosomes for Human Disease and Tissue Regeneration. Front. Pharmacol. 2020, 11, 158. [Google Scholar] [CrossRef]
- Bora, P.; Majumdar, A.S. Adipose tissue-derived stromal vascular fraction in regenerative medicine: A brief review on biology and translation. Stem Cell Res. Ther. 2017, 8, 158. [Google Scholar] [CrossRef]
- Jatana, K.R.; Smith, S.P., Jr. The scientific basis for lipotransfer: Is it the ideal filler? Facial Plast. Surg. Clin. N. Am. 2008, 16, 443–448. [Google Scholar] [CrossRef]
- Coleman, S.R. Structural fat grafting: More than a permanent filler. Plast. Reconstr. Surg. 2006, 118, 108S–120S. [Google Scholar] [CrossRef]
- Del Vecchio, D.; Wall, S., Jr. Expansion Vibration Lipofilling: A New Technique in Large-Volume Fat Transplantation. Plast. Reconstr. Surg. 2018, 141, 639e–649e. [Google Scholar] [CrossRef] [PubMed]
- Simonacci, F.; Bertozzi, N.; Grieco, M.P.; Grignaffini, E.; Raposio, E. Procedure, applications, and outcomes of autologous fat grafting. Ann. Med. Surg. 2017, 27, 49–60. [Google Scholar] [CrossRef]
- Gause, T.M., 2nd; Kling, R.E.; Sivak, W.N.; Marra, K.G.; Rubin, J.P.; Kokai, L.E. Particle size in fat graft retention: A review on the impact of harvesting technique in lipofilling surgical outcomes. Adipocyte 2014, 3, 273–279. [Google Scholar] [CrossRef]
- Gentile, P.; De Angelis, B.; Di Pietro, V.; Amorosi, V.; Scioli, M.G.; Orlandi, A.; Cervelli, V. Gentle is better: The original “gentle technique” for fat placement in breast lipofilling. J. Cutan. Aesthetic Surg. 2018, 11, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Peer, L.A. Loss of weight and volume in human fat grafts: With postulation of a “cell survival theory”. Plast. Reconstr. Surg. 1950, 5, 217–230. [Google Scholar] [CrossRef]
- Nguyen, A.; Pasyk, K.A.; Bouvier, T.N.; Hassett, C.A.; Argenta, L.C. Comparative study of survival of autologous adipose tissue taken and transplanted by different techniques. Plast. Reconstr. Surg. 1990, 85, 378–386. [Google Scholar] [CrossRef]
- Epply, B.A.; Sidner, R.A.; Platis, J.M. Bioactivation of free-fat transfers: A potential new approach to improving graft survival. Plast. Reconstr. Surg. 1992, 90, 1021–1030. [Google Scholar] [CrossRef]
- Saunders, M.C.; Keller, J.T.; Dunsker, S.B.; Mayfield, F.H. Survival of Autologous Fat Grafts in Humans and in Mice. Connect. Tissue Res. 1981, 8, 85–91. [Google Scholar] [CrossRef]
- Gurney, C.E. Experimental study of the behavior of free fat transplants. Surgery 1938, 3, 680. [Google Scholar]
- Johnson, G.W. Body Contouring by Macroinjection of Autogenous Fat. Am. J. Cosmet. Surg. 1987, 4, 103–109. [Google Scholar] [CrossRef]
- Gormley, D.E.; Eremia, S. Quantitative Assessment of Augmentation Therapy. J. Dermatol. Surg. Oncol. 1990, 16, 1147–1151. [Google Scholar] [CrossRef]
- Hörl, H.W.; Feller, A.-M.; Biemer, E. Technique for Liposuction Fat Reimplantation and Long-Term Volume Evaluation by Magnetic Resonance Imaging. Ann. Plast. Surg. 1991, 26, 248–258. [Google Scholar] [CrossRef]
- Rigotti, G.; Marchi, A.; Micciolo, R.; Baroni, G. Autologous fat grafting in breast cancer patients. Breast 2012, 21, 690. [Google Scholar] [CrossRef]
- Zarei, F.; Abbaszadeh, A. Application of Cell Therapy for Anti-Aging Facial Skin. Curr. Stem. Cell Res. Ther. 2019, 14, 244–248. [Google Scholar] [CrossRef]
- Naderi, N.; Combellack, E.J.; Griffin, M.; Sedaghati, T.; Javed, M.; Findlay, M.W.; Wallace, C.G.; Mosahebi, A.; Butler, P.E.; Seifalian, A.M.; et al. The regenerative role of adipose-derived stem cells (ADSC) in plastic and reconstructive surgery. Int. Wound J. 2016, 14, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Won, C.H.; Park, G.-H.; Wu, X.; Tran, T.-N.; Park, K.-Y.; Park, B.-S.; Kim, D.Y.; Kwon, O.; Kim, K.-H. The Basic Mechanism of Hair Growth Stimulation by Adipose-derived Stem Cells and Their Secretory Factors. Curr. Stem Cell Res. Ther. 2017, 12, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Addeman, B.T.; Kutty, S.; Perkins, T.G.; Soliman, A.S.; Wiens, C.N.; McCurdy, C.M.; Beaton, M.D.; Hegele, R.A.; McKenzie, C.A. Validation of volumetric and single-slice MRI adipose analysis using a novel fully automated segmentation method. J. Magn. Reson. Imaging 2014, 41, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Glogau, R.G. Microlipoinjection. Arch. Dermatol. 1988, 124, 1340–1343. [Google Scholar] [CrossRef] [PubMed]
- Haiavy, J.; Elias, H. Injectable Fillers in the Upper Face. Atlas. Oral Maxillofac. Surg. Clin. N. Am. 2016, 24, 105–116. [Google Scholar] [CrossRef]
- Pu, L.L. Mechanisms of Fat Graft Survival. Ann. Plast. Surg. 2016, 77, S84–S86. [Google Scholar] [CrossRef] [PubMed]
- Truswell, W. Aging Changes of the Periorbita, Cheeks, and Midface. Facial Plast. Surg. 2013, 29, 3–12. [Google Scholar] [CrossRef]
- Herold, C.; Ueberreiter, K.; Busche, M.N.; Vogt, P.M. Autologous Fat Transplantation: Volumetric Tools for Estimation of Volume Survival. A Systematic Review. Aesthetic Plast. Surg. 2013, 37, 380–387. [Google Scholar] [CrossRef]
- Kaufman, M.R.; Bradley, J.P.; Dickinson, B.; Heller, J.B.; Wasson, K.; O’Hara, C.; Huang, C.; Gabbay, J.; Ghadjar, K.; Miller, T.A. Autologous Fat Transfer National Consensus Survey: Trends in Techniques for Harvest, Preparation, and Application, and Perception of Short- and Long-Term Results. Plast. Reconstr. Surg. 2007, 119, 323–331. [Google Scholar] [CrossRef]
- Li, Y.; Mou, S.; Xiao, P.; Li, G.; Li, J.; Tong, J.; Wang, J.; Yang, J.; Sun, J.; Wang, Z. Delayed two steps PRP injection strategy for the improvement of fat graft survival with superior angiogenesis. Sci. Rep. 2020, 10, 5231–5238. [Google Scholar] [CrossRef]
- Modarressi, A. Platlet Rich Plasma (PRP) Improves Fat Grafting Outcomes. World J. Plast. Surg. 2013, 2, 6–13. [Google Scholar]
- Kanchwala, S.K.; Holloway, L.; Bucky, L.P. Reliable soft tissue augmentation: A clinical comparison of injectable soft-tissue fillers for facial-volume augmentation. Ann. Plast. Surg. 2005, 55, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Maamari, R.N.; Massry, G.G.; Holds, J.B. Complications Associated with Fat Grafting to the Lower Eyelid. Facial Plast. Surg. Clin. N. Am. 2019, 27, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Cuzalina, A.; Guerrero, A.V. Complications in Fat Grafting. Atlas. Oral Maxillofac. Surg. Clin. N. Am. 2018, 26, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Belyea, J.; Hart, R.; Trites, J.; Taylor, S.M. A case of autologous microfat grafting in lip reconstruction of a whistle deformity following cancer treatment. Can. J. Plast. Surg. 2010, 18, e53–e54. [Google Scholar] [CrossRef] [PubMed]
- Ueland, H.O.; Nilsen, R.M.; Rødahl, E.; Jensen, S.A. Hyaluronic acid is superior to autologous fat for treatment of temporal hollowing after lateral orbital wall decompression: A prospective interventional trial. J. Plast. Reconstr. Aesthetic Surg. 2019, 72, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Shuck, J.; Iorio, M.L.; Hung, R.; Davison, S.P. Autologous fat grafting and injectable dermal fillers for human immunodeficiency virus-associated facial lipodystrophy: A comparison of safety, efficacy, and long-term treatment outcomes. Plast. Reconstr. Surg. 2013, 131, 499–506. [Google Scholar] [CrossRef]
- Rohrich, R.J.; Pessa, J.E.; Ristow, B. The Youthful Cheek and the Deep Medial Fat Compartment. Plast. Reconstr. Surg. 2008, 121, 2107–2112. [Google Scholar] [CrossRef]
- Wang, F.; Garza, L.A.; Kang, S.; Varani, J.; Orringer, J.S.; Fisher, G.J.; Voorhees, J.J. In Vivo stimulation of de novo collagen production caused by cross-linked hyaluronic acid dermal filler injections in photo-damaged human skin. Arch. Dermatol. 2007, 143, 155–163. [Google Scholar] [CrossRef]
- Zerbinati, N.; Sommatis, S.; Maccario, C.; Capillo, M.; Grimaldi, G.; Alonci, G.; Protasoni, M.; Rauso, R.; Mocchi, R. Toward Physicochemical and Rheological Characterization of Different Injectable Hyaluronic Acid Dermal Fillers Cross-Linked with Polyethylene Glycol Diglycidyl Ether. Polymers 2021, 13, 948. [Google Scholar] [CrossRef]
- Zerbinati, N.; Esposito, C.; Cipolla, G.; Calligaro, A.; Monticelli, D.; Martina, V.; Golubovic, M.; Binic, I.; Sigova, J.; Gallo, A.L.; et al. Chemical and mechanical characterization of hyaluronic acid hydrogel cross-linked with polyethylen glycol and its use in dermatology. Dermatol. Ther. 2020, 33, e13747. [Google Scholar] [CrossRef] [PubMed]
- Monticelli, D.; Martina, V.; Mocchi, R.; Rauso, R.; Zerbinati, U.; Cipolla, G.; Zerbinati, N. Chemical Characterization of Hydrogels Crosslinked with Polyethylene Glycol for Soft Tissue Augmentation. Open Access Maced. J. Med. Sci. 2019, 7, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Marino, F.; Cosentino, M.; Legnaro, M.; Luini, A.; Sigova, J.; Mocchi, R.; Lotti, T.; Zerbinati, N.; Gasperina, D.; Tirant, M.; et al. Immune profile of hyaluronic acid hydrogel polyethylene glycol crosslinked: An in vitro evaluation in human polymorphonuclear leukocytes. Dermatol. Ther. 2020, 33, e13388. [Google Scholar] [CrossRef]
- Zerbinati, N.; D’Este, E.; Farina, A.; Rauso, R.; Cherubino, M.; Calligaro, A. Morphological evidences following pegylated filler treatment in human skin. J. Boil. Regul. Homeost. Agents 2017, 31, 79–85. [Google Scholar]
- Zerbinati, N.; Lotti, T.; Monticelli, D.; Martina, V.; Cipolla, G.; D’Este, E.; Calligaro, A.; Mocchi, R.; Maccario, C.; Sommatis, S.; et al. In Vitro Evaluation of the Sensitivity of a Hyaluronic Acid PEG Cross-Linked to Bovine Testes Hyaluronidase. Open Access Maced. J. Med. Sci. 2018, 6, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Bove, P.; Rugge, L.; Chirico, F. Unusual Intraoral Necrosis After Hyaluronic Acid Injections. Dermatol. Surg. 2021, 47, 1158–1160. [Google Scholar] [CrossRef]
- Rauso, R.; Zerbinati, N.; Franco, R.; Chirico, F.; Ronchi, A.; Sesenna, E.; Colella, G.; Tartaro, G. Cross-linked hyaluronic acid filler hydrolysis with hyaluronidase: Different settings to reproduce different clinical scenarios. Dermatol. Ther. 2020, 33, e13269. [Google Scholar] [CrossRef]
- Rauso, R.; Colella, G.; Franco, R.; Chirico, F.; Ronchi, A.; Federico, F.; Volpicelli, A.; Tartaro, G. Is hyaluronidase able to reverse embolism associated with hyaluronic acid filler? An anatomical case study. J. Biol. Regul. Homeost. Agents 2020, 33, 1927–1930. [Google Scholar]
- Rauso, R.; Sesenna, E.; Fragola, R.; Zerbinati, N.; Nicoletti, G.F.; Tartaro, G. Skin Necrosis and Vision Loss or Impairment After Facial Filler Injection. J. Craniofac. Surg. 2020, 31, 2289–2293. [Google Scholar] [CrossRef]
- Zerbinati, N.; Mocchi, R.; Galadari, H.; Maccario, C.; Maggi, M.; Rauso, R.; Passi, A.; Esposito, C.; Sommatis, S. In Vitro Evaluation of the Biological Availability of Hyaluronic Acid Polyethylene Glycols-Cross-Linked Hydrogels to Bovine Testes Hyaluronidase. BioMed Res. Int. 2019. [Google Scholar] [CrossRef]
- Zerbinati, N.; Lotti, T.; Monticelli, D.; Rauso, R.; González-Isaza, P.; D’Este, E.; Calligaro, A.; Sommatis, S.; Maccario, C.; Mocchi, R.; et al. In Vitro Evaluation of the Biosafety of Hyaluronic Acid PEG Cross-Linked with Micromolecules of Calcium Hydroxyapatite in Low Concentration. Open Access Maced. J. Med. Sci. 2018, 6, 15–19. [Google Scholar] [CrossRef]
- Artzi, O.; Loizides, C.; Verner, I.; Landau, M. Resistant and Recurrent Late Reaction to Hyaluronic Acid–Based Gel. Dermatol. Surg. 2016, 42, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Friedman, P.M.; Mafong, E.A.; Kauvar, A.N.; Geronemus, R.G. Safety data of injectable nonanimal stabilized hyaluronic acid gel for soft tissue augmentation. Dermatol. Surg. 2002, 28, 491–494. [Google Scholar] [PubMed]
- Becker, L.C.; Bergfeld, W.F.; Belsito, D.V.; Klaassen, C.D.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; Panel, W.C.I.R.E.; Andersen, F.A. Final Report of the Safety Assessment of Hyaluronic Acid, Potassium Hyaluronate, and Sodium Hyaluronate. Int. J. Toxicol. 2009, 28, 5–67. [Google Scholar] [CrossRef] [PubMed]
- Salti, G.; Fundarò, S.P. Evaluation of the Rheologic and Physicochemical Properties of a Novel Hyaluronic Acid Filler Range with eXcellent Three-Dimensional Reticulation (XTR™) Technology. Polymers 2020, 12, 1644. [Google Scholar] [CrossRef]
- Fallacara, A.; Durini, E.; Vertuani, S.; Manfredini, S. Hyaluronic Acid Fillers in Soft Tissue Regeneration. Facial Plast. Surg. 2017, 33, 087–096. [Google Scholar] [CrossRef] [PubMed]
- Mashiko, T.; Mori, H.; Kato, H.; Doi, K.; Kuno, S.; Kinoshita, K.; Kunimatsu, A.; Ohtomo, K.; Yoshimura, K. Semipermant volumization by an absorbable filler: Onlay injection technique to the bone. Plast. Reconstr. Surg. Glob. Open 2013, 1, e4–e14. [Google Scholar] [CrossRef]
- Eccleston, D.; Murphy, D.K. Juvederm VOLBELLA in the peri-oral area: A 12 month prospective, multicenter, open label study. Clin. Cosmet. Investig. Dermatol. 2012, 5, 1–6. [Google Scholar]
- Pierre, S.; Liew, S.; Bernardin, A. Basics of dermal filler rheology. Dermatol. Surg. 2015, 41, S120–S126. [Google Scholar] [CrossRef]
- Salti, G.; Rauso, R. Facial rejuvenation with fillers: The dual plane technique. J. Cutan. Aesthetic Surg. 2015, 8, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Hee, C.K.; Shumate, G.T.; Narurkar, V.; Bernardin, A.; Messina, D.J. Rheological Properties and In Vivo Performance Characteristics of Soft Tissue Fillers. Dermatol. Surg. 2015, 41, S373–S381. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Federico, F.; Zerbinati, N.; De Cicco, D.; Nicoletti, G.F.; Tartaro, G. Hyaluronic Acid Injections to Correct Lips Deformity Following Surgical Removal of Permanent Implant. J. Craniofac. Surg. 2020, 31, e604–e606. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Califano, L.; Rugge, L.; Chirico, F.; Tartaro, G. Surgical Lip Remodeling After Injection of Permanent Filler. Aesthetic Surg. J. 2018, 39, 565–571. [Google Scholar] [CrossRef]
- Schweiger, E.S.; Riddle, C.C.; Tonkovic-Capin, V.; Aires, D.J. Successful treatment with injected hyaluronic acid in a patient with lip asymmetry after surgical correction of cleft lip. Dermatol. Surg. 2008, 34, 717–719. [Google Scholar] [PubMed]
- Stolic, D.; Jankovic, M.; Draskovic, M.; Georgiev, S.; Stolic, M. The Surgical Lips Deformity Corrected with Hyaluronic Fillers: A Case Report. Open Access Maced. J. Med. Sci. 2015, 3, 423–425. [Google Scholar] [CrossRef]
- Rahman, E.; Kandhari, R.; Goodman, G.J.; Signorini, M. Use of a hyaluronic acid soft-tissue filler to correct congenital and post-traumatic lip asymmetry. J. Cutan. Aesthetic Surg. 2017, 10, 153–156. [Google Scholar] [CrossRef]
- Rauso, R.; Colella, G.; Franco, R.; Ronchi, A.; Chirico, F. Ossified Carcinoma Ex Pleomorphic Adenoma in accessory lobe of parotid gland: Complexity in clinical, imaging and histologic diagnosis and minimally invasive surgery. Oral Oncol. 2019, 92, 95–98. [Google Scholar] [CrossRef]
- Tamplen, M.; Knott, P.D.; Fritz, M.A.; Seth, R. Controversies in Parotid Defect Reconstruction. Facial Plast. Surg. Clin. N. Am. 2016, 24, 235–243. [Google Scholar] [CrossRef]
- Cannady, S.B.; Seth, R.; Fritz, M.A.; Alam, D.S.; Wax, M.K. Total parotidectomy defect reconstruction using the buried free flap. Otolaryngol. Head Neck Surg. 2010, 143, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Clauser, L.; Polito, J.; Mandrioli, S.; Tieghi, R.; Denes, S.A.; Galiè, M. Structural Fat Grafting in Complex Reconstructive Surgery. J. Craniofac. Surg. 2008, 19, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Curry, J.M.; Fisher, K.W.; Heffelfinger, R.N.; Rosen, M.R.; Keane, W.M.; Pribitkin, E.A. Superficial Musculoaponeurotic System Elevation and Fat Graft Reconstruction After Superficial Parotidectomy. Laryngoscope 2008, 118, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Natghian, H.; Song, M.; Jayamohan, J.; Johnson, D.; Magdum, S.; Richards, P.; Wall, S. Long-Term Results in Isolated Metopic Synostosis: The Oxford Experience over 22 Years. Plast. Reconstr. Surg. 2018, 142, 509e–515e. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.R.; Kawamoto, H.K., Jr.; Burstein, F.; Peacock, W.J. Advancement-onlay: An improved technique of fronto-orbital remodeling in craniosynostosis. Childs Nerv. Syst. 1991, 7, 264–271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deyerle, W.M.; Crossland, S.; Sullivan, H. Methylmethacrylate: Uses and complications. AORN J. 1979, 29, 696–712. [Google Scholar] [CrossRef]
- Shapiro, S.A. Cranioplasty, Vertebral Body Replacement, and Spinal Fusion with Tobramycin-impregnated Methylmethacrylate. Neurosurgery 1991, 28, 789–791. [Google Scholar] [CrossRef]
- Blum, K.S.; Schneider, S.J.; Rosenthal, A.D. Methylmethacrylate cranioplasty in children: Long-term results. Pediatr. Neurosurg. 1997, 26, 33–35. [Google Scholar] [CrossRef]
- Replogle, R.E.; Lanzino, G.; Francel, P.; Henson, S.; Lin, K.; Jane, J.A. Acrylic Cranioplasty Using Miniplate Struts. Neurosurgery 1996, 39, 747–749. [Google Scholar] [CrossRef] [PubMed]
- Constantino, P.D.; Friedman, C.D.; Jones, K.; Chow, L.C.; Pelzer, H.J.; Sisson, G.A., Sr. Hydroxyapatite cement. I. Basic chemistry and histologic properties. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Friedman, C.D.; Costantino, P.D.; Takagi, S.; Chow, L.C. BoneSource? hydroxyapatite cement: A novel biomaterial for craniofacial skeletal tissue engineering and reconstruction. J. Biomed. Mater. Res. 1998, 43, 428–432. [Google Scholar] [CrossRef]
- Al-Nuaimi, Y.; Sherratt, M.J.; Griffiths, C.E.M. Skin health in older age. Maturitas 2014, 79, 256–264. [Google Scholar] [CrossRef]
- Greco, R.M.; Iocono, J.A.; Ehrlich, H.P. Hyaluronic acid stimulates human fibroblast proliferation within a collagen matrix. J. Cell. Physiol. 1998, 177, 465–473. [Google Scholar] [CrossRef]
- Mast, B.A.; Diegelmann, R.F.; Krummel, T.M.; Cohen, I.K. Hyaluronic Acid Modulates Proliferation, Collagen and Protein Synthesis of Cultured Fetal Fibroblasts. Matrix 1993, 13, 441–446. [Google Scholar] [CrossRef]
- Croce, M.; Dyne, K.; Boraldi, F.; Quaglino, D.; Cetta, G.; Tiozzo, R.; Ronchetti, I.P. Hyaluronan affects protein and collagen synthesis by in vitro human skin fibroblasts. Tissue Cell 2001, 33, 326–331. [Google Scholar] [CrossRef]
- Huang-Lee, L.L.; Nimni, M.E. Fibroblast contraction of collagen matrices with and without covalently bound hyaluronan. J. Biomater. Sci. Polym. Ed. 1994, 5, 99–109. [Google Scholar] [CrossRef]
- Iocono, J.A.; Krummel, T.M.; Keefer, K.A.; Allison, G.M.; Paul Ehrlich, H. Repeated additions of hyaluronan alters granulation tissue deposition in sponge implants in mice. Wound Repair Regen. 1998, 6, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Paliwal, S.; Fagien, S.; Sun, X.; Holt, T.; Kim, T.; Hee, C.K.; Van Epps, D.; Messina, D.J. Skin extracellular matrix stimulation following injection of a hya-luronic acid-based dermal filler in a rat model. Plast. Reconstr. Surg. 2014, 134, 1224–1233. [Google Scholar] [CrossRef]
- Quan, T.; Wang, F.; Shao, Y.; Rittié, L.; Xia, W.; Orringer, J.S.; Voorhees, J.J.; Fisher, G.J. Enhancing Structural Support of the Dermal Microenvironment Activates Fibroblasts, Endothelial Cells, and Keratinocytes in Aged Human Skin In Vivo. J. Investig. Dermatol. 2013, 133, 658–667. [Google Scholar] [CrossRef]
- Zerbinati, N.; D’Este, E.; Parodi, P.C.; Calligaro, A. Microscopic and ultrastructural evidences in human skin following calcium hydroxylapatite filler treatment. Arch. Dermatol. Res. 2017, 309, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Maytin, E.V. Hyaluronan: More than just a wrinkle filler. Glycobiology 2016, 26, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Nicoletti, G.F.; Sesenna, E.; Faro, C.L.; Chirico, F.; Fragola, R.; Giudice, G.L.; Tartaro, G. Superficial Temporal Artery Perforator Flap: Indications, Surgical Outcomes, and Donor Site Morbidity. Dent. J. 2020, 8, 117. [Google Scholar] [CrossRef] [PubMed]
- Ohlsén, L.; Skoog, T.; Sohn, S.A. The pathogenesis of cauliflower ear. An experimental study in rabbits. Scand. J. Plast. Reconstr. Surg. 1975, 9, 34–39. [Google Scholar] [CrossRef]
- Benecke, J.E., Jr.; Gadre, A.K.; Linthicum, F.H., Jr. Chondrogenic potential of tragal perichondrium: A cause of hearing loss following stapedectomy. Laryngoscope 1990, 100, 1292–1293. [Google Scholar] [CrossRef]
- Abrahamsson, P.; Isaksson, S.; Gordh, M.; Andersson, G. Periosteal expansion of rabbit mandible with an osmotic self-inflatable expander. Scand. J. Plast. Reconstr. Surg. Hand Surg. 2009, 43, 121–125. [Google Scholar] [CrossRef]
- Sgaramella, N.; Wilms, T.; Boldrup, L.; Loljung, L.; Gu, X.; Coates, P.; Califano, L.; Muzio, L.L.; Spaak, L.N.; Franco, R.; et al. Ethnicity based variation in expression of E-cadherin in patients with squamous cell carcinoma of the oral tongue. Oncol. Lett. 2018, 16, 6603–6607. [Google Scholar] [CrossRef]
- Hanasono, M.M.; Silva, A.; Yu, P.; Skoracki, R.J. A Comprehensive Algorithm for Oncologic Maxillary Reconstruction. Plast. Reconstr. Surg. 2013, 131, 47–60. [Google Scholar] [CrossRef]
- Rauso, R.; Colella, G.; Chirico, F.; Fragola, R.; Nicoletti, G.F.; Faro, C.L.; Tartaro, G. Alar facial sulcus and nasal ala reconstruction: A challenging anatomical area. Minerva Dent. Oral Sci. 2021, 70, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, P.G.; Santamaria, E. A Classification System and Algorithm for Reconstruction of Maxillectomy and Midfacial Defects. Plast. Reconstr. Surg. 2000, 105, 2331–2346. [Google Scholar] [CrossRef] [PubMed]
- Pipkorn, P.; Rosenquist, K.; Zenga, J. Functional considerations in oral cavity reconstruction. Curr. Opin. Otolaryngol. Head Neck Surg. 2018, 26, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Jaquet, Y.; Enepekides, D.J.; Torgerson, C.; Higgins, K.M. Radial Forearm Free Flap Donor Site MorbidityUlnar-Based Transposition Flap vs Split-Thickness Skin Graft. Arch. Otolaryngol. Head Neck Surg. 2012, 138, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Colella, G.; Rugge, L.; Chirico, F.; Tartaro, G. Alternative suture technique for anastomosis of veins with different diameters. Br. J. Oral Maxillofac. Surg. 2019, 57, 682–684. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Chirico, F.; Federico, F.; Nicoletti, G.F.; Colella, G.; Fragola, R.; Pafundi, P.C.; Tartaro, G. Maxillo-facial reconstruction following cancer ablation during COVID-19 pandemic in southern Italy. Oral Oncol. 2020, 115, 105114. [Google Scholar] [CrossRef] [PubMed]
- Massarelli, O.; Vaira, L.; Gobbi, R.; Biglio, A.; Orabona, G.D.; De Riu, G. Soft palate functional reconstruction with buccinator myomucosal island flaps. Int. J. Oral Maxillofac. Surg. 2018, 47, 316–323. [Google Scholar] [CrossRef]







| Subject (Case) | Sex | Age | Anatomical Area | Anamnesis | Number of Sessions | HA Amount per Session |
|---|---|---|---|---|---|---|
| 1 | M | 26 | Right Malar area | Facial trauma (zygomatic arch fracture) | 1 | 2 mL |
| 2 | F | 54 | Lip | Labial incompetence (permanent implant removal surgery sequelae) | 1 | 3 mL |
| 3 | M | 34 | Temporoparietal area | Facial malformation surgery sequelae | 5 | 4 mL |
| 4 | F | 59 | Left parotid area | Nerve-sparing total parotidectomy sequelae | 1 | 4.5 mL |
| 5 | M | 74 | Left forehead region | Reconstructive surgery sequelae (STAP flap) | 5 | 0.2 mL |
| 6 | M | 55 | Right cheek region | Reconstructive surgery sequelae (t-FAMMIF) | 2 | 1.8 mL |
| Impending Necrosis | Visual Loss | Blindness | Bruising | Swelling | |
|---|---|---|---|---|---|
| Number of Patients | 0 (0%) | 0 (0%) | 0 (0%) | 1 (17%) | 1 (17%) |
| VAS Score | 0–74 | 75–79 | 80–89 | 90–99 | 100 |
|---|---|---|---|---|---|
| Number of Patients | 0 | 0 | 2 (33%) | 0 | 4 (67%) |
| VAS Score | 0–74 | 75–79 | 80–89 | 90–99 | 100 |
|---|---|---|---|---|---|
| Number of Patients | 0 | 0 | 0 | 2 (33%) | 4 (67%) |
| VAS Score | 0–74 | 75–79 | 80–89 | 90–99 | 100 |
|---|---|---|---|---|---|
| Number of Patients | 0 | 0 | 0 | 0 | 6 (100%) |
| VAS Score | 0–74 | 75–79 | 80–89 | 90–99 | 100 |
|---|---|---|---|---|---|
| Number of Patients | 0 | 0 | 0 | 0 | 6 (100%) |
| VAS Score | 0–74 | 75–79 | 80–89 | 90–99 | 100 |
|---|---|---|---|---|---|
| Number of Patients | 0 | 0 | 0 | 0 | 6 (100%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chirico, F.; Colella, G.; Cortese, A.; Bove, P.; Fragola, R.; Rugge, L.; Audino, G.; Sgaramella, N.; Tartaro, G. Non-Surgical Touch-Up with Hyaluronic Acid Fillers Following Facial Reconstructive Surgery. Appl. Sci. 2021, 11, 7507. https://doi.org/10.3390/app11167507
Chirico F, Colella G, Cortese A, Bove P, Fragola R, Rugge L, Audino G, Sgaramella N, Tartaro G. Non-Surgical Touch-Up with Hyaluronic Acid Fillers Following Facial Reconstructive Surgery. Applied Sciences. 2021; 11(16):7507. https://doi.org/10.3390/app11167507
Chicago/Turabian StyleChirico, Fabrizio, Giuseppe Colella, Antonio Cortese, Pierfrancesco Bove, Romolo Fragola, Luigi Rugge, Giovanni Audino, Nicola Sgaramella, and Gianpaolo Tartaro. 2021. "Non-Surgical Touch-Up with Hyaluronic Acid Fillers Following Facial Reconstructive Surgery" Applied Sciences 11, no. 16: 7507. https://doi.org/10.3390/app11167507
APA StyleChirico, F., Colella, G., Cortese, A., Bove, P., Fragola, R., Rugge, L., Audino, G., Sgaramella, N., & Tartaro, G. (2021). Non-Surgical Touch-Up with Hyaluronic Acid Fillers Following Facial Reconstructive Surgery. Applied Sciences, 11(16), 7507. https://doi.org/10.3390/app11167507







