The Effect of Pesticides on the Tomato Bacterial Speck Disease Pathogen Pseudomonas Syringae pv. Tomato
Abstract
1. Introduction
2. Materials and Methods
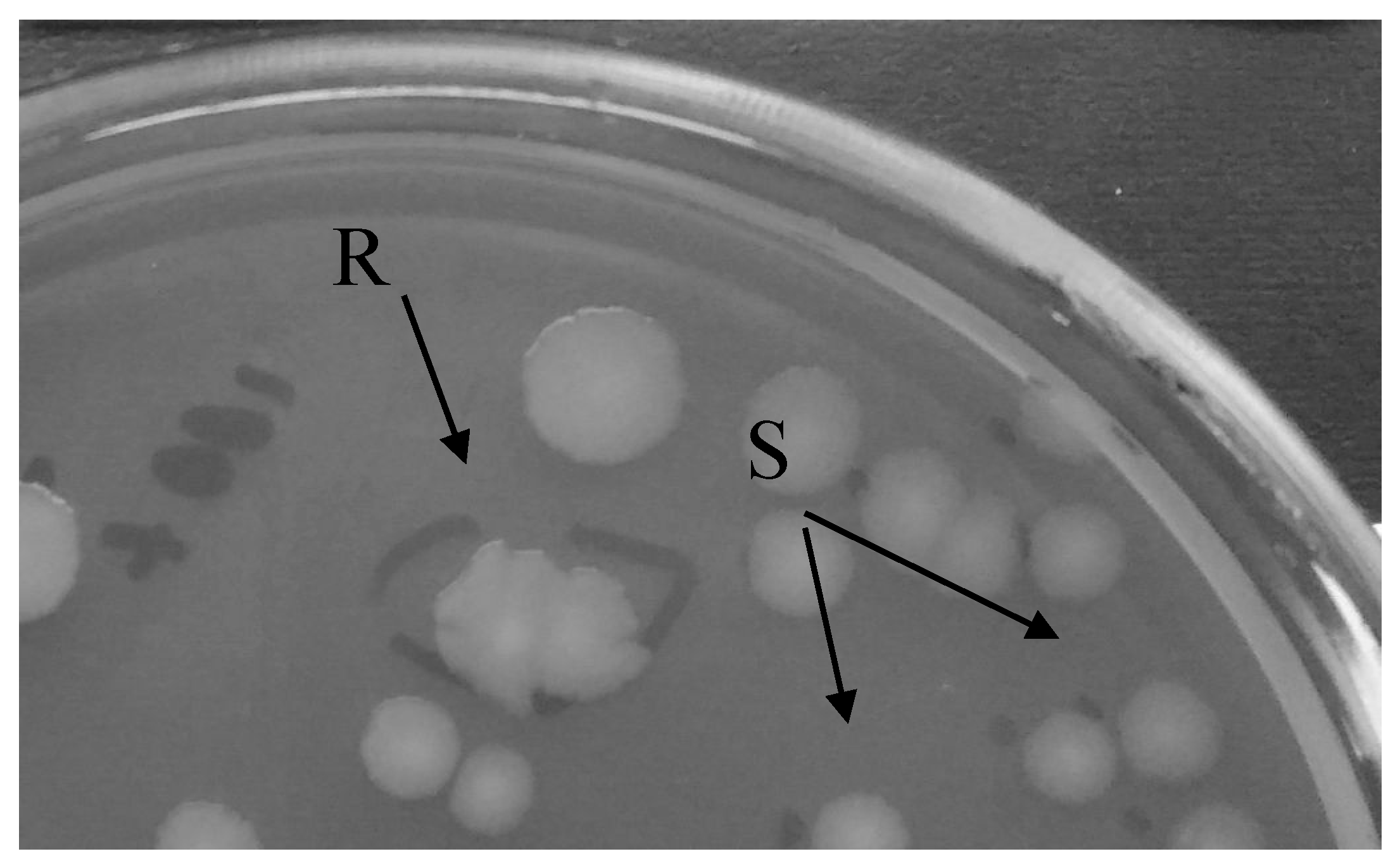
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Eichenlaub, R.; Gartemann, K.-H. The Clavibacter michiganensis subspecies: Molecular investigation of Gram-positive bacterial plant pathogens. Annu. Rev. Phytopathol. 2011, 49, 445–464. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.; Gao, Z.; Yang, W. Breeding for Resistance to Tomato Bacterial Diseases in China: Challenges and Prospects. Hortic. Plant J. 2018, 4, 193–207. [Google Scholar] [CrossRef]
- Stall, R.E.; Jones, J.B.; Minsavage, G.V. Durability of resistance in tomato and pepper to Xanthomonads causing bacterial spot. Annu. Rev. Phytopathol. 2009, 47, 265–284. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, D.R.; Kumar, K.; Birah, A. Eco-friendly management modules for bacterial wilt (Ralstonia solanacearum) of tomato for protected cultivation in a tropical island ecosystem. Biol. Agric. Hortic. 2014, 30, 219–227. [Google Scholar] [CrossRef]
- Kolomiets, J.; Grygoryuk, I.; Butsenko, L. Bacterial diseases of tomato plants in terms of open and covered growing of Ukraine. Ann. Agrar. Sci. 2017, 15, 213–216. [Google Scholar] [CrossRef]
- Hollomon, D.W. Fungicide resistance: Facing the challenge—A review. Plant Prot. Sci. 2015, 51, 170–176. [Google Scholar] [CrossRef]
- Corio-Costet, M.-F.; Dufour, M.-C.; Cigna, J.; Abadie, P.; Chen, W. Diversity and fitness of Plasmopara viticola isolates resistant to QoI fungicides. Eur. J. Plant Pathol. 2011, 129, 315–329. [Google Scholar] [CrossRef]
- Karaoglanidis, G.S.; Thanassoulopoulos, C.C. Phenotypic Instability of Cercospora beticola Sacc. Strains Expressing Resistance to the Sterol Demethylation-Inhibiting (DMI) Fungicide Flutriafol after Cold Exposure. J. Phytopathol. 2002, 150, 692–696. [Google Scholar] [CrossRef]
- Asad, M.; Lavoie, M.; Song, H.; Jin, Y.; Fu, Z.; Qian, H. Interaction of chiral herbicides with soil microorganisms, algae and vascular plants. Sci. Total Environ. 2017, 580, 1287–1299. [Google Scholar] [CrossRef]
- Gu, L.; Bai, Z.; Jin, B.; Hu, Q.; Wang, H.; Zhuang, G.; Zhang, H. Assessing the impact of fungicide enostroburin application on bacterial community in wheat phyllosphere. J. Environ. Sci. 2010, 22, 134–141. [Google Scholar] [CrossRef]
- Zhang, B.; Bai, Z.; Hoefel, D.; Tang, L.; Wang, X.; Li, B.; Li, Z.; Zhuang, G. The impacts of cypermethrin pesticide application on the non-target microbial community of the pepper plant phyllosphere. Sci. Total Environ. 2009, 407, 1915–1922. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xu, Z.; Teng, Y.; Christie, P.; Wang, J.; Ren, W.; Luo, Y.; Li, Z. Non-target effects of repeated chlorothalonil application on soil nitrogen cycling: The key functional gene study. Sci. Total Environ. 2016, 543, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Bull, S.; Fletcher, K.; Boobis, A.R.; Battershill, J.M. Evidence for genotoxicity of pesticides in pesticide applicators: A review. Mutagenesis 2006, 21, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Buletsa, N.; Butsenko, L.; Pasichnyk, L.; Patyka, V. Physiology of growth Pseudomonas syringae pv. atrofaciens for the effects of pesticides. Mikrobiolohichnyi Zhurnal 2016, 78, 52–60. [Google Scholar] [CrossRef]
- Butsenko, L.; Pasichnyk, L.; Buletsa, N.; Patyka, V. Effect of insecticide Alpha Super on phytopathogenic bacteria Pseudomonas syringae of agrophytocenosis of wheat. Bull. Agric. Sci. 2017, 3, 18–22. [Google Scholar] [CrossRef]
- Lukáčová, M.; Barák, I.; Kazár, J. Role of structural variations of polysaccharide antigens in the pathogenicity of Gram-negative bacteria. Clin. Microbiol. Infect. 2008, 14, 200–206. [Google Scholar] [CrossRef]
- Sułowicz, S.; Cycoń, M.; Piotrowska-Seget, Z. Non-target impact of fungicide tetraconazole on microbial communities in soils with different agricultural management. Ecotoxicology 2016, 25, 1047–1060. [Google Scholar] [CrossRef]
- State Register of Pesticides and Agrochemicals Permitted for Use in Ukraine. (In Ukr.). Available online: https://data.gov.ua/dataset/389ddb5a-ac73-44bb-9252-f899e4a97588 (accessed on 27 February 2020).
- Dankevych, L.A.; Gnatiuk, T.T.; Huliaieva, H.B.; Tokovenko, I.P.; Kalinichenko, A.V. Express diagnostics of phytopathogenic bacteria and phytoplasmas in agrophytocenosis. In Guidelines; Suszanowich, D., Patyka, V., Eds.; Wyd-wo I Drukarnia Swietego Krzyza: Opole, Poland, 2019; Available online: https://www.researchgate.net/publication/335692227_EXPRESS_DIAGNOSTICS_OF_PHYTOPATHOGENIC_BACTERIA_AND_PHYTOPLASMAS_IN_AGROPHYTOCENOSES (accessed on 10 November 2019).
- Patyka, V.; Buletsa, N.; Pasichnyk, L.; Zhitkevich, N.; Kalinichenko, A.; Gnatiuk, T.; Butsenko, L. Specifics of pesticides effects on the phytopathogenic bacteria. Ecol. Chem. Eng. S 2016, 23, 311–331. [Google Scholar] [CrossRef]
- Van den Broek, D.; Bloemberg, G.V.; Lugtenberg, B. The role of phenotypic variation in rhizosphere Pseudomonas bacteria. Environ. Microbiol. 2005, 7, 1686–1697. [Google Scholar] [CrossRef]
- Esson, D.; Mather, A.E.; Scanlan, E.; Gupta, S.; De Vries, S.P.W.; Bailey, D.; Harris, S.R.; McKinley, T.J.; Méric, G.; Berry, S.K.; et al. Genomic variations leading to alterations in cell morphology of Campylobacter spp. Sci. Rep. 2016, 6, 38303. [Google Scholar] [CrossRef]
- Seaton, S.C.; Silby, M.W.; Levy, S.B. Pleiotropic effects of GacA on Pseudomonas fluorescens Pf0-1 in vitro and in soil. Appl. Environ. Microbiol. 2013, 79, 5405–5410. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wong, C.S. Environmental fate processes and biochemical transformations of chiral emerging organic pollutants. Anal. Bioanal. Chem. 2006, 386, 544–558. [Google Scholar] [CrossRef] [PubMed]
- Maltby, L.; Brock, T.; van den Brink, P. Fungicide Risk Assessment for Aquatic Ecosystems: Importance of Interspecific Variation, Toxic Mode of Action, and Exposure Regime. Environ. Sci. Technol. 2009, 43, 7556–7563. [Google Scholar] [CrossRef] [PubMed]
- Ono, T.; Norimatsu, M.; Yoshimura, H. Induction of chromosome aberrations by pyrimethamine in cultured Chinese hamster (CHL) cells. Mutat. Res. Lett. 1994, 323, 197–201. [Google Scholar] [CrossRef]
- Ozkan, O.; Ustuner, O. Investigations about genotoxicity of deltamethrin. Kafkas Univ. Vet Fak Derg. 2012, 18, 69–74. [Google Scholar] [CrossRef]
- Grisolia, C. A comparison between mouse and fish micronucleus test using cyclophosphamide, mitomycin C and various pesticides. Mutat. Res. 2002, 518, 145–150. [Google Scholar] [CrossRef]
- Oztas, E.; Ulus, B.; Özhan, G. In Vitro Investigation on the Toxic Potentials of Commonly Used Synthetic Pyrethroids, Especially Esbiothrin. Appl. In Vitro Toxicol. 2015, 1, 302–307. [Google Scholar] [CrossRef]
- Quest, J.A.; Hamernik, K.L.; Engler, R.; Burnam, W.L.; Fenner-Crisp, P.A. Evaluation of the carcinogenic potential of pesticides. 3. Aliette. Regul. Toxicol. Pharmacol. 1991, 14, 3–11. [Google Scholar] [CrossRef]
- Fosetyl. EU Pesticides Database. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/?event=activesubstance.detail&language=EN&selectedID=1419 (accessed on 10 November 2019).
- Curutiu, C.; Lazar, V.; Chifiriuc, M. Pesicides and antimicrobial resistance: From environmental compartments to animal and human infections. In New Pesticides and Soil Sensors; Grumezescu, A.M., Ed.; Academic Press: Bucharest, Romania, 2017; pp. 373–392. [Google Scholar] [CrossRef]
- Rangasamy, K.; Athiappan, M.; Devarajan, N.; Parray, J.A. Emergence of multi drug resistance among soil bacteria exposing to insecticides. Microb. Pathog. 2017, 105, 153–165. [Google Scholar] [CrossRef]
- Viljoen, A.; Herrmann, J.-L.; Onajole, O.; Stec, J.; Kozikowski, A.; Kremer, L. Controlling extra- and intramacrophagic Mycobacterium abscessus by targeting mycolic acid transport. Front. Cell. Infect. Microbiol. 2017, 7, 388. [Google Scholar] [CrossRef]
- Drenkard, E.; Ausubel, F.M. Pseudomonas biofilm formation and antibiotic resistance are linked to phenotypic variation. Nature 2002, 416, 740–743. [Google Scholar] [CrossRef] [PubMed]
- Kirisits, M.J.; Prost, L.; Starkey, M.; Parsek, M.R. Characterization of colony morphology variants isolated from Pseudomonas aeruginosa biofilms. Appl. Environ. Microbiol. 2005, 71, 4809–4821. [Google Scholar] [CrossRef] [PubMed]
- Malone, J.; Jaeger, T.; Spangler, C.; Ritz, D.; Spang, A.; Arrieumerlou, C.; Kaever, V.; Landmann, R.; Jenal, U. YfiBNR mediates cyclic di-GMP dependent small colony variant formation and persistence in Pseudomonas aeruginosa. PLoS Pathog. 2010, 6, e1000804. [Google Scholar] [CrossRef] [PubMed]
- Clark, A. The Occupational Opportunist: An Update on Erysipelothrix rhusiopathiae Infection, Disease Pathogenesis, and Microbiology. Clin. Microbiol. Newsl. 2015, 37, 143–151. [Google Scholar] [CrossRef]
- Dankevych, L.; Leonova, N.; Dragovoz, I.; Patyka, V.; Kalinichenko, A.; Włodarczyk, P.; Włodarczyk, B. The synthesis of plant growth stimulators by phytopathogenic bacteria as factor of pathogenicity. Appl. Ecol. Environ. Res. 2018, 16, 1581–1593. [Google Scholar] [CrossRef]


| Pesticide | Pesticide Concentration, mg/L | Frequency of Occurrence of StrR Mutants of Strains, ×10−8 | |
|---|---|---|---|
| P. syringae pv. tomato IZ28 | P. syringae pv. tomato IZ46 | ||
| Aluminium phosethyl | 400.0 | – | – |
| 40.0 | 1.0 ± 0.02 | 1.0 ± 0.07 | |
| 4.0 | 1.0 ± 0.1 | 2.0 ± 0.1 | |
| Deltamethrin | 2.5 | 10.0 ± 0.2 * | 8.0 ± 0.1 * |
| 0.25 | 15.0 ± 0.3 * | 12.0 ± 0.4 * | |
| 0.025 | 14.0 ± 0.2 * | 15.0 ± 0.2 * | |
| Control ** | 1.5 ± 0.1 | 2.0 ± 0.1 | |
| Test | StrS (Original Form) P. syringae pv. tomato IZ28 and IZ46 | StrR Mutants P. syringae pv. tomato IZ28 and IZ46 | |
|---|---|---|---|
| Induced by Aluminium Phosethyl | Induced by Deltamethrin | ||
| Gram staining | - | - | - |
| Oxidase | - | - | - |
| Growth on LB Broth | Uniform turbidity | Uniform turbidity | Uniform turbidity |
| Fermentation: | |||
| glucose, mannose, arabinose, sorbitol, inositol | + | + | + |
| glucose (anaerobic), lactose, maltose | - | - | - |
| HR on tobacco | + | + | + |
| Microagglutination reaction with antiserum in dilution: | |||
| 1:20 | + | + | + |
| Pesticide | Pesticide Concentration, mg/L | Frequency of Occurrence of R Forms of Strains | |
|---|---|---|---|
| P. syringae pv. tomato IZ28 | P. syringae pv. tomato IZ46 | ||
| Deltamethrin | 2.5 | 8 × 10−2 * | 3 × 10−2 * |
| 0.25 | 1 × 10−2 * | 1 × 10−2 * | |
| 0.025 | 4 × 10−3 * | 2 × 10−2 * | |
| Control ** | 1 × 10−4 | 1 × 10−4 | |
| Test | S Form P. syringae pv. tomato IZ28 and IZ46 | R Form P. syringae pv. tomato IZ28 and IZ46 | |
|---|---|---|---|
| Spontaneous | Induced by Deltamethrin | ||
| Gram staining | - | - | - |
| Oxidase | - | - | - |
| Growth on LB Broth | Uniform turbidity | Film, sediment | Film, sediment |
| Fermentation: | |||
| glucose, mannose, arabinose, sorbitol, inositol | + | + | + |
| glucose (anaerobic), lactose, maltose | - | - | - |
| HR on tobacco | + | + | + |
| Microagglutination reaction with antiserum in dilution: | |||
| 1:20 | + | + | + |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butsenko, L.; Pasichnyk, L.; Kolomiiets, Y.; Kalinichenko, A. The Effect of Pesticides on the Tomato Bacterial Speck Disease Pathogen Pseudomonas Syringae pv. Tomato. Appl. Sci. 2020, 10, 3263. https://doi.org/10.3390/app10093263
Butsenko L, Pasichnyk L, Kolomiiets Y, Kalinichenko A. The Effect of Pesticides on the Tomato Bacterial Speck Disease Pathogen Pseudomonas Syringae pv. Tomato. Applied Sciences. 2020; 10(9):3263. https://doi.org/10.3390/app10093263
Chicago/Turabian StyleButsenko, Liudmyla, Lidiia Pasichnyk, Yuliia Kolomiiets, and Antonina Kalinichenko. 2020. "The Effect of Pesticides on the Tomato Bacterial Speck Disease Pathogen Pseudomonas Syringae pv. Tomato" Applied Sciences 10, no. 9: 3263. https://doi.org/10.3390/app10093263
APA StyleButsenko, L., Pasichnyk, L., Kolomiiets, Y., & Kalinichenko, A. (2020). The Effect of Pesticides on the Tomato Bacterial Speck Disease Pathogen Pseudomonas Syringae pv. Tomato. Applied Sciences, 10(9), 3263. https://doi.org/10.3390/app10093263






