Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals
Abstract
1. Introduction
2. Materials
2.1. Preparation of Extracts
2.2. Extraction Yield of Each Extract of C. sativus L Petals
2.3. Estimation of Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
2.4. In-vitro Antioxidant Activity
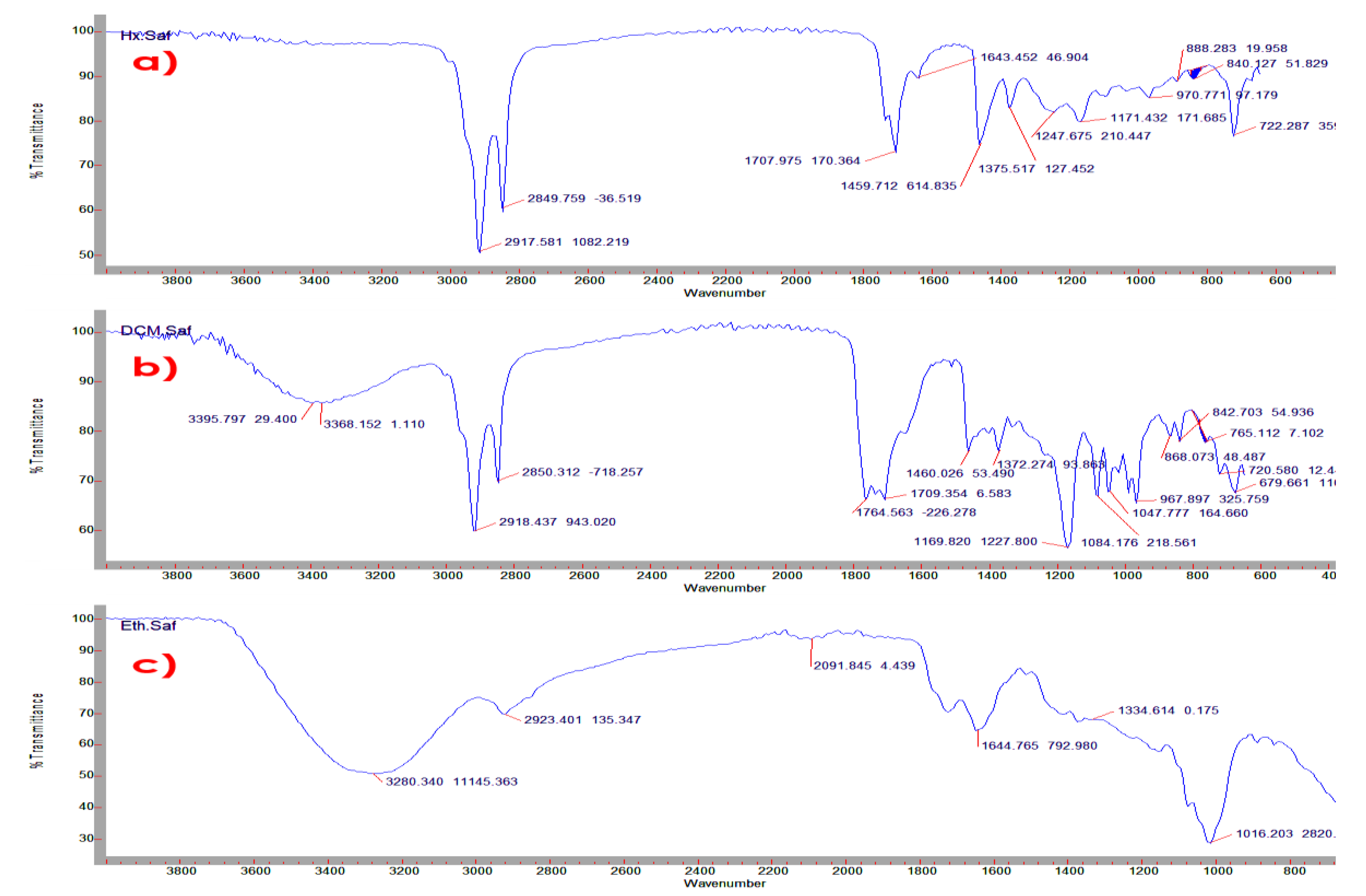
2.5. Fourier Transform Infrared Spectrometry (FTIR) Analysis of C. sativus L. Petals Extracts
2.6. Antimicrobial Activity
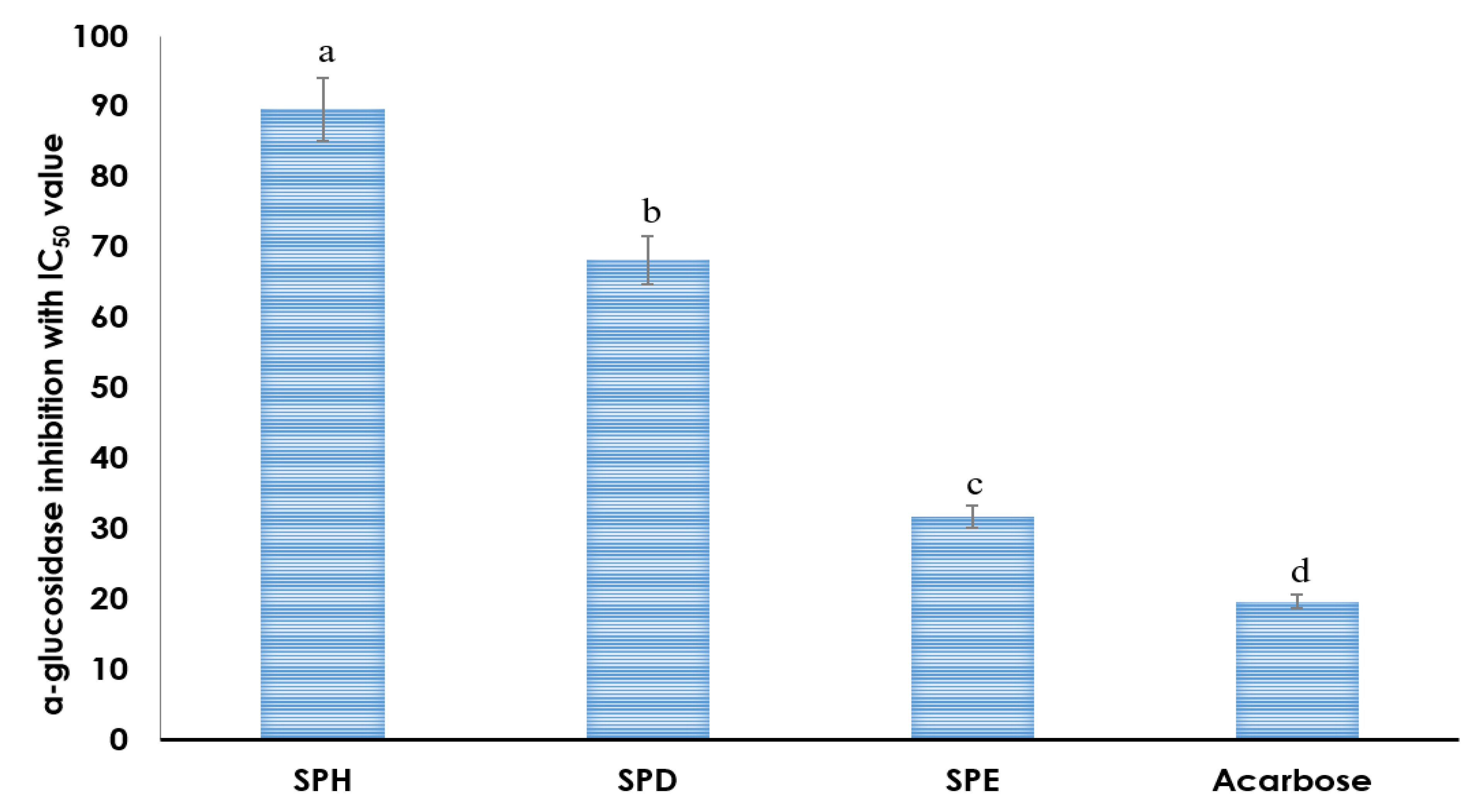
2.7. Antidiabetic Study
2.8. Cytotoxic Activity
3. Statistical Analysis
4. Results
4.1. Extraction Yield:
4.2. Estimation of Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
4.3. In-Vitro Antioxidant Activity
4.4. Identified Functional Groups in the Extracts
4.5. Antimicrobial Activity
4.6. Antidiabetic Study
4.7. Cytotoxic Activity
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SPH | Saffron petals hexane extract |
| SPD | Saffron petals dichloromethane extract |
| SPE | Saffron petals ethanol extract |
| TPC | phenolic content |
| TFC | Total flavonoid content |
| GAE/g | Gallic acid equivalent per gram |
| QE/g | Quercetin equivalent per gram |
| DPPH | 2, 2-diphenyl-1-picry1hydrazy1 |
| ABTS | (2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| BHA | Butylated hydroxyanisole. |
| R2 | Regression |
| IC50 | Half maximal inhibitory concentration |
| FTIR | Fourier transform infrared spectrometry |
| ATCC | American Type Culture Collection |
| NCPF | National Collection of Pathogenic Fungi |
| NCCLS | National Committee for Clinical Laboratory Standards |
| DMSO | Dimethyl sulfoxide |
| µg/mL | microgram/mL |
| °C | Celsius |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| MDA-MB-231 | Triple negative breast cancer cell line |
| LC-ESI-Q-TOFMS | Liquid chromatography coupled with electrospray ionization-quadrupole-time of flight-mass spectrometry |
References
- World Health Organization (WHO). List of Blueprint Priority Diseases. 2018. Available online: www.who.int/blueprint/priority-diseases/en/ (accessed on 7 February 2019).
- Cosgrove, S.E.; Qi, Y.; Kaye, K.S.; Harbarth, S.; Karchmer, A.W.; Carmeli, Y. The impact of methicillin resistance in Staphylococcus aureus bacteremia on patient outcomes: Mortality, length of stay, and hospital charges Infect. Control. Hosp. Epidemiol. 2005, 26, 166–174. [Google Scholar] [CrossRef]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. P & T Peer-Rev. J. Formul. Manag. 2015, 40, 277–283. [Google Scholar]
- Jesus, G.D.; Emmy, T.; Julio, C.E.A.; Gabriel, L.M.; Paul, C.; Luc, P. Antimicrobial activity of leaf extracts and isolated constituents of Croton linearis. J. Ethnopharmacol. 2019, 236, 250–257. [Google Scholar]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Rasool, M.H.; Salamat, M.K.F. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645–1658. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.U.; Wali, A.F.; Ahmad, A.; Shakeel, S.; Rasool, S.; Ali, R.; Khan, R. Neuroprotective Strategies for Neurological Disorders by Natural Products: An update. Curr. Neuropharmacol. 2018, 17, 247–267. [Google Scholar] [CrossRef] [PubMed]
- Mehlhorn, H. The Continued Rise in the Magnitude of Bacterial Resistance to Traditional Antibiotics is a Concern of Global Significance, 6th ed.; Encyclopedic Reference of Parasitology; Springer Press: Berlin, Germany, 2014. [Google Scholar]
- Verica, A.; Sabo, P.K. Antimicrobial activity of Eucalyptus camaldulensis Dehn. plant extracts and essential oils: A review. Ind. Crops Prod. 2019, 32, 413–429. [Google Scholar]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Othman, L.; Sleiman, A.; Abdel-Massih, R.M. Antimicrobial Activity of Polyphenols and Alkaloids in Middle Eastern Plants. Front. Microbiol. 2019, 10, 911. [Google Scholar] [CrossRef] [PubMed]
- Dorman, H.; Deans, S. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2008, 88, 308–316. [Google Scholar] [CrossRef]
- Eruygur, N.; Koçyigit, U.M.; Taslimi, P.; Atas, M.; Tekinf, M.; Gulçin, I. Screening the in vitro antioxidant, antimicrobial, anticholinesterase, antidiabetic activities of endemic Achillea cucullata (Asteraceae) ethanol extract. S. Afr. J. Bot. 2019, 120, 141–145. [Google Scholar] [CrossRef]
- Lim, T.K. Edible Medicinal and Non Medicinal Plants. Flowers Crocus sativus; Springer: Amsterdam, The Netherlands; New York, NY, USA; London, UK, 2014; Volume 8. [Google Scholar]
- Melnyk, J.P.; Wang, S.; Marcone, M.F. Chemical and biological properties of the world’s most expensive spice: Saffron. Food Res. Int. 2010, 43, 1981–1989. [Google Scholar] [CrossRef]
- Liakopoulou-Kyriakides, M.; Kyriakidis, D. Crocus sativus biological active constituents. Stud. Nat. Prod. Chem. 2002, 16, 293–312. [Google Scholar]
- Carmona, M.; Zalacain, A.; Salinas, M.R.; Alonso, G.L. A new approach to saffron aroma. CRC Crit. Rev. Food Sci. Nutr. 2007, 47, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Bagri, J.; Yadav, A.; Anwar, K.; Dkhar, J.; Singla-Pareek, S.L.; Pareek, A. Metabolic shift in sugars and amino acids regulates sprouting in Saffron corm. Sci. Rep. 2017, 7, 11904–11914. [Google Scholar] [CrossRef]
- Mokhtari-Zaer, A.; Khazdair, M.R.; Boskabady, M.H. Smooth muscle relaxant activity of Crocus sativus (saffron) and its constituents: Possible mechanisms. Avicenna J. Phytomed. 2015, 5, 365–375. [Google Scholar]
- Rahaie, S.; Gharibzahedi, S.M.T.; Razavi, S.H.; Jafari, S.M. Recent developments on new formulations based on nutrient-dense ingredients for the production of healthy-functional bread: A review. J. Food Sci. Technol. 2012, 51, 2896–2906. [Google Scholar] [CrossRef]
- Mollaei, H.; Safaralizadeh, R.; Babaei, E.; Abedini, M.R.; Hoshyar, R. The anti-proliferative and apoptotic effects of crocin on chemosensitive and chemoresistant cervical cancer cells. Biomed. Pharmacother. 2017, 94, 307–316. [Google Scholar] [CrossRef]
- Abdel-Rahman, R.F.; El Awdan, S.A.; Hegazy, R.R.; Mansour, D.F.; Ogaly, H.A.; Abdelbaset, M. Neuroprotective effect of Crocus sativus against cerebral ischemia in rats. Metab. Brain Dis. 2019, 1–13. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Samini, F. Ameliorative effect of saffron aqueous extract on hyperglycemia, hyperlipidemia, and oxidative stress on diabetic encephalopathy in streptozotocin induced experimental diabetes mellitus. BioMed Res. Int. 2014, 1–12. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Ziaee, T.; Sadeghi, A. The effect of saffron, Crocus sativus stigma, extract and its constituents, safranal and crocin on sexual behaviors in normal male rats. Phytomedicine 2008, 15, 491–495. [Google Scholar] [CrossRef]
- Asdaq, S.M.B.; Inamdar, M.N. Potential of Crocus sativus (saffron) and its constituent, crocin, as hypolipidemic and antioxidant in rats. Appl. Biochem. Biotechnol. 2010, 162, 358–372. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Zhong, Y.; Qi, Y.; Cheng, W.; Li, S.; Yan, X. Wang Anti-arthritic effects of crocin in interleukin-1β-treated articular chondrocytes and cartilage in a rabbit osteoarthritic model. Inflamm. Res. 2013, 62, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Javid, A.P.; Azra, N.K.; Rehana, H.; Zafar, A.R.; Raies, A.Q. Antibacterial and antioxidant activity of methanol extracts of Crocus sativus L. c.v. Kashmirianus. Front. Life Sci. 2014, 8, 40–46. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoids content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Kaur, C.; Dalamu, M.; Singh, S.; Walia, S.; Joshi, A.D. Variations in phenolics and antioxidants in Indian onions (Allium cepa L.) Genotype selection for breeding. Nutr. Food Sci. 2004, 40, 6–19. [Google Scholar]
- Wang, D.M.; Zhang, Y.J.; Wang, S.S.; Li, D.W. Antioxidant and antifungal activities of extracts and fractions from Anemone taipaiensis, China. Allelopath. J. 2013, 32, 67–68. [Google Scholar]
- Tan, X.Q.; Guo, L.J.; Chen, H.S.; Wu, L.S.; Kong, F.F. Study on the flavonoids constituents of Trachelospermum jasminoides. J. Chin. Med. Mater. 2010, 33, 58–60. [Google Scholar]
- Rahman, A.A.; Samoylenko, V.; Jain, S.K.; Tekwani, B.L.; Khan, S.I.; Jacob, M.R.; Muhammad, I. Antiparasitic and antimicrobial isoflavanquinones from Abrus schimperi. Nat. Prod. Commun. 2012, 6, 1645–1650. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, Y.; Cheng, Y.; Wang, Y. Rapid screening and identification of α- glucosidase inhibitors from mulberry leaves using enzyme-immobilizedmagnetic beads coupled with HPLC/MS and NMR. BioMed Chromatogr. 2013, 27, 148–155. [Google Scholar] [CrossRef]
- Yusra, A.; Ali, E.; Synan, A.Q.; Samir, A.; Mohammad, K.; Ghenima, A.; Soleiman, H.; Rabah, I. Mitotic Arrest and Apoptosis in Breast Cancer Cells Induced by Origanum majorana Extract: Upregulation of TNF-α and Downregulation of Survivin and Mutant p53. PLoS ONE 2013, 8, e56649. [Google Scholar]
- Mzabri, I.; Addi, M.; Berrichi, A. Traditional and Modern Uses of Saffron (Crocus Sativus). Cosmetics 2019, 6, 63. [Google Scholar] [CrossRef]
- Somayeh, R.; Sohrab, M.; Maryam, H.; Seyed, A.S. Evaluation of antioxidant activities of bioactive compounds and various extracts obtained from saffron (Crocus sativus L.): A review. J. Food Sci. Technol. 2015, 52, 1881–1888. [Google Scholar]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef] [PubMed]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z. Evaluation of Crocus sativus L. stigma phenolic and flavonoid compounds and its antioxidant activity. Molecules 2010, 6, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
- Duangjai, T.; Areeya, T.; Apinan, P.; Aujana, Y. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Forni, C.; Facchiano, F.; Bartoli, M. Beneficial Role of Phytochemicals on Oxidative Stress and Age-Related Diseases. BioMed Res. Int. 2019, 7, 8748253. [Google Scholar] [CrossRef]
- Nowacka, N.; Nowak, R.; Drozd, M.; Olech, M.; Los, R.; Malm, A. Antibacterial, antiradical potential and phenolic compounds of thirty-one polish mushrooms. PLoS ONE 2015, 10, 0140355. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Baptista, P.; Vilas-Boas, M.; Barros, L. Free-radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal: Individual cap and stipe activity. Food Chem. 2007, 100, 511–1516. [Google Scholar] [CrossRef]
- Mathew, S.; Abraham, T.E.; Zakaria, Z.A. Reactivity of phenolic compounds towards free radicals under in vitro conditions. J. Food Sci. Technol. 2015, 52, 5790–5798. [Google Scholar] [CrossRef]
- Baba, S.A.; Malik, A.H.; Wani, Z.A.; Mohiuddin, T.; Shah, Z.; Abbas, N.; Ashraf, N. Phytochemical analysis and antioxidant activity of different tissue types of Crocus sativus and oxidative stress alleviating potential of saffron extract in plants, bacteria, and yeast. S. Afr. J. Bot. 2015, 99, 80–87. [Google Scholar] [CrossRef]
- Lahmass, I.; Ouahhoud, S.; Elmansuri, M.; Sabouni, A.; Elyoubi, M.; Benabbas, R.; Saalaoui, E. Determination of Antioxidant Properties of Six By-Products of Crocus sativus L. (Saffron) Plant Products. Waste Biomass Valoriz. 2018, 8, 1349–1357. [Google Scholar] [CrossRef]
- Fei, Y.L.; Thet, T.H.; Gabriel, A.A. ATR-FTIR and spectrometric methods for the assay of Crocin in commercial saffron spices (Crocus savitus L.). Int. J. Food Prop. 2015, 18, 1773–1783. [Google Scholar]
- Maryam, A.; Afshin, R.; Seyed, S.S.; Hamdollah, M. The effects of aqueous and alcoholic saffron (crocus sativus) tepal extracts on quality and shelflife of pacific white shrimp (litopeneous vannamei) during iced storage. J. Food Qual. 2016, 39, 732–742. [Google Scholar]
- Cenci-Goga, B.; Torricelli, R.; Hosseinzadeh, G.Y. In vitro bactericidal activities of various extracts of saffron (Crocus sativus L.) stigmas from Torbat-e Heydarieh, Gonabad and Khorasan, Iran. Microbiol. Res. 2018, 9. [Google Scholar] [CrossRef]
- Wali, A.F.; Pillai, J.R.; Al Dhaheri, Y.; Rehman, M.U.; Shoaib, A.; Sarheed, O.; Ahmad, P. Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury. Plants 2020, 9, 167. [Google Scholar] [CrossRef] [PubMed]
- Syed, M.; Sajad, A.R.; Khaliquz, Z.K. In vitro bactericidal and fungicidal activities of various extracts of saffron (Crocus sativus L.) stigmas from Jammu & Kashmir, India. Cogent. Food Agric. 2016, 2, 1158999. [Google Scholar]
- Eleni, K.; Dimitra, D.; Spiros, P.; Konstantina, A.; Eleftherios, H.D.; Moschos, G.P. Crocus sativus L. tepals: The natural source of antioxidant and antimicrobial factors. J. Appl. Res. Med. Aromat. Plants 2017, 4, 66–74. [Google Scholar]
- Carmona, M.; Zalacain, A.; Alonso, G.L. The Chemical Composition of Saffron: Colour, Taste and Aroma, 1st ed.; Bomarzo: Albacet, Spain, 2006. [Google Scholar]
- Gandomi, H.; Misaghi, A.; Abbaszadeh, S.; Azami, L.; Shariatifar, N.; Tayyar, N. Antibacterial effect of aqueous and alcoholic extracts from petal of saffron (Crocus sativus) on some foodborne bacterial pathogens. J. Med. Plants 2012, 11, 189–196. [Google Scholar]
- Amin, M.U.; Khurram, M.; Khattak, B.; Khan, J. Antibiotic additive and synergistic action of rutin, morin and quercetin against methicillin resistant Staphylococcus aureus. BMC Complement. Altern. Med. 2015, 15, 59. [Google Scholar] [CrossRef]
- Chukwujekwu, J.C.; Amoo, S.O.; de Kock, C.A.; Smith, P.J.; Van Staden, J. Antiplasmodial, acetylcholinesterase and alpha-glucosidase inhibitory and cytotoxicity properties of Buddleja saligna. S. Afr. J. Bot. 2014, 94, 6–8. [Google Scholar] [CrossRef][Green Version]
- Kondhare, D.; Lade, H. Phytochemical profile, aldose reductase inhibitory, and antioxidant activities of indian traditional medicinal Coccinia grandis (L.) fruit extract. 3 Biotech. 2017, 7, 378. [Google Scholar] [CrossRef] [PubMed]
- Arul, D.; Subramanian, P. Inhibitory effect of naringenin (citrus flavonone) on N-nitrosodiethylamine induced hepatocarcinogenesis in rats. Biochem. Biophys. Res. Commun. 2013, 434, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.H.; Tavakkol-Afshari, J.; Brook, A.; Jafari-Anarkooli, I. Role of caspases and Bax protein in saffron-induced apoptosis in MCF-7 cells. Food Chem. Toxicol. 2009, 47, 1909–1913. [Google Scholar] [CrossRef] [PubMed]
| Assay | Hexane (SPH) | R2 | Dichloromethane (SPD) | R2 | Ethanol (SPE) | R2 |
|---|---|---|---|---|---|---|
| Total phenolic content (mg GAE a/g of extract) | 45.73 ± 0.72 | 0.970 | 59.23 ± 1.25 | 0.989 | 83.98 ± 1.30 | 0.999 |
| Total flavonoids content (μg QE b/g of extract) | 32.23 ± 0.92 | 0.999 | 49.84 ± 1.15 | 0.978 | 67.93 ± 1.17 | 0.988 |
| Assay | SPH IC50 (μg/mL) | R2 | SPD IC50 (μg/mL) | R2 | SPE IC50 (μg/mL) | R2 | BHA IC50 (μg/mL) | R2 |
|---|---|---|---|---|---|---|---|---|
| DPPH | 129.54 ± 5.92 | 0.952 | 102.01 ± 4.21 | 0.961 | 86.63 ± 7.53 | 0.953 | 70.32 ± 4.19 | 0.965 |
| ABTS | 156.28 ± 6.30 | 0.936 | 126.02 ± 5.28 | 0.986 | 101.32 ± 6.83 | 0.963 | 85.84 ± 6.97 | 0.929 |
| Extracts | Conc. µg/mL | Growth Inhibition Zone (mm) | |||
|---|---|---|---|---|---|
| Gram-Positive Bacteria | Gram-Negative Bacteria | Fungi | |||
| S. aureus | P. aeruginosa | E. coli | C. albicans | ||
| SPH | 500 | 10.6 ± 0.90 | 12.8 ± 0.64 | 5.7 ± 0.49 | 2.6 ± 0.36 |
| 250 | 8.9 ± 0.67 | 10.2 ± −0.88 | 1.8 ± 0.93 | 0 ± 00 | |
| 125 | 6.1 ± 0.83 | 7.9 ± 0.96 | 0 ± 00 | 0 ± 00 | |
| 62.5 | 3.7 ± 0.70 | 5.3 ± 0.93 | 0 ± 00 | 0 ± 00 | |
| 31.25 | 0 ± 00 | 0 ± 00 | 0 ± 00 | 0 ± 00 | |
| 15.63 | 0 ± 00 | 0 ± 00 | 0 ± 00 | 0 ± 00 | |
| SPD | 500 | 15.4 ± 0.54 | 13.3 ± 0.90 | 6.5 ± 0.69 | 5.4 ± 0.65 |
| 250 | 13.8 ± 0.98 | 10.7 ± 0.67 | 3.0 ± 0.35 | 1.9 ± 0.90 | |
| 125 | 10.5 ± 0.77 | 7.8 ± 0.83 | 0 ± 00 | 0 ± 00 | |
| 62.5 | 8.9 ± 0.34 | 4.5 ± 0.70 | 0 ± 00 | 0 ± 00 | |
| 31.25 | 5.3 ± 0.80 | 2.4 ± 0.80 | 0 ± 00 | 0 ± 00 | |
| 15.63 | 0 ± 00 | 0 ± 00 | 0 ± 00 | 0 ± 00 | |
| SPE | 500 | 18.2 ± 0.98 | 14.8 ± 0.64 | 9.7 ± 0.90 | 8.7 ± 0.54 |
| 250 | 16.5 ± 0.53 | 11.2 ± −0.88 | 7.4 ± 0.64 | 3.6 ± 0.78 | |
| 125 | 12.2± 0.89 | 9.3 ± 0.96 | 2.0 ± 0.90 | 0 ± 00 | |
| 62.5 | 11.5± 0.43 | 7.9 ± 0.93 | 0 ± 00 | 0 ± 00 | |
| 31.25 | 10.8± 0.48 | 4.6 ± 0.67 | 0 ± 00 | 0 ± 00 | |
| 15.63 | 8.3 ± 0.77 | 2.0 ± 0.74 | 0 ± 00 | 0 ± 00 | |
| Gentamicin | 10 | 22.19 ± 0.55 | 16.56 ± 0.60 | 12.43 ± 0.89 | − |
| Fluconazole | 10 | − | − | − | 11.3 ± 0.54 |
| Extracts | IC50 (mg/mL) | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| SPH | 110.56 ± 1.67 @ | 56.65 ± 1.22 @ | 39.12 ± 1.45 @ |
| SPD | 71.29 ± 0.87 * | 40.43 ± 1.02 * | 22.64 ± 1.33 * |
| SPE | 35.12 ± 1.56 # | 19.35 ± 1.54 # | 12.98 ± 2.53 # |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wali, A.F.; Alchamat, H.A.A.; Hariri, H.K.; Hariri, B.K.; Menezes, G.A.; Zehra, U.; Rehman, M.U.; Ahmad, P. Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals. Appl. Sci. 2020, 10, 1519. https://doi.org/10.3390/app10041519
Wali AF, Alchamat HAA, Hariri HK, Hariri BK, Menezes GA, Zehra U, Rehman MU, Ahmad P. Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals. Applied Sciences. 2020; 10(4):1519. https://doi.org/10.3390/app10041519
Chicago/Turabian StyleWali, Adil Farooq, Houda Ahmed Abou Alchamat, Huda Khaled Hariri, Bushra Khaled Hariri, Godfred A. Menezes, Uzma Zehra, Muneeb U. Rehman, and Parvaiz Ahmad. 2020. "Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals" Applied Sciences 10, no. 4: 1519. https://doi.org/10.3390/app10041519
APA StyleWali, A. F., Alchamat, H. A. A., Hariri, H. K., Hariri, B. K., Menezes, G. A., Zehra, U., Rehman, M. U., & Ahmad, P. (2020). Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals. Applied Sciences, 10(4), 1519. https://doi.org/10.3390/app10041519






