Fluoride Exposure and the Effect of Tobacco Smoking on Urinary Fluoride Levels in Primary Aluminum Workers
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Study Site
2.3. Study Participants
2.4. Air Sampling and Analyses
2.5. Urine Sampling and Analyses
2.6. Statistical Analysis
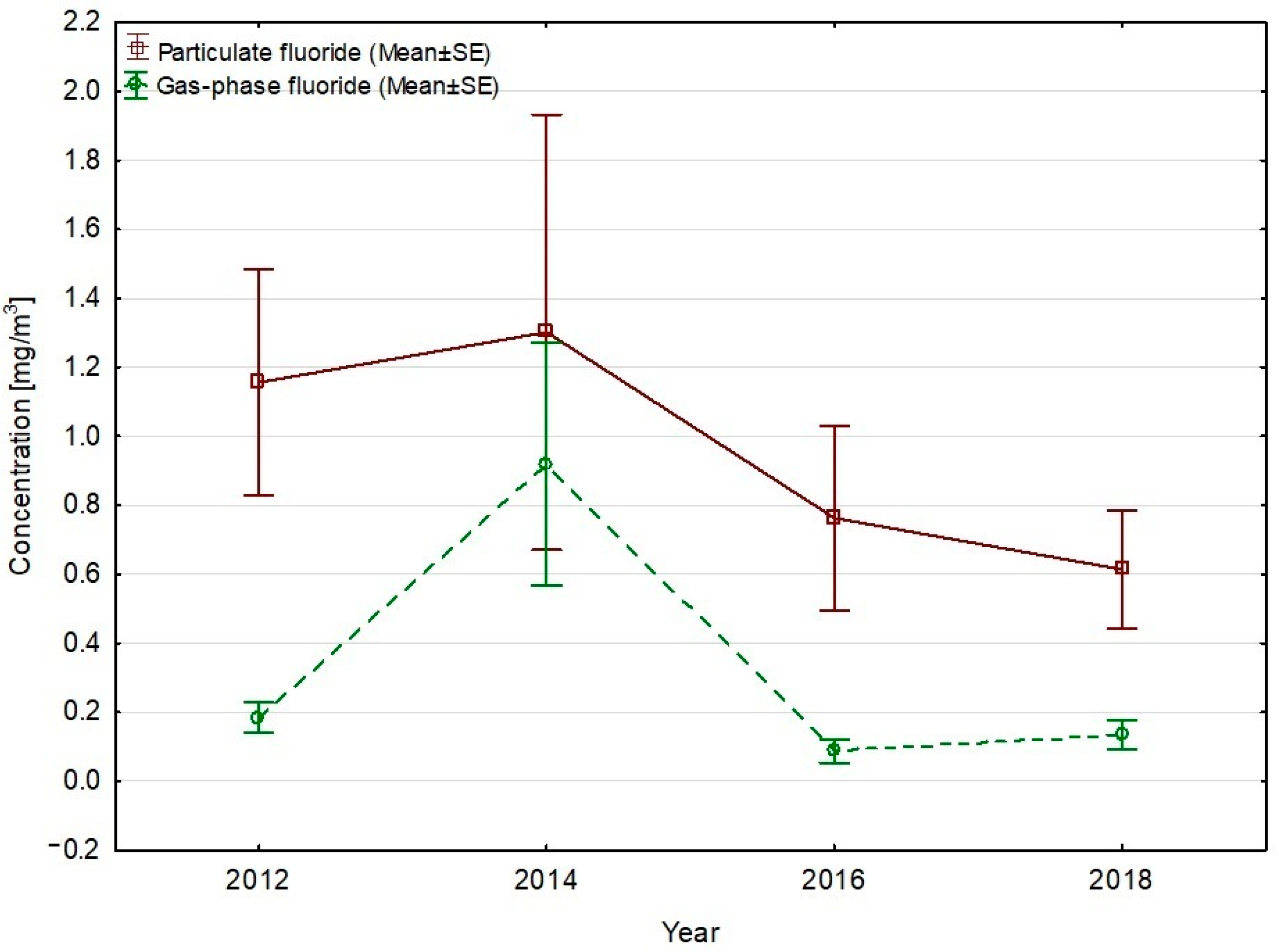
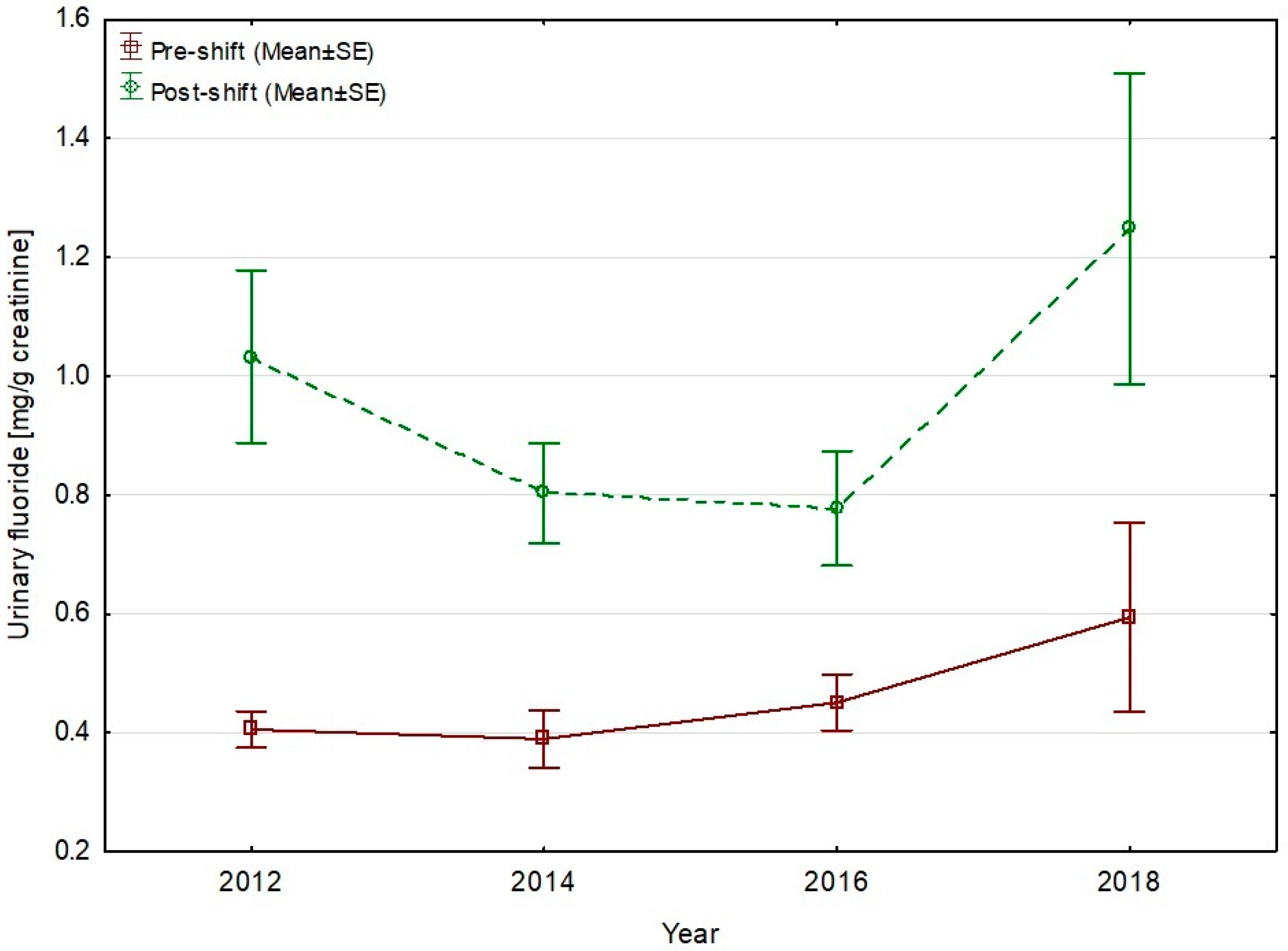
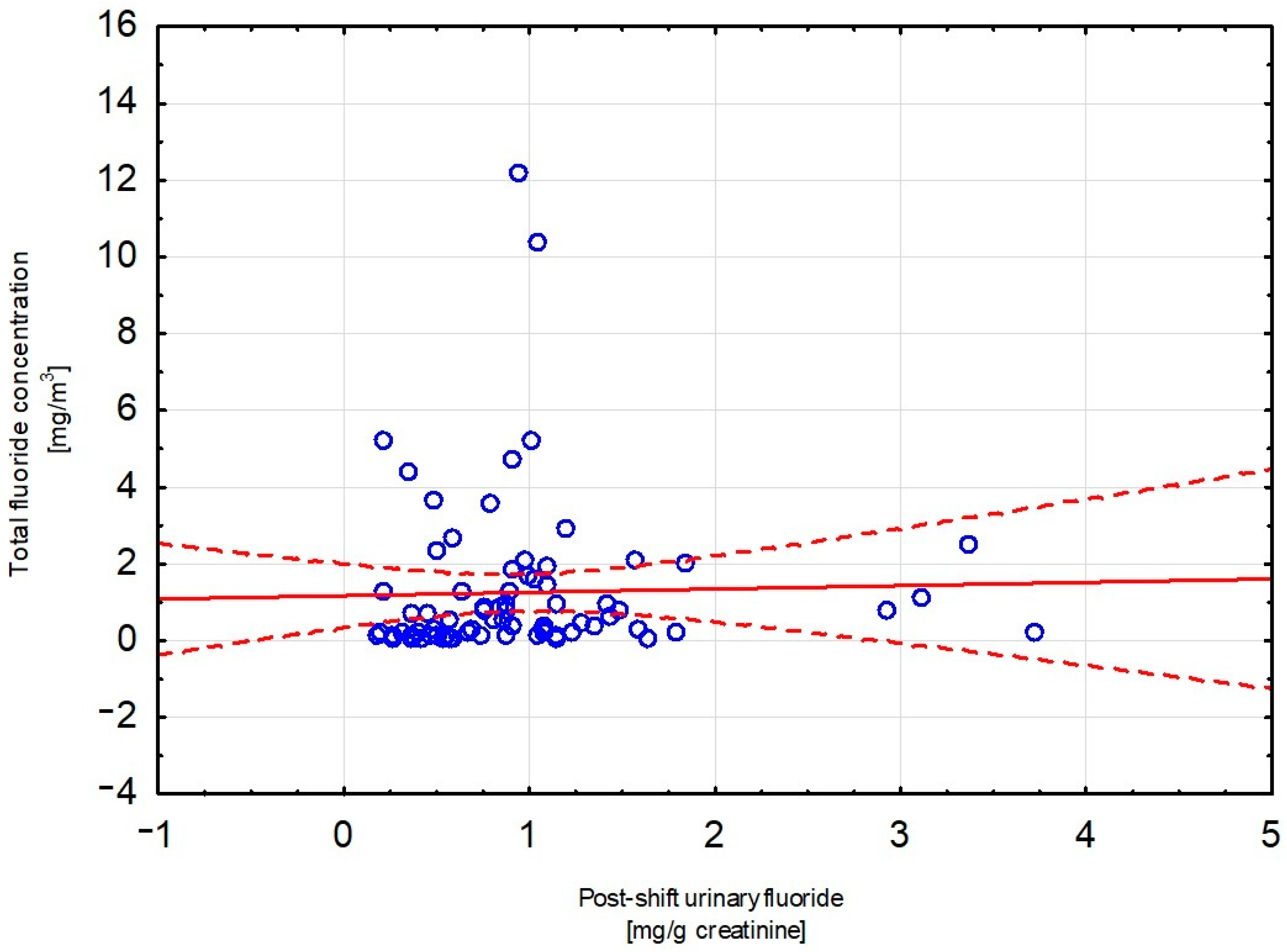
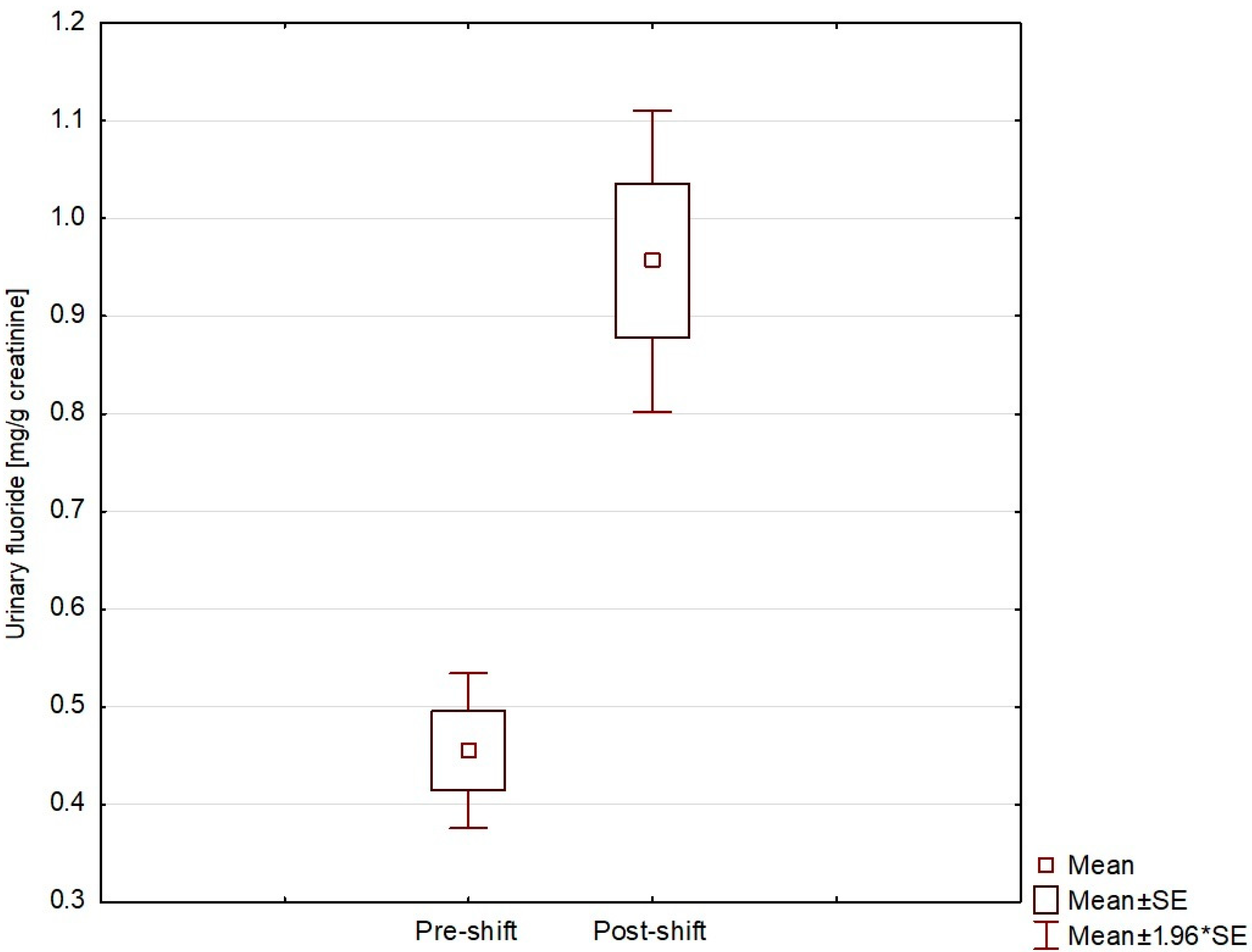
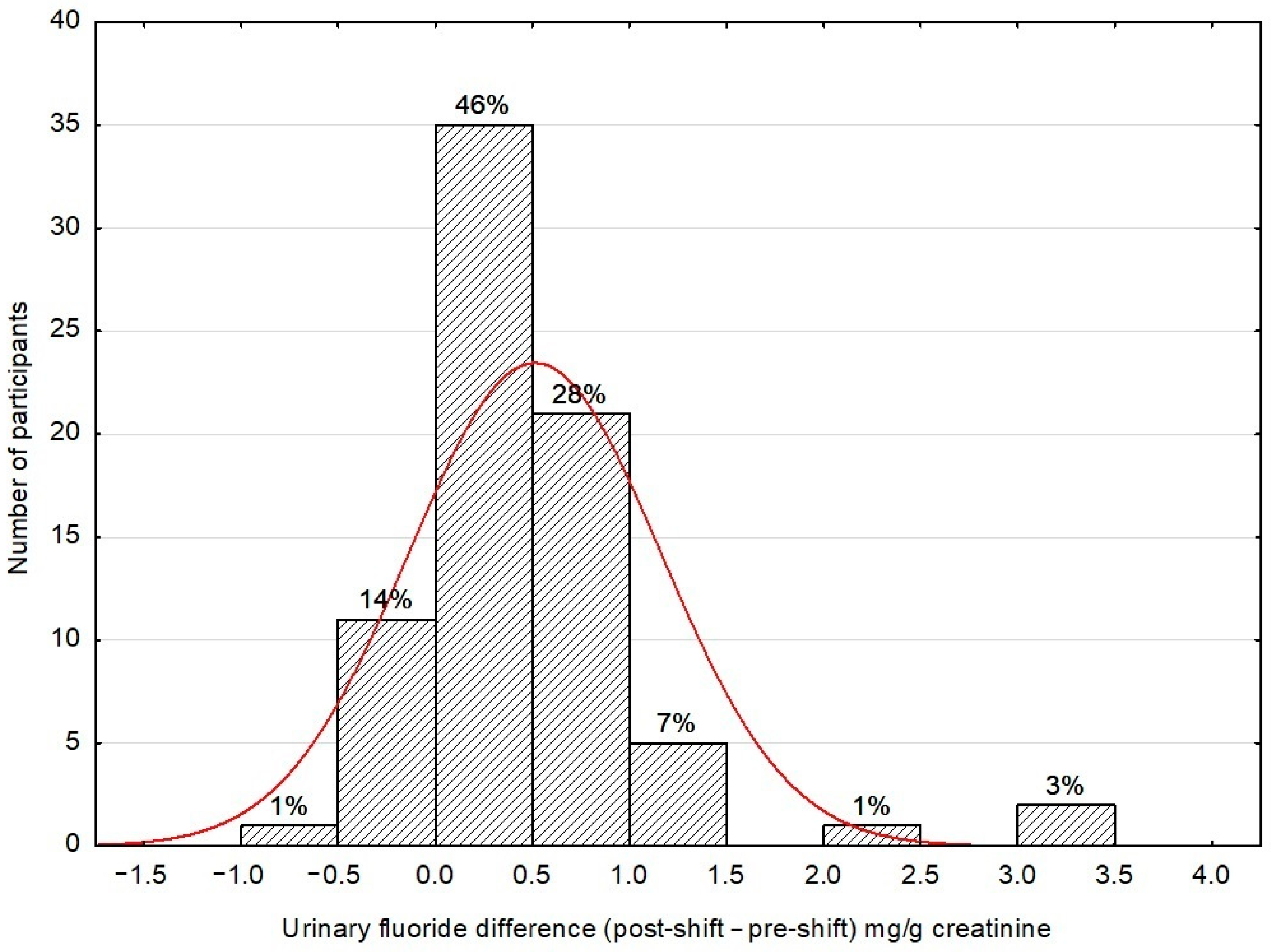
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Participant | Year | Particulate Fluoride Concentration [mg/m3] | Gas-Phase Fluoride (HF) Concentration [mg/m3] | Pre-Shift Urinary Fluoride [mg/g Creatinine] | Post-Shift Urinary Fluoride [mg/g Creatinine] | Tobacco Smoking | Work Position |
|---|---|---|---|---|---|---|---|
| 1. | 2012 | 1.30 | 0.14 | 0.54 | 1.11 | smoker | anode replacement |
| 2. | 2012 | 2.04 | 0.46 | 0.28 | 3.37 | non-smoker | anode replacement |
| 3. | 2012 | 0.39 | 0.15 | 0.43 | 0.57 | non-smoker | crane operator |
| 4. | 2012 | 4.82 | 0.36 | 0.56 | 0.22 | non-smoker | forklift operator |
| 5. | 2012 | 1.92 | 0.18 | 0.60 | 0.99 | non-smoker | anode replacement |
| 6. | 2012 | 0.26 | 0.15 | 0.47 | 1.29 | unknown | forklift operator |
| 7. | 2012 | 0.44 | 0.15 | 0.73 | 1.44 | non-smoker | anode replacement |
| 8. | 2012 | 0.08 | 0.01 | 0.38 | 0.38 | non-smoker | pumping wagon operator |
| 9. | 2012 | 0.03 | 0.04 | 0.25 | 1.05 | smoker | casting of alloys |
| 10. | 2012 | 0.03 | 0.01 | 0.43 | 0.42 | non-smoker | preparation of smelting in the foundry, transport of dross |
| 11. | 2012 | 0.04 | 0.01 | 0.37 | 1.16 | unknown | preparation of smelting in the foundry, transport of dross |
| 12. | 2012 | 0.17 | 0.03 | 0.41 | 1.24 | non-smoker | primary refining |
| 13. | 2012 | 0.10 | 0.02 | 0.36 | 0.56 | smoker | pumping wagon operator |
| 14. | 2012 | 0.21 | 0.09 | 0.36 | 1.09 | smoker | cell start |
| 15. | 2012 | 1.74 | 0.21 | 0.31 | 1.11 | non-smoker | preparation of smelting in the foundry, transport of dross |
| 16. | 2012 | 3.26 | 0.25 | 0.28 | 0.80 | non-smoker | anode replacement |
| 17. | 2012 | 0.63 | 0.23 | 0.46 | 0.85 | non-smoker | cell operator |
| 18. | 2012 | 0.20 | 0.19 | 0.10 | 1.35 | non-smoker | anode replacement |
| 19. | 2012 | 4.34 | 0.85 | 0.43 | 1.01 | smoker | pumping wagon operator |
| 20. | 2012 | 1.12 | 0.14 | 0.36 | 0.65 | non-smoker | cell operator |
| 21. | 2014 | 0.009 | 0.66 | 0.12 | 0.45 | non-smoker | crane operator |
| 22. | 2014 | 10.29 | 0.10 | 0.74 | 1.05 | non-smoker | crane operator |
| 23. | 2014 | 0.009 | 0.95 | 0.39 | 1.15 | non-smoker | forklift operator |
| 24. | 2014 | 0.89 | 0.36 | 0.16 | 0.22 | non-smoker | forklift operator |
| 25. | 2014 | - | - | 0.12 | 0.60 | smoker | anode replacement |
| 26. | 2014 | - | - | 0.33 | 1.64 | smoker | forklift operator |
| 27. | 2014 | 0.009 | 0.13 | 0.18 | 0.89 | smoker | anode replacement |
| 28. | 2014 | 0.009 | 0.22 | 0.25 | 0.50 | non-smoker | anode replacement |
| 29. | 2014 | 0.91 | 2.68 | 0.14 | 0.50 | smoker | cell operator |
| 30. | 2014 | 0.009 | 0.14 | 0.12 | 0.21 | non-smoker | anode transport |
| 31. | 2014 | 0.72 | 0.23 | 0.57 | 0.89 | smoker | crane operator |
| 32. | 2014 | 0.53 | 0.38 | 0.84 | 1.42 | smoker | crane operator |
| 33. | 2014 | 0.009 | 0.31 | 0.63 | 1.09 | smoker | forklift operator |
| 34. | 2014 | 0.34 | 1.31 | 0.55 | 1.00 | non-smoker | anode replacement |
| 35. | 2014 | 5.91 | 6.21 | 0.46 | 0.95 | smoker | anode replacement |
| 36. | 2014 | 0.31 | 0.37 | 0.35 | 0.38 | smoker | cell operator |
| 37. | 2014 | 0.51 | 0.009 | 0.57 | 0.87 | non-smoker | cell operator |
| 38. | 2014 | 2.95 | 1.72 | 0.5 | 0.91 | non-smoker | anode transport |
| 39. | 2014 | 0.009 | 0.74 | 0.43 | 0.76 | smoker | pumping and transport of liquid metal |
| 40. | 2014 | 0.009 | 0.03 | 0.34 | 0.58 | smoker | preparation of smelting in the foundry, transport of dross |
| 41. | 2016 | 0.15 | 0.03 | 0.35 | 0.33 | smoker | crane operator |
| 42. | 2016 | 0.06 | 0.04 | 0.49 | 0.51 | non-smoker | unknown |
| 43. | 2016 | 0.08 | 0.06 | 0.18 | 1.16 | smoker | forklift operator |
| 44. | 2016 | 0.10 | 0.03 | 0.20 | 0.48 | non-smoker | cell operator |
| 45. | 2016 | 4.36 | 0.04 | 0.33 | 0.35 | smoker | anode replacement |
| 46. | 2016 | 0.80 | 0.04 | 0.82 | 0.77 | non-smoker | cell operator |
| 47. | 2016 | 2.67 | 0.23 | 0.32 | 1.20 | non-smoker | anode replacement |
| 48. | 2016 | 0.009 | 0.05 | 0.56 | 0.55 | non-smoker | pumping wagon operator |
| 49. | 2016 | 0.22 | 0.009 | 0.27 | 0.70 | smoker | pumping wagon operator |
| 50. | 2016 | 0.07 | 0.091 | 0.26 | 0.41 | non-smoker | preparation of smelting in the foundry, transport of dross |
| 51. | 2016 | 0.71 | 0.082 | 0.50 | 1.50 | smoker | cell operator |
| 52. | 2016 | 1.87 | 0.15 | 0.48 | 1.85 | smoker | cell operator |
| 53. | 2016 | 2.02 | 0.65 | 0.85 | 0.60 | smoker | forklift operator |
| 54. | 2016 | 0.009 | 0.009 | 0.75 | 0.43 | non-smoker | crane operator |
| 55. | 2016 | 0.37 | 0.009 | 0.47 | 0.91 | non-smoker | anode replacement |
| 56. | 2016 | 0.66 | 0.07 | 0.36 | 0.89 | smoker | anode replacement |
| 57. | 2016 | 0.009 | 0.009 | 0.21 | 0.37 | non-smoker | preparation of smelting in the foundry, transport of dross |
| 58. | 2016 | 0.19 | 0.03 | 0.54 | 0.68 | smoker | pumping wagon operator |
| 59. | 2016 | 0.14 | 0.03 | 0.63 | 1.08 | non-smoker | pumping wagon operator |
| 60. | 2018 | 0.07 | 0.06 | 0.23 | 0.74 | smoker | anode replacement |
| 61. | 2018 | 0.11 | 0.00 | 0.20 | 0.18 | non-smoker | forklift operator |
| 62. | 2018 | 2.24 | 0.07 | 0.51 | 0.51 | smoker | anode replacement |
| 63. | 2018 | 0.24 | 0.04 | 2.42 | 1.60 | non-smoker | cell start |
| 64. | 2018 | 1.92 | 0.17 | 0.19 | 1.57 | smoker | cell operator |
| 65. | 2018 | 0.91 | 0.64 | 0.48 | 1.03 | non-smoker | measurement technician |
| 66. | 2018 | 0.19 | 0.02 | 0.22 | 0.41 | non-smoker | pumping and transport of liquid metal |
| 67. | 2018 | 0.12 | 0.04 | 0.35 | 3.73 | smoker | pumping and transport of liquid metal |
| 68. | 2018 | 0.02 | 0.01 | 0.20 | 0.28 | non-smoker | preparation of smelting in the foundry, transport of dross |
| 69. | 2018 | 0.37 | 0.104 | 0.36 | 0.81 | smoker | forklift operator |
| 70. | 2018 | 0.13 | 0.08 | 0.45 | 0.41 | non-smoker | anode replacement |
| 71. | 2018 | 0.92 | 0.31 | 0.22 | 0.90 | smoker | cell operator |
| 72. | 2018 | 1.68 | 0.12 | 0.64 | 0.91 | smoker | anode replacement |
| 73. | 2018 | 0.73 | 0.38 | 0.89 | 3.12 | non-smoker | cell start |
| 74. | 2018 | 0.06 | 0.03 | 0.18 | 0.28 | smoker | preparation of smelting in the foundry, transport of dross |
| 75. | 2018 | 0.19 | 0.02 | 0.47 | 1.80 | smoker | pumping and transport of liquid metal |
| 76. | 2018 | 0.53 | 0.20 | 2.09 | 2.93 | smoker | pumping and transport of liquid metal |
References
- International Aluminium Institute. Available online: http://www.world-aluminium.org/statistics/ (accessed on 1 September 2020).
- Berdowski, J.; van der Most, P.; Mulder, W.; Bloos, J.P.; Payna, J.M.; Rentz, O.; Oertel, D.; Woodfield, M.; Pulles, T.; Appelman, W. Aluminium production. In 2016 EMEP/EEA Emission Inventory Guidebook; European Environment Agency: Copenhagen, Denmark, 2016. [Google Scholar]
- Kvande, H. The aluminium smelting process. J. Occup. Environ. Med. 2014, 56 (Suppl. S5), S2–S4. [Google Scholar] [CrossRef] [PubMed]
- European Chemicals Agency. Available online: https://echa.europa.eu/documents/10162/c53c81ed-9e4e-4cb7-9930-f22ca736708c (accessed on 9 April 2020).
- Ranjan, R.; Ranjan, A. Fluoride Toxicity in Animals, 1st ed.; Springer International Publishing: New York, NY, USA, 2015; pp. 11–20. [Google Scholar]
- Susheela, A.K.; Mondai, N.K.; Singh, A. Exposure to fluoride in smelter workers in a primary aluminium industry in India. Int. J. Occup. Environ. Med. 2013, 4, 61–72. [Google Scholar] [PubMed]
- Villa, A.; Anabalon, M.; Zohouri, V.; Maguire, A.; Franco, A.M.; Rugg-Gunn, A. Relationships between fluoride intake, urinary fluoride excretion and fluoride retention in children and adults: An analysis of available data. Caries Res. 2010, 44, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Fluorides, Hydrogen Fluoride, and Fluorine; U.S. Department of Health and Human Services, Public Health Service: Atlanta, GA, USA, 2003.
- Seixas, N.S.; Cohen, M.; Zevenbergen, B.; Cotey, M.; Carter, S.; Kaufam, J. Urinary fluoride as an exposure index in aluminium smelting. J. Sci. Occup. Environ. Health Saf. 2010, 61, 89–94. [Google Scholar]
- Aylward, L.L.; Hays, S.M.; Vezina, A.; Deveau, M.; St-Amand, A.; Nong, A. Biomonitoring Equivalents for interpretation of urinary fluoride. Regul. Toxicol. Pharmacol. 2015, 72, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Idowu, O.S.; Azevedo, L.B.; Valentine, R.A.; Swan, J.; Vasantavada, P.V.; Maguire, A. The use of urinary fluoride excretion to facilitate monitoring fluoride intake: A systematic scoping review. PLoS ONE 2019, 14, e0222260. [Google Scholar] [CrossRef]
- Schwarz, M. Monitoring of fluorine in foodstuffs within the district of Žiar nad Hronom in 1986–1997. Food Res. Inst. Bratisl. 1998, 37, 53–64. [Google Scholar]
- Koç, E.; Karademir, B.; Soomro, N.; Uzun, F. The Effects, both separate and interactive, of smoking and tea consumption on urinary fluoride levels. Fluoride 2018, 51, 84–96. [Google Scholar]
- Kocyigit, A.; Erel, O.; Gur, S. Effects of tobacco smoking on plasma selenium, zinc, copper and iron concentrations and related antioxidative enzyme activities. Clin. Biochem. 2001, 34, 629–633. [Google Scholar] [CrossRef]
- Özden, T.A.; Gökçay, G.; Ertem, H.V.; Süoğlu, Ö.D.; Kılıç, A.; Sökücü, S. Elevated hair levels of cadmium and lead in school children exposed to smoking and in highways near schools. Clin. Biochem. 2007, 40, 52–56. [Google Scholar] [CrossRef]
- Slov-Lex Legislative and Information Portal. Ordinance No. 355/2006 on Protection of Employees against Risks due to Exposure to Chemical Factors at Work. Available online: https://www.slov-lex.sk/pravne-predpisy/SK/ZZ/2006/355/20201001 (accessed on 26 December 2020).
- National Institute for Occupational Safety and Health. Fluorides, aerosol and gas by ISE. In 1994 Method: 7902, Issue 2, 4th ed.; NIOSH: Washington, DC, USA, 1994. [Google Scholar]
- National Institute for Occupational Safety and Health. Fluoride in Urine. In 1994 Method: 8308, Issue 2; NIOSH: Washington, DC, USA, 1994. [Google Scholar]
- Barr, D.B.; Wilder, L.C.; Caudill, S.P.; Gonzalez, A.J.; Needham, L.L.; Pirkle, J.L. Urinary creatinine concentrations in the U.S. population: Implications for urinary biologic monitoring measurements. Environ. Health Perspect. 2005, 113, 192–200. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Occupational Safety and Health. Occupational Exposure to Inorganic Fluorides; NIOSH: Washington, DC, USA, 1976; Publication No. 76103. [Google Scholar]
- National Institute for Occupational Safety and Health. Occupational Exposure to Hydrogen Fluoride; NIOSH: Washington, DC, USA, 1976; Publication No. 76143. [Google Scholar]
- Ehrnebo, M.; Ekstrand, J. Occupational fluoride exposure and plasma fluoride levels in man. Int. Arch. Occup. Environ. Health 1986, 58, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Aarhaug, T.A.; Ratvik, A.P. Aluminium Primary Production Off-Gas Composition and Emissions: An Overview. JOM 2019, 71, 2966–2977. [Google Scholar] [CrossRef]
- Occupational Safety and Health Administration (OSHA). Available online: https://www.osha.gov/annotated-pels/table-z-1 (accessed on 9 December 2020).
- European Commission Website. Available online: https://ec.europa.eu/health/scientific_committees/opinions_layman/fluoridation/en/l-3/4.htm (accessed on 10 December 2020).
- Slovak Hydrometeorological Institute. Available online: http://www.shmu.sk/File/oko/rocenky/SHMU_Air_Pollution_in_the_SR_2019_v1.pdf (accessed on 10 December 2020).
- Pierre, F.; Baruthio, F.; Diebold, F.; Biette, P. Effect of different exposure compounds on urinary kinetics of aluminium and fluoride in industrially exposed workers. Occup. Environ. Med. 1995, 52, 396–403. [Google Scholar] [CrossRef]
- Kono, K.; Yoshida, Y.; Yamagata, H.; Watanabe, M.; Shibuya, Y.; Doi, K. Urinary fluoride monitoring of industrial hydrofluoric acid exposure. Environ. Res. 1987, 42, 415–520. [Google Scholar] [CrossRef]
- Waugh, D.T.; Godfrey, M.; Limeback, H.; Potter, W. Black Tea Source, Production, and Consumption: Assessment of Health Risks of Fluoride Intake in New Zealand. J. Environ. Public Health 2017, 2017, 1–27. [Google Scholar] [CrossRef]
- Whitford, G.M. The Metabolism and Toxicity of Fluoride. Monogr. Oral Sci. 1996, 16, 1–153. [Google Scholar] [CrossRef]
- World Health Organization. Basic Methods for Assessing Renal Fluoride Excretion in Community Prevention Programmes for Oral Health; World Health Organization: Geneva, Switzerland, 2014; Available online: https://apps.who.int/iris/handle/10665/112662 (accessed on 6 December 2020).
- Rees, D.; Rama, D.B.; Yousefi, V. Fluoride in workplace air and in urine of workers concentrating fluorspar. Am. J. Ind. Med. 1990, 17, 311–320. [Google Scholar] [CrossRef]
- Zohouri, F.V.; Swinbank, C.M.; Maguire, A.; Moynihan, P.J. Is the fluoride/creatinine ratio of a spot urine sample indicative of 24-h urinary fluoride? Community Dent. Oral. Epidemiol. 2006, 34, 130–138. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Available online: https://www.cdc.gov/niosh/docs/2003-154/pdfs/8308.pdf (accessed on 8 December 2020).
- Lauwerys, R.R.; Hoet, P. Industrial Chemical Exposure Guidelines for Biological Monitoring, 3rd ed.; CRC Press, Inc.: Boca Raton, FL, USA, 2001; pp. 97–102. [Google Scholar]
- National Research Council. Fluoride in Drinking Water: A Scientific Review of EPA’s Standards, 1st ed.; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar] [CrossRef]
- Laisalmi, M.; Soikkeli, A.; Kokki, H.; Markkanen, H.; Yli-Hankala, A.; Rosenberg, P.; Lindgren, L. Fluoride metabolism in smokers and non-smokers following enflurane anaesthesia. Br. J. Anaesth. 2003, 91, 800–804. [Google Scholar] [CrossRef][Green Version]
- Radon, K.; Nowak, D.; Szadkowski, D. Lack of Combined Effects of Exposure and Smoking on Respiratory Health in Aluminium Potroom Workers. Occup. Environ. Med. 1999, 56, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Liu, K.; Ran, L.; Pan, X. Fluoride burden of aluminium plant workers. Iran. J. Public Health 2015, 44, 583–585. [Google Scholar] [PubMed]

| Number of Participants | Work Position |
|---|---|
| n = 7 | Crane operator |
| n = 10 | Forklift operator |
| n = 2 | Anode transport |
| n = 1 | Casting of alloys |
| n = 8 | Preparation of smelting in the foundry, transport of dross |
| n = 11 | Cell operator |
| n = 19 | Anode replacement |
| n = 7 | Pumping wagon operator |
| n = 1 | Primary refining |
| n = 3 | Cell start |
| n = 1 | Measurement technician |
| n = 5 | Pumping and transport of liquid metal |
| Group | Statistical Indicator | Particulate Fluoride Concentration [mg/m3] | Gas-Phase Fluoride (HF) Concentration [mg/m3] | Pre-Shift Urinary Fluoride [mg/g Creatinine] | Post-Shift Urinary Fluoride [mg/g Creatinine] |
|---|---|---|---|---|---|
| All participants | Mean ± SD | 0.966 ± 1.658 | 0.327 ± 0.809 | 0.455 ± 0.353 | 0.957 ± 0.686 |
| Median | 0.285 | 0.112 | 0.385 | 0.880 | |
| Range | 0.009–10.290 | 0–6.210 | 0.100–2.420 | 0.180–3.730 | |
| n = 74 1 | n = 74 1 | n = 76 | n = 76 | ||
| Smokers | Mean ± SD | 0.992 ± 1.427 | 0.438 ± 1.143 | 0.441 ± 0.338 | 1.050 ± 0.708 |
| Median | 0.370 | 0.104 | 0.360 | 0.890 | |
| Range | 0.009–5.910 | 0.009–6.210 | 0.120–2.090 | 0.280–3.730 | |
| n = 33 | n = 33 | n = 35 | n = 35 | ||
| Non-smokers | Mean ± SD | 0.985 ± 1.879 | 0.245 ± 0.368 | 0.469 ± 0.379 | 0.859 ± 0.677 |
| Median | 0.240 | 0.140 | 0.430 | 0.770 | |
| Range | 0.009–10.290 | 0–1.720 | 0.100–2.420 | 0.180–3.370 | |
| n = 39 | n = 39 | n = 39 | n = 39 |
| Parameter | Parameter |
|---|---|
| Post-Shift Urinary Fluoride [mg/g creatinine] | |
| Particulate fluoride concentration [mg/m3] | 0.039 (p = 0.74) |
| Gas-phase fluoride concentration [mg/m3] | 0.005 (p = 0.96) |
| Total fluoride concentration [mg/m3] | 0.029 (p = 0.81) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwarz, M.; Salva, J.; Vanek, M.; Rasulov, O.; Darmová, I. Fluoride Exposure and the Effect of Tobacco Smoking on Urinary Fluoride Levels in Primary Aluminum Workers. Appl. Sci. 2021, 11, 156. https://doi.org/10.3390/app11010156
Schwarz M, Salva J, Vanek M, Rasulov O, Darmová I. Fluoride Exposure and the Effect of Tobacco Smoking on Urinary Fluoride Levels in Primary Aluminum Workers. Applied Sciences. 2021; 11(1):156. https://doi.org/10.3390/app11010156
Chicago/Turabian StyleSchwarz, Marián, Jozef Salva, Miroslav Vanek, Oqil Rasulov, and Ivana Darmová. 2021. "Fluoride Exposure and the Effect of Tobacco Smoking on Urinary Fluoride Levels in Primary Aluminum Workers" Applied Sciences 11, no. 1: 156. https://doi.org/10.3390/app11010156
APA StyleSchwarz, M., Salva, J., Vanek, M., Rasulov, O., & Darmová, I. (2021). Fluoride Exposure and the Effect of Tobacco Smoking on Urinary Fluoride Levels in Primary Aluminum Workers. Applied Sciences, 11(1), 156. https://doi.org/10.3390/app11010156





