Relative Environmental, Economic, and Energy Performance Indicators of Fuel Compositions with Biomass
Abstract
1. Introduction
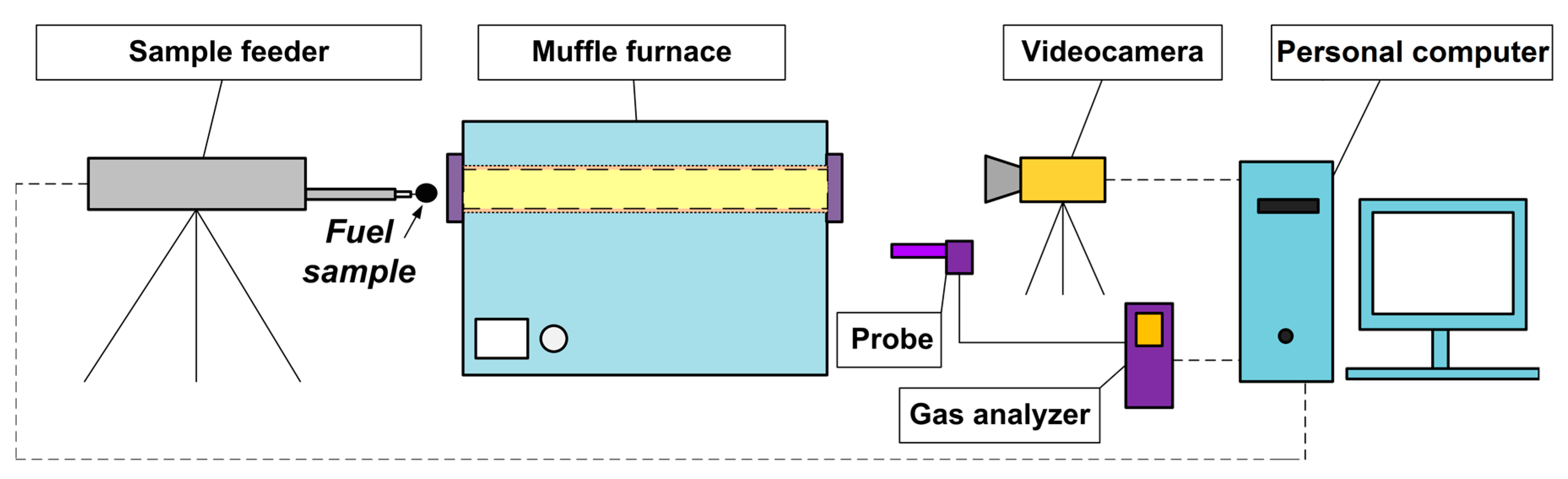
2. Materials and Methods
3. Results and Discussion
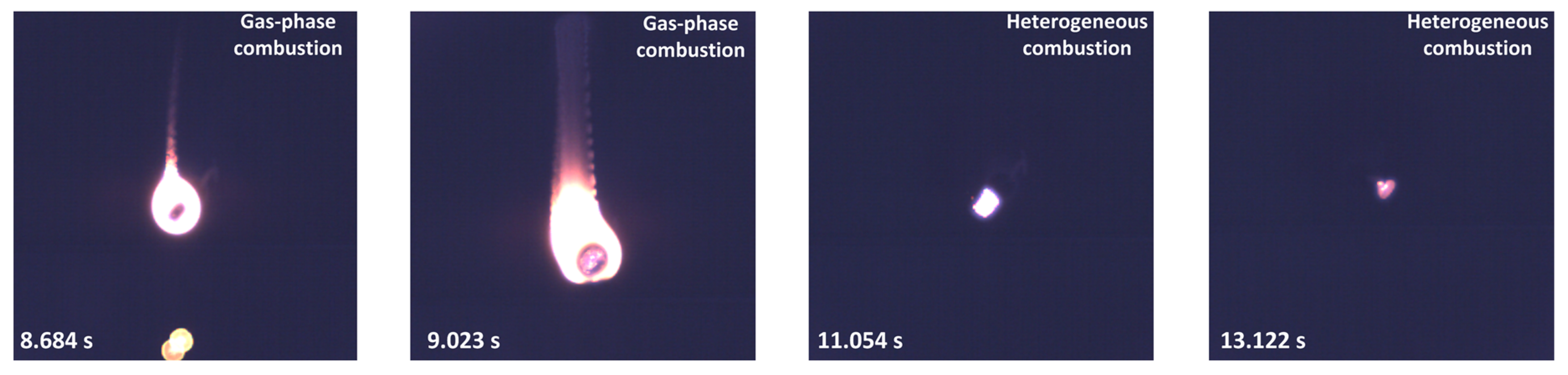
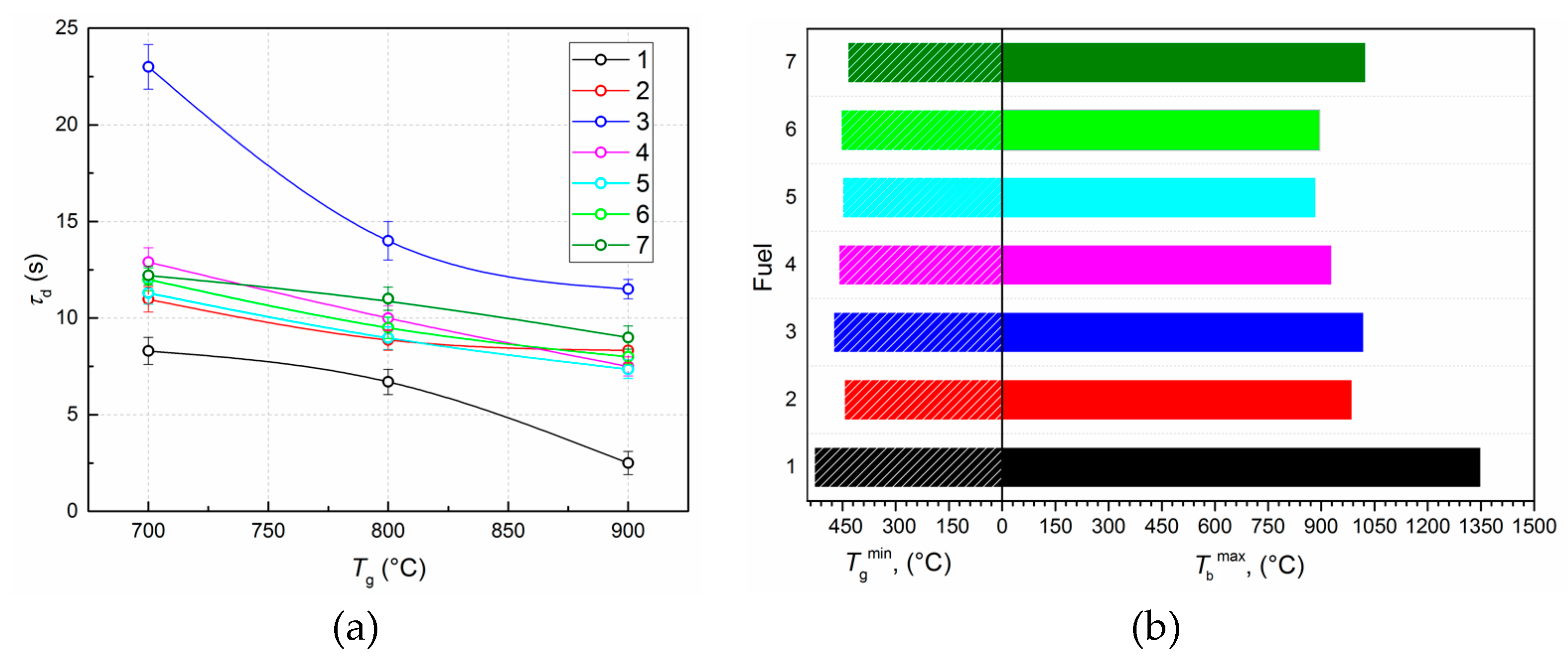
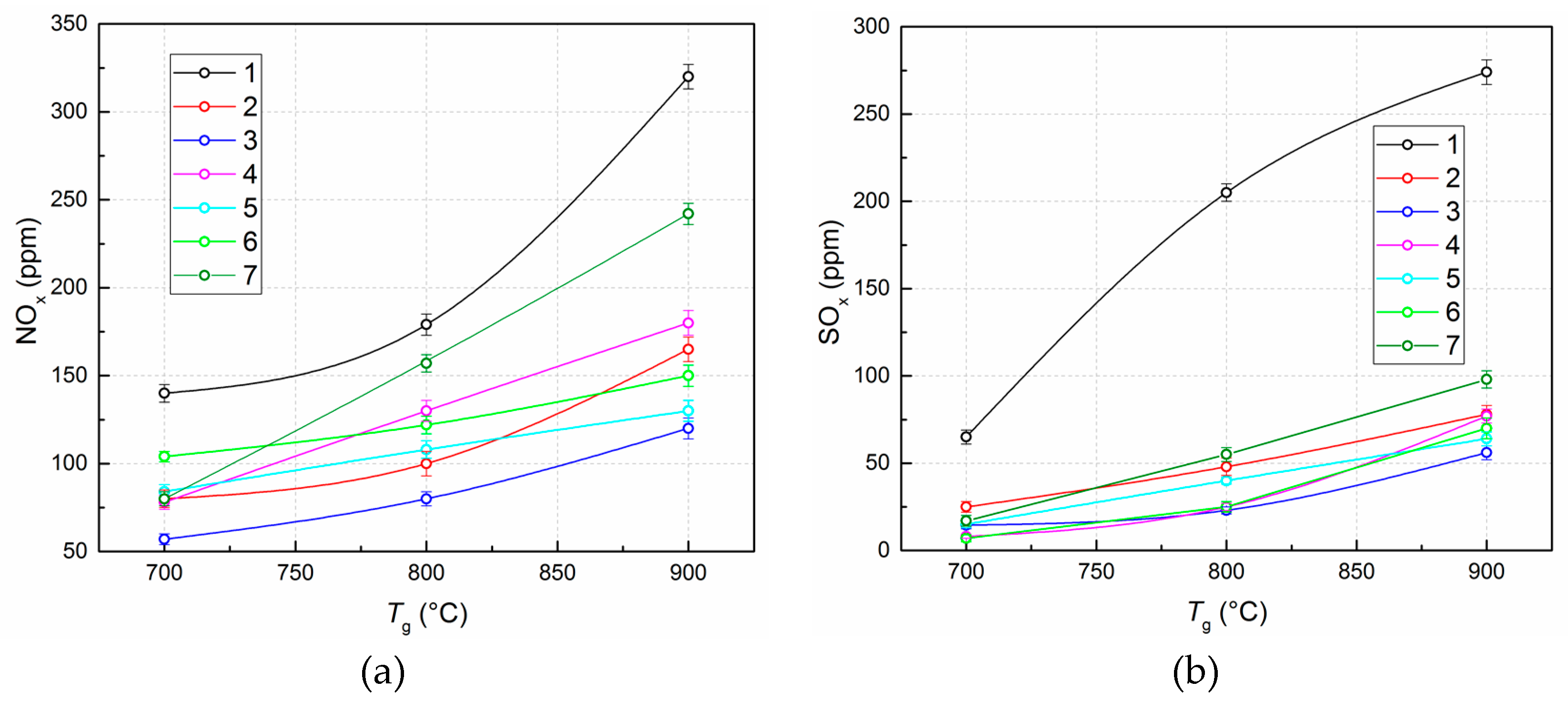
3.1. Experimental Results
3.2. MCDM Principles and Selected Criteria
3.3. Efficiency Indicators of Combustion of Slurry Fuels with Biomass
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| An | relative complex parameter |
| Ad | ash content, % |
| C | fuel cost, USD/kg |
| Cdaf, Hdaf, Ndaf, Odaf | fraction of carbon, hydrogen, nitrogen, oxygen in the sample converted to a dry ash free state, % |
| N | capital costs, USD |
| Qas | heat of combustion, MJ/kg |
| Std | fraction of sulfur in the sample converted to a dry state, % |
| Tbmax | maximum combustion temperature, °C |
| Tg | temperature in the combustion chamber, °C |
| Tgmin | minimum ignition temperature, °C |
| Tf | flash-point of liquid combustible component, °C |
| Tign | ignition temperature of liquid combustible component, °C |
| Vdaf | yield of volatiles of the sample in a dry ash-free state, % |
| Wa | moisture content of analytical sample in an air-dry state, % |
| τ | time, s |
| τd | ignition delay time, s |
References
- Malico, I.; Nepomuceno Pereira, R.; Gonçalves, A.C.; Sousa, A.M.O. Current status and future perspectives for energy production from solid biomass in the European industry. Renew. Sustain. Energy Rev. 2019, 112, 960–977. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Zawawi, N.A.; Kasim, F.H.; Inayat, A.; Khasri, A. Assessing the gasification performance of biomass: A review on biomass gasification process conditions, optimization and economic evaluation. Renew. Sustain. Energy Rev. 2016, 53, 1333–1347. [Google Scholar] [CrossRef]
- Wang, C.; Feng, Q.; Lv, Q.; Zhao, L.; Du, Y.; Wang, P.; Zhang, J.; Che, D. Numerical Investigation on Co-firing Characteristics of Semi-Coke and Lean Coal in a 600 MW Supercritical Wall-Fired Boiler. Appl. Sci. 2019, 9, 889. [Google Scholar] [CrossRef]
- AlNouss, A.; McKay, G.; Al-Ansari, T. Production of syngas via gasification using optimum blends of biomass. J. Clean. Prod. 2020, 242, 118499. [Google Scholar] [CrossRef]
- Sikarwar, V.S.; Zhao, M.; Clough, P.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.J.; Fennell, P.S. An overview of advances in biomass gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar] [CrossRef]
- Nipattummakul, N.; Ahmed, I.I.; Kerdsuwan, S.; Gupta, A.K. Hydrogen and syngas production from sewage sludge via steam gasification. Int. J. Hydrog. Energy 2010, 35, 11738–11745. [Google Scholar] [CrossRef]
- Bhuiyan, A.A.; Blicblau, A.S.; Islam, A.K.M.S.; Naser, J. A review on thermo-chemical characteristics of coal/biomass co-firing in industrial furnace. J. Energy Inst. 2018, 91, 1–18. [Google Scholar] [CrossRef]
- Kopczyński, M.; Lasek, J.A.; Iluk, A.; Zuwała, J. The co-combustion of hard coal with raw and torrefied biomasses (willow (Salix viminalis), olive oil residue and waste wood from furniture manufacturing). Energy 2017, 140, 1316–1325. [Google Scholar] [CrossRef]
- Fu, B.; Liu, G.; Mian, M.M.; Zhou, C.; Sun, M.; Wu, D.; Liu, Y. Co-combustion of industrial coal slurry and sewage sludge: Thermochemical and emission behavior of heavy metals. Chemosphere 2019, 233, 440–451. [Google Scholar] [CrossRef]
- Nyashina, G.S.; Kuznetsov, G.V.; Strizhak, P.A. Effects of plant additives on the concentration of sulfur and nitrogen oxides in the combustion products of coal-water slurries containing petrochemicals. Environ. Pollut. 2020, 258, 113682. [Google Scholar] [CrossRef]
- Peng, W.X.; Ge, S.B.; Ebadi, A.G.; Hisoriev, H.; Esfahani, M.J. Syngas production by catalytic co-gasification of coal-biomass blends in a circulating fluidized bed gasifier. J. Clean. Prod. 2017, 168, 1513–1517. [Google Scholar] [CrossRef]
- Trippe, F.; Fröhling, M.; Schultmann, F.; Stahl, R.; Henrich, E. Techno-economic assessment of gasification as a process step within biomass-to-liquid (BtL) fuel and chemicals production. Fuel Process. Technol. 2011, 92, 2169–2184. [Google Scholar] [CrossRef]
- Chyou, Y.-P.; Chang, D.-M.; Chen, P.-C.; Chien, H.-Y.; Wu, K.-T.; Chein, R.-Y. Development of Biomass Gasification Technology with Fluidized-Bed Reactors for Enhancing Hydrogen Generation: Part I, Hydrodynamic Characterization of Dual Fluidized-Bed Gasifiers. Appl. Sci. 2019, 10, 2. [Google Scholar] [CrossRef]
- AlNouss, A.; McKay, G.; Al-Ansari, T. A techno-economic-environmental study evaluating the potential of oxygen-steam biomass gasification for the generation of value-added products. Energy Convers. Manag. 2019, 196, 664–676. [Google Scholar] [CrossRef]
- Nyashina, G.S.; Kurgankina, M.A.; Strizhak, P.A. Environmental, economic and energetic benefits of using coal and oil processing waste instead of coal to produce the same amount of energy. Energy Convers. Manag. 2018, 174, 175–187. [Google Scholar] [CrossRef]
- Kurgankina, M.A.; Nyashina, G.S.; Strizhak, P.A. Advantages of switching coal-burning power plants to coal-water slurries containing petrochemicals. Appl. Therm. Eng. 2019, 147, 998–1008. [Google Scholar] [CrossRef]
- Kurgankina, M.A.; Nyashina, G.S.; Strizhak, P.A. Prospects of thermal power plants switching from traditional fuels to coal-water slurries containing petrochemicals. Sci. Total Environ. 2019, 671, 568–577. [Google Scholar] [CrossRef]
- Nyashina, G.S.; Shlegel, N.E.; Vershinina, K.Y.; Strizhak, P.A. Industrial Waste as Part of Coal-Water Slurry Fuels. Energy Fuels 2018, 32, 11398–11410. [Google Scholar] [CrossRef]
- Zaitsev, A.S.; Egorov, R.I.; Strizhak, P.A. Light-induced gasification of the coal-processing waste: Possible products and regimes. Fuel 2018, 212, 347–352. [Google Scholar] [CrossRef]
- Nyashina, G.; Strizhak, P. Impact of Forest Fuels on Gas Emissions in Coal Slurry Fuel Combustion. Energies 2018, 11, 2491. [Google Scholar] [CrossRef]
- Kumar, A.; Sah, B.; Singh, A.R.; Deng, Y.; He, X.; Kumar, P.; Bansal, R.C. A review of multi criteria decision making (MCDM) towards sustainable renewable energy development. Renew. Sustain. Energy Rev. 2017, 69, 596–609. [Google Scholar] [CrossRef]
- Haddad, B.; Liazid, A.; Ferreira, P. A multi-criteria approach to rank renewables for the Algerian electricity system. Renew. Energy 2017, 107, 462–472. [Google Scholar] [CrossRef]
- Sahu, S.G.; Chakraborty, N.; Sarkar, P. Coal-biomass co-combustion: An overview. Renew. Sustain. Energy Rev. 2014, 39, 575–586. [Google Scholar] [CrossRef]
- Sh, L.; Lee, B.-H.; Lee, Y.-J.; Jeon, C.-H. Comparing the Physicochemical Properties of Upgraded Biomass Fuel by Torrefaction and the Ashless Technique. Appl. Sci. 2019, 9, 5519. [Google Scholar] [CrossRef]
- Socci, J.; Saraeian, A.; Stefanidis, S.D.; Banks, S.W.; Shanks, B.H.; Bridgwater, T. The role of catalyst acidity and shape selectivity on products from the catalytic fast pyrolysis of beech wood. J. Anal. Appl. Pyrolysis 2019. [Google Scholar] [CrossRef]
- Johansson, A.C.; Sandström, L.; Öhrman, O.G.W.; Jilvero, H. Co-pyrolysis of woody biomass and plastic waste in both analytical and pilot scale. J. Anal. Appl. Pyrolysis 2018, 134, 102–113. [Google Scholar] [CrossRef]
- Riaza, J.; Mason, P.; Jones, J.M.; Gibbins, J.; Chalmers, H. High temperature volatile yield and nitrogen partitioning during pyrolysis of coal and biomass fuels. Fuel 2019, 248, 215–220. [Google Scholar] [CrossRef]
- Pighinelli, A.L.M.T.; Boateng, A.A.; Mullen, C.A.; Elkasabi, Y. Evaluation of Brazilian biomasses as feedstocks for fuel production via fast pyrolysis. Energy Sustain. Dev. 2014, 21, 42–50. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mohanty, K. Thermal and catalytic pyrolysis of pine sawdust (Pinus ponderosa) and Gulmohar seed (Delonix regia) towards production of fuel and chemicals. Mater. Sci. Energy Technol. 2019, 2, 139–149. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mohanty, K. Pyrolysis kinetics and thermal behavior of waste sawdust biomass using thermogravimetric analysis. Bioresour. Technol. 2018, 251, 63–74. [Google Scholar] [CrossRef]
- Kozlov, A. The study of the kinetics of formation of gaseous products during thermochemical conversion of woody biomass. E3S Web Conf. 2019, 114, 07008. [Google Scholar] [CrossRef]
- Zubkova, V.; Strojwas, A.; Bielecki, M.; Kieush, L.; Koverya, A. Comparative study of pyrolytic behavior of the biomass wastes originating in the Ukraine and potential application of such biomass. Part 1. Analysis of the course of pyrolysis process and the composition of formed products. Fuel 2019, 254, 115688. [Google Scholar] [CrossRef]
- Akubo, K.; Nahil, M.A.; Williams, P.T. Pyrolysis-catalytic steam reforming of agricultural biomass wastes and biomass components for production of hydrogen/syngas. J. Energy Inst. 2019, 92, 1987–1996. [Google Scholar] [CrossRef]
- Liu, X.; Luo, Z.; Yu, C.; Xie, G. Conversion mechanism of fuel-N during pyrolysis of biomass wastes. Fuel 2019, 246, 42–50. [Google Scholar] [CrossRef]
- Tian, H.; Jiao, H.; Cai, J.; Wang, J.; Yang, Y.; Bridgwater, A.V. Co-pyrolysis of Miscanthus Sacchariflorus and coals: A systematic study on the synergies in thermal decomposition, kinetics and vapour phase products. Fuel 2020, 262, 116603. [Google Scholar] [CrossRef]
- Pelaez-Samaniego, M.R.; Mesa-Pérez, J.; Cortez, L.A.B.; Rocha, J.D.; Sanchez, C.G.; Marín, H. Use of blends of gasoline with biomass pyrolysis-oil derived fractions as fuels in an Otto engine. Energy Sustain. Dev. 2011, 15, 376–381. [Google Scholar] [CrossRef]
- Yi, L.; Liu, H.; Li, S.; Li, M.; Wang, G.; Man, G.; Yao, H. Catalytic pyrolysis of biomass wastes over Org-CaO/Nano-ZSM-5 to produce aromatics: Influence of catalyst properties. Bioresour. Technol. 2019, 294, 122186. [Google Scholar] [CrossRef]
- Li, J.; Qiao, Y.; Zong, P.; Qin, S.; Wang, C.; Tian, Y. Fast pyrolysis characteristics of two typical coastal zone biomass fuels by thermal gravimetric analyzer and down tube reactor. Bioresour. Technol. 2019, 283, 96–105. [Google Scholar] [CrossRef]
- David, E.; Kopac, J. Pyrolysis of rapeseed oil cake in a fixed bed reactor to produce bio-oil. J. Anal. Appl. Pyrolysis 2018, 134, 495–502. [Google Scholar] [CrossRef]
- Chang, Z.; Duan, P.; Xu, Y. Catalytic hydropyrolysis of microalgae: Influence of operating variables on the formation and composition of bio-oil. Bioresour. Technol. 2015, 184, 349–354. [Google Scholar] [CrossRef]
- Li, D.; Wu, D.; Xu, F.; Lai, J.; Shao, L. Literature overview of Chinese research in the field of better coal utilization. J. Clean. Prod. 2018, 185, 959–980. [Google Scholar] [CrossRef]
- Zhao, C.; Luo, K. Sulfur, arsenic, fluorine and mercury emissions resulting from coal-washing byproducts: A critical component of China’s emission inventory. Atmos. Environ. 2017, 152, 270–278. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, Y.; Liu, L.; Wang, X.; Zhang, Z. Environmental investigation on co-combustion of sewage sludge and coal gangue: SO2, NOx and trace elements emissions. Waste Manag. 2016, 50, 213–221. [Google Scholar] [CrossRef] [PubMed]
- McIlveen-Wright, D.R.; Pinto, F.; Armesto, L.; Caballero, M.A.; Aznar, M.P.; Cabanillas, A.; Huang, Y.; Franco, C.; Gulyurtlu, I.; McMullan, J.T. A comparison of circulating fluidised bed combustion and gasification power plant technologies for processing mixtures of coal, biomass and plastic waste. Fuel Process. Technol. 2006, 87, 793–801. [Google Scholar] [CrossRef]
- Haykiri-Acma, H.; Turan, A.Z.; Yaman, S.; Kucukbayrak, S. Controlling the excess heat from oxy-combustion of coal by blending with biomass. Fuel Process. Technol. 2010, 91, 1569–1575. [Google Scholar] [CrossRef]
- Staroń, A.; Kowalski, Z.; Staroń, P.; Banach, M. Analysis of the useable properties of coal-water fuel modified with chemical compounds. Fuel Process. Technol. 2016, 152, 183–191. [Google Scholar] [CrossRef]
- Yanik, J.; Duman, G.; Karlström, O.; Brink, A. NO and SO2 emissions from combustion of raw and torrefied biomasses and their blends with lignite. J. Environ. Manag. 2018, 227, 155–161. [Google Scholar] [CrossRef]
- Burdukov, A.P.; Popov, V.I.; Tomilov, V.G.; Fedosenko, V.D. The rheodynamics and combustion of coal–water mixtures. Fuel 2002, 81, 927–933. [Google Scholar] [CrossRef]
- Daood, S.S.; Ord, G.; Wilkinson, T.; Nimmo, W. Fuel additive technology—NOx reduction, combustion efficiency and fly ash improvement for coal fired power stations. Fuel 2014, 134, 293–306. [Google Scholar] [CrossRef]
- Shahzad, K.; Saleem, M.; Kazmi, M.; Ali, Z.; Hussain, S.; Akhtar, N.A. Effect of Hydrodynamic Conditions on Emissions of NOx, SO2, and CO from Co-Combustion of Wheat Straw and Coal Under Fast Fluidized Bed Condition. Combust. Sci. Technol. 2016, 188, 1303–1318. [Google Scholar] [CrossRef]
- Zhao, B.; Su, Y.; Liu, D.; Zhang, H.; Liu, W.; Cui, G. SO2/NOx emissions and ash formation from algae biomass combustion: Process characteristics and mechanisms. Energy 2016, 113, 821–830. [Google Scholar] [CrossRef]
- Rokni, E.; Ren, X.; Panahi, A.; Levendis, Y.A. Emissions of SO2, NOx, CO2, and HCl from Co-firing of coals with raw and torrefied biomass fuels. Fuel 2018, 211, 363–374. [Google Scholar] [CrossRef]
- Armesto, L.; Bahillo, A.; Cabanillas, A.; Veijonen, K.; Otero, J.; Plumed, A.; Salvador, L. Co-combustion of coal and olive oil industry residues in fluidised bed. Fuel 2003, 82, 993–1000. [Google Scholar] [CrossRef]
- Zhang, Z.; Zeng, Q.; Hao, R.; He, H.; Yang, F.; Mao, X.; Mao, Y.; Zhao, P. Combustion behavior, emission characteristics of SO2, SO3 and NO, and in situ control of SO2 and NO during the co-combustion of anthracite and dried sawdust sludge. Sci. Total Environ. 2019, 646, 716–726. [Google Scholar] [CrossRef]
- Glushkov, D.O.; Paushkina, K.K.; Shabardin, D.P. Co-combustion of coal processing waste, oil refining waste and municipal solid waste: Mechanism, characteristics, emissions. Chemosphere 2020, 240, 124892. [Google Scholar] [CrossRef]
- McKay, G. Dioxin characterisation, formation and minimisation during municipal solid waste (MSW) incineration: Review. Chem. Eng. J. 2002, 86, 343–368. [Google Scholar] [CrossRef]
- Mukherjee, A.; Debnath, B.; Ghosh, S.K. A Review on Technologies of Removal of Dioxins and Furans from Incinerator Flue Gas. Procedia Environ. Sci. 2016, 35, 528–540. [Google Scholar] [CrossRef]
- Goering, C.E.; Schwab, A.W.; Daugherty, M.J.; Pryde, E.H.; Heakin, A.J. Fuel Properties of Eleven Vegetable Oils. Trans. ASAE 1982, 25, 1472–1477. [Google Scholar] [CrossRef]
- BP Statistical Review of World Energy 2019; Pureprint Group Limited: London, UK, 2019.
- Cho, M.J.; Choi, Y.S.; Paik, S.H.; Mun, H.S.; Cha, D.S.; Han, S.K.; Oh, J.H. Comparison of productivity and cost between two integrated harvesting systems in South Korea. Forests 2019, 10, 763. [Google Scholar] [CrossRef]
- Perea-Moreno, M.A.; Manzano-Agugliaro, F.; Perea-Moreno, A.J. Sustainable energy based on sunflower seed husk boiler for residential buildings. Sustain. 2018, 10, 3407. [Google Scholar] [CrossRef]
| Biomass | Country | Ultimate Analysis (wt %) | Proximate Analysis (wt %) | Ref. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | H | O | N | S | Moisture | Volatile Matter | Fixed Carbon | Ash | Heat of Combustion (MJ/kg) | |||
| Wood biomass | ||||||||||||
| Beech sawdust | Europe, Russia | 44.1 | 6.3 | 49.4 | 0.2 | 0 | 4.7 | 87.6 | 8 | 0.8 | 19.5 | [25] |
| Woody biomass (pine, fir) | Sweden | 51.6 | 6.2 | 42 | 0.1 | 0.021 | 4.3 | 83.8 | – | 0.3 | 19.36 | [26] |
| Wood pellet | UK | 52.34 | 6.8 | 40.7 | 0.16 | – | 6.7 | 84.3 | 15.7 | 0.8 | 20.8 | [27] |
| Eucalyptus wood | Brazil | 56.38 | 4.77 | 38.78 | 0.07 | – | 11.56 | 74.97 | 12.98 | 0.45 | 17.91 | [28] |
| Pine sawdust | India | 56.53 | 6.93 | 32.55 | 3.33 | 0.66 | 7.85 ± 0.05 | 77.27 ± 0.65 | 12.20 ± 0.15 | 2.78 ± 0.12 | 18.55 ± 0.43 | [29] |
| Pine sawdust | India | 50.32 | 6 | 42.99 | 0.69 | – | 6.09 ± 0.3 | 78.03 ± 0.2 | 12.16 ± 0.1 | 2.07 ± 0.03 | 18.44 ± 09 | [30] |
| Pine sawdust | Russian | 54.4 | 5.2 | 40.0 | 0.4 | – | 3.5 | 80.1 | 15.1 | 1.1 | – | [31] |
| Pine sawdust | Korea | 46.92 | 5.78 | 46.73 | 0.08 | 0.49 | 8.31 | 79.41 | 19.46 | 1.13 | – | [24] |
| Agricultural biomass | ||||||||||||
| Wheat straw | Ukraine | 51.5 | 5.75 | 41.97 | 0.65 | 0.13 | 6.84 | – | – | 11.59 ± 0.76 | 16.12 ± 0.19 | [32] |
| Wheat straw | UK | 40.58 | 4.84 | 53.84 | 0.74 | 0 | 5.19 | 64.24 | 15.60 | 14.97 | – | [33] |
| Wheat straw | China | 54.08 | 5.83 | 39.08 | 0.56 | 0.45 | 2.50 | 72.36 | 18.00 | 5.64 | – | [34] |
| Rice straw | China | 55.48 | 5.68 | 37.37 | 1.03 | 0.44 | 1.51 | 69.09 | 18.09 | 11.31 | – | [34] |
| Rice Husk | China | 37.6 | 5.26 | 55.45 | 1.69 | 0 | 8.02 | 61.43 | 12.53 | 18.02 | – | [33] |
| Energy crops | Northeast Asia, China | 50.73 | 7.08 | 41.95 | 0.14 | 0.10 | 10.67 | 65.65 | 18.34 | 5.34 | 17.00 | [35] |
| Sugar cane bagasse | India | 49.4 | 6.2 | 43.2 | 0.4 | 0.8 | 10 | 76 | 9.6 | 4.4 | 17.2 | [30] |
| Waste of sugar cane bagasse | Brazil | 44.7 | 5.8 | 48.97 | 0.45 | 0.08 | 9.92 | 81.55 | 6.90 | 11.57 | 17.74 | [36] |
| Corn stalks | Ukraine | 48.84 | 5.40 | 44.08 | 1.68 | 0.16 | 8.13 | – | – | 12.30 ± 0.87 | 14.24 ± 0.46 | [32] |
| Corn stalks | China | 46.6 | 5.8 | 47.0 | 0.4 | 0.2 | – | 86.9 | 11.8 | 1.3 | – | [37] |
| Corn waste (cob) | India | 49.28 | 5.9 | 44.2 | 0.54 | 0.08 | 10.2 | 80 | 4.2 | 5.7 | 15.5 | [30] |
| Jerusalem artichoke stalks | China | 45.36 | 6.11 | 47.26 | 0.75 | 0.52 | 15.76 | 67.4 | 13.5 | 3.34 | 15.69 | [38] |
| Oilseed waste | ||||||||||||
| Palm fruit bunches | United Arab Emirates | 44.06 | 5.97 | 49.05 | 0.74 | 0.18 | 8.73 | 67.51 | 17.47 | 6.28 | 17.2 | [5] |
| Date palm waste (leaves) | United Arab Emirates | 40.75 | 5.55 | 52.14 | 1.32 | 0.24 | 12.03 | 58.17 | 15.41 | 14.4 | 18.9 | [5] |
| Palm kernel cake | China | 57.22 | 5.93 | 34.10 | 2.46 | 0.29 | 2.88 | 75.83 | 15.99 | 5.30 | – | [34] |
| Sunflower husks | Ukraine | 52.76 | 6.32 | 38.31 | 2.61 | 0.14 | 6.1 | – | – | 6.81 ± 0.51 | 19.31 ± 0.13 | [32] |
| Seeds de-oil cake | China | 45.3 | 6.2 | 43.8 | 4.5 | 0.2 | – | 73.5 | 18.2 | 7.3 | – | [37] |
| Olive waste | UK | 52.8 | 6.5 | 39.1 | 1.6 | – | 5.9 | 80.1 | 19.9 | 7.6 | 20.1 | [27] |
| Rapeseed oil cake | Romania | 45.95 | 6.21 | 40.17 | 6.76 | 0.91 | 10.02 | 67.84 | 15.47 | 6.77 | 25.41 | [39] |
| Shell and husk | ||||||||||||
| Nut shell (areca) | India | 48.71 | 5.79 | 43.45 | 1.95 | 0.1 | 7.43 ± 0.1 | 74.05 ± 0.2 | 15.55 ± 0.3 | 2.48 ± 0.05 | 18.21 ± 09 | [30] |
| Coconut shell | UK | 48.31 | 5.26 | 46.14 | 0.29 | 0 | 7.16 | 68.58 | 22.00 | 2.26 | – | [33] |
| Walnut shell | Ukraine | 51.56 | 5.66 | 48.44 | 1.98. | 0.11. | 4.1 | – | – | 0.41 ± 0.11 | 16.79 ± 0.08 | [32] |
| Peanut shell | China | 49.8 | 5.8 | 43.7 | 0.6 | 0.1 | – | 84.1 | 14.5 | 1.4 | – | [37] |
| Palm kernel shell | UK | 50.1 | 6.24 | 42.16 | 1.50 | 0 | 6.70 | 67.52 | 22.13 | 3.65 | – | [33] |
| Other | ||||||||||||
| Algae | China | 58.9 | 7.0 | 25.4 | 8.2 | 0.5 | 10 | 81 | 16 | 9 | – | [40] |
| Cotton stalk | India | 46.3 | 6.4 | 46.8 | 0.3 | 0.2 | 8.9 | 71 | 16.6 | 3.5 | 19.2 | [30] |
| Component | Proximate Analysis | Ultimate Snalysis | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Water Fraction, % | Ad, % | Vdaf, % | Qas, MJ/kg | Cdaf, % | Hdaf, % | Ndaf, % | Std, % | Odaf, % | |
| Solid components in dry state | |||||||||
| Coal | 2.05 | 14.65 | 27.03 | 29.76 | 80.10 | 4.49 | 1.84 | 0.87 | 12.70 |
| Filter cake | 43.5 | 26.46 | 23.08 | 24.83 | 87.38 | 5.09 | 2.05 | 1.02 | 4.46 |
| Leaves (birch) | 6.95 | 6.25 | 76.85 | 17.05 | 49.91 | 5.92 | 0.86 | 0.09 | 43.22 |
| Straw (wheat) | 7.0 | 2.8 | 78.5 | 17.7 | 50.20 | 6.36 | 1.09 | traces | 42.35 |
| Sunflower cake | 11.5 | 5.9 | 81 | 16.6 | 48.44 | 6.92 | 3.05 | traces | 41.59 |
| Sawdust (pine) | 7.0 | 1.6 | 83.4 | 18.1 | 52.5 | 6.58 | 0.22 | traces | 40.70 |
| Liquid component | |||||||||
| Tf, oC | Tign, oC | Qas, MJ/kg | C, % | H, % | N, % | O, % | S, % | ||
| Rapeseed oil | 225 | 260 | 42.1 | 79.6 | 11.4 | 0 | 8.97 | 0.03 | |
| No. of Fuel Composition | Fuel Components |
|---|---|
| 1 | 100% coal |
| 2 | 90% filter cake, 10% sawdust |
| 3 | 90% filter cake, 10% rapeseed oil |
| 4 | 90% filter cake, 10% leaves |
| 5 | 90% filter cake, 10% sunflower cake |
| 6 | 90% filter cake, 10% straw |
| 7 | 100% filter cake |
| Group | Criteria |
|---|---|
| Energy performance | Ignition delay times, τd (s) |
| Maximum combustion temperature, Tbmax (°C) | |
| Minimum ignition temperature, Tgmin (°C) | |
| Heat of combustion, Qas (MJ/kg) | |
| Ash content, Ad (%) | |
| Environmental | Nitrogen oxide emissions, NOx (ppm) |
| Sulfur oxide emissions, SOx (ppm) | |
| Economic | Fuel cost, C (USD/kg) |
| Capital costs, N (USD) |
| Criteria | Fuel * | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | |||||||||
| abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | ||
| τd, s | at 700 °C | 8.3 | 1 | 10.97 | 0.76 | 23 | 0.36 | 12.9 | 0.64 | 11.3 | 0.74 | 12 | 0.69 | 12.2 | 0.68 |
| at 800 °C | 6.7 | 1 | 8.87 | 0.76 | 14 | 0.48 | 10 | 0.67 | 8.97 | 0.75 | 9.5 | 0.71 | 11.3 | 0.59 | |
| at 900 °C | 2.5 | 1 | 8.33 | 0.30 | 11.5 | 0.22 | 7.5 | 0.33 | 7.36 | 0.34 | 8 | 0.31 | 9.5 | 0.26 | |
| Tbmax, °C | 1350 | 1 | 987 | 0.73 | 1020 | 0.76 | 930 | 0.69 | 885 | 0.66 | 896 | 0.66 | 1025 | 0.76 | |
| Tgmin, °C | 530 | 0.84 | 445 | 1 | 475 | 0.94 | 461 | 0.96 | 449 | 0.99 | 454 | 0.98 | 448 | 0.99 | |
| Qas, MJ/kg | 29.76 | 1 | 14.55 | 0.49 | 18.96 | 0.64 | 14.44 | 0.49 | 14.4 | 0.34 | 14.51 | 0.49 | 14,15 | 0.47 | |
| Ad, % | 14.65 | 0.79 | 13.61 | 0.85 | 11.64 | 1 | 14.08 | 0.83 | 14.04 | 0.83 | 13.73 | 0.85 | 14.95 | 0.78 | |
| NOx, ppm | at 700 °C | 140 | 0.41 | 80 | 0.71 | 57 | 1 | 78 | 0.73 | 84 | 0.68 | 104 | 0.55 | 80 | 0.71 |
| at 800 °C | 179 | 0.45 | 100 | 0.8 | 80 | 1 | 130 | 0.53 | 108 | 0.74 | 122 | 0.66 | 157 | 0.51 | |
| at 900 °C | 320 | 0.38 | 165 | 0.73 | 120 | 1 | 180 | 0.57 | 130 | 0.92 | 150 | 0.8 | 242 | 0.49 | |
| SOx, ppm | at 700 °C | 65 | 0.11 | 25 | 0.28 | 14.5 | 0.48 | 8 | 0.88 | 15 | 0.47 | 7 | 1 | 17 | 0.41 |
| at 800 °C | 205 | 0.10 | 48 | 0.42 | 23 | 0.87 | 25 | 0.8 | 40 | 0.5 | 20 | 1 | 55 | 0.36 | |
| at 900 °C | 274 | 0.20 | 78 | 0.72 | 56 | 1 | 77 | 0.73 | 64 | 0.88 | 75 | 0.75 | 98 | 0.57 | |
| C, USD/kg | 0.08 | 0.125 | 0.019 | 0.52 | 0.090 | 0.11 | 0.013 | 0.75 | 0.016 | 0.62 | 0.012 | 0.85 | 0.01 | 1 | |
| N, USD | 665.000 | 0.43 | 290.000 | 1 | 290.000 | 1 | 290.000 | 1 | 290.000 | 1 | 290.000 | 1 | 290.000 | 1 | |
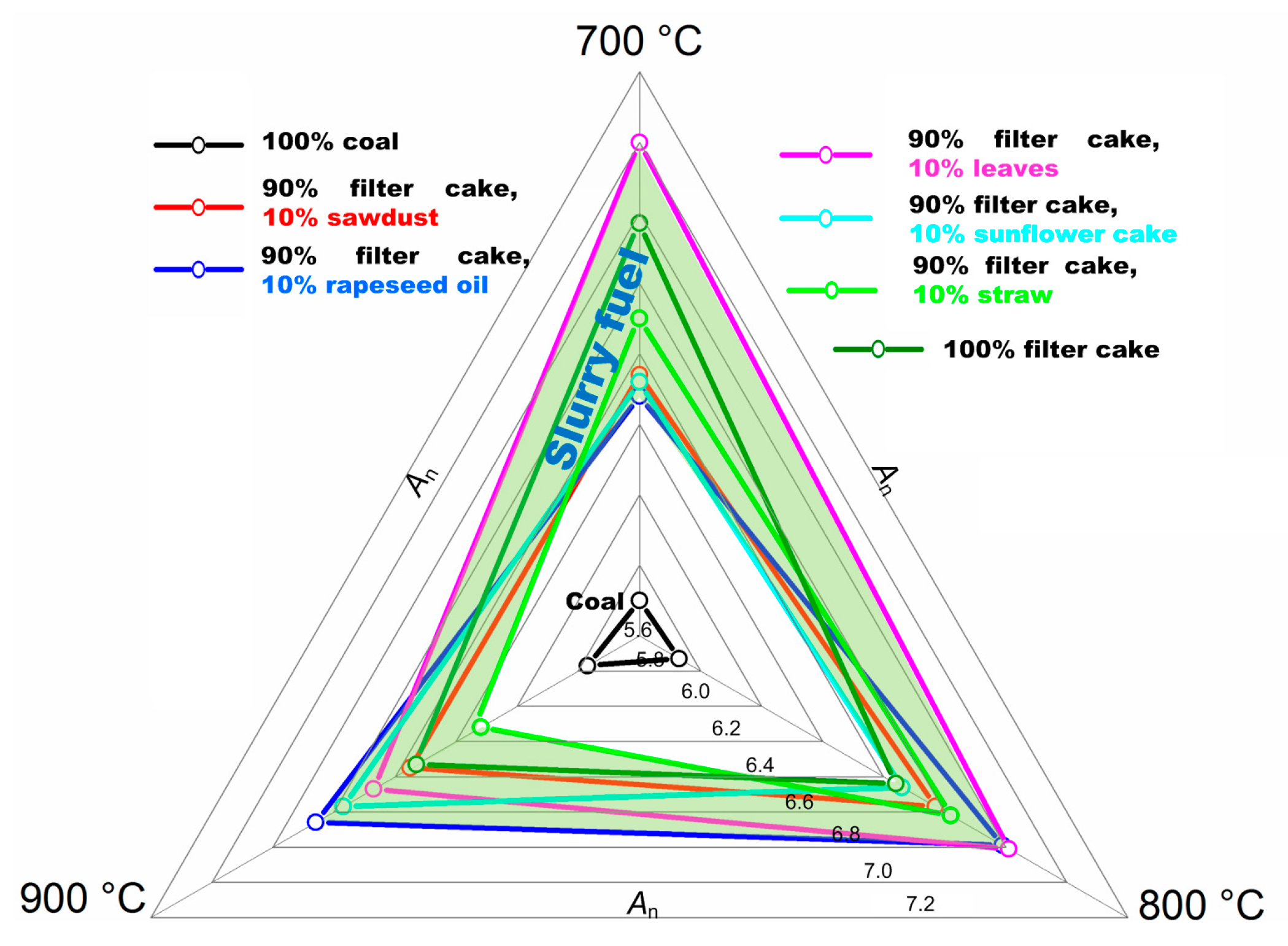
| Temperature | An | ||||||
|---|---|---|---|---|---|---|---|
| 100% Coal | 90% Filter Cake, 10% Sawdust | 90% Filter Cake, 10% Rapeseed Oil | 90% Filter Cake, 10% Leaves | 90% Filter Cake, 10% Sunflower Cake | 90% Filter Cake, 10% Straw | 100% Filter Cake | |
| 700 °C | 5.70 | 6.34 | 6.28 | 7.0 | 6.32 | 6.50 | 6.77 |
| 800 °C | 5.73 | 6.57 | 6.79 | 6.81 | 6.46 | 6.62 | 6.44 |
| 900 °C | 5.77 | 6.35 | 6.66 | 6.47 | 6.57 | 6.12 | 6.33 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glushkov, D.; Nyashina, G.; Medvedev, V.; Vershinina, K. Relative Environmental, Economic, and Energy Performance Indicators of Fuel Compositions with Biomass. Appl. Sci. 2020, 10, 2092. https://doi.org/10.3390/app10062092
Glushkov D, Nyashina G, Medvedev V, Vershinina K. Relative Environmental, Economic, and Energy Performance Indicators of Fuel Compositions with Biomass. Applied Sciences. 2020; 10(6):2092. https://doi.org/10.3390/app10062092
Chicago/Turabian StyleGlushkov, Dmitrii, Galina Nyashina, Valeriy Medvedev, and Kseniya Vershinina. 2020. "Relative Environmental, Economic, and Energy Performance Indicators of Fuel Compositions with Biomass" Applied Sciences 10, no. 6: 2092. https://doi.org/10.3390/app10062092
APA StyleGlushkov, D., Nyashina, G., Medvedev, V., & Vershinina, K. (2020). Relative Environmental, Economic, and Energy Performance Indicators of Fuel Compositions with Biomass. Applied Sciences, 10(6), 2092. https://doi.org/10.3390/app10062092






