Potential Impacts of Climate Change on Shrimps Distribution of Commercial Importance in the Gulf of California
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Habitat Suitability Models
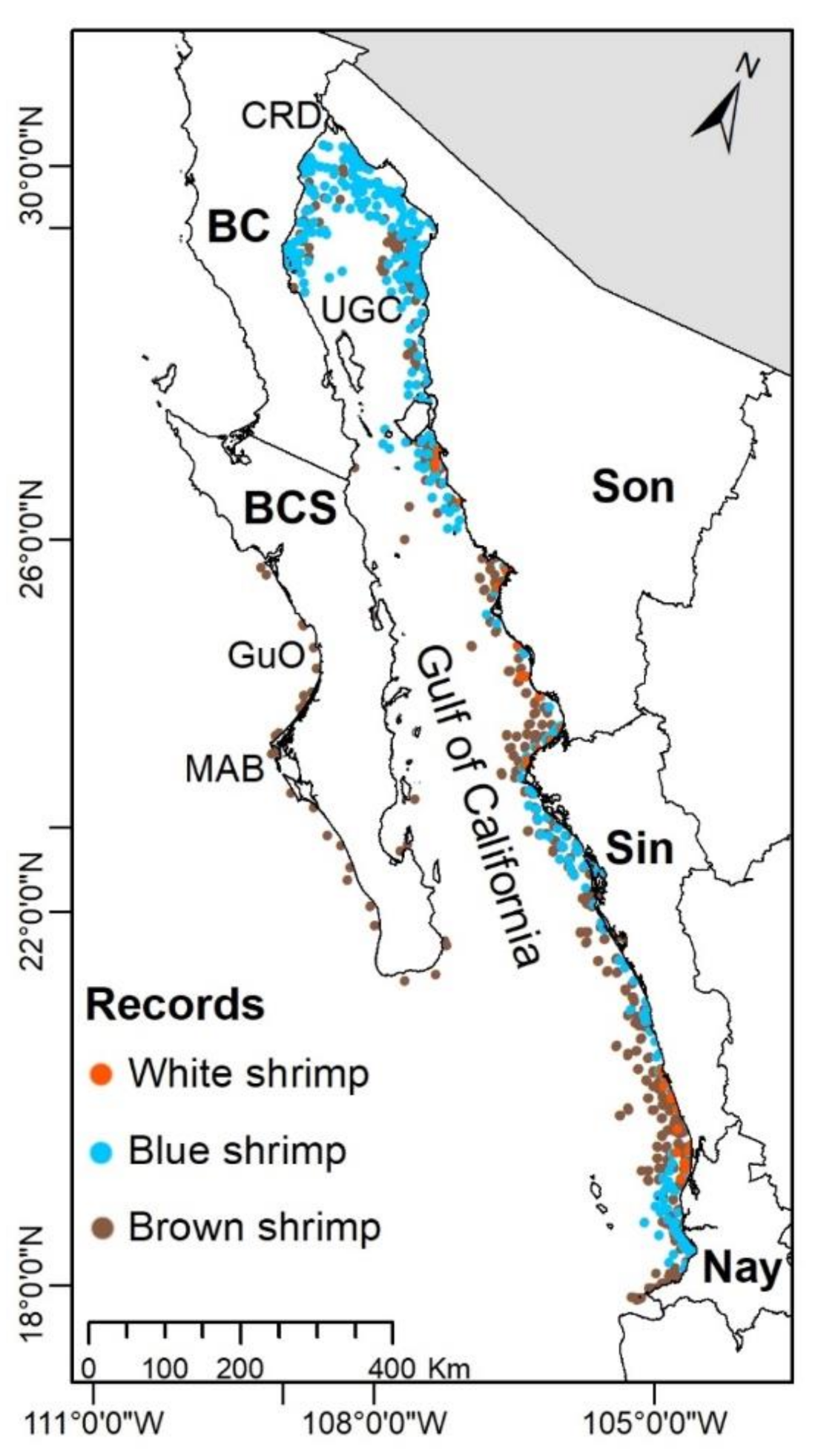
2.2.1. Occurrence Records of Shrimps
2.2.2. Environmental Database
2.2.3. Current Shrimps’ Distribution Model
2.3. Climate Change Scenarios
3. Results
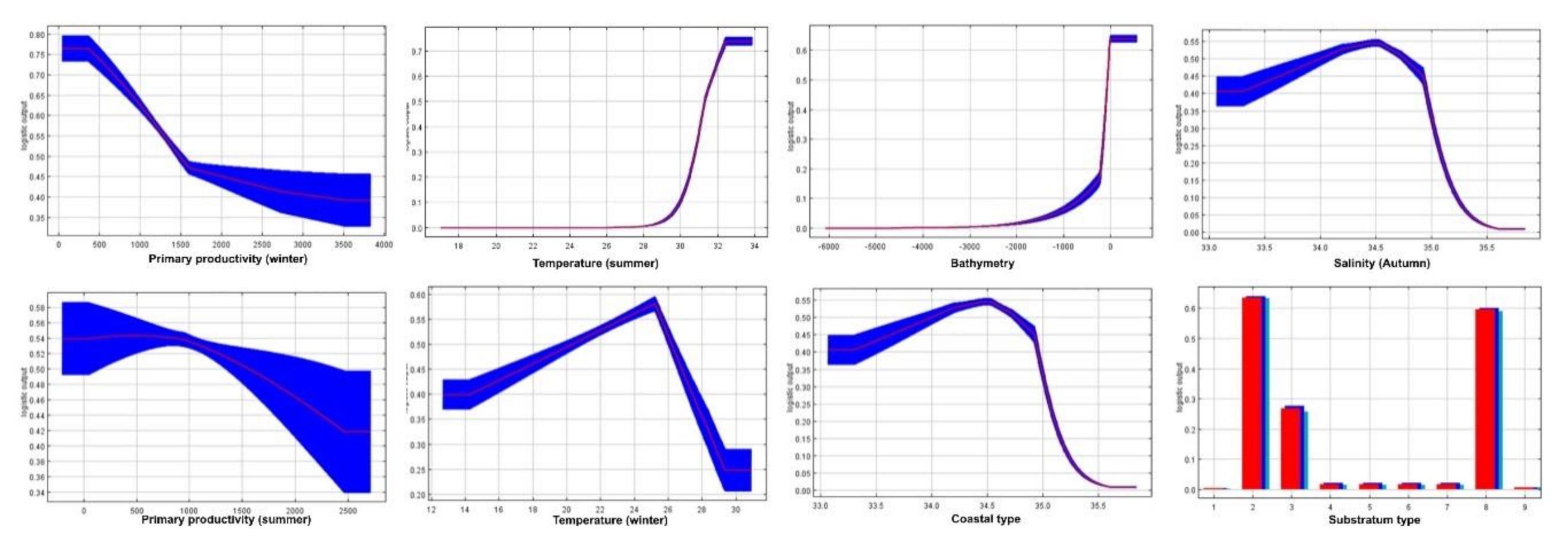
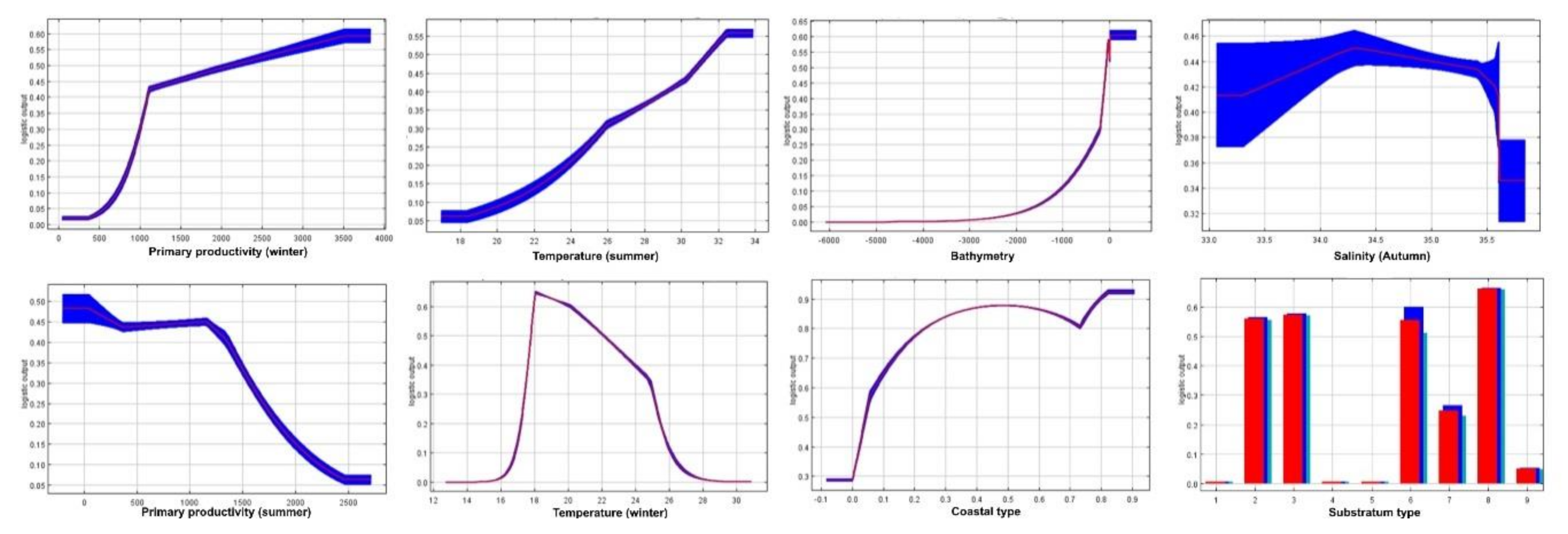
3.1. Environmental Contribution on Shrimps’ Habitat Suitability Models
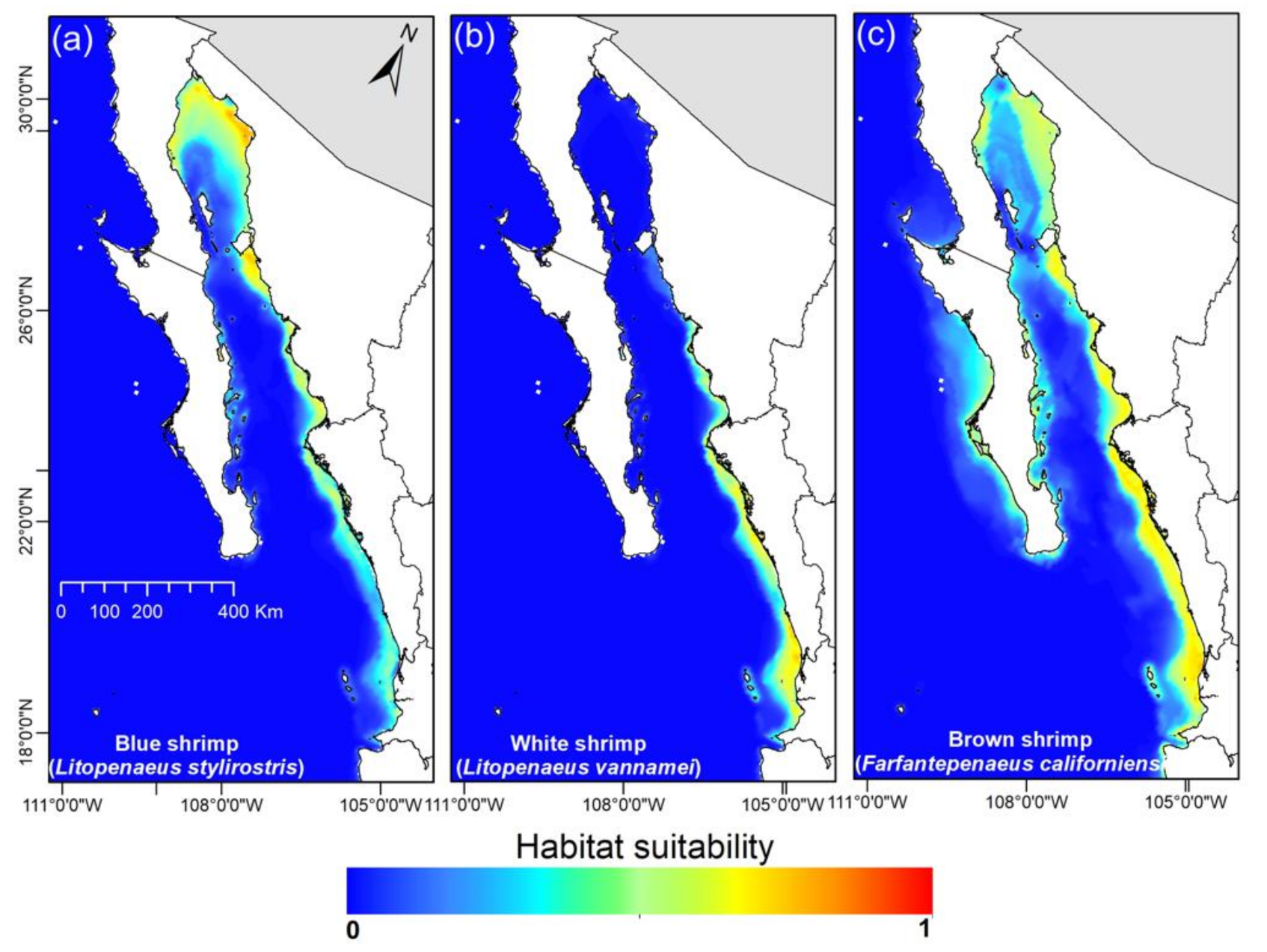
3.2. Current Shrimps’ Distribution
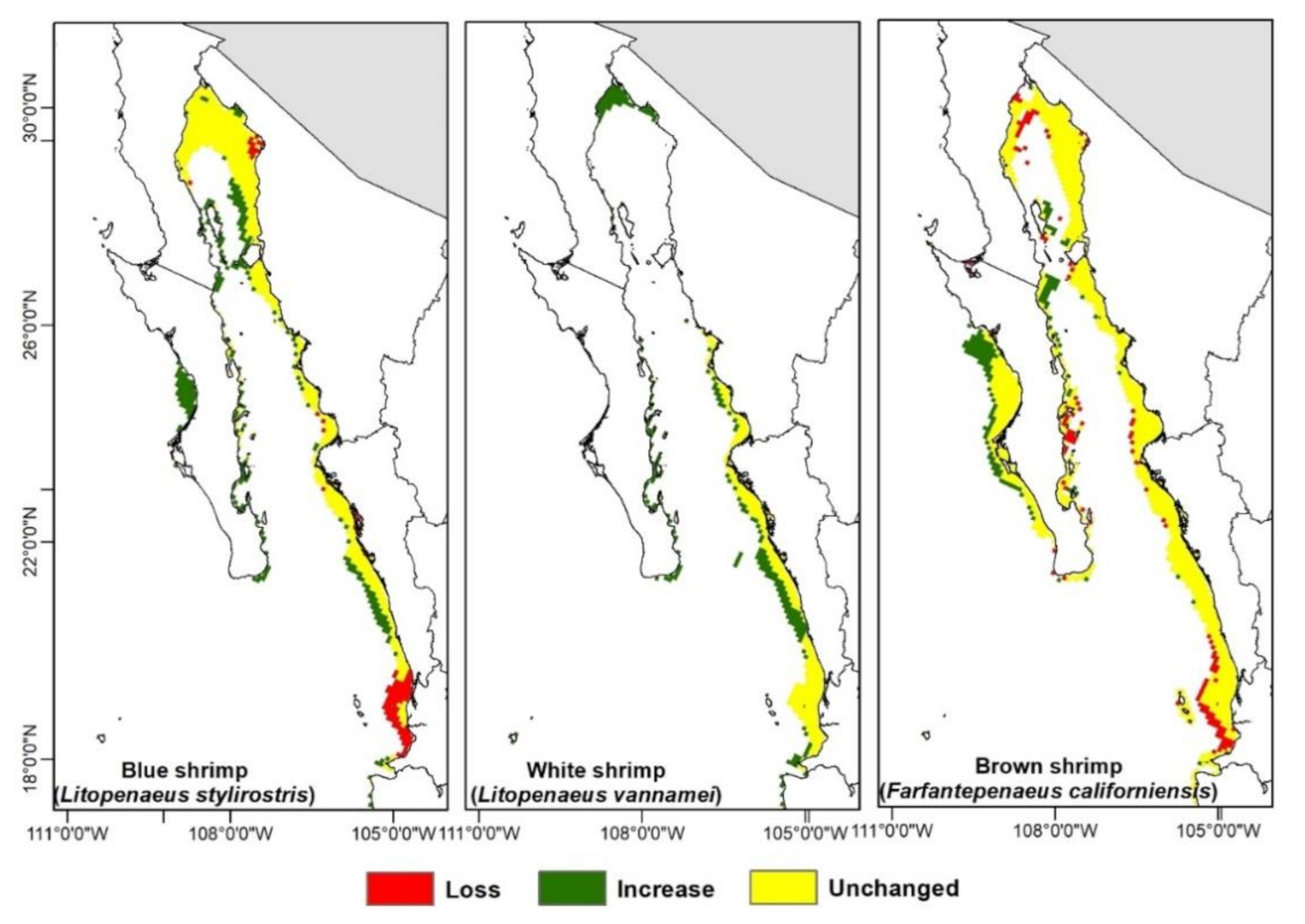
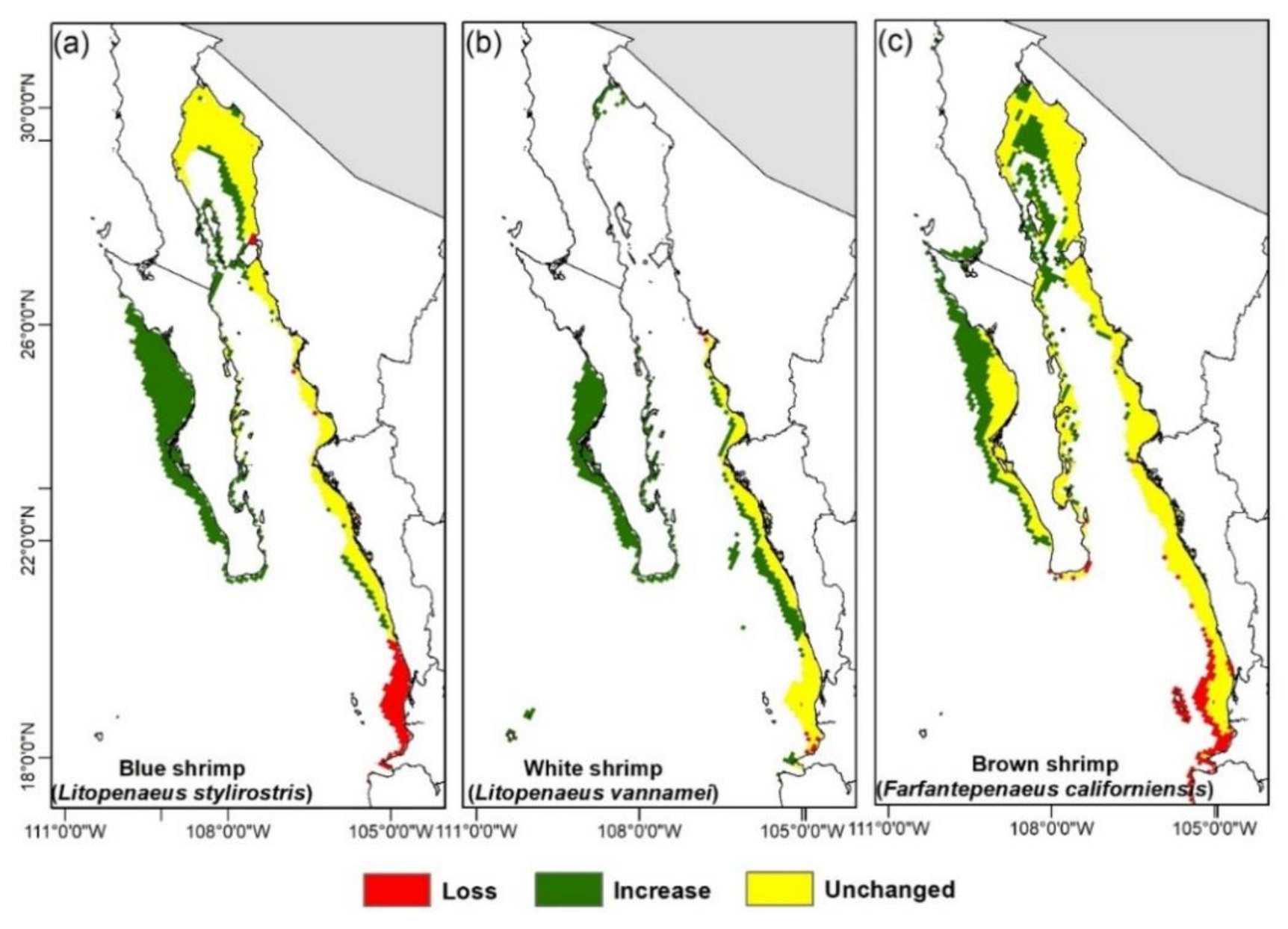
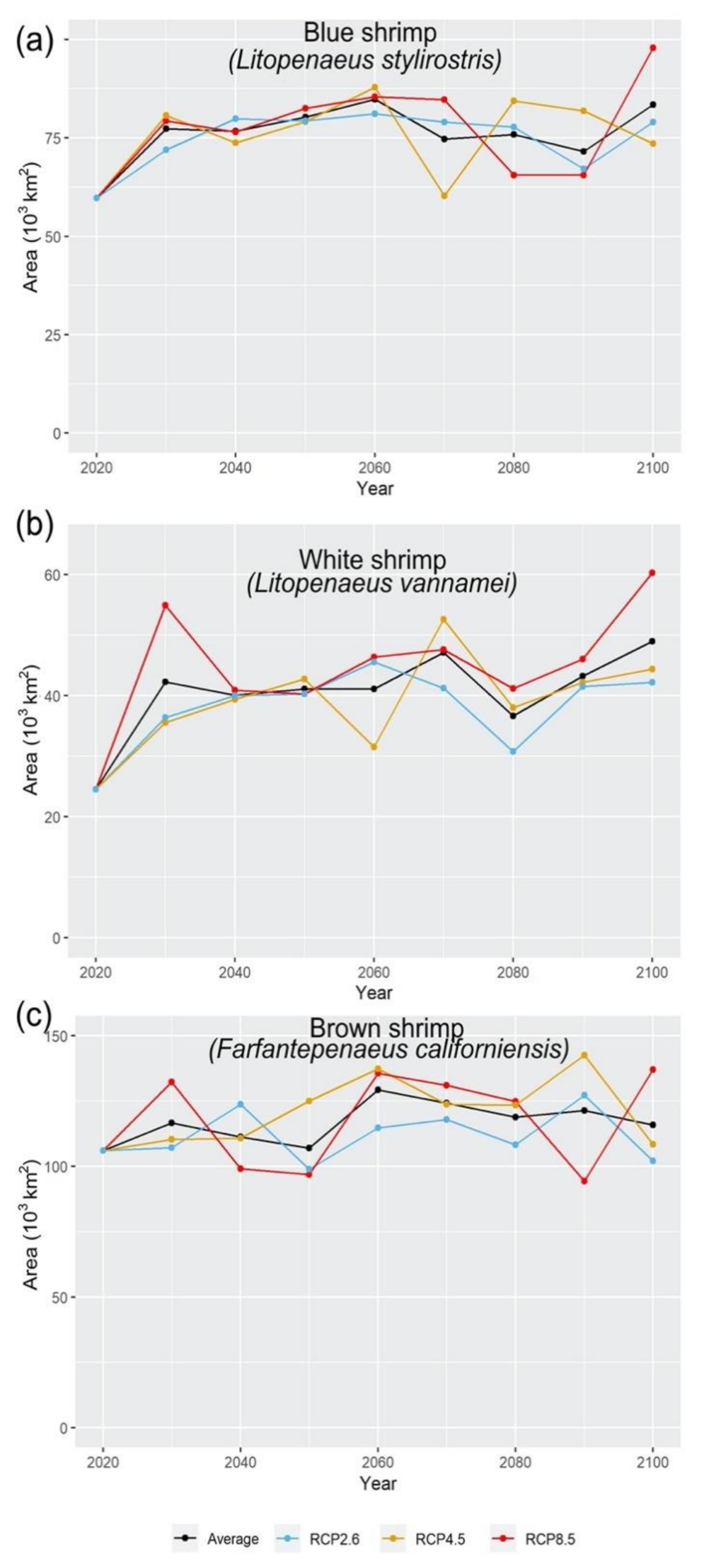
3.3. Climate Change Effects on Shrimps’ Distribution
4. Discussion
4.1. Uncertainties and Assumptions
4.2. Model Accuracy and Current Distribution
4.3. Environmental Drivers of Shrimps’ Distribution
4.4. Range Shift in Response to Climate Change
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Layer | NPP Winter | NPP Spring | NPP Summer | NPP Autumn | SST Winter | SST Spring | SST Summer | SST Autumn | SSS Winter | SSS Spring | SSS Summer | SSS Autumn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NPP winter | 1.00 | |||||||||||
| NPP spring | 0.76 | 1.00 | ||||||||||
| NPP summer | 0.67 | 0.94 | 1.00 | |||||||||
| NPP autumn | 0.91 | 0.78 | 0.79 | 1.00 | ||||||||
| SST winter | 0.33 | 0.03 | −0.07 | 0.17 | 1.00 | |||||||
| SST spring | 0.44 | 0.18 | 0.06 | 0.28 | 0.95 | 1.00 | ||||||
| SST summer | 0.55 | 0.39 | 0.25 | 0.38 | 0.85 | 0.95 | 1.00 | |||||
| SST autumn | 0.45 | 0.20 | 0.07 | 0.27 | 0.97 | 0.97 | 0.95 | 1.00 | ||||
| SSS winter | 0.27 | 0.33 | 0.23 | 0.17 | 0.48 | 0.61 | 0.76 | 0.65 | 1.00 | |||
| SSS spring | 0.38 | 0.35 | 0.24 | 0.25 | 0.65 | 0.78 | 0.88 | 0.79 | 0.96 | 1.00 | ||
| SSS summer | 0.27 | 0.31 | 0.21 | 0.14 | 0.55 | 0.67 | 0.79 | 0.70 | 0.97 | 0.97 | 1.00 | |
| SSS autumn | 0.20 | 0.31 | 0.22 | 0.09 | 0.39 | 0.52 | 0.68 | 0.56 | 0.97 | 0.91 | 0.97 | 1.00 |


References
- Instituto Nacional de Pesca. Evaluación Biológica de las Poblaciones de Camarón Durante la Veda 2014 en el Litoral del Pacífico Mexicano; INAPESCA: Mazatlán, Mexico, 2014; pp. 1–96.
- Aranceta-Garza, F.; Arreguín-Sánchez, F.; Seijo, J.C.; Ponce-Díaz, G.; Lluch-Cota, D.; del Monte-Luna, P. Determination of catchability-at-age for the Mexican Pacific shrimp fishery in the southern Gulf of California. Reg. Stud. Mar. Sci. 2020, 33, 100967. [Google Scholar] [CrossRef]
- Instituto Nacional de Pesca. La Pesquería de Camarón del Pacifico; INAPESCA: La Paz, Mexico, 2012; pp. 1–50.
- López-Martínez, J.; Hernández-Vásquez, S.; Herrera-Valdivia, E.; Rodríguez-Romero, J.; Chávez, E.A. Influencia Ambiental en la Pesquería de Camarón. In Variabilidad Ambiental y Pesquería en Mexico; Comision Nacional de Acuacultura y Pesca: Zacatepec, Morelos, Mexico, 2008; pp. 111–123. [Google Scholar]
- García-Borbón, J.A.; Balart, E.F.; Gallo-Ramírez, J.J.; Loreto-Campos, P.A. La Pesquería de Camarón. In Estudio del Potencial Pesquero y Acuícola de Baja California Sur; Casas-Valdéz, M., Ponce-Diaz, G., Eds.; INAPESCA: La Paz, Mexico, 1996; Volume 1, pp. 187–206. [Google Scholar]
- Instituto Nacional de Pesca. Evaluación y Manejo de la Pesquería de Camarón del Pacifico Mexicano; INAPESCA: La Paz, Mexico, 2016; pp. 1–42.
- López-Martínez, J.; Morales-Bojorques, E.; Paredes-Mallon, F.; Lluch-Belda, D.; Cervantes, C. La Pesquería de Camarón de Altamar en Sonora; Centros de Actividad Biológica del Pacífico Mexicano: La Paz, Mexico, 2001; pp. 301–312. [Google Scholar]
- Rodríguez de la Cruz, C.; Chávez, E. La pesquería de camarón en altamar. Pacífico de Mexico. Memorias 1994, 35, 11. [Google Scholar]
- García, S.; Le Reste, L. Ciclos Vitales, Dinámica, Explotación y Ordenación de las Poblaciones de Camarones Peneidos Costeros; FAO Doc. Téc. Pesca; Food and Agricultural Organization of United Nations: Rome, Italy, 1987; 180p. [Google Scholar]
- Mendo, J.; Tam, J. Multiple environmental states affecting penaeid shrimp production in Peru. Naga ICLARM Q. 1993, 16, 44–47. [Google Scholar]
- Sheridan, P. Forecasting the fishery for pink shrimp, Penaeus duorarum, on the Tortugas Grounds, Florida. Fish. Bull. 1996, 94, 743–755. [Google Scholar]
- Criales, M.M.; Lee, T.N. Larval distribution and transport of penaeoid shrimps during the presence of the Tortugas Gyre in May–June 1991. Oceanogr. Lit. Rev. 1996, 6, 471–482. [Google Scholar]
- García, S. Tropical Penaeid Prawns. In Fish Population Dynamics, 2nd ed.; Gulland, J.A., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 1988; pp. 219–249. [Google Scholar]
- Hannah, R.W. Influence of environmental variation and spawning stock levels on recruitment of ocean shrimp (Pandalus jordani). Can. J. Fish. Aquat. Sci. 1993, 50, 612–622. [Google Scholar] [CrossRef]
- Pachauri, R.K.; Allen, M.R.; Barros, V.R.; Broome, J.; Cramer, W.; Christ, R.; Dubash, N.K. Climate Change 2014: Synthesis Report—Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef]
- Petatán-Ramírez, D.; Ojeda-Ruiz, M.Á.; Sánchez-Velasco, L.; Rivas, D.; Reyes-Bonilla, H.; Cruz-Piñón, G.; Salvadeo, C. Potential changes in the distribution of suitable habitat for Pacific sardine (Sardinops sagax) under climate change scenarios. Deep Sea Res. Part. II Top. Stud. Oceanogr. 2019, 169, 104632. [Google Scholar] [CrossRef]
- Rahel, F.J.; Olden, J.D. Assessing the effects of climate change on aquatic invasive species. Conserv. Biol. 2008, 22, 521–533. [Google Scholar] [CrossRef]
- Magaña-Rueda, V.O.; García, C.G. Vulnerabilidad y adaptación regional ante el cambio climático y sus impactos ambientales, sociales y económicos. Gaceta Ecológica 2002, 65, 7–23. [Google Scholar]
- López-Martínez, J.; Herrera-Valdivia, E.; Hernández-Saavedra, N.; Serviere-Zaragoza, E.; Rodríguez-Romero, J.; Rábago-Quiroz, C.H.; Pedrín-Aviles, S. Efectos de la Pesca de Arrastre del Camarón en el Golfo de California: Síntesis de las Investigaciones Desarrolladas por el Centro de Investigaciones Biológicas del Noroeste SC. In Efectos de la Pesca de Arrastre en el Golfo de California; López-Martínez, J., Morales-Bojórquez, E., Eds.; Cibnor: La Paz, Mexico, 2012; pp. 15–26. [Google Scholar]
- Sosa-Rodríguez, F.S. Política del cambio climático en Mexico: Avances, obstáculos y retos. Revista Internacional de Estadística y Geografía 2015, 6, 4–23. [Google Scholar]
- Morales-Bojórquez, E.; Madrid-Vera, J.; Díaz-Uribe, J.G.; Aguirre-Villaseñor, H.; Liedo-Galindo, A.; Chávez-Herrera, D.; Hernández-López, A. Distribución y Abundancia de Camarón Café (Fanfantepenaeus californiensis) en el Norte de Sinaloa, Mexico. In Efectos de la Pesca de Arrastre en el Golfo de California; López-Martínez, J., Morales-Bojórquez, E., Eds.; Cibnor: La Paz, Mexico, 2012; pp. 385–398. [Google Scholar]
- Mateo, R.G.; Felicísimo, Á.M.; Muñoz, J. Modelos de distribución de especies y su potencialidad como recurso educativo interdisciplinar. Reduca 2012, 5, 137–153. [Google Scholar]
- Cuspilici, A.; Monforte, P.; Ragusa, M.A. Study of Saharan dust influence on PM10 measures in Sicily from 2013 to 2015. Ecol. Indic. 2017, 76, 297–303. [Google Scholar] [CrossRef]
- López-Martínez, J.; Rábago-Quiroz, C.H.; Herrera-Valdivia, E.; Morales-Azpeitia, R.; Padilla-Serrato, J.G. Distribution and abundance of Sicyonia shrimp in the Gulf of California and western coast of the Baja California Peninsula, Mexico. Reg. Stud. Mar. Sci. 2020, 38, 101381. [Google Scholar] [CrossRef]
- Wilson, K.A.; Westphal, M.I.; Possingham, H.P.; Elith, J. Sensitivity of conservation planning to different approaches to using predicted species distribution data. Biol. Conserv. 2005, 122, 99–112. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Calmus, T.; Burquez, A.; Martínez-Yrizar, A. El Golfo de California: Un océano joven, región megadiversa, vínculo entre tectónica y ecología. Ciencia UANL 2017, 85, 59–64. [Google Scholar]
- Soto-Mardones, L.; Marinone, S.; Parés-Sierra, A. Variabilidad espaciotemporal de la temperatura superficial del mar en el Golfo de California. Ciencias Marinas 1999, 25, 1–30. [Google Scholar] [CrossRef]
- Álvarez-Borrego, S.; Lara-Lara, J.R. The Physical Environment and Productivity of the Gulf of California. In The Gulf and Peninsular Province of the Californias; Dauphin, J.P., Simoneit, B., Eds.; American Association of Petroleum Geologists: Tulsa, OK, USA, 1991; pp. 555–567. [Google Scholar]
- Veloz, S.D. Spatially autocorrelated sampling falsely inflates measures of accuracy for presence-only niche models. J. Biogeogr. 2009, 36, 2290–2299. [Google Scholar] [CrossRef]
- Radosavljevic, A.; Anderson, R.P. Making better Maxent models of species distributions: Complexity, overfitting and evaluation. J. Biogeogr. 2014, 41, 629–643. [Google Scholar] [CrossRef]
- Westberry, T.; Behrenfeld, M.J.; Siegel, D.A.; Boss, E. Carbon-based primary productivity modeling with vertically resolved photoacclimation. Global Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Chassignet, E.P.; Hurlburt, H.E.; Smedstad, O.M.; Halliwell, G.R.; Hogan, P.J.; Wallcraft, A.J.; Bleck, R. The HYCOM (hybrid coordinate ocean model) data assimilative system. J. Mar. Syst. 2007, 65, 60–83. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing. 2021. Available online: https://www.r-project.org (accessed on 6 February 2021).
- Gilani, H.; Goheer, M.A.; Ahmad, H.; Hussain, K. Under predicted climate change: Distribution and ecological niche modelling of six native tree species in Gilgit-Baltistan, Pakistan. Ecol. Indic. 2020, 111, 106049. [Google Scholar] [CrossRef]
- Heikkinen, R.K.; Luoto, M.; Araújo, M.B.; Virkkala, R.; Thuiller, W.; Sykes, M.T. Methods and uncertainties in bioclimatic envelope modelling under climate change. Prog. Phys. Geogr. 2006, 30, 751–777. [Google Scholar] [CrossRef]
- Euán-Avila, J.I.; Cuevas-Jiménez, A. Regionalizing Coastal Zones with Geospatial Tools for Integrated Coastal Zone Management. In Coastal and Marine Geospatial Technologies; Green, D.R., Ed.; Springer Press: Dordrecht, The Netherlands, 2010; pp. 139–151. [Google Scholar] [CrossRef]
- Petatán-Ramírez, D.; Hernández, L.; Becerril-García, E.E.; Berúmen-Solórzano, P.; Auliz-Ortiz, D.; Reyes-Bonilla, H. Potential distribution of the tiger shrimp Penaeus monodon (Decapoda: Penaeidae), an invasive species in the Atlantic Ocean. Rev. Biol. Trop. 2020, 68, 156–166. [Google Scholar] [CrossRef]
- Instituto Nacional de Pesca. Atlas Pesquero de Mexico; INAPESCA: Ciudad de Mexico, Mexico, 1994.
- Intergovernmental Panel on Climate Change (IPCC). Resumen para Responsables de Políticas. In Cambio Climático 2013: Bases Físicas; Contribución del Cambio Climático; IPCC: Geneva, Switzerland, 2013. [Google Scholar]
- Gudmundsson, L.; Bremnes, J.B.; Haugen, J.E.; Engen-Skaugen, T. Downscaling RCM precipitation to the station scale using statistical transformations—A comparison of methods. Hydrol. Earth Syst. Sci. 2012, 16, 3383–3390. [Google Scholar] [CrossRef]
- Melo-Merino, S.M.; Reyes-Bonilla, H.; Lira-Noriega, A. Ecological niche models and species distribution models in marine environments: A literature review and spatial analysis of evidence. Ecol. Model. 2020, 415, 108837. [Google Scholar] [CrossRef]
- Mann, K.H.; Lazier, J.R. Dynamics of Marine Ecosystems: Biological-Physical Interactions in the Oceans; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Pearson, R.G.; Raxworthy, C.J.; Nakamura, M.; Peterson, A.T. Predicting species distributions from small numbers of occurrence records: A test case using cryptic geckos in Madagascar. J. Biogeogr. 2007, 34, 102–117. [Google Scholar] [CrossRef]
- Basher, Z.; Bowden, D.A.; Costello, M.J. Diversity and distribution of deep-sea shrimps in the Ross Sea region of Antarctica. PLoS ONE 2014, 9, e103195. [Google Scholar] [CrossRef]
- Peterson, A.; Papeş, M.; Eaton, M. Transferability and model evaluation in ecological niche modeling: A comparison of GARP and Maxent. Ecography 2007, 30, 550–560. [Google Scholar] [CrossRef]
- Elith, J. Quantitative Methods for Modeling Species Habitat: Comparative Performance and an Application to Australian plants. In Quantitative Methods for Conservation Biology; Ferson, S., Burgman, M., Eds.; Springer: New York, NY, USA, 2000; pp. 39–58. [Google Scholar] [CrossRef]
- García-Juárez, A.R.; Rodríguez-Domínguez, G.; Lluch-Cota, D.B. La cuota de captura de camarón azul (Litopenaeus stylirostris) como instrumento de gestión en el Alto Golfo de California. Ciencias Marinas 2009, 35, 297–306. [Google Scholar] [CrossRef]
- Gell, F.R.; Roberts, C.M. Benefits beyond boundaries: The fishery effects of marine reserves. Trends Ecol. Evol. 2003, 18, 448–455. [Google Scholar] [CrossRef]
- Hilborn, R.; Stokes, K.; Maguire, J.J.; Smith, T.; Botsford, L.W.; Mangel, M.; Cochrane, K.L. When can marine reserves improve fisheries management? Ocean. Coast. Manag. 2004, 47, 197–205. [Google Scholar] [CrossRef]
- Balmori-Ramírez, A.; Cervantes-Higuera, H. La Fauna Acompañante de la Pesca Ribereña de Camarón en Sonora 2019. Available online: https://fisheryprogress.org/sites/default/files/documents_actions/Gillnet%20bycatch%20evaluation%202018-19.pdf (accessed on 6 February 2021).
- Berlanga-Robles, C.; Ruiz-Luna, A. Los Sistemas Acuáticos Costeros de Sinaloa. In Atlas de los Ecosistemas de Sinaloa; Cifuentes-Lemus, J.L., Gaxiola-López, J., Eds.; El Colegio de Sinaloa: Culiacán, Mexico, 2003; pp. 197–214. [Google Scholar]
- Basher, Z.; Costello, M.J. The past, present and future distribution of a deep-sea shrimp in the Southern Ocean. PeerJ 2016, 4, e1713. [Google Scholar] [CrossRef]
- Dambach, J.; Thatje, S.; Roedder, D.; Basher, Z.; Raupach, M.J. Effects of Late-Cenozoic glaciation on habitat availability in Antarctic benthic shrimps (Crustacea: Decapoda: Caridea). PLoS ONE 2012, 7, e46283. [Google Scholar] [CrossRef]
- Vargas-Albores, F.; Hinojosa-Baltazar, P.; Portillo-Clark, G.; Magallon-Barajas, F. Influence of temperature and salinity on the yellowleg shrimp, Penaeus californieinsis Holmes, 1900, prophenoloxidase system. Aquac. Res. 1998, 29, 549–553. [Google Scholar] [CrossRef]
- Lluch, B.D. La Pesquería del Camarón del Pacífico (Diagnosis Monográfica de los Conocimientos Existentes); Centro de Investigaciones Bilologicas del Noroeste; S.C. Centro Interdisciplinario de Ciencias Marinas: La Paz, Mexico; Consejo Nacional deCiencia y Tecnologia: Cocoyoc, Mexico, 1982. [Google Scholar]
- Hernández-Covarrubias, V.; Muñoz-Rubí, H.A.; Madrid-Vera, J.; Chávez-Herrera, D. Fecundidad del camarón blanco Litopenaeus vannamei de la plataforma continental de Sinaloa, Mexico. Ciencia Pesquera 2012, 20, 17–21. [Google Scholar]
- Ponce-Palafox, J.; Martinez-Palacios, C.A.; Ross, L.G. The effects of salinity and temperature on the growth and survival rates of juvenile white shrimp, Penaeus vannamei, Boone, 1931. Aquaculture 1997, 157, 107–115. [Google Scholar] [CrossRef]
- Magallón, B.F.; Portillo, G.; Campos, R.; Porchas, M.A.; Naranjo, J.; Muhlia, A. Penaeus californiensis as a cold tolerant species in Baja California Sur, Mexico. In Book of Abstracts: World Aquaculture ‘94 New Orleans LA, USA, 14–18 January 1994; World Aquaculture Society: Sorrento, LA, USA, 1994; p. 84. [Google Scholar]
- Villarreal, H.; Hernandez-Llamas, A.; Hewitt, R. Effect of salinity on growth, survival and oxygen consumption of juvenile brown shrimp, Farfantepenaeus californiensis (Holmes 1900). Aquac. Res. 2003, 34, 187–193. [Google Scholar] [CrossRef]
- Romero-Sedano, J.C.; Aragón-Noriega, E.A.; Manzano-Sarabia, M.M.; Salinas-Zavala, C.A.; García-Juárez, A.R. Periodo reproductivo del camarón café Farfantepenaeus californiensis (Holmes, 1900) en la laguna costera de Agiabampo, Sonora/Sinaloa, Mexico. Ciencias Marinas 2004, 30, 465–475. [Google Scholar] [CrossRef]
- Aragón-Noriega, E.A.; Alcántara-Razo, E. Influence of sea surface temperature on reproductive period and size at maturity of brown shrimp (Farfantepenaeus californiensis) in the Gulf of California. Mar. Biol. 2005, 146, 373–379. [Google Scholar] [CrossRef]
- Aragón-Noriega, E.A. Coupling the reproductive period of blue shrimp Litopenaeus stylirostris Stimpson, 1874 (Decapoda: Penaeidae) and sea surface temperature in the Gulf of California. Rev. Biol. Mar. Oceanogr. 2007, 42, 167–175. [Google Scholar] [CrossRef]
- Aragón-Noriega, E.; Pérez-Arvizu, E.; Valenzuela-Quiñonez, W. Latitudinal variation in reproduction timing of whiteleg shrimp Litopenaeus vannamei (Decapoda, Penaeidae) of the Mexican Pacific coast. Crustaceana 2012, 85, 287–300. [Google Scholar] [CrossRef]
- García-Martínez, S.; Chávez-Ortiz, E.A. La Pesquería de Camarón en Puerto San Carlos, Bahía Magdalena: Una Perspectiva Socioeconómica. In Estudios Ecológicos en Bahía Magdalena; Funes-Rodríguez, R., Gómez-Gutiérrez, J., Palomares-García, R.S., Eds.; Centro de Investigaciones Biológicas del Noroeste: La Paz, Mexico, 2007; pp. 277–287. [Google Scholar]
- Doode, S.; Wong, P. El Golfo de California: Surgimiento de nuevos actores sociales, ambientalismo y región. Estudios Sociales 2001, 11, 25–56. [Google Scholar]
- Rodriguez Quiroz, G.; Bracamontes-Sierra, A. Pertinencia de las ANP como política de conservación y mejoramiento de la calidad de vida: Análisis de percepción en la Reserva de la Biosfera del Alto Golfo de California y Delta del Río Colorado. Estudios Sociales 2008, 16, 141–176. [Google Scholar]
- Morzaria-Luna HNTurk-Boyer, P.; Hernández, J.M.D.; Polanco-Mizquez, E.; Downton-Hoffmann, C.; Cruz-Piñón, G.; Munguia-Vega, A. Fisheries management tools to support coastal and marine spatial planning: A case study from the Northern Gulf of California, Mexico. MethodsX 2020, 7, 101108. [Google Scholar] [CrossRef]

| Data | Source | Period |
|---|---|---|
| Ocurrence records | Ruiz-Luna (2010) | 2010 |
| Amezcua et al. (2006) | 2001–2002 | |
| Manzano Sarabia et al. (2007) | 2003 | |
| Morales-Bojórquez et al. (2012) | 1992–2006 | |
| Hernández-Padilla et al. (2018) | 2005–2006 | |
| Rábago-Quiroz et al. (2011) | 2004–2007 | |
| López-Martínez et al. (2012) | 2004–2005 | |
| García-Juárez (2012) | 2012 | |
| INAPESCA (2014) | 2014 | |
| Herrera-Valdivia et al. (2016) | 2009–2011 | |
| GBIF.org (accessed on 28 February 2019) GBIF Occurrence Download (https://doi.org/10.15468/39omei) | 1990–2018 | |
| OBIS (https://obis.org/; accessed on 13 September 2020) | 1990–2014 | |
| INaturalist (https://www.inaturalist.org/; accessed on 13 September 2020) | 2016–2019 | |
| Primary Production | Oregon State University (https://www.science.oregonstate.edu/ocean.productivity/; accessed on 13 September 2020) | 2003–2019 |
| Sea Surface Salinity | Hycom (https://hycom.org/; accessed on 13 September 2020) | 2003–2019 |
| Sea Surface Temperature | Modis Aqua (https://oceancolor.gsfc.nasa.gov/; accessed on 13 September 2020) | 2003–2019 |
| Bathymetry | GEBCO (https://www.gebco.net/; accessed on 13 September 2020) | |
| Primary Production (RCP models) | Max-Planck-Institut für Meteorologie (https://esgf-node.llnl.gov/projects/esgf-llnl/; accessed on 13 September 2020) | 2006–2100 |
| Sea Surface Temperature (RCP models) | Max-Planck-Institut für Meteorologie (https://esgf-node.llnl.gov/projects/esgf-llnl/; accessed on 13 September 2020) | 2006–2100 |
| Sea Surface Salinity (RCP models) | Max-Planck-Institut für Meteorologie (https://esgf-node.llnl.gov/projects/esgf-llnl/; accessed on 13 September 2020) | 2006–2100 |
| Environmental Variable | Blue Shrimp | White Shrimp | Brown Shrimp |
|---|---|---|---|
| Bathymetry | 79.5% | 68.6% | 83% |
| SST summer | 11.2% | 16.1% | 7.6% |
| SSS Autumn | 5.5% | 9.7% | 3.4% |
| Substratum | 2.8% | 4.2% | 2.5% |
| PP winter | 0.6% | 0.9% | 2% |
| SST summer | 0.2% | 0.3% | 0.7% |
| PP summer | 0.1% | 0.1% | 0.4% |
| Coastal type | 0% | 0.1% | 0.3% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cota-Durán, A.; Petatán-Ramírez, D.; Ojeda-Ruiz, M.Á.; Marín-Monroy, E.A. Potential Impacts of Climate Change on Shrimps Distribution of Commercial Importance in the Gulf of California. Appl. Sci. 2021, 11, 5506. https://doi.org/10.3390/app11125506
Cota-Durán A, Petatán-Ramírez D, Ojeda-Ruiz MÁ, Marín-Monroy EA. Potential Impacts of Climate Change on Shrimps Distribution of Commercial Importance in the Gulf of California. Applied Sciences. 2021; 11(12):5506. https://doi.org/10.3390/app11125506
Chicago/Turabian StyleCota-Durán, Andres, David Petatán-Ramírez, Miguel Ángel Ojeda-Ruiz, and Elvia Aida Marín-Monroy. 2021. "Potential Impacts of Climate Change on Shrimps Distribution of Commercial Importance in the Gulf of California" Applied Sciences 11, no. 12: 5506. https://doi.org/10.3390/app11125506
APA StyleCota-Durán, A., Petatán-Ramírez, D., Ojeda-Ruiz, M. Á., & Marín-Monroy, E. A. (2021). Potential Impacts of Climate Change on Shrimps Distribution of Commercial Importance in the Gulf of California. Applied Sciences, 11(12), 5506. https://doi.org/10.3390/app11125506





