Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.)
Abstract
:1. Introduction
2. Materials and Methods
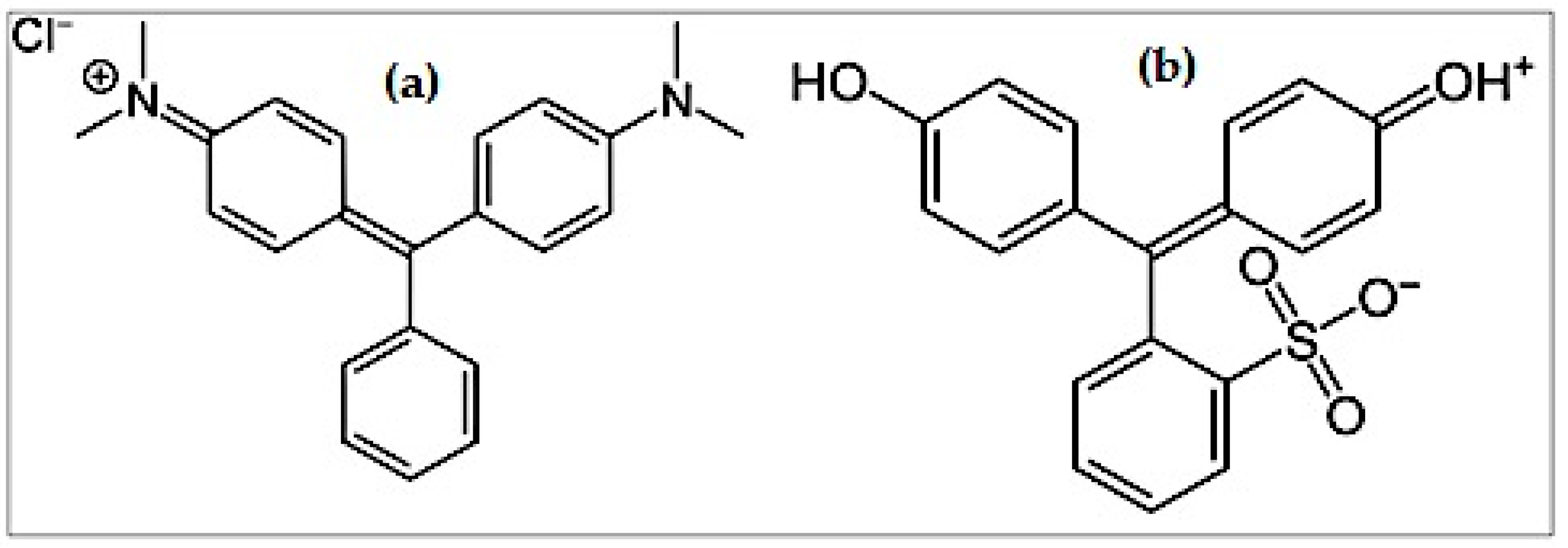
2.1. Materials
2.2. Characterization of BS-HVL and BC-HVL
2.3. Adsorption Process
2.3.1. Adsorption Kinetics
- qe is the amount adsorbed at equilibrium (mg g−1),
- qt is the quantity adsorbed at time t (mg g−1),
- KS1 is the rate constant of first order sorption (min−1),
- Ki is the intraparticle diffusion rate constant,
- The value of the ordinate at the origin C provides an indication of the thickness of the boundary layer,
- K2 (g mg−1.min−1) is the adsorption rate,
- α is the initial adsorption capacity (mg g−1 min),
- β is the desorption constant (g mg−1),
- t is the time (min).
2.3.2. Adsorption Isotherm
- C0 is the initial concentration of the adsorbate (mg L−1),
- KL is the Langmuir constant (L mg−1),
- RL > 1 indicates that the adsorption is unfavorable,
- RL = 1 indicates that the adsorption is linear,
- 0 < RL < 1 indicates that the adsorption is favorable,
- RL = 0 indicates that the adsorption is irreversible.
3. Results and Discussion
3.1. Interpretation of Analysis
3.2. Effect of pH on the Adsorption Capacity of X-PR and MGC
3.3. Effect of the Adsorbent Dose on the Adsorption Capacity of X-PR and MGC
3.4. Effect of Initial Concentration
3.5. Effect of Contact Time
3.6. Adsorption Kinetics
3.7. Adsorption Isotherms
3.8. Statistical Analysis
3.9. Comparison of Adsorption Capacity with Various Adsorbents
3.10. Adsorption Thermodynamics Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- EL Mansouri, F.; Palma Lovillo, M.; El Farissi, H.; Oufdou, H.; Brigui, J. Extraction, analysis of polyphenols and antioxidant properties of morrocan barley seed extracts (Hordeum vulgare L.). Mater. Today Proc. 2021, 43, 1896–1902. [Google Scholar] [CrossRef]
- Crini, G.; Badot, P.M. Traitement et Epuration des Eaux Industrielles Polluées; Presses Universitaires de Franche-Comté: Besançon, France, 2007; 353p. [Google Scholar]
- Crini, G. Non-conventional low-cost adsorbents for dye removal: A review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Rastogi, V.K. Biosorption of lead (II) from aqueous solutions by non-living algal biomass Oedogonium sp. and Nostoc sp.—a comparative study. Colloids Surf. 2008, 64, 170–178. [Google Scholar] [CrossRef]
- Saini, J.; Garg, V.K.; Gupta, R.K.; Kataria, N. Removal of Orange G and Rhodamine B dyes from aqueous system using hydrothermally synthesized zinc oxide loaded activated carbon (ZnO-AC). J. Environ. Chem. Eng. 2017, 5, 884–892. [Google Scholar] [CrossRef]
- Gupta, A.; Rastogi, V.K.; Nayak, A. Biosorption of nickel onto treated alga (Oedo- gonium hatei): Application of isotherm and kinetic models. J. Colloid Interface Sci. 2010, 342, 533–539. [Google Scholar] [CrossRef]
- Gupta, P.R.; Gogate, H. Intensified removal of copper from waste water using activated watermelon based biosorbent in the presence of ultrasound. Ultrason. Sonochem. 2016, 30, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, M.; Mashhadi, S.; Asif, M.; Tyagi, I.; Agarwal, S.; Kumar, V. Microwave-assisted synthesis of tetraethylenepentamine functionalized activated carbon with high adsorption capacity for Malachite green dye. J. Mol. Liq. 2016, 213, 317–325. [Google Scholar] [CrossRef]
- Parlayici, Ş. Alginate-coated perlite beads for the efficient removal of methylene blue, malachite green, and methyl violet from aqueous solutions: Kinetic, thermodynamic, and equilibrium studies. J. Anal. Sci. Technol. 2019, 10, 4. [Google Scholar] [CrossRef]
- Ismail, S.N.A.S.; Rahman, W.A.; Rahim, N.A.A.; Masdar, N.D.; Kamal, M.L. Adsorption of malachite green dye from aqueous solution using corn cob. AIP Conf. Proc. 2018, 2031, 020036. [Google Scholar]
- My Linh, N.L.; Duong, T.; Van Duc, H.; Thi Anh Thu, N.; Khac Lieu, P.; Van Hung, N.; Hoa, L.T.; Quang Khieu, D. Phenol Red Adsorption from Aqueous Solution on the Modified Bentonite. J. Chem. 2020, 2020, 1–14. [Google Scholar] [CrossRef]
- Masoudian, N.; Rajabi, M.; Ghaedi, M. Titanium oxide nanoparticles loaded onto activated carbon prepared from bio-waste watermelon rind for the efficient ultrasonic-assisted adsorption of congo red and phenol red dyes from wastewaters. Polyhedron 2019, 173, 114105. [Google Scholar] [CrossRef]
- Alebachew, N.; Yadav, O.P. Lokesh, Removal of Phenol Red Dye from Contaminated Water Using Barley (Hordeum vulgare L.) Husk-Derived Activated Carbon. Sci. Int. 2017, 5, 7–16. [Google Scholar]
- Grady, H.L.; Nonneman, D.J.; Rottinghaus, G.E.; Welshons, W.V. pH-dependent cytotoxicity of contaminants of phenol red for MCF-7 breast cancer cells. Endocrinology 1991, 129, 3321–3330. [Google Scholar] [CrossRef] [PubMed]
- Steffens, W.; Leider, U.; Wehring, D.; Hattop, W.H. Moglichkeiten und Gefahrn der Anwendung von Malachitgrun in der fischerei. Zeitshrift Fish. 1961, 10, 745–771. [Google Scholar]
- Werth, G.; Boiteaux, A. The toxicity of the triphenylmethane dyestuff malachite green, as an uncoupler of oxidative phosphorylation in vivo and in vitro. Arch. Toxicol. 1967, 23, 82–103. [Google Scholar]
- Meyer, F.P.; Jorgensen, T.A. Teratological and other effects of malachite green on the development of rainbow trout and rabbits. Trans. Am. Fish. Soc. 1983, 112, 818–824. [Google Scholar] [CrossRef]
- Srivastava, S.; Sinha, R.; Roy, D. Toxicological effects of malachite green. Aquat. Toxicol. 2004, 66, 319–329. [Google Scholar] [CrossRef]
- Hormazabal, V.; Steffenak, I.; Yndestad, M. A time and cost-effective assay for the determination of residues of malachite green in fish tissues by HPLC. J. Liquid Chromatogr. 1992, 15, 2035–2044. [Google Scholar] [CrossRef]
- Alderman, D.J.; Clifton-Hadley, R.S. Malachite green: A pharmacokinetic study in rainbow trout, Oncorhynchus mykiss (Walbaum). J. Fish Dis. 1993, 16, 297–311. [Google Scholar] [CrossRef]
- El Farissi, H.; Lakhmiri, R.; Albourine, A.; Safi, M. The adsorption of the orange acid dye 52 in aqueous solutions by the biochar of the seeds and shells of Cistus Ladaniferus. Int. J. Sci. Eng. Res. 2018, 9, 563–571. [Google Scholar]
- El Fargani, H.; Lakhmiri, R.; El Farissi, H.; Albourine, A.; Safi, M.; Cherkaoui, O. Modified Chitosan Immobilized on Modified Sand for Industrial Wastewater Treatment in Multicomponent Sorption: Shrimp Biowaste Processing. Chem. Mater. Res. 2017, 9, 20–42. [Google Scholar]
- El Farissi, H.; Lakhmiri, R.; Albourine, A.; Safi, M.; Cherkaoui, O. Removal of anionic dyes from aqueous solutions by cistus ladaniferus shells and their biochar: Isotherms, kinetic and thermodynamic studies. Int. J. Sci. Eng. Res. 2018, 9, 200–211. [Google Scholar] [CrossRef]
- El Farissi, H.; Lakhmiri, R.; Albourine, A.; Safi, M.; Cherkaoui, O. Adsorption study of charcoal of cistus ladaniferus shell modified by H3PO4 and NaOH used as a low-cost adsorbent for the removal of toxic reactive red 23 dye: Kinetics and thermodynamics. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Lagergren, S. Zur theorie der sogenannten adsorption gel osterstoffe. Kungliga Svenska Vetenskapsakademiens. Handlingar 1898, 124, 1–13. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Chien, S.H.; Clayton, W.R. Application of Elovich equation to the kinetics of phosphate release and sorption in soils. Soil Sci. Soc. Am. J. 1980, 44, 265–268. [Google Scholar] [CrossRef]
- Urano, K.; Hirotaka, T. Process Development for Removal and Recovery of Phosphorus from Wastewater by a New Adsorbent. 2. Adsorption Rates and Breakthrough Curves. Ind. Eng. Chem. Res. 1991, 30, 1897–1899. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Das, S.K.; Bhowal, J.; Das, A.R.; Guha, A.K. Adsorption Behavior of Rhodamine B on Rhizopus oryzae Biomass. Langmuir 2006, 22, 7265–7272. [Google Scholar] [CrossRef]
- Sewu, D.D.; Boakye, P.; Woo, S.H. Highly efficient adsorption of cationic dye by biochar produced with Korean cabbage waste. Bioresour. Technol. 2017, 224, 206–213. [Google Scholar] [CrossRef]
- Popa, N.; Visa, M. The synthesis, activation and characterization of charcoal powder for the removal of methylene blue and cadmium from wastewater. Adv. Powder Technol. 2017, 28, 1866–1876. [Google Scholar] [CrossRef]
- Islam, M.T.; Hyder, A.G.; Saenz-Arana, R.; Hernandez, C.; Guinto, T.; Ahsan, M.A.; Alvarado-Tenorio, B.; Noveron, J.C. Removal of methylene blue and tetracycline from water using peanut shell derived adsorbent prepared by sulfuric acid reflux. J. Environ. Chem. Eng. 2019, 7, 102816. [Google Scholar] [CrossRef]
- Isa, K.M.; Daud, S.; Hamidin, N.; Ismail, K.; Saad, S.A.; Kasim, F.H. Thermogravimetric analysis and the optimisation of bio-oil yield from fixed-bed pyrolysis of rice husk using response surface methodology (RSM). Ind. Crops Prod. 2011, 33, 481–487. [Google Scholar] [CrossRef]
- Núñez-gómez, D.; Rodrigues, C.; Rubens, F. Adsorption of heavy metals from coal acid mine drainage by shrimp shell waste: Isotherm and continuous-flow studies. J. Environ. Chem. Eng. 2019, 7, 1–10. [Google Scholar] [CrossRef]
- Dash, S.; Chaudhuri, H.; Gupta, R.; Nair, U.G. Adsorption Study of Modified Coal Fly Ash with Sulfonic Acid as a Potential Adsorbent for the Removal of Toxic Reactive Dyes from Aqueous Solution: Kinetics and Thermodynamics. J. Environ. Chem. Eng. 2018, 6, 5897–5905. [Google Scholar] [CrossRef]
- El Farissi, H.; Lakhmiri, R.; Albourine, A.; Safi, M.; Cherkaoui, O. Removal of RR-23 dye from industrial textile wastewater by adsorption on cistus ladaniferus seeds and their biochar. J. Environ. Earth Sci. 2017, 7, 105–118. [Google Scholar]
- El Fargani, H.; Lakhmiri, R.; El Farissi, H.; Albourine, A.; Safi, M.; Cherkaoui, O. Removal of anionic dyes by silica-chitosan composite in single and binary systems: Valorization of shrimp co- product Crangon-Crangon and Pandalus Borealis. J. Mater. Environ. Sci. 2017, 8, 724–739. [Google Scholar]
- Chen, S.; Zhang, J.; Zhang, C.; Yue, Q.; Li, Y.; Li, C. Equilibrium and kinetic studies of methyl orange and methyl violet adsorption on activated carbon derived from Phragmites australis. Desalination 2010, 252, 149–156. [Google Scholar] [CrossRef]
- Zhao, D.; Zhang, W.; Chen, C.; Wang, X. Adsorption of Methyl Orange Dye Onto Multiwalled Carbon Nanotubes. Procedia Environ. Sci. 2013, 18, 890–895. [Google Scholar] [CrossRef] [Green Version]
- Nourmoradi, H.; Moghadam, K.F.; Jafari, A.; Kamarehie, B. Removal of Acetaminophen and Ibuprofen from Aqueous Solutions by Activated Carbon Derived from Quercus Brantii (Oak) Acorn as a Low-cost Biosorbent. J. Environ. Chem. Eng. 2018, 6, 6807–6815. [Google Scholar] [CrossRef]
- Jin, G.P.; Wang, X.L.; Fu, Y.; Do, Y. Preparation of tetraoxalyl ethylenediamine melamine resin grafted-carbon fibers for nano-nickel recovery from spent electroless nickel plating baths. Chem. Eng. J. 2012, 203, 440–446. [Google Scholar] [CrossRef]
- Mahjoub, B.; Ncibi, M.C.; Seffen, M. Adsorption of a Reactive Textile Dye on a Non-Conventional Biosorbent: The Fibers of Posidonia Oceanica (L.) Delile. Can. J. Chem. Eng. 2008, 86, 23–29. (In French) [Google Scholar] [CrossRef]
- Mane, V.S.; Vijay Babu, P.V. Studies on the adsorption of Brilliant Green dye from aqueous solution onto low-cost NaOH treated saw dust. Desalination 2011, 273, 321–329. [Google Scholar] [CrossRef]

















| Models | BS-HVL | BC-HVL | |||
|---|---|---|---|---|---|
| X-PR | MGC | X-PR | MGC | ||
| Pseudo-first-order | R2 | 0.9179 | 0.9546 | 0.9127 | 0.9333 |
| K1 (min−1) | 0.045 | 0.0565 | 0.0401 | 0.0551 | |
| qe (mg g−1) | 44.7817 | 72.3633 | 40.8007 | 66.4201 | |
| Pseudo-second-order | R2 | 0.9379 | 0.9969 | 0.9647 | 0.9692 |
| K2 (g mg−1 min−1) | 0.0005 | 0.0011 | 0.0006 | 0.0007 | |
| qe (mg g−1) | 61.7283 | 54.0540 | 41.6667 | 41.8410 | |
| Elovich | R2 | 0.9101 | 0.9742 | 0.9429 | 0.9341 |
| α (mg g−1 min−1) | 3.6528 | 7.8653 | 2.1251 | 2.6571 | |
| β (g mg−1) | 0.0674 | 0.0913 | 0.1071 | 0.1073 | |
| Intraparticle diffusion | R2 | 0.7969 | 0.9191 | 0.9519 | 0.9306 |
| Ki (mg g−1 min0.5) | 4.2488 | 3.2554 | 2.8709 | 3.3419 | |
| C (mg g−1) | 5.3228 | 14.733 | 1.0959 | 3.3419 | |
| Models | Constants | BS-HVL | BC-HVL | ||
|---|---|---|---|---|---|
| X-PR | MGC | X-PR | MGC | ||
| Langmuir | R2 | 0.9954 | 0.8953 | 0.6271 | 0.8954 |
| RL | 0.1862–0.6958 | 0.0089–0.0823 | 0.0405–0.2968 | 0.0099–0.9091 | |
| KL (L mg−1) | 0.0437 | 1.115 | 0.2369 | 1 | |
| qm (mg g−1) | 71.642 | 50 | 44.843 | 121.95 | |
| Freundlich | R2 | 0.9954 | 0.977 | 0.7529 | 0.9927 |
| KF (L mg−1) | 4.4017 | 5.1908 | 13.5637 | 6.2883 | |
| n | 0.9975 | 0.9876 | 3.5385 | 0.8011 | |
| Temkin | R2 | 0.8793 | 0.9195 | 0.5801 | 0.9441 |
| KT (L mg−1) | 0.1074 | 0.1117 | 0.1104 | 0.6773 | |
| B1 (J mol−1) | 7.498 | 6.427 | 6.6877 | 45.011 | |
| b | 325.3889 | 380.1369 | 365.3184 | 54.2787 | |
| Dubinin– Radushkevich | R2 | 0.6943 | 0.7648 | 0.4148 | 0.8968 |
| Kad (mol2 KJ−2) | 20 × 10−5 | 30 × 10−5 | 0.04 × 10−5 | 1 × 10−5 | |
| E (KJ mol−1) | 15.8114 | 12.9099 | 35.3563 | 70.7108 | |
| qm (mg g−1) | 11.5802 | 10.7124 | 31.5004 | 71.715 | |
| Type of Analysis | Parameter Study | Type of Sample | Mean | Std. Error | 95% Confidence Interval | Test ANOVA | ||
|---|---|---|---|---|---|---|---|---|
| Lower Bound | Upper Bound | F | Sig. | |||||
| Effect of pH on the adsorption capacity of X-PR and MGC | Adsorption capacity, X-PR, (mg g−1) | BS-HVL | 30.737 | 2.244 | 24.966 | 36.5080 | 19.735 | 0.001 S |
| BC-HVL | 14.918 | 2.764 | 7.812 | 22.023 | ||||
| Adsorption capacity, MGC, (mg g−1) | BS-HVL | 22.690 | 4.104 | 12.139 | 33.241 | 1.784 | 0.211 NS | |
| BC-HVL | 16.011 | 2.855 | 8.670 | 23.353 | ||||
| Effect of adsorbent dose on adsorption yield of X-PR and MGC | Adsorption yield, X-PR, (%) | BS-HVL | 46.300 | 5.621 | 33.58 | 59.02 | 0.360 | 0.556 NS |
| BC-HVL | 42.100 | 4.165 | 32.68 | 51.52 | ||||
| Adsorption yield, MGC, (%) | BS-HVL | 63.300 | 8.013 | 45.17 | 81.43 | 0.034 | 0.855 NS | |
| BC-HVL | 61.200 | 8.023 | 43.05 | 79.35 | ||||
| Effect of concentration on the adsorption capacity of X-PR and MGC | Adsorption capacity, X-PR, (mg g−1) | BS-HVL | 51.578 | 9.270 | 22.284 | 80.871 | 3.537 | 0.076 NS |
| BC-HVL | 31.897 | 4.872 | 16.498 | 47.295 | ||||
| Adsorption capacity, MGC, (mg g−1) | BS-HVL | 46.072 | 7.715 | 21.691 | 70.452 | 0.105 | 0.749 NS | |
| BC-HVL | 49.859 | 8.774 | 22.131 | 77.587 | ||||
| Effect of contact time on adsorption capacity of X-PR and MGC | Adsorption capacity, X-PR, (mg g−1) | BS-HVL | 37.920 | 3.669 | 26.323 | 49.517 | 13.670 | 0.001 S |
| BC-HVL | 23.250 | 2.333 | 15.875 | 30.625 | ||||
| Adsorption capacity, MGC, (mg g−1) | BS-HVL | 39.750 | 2.655 | 31.358 | 48.142 | 20.278 | 0.000 S | |
| BC-HVL | 25.33 | 2.330 | 17.965 | 32.695 | ||||
| Effect of temperature on adsorption of X-PR and MGC | Adsorption capacity, X-PR, (mg g−1) | BS-HVL | 43.02 | 1.098 | 39.548 | 46.492 | 13.003 | 0.011 S |
| BC-HVL | 49.3135 | 0.10 | 48.966 | 49.660 | ||||
| Adsorption capacity, MGC, (mg g−1) | BS-HVL | 32.87 | 1.158 | 29.210 | 36.530 | 75.522 | 0.000 S | |
| BC-HVL | 48.8510 | 0.115 | 48.484 | 49.217 | ||||
| Adsorbate | Adsorbent Pollutants | Dose (mg) | C0 (mg L−1) | pH | Kinetic | Isotherm | qm (mg g−1) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Zinc oxide loaded activated char (ZnO-AC) | OG Rh-b | 8–30 | 50 | 7 | Pseudo-second-order | Langmuir | 153.8 128.2 | [5] |
| Rice straw (RS) biochar Wood chip (WC) biochar | CV-CR | 01 | 500 | 7 | Pseudo-second-order | Langmuir | 620.3 195.6 | [31] |
| Charcoal (tree branches) (BCA-TiO2) | MB Cd2+ | ** ** | 0.4 600 | 7 8 | Pseudo-second-order | ** | 200 250 | [32] |
| Sulfonated peanut shell (PNS-SO3H) | MB TC | 20 | 900 ppm | 10 | Pseudo-second-order | Langmuir | 1250 303 | [33,34] |
| Shrimp shell (SS) Coal acid mine drainage (AMD) | Mn Fe | ** | ≤1 ≤15 | 6–9 5–9 | Pseudo-second-order | Frendlich | 17.43 3.87 | [35] |
| Coal fly ash (CFA) | MG RG | 40 30 | 100 ppm | 8 | Pseudo-second-order | Langmuir | 233.3 381.7 | [36] |
| Biomass CLS Biochar (BCCLS) | RR-23 | 50 | 50 | 7 | Pseudo-second-order | Langmuir | 62.5 166.67 | [37] |
| Biomass CLSh Biochar (BCCLSh) | RR-23 | 50 | 50 | 7 | Pseudo-second-order | Langmuir | 90.91 354.82 | [24] |
| Biochar BC CLS Biochar (BCCLSh) | AO-52 | 50 | 300 | 7 | Pseudo-second-order | Langmuir | 333.33 500 | [22] |
| Silica-Chitosan Composite | RR-23 RB19 | 40 | 60 | 7 | Pseudo-second-order | Langmuir | 128.2 156.25 | [38] |
| Chitosan Composite MCs/MS | RR-23 RB19 Fe2+ | 70 | 50 | 7 | Pseudo-second-order | Langmuir Frendlich Frendlich | 71.94 175.44 62.11 | [23] |
| Activated Carbon derived from Phragmites Australis | MO MV | 50 50 | 500 400 | ** | Pseudo-second-order | Langmuir | 238.11 476.19 | [39] |
| Carbon nanotubes (CNTs) | MO | 200 mg L−1 | 10 | ** | Pseudo-second-order | Langmuir | 55.2 | [40] |
| Carbon of Quercus Brantii (Oak) | ACT IBP | 1 g L−1 | 100 | 7 | Pseudo-second-order | Freundlich | 45.45 96.15 | [41] |
| BS−HVL | X−PR | 50 | 50 | 7 | Pseudo-second-order | Langmuir-Freundlich | 71.642 | This work |
| BS−HVL | MGC | 50 | 50 | 7 | Pseudo-second-order | Freundlich | 44.843 | |
| BS−HVL | X−PR | 50 | 50 | 7 | Pseudo-second-order | Freundlich | 44.843 | |
| BS−HVL | MGC | 50 | 50 | 7 | Pseudo-second-order | Freundlich | 121.95 |
| Parameters | BS-HVL | BC-HVL | |||
|---|---|---|---|---|---|
| X-PR | MGC | X-PR | MGC | ||
| ΔH° (kJ mol−1) | 23.55 | 12.757 | 21.179 | 13.386 | |
| ΔS° (J mol−1 K−1) | 90.797 | 46.074 | 103.734 | 74.759 | |
| ΔG° (kJ mol−1) | T = 294 K | −27.034 | −13.533 | −30.477 | −21.966 |
| T= 303 K | −27.942 | −13.948 | −31.410 | −22.639 | |
| T= 323 K | −29.758 | −14.869 | −33.485 | −24.134 | |
| T= 343 K | −30.666 | −15.791 | −35.560 | −25.629 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mansouri, F.E.; Farissi, H.E.; Zerrouk, M.H.; Cacciola, F.; Bakkali, C.; Brigui, J.; Lovillo, M.P.; Esteves da Silva, J.C.G. Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.). Appl. Sci. 2021, 11, 5125. https://doi.org/10.3390/app11115125
Mansouri FE, Farissi HE, Zerrouk MH, Cacciola F, Bakkali C, Brigui J, Lovillo MP, Esteves da Silva JCG. Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.). Applied Sciences. 2021; 11(11):5125. https://doi.org/10.3390/app11115125
Chicago/Turabian StyleMansouri, Fouad El, Hammadi El Farissi, Mohamed Hassani Zerrouk, Francesco Cacciola, Chaimae Bakkali, Jamal Brigui, Miguel Palma Lovillo, and Joaquim C. G. Esteves da Silva. 2021. "Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.)" Applied Sciences 11, no. 11: 5125. https://doi.org/10.3390/app11115125
APA StyleMansouri, F. E., Farissi, H. E., Zerrouk, M. H., Cacciola, F., Bakkali, C., Brigui, J., Lovillo, M. P., & Esteves da Silva, J. C. G. (2021). Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.). Applied Sciences, 11(11), 5125. https://doi.org/10.3390/app11115125










