The Effect of Noble Metals on Co Gas Sensing Properties of In2O3 Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Decorated In2O3 Particles (Au/In2O3, Pd/In2O3, Pt/In2O3)
2.2. Characterization
3. Results
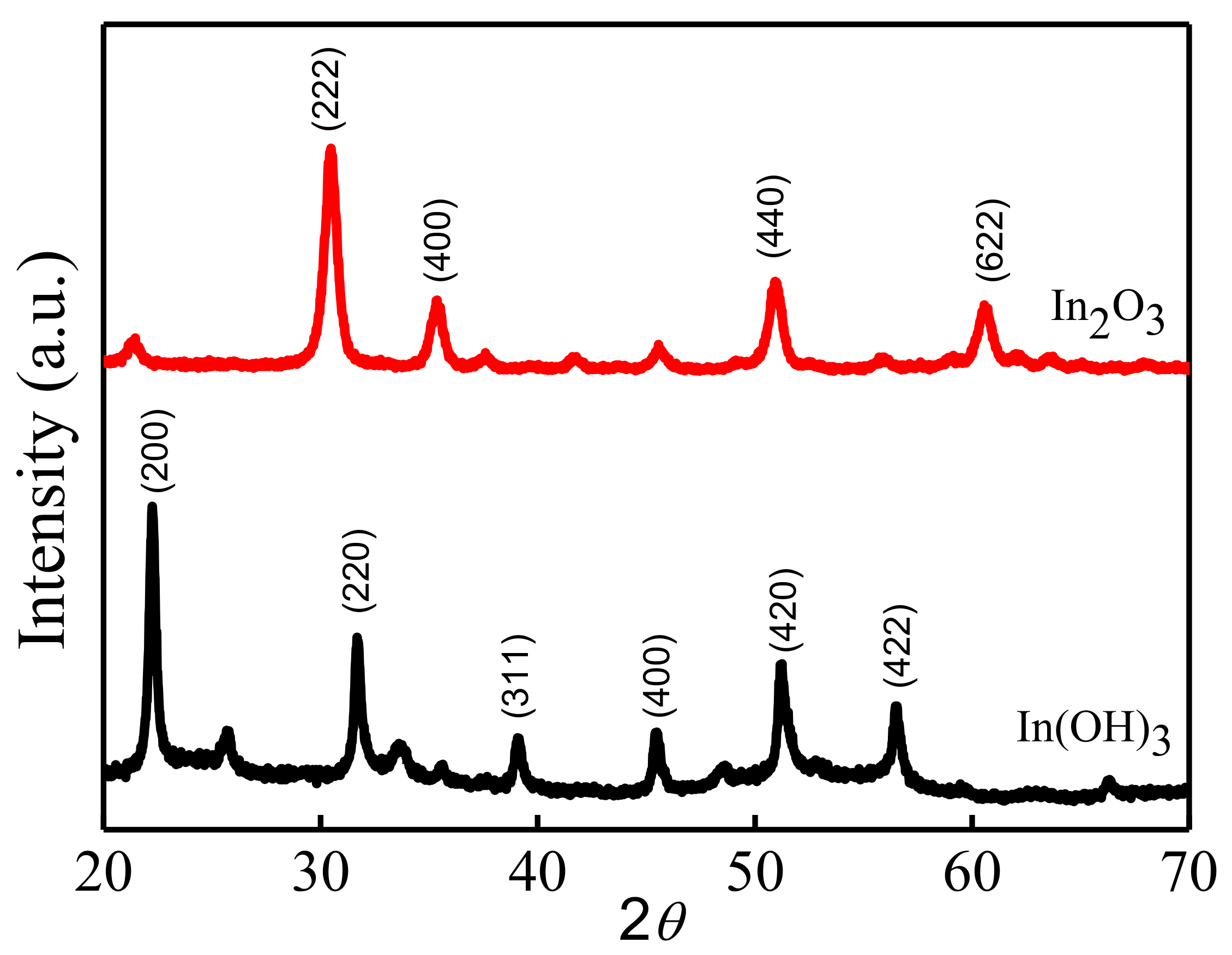
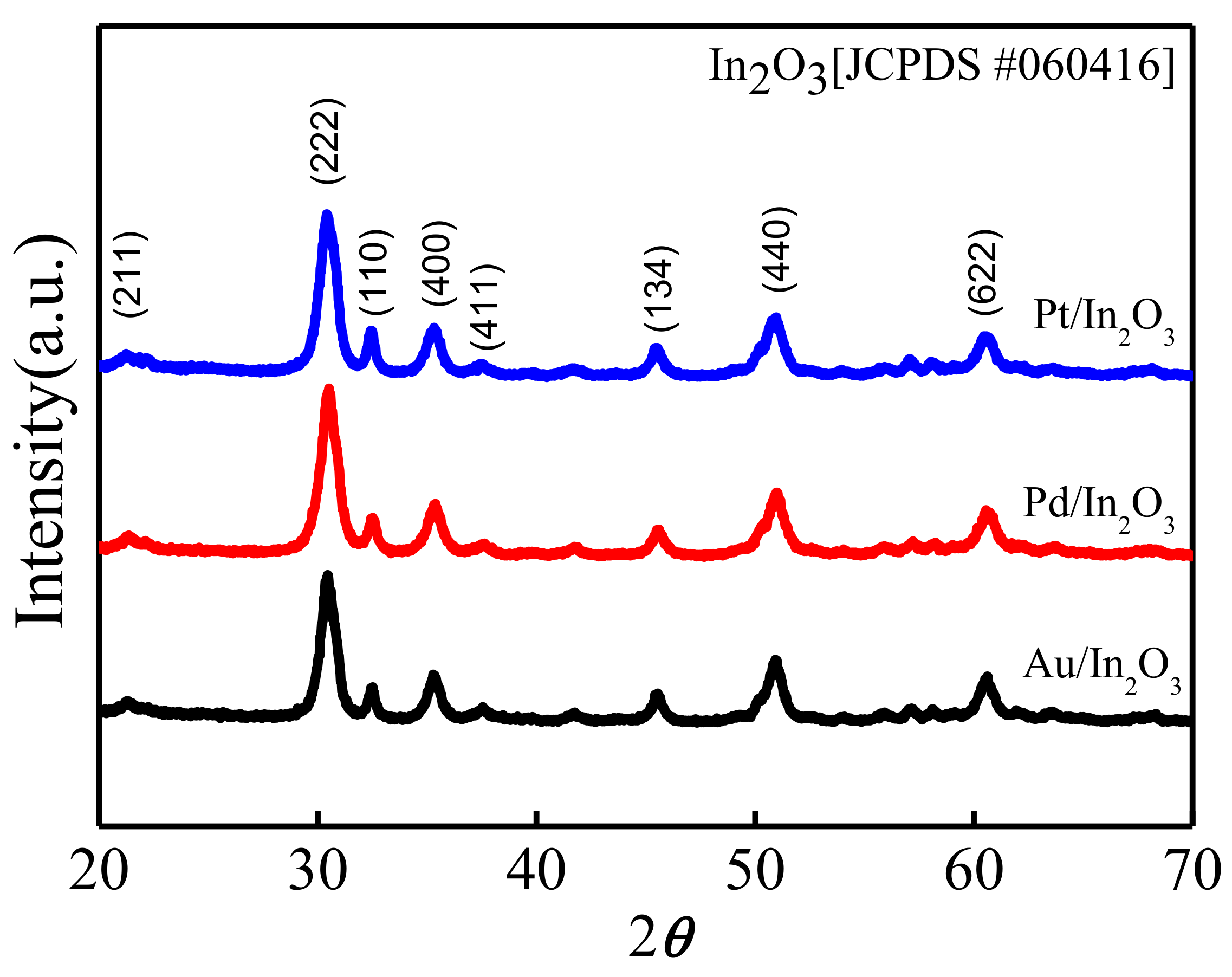
3.1. Structural and Morphological Characteristics
3.2. XPS Analysis of Noble Metal-Decorated In2O3 Particles
3.3. Gas Sensing Properties
3.4. Gas Sensing Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McCue, J.T.; Ying, J.Y. SnO2-In2O3 nanocomposites as semiconductor gas sensors for CO and NOx etection. Chem. Mater. 2007, 19, 1009–1015. [Google Scholar] [CrossRef]
- Goto, T.; Itoh, T.; Akamatsu, T.; Izu, N.; Shin, W. CO sensing properties of Au/SnO2–Co3O4 catalysts on a micro thermoelectric gas sensor. Sens. Actuators B Chem. 2016, 223, 774–783. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Pandey, A.; Satpati, B.; Tomar, M.; Gupta, V.; Mohapatra, S. Enhanced CO gas sensing properties of Cu doped SnO2 nanostructures prepared by a facile wet chemical method. Phys. Chem. Chem. Phys. 2016, 18, 18846–18854. [Google Scholar] [CrossRef]
- Kumar, A.; Sanger, A.; Kumar, A.; Chandra, R. Highly sensitive and selective CO gas sensor based on hydrophobic SnO2/CuO bilayer. RSC Adv. 2016, 6, 47178–47184. [Google Scholar] [CrossRef]
- Tee, T.S.; Hui, T.C.; Yi, C.W.; Chin, Y.C.; Umar, A.A.; Titian, G.R.; Beng, L.H.K.; Sing, L.K.; Yahaya, M.; Salleh, M.M. Microwave-assisted hydrolysis preparation of highly crystalline ZnO nanorod array for room temperature photoluminescence-based CO gas sensor. Sens. Actuators B Chem. 2016, 227, 304–312. [Google Scholar]
- Barsan, N.; Simion, C.; Heine, T.; Pokhrel, S.; Weimer, U. Modeling of sensing and transduction for p-type semiconducting metal oxide based gas sensors. J. Electroceram. 2010, 25, 11–19. [Google Scholar] [CrossRef]
- Lee, J.S.; Ha, T.J.; Hong, M.H.; Park, C.S.; Park, H.H. The effect of multiwalled carbon nanotube doping on the CO gas sensitivity of TiO2 xerogel composite film. Appl. Surf. Sci. 2013, 269, 125–128. [Google Scholar] [CrossRef]
- Hjiri, M.; El Mir, L.; Leonardi, S.G.; Pistone, A.; Mavilia, L.; Neri, G. Al-doped ZnO for highly sensitive CO gas sensors. Sens. Actuators B Chem. 2014, 196, 413–420. [Google Scholar] [CrossRef]
- Fu, H.; Hou, C.; Gu, F.; Han, D.; Wang, Z. Facile preparation of rod-like Au/In2O3 nanocomposites exhibiting high response to CO at room temperature. Sens. Actuators B Chem. 2017, 243, 516–524. [Google Scholar] [CrossRef]
- Hübner, M.; Simion, C.E.; Haensch, A.; Barsan, N.; Weimar, U. CO sensing mechanism with WO3 based gas sensors. Sens. Actuators B Chem. 2010, 151, 103–106. [Google Scholar] [CrossRef]
- Patil, D.; Patil, P.; Subramanian, V.; Joy, P.A.; Potdar, H.S. Highly sensitive and fast responding CO sensor based on Co3O4 nanorods. Talanta 2010, 81, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Steinhauer, S.; Brunet, E.; Maier, T.; Mutinati, G.C.; Köck, A.; Köck, A. CuO nanowire gas sensors for CO detection in humid atmosphere. In Proceedings of the 2013 Transducers & Eurosensors XXVII: The 17th International Conference on Solid-State Sensors, Actuators and Microsystems, Barcelona, Spain, 16–20 June 2013; pp. 1095–1098. [Google Scholar]
- Batzill, M.; Diebold, U. The surface and materials science of tin oxide. Prog. Surf. Sci. 2005, 79, 47–154. [Google Scholar] [CrossRef]
- Moos, R.; Sahner, K.; Fleischer, M.; Guth, U. Solid State Gas Sensor Research in Germany—A Status Report. Sensors 2009, 9, 4323–4365. [Google Scholar] [CrossRef] [PubMed]
- Rumyantseva, M.N.; Gas’kov, A.M. Chemical Modification of Nanocrystalline Metal Oxides: Effect of the Real Structure and Surface Chemistry on the Sensor Properties. Russ. Chem. Bull. 2008, 57, 1106–1125. [Google Scholar] [CrossRef]
- Lee, J.H. Gas Sensors Using Hierarchical and Hollow Oxide Nanostructures: Overview. Sens. Actuators B Chem. 2009, 140, 319–336. [Google Scholar] [CrossRef]
- Noguchi, S.; Sakata, H. Electrical properties of undoped In2O3 films prepared by reactive evaporation. J. Phys. D Appl. Phys. 1980, 13, 1129–1133. [Google Scholar] [CrossRef]
- Shigesato, Y.; Takaki, S.; Haranoh, T. Electrical and structural properties of low resistivity tin-doped indium oxide films. J. Appl. Phys. 1992, 71, 3356–3364. [Google Scholar] [CrossRef]
- Shanmugasundaram, A.; Basak, P.; Satyanarayana, L.; Manorama, S.V. Hierarchical SnO/SnO2 nanocomposites: Formation of in situ p–n junctions and enhanced H2 sensing. Sens. Actuators B Chem. 2013, 185, 265–273. [Google Scholar] [CrossRef]
- Kawasaki, H.; Ueda, T.; Suda, Y.; Ohshima, T. Properties of metal doped tungsten oxide thin films for NOx gas sensors grown by PLD method combined with sputtering process. Sens. Actuators B Chem. 2004, 100, 266–269. [Google Scholar] [CrossRef]
- Hieua, N.V.; Thuy, L.T.B.; Chien, N.D. Highly sensitive thin film NH3 gas sensor operating at room temperature based on SnO2/MWCNTs composite. Sens. Actuators B Chem. 2008, 129, 888–895. [Google Scholar] [CrossRef]
- Lin, Q.; Li, Y.; Yang, M. Tin oxide/graphene composite fabricated via a hydrothermal method for gas sensors working at room temperature. Sens. Actuators B Chem. 2012, 173, 139–147. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, J.; Guo, X.; Wu, S.; Wang, S. Amino acid-assisted one-pot assembly of Au, Pt nanoparticles onto one-dimensional ZnO microrods. Nanoscale 2010, 2, 1178–1184. [Google Scholar] [CrossRef]
- Gogurla, N.; Sinha, A.K.; Santra, S.; Manna, S.; Ray, S.K. Multifunctional Au-ZnO plasmonic nanostructures for enhanced UV photodetector and room temperature NO sensing devices. Sci. Rep. 2014, 4, 6483. [Google Scholar] [CrossRef] [Green Version]
- Santra, S.; Guha, P.K.; Ali, S.Z.; Hiralal, P.; Unalan, H.E.; Covington, J.A.; Amaratunga, G.A.J.; Milne, W.I.; Gardner, J.W.; Udrea, F. ZnO nanowires grown on SOI CMOS substrate for ethanol sensing. Sens. Actuators B Chem. 2010, 146, 559–565. [Google Scholar] [CrossRef]
- Phani, A.R.; Manorama, S.V.; Rao, V.J. X-ray photoelectron spectroscopy studies on Pd doped SnO2 liquid petroleum gas sensor. Appl. Phys. Lett. 1997, 71, 2358–2360. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, X.H.; Guo, X.Z.; Wu, S.H.; Wang, S.R. A general approach to fabricate diverse noble-metal (Au, Pt Ag, Pt/Au)/Fe2O3 hybrid nanomaterials. Chem. Eur. J. 2010, 16, 8108–8116. [Google Scholar] [CrossRef] [PubMed]
- Kolmakov, A.; Klenov, D.O.; Lilach, Y.; Stemmer, S.; Moskovits, M. Enhanced gas sensing by individual SnO2 nanowires and nanobelts functionalized with Pd Catalyst Particles. Nano Lett. 2005, 5, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Vander Wal, R.L.; Hunter, G.W.; Xu, J.C.; Kulis, M.J.; Berger, G.M.; Ticich, T.M. Metal-oxide nanostructure and gas-sensing performance. Sens. Actuators B Chem. 2009, 138, 113–119. [Google Scholar] [CrossRef]
- Joshi, R.K.; Hu, Q.; Alvi, F.; Joshi, N.; Kumar, A. Au decorated zinc oxide nanowires for CO sensing. J. Phys. Chem. C. 2009, 113, 16199–16202. [Google Scholar] [CrossRef]
- Romanovskaya, V.; Ivanovskaya, M.; Bogdanov, P. A study of sensing properties of Pt- and Au-loaded In2O3 ceramics. Sens. Actuators B Chem 1999, 56, 31–36. [Google Scholar] [CrossRef]
- Shen, Y.; Yamazaki, T.; Liu, Z.; Meng, D.; Kikuta, T. Hydrogen sensors made of undoped and Pt-doped SnO2 nanowires. J. Alloys Compd. 2009, 488, 21–25. [Google Scholar] [CrossRef]
- Han, X.G.; Jin, M.S.; Xie, S.F.; Kuang, Q.; Jiang, Z.Y.; Jiang, Y.Q.; Xie, Z.X.; Zheng, L.; Angew, S. Synthesis of trapezohedral indium oxide nanoparticles with high-index {211} facets and high gas sensing activity. Chem. Int. Ed. 2009, 48, 9180–91832009. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Han, X.G.; Xie, S.F.; Kuang, Q.; Jiang, Y.Q.; Zhang, S.B.; Mu, X.L.; Chen, X.G.; Xie, Z.X.; Zheng, L.S. Controlled synthesis and enhanced catalytic and gas-sensing properties of tin dioxide nanoparticles with exposed high-energy facets. Chem. Eur. J. 2012, 18, 2283–2289. [Google Scholar] [CrossRef] [PubMed]
- Valden, M.; Pak, S.; Lai, X.; Goodman, D.W. Structure sensitivity of CO oxidation over model Au/TiO2 2 catalysts. Catal. Lett. 1998, 56, 7–10. [Google Scholar] [CrossRef]
- Schubert, M.M.; Hackenberg, S.; Veen, A.C.V.; Muhler, M.; Plzak, V.; Behm, R.J. CO Oxidation over Supported Gold Catalysts—“Inert” and “Active” Support Materials and Their Role for the Oxygen Supply. J. Catal. 2001, 197, 113–122. [Google Scholar] [CrossRef]
- Grunwaldt, J.D.; Bailker, A. Gold/Titania Interfaces and Their Role in Carbon Monoxide Oxidation. Phys. Chem. B 1999, 103, 1002–1012. [Google Scholar] [CrossRef]
- Liu, H.; Kozlov, A.I.; Kozlov, A.P.; Shido, T.; Asakura, K. Iwasawa, Active Oxygen Species and Mechanism for LowTemperature CO Oxidation Reaction on a TiO2-Supported Au Catalyst Prepared from Au(PPh3)(NO3) and As-Precipitated Titanium Hydroxide. J. Catal. 1999, 185, 252–264. [Google Scholar] [CrossRef]
- Guggenheim, S.; Bain, D.C.; Bergaya, F.; Brigatti, M.F.; Drits, V.A.; Eberl, D.D.; Formoso, M.L.L.; Galán, E.; Merriman, R.J.; Peacor, D.E. Report of the Association Internationale pour l’Etude des Argiles (AIPEA) Nomenclature Committee for 2001: Order, disorder and crystallinity in phyllosilicates and the use of the ‘Crystallinity Index’. Clay Miner. 2002, 37, 389–393. [Google Scholar] [CrossRef]
- Sun, Y.; Zhuang, L.; Lu, J.; Hong, X.; Liu, P. Collapse in Crystalline Structure and Decline in Catalytic Activity of Pt Nanoparticles on Reducing Particle Size to 1nm. J. Am. Chem. Soc. 2007, 129, 15465–15467. [Google Scholar] [CrossRef]
- Jaramillo, T.F.; Baeck, S.H.; Cuenya, B.R.; McFarland, E.W. Catalytic Activity of Supported Au Nanoparticles Deposited from Block Copolymer Micelles. J. Am. Chem. Soc. 2003, 125, 7148–7149. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Kreider, P.B.; Chang, C.H.; Park, C.M.; Ahn, H.G. Visible-light-sensitive nanoscale Au-ZnO photocatalysts. J. Nanopart. Res. 2013, 42, 1–11. [Google Scholar] [CrossRef]
- Minicò, S.; Scirè, S.; Crisafulli, C.; Galvagno, S. Influence of catalyst pretreatments on volatile organic compounds oxidation over gold/iron oxide. Appl. Catal. B Environ. 2001, 34, 277–285. [Google Scholar] [CrossRef]
- Yang, D.J.; Kamienchick, I.; Youn, D.Y.; Rothschild, A.; Kim, I.D. Ultrasensitive and Highly Selective Gas Sensors Based on Electrospun SnO2 Nanofibers Modified by Pd Loading. Adv. Funct. Mater. 2010, 20, 4258–4264. [Google Scholar] [CrossRef]
- Liu, J.; Chen, G.; Yu, Y.; Wu, Y.; Zhou, M.; Zhang, H.; Lv, C.; Zheng, Y.; He, F. Controllable synthesis of In2O3 octodecahedra exposing {110} facets with enhanced gas sensing performance. RSC Adv. 2015, 5, 44306–44312. [Google Scholar] [CrossRef]
- Zhang, Y.; Yueying, L.; Linsheng, Z.; Deye, L.; Fengmin, L.; Fangmeng, L.; Xishuang, L.; Xu, Y.; Yuan, G.; Geyu, L. The role of Ce doping in enhancing sensing performance of ZnO-based gas sensor by adjusting the proportion of oxygen species. Sens. Actuators B Chem. 2018, 273, 991–998. [Google Scholar] [CrossRef]
- Lei, F.; Sun, Y.; Liu, K.; Gao, S.; Liang, L.; Pan, B.; Xie, Y. Oxygen vacancies confined in ultrathin indium oxide porous sheets for promoted visible-light water splitting. J. Am. Chem. Soc. 2014, 136, 6826–6829. [Google Scholar] [CrossRef]
- Gan, J.; Lu, X.; Wu, J.; Xie, S.; Zhai, T.; Yu, M.; Zhang, Z.; Mao, Y.; Wang, S.C.I.; Shen, Y.; et al. Oxygen vacancies promoting photoelectrochemical performance of In2O3 nanocubes. Sci. Rep. 2013, 3, 1021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Kuang, Q.; Xie, Z.X.; Zheng, L.S. The effect of noble metal (Au, Pd and Pt) nanoparticles on the gas sensing performance of SnO2-based sensors: A case study on the {221} high-index faceted SnO2 octahedra. CrystEngComm 2015, 17, 6308–6313. [Google Scholar] [CrossRef]
- Ma, N.; Suematsu, K.; Yuasa, M.; Kida, T.; Shimanoe, K. Effect of water vapor on Pd-loaded SnO2 nanoparticles gas sensor. ACS Appl. Mater. Interfaces 2015, 7, 5863–5869. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Meng, G.F.; Zhao, H.B.; Zhang, Y.; Li, H.; Ma, W.J.; Xu, J.Q. Au nanoparticle modified WO3 nanorods with their enhanced properties for photocatalysis and gas Sensing. J. Phys. Chem. C 2010, 114, 2049–2055. [Google Scholar] [CrossRef]
- Gong, H.; Hu, J.Q.; Wang, J.H.; Ong, C.H.; Zhu, F.R. Nano-crystalline Cu-doped ZnO thin film gas sensor for CO. Sens. Actuators B Chem. 2006, 115, 247–251. [Google Scholar] [CrossRef]
- Wei, S.; Yu, Y.; Zhou, M. CO gas sensing of Pd-doped ZnO nanofibers synthesized by electrospinning method. Mater. Lett. 2010, 64, 2284–2286. [Google Scholar] [CrossRef]
- Hubner, M.; Koziej, D.; Grunwaldt, J.D.; Weimar, U.; Barsan, N. An Au clusters related spill-over sensitization mechanism in SnO2-based gas sensors identified by operando HERFD-XAS, work function changes, DC resistance and catalytic conversion studies. Phys. Chem. Chem. Phys. 2012, 14, 13249–13254. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Wagner, T. Interaction of nanostructured metal overlayers with oxide surfaces. Surf. Sci. Rep. 2007, 62, 431–498. [Google Scholar] [CrossRef]
- Li, X.; Feng, W.; Xiao, Y.; Sun, P.; Hu, X.; Shimanoe, K.; Lu, G.; Yamazoe, N. Hollow Zinc oxide microspheres functionalized by Au nanoparticles for gas sensors. RSC Adv. 2014, 4, 28005–28010. [Google Scholar] [CrossRef]
- Min, B.K.; Friend, C.M. Heterogeneous Gold-Based Catalysis for Green Chemistry:Low-Temperature CO Oxidation and Propene Oxidation. Chem. Rev. 2007, 107, 2709–2724. [Google Scholar] [CrossRef] [PubMed]






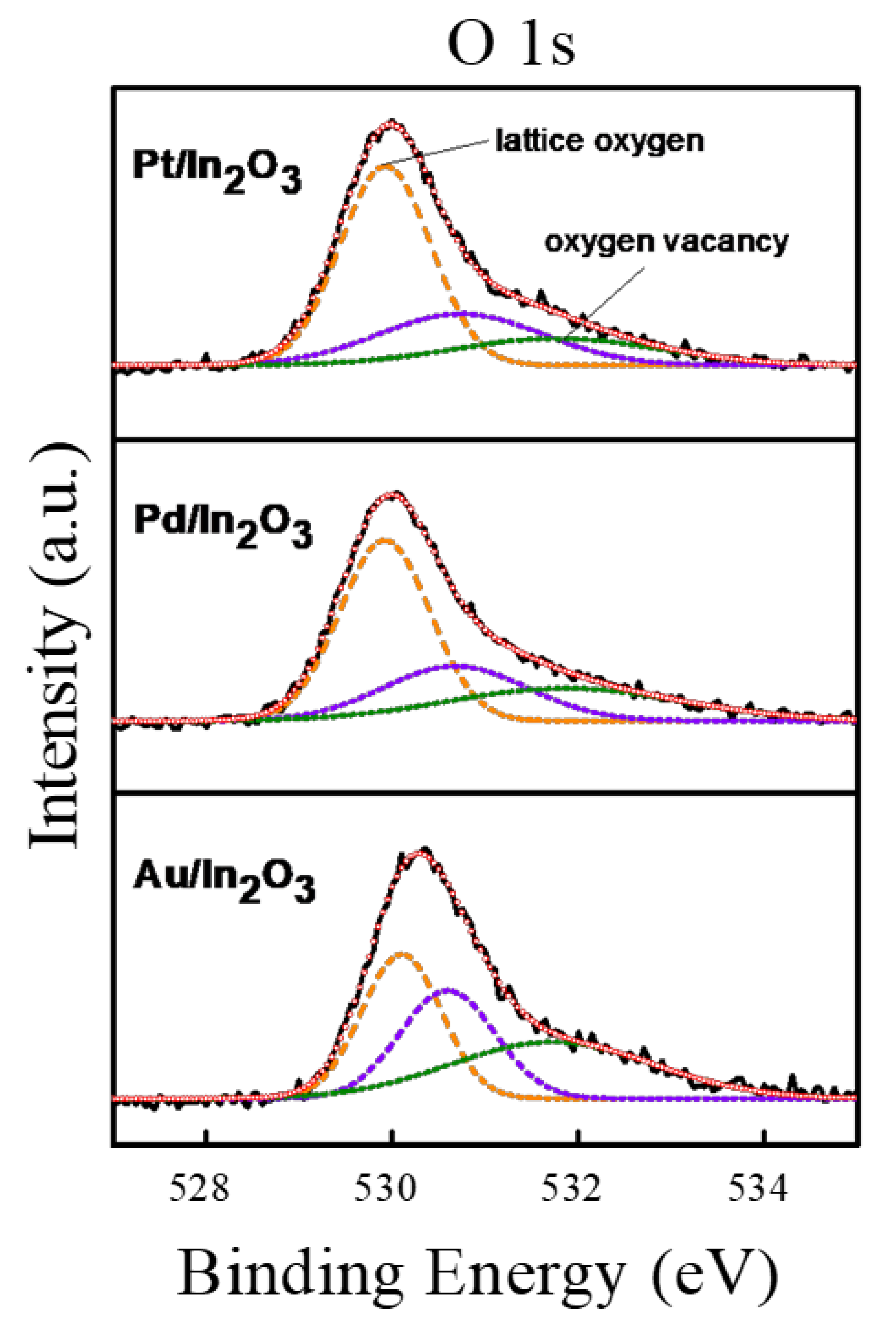



| Gas Sensors | OI (Lattice Oxygen) | OII (Oxygen Vacancy) | ||
|---|---|---|---|---|
| Peak (eV) | Percentage (%) | Peak (eV) | Percentage (%) | |
| Au/In2O3 | 530.1 | 35 | 530.6 | 65 |
| Pd/In2O3 | 529.9 | 51 | 530.7 | 49 |
| Pt/In2O3 | 529.9 | 56 | 530.8 | 44 |
| ppm | Pt/In2O3 | Pd/In2O3 | Au/In2O3 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| °C | 1 | 5 | 10 | 50 | 1 | 5 | 10 | 50 | 1 | 5 | 10 | 50 | |
| 50 | 1.00 | 1.06 | 1.07 | 1.75 | 1.2 | 1.9 | 2.20 | 4.5 | 1.31 | 3.87 | 5.59 | 11.49 | |
| 100 | 1.00 | 1.67 | 1.72 | 6.09 | 1.0 | 1.4 | 1.60 | 3.6 | 1.44 | 5.54 | 8.92 | 19.88 | |
| 150 | 1.00 | 1.14 | 1.34 | 1.88 | 1.1 | 1.3 | 1.60 | 3.5 | 1.32 | 5.36 | 7.68 | 13.02 | |
| 200 | 1.00 | 1.16 | 1.41 | 2.24 | 1.3 | 2.2 | 2.60 | 5.2 | 1.39 | 3.41 | 5.12 | 9.58 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, J.; Jung, H.; Shin, H.-S.; Kim, D.-S.; Kim, D.S.; Ju, B.-K.; Chun, M. The Effect of Noble Metals on Co Gas Sensing Properties of In2O3 Nanoparticles. Appl. Sci. 2021, 11, 4903. https://doi.org/10.3390/app11114903
Hwang J, Jung H, Shin H-S, Kim D-S, Kim DS, Ju B-K, Chun M. The Effect of Noble Metals on Co Gas Sensing Properties of In2O3 Nanoparticles. Applied Sciences. 2021; 11(11):4903. https://doi.org/10.3390/app11114903
Chicago/Turabian StyleHwang, JinAh, Hyunsung Jung, Hyo-Soon Shin, Dae-Sung Kim, Dong Soo Kim, Byeong-Kwon Ju, and MyoungPyo Chun. 2021. "The Effect of Noble Metals on Co Gas Sensing Properties of In2O3 Nanoparticles" Applied Sciences 11, no. 11: 4903. https://doi.org/10.3390/app11114903
APA StyleHwang, J., Jung, H., Shin, H.-S., Kim, D.-S., Kim, D. S., Ju, B.-K., & Chun, M. (2021). The Effect of Noble Metals on Co Gas Sensing Properties of In2O3 Nanoparticles. Applied Sciences, 11(11), 4903. https://doi.org/10.3390/app11114903






