Changing Isotopic Food Webs of Two Economically Important Fish in Mediterranean Coastal Lakes with Different Trophic Status
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Field Collections
2.3. Stable Isotope Analysis (SIA)
2.4. Data Analysis
3. Results
3.1. Community Composition and Isotopic Signatures
3.2. Niche Metrics and Diet of Anguilla anguilla
3.3. Niche Metrics and Diet of Diplodus annularis
4. Discussion
5. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Basset, A.; Sabetta, L.; Fonnesu, A.; Mouillot, D.; Chi, T.D.; Viaroli, P.; Giordani, G.; Reizopoulou, S.; Abbiati, M.; Carrada, G.C. Typology in Mediterranean transitional waters: New challenges and perspectives. Aquat. Conserv. Mar. Freshw. Ecosyst. 2006, 16, 441–455. [Google Scholar] [CrossRef]
- Santoro, R.; Bentivoglio, F.; Carlino, P.; Calizza, E.; Costantini, M.L.; Rossi, L. Sensitivity of food webs to nitrogen pollution: A study of three transitional water ecosystems embedded in agricultural landscapes. Transit. Waters Bull. 2014, 8, 84–97. [Google Scholar]
- Jona-Lasinio, G.; Costantini, M.L.; Calizza, E.; Pollice, A.; Bentivoglio, F.; Orlandi, L.; Careddu, G.; Rossi, L. Stable isotope-based statistical tools as ecological indicator of pollution sources in Mediterranean transitional water ecosystems. Ecol. Indic. 2015, 55, 23–31. [Google Scholar] [CrossRef]
- Elliott, M.; Whitfield, A.K.; Potter, I.C.; Blaber, S.J.M.; Cyrus, D.P.; Nordlie, F.G.; Harrison, T.D. The guild approach to categorizing estuarine fish assemblages: A global review. Fish Fish. 2007, 8, 241–268. [Google Scholar] [CrossRef]
- Chapman, P.M. Management of coastal lagoons under climate change. Estuar. Coast. Shelf Sci. 2012, 110, 32–35. [Google Scholar] [CrossRef]
- Orlandi, L.; Bentivoglio, F.; Carlino, P.; Calizza, E.; Rossi, D.; Costantini, M.L.; Rossi, L. δ15N variation in Ulva lactuca as a proxy for anthropogenic nitrogen inputs in coastal areas of Gulf of Gaeta (Mediterranean Sea). Mar. Pollut. Bull. 2014, 84, 76–82. [Google Scholar] [CrossRef]
- Careddu, G.; Costantini, M.L.; Calizza, E.; Carlino, P.; Bentivoglio, F.; Orlandi, L.; Rossi, L. Effects of terrestrial input on macrobenthic food webs of coastal sea are detected by stable isotope analysis in Gaeta Gulf. Estuar. Coast. Shelf Sci. 2015, 154, 158–168. [Google Scholar] [CrossRef]
- Fiorentino, F.; Cicala, D.; Careddu, G.; Calizza, E.; Jona-Lasinio, G.; Rossi, L.; Costantini, M.L. Epilithon δ15N signatures indicate the origins of nitrogen loading and its seasonal dynamics in a volcanic Lake. Ecol. Indic. 2017, 79, 19–27. [Google Scholar] [CrossRef]
- Howarth, R.W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae 2008, 8, 14–20. [Google Scholar] [CrossRef]
- Calizza, E.; Favero, F.; Rossi, D.; Careddu, G.; Fiorentino, F.; Sporta Caputi, S.; Rossi, L.; Costantini, M.L. Isotopic biomonitoring of N pollution in rivers embedded in complex human landscapes. Sci. Total Environ. 2020, 706, 136081. [Google Scholar] [CrossRef]
- Fox, S.E.; Teichberg, M.; Olsen, Y.S.; Heffner, L.; Valiela, I. Restructuring of benthic communities in eutrophic estuaries: Lower abundance of prey leads to trophic shifts from omnivory to grazing. Mar. Ecol. Prog. Ser. 2009, 380, 43–57. [Google Scholar] [CrossRef]
- Martínez-Durazo, A.; García-Hernández, J.; Páez-Osuna, F.; Soto-Jiménez, M.F.; Jara-Marini, M.E. The influence of anthropogenic organic matter and nutrient inputs on the food web structure in a coastal lagoon receiving agriculture and shrimp farming effluents. Sci. Total Environ. 2019, 664, 635–646. [Google Scholar] [CrossRef]
- Calizza, E.; Costantini, M.L.; Rossi, L. Effect of multiple disturbances on food web vulnerability to biodiversity loss in detritus-based systems. Ecosphere 2015, 6, 1–20. [Google Scholar] [CrossRef]
- Carlier, A.; Riera, P.; Amouroux, J.-M.; Bodiou, J.-Y.; Desmalades, M.; Grémare, A. Food web structure of two Mediterranean lagoons under varying degree of eutrophication. J. Sea Res. 2008, 60, 264–275. [Google Scholar] [CrossRef]
- Capoccioni, F.; Costa, C.; Canali, E.; Aguzzi, J.; Antonucci, F.; Ragonese, S.; Bianchini, M.L. The potential reproductive contribution of Mediterranean migrating eels to the Anguilla anguilla stock. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef]
- Trojette, M.; Faleh, A.B.; Fatnassi, M.; Marsaoui, B.; Mahouachi, H.; El, N.; Chalh, A.; Quignard, J.P.; Trabelsi, M. Stock discrimination of two insular populations of Diplodus annularis (ACTINOPTERYGII: PERCIFORMES: SPARIDAE) along the coast of Tunisia by analysis of otolith shape. Acta Ichtyologica et Piscicatoria 2015, 45, 363–372. [Google Scholar] [CrossRef]
- Bilotta, G.S.; Sibley, P.; Hateley, J.; Don, A. The decline of the European eel Anguilla anguilla: Quantifying and managing escapement to support conservation. J. Fish Biol. 2011, 78, 23–38. [Google Scholar] [CrossRef]
- Harrod, C.; Grey, J.; McCarthy, T.K.; Morrissey, M. Stable isotope analyses provide new insights into ecological plasticity in a mixohaline population of European eel. Oecologia 2005, 144, 673–683. [Google Scholar] [CrossRef]
- Chaouch, H.; Ben Abdallah-Ben Hadj Hamida, O.; Ghorbel, M.; Jarboui, O. Feeding habits of the annular seabream, Diplodus annularis (Linnaeus, 1758) (Pisces: Sparidae), in the Gulf of Gabes (Central Mediterranean). Cahiers de Biologie Marine 2014, 55, 13–19. [Google Scholar]
- Vizzini, S.; Mazzola, A. Seasonal variations in the stable carbon and nitrogen isotope ratios (13C/12C and 15N/14N) of primary producers and consumers in a western Mediterranean coastal lagoon. Mar. Biol. 2003, 142, 1009–1018. [Google Scholar] [CrossRef]
- Persic, A.; Roche, H.; Ramade, F. Stable carbon and nitrogen isotope quantitative structural assessment of dominant species from the Vaccarès Lagoon trophic web (Camargue Biosphere Reserve, France). Estuar. Coast. Shelf Sci. 2004, 60, 261–272. [Google Scholar] [CrossRef]
- Mariani, S.; Maccaroni, A.; Massa, F.; Rampacci, M.; Tancioni, L. Lack of consistency between the trophic interrelationships of five sparid species in two adjacent central Mediterranean coastal lagoons. J. Fish Biol. 2002, 61, 138–147. [Google Scholar] [CrossRef]
- Pita, C.; Gamito, S.; Erzini, K. Feeding habits of the gilthead seabream (Sparus aurata) from the Ria Formosa (southern Portugal) as compared to the black seabream (Spondyliosoma cantharus) and the annular seabream (Diplodus annularis). J. Appl. Ichthyol. 2002, 18, 81–86. [Google Scholar] [CrossRef]
- Bouchereau, J.L.; Marques, C.; Pereira, P.; Guélorget, O.; Lourié, S.M.; Vergne, Y. Feeding behaviour of Anguilla anguilla and trophic resources in the Ingril Lagoon (Mediterranean, France). Cahiers de Biologie Marine 2009, 50, 319. [Google Scholar]
- Bouchereau, J.L.; Marques, C.; Pereira, P.; Guélorget, O.; Vergne, Y. Food of the European eel Anguilla anguilla in the Mauguio lagoon (Mediterranean, France). Acta Adriat. 2009, 50, 159–170. [Google Scholar]
- Pasquaud, S.; Elie, P.; Jeantet, C.; Billy, I.; Martinez, P.; Girardin, M. A preliminary investigation of the fish food web in the Gironde estuary, France, using dietary and stable isotope analyses. Estuar. Coast. Shelf Sci. 2008, 78, 267–279. [Google Scholar] [CrossRef]
- França, S.; Vasconcelos, R.P.; Tanner, S.; Máguas, C.; Costa, M.J.; Cabral, H.N. Assessing food web dynamics and relative importance of organic matter sources for fish species in two Portuguese estuaries: A stable isotope approach. Mar. Environ. Res. 2011, 72, 204–215. [Google Scholar] [CrossRef]
- Woodward, G.; Hildrew, A.G. Invasion of a stream food web by a new top predator. J. Anim. Ecol. 2001, 70, 273–288. [Google Scholar] [CrossRef]
- Peterson, B.J.; Fry, B. Stable Isotopes in Ecosystem Studies. Annu. Rev. Ecol. Syst. 1987, 18, 293–320. [Google Scholar] [CrossRef]
- Kwak, T.J.; Zedler, J.B. Food web analysis of southern California coastal wetlands using multiple stable isotopes. Oecologia 1997, 110, 262–277. [Google Scholar] [CrossRef]
- Rossi, L.; Sporta Caputi, S.; Calizza, E.; Careddu, G.; Oliverio, M.; Schiaparelli, S.; Costantini, M.L. Antarctic food web architecture under varying dynamics of sea ice cover. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Fry, B. Stable Isotope Ecology; Springer: New York, NY, USA, 2006; Volume 521. [Google Scholar]
- Fry, B. Coupled N, C and S stable isotope measurements using a dual-column gas chromatography system. Rapid Commun. Mass Spectrom. 2007, 21, 750–756. [Google Scholar] [CrossRef]
- Rossi, L.; Costantini, M.L.; Carlino, P.; di Lascio, A.; Rossi, D. Autochthonous and allochthonous plant contributions to coastal benthic detritus deposits: A dual-stable isotope study in a volcanic lake. Aquat. Sci. 2010, 72, 227–236. [Google Scholar] [CrossRef]
- Careddu, G.; Calizza, E.; Costantini, M.L.; Rossi, L. Isotopic determination of the trophic ecology of a ubiquitous key species—The crab Liocarcinus depurator (Brachyura: Portunidae). Estuar. Coast. Shelf Sci. 2017, 191, 106–114. [Google Scholar] [CrossRef]
- Calizza, E.; Careddu, G.; Sporta Caputi, S.; Rossi, L.; Costantini, M.L. Time- and depth wise trophic niche shifts in Antarctic benthos. PLoS ONE 2018, 13, e0194796. [Google Scholar] [CrossRef]
- Post, D.M. Using Stable Isotopes to Estimate Trophic Position: Models, Methods, and Assumptions. Ecology 2002, 83, 703–718. [Google Scholar] [CrossRef]
- Bentivoglio, F.; Calizza, E.; Rossi, D.; Carlino, P.; Careddu, G.; Rossi, L.; Costantini, M.L. Site-scale isotopic variations along a river course help localize drainage basin influence on river food webs. Hydrobiologia 2016, 770, 257–272. [Google Scholar] [CrossRef]
- Dailer, M.L.; Knox, R.S.; Smith, J.E.; Napier, M.; Smith, C.M. Using δ15N values in algal tissue to map locations and potential sources of anthropogenic nutrient inputs on the island of Maui, Hawai‘i, USA. Mar. Pollut. Bull. 2010, 60, 655–671. [Google Scholar] [CrossRef]
- Rossi, L.; Calizza, E.; Careddu, G.; Rossi, D.; Orlandi, L.; Jona-Lasinio, G.; Aguzzi, L.; Costantini, M.L. Space-time monitoring of coastal pollution in the Gulf of Gaeta, Italy, using δ15N values of Ulva lactuca, landscape hydromorphology, and Bayesian Kriging modelling. Mar. Pollut. Bull. 2018, 126, 479–487. [Google Scholar] [CrossRef]
- Orlandi, L.; Calizza, E.; Careddu, G.; Carlino, P.; Costantini, M.L.; Rossi, L. The effects of nitrogen pollutants on the isotopic signal (δ15N) of Ulva lactuca: Microcosm experiments. Mar. Pollut. Bull. 2017, 115, 429–435. [Google Scholar] [CrossRef]
- Rosecchi, E. L’alimentation de Diplodus annularis, Diplodus sargus, Diplodus vulgaris et Sparus aurata (Pisces, Sparidae) dans le golfe du Lion et les lagunes littorales. Revue des Travaux de l’Institut des Pêches maritimes 1987, 49, 125–141. [Google Scholar]
- Rodriguez-Ruiz, S.; Sànchez-Lizaso, J.L.; Ramos-Esplà, A.A. Feeding of Diplodus annularis in Posidonia oceanica meadows: Ontogenetic, diel and habitat related dietary shifts. Bull. Mar. Sci. 2002, 71, 1353–1360. [Google Scholar]
- Lazio Region, Piano di Tutela delle Acque Regionale (PTAR) Aggiornamento. Available online: http://www.regione.lazio.it/binary/prl_ambiente/tbl_contenuti/AMB_Piano_tutela_delle_acque_PTAR_aggiornamento.pdf (accessed on 20 March 2020).
- Cataudella, S.; Crosetti, D.; Massa, F. Mediterranean Coastal Lagoons: Sustainable Management and Interactions among Aquaculture, Capture Fisheries and the Environment; General Fisheries Commission for the Mediterranean: Studies and Reviews; FAO: Rome, Italy, 2015; Volume 95. [Google Scholar]
- Basset, A.; Galuppo, N.; Sabetta, L. Environmental heterogeneity and benthic macroinvertebrate guilds in Italian lagoons. Transit. Waters Bull. 2007, 1, 48–63. [Google Scholar]
- Cioffi, F.; Gallerano, F. Management strategies for the control of eutrophication processes in Fogliano lagoon (Italy): A long-term analysis using a mathematical model. Appl. Math. Model. 2001, 25, 385–426. [Google Scholar] [CrossRef]
- Jacob, U.; Mintenbeck, K.; Brey, T.; Knust, R.; Beyer, K. Stable isotope food web studies: A case for standardized sample treatment. Mar. Ecol. Progr. 2005, 287, 251–253. [Google Scholar] [CrossRef]
- McCutchan, J.H.; Lewis, W.M.; Kendall, C.; McGrath, C.C. Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 2003, 102, 378–390. [Google Scholar] [CrossRef]
- Vander Zanden, M.J.; Rasmussen, J.B. A trophic position model of pelagic food webs: Impact on contaminant bioaccumulation in lake trout. Ecol. Monogr. 1996, 66, 451–477. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics; Version 3.0; National History Museum, University of Oslo: Oslo, Norway, 2013. [Google Scholar]
- Parnell, A.; Inger, R. Simmr: A Stable Isotope Mixing Model; R Package Version 0.3. R. 2016. Available online: https://CRAN.R-project.org/package=simmr (accessed on 20 June 2019).
- DeNiro, M.J.; Epstein, S. Influence of diet on the distribution of carbon isotopes in animals. Geochim. Cosmochim. Acta 1978, 42, 495–506. [Google Scholar] [CrossRef]
- Minagawa, M.; Wada, E. Stepwise enrichment of 15N along food chains: Further evidence and the relation between δ15N and animal age. Geochim. Cosmochim. Acta 1984, 48, 1135–1140. [Google Scholar] [CrossRef]
- Bardonnet, A.; Riera, P. Feeding of glass eels (Anguilla anguilla) in the course of their estuarine migration: New insights from stable isotope analysis. Estuar. Coast. Shelf Sci. 2005, 63, 201–209. [Google Scholar] [CrossRef]
- Costantini, M.L.; Carlino, P.; Calizza, E.; Careddu, G.; Cicala, D.; Sporta Caputi, S.; Fiorentino, F.; Rossi, L. The role of alien fish (the centrarchid Micropterus salmoides) in lake food webs highlighted by stable isotope analysis. Freshw. Biol. 2018, 63, 1130–1142. [Google Scholar] [CrossRef]
- McArthur, R.; Levin, R. The limiting similarity, convergence, and divergence of coexisting species. Am. Nat. 1967, 101, 377–385. [Google Scholar] [CrossRef]
- Levins, R. Evolution in Changing Environments: Some Theoretical Explorations (No. 2); Princeton University Press: Princeton, NJ, USA, 1968. [Google Scholar]
- Pianka, E.R. Sympatry of Desert Lizards (Ctenotus) in Western Australia. Ecology 1969, 50, 1012–1030. [Google Scholar] [CrossRef]
- Chesson, J. Measuring preference in selective predation. Ecology 1978, 59, 211–215. [Google Scholar] [CrossRef]
- Costantini, M.L.; Rossi, L. Laboratory study of the grass shrimp feeding preferences. Hydrobiologia 2010, 443, 129–136. [Google Scholar] [CrossRef]
- Layman, C.A.; Arrington, D.A.; Montaña, C.G.; Post, D.M. Can Stable Isotope Ratios Provide for Community-Wide Measures of Trophic Structure? Ecology 2007, 88, 42–48. [Google Scholar] [CrossRef]
- Bearhop, S.; Adams, C.E.; Waldron, S.; Fuller, R.A.; Macleod, H. Determining trophic niche width: A novel approach using stable isotope analysis. J. Anim. Ecol. 2004, 73, 1007–1012. [Google Scholar] [CrossRef]
- Jackson, A.L.; Inger, R.; Parnell, A.C.; Bearhop, S. Comparing isotopic niche widths among and within communities: SIBER—Stable Isotope Bayesian Ellipses in R. J. Ecol. 2011, 595–602. [Google Scholar] [CrossRef]
- Parnell, A.C.; Inger, R.; Bearhop, S.; Jackson, A.L. Source partitioning using stable isotopes: Coping with too much variation. PLoS ONE 2010, 5, e9672. [Google Scholar] [CrossRef]
- Brey, T.; Rumohr, H.; Ankar, S. Energy content of macrobenthic invertebrates: General conversion factors from weight to energy. Mar. Ecol. Prog. Ser. 1988, 117, 271–278. [Google Scholar] [CrossRef]
- Neuhoff, H.G. Influence of temperature and salinity on food conversion and growth of different Nereis species (Polychaeta, Annelida). Mar. Ecol. Prog. 1979, 1, e262. [Google Scholar] [CrossRef]
- Ryan, P.A. Seasonal and size-related changes in the food of the short-finned eel, Anguilla australis in Lake Ellesmere, Canterbury, New Zealand. Environ. Biol. Fishes 1986, 15, 47–58. [Google Scholar] [CrossRef]
- Whittaker, R.H.; Levin, S.A. Niche: Theory and application; Stroudsbourg, PA: Dowden, Hutchison and Ross; Distributed by Halsted Press: New York, NY, USA, 1975. [Google Scholar]
- Calizza, E.; Costantini, M.L.; Careddu, G.; Rossi, L. Effect of habitat degradation on competition, carrying capacity, and species assemblage stability. Ecol. Evol. 2017, 7, 5784–5796. [Google Scholar] [CrossRef] [PubMed]
- McClelland, J.W.; Valiela, I.; Michener, R.H. Nitrogen-stable isotope signatures in estuarine food webs: A record of increasing urbanization in coastal watersheds. Limnol. Oceanogr. 1997, 42, 930–937. [Google Scholar] [CrossRef]
- Di Lascio, A.; Rossi, L.; Carlino, P.; Calizza, E.; Rossi, D.; Costantini, M.L. Stable isotope variation in macroinvertebrates indicates anthropogenic disturbance along an urban stretch of the river Tiber (Rome, Italy). Ecol. Indic. 2013, 28, 107–114. [Google Scholar] [CrossRef]
- Vizzini, S.; Mazzola, A. The effects of anthropogenic organic matter inputs on stable carbon and nitrogen isotopes in organisms from different trophic levels in a southern Mediterranean coastal area. Sci. Total Environ. 2006, 368, 723–731. [Google Scholar] [CrossRef]
- Calizza, E.; Aktan, Y.; Costantini, M.L.; Rossi, L. Stable isotope variations in benthic primary producers along the Bosphorus (Turkey): A preliminary study. Mar. Pollut. Bull. 2015, 97, 535–538. [Google Scholar] [CrossRef]
- Vizzini, S.; Mazzola, A. The fate of organic matter sources in coastal environments: A comparison of three Mediterranean lagoons. Hydrobiologia 2008, 611, 67–79. [Google Scholar] [CrossRef]
- Tapia González, F.U.; Herrera-Silveira, J.A.; Aguirre-Macedo, M.L. Water quality variability and eutrophic trends in karstic tropical coastal lagoons of the Yucatán Peninsula. Estuar. Coast. Shelf Sci. 2008, 76, 418–430. [Google Scholar] [CrossRef]
- Tagliapietra, D.; Pavan, M.; Wagner, C. Macrobenthic Community Changes Related to Eutrophication in Palude della Rosa (Venetian Lagoon, Italy). Estuar. Coast. Shelf Sci. 1998, 47, 217–226. [Google Scholar] [CrossRef]
- Mehner, T.; Arlinghaus, R.; Berg, S.; Dörner, H.; Jacobsen, L.; Kasprzak, P.; Koschel, R.; Schulze, T.; Skov, C.; Wolter, C.; et al. How to link biomanipulation and sustainable fisheries management: A step-by-step guideline for lakes of the European temperate zone. Fish. Manag. Ecol. 2004, 11, 261–275. [Google Scholar] [CrossRef]
- Cicala, D.; Calizza, E.; Careddu, G.; Fiorentino, F.; Sporta Caputi, S.; Rossi, L.; Costantini, M.L. Spatial variation in the feeding strategies of Mediterranean fish: Flatfish and mullet in the Gulf of Gaeta (Italy). Aquat. Ecol. 2019, 53, 529–541. [Google Scholar] [CrossRef]
- Dörner, H.; Skov, C.; Berg, S.; Schulze, T.; Beare, D.J.; Van der Velde, G. Piscivory and trophic position of Anguilla anguilla in two lakes: Importance of macrozoobenthos density. J. Fish Biol. 2009, 74, 2115–2131. [Google Scholar] [CrossRef]
- Deudero, S.; Pinnegar, J.K.; Polunin, N.V.C.; Morey, G.; Morales-Nin, B. Spatial variation and ontogenic shifts in the isotopic composition of Mediterranean littoral fishes. Mar. Biol. 2004, 145, 971–981. [Google Scholar] [CrossRef]
- Jennings, S.; Barnes, C.; Sweeting, C.J.; Polunin, N.V. Application of nitrogen stable isotope analysis in size-based marine food web and macroecological research. Rapid Commun. Mass Spectrom. 2008, 22, 1673–1680. [Google Scholar] [CrossRef]
- Galvan, D.E.; Sweeting, C.J.; Reid, W.D.K. Power of stable isotope techniques to detect size-based feeding in marine fishes. Mar. Ecol. Prog. Ser. 2010, 407, 271–278. [Google Scholar] [CrossRef]
- Lammens, E.H.R.R.; Nie, H.W.; de Vijverberg, J.; Densen, W.L.T. Resource Partitioning and Niche Shifts of Bream (Abramis brama) and Eel (Anguilla anguilla) Mediated by Predation of Smelt (Osmerus eperlanus) on Daphnia hyalina. Can. J. Fish. Aquat. Sci. 1985, 42, 1342–1351. [Google Scholar] [CrossRef]
- De Nie, H.W. Food, feeding periodicity and consumption of the eel Anguilla anguilla (L.) in the shallow eutrophic Tjeukemeer (The Netherlands). Archiv für Hydrobiologie 1987, 109, 421–443. [Google Scholar]
- Polis, G.A.; Strong, D.R. Food Web Complexity and Community Dynamics. Am. Nat. 1996, 147, 813–846. [Google Scholar] [CrossRef]
- Layman, C.A.; Araujo, M.S.; Boucek, R.; Hammerschlag-Peyer, C.M.; Harrison, E.; Jud, Z.R.; Matich, P.; Rosenblatt, A.E.; Vaudo, J.J.; Yeager, L.A.; et al. Applying stable isotopes to examine food-web structure: An overview of analytical tools. Biol. Rev. 2012, 87, 545–562. [Google Scholar] [CrossRef]
- Calizza, E.; Costantini, M.L.; Carlino, P.; Bentivoglio, F.; Orlandi, L.; Rossi, L. Posidonia oceanica habitat loss and changes in litter-associated biodiversity organization: A stable isotope-based preliminary study. Estuar. Coast. Shelf Sci. 2013, 135, 137–145. [Google Scholar] [CrossRef]
- Calizza, E.; Costantini, M.L.; Rossi, D.; Carlino, P.; Rossi, L. Effects of disturbance on an urban river food web. Freshw. Biol. 2012, 57, 2613–2628. [Google Scholar] [CrossRef]

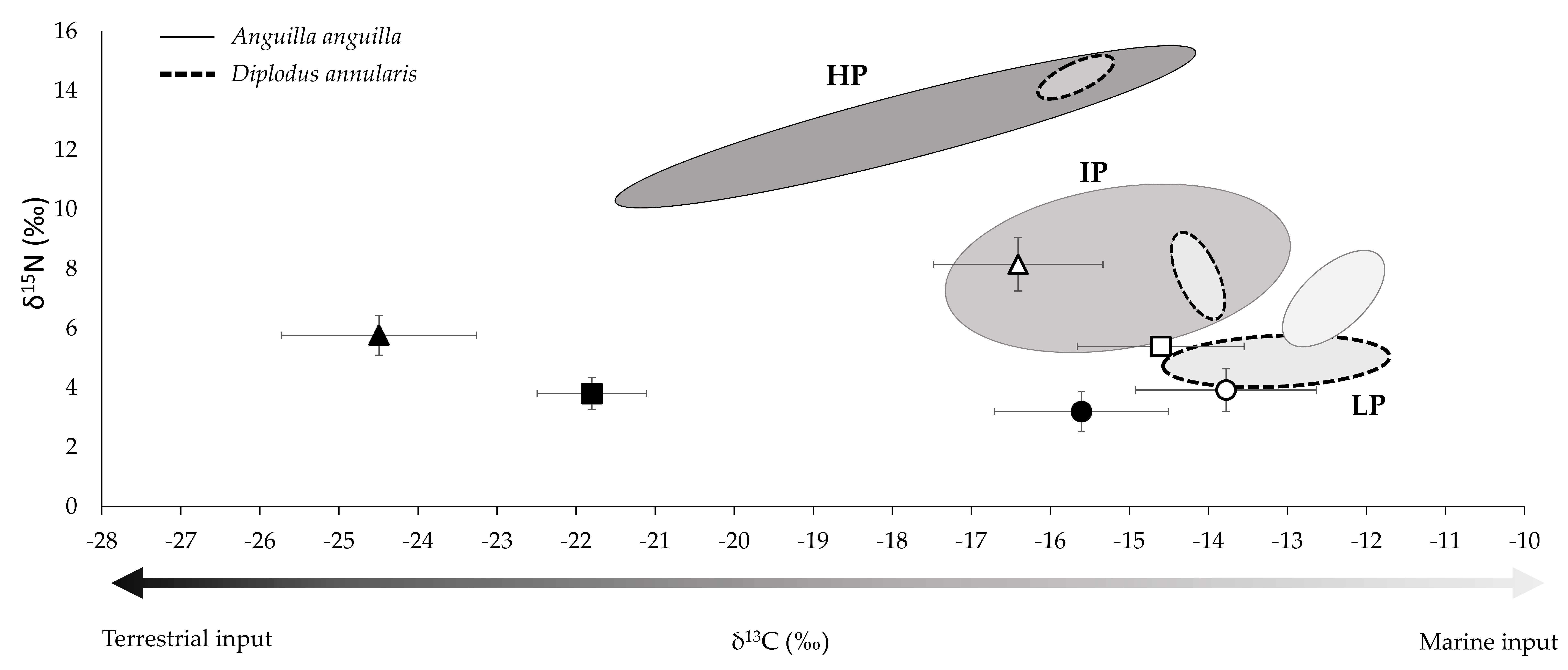
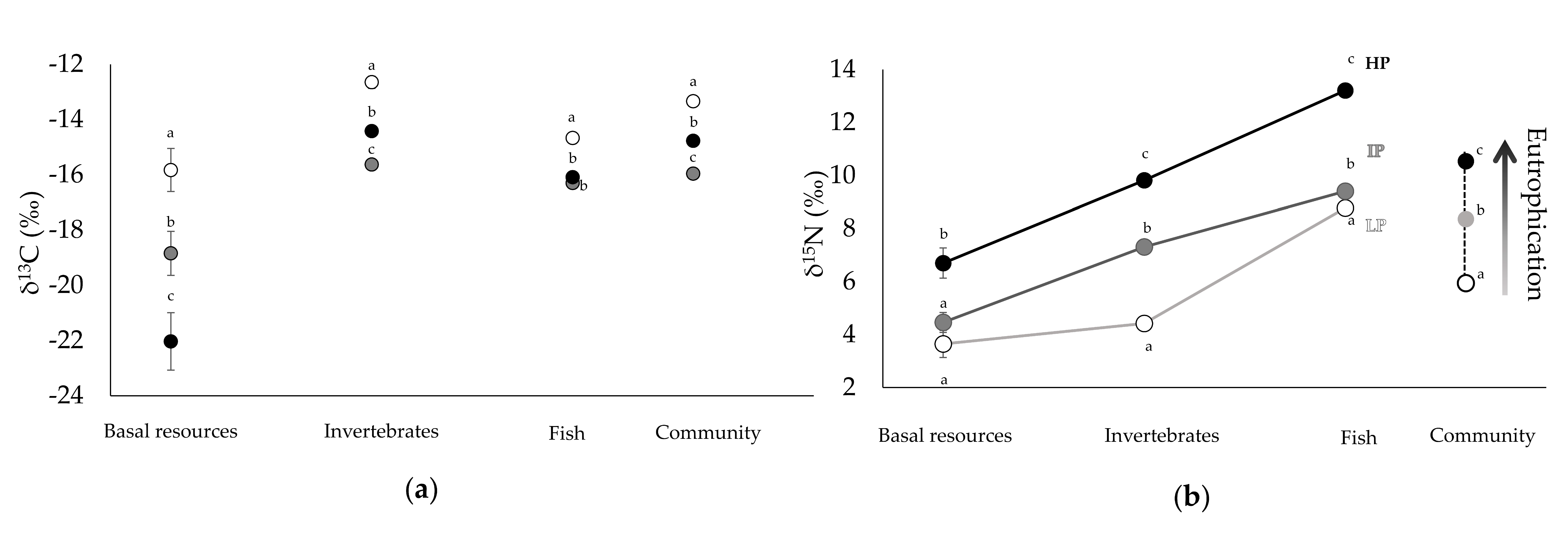
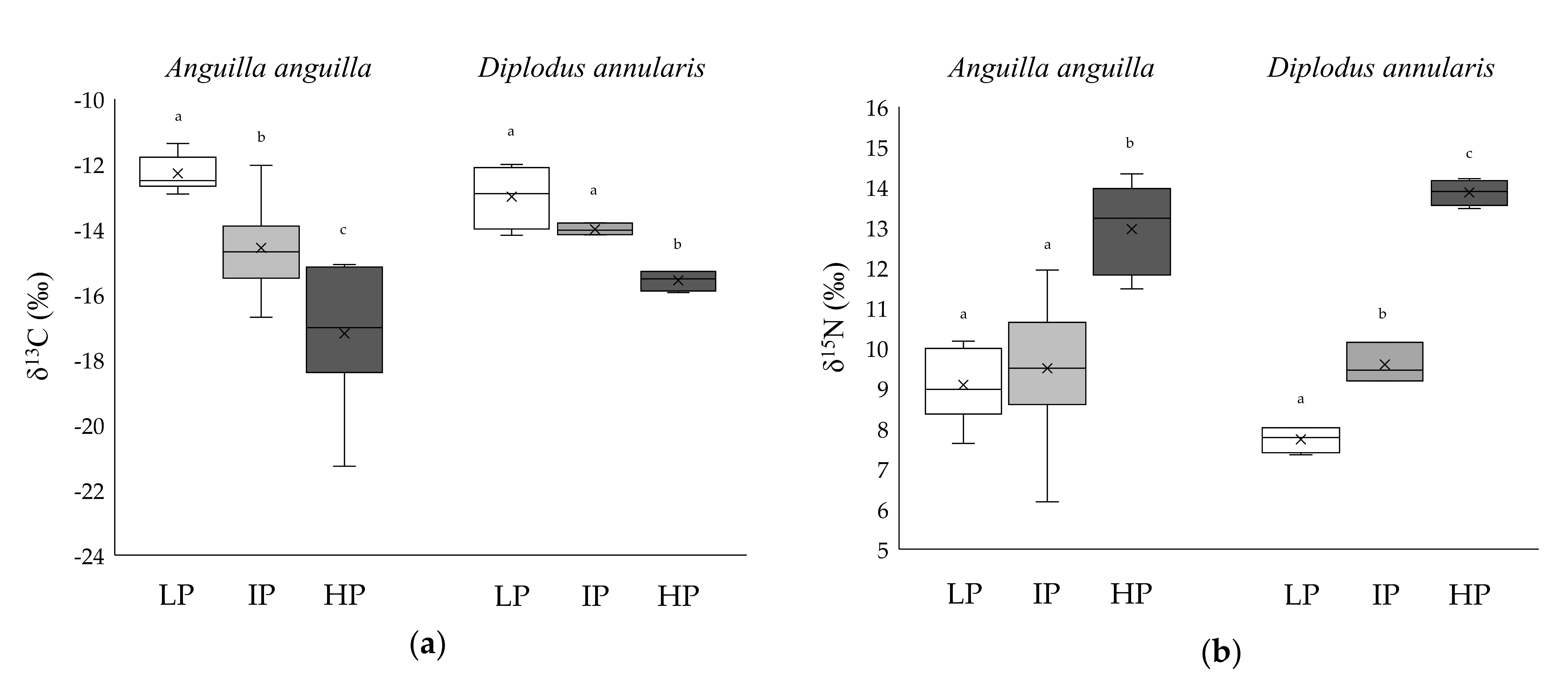

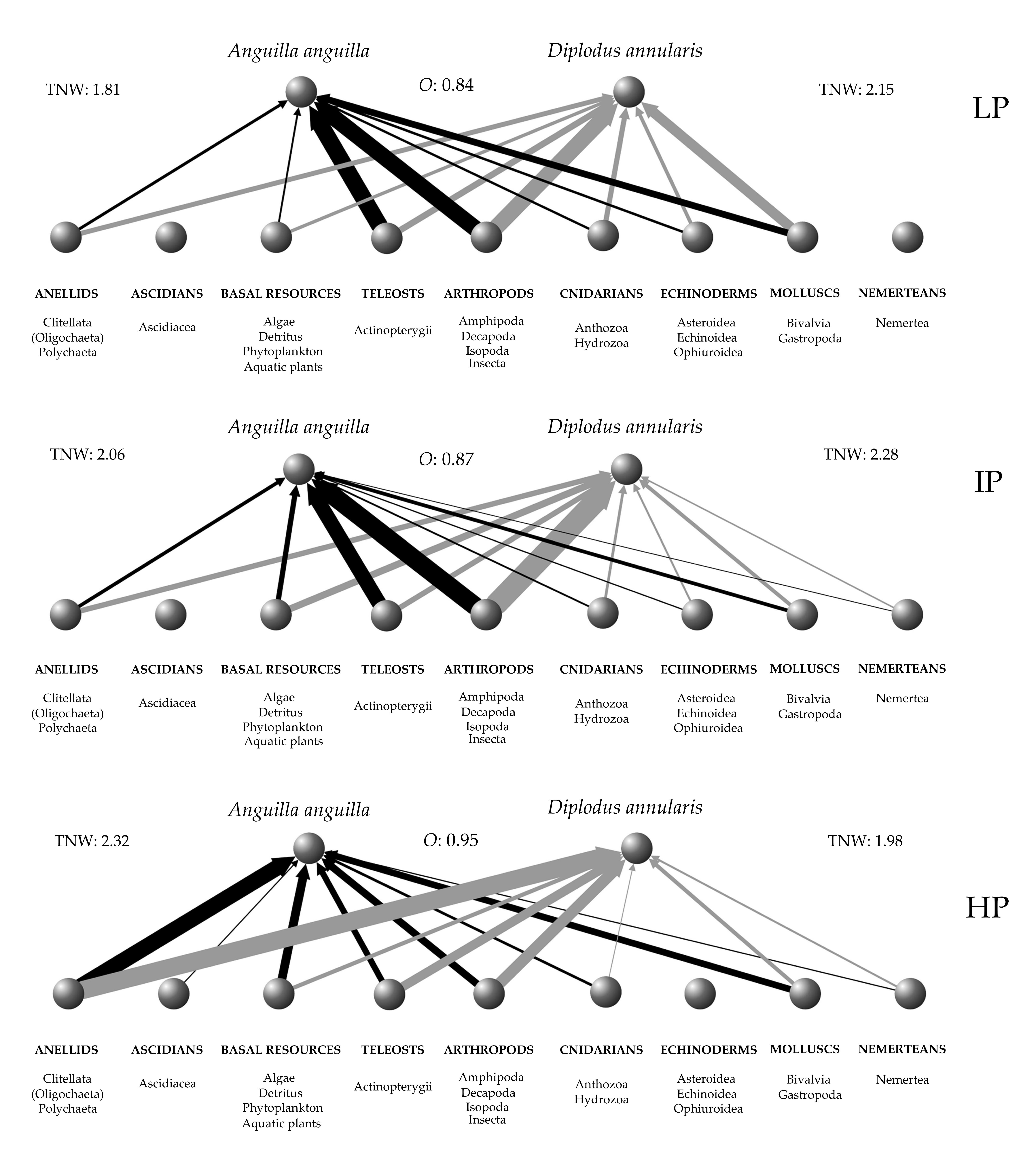
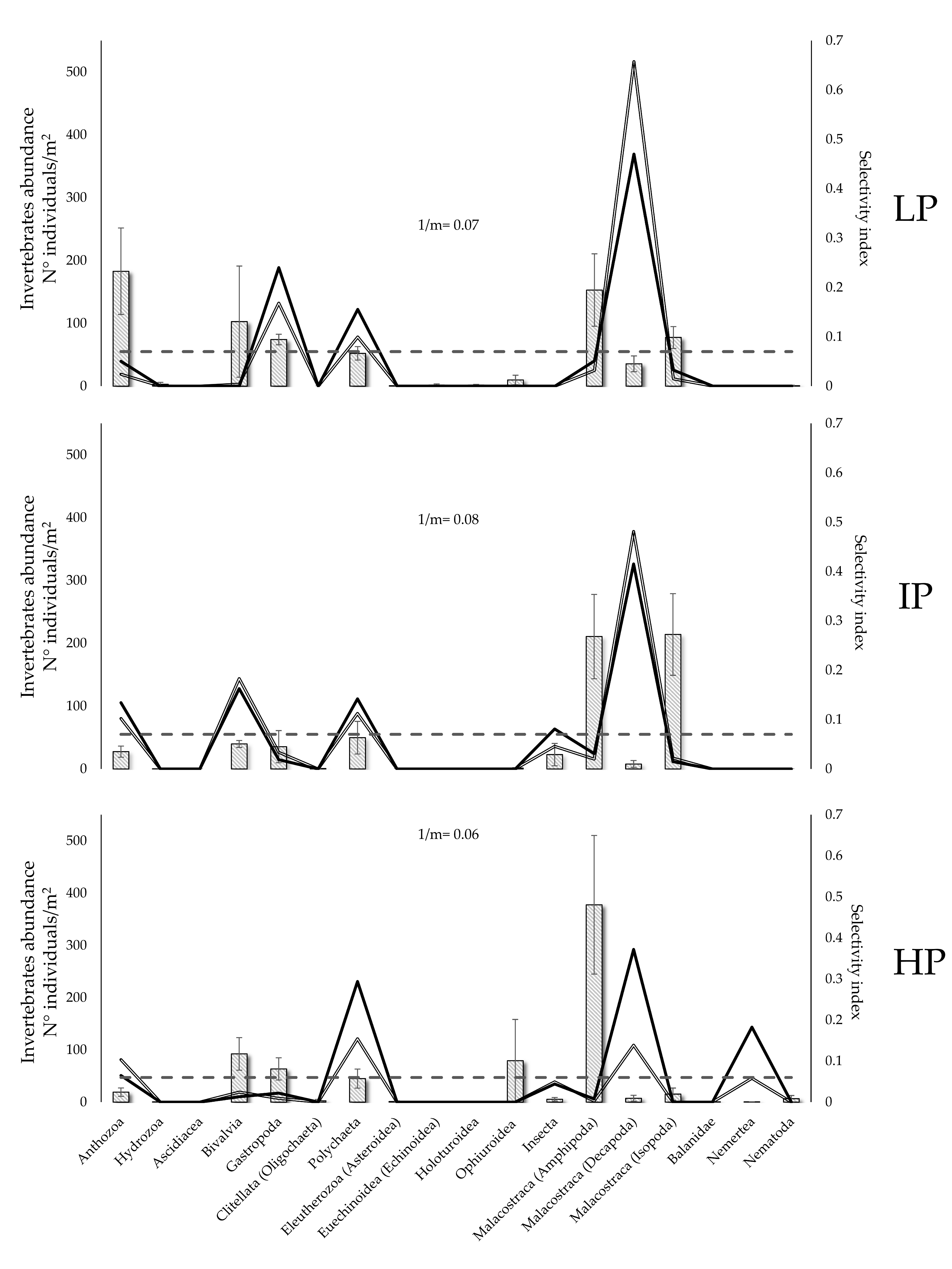
| LP | IP | HP | |
|---|---|---|---|
| N° | |||
| Community | 2942 (417) | 2777 (502) | 2926 (340) |
| Basal resources | 28 (28) | 51 (51) | 28 (28) |
| Invertebrates | 2793 (268) a | 2526 (251) b | 2848 (262) a |
| Fish | 149 (145) a | 251 (251) b | 78 (78) c |
| δ13C (‰) | |||
| Community | −13.34 ± 0.17 a | −15.96 ± 0.13 b | −14.77 ± 0.15 c |
| Basal resources | −15.83 ± 0.78 a | −18.84 ± 0.80 b | −22.03 ± 1.04 c |
| Invertebrates | −12.65 ± 0.22 a | −15.63 ± 0.19 b | −14.42 ± 0.18 c |
| Fish | −14.67 ± 0.21 a | −16.30 ± 0.17 b | −16.09 ± 0.15 b |
| δ15N (‰) | |||
| Community | 5.94 ± 0.16 a | 8.36 ± 0.14 b | 10.54 ± 0.14 c |
| Basal resources | 3.65 ± 0.51 a | 4.46 ± 0.38 a | 6.70 ± 0.57 b |
| Invertebrates | 4.42 ± 0.16 a | 7.31 ± 0.21 b | 9.82 ± 0.14 c |
| Fish | 8.79 ± 0.17 a | 9.41 ± 0.15 b | 13.21 ± 0.16 c |
| Anguilla anguilla | Diplodus annularis | |||||
|---|---|---|---|---|---|---|
| LP | IP | HP | LP | IP | HP | |
| N | 8 | 16 | 10 | 8 | 6 | 8 |
| ITUs | ||||||
| δ13C (‰) | −12.76 ± 0.11 a | −15.07 ± 0.15 b | −16.31 ± 0.16 c | −13.49 ± 0.16 a | −16.23 ± 0.14 a | −15.34 ± 0.19 b |
| δ15N (‰) | 8.27 ± 0.19 a | 9.62 ± 0.12 a | 13.58 ± 0.11 b | 5.81 ± 0.15 a | 7.99 ± 0.14 b | 10.24 ± 0.15 c |
| CR | 2.05 | 9.52 | 6.19 | 2.17 | 0.35 | 0.65 |
| NR | 3.15 | 5.79 | 3.51 | 0.67 | 0.96 | 0.74 |
| Taxa | ||||||
| δ13C (‰) | −12.29 ± 0.19 a | −15.01 ± 0.53 b | −17.20 ± 1.12 c | −13.00 ± 0.49 a | −14.00 ± 0.10 a | −15.57 ± 0.16 b |
| δ15N (‰) | 9.08 ± 0.32 a | 9.50 ± 0.36 a | 12.96 ± 0.51 b | 7.73 ± 0.17 a | 9.59 ± 0.29 b | 13.87 ± 0.16 c |
| CR | 1.55 | 4.66 | 6.19 | 2.17 | 0.35 | 0.65 |
| NR | 2.54 | 2.59 | 2.86 | 0.67 | 0.96 | 0.74 |
| L | 30 | 84 | 33 | 28 | 13 | 14 |
| S | 19 | 42 | 22 | 21 | 10 | 11 |
| L/S | 1.6 | 2.0 | 1.5 | 1.3 | 1.3 | 1.3 |
| SEAc | 1.46 | 9.62 | 4.84 | 1.55 | 0.45 | 0.32 |
| TNW | 1.81 | 2.06 | 2.32 | 2.15 | 2.28 | 1.98 |
| LP | IP | HP | ||||
|---|---|---|---|---|---|---|
| Food Sources | Taxa | Contribution | Taxa | Contribution | Taxa | Contribution |
| TELEOSTS | ||||||
| Actinopterygii | 7 | 34.76 ± 1.90 | 12 | 30.65 ± 0.50 | 3 | 12.18 ± 1.50 |
| CNIDARIANS | ||||||
| Anthozoa | 2 | 4.40 ± 0.60 | 4 | 3.17 ± 0.10 | 2 | 3.39 ± 0.20 |
| Hydrozoa | - | - | - | - | 1 | 1.67 ± 0.10 |
| ASCIDIANS | ||||||
| Ascidiacea | - | - | - | - | 1 | 1.82 ± 0.10 |
| BASAL RESOURCES | ||||||
| Algae | 1 | 0.75 ± 0.10 | - | - | 1 | 1.55 ± 0.10 |
| Detritus | 2 | 1.81 ± 0.40 | 4 | 5.33 ± 0.60 | 2 | 9.03 ± 0.50 |
| Phytoplankton | - | - | 1 | 0.42 ± 0.10 | 1 | 3.10 ± 0.10 |
| Aquatic plants | 1 | 0.75 ± 0.10 | 4 | 4.68 ± 0.40 | 1 | 4.19 ± 0.10 |
| MOLLUSCS | ||||||
| Bivalvia | 1 | 0.43 ± 0.10 | 4 | 5.68 ± 0.40 | 4 | 10.35 ± 0.90 |
| Gastropoda | 8 | 13.06 ± 0.40 | 3 | 1.57 ± 0.20 | 2 | 2.95 ± 1.00 |
| ANELLIDA | ||||||
| Clitellata (Oligochaeta) | - | - | - | - | 1 | 2.60 ± 0.10 |
| Polychaeta | 6 | 5.62 ± 0.20 | 5 | 6.99 ± 0.20 | 13 | 30.41 ± 0.60 |
| ECHINODERMS | ||||||
| Eleutherozoa (Asteroidea) | 1 | 0.29 ± 0.10 | - | - | - | - |
| Euechinoidea (Echinoidea) | 1 | 4.40 ± 0.10 | - | - | - | - |
| Ophiuroidea | - | - | 1 | 2.01 ± 0.10 | - | - |
| ARTHROPODS | ||||||
| Insecta | - | - | 1 | 1.42 ± 0.10 | 1 | 1.62 ± 0.10 |
| Malacostraca | ||||||
| Amphipoda | 4 | 4.74 ± 0.30 | 4 | 5.10 ± 0.40 | 2 | 4.13 ± 0.50 |
| Decapoda | 4 | 27.84 ± 4.60 | 7 | 26.13 ± 1.90 | 2 | 9.11 ± 1.90 |
| Isopoda | 2 | 1.15 ± 1.70 | 4 | 5.78 ± 0.30 | - | - |
| NEMERTEANS | ||||||
| Nemertea | - | - | 1 | 1.08 ± 0.10 | 1 | 1.91 ± 0.10 |
| BASAL RESOURCES | 3.31 ± 0.35 a | 10.43 ± 1.54 ab | 17.87 ± 1.60 b | |||
| INVERTEBRATES | 61.93 ± 2.92 | 58.93 ± 2.15 | 69.96 ± 2.56 | |||
| FISH | 34.76 ± 1.90 a | 30.65 ± 0.50 a | 16.18 ± 1.50 b | |||
| LP | IP | HP | ||||
|---|---|---|---|---|---|---|
| Food Sources | Taxa | Contribution | Taxa | Contribution | Taxa | Contribution |
| TELEOSTS | ||||||
| Actinopterygii | 4 | 12.69 ± 0.49 | 5 | 10.84 ± 0.28 | 3 | 18.56 ± 0.78 |
| CNIDARIANS | ||||||
| Anthozoa | 2 | 10.21 ± 1.31 | 3 | 5.15 ± 0.69 | 1 | 1.28 ± 4.75 |
| Hydrozoa | - | - | - | - | - | - |
| BASAL RESOURCES | ||||||
| Algae | 1 | 2.01 ± 0.10 | - | - | - | - |
| Detritus | 1 | 1.56 ± 0.58 | 2 | 5.91 ± 0.29 | 1 | 3.02 ± 5.00 |
| Phytoplankton | - | - | - | - | - | - |
| Aquatic plants | 2 | 2.88 ± 0.91 | 4 | 8.07 ± 0.10 | 1 | 5.05 ± 0.28 |
| MOLLUSCS | ||||||
| Bivalvia | - | - | 2 | 6.24 ± 0.76 | 2 | 3.37 ± 0.44 |
| Gastropoda | 6 | 20.65 ± 1.06 | 1 | 1.08 ± 5.00 | 2 | 4.29 ± 1.23 |
| ANELLIDS | ||||||
| Clitellata (Oligochaeta) | - | - | - | - | 1 | 2.73 ± 5.00 |
| Polychaeta | 8 | 9.71 ± 0.55 | 3 | 10.89 ± 0.69 | 8 | 34.76 ± 1.12 |
| ECHINODERMS | ||||||
| Eleutherozoa (Asteroidea) | 1 | 0.99 ± 5.00 | - | - | - | - |
| Euechinoidea (Echinoidea) | 1 | 6.04 ± 5.00 | - | - | - | - |
| Ophiuroidea | - | - | 1 | 3.54 ± 0.75 | - | - |
| ARTHROPODS | ||||||
| Insecta | - | - | 1 | 3.1 ± 1.06 | 1 | 0.87 ± 5.00 |
| Malacostraca | ||||||
| Amphipoda | 3 | 8.43 ± 1.13 | 3 | 9.86 ± 1.54 | 2 | 7.8 ± 1.10 |
| Decapoda | 5 | 22.03 ± 1.37 | 5 | 27.86 ± 1.69 | 2 | 14.67 ± 1.03 |
| Isopoda | 3 | 2.8 ± 0.31 | 4 | 4.85 ± 0.20 | - | - |
| NEMERTEANS | ||||||
| Nemertea | - | - | 1 | 2.61 ± 0.71 | 1 | 3.6 ± 0.74 |
| BASAL RESOURCES | 6.45 ± 0.39 | 13.98 ± 1.08 | 8.07 ± 1.02 | |||
| INVERTEBRATES | 80.86 ± 2.7 | 75.18 ± 2.46 | 73.37 ± 3.61 | |||
| FISH | 12.68 ± 0.49 | 10.82 ± 0.28 | 18.57 ± 0.78 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sporta Caputi, S.; Careddu, G.; Calizza, E.; Fiorentino, F.; Maccapan, D.; Rossi, L.; Costantini, M.L. Changing Isotopic Food Webs of Two Economically Important Fish in Mediterranean Coastal Lakes with Different Trophic Status. Appl. Sci. 2020, 10, 2756. https://doi.org/10.3390/app10082756
Sporta Caputi S, Careddu G, Calizza E, Fiorentino F, Maccapan D, Rossi L, Costantini ML. Changing Isotopic Food Webs of Two Economically Important Fish in Mediterranean Coastal Lakes with Different Trophic Status. Applied Sciences. 2020; 10(8):2756. https://doi.org/10.3390/app10082756
Chicago/Turabian StyleSporta Caputi, Simona, Giulio Careddu, Edoardo Calizza, Federico Fiorentino, Deborah Maccapan, Loreto Rossi, and Maria Letizia Costantini. 2020. "Changing Isotopic Food Webs of Two Economically Important Fish in Mediterranean Coastal Lakes with Different Trophic Status" Applied Sciences 10, no. 8: 2756. https://doi.org/10.3390/app10082756
APA StyleSporta Caputi, S., Careddu, G., Calizza, E., Fiorentino, F., Maccapan, D., Rossi, L., & Costantini, M. L. (2020). Changing Isotopic Food Webs of Two Economically Important Fish in Mediterranean Coastal Lakes with Different Trophic Status. Applied Sciences, 10(8), 2756. https://doi.org/10.3390/app10082756






