Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability
Abstract
1. Introduction
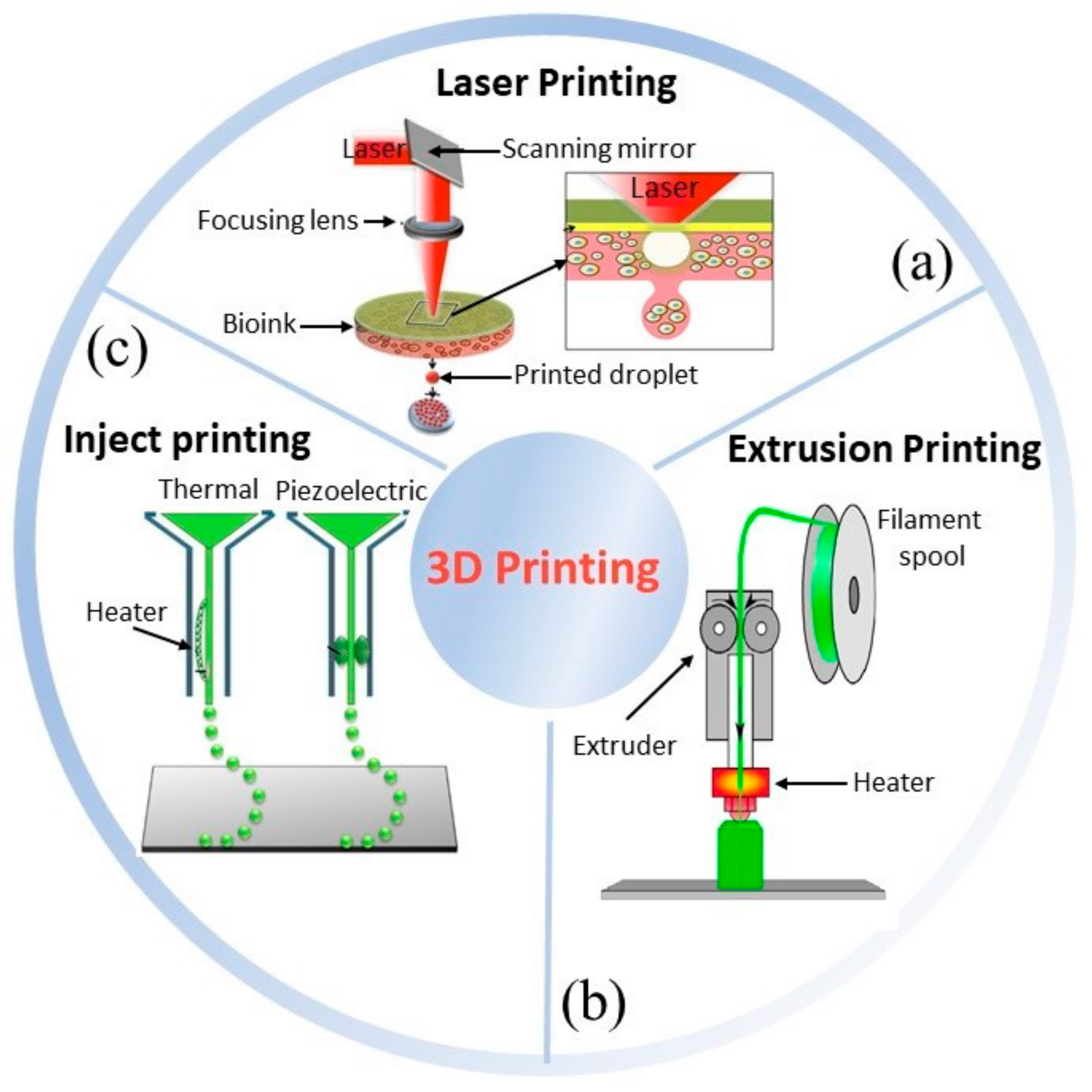
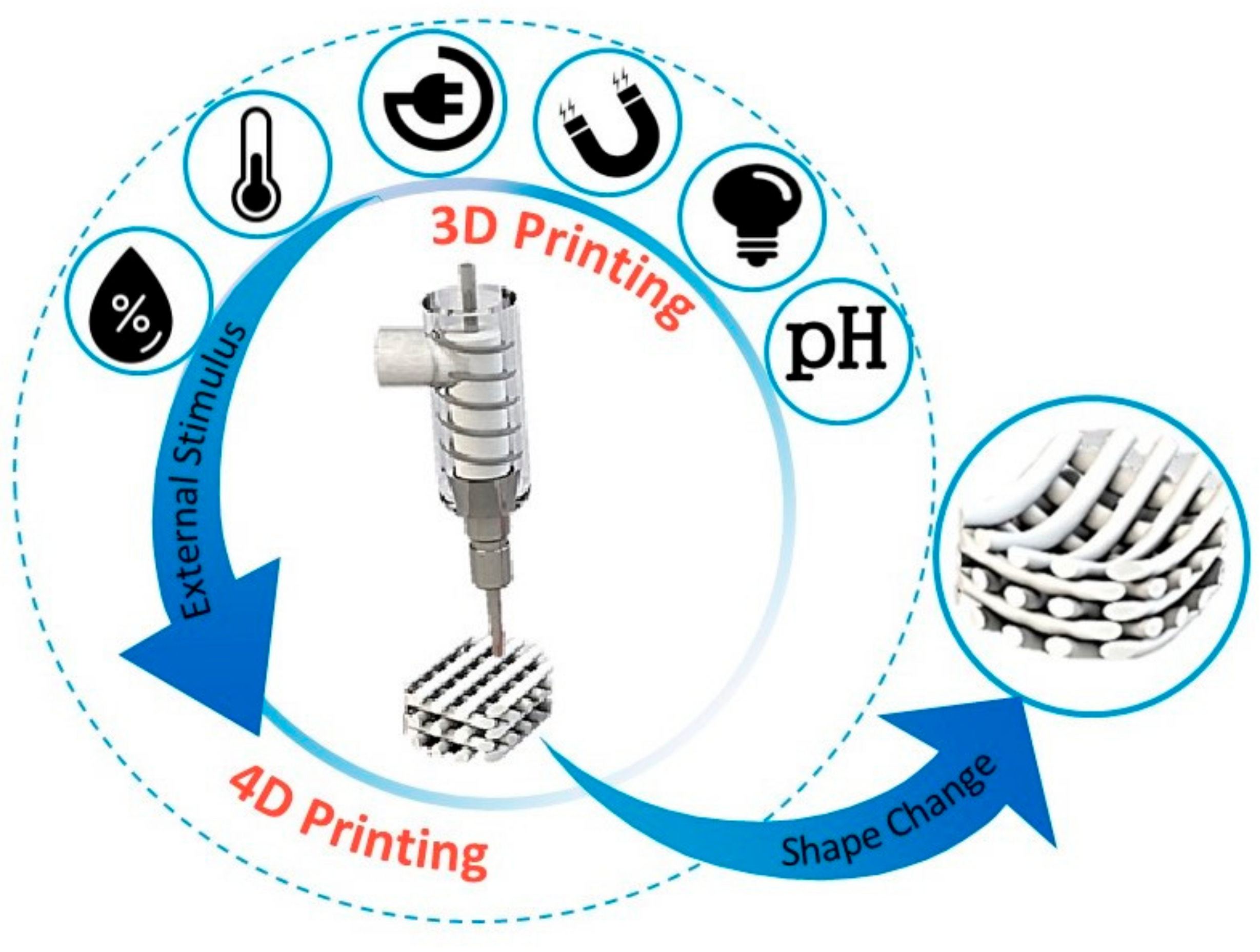
2. Four-Dimensional (Bio-)printing
3. Stimuli-Responsive Mechanisms: Avenues for 4D (Bio-)printing
3.1. Humidity-Responsive Materials
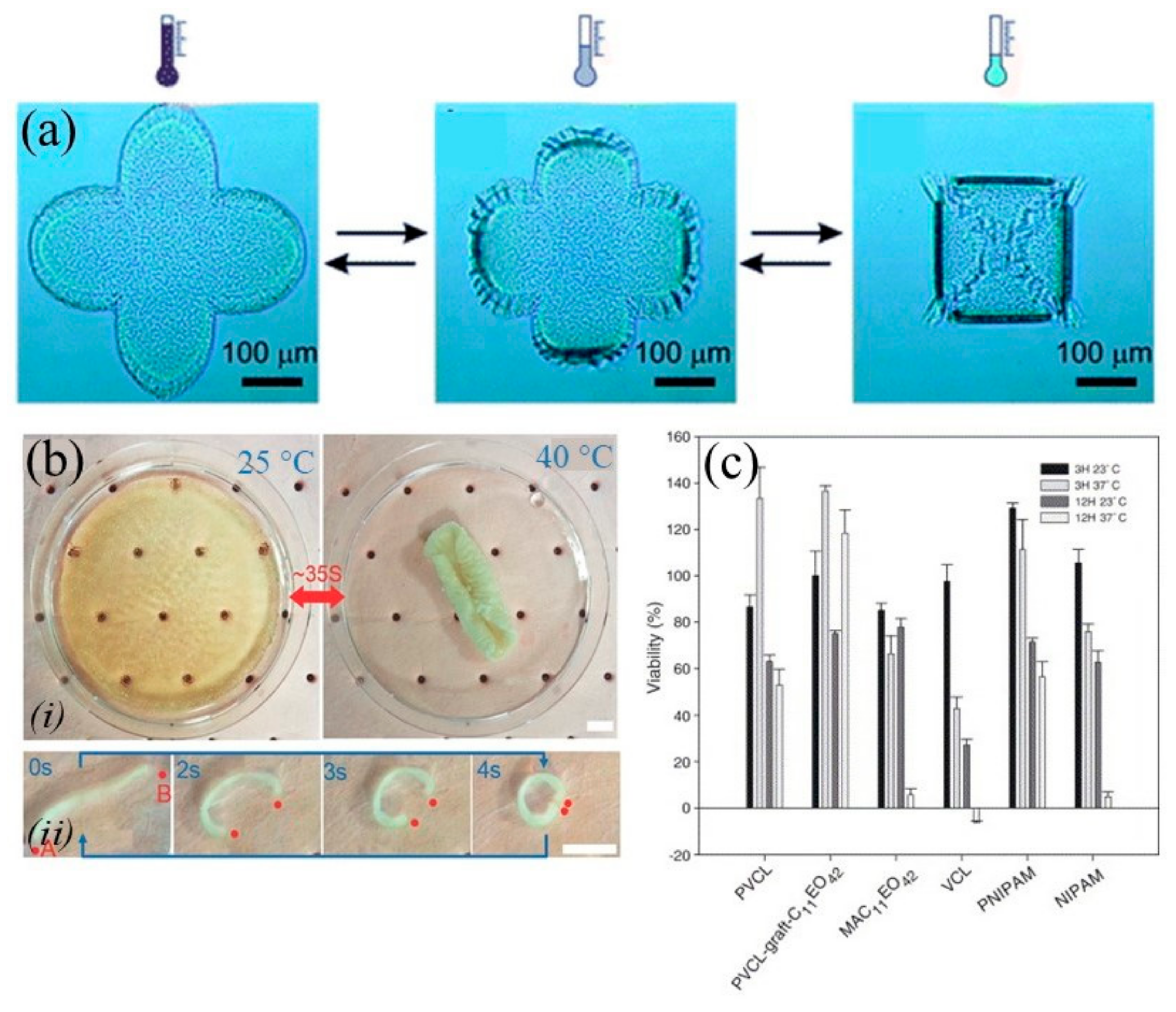
3.2. Temperature-Responsive Materials
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Poly(N-alkyl substituted acrylamides) | Temperature | NA | [65] |
| Poly(N-isopropylacrylamide) (PNIPAAm) | Temperature | Drug delivery, tissue regeneration, yeast cell encapsulation, artificial muscle, programmable actuators | [24,63,65,67,70,73,74,85,86,87] |
| Poly(N-vinylalkylamides) | Temperature | NA | [65,79] |
| Poly(N-vinylcaprolactam) (PNVC) | Temperature | Stabilization of proteases and controlled drug delivery | [80,88] |
| Copolymer of N-isopropylacrylamide (NiPAAm) and N-tert-butylacrylamide (NtBAAm) | Temperature | Potential vehicles for delivery of an antimitotic agent to vascular smooth muscle cells | [78] |
| Poly(ether-amide) grafting with PNIPAAm and PEG | Temperature | Controlled drug release | [79] |
| Poly(acrylic acid)–PNIPAAm and ceramic powder (Al2O3) | Temperature | Drug delivery and tissue regeneration | [68] |
| Poly(ɩ-lactic acid)-poly(ethylene glycol) Poly(ɩ-lactic acid) (PLLA-PEG-PLLA) | Temperature | Potential anticancer drug carrier, drug delivery | [65,89] |
| Poly(ethylene oxide)–poly (propylene oxide)–poly (ethylene oxide) (PEO-PPO-PEO) | Temperature | Controlled drug delivery | [82,83,90] |
| Epoxy based UV curable shape memory polymers (SMP) | Temperature | Medical, civil, and industrial | [91] |
| Poly-(D,L-lactide-co-glycolide)-block-poly(ethylene glycol)-block-poly-(D,L-lactide-co-glycolide) (PLGA-b-PEG-b-PLGA) triblock copolymer thermogels | Temperature | Potential anticancer drug carrier | [84] |
| Methacrylate based copolymer | Temperature | Biomedical devices | [92] |
| (PNIPAAm; PCL); alumina ceramics | Transition temperature | Pharmaceutical formulations, skeletal muscle-like linear actuation | [24,67,70] |
| Methyl cellulose hydrogel | Temperature | Cell sheet engineering | [93] |
3.3. Electrically and Mechanoelectrically Responsive Materials
3.4. Magnetically Responsive Materials
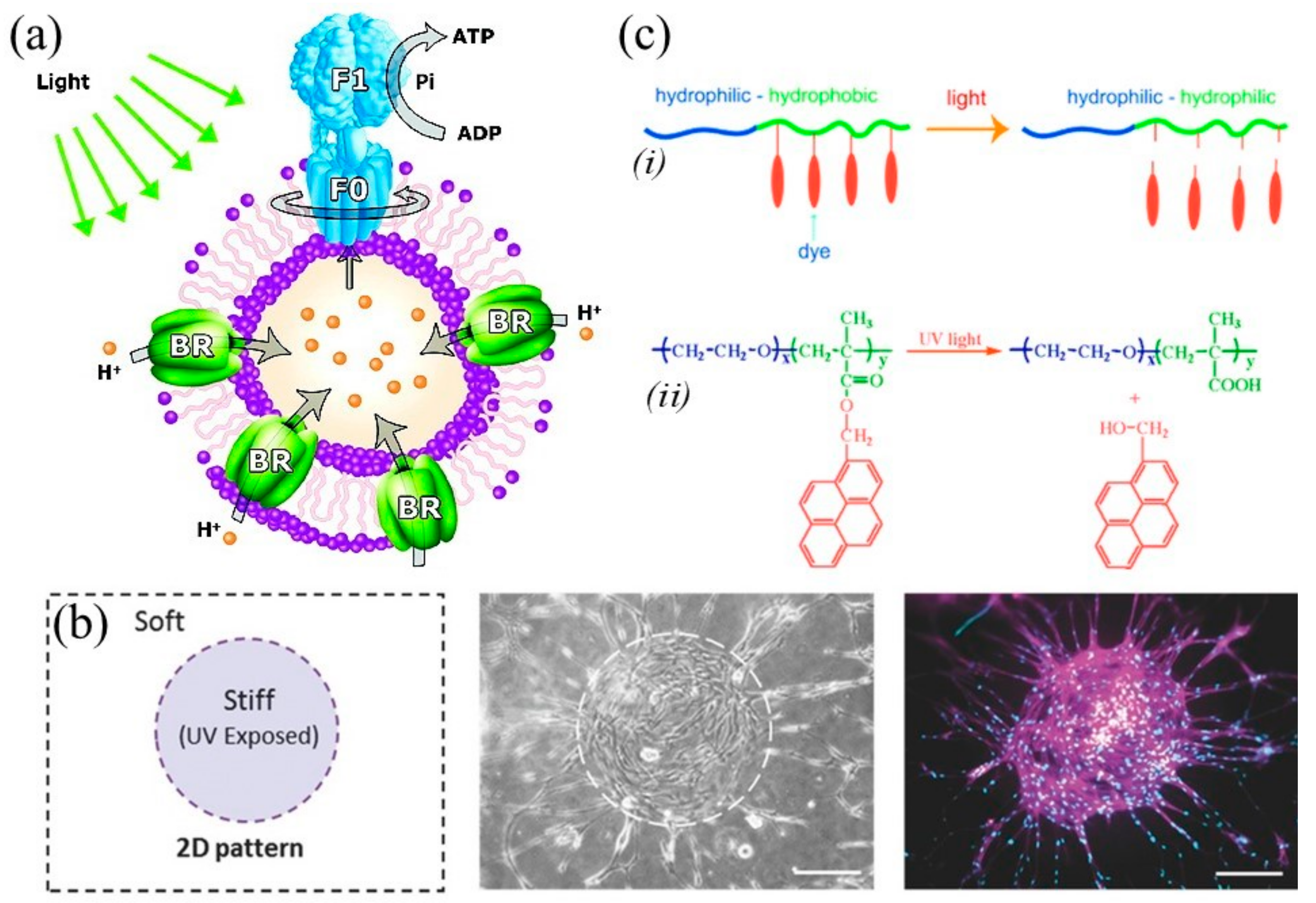
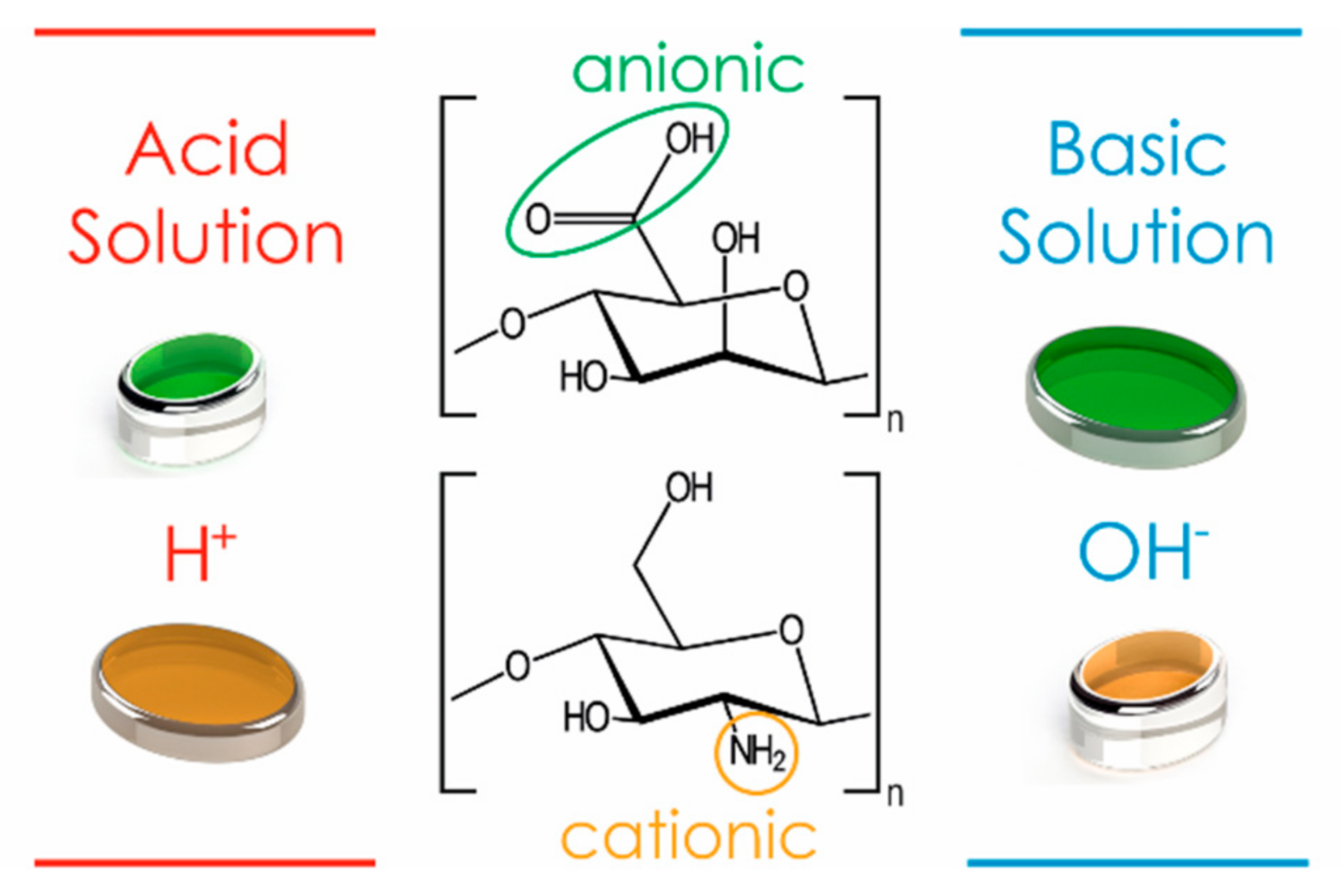
3.5. pH-Responsive Materials
3.6. Other Stimuli-Responsive Materials
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Glucose-responsive hydrogels | Chemistry | Diabetes monitoring | [41,65,158] |
| Glucose oxidase | Chemistry | Drug delivery | [159] |
| Glucose oxidase | Chemistry | Controlled release of insulin | [160] |
| Cysteine-containing hydrogel | Mechanical force | Injectable biomaterial | [157] |
| Barium titanate (BaTiO3, BTO) nanoparticles into a polyethylene glycol diacrylate (PEGDA) matrix | Stress (piezoelectric) | Implantable medical devices | [176] |
| BaTiO3 (BTO) filler in a poly(vinylidene) fluoride (PVDF) | Mechanical force (piezoelectric) | Pressure sensor | [179] |
| Ultraviolet-curable resins (acrylate ester for Resin 1, and methacrylated monomers/oligomers for Resin 2) | Mechanical force (piezoelectric) | N/A | [172] |
| BaTiO3 powder and photocurable resin SI500 | Mechanical force (piezoelectric) | Energy focusing and ultrasonic sensing | [173] |
| BaTiO3 nanoparticles in nucleating piezoelectric β-polymorph in 3D printable polyvinylidene fluoride (PVDF) | Mechanical force (piezoelectric) | Integrable sensors | [171] |
| Multiwall carbon nanotubes (MWCNTs)/PVDF/BaTiO3 nanoparticles | Mechanical force (piezoelectric) | Sensor | [174] |
| Shape memory polymer | Thermomechanical | Active origami | [177] |
| Glassy shape memory polymer | Thermomechanical | Active origami | [178] |
4. 4D (Bio-)printing: Cutting Edge Applications and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Miyoshi, H. Smart Design of Materials for Tissue Engineering. In Smart Materials for Tissue Engineering: Fundamental Principles; The Royal Society of Chemistry: London, UK, 2016; ISBN 9789533070797. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. 2008, 17. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Stimuli-responsive polymers: Biomedical applications and challenges for clinical translation. Adv. Drug Deliv. Rev. 2013, 65, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Stuart, M.A.C.; Huck, W.T.S.; Genzer, J.; Müller, M.; Ober, C.; Stamm, M.; Sukhorukov, G.B.; Szleifer, I.; Tsukruk, V.V.; Urban, M.; et al. Emerging applications of stimuli-responsive polymer materials. Nat. Mater. 2010, 9, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, L.; Lim, K.H.; Gonuguntla, S.; Lim, K.W.; Pranantyo, D.; Yong, W.P.; Yam, W.J.T.; Low, Z.; Teo, W.J.; et al. The Pathway to Intelligence: Using Stimuli-Responsive Materials as Building Blocks for Constructing Smart and Functional Systems. Adv. Mater. 2019, 31, 1804540. [Google Scholar] [CrossRef] [PubMed]
- Sears, N.A.; Seshadri, D.R.; Dhavalikar, P.S.; Cosgriff-Hernandez, E. A Review of Three-Dimensional Printing in Tissue Engineering. Tissue Eng. Part B Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Morouço, P.G. The Usefulness of Direct Digital Manufacturing for Biomedical Applications. In Intensification of Biobased Processes; The Royal Society of Chemistry: London, UK, 2018; pp. 478–487. [Google Scholar]
- Hull, C.W. Method of and Apparatus for Production of Three Dimensional Objects by Stereolithography. U.S. Patent No 5,236,637, 17 August 1993. [Google Scholar]
- Hutmacher, D.W.; Schantz, T.; Zein, I.; Ng, K.W.; Teoh, S.H.; Tan, K.C. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J. Biomed. Mater. Res. 2001, 55, 203–216. [Google Scholar] [CrossRef]
- Mullen, L.; Stamp, R.C.; Brooks, W.K.; Jones, E.; Sutcliffe, C.J. Selective laser melting: A regular unit cell approach for the manufacture of porous, titanium, bone in-growth constructs, suitable for orthopedic applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 325–334. [Google Scholar] [CrossRef]
- Nakamura, M.; Iwanaga, S.; Henmi, C.; Arai, K.; Nishiyama, Y. Biomatrices and biomaterials for future developments of bioprinting and biofabrication. Biofabrication 2010, 2, 014110. [Google Scholar] [CrossRef]
- Castro, N.J.; Meinert, C.; Levett, P.; Hutmacher, D.W. Current developments in multifunctional smart materials for 3D/4D bioprinting. Curr. Opin. Biomed. Eng. 2017, 2, 67–75. [Google Scholar] [CrossRef]
- Milazzo, M.; Contessi Negrini, N.; Scialla, S.; Marelli, B.; Farè, S.; Danti, S.; Buehler, M.J. Additive Manufacturing Approaches for Hydroxyapatite-Reinforced Composites. Adv. Funct. Mater. 2019, 29, 1903055. [Google Scholar] [CrossRef]
- Khoo, Z.X.; Teoh, J.E.M.; Liu, Y.; Chua, C.K.; Yang, S.; An, J.; Leong, K.F.; Yeong, W.Y. 3D printing of smart materials: A review on recent progresses in 4D printing. Virtual Phys. Prototyp. 2015, 10, 103–122. [Google Scholar] [CrossRef]
- Li, Y.-C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication 2016, 9, 12001. [Google Scholar] [CrossRef] [PubMed]
- Ashammakhi, N.; Ahadian, S.; Zengjie, F.; Suthiwanich, K.; Lorestani, F.; Orive, G.; Ostrovidov, S.; Khademhosseini, A. Advances and future perspectives in 4D bioprinting. Biotechnol. J. 2018, 13, 1800148. [Google Scholar] [CrossRef]
- Keriquel, V.; Oliveira, H.; Rémy, M.; Ziane, S.; Delmond, S.; Rousseau, B.; Rey, S.; Catros, S.; Amédée, J.; Guillemot, F.; et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Liu, F.; Wang, X. Synthetic Polymers for Organ 3D Printing. Polymers (Basel) 2020, 12, 1765. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Fuh, J.Y.H.; Lu, W.F. 3D printing and 3D bioprinting in pediatrics. Bioengineering 2017, 4, 63. [Google Scholar] [CrossRef]
- Groll, J.; Boland, T.; Blunk, T.; Burdick, J.A.; Cho, D.-W.; Dalton, P.D.; Derby, B.; Forgacs, G.; Li, Q.; Mironov, V.A.; et al. Biofabrication: Reappraising the definition of an evolving field. Biofabrication 2016, 8, 13001. [Google Scholar] [CrossRef]
- Leist, S.K.; Zhou, J. Current status of 4D printing technology and the potential of light-reactive smart materials as 4D printable materials. Virtual Phys. Prototyp. 2016, 2759, 1–14. [Google Scholar] [CrossRef]
- Mandon, C.A.; Blum, L.J.; Marquette, C.A. 3D-4D printed objects: New bioactive material opportunities. Micromachines 2017, 8, 102. [Google Scholar] [CrossRef]
- Gao, B.; Yang, Q.; Zhao, X.; Jin, G.; Ma, Y.; Xu, F. 4D bioprinting for biomedical applications. Trends Biotechnol. 2016, 34, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Morouço, P.; Lattanzi, W.; Alves, N. Four-dimensional bioprinting as a new era for tissue engineering and regenerative medicine. Front. Bioeng. Biotechnol. 2017, 5, 61. [Google Scholar] [CrossRef] [PubMed]
- Momeni, F.M.; Mehdi Hassani, N.S.; Liu, X.; Ni, J. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- An, J.; Chua, C.K.; Mironov, V. A perspective on 4D bioprinting. Int. J. Bioprinting 2016, 2, 3–5. [Google Scholar] [CrossRef]
- Moroni, L.; Boland, T.; Burdick, J.A.; De Maria, C.; Derby, B.; Forgacs, G.; Groll, J.; Li, Q.; Malda, J.; Mironov, V.A.; et al. Biofabrication: A guide to technology and terminology. Trends Biotechnol. 2018, 36, 384–402. [Google Scholar] [CrossRef]
- Wang, S.; Lee, J.M.; Yeong, W.Y. Smart hydrogels for 3D bioprinting. Int. J. Bioprinting 2015, 1, 3–14. Available online: http://ijb.whioce.com/index.php/int-j-bioprinting/article/view/01005 (accessed on 23 October 2020). [CrossRef]
- Negrini, N.C.; Bonetti, L.; Contili, L.; Farè, S. 3D printing of methylcellulose-based hydrogels. Bioprinting 2018, 10, e00024. [Google Scholar] [CrossRef]
- Negrini, N.C.; Celikkin, N.; Tarsini, P.; Farè, S.; Święszkowski, W. Three-dimensional printing of chemically crosslinked gelatin hydrogels for adipose tissue engineering. Biofabrication 2020, 12, 25001. [Google Scholar] [CrossRef]
- Ikegami, T.; Maehara, Y. Transplantation: 3D printing of the liver in living donor liver transplantation. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 697–698. [Google Scholar] [CrossRef]
- Valverde, I.; Gomez, G.; Suarez-mejias, C.; Hosseinpour, A.; Hazekamp, M.; Roest, A.; Vazquez-jimenez, J.F.; El-rassi, I.; Uribe, S.; Gomez-cia, T. 3D printed cardiovascular models for surgical planning in complex congenital heart diseases. J. Cardiovasc. Magn. Reson. 2015, 17, P196. [Google Scholar] [CrossRef]
- Gao, G.; Cui, X. Three-dimensional bioprinting in tissue engineering and regenerative medicine. Biotechnol. Lett. 2016, 38, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.-C. Mechanobiology of tendon. J. Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef] [PubMed]
- Milazzo, M.; Jung, G.S.; Danti, S.; Buehler, M.J. Mechanics of mineralized collagen fibrils upon transient loads. ACS Nano 2020, 14, 8307–8316. [Google Scholar] [CrossRef] [PubMed]
- Milazzo, M.; Jung, G.S.; Danti, S.; Buehler, M.J. Wave propagation and energy dissipation in collagen molecules. ACS Biomater. Sci. Eng. 2020, 6, 1367–1374. [Google Scholar] [CrossRef]
- Ekwueme, E.C.; Patel, J.M.; Freeman, J.W.; Danti, S. Applications of bioresorbable polymers in the skeletal systems (cartilages, tendons, bones). In Bioresorbable Polymers for Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2017; pp. 391–422. [Google Scholar]
- Zhu, L.; Qiu, J.; Sakai, E. A high modulus hydrogel obtained from hydrogen bond reconstruction and its application in vibration damper. RSC Adv. 2017, 7, 43755–43763. [Google Scholar] [CrossRef]
- Meng, H.; Li, G. A review of stimuli-responsive shape memory polymer composites. Polymer (United Kingdom) 2013, 54, 2199–2221. [Google Scholar] [CrossRef]
- Roy, D.; Cambre, J.N.; Sumerlin, B.S. Future perspectives and recent advances in stimuli-responsive materials. Prog. Polym. Sci. 2010, 35, 278–301. [Google Scholar] [CrossRef]
- Morouço, P.; Gil, J. Four-Dimensional Bioprinting for Regenerative Medicine: Mechanisms to Induce Shape Variation and Potential Applications. Innovations 2019, 3, 36–43. [Google Scholar] [CrossRef]
- Koetting, M.C.; Peters, J.T.; Steichen, S.D.; Peppas, N.A. Stimulus-responsive hydrogels: Theory, modern advances, and applications. Mater. Sci. Eng. R Rep. 2015, 93, 1–49. [Google Scholar] [CrossRef]
- Bertassoni, L.E.; Cardoso, J.C.; Manoharan, V.; Cristino, A.L.; Bhise, N.S.; Araujo, W.A.; Zorlutuna, P.; Vrana, N.E.; Ghaemmaghami, A.M.; Dokmeci, M.R.; et al. Direct-write bioprinting of cell-laden methacrylated gelatin hydrogels. Biofabrication 2014, 6, 024105. [Google Scholar] [CrossRef]
- Chang, C.C.; Boland, E.D.; Williams, S.K.; Hoying, J.B. Direct-write bioprinting three-dimensional biohybrid systems for future regenerative therapies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011. [Google Scholar] [CrossRef] [PubMed]
- Woodfield, T.; Lim, K.; Morouço, P.; Levato, R.; Malda, J.; Melchels, F. Biofabrication in Tissue Engineering. In Comprehensive Biomaterials II; Springer International Publishing: Cham, Switzerland, 2017; pp. 236–266. ISBN 9780128035818. [Google Scholar]
- Dai, M.; Picot, O.T.; Verjans, J.M.N.; De Haan, L.T.; Schenning, A.P.H.J.; Peijs, T.; Bastiaansen, C.W.M. Humidity-responsive bilayer actuators based on a liquid-crystalline polymer network. ACS Appl. Mater. Interfaces 2013, 5, 4945–4950. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liang, H.; Jacob, J.; Naumov, P. Photogated humidity-driven motility. Nat. Commun. 2015, 6, 7429. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Y.; Yang, G.; Zheng, X.; Zhou, S. Multi-stimulus-responsive shape-memory polymer nanocomposite network cross-linked by cellulose nanocrystals. ACS Appl. Mater. Interfaces 2015, 7, 4118–4126. [Google Scholar] [CrossRef]
- Torbati, A.H.; Mather, P.T. A hydrogel-forming liquid crystalline elastomer exhibiting soft shape memory. J. Polym. Sci. Part B Polym. Phys. 2016, 54, 38–52. [Google Scholar] [CrossRef]
- Lv, C.; Sun, X.-C.; Xia, H.; Yu, Y.-H.; Wang, G.; Cao, X.-W.; Li, S.-X.; Wang, Y.-S.; Chen, Q.-D.; Yu, Y.-D. Humidity-responsive actuation of programmable hydrogel microstructures based on 3D printing. Sens. Actuators B Chem. 2018, 259, 736–744. [Google Scholar] [CrossRef]
- Lv, C.; Xia, H.; Shi, Q.; Wang, G.; Wang, Y.; Chen, Q.; Zhang, Y.; Liu, L.; Sun, H. Sensitively Humidity-Driven Actuator Based on Photopolymerizable PEG-DA Films. Adv. Mater. Interfaces 2017, 4. [Google Scholar] [CrossRef]
- Jamal, M.; Kadam, S.S.; Xiao, R.; Jivan, F.; Onn, T.; Fernandes, R.; Nguyen, T.D.; Gracias, D.H. Bio-Origami Hydrogel Scaffolds Composed of Photocrosslinked PEG Bilayers. Adv. Healthc. Mater. 2013, 2, 1142–1150. [Google Scholar] [CrossRef]
- Ma, M.; Guo, L.; Anderson, D.G.; Langer, R. Bio-inspired polymer composite actuator and generator driven by water gradients. Science 2013, 339, 186–189. [Google Scholar] [CrossRef]
- Zhang, K.; Geissler, A.; Standhardt, M.; Mehlhase, S.; Gallei, M.; Chen, L.; Thiele, C.M. Moisture-responsive films of cellulose stearoyl esters showing reversible shape transitions. Sci. Rep. 2015, 5, 11011. [Google Scholar] [CrossRef]
- Wang, G.; Xia, H.; Sun, X.-C.; Lv, C.; Li, S.-X.; Han, B.; Guo, Q.; Shi, Q.; Wang, Y.-S.; Sun, H.-B. Actuator and generator based on moisture-responsive PEDOT: PSS/PVDF composite film. Sens. Actuators B Chem. 2018, 255, 1415–1421. [Google Scholar] [CrossRef]
- Raviv, D.; Zhao, W.; McKnelly, C.; Papadopoulou, A.; Kadambi, A.; Shi, B.; Hirsch, S.; Dikovsky, D.; Zyracki, M.; Olguin, C. Active printed materials for complex self-evolving deformations. Sci. Rep. 2014, 4, 7422. [Google Scholar] [CrossRef] [PubMed]
- Gladman, A.S.; Matsumoto, E.A.; Nuzzo, R.G.; Mahadevan, L.; Lewis, J.A. Biomimetic 4D printing. Nat. Mater. 2016, 15, 413. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.; Lu, F.; Zhao, M.; Sun, N.; Shi, L.; Zheng, L. Photo and Humidity Responsive Mesoporous Poly (ionic Liquid) Membrane for Selective Dye Adsorption. ChemistrySelect 2017, 2, 1878–1884. [Google Scholar] [CrossRef]
- EPA, U.S. United States Environmental Protection Agency. Qual. Assur. Guid. Doc. Qual. Assur. Proj. Plan PM Ambient Air 2001, 2. Available online: https://www3.epa.gov/ttnamti1/files/ambient/pm25/qa/Final%20Handbook%20Document%201_17.pdf (accessed on 23 October 2020).
- Zhou, S.; Deng, X.; Yang, H. Biodegradable poly(ε-caprolactone)-poly(ethylene glycol) block copolymers: Characterization and their use as drug carriers for a controlled delivery system. Biomaterials 2003, 24, 3563–3570. [Google Scholar] [CrossRef]
- AndrÃ, J.-C. From Additive Manufacturing to 3D/4D Printing: Breakthrough Innovations: Programmable Material, 4D Printing and Bio-Printing; John Wiley & Sons: Hoboken, NJ, USA, 2017; ISBN 1119482771. [Google Scholar]
- Zhang, X.; Pint, C.L.; Lee, M.H.; Schubert, B.E.; Jamshidi, A.; Takei, K.; Ko, H.; Gillies, A.; Bardhan, R.; Urban, J.J. Optically-and thermally-responsive programmable materials based on carbon nanotube-hydrogel polymer composites. Nano Lett. 2011, 11, 3239–3244. [Google Scholar] [CrossRef]
- Romano, L.; Portone, L.; Coltelli, M.B.; Patti, F.; Saija, R.; Iatì, M.A.; Gallone, G.; Lazzeri, A.; Danti, S.; Marago, O.; et al. Intelligent non-colorimetric indicators for the perishable supply chain by non-wovens with photo-programmed thermal response. Nat. Commun. 2020, 11, 5991. [Google Scholar] [CrossRef]
- Cabane, E.; Zhang, X.; Langowska, K.; Palivan, C.G.; Meier, W.; Cabane, E.; Zhang, X.; Langowska, K.; Palivan, C.G.; Meier, W. Stimuli-responsive polymers and their applications in nanomedicine. Biointerphases 2012, 7, 1–27. [Google Scholar] [CrossRef]
- Bakarich, S.E.; Gorkin, R., III; Panhuis, M.I.; Spinks, G.M. 4D Printing with Mechanically Robust, Thermally Actuating Hydrogels. Macromol. Rapid Commun. 2015, 36, 1211–1217. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Wang, X.; Sun, Y.; Peng, C.; Luo, H.; Wang, R.; Zhang, D. Transitional Suspensions Containing Thermosensitive Dispersant for Three-Dimensional Printing. ACS Appl. Mater. Interfaces 2015, 7, 26131–26136. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.H.; Jeon, O.; Kwon, I.C.; Park, K. Engineered polymers for advanced drug delivery. Eur. J. Pharm. Biopharm. 2009, 71, 420–430. [Google Scholar] [CrossRef] [PubMed]
- Stoychev, G.; Puretskiy, N.; Ionov, L. Self-folding all-polymer thermoresponsive microcapsules. Soft Matter 2011, 7, 3277. [Google Scholar] [CrossRef]
- Luo, R.; Wu, J.; Dinh, N.-D.; Chen, C.-H. Gradient porous elastic hydrogels with shape-memory property and anisotropic responses for programmable locomotion. Adv. Funct. Mater. 2015, 25, 7272–7279. [Google Scholar] [CrossRef]
- Xu, X.; Liu, Y.; Fu, W.; Yao, M.; Ding, Z.; Xuan, J.; Li, D.; Wang, S.; Xia, Y.; Cao, M. Poly (N-isopropylacrylamide)-based thermoresponsive composite hydrogels for biomedical applications. Polymers (Basel) 2020, 12, 580. [Google Scholar] [CrossRef]
- Chastek, T.T.; Wadajkar, A.; Nguyen, K.T.; Hudson, S.D.; Chastek, T.Q. Polyglycol-templated synthesis of poly(N-isopropyl acrylamide) microgels with improved biocompatibility. Colloid Polym. Sci. 2010, 288, 105–114. [Google Scholar] [CrossRef]
- Cooperstein, M.A.; Canavan, H.E. Assessment of cytotoxicity of (N-isopropyl acrylamide ) and Poly (N-isopropyl acrylamide )—Coated surfaces. Biointerphases 2013, 8, 1–12. [Google Scholar] [CrossRef]
- Sarkar, J.; Kumari, J.; Tonello, J.M.; Kamihira, M.; Kumar, A. Enhanced hepatic functions of genetically modified mouse hepatoma cells by spheroid culture for drug toxicity screening. Biotechnol. J. 2017, 12, 1700274. [Google Scholar] [CrossRef]
- Ricci, C.; Moroni, L.; Danti, S. Cancer tissue engineering-new perspectives in understanding the biology of solid tumours-a critical review. OA Tissue Eng. 2013, 1, 1–4. [Google Scholar] [CrossRef]
- Chew, S.A.; Moscato, S.; George, S.; Azimi, B.; Danti, S. Liver cancer: Current and future trends using biomaterials. Cancers (Basel) 2019, 11, 2026. [Google Scholar] [CrossRef] [PubMed]
- Doorty, K.B.; Golubeva, T.A.; Gorelov, A.V.; Rochev, Y.A.; Allen, L.T.; Dawson, K.A.; Gallagher, W.M.; Keenan, A.K. Poly (N-isopropylacrylamide) co-polymer films as potential vehicles for delivery of an antimitotic agent to vascular smooth muscle cells. Cardiovasc. Pathol. 2003, 12, 105–110. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, W.; Zou, J.; Shi, W. Synthesis and thermally responsive characteristics of dendritic poly (ether-amide) grafting with PNIPAAm and PEG. Polymer (Guildf) 2007, 48, 931–938. [Google Scholar] [CrossRef]
- Vihola, H.; Laukkanen, A.; Valtola, L.; Tenhu, H.; Hirvonen, J. Cytotoxicity of thermosensitive polymers poly(N-isopropylacrylamide), poly(N-vinylcaprolactam) and amphiphilically modified poly(N-vinylcaprolactam). Biomaterials 2005, 26, 3055–3064. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.Z.; Zeng, Z.W.; Zhou, G.L.; Wang, J.J.; Li, F.Z.; Wang, A.M. Recent advances in PEG-PLA block copolymer nanoparticles. Int. J. Nanomed. 2010, 5, 1057–1065. [Google Scholar] [CrossRef]
- Redhead, M.; Mantovani, G.; Nawaz, S.; Carbone, P.; Gorecki, D.C.; Alexander, C.; Bosquillon, C. Relationship between the affinity of PEO-PPO-PEO block copolymers for biological membranes and their cellular effects. Pharm. Res. 2012, 29, 1908–1918. [Google Scholar] [CrossRef]
- Sosnik, A.; Cohn, D. Ethoxysilane-capped PEO–PPO–PEO triblocks: A new family of reverse thermo-responsive polymers. Biomaterials 2004, 25, 2851–2858. [Google Scholar] [CrossRef]
- Cho, H.; Kwon, G.S. Thermosensitive poly-(d, l-lactide-co-glycolide)-block-poly (ethylene glycol)-block-poly-(d, l-lactide-co-glycolide) hydrogels for multi-drug delivery. J. Drug Target. 2014, 22, 669–677. [Google Scholar] [CrossRef]
- Magdanz, V.; Stoychev, G.; Ionov, L.; Sanchez, S.; Schmidt, O.G. Stimuli-responsive microjets with reconfigurable shape. Angew. Chemie 2014, 126, 2711–2715. [Google Scholar] [CrossRef]
- Ohya, S.; Sonoda, H.; Nakayama, Y.; Matsuda, T. The potential of poly (N-isopropylacrylamide)(PNIPAM)-grafted hyaluronan and PNIPAM-grafted gelatin in the control of post-surgical tissue adhesions. Biomaterials 2005, 26, 655–659. [Google Scholar] [CrossRef]
- Liu, Y.; Meng, L.; Lu, X.; Zhang, L.; He, Y. Thermo and pH sensitive fluorescent polymer sensor for metal cations in aqueous solution. Polym. Adv. Technol. 2008, 19, 137–143. [Google Scholar] [CrossRef]
- Liang, X.; Kozlovskaya, V.; Chen, Y.; Zavgorodnya, O.; Kharlampieva, E. Thermosensitive multilayer hydrogels of poly (N-vinylcaprolactam) as nanothin films and shaped capsules. Chem. Mater. 2012, 24, 3707–3719. [Google Scholar] [CrossRef] [PubMed]
- Na, K.; Lee, K.H.; Lee, D.H.; Bae, Y.H. Biodegradable thermo-sensitive nanoparticles from poly (L-lactic acid)/poly (ethylene glycol) alternating multi-block copolymer for potential anti-cancer drug carrier. Eur. J. Pharm. Sci. 2006, 27, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Cohn, D.; Lando, G.; Sosnik, A.; Garty, S.; Levi, A. PEO—PPO—PEO-based poly (ether ester urethane) s as degradable reverse thermo-responsive multiblock copolymers. Biomaterials 2006, 27, 1718–1727. [Google Scholar] [CrossRef]
- Yu, K.; Ritchie, A.; Mao, Y.; Dunn, M.L.; Qi, H.J. Controlled sequential shape changing components by 3D printing of shape memory polymer multimaterials. Procedia IUTAM 2015, 12, 193–203. [Google Scholar] [CrossRef]
- Ge, Q.; Sakhaei, A.H.; Lee, H.; Dunn, C.K.; Fang, N.X.; Dunn, M.L. Multimaterial 4D printing with tailorable shape memory polymers. Sci. Rep. 2016, 6, 31110. [Google Scholar] [CrossRef]
- Contessi, N.; Altomare, L.; Filipponi, A.; Farè, S. Thermo-responsive properties of methylcellulose hydrogels for cell sheet engineering. Mater. Lett. 2017, 207, 157–160. [Google Scholar] [CrossRef]
- Pillay, V.; Tsai, T.-S.; Choonara, Y.E.; du Toit, L.C.; Kumar, P.; Modi, G.; Naidoo, D.; Tomar, L.K.; Tyagi, C.; Ndesendo, V.M.K. A review of integrating electroactive polymers as responsive systems for specialized drug delivery applications. J. Biomed. Mater. Res. Part A 2014, 102, 2039–2054. [Google Scholar] [CrossRef]
- Sista, P.; Ghosh, K.; Martinez, J.S.; Rocha, R.C. Polythiophenes in biological applications. J. Nanosci. Nanotechnol. 2014, 14, 250–272. [Google Scholar] [CrossRef]
- Herold, B.C.; Bourne, N.; Marcellino, D.; Kirkpatrick, R.; Mosoian, A.; Klotman, M.; Strauss, D.; Anderson, R.; Zanenveld, L.; Cooper, M.; et al. Polystyrene sulfonate is a safe and effective candidate topical antimicrobial for the prevention of sexually transmitted diseases. Pediatr. Res. 1999, 45, 163. [Google Scholar] [CrossRef][Green Version]
- Alzari, V.; Nuvoli, D.; Sanna, R.; Peponi, L.; Piccinini, M.; Bon, S.B.; Marceddu, S.; Valentini, L.; Kenny, J.M.; Mariani, A. Multistimuli-responsive hydrogels of poly (2-acrylamido-2-methyl-1-propanesulfonic acid) containing graphene. Colloid Polym. Sci. 2013, 291, 2681–2687. [Google Scholar] [CrossRef]
- D’Emanuele, A.; Staniforth, J.N. An electrically modulated drug delivery device: I. Pharm. Res. 1991, 8, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Rouabhia, M.; Chiffot, N.; King, M.W.; Zhang, Z. An electrically conductive 3D scaffold based on a nonwoven web of poly (l-lactic acid) and conductive poly (3, 4-ethylenedioxythiophene). J. Biomed. Mater. Res. Part A 2015, 103, 2635–2644. [Google Scholar] [CrossRef] [PubMed]
- Duc, C.; Vlandas, A.; Malliaras, G.G.; Senez, V. Study of the electro-responsiveness and surface texturing of PEDOT: PSS for smart MEMS interface applications. In Proceedings of the 2017 19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 1391–1394. [Google Scholar]
- Pires, F.; Ferreira, Q.; Rodrigues, C.A.V.; Morgado, J.; Ferreira, F.C. Neural stem cell differentiation by electrical stimulation using a cross-linked PEDOT substrate: Expanding the use of biocompatible conjugated conductive polymers for neural tissue engineering. Biochim. Biophys. Acta (BBA) General Subj. 2015, 1850, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Venugopalan, V.; Divya, M.S.; Rasheed, V.A.; James, J.; Narayan, K.S. Neuronal differentiation of embryonic stem cell derived neuronal progenitors can be regulated by stretchable conducting polymers. Tissue Eng. Part A 2013, 19, 1984–1993. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Urban, M.W. Recent advances and challenges in designing stimuli-responsive polymers. Prog. Polym. Sci. 2010, 35, 3–23. [Google Scholar] [CrossRef]
- Scialla, S.; Barca, A.; Palazzo, B.; D’Amora, U.; Russo, T.; Gloria, A.; De Santis, R.; Verri, T.; Sannino, A.; Ambrosio, L.; et al. Bioactive Chitosan-Based Scaffolds with Improved Properties Induced by Dextran-Grafted Nano-Maghemite and L-Arginine Amino Acid. J. Biomed. Mater. Res. Part A 2019, 107, 1244–1252. [Google Scholar] [CrossRef]
- Szabo, D.; Szeghy, G.; Zrinyi, M. Shape transition of magnetic field sensitive polymer gels. Macromolecules 1998, 31, 6541–6548. [Google Scholar] [CrossRef]
- Zrinyi, M. Intelligent polymer gels controlled by magnetic fields. Colloid Polym. Sci. 2000, 278, 98–103. [Google Scholar] [CrossRef]
- Bowles, S.E.; Wu, W.; Kowalewski, T.; Schalnat, M.C.; Davis, R.J.; Pemberton, J.E.; Shim, I.; Korth, B.D.; Pyun, J. Magnetic assembly and pyrolysis of functional ferromagnetic colloids into one-dimensional carbon nanostructures. J. Am. Chem. Soc. 2007, 129, 8694–8695. [Google Scholar] [CrossRef]
- Giani, G.; Fedi, S.; Barbucci, R. Hybrid magnetic hydrogel: A potential system for controlled drug delivery by means of alternating magnetic fields. Polymers (Basel) 2012, 4, 1157–1169. [Google Scholar] [CrossRef]
- Brugger, B.; Richtering, W. Magnetic, thermosensitive microgels as stimuli-responsive emulsifiers allowing for remote control of separability and stability of oil in water-emulsions. Adv. Mater. 2007, 19, 2973–2978. [Google Scholar] [CrossRef]
- Xulu, P.M.; Filipcsei, G.; Zrinyi, M. Preparation and responsive properties of magnetically soft poly (N-isopropylacrylamide) gels. Macromolecules 2000, 33, 1716–1719. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, S.; Kumacheva, E. Polymer microgels: Reactors for semiconductor, metal, and magnetic nanoparticles. J. Am. Chem. Soc. 2004, 126, 7908–7914. [Google Scholar] [CrossRef]
- Zhao, X.; Kim, J.; Cezar, C.A.; Huebsch, N.; Lee, K.; Bouhadir, K.; Mooney, D.J. Active scaffolds for on-demand drug and cell delivery. Proc. Natl. Acad. Sci. USA 2011, 108, 67–72. [Google Scholar] [CrossRef]
- Czaun, M.; Hevesi, L.; Takafuji, M.; Ihara, H. A novel approach to magneto-responsive polymeric gels assisted by iron nanoparticles as nano cross-linkers. Chem. Commun. 2008, 2124–2126. [Google Scholar] [CrossRef]
- Pašukonienė, V.; Mlynska, A.; Steponkienė, S.; Poderys, V.; Matulionytė, M.; Karabanovas, V.; Statkutė, U.; Purvinienė, R.; Kraśko, J.A.; Jagminas, A.; et al. Accumulation and biological effects of cobalt ferrite nanoparticles in human pancreatic and ovarian cancer cells. Medicina (B. Aires) 2014, 50, 237–244. [Google Scholar] [CrossRef]
- Li, Y.; Huang, G.; Zhang, X.; Li, B.; Chen, Y.; Lu, T.; Lu, T.J.; Xu, F. Magnetic hydrogels and their potential biomedical applications. Adv. Funct. Mater. 2013, 23, 660–672. [Google Scholar] [CrossRef]
- Della Giustina, G.; Gandin, A.; Brigo, L.; Panciera, T.; Giulitti, S.; Sgarbossa, P.; D’Alessandro, D.; Trombi, L.; Danti, S.; Brusatin, G. Polysaccharide hydrogels for multiscale 3D printing of pullulan scaffolds. Mater. Des. 2019, 165, 107566. [Google Scholar] [CrossRef]
- Yavuz, M.S.; Cheng, Y.; Chen, J.; Cobley, C.M.; Zhang, Q.; Rycenga, M.; Xie, J.; Kim, C.; Song, K.H.; Schwartz, A.G.; et al. Gold nanocages covered by smart polymers for controlled release with near-infrared light. Nat. Mater. 2009, 8, 935–939. [Google Scholar] [CrossRef]
- D’Acunto, M.; Trombi, L.; D’Alessandro, D.; Danti, S. Raman spectroscopy of osteosarcoma cells. Phys. Biol. 2018, 16, 16007. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Lv, J.; Tian, X.; Wang, Y.; Yu, Y.; Liu, J. Miniaturized swimming soft robot with complex movement actuated and controlled by remote light signals. Sci. Rep. 2015, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ichimura, K.; Oh, S.-K.; Nakagawa, M. Light-driven motion of liquids on a photoresponsive surface. Science 2000, 288, 1624–1626. [Google Scholar] [CrossRef] [PubMed]
- Mahimwalla, Z. Characterization of Photo-Induced Mechanical Responses in Azobenzene Polymers; McGill University Libraries: Montreal, QC, Canada, 2013. [Google Scholar]
- Wang, J.; Sun, L.; Zou, M.; Gao, W.; Liu, C.; Shang, L.; Gu, Z.; Zhao, Y. Bioinspired shape-memory graphene film with tunable wettability. Sci. Adv. 2017, 3, e1700004. [Google Scholar] [CrossRef]
- Yoshida, M.; Lahann, J. Smart nanomaterials. ACS Nano 2008, 2, 1101–1107. [Google Scholar] [CrossRef]
- Gumbley, P.; Koylu, D.; Thomas, S.W., III. Photoresponsive polymers containing nitrobenzyl esters via ring-opening metathesis polymerization. Macromolecules 2011, 44, 7956–7961. [Google Scholar] [CrossRef]
- Zakrevskyy, Y.; Cywinski, P.; Cywinska, M.; Paasche, J.; Lomadze, N.; Reich, O.; Löhmannsröben, H.-G.; Santer, S. Interaction of photosensitive surfactant with DNA and poly acrylic acid. J. Chem. Phys. 2014, 140, 01B613_1. [Google Scholar] [CrossRef]
- Konak, C.; Kopeckova, P.; Kopecek, J. Photoregulated association of N-(2-hydroxypropyl) methacrylamide copolymers with azobenzene-containing side chains. Macromolecules 1992, 25, 5451–5456. [Google Scholar] [CrossRef]
- Sugiyama, K.; Sono, K. Characterization of photo-and thermoresponsible amphiphilic copolymers having azobenzene moieties as side groups. J. Appl. Polym. Sci. 2001, 81, 3056–3063. [Google Scholar] [CrossRef]
- Cao, P.-F.; Mangadlao, J.D.; Advincula, R.C. Stimuli-responsive polymers and their potential applications in oil-gas industry. Polym. Rev. 2015, 55, 706–733. [Google Scholar] [CrossRef]
- Motornov, M.; Roiter, Y.; Tokarev, I.; Minko, S. Stimuli-responsive nanoparticles, nanogels and capsules for integrated multifunctional intelligent systems. Prog. Polym. Sci. 2010, 35, 174–211. [Google Scholar] [CrossRef]
- Giustina, G.D.; Giulitti, S.; Brigo, L.; Zanatta, M.; Tromayer, M.; Liska, R.; Elvassore, N.; Brusatin, G. Hydrogel with Orthogonal Reactive Units: 2D and 3D Cross-Linking Modulation. Macromol. Rapid Commun. 2017, 38, 1600570. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, R.R.; Elella, M.H.A.; Sabaa, M.W. Cytotoxicity and metal ions removal using antibacterial biodegradable hydrogels based on N-quaternized chitosan/poly (acrylic acid). Int. J. Biol. Macromol. 2017, 98, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.W.; Hennink, W.E. Polymeric micelles in anticancer therapy: Targeting, imaging and triggered release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Shimoboji, T.; Ding, Z.L.; Stayton, P.S.; Hoffman, A.S. Photoswitching of ligand association with a photoresponsive polymer- protein conjugate. Bioconjug. Chem. 2002, 13, 915–919. [Google Scholar] [CrossRef]
- Lee, M.-J.; Jung, D.-H.; Han, Y.-K. Photo-responsive polymers and their applications to optical memory. Mol. Cryst. Liq. Cryst. 2006, 444, 41–50. [Google Scholar] [CrossRef]
- Babin, J.; Pelletier, M.; Lepage, M.; Allard, J.-F.; Morris, D.; Zhao, Y. A new two-photon-sensitive block copolymer nanocarrier. Angew. Chem. Int. Ed. 2009, 48, 3329–3332. [Google Scholar] [CrossRef]
- Jiang, J.; Tong, X.; Zhao, Y. A new design for light-breakable polymer micelles. J. Am. Chem. Soc. 2005, 127, 8290–8291. [Google Scholar] [CrossRef]
- Zhao, Y. Rational design of light-controllable polymer micelles. Chem. Rec. 2007, 7, 286–294. [Google Scholar] [CrossRef]
- Choi, H.-J.; Montemagno, C.D. Artificial organelle: ATP synthesis from cellular mimetic polymersomes. Nano Lett. 2005, 5, 2538–2542. [Google Scholar] [CrossRef]
- Amiji, M.; Tailor, R.; Ly, M.-K.; Goreham, J. Gelatin-poly (ethylene oxide) semi-interpenetrating polymer network with pH-sensitive swelling and enzyme-degradable properties for oral drug delivery. Drug Dev. Ind. Pharm. 1997, 23, 575–582. [Google Scholar] [CrossRef]
- Bilia, A.; Carelli, V.; Di Colo, G.; Nannipieri, E. In vitro evaluation of a pH-sensitive hydrogel for control of GI drug delivery from silicone-based matrices. Int. J. Pharm. 1996, 130, 83–92. [Google Scholar] [CrossRef]
- Bonina, P.; Petrova, T.S.; Manolova, N. pH-sensitive hydrogels composed of chitosan and polyacrylamide--preparation and properties. J. Bioact. Compat. Polym. 2004, 19, 101–116. [Google Scholar] [CrossRef]
- Ferreira, L.; Vidal, M.M.; Gil, M.H. Evaluation of poly (2-hydroxyethyl methacrylate) gels as drug delivery systems at different pH values. Int. J. Pharm. 2000, 194, 169–180. [Google Scholar] [CrossRef]
- Gupta, P.; Vermani, K.; Garg, S. Hydrogels: From controlled release to pH-responsive drug delivery. Drug Discov. Today 2002, 7, 569–579. [Google Scholar] [CrossRef]
- Mishra, R.K.; Ramasamy, K.; Majeed, A.B.A. pH-responsive poly (DMAPMA-co-HEMA)-based hydrogels for prolonged release of 5-fluorouracil. J. Appl. Polym. Sci. 2012, 126, E98–E107. [Google Scholar] [CrossRef]
- Patel, V.R.; Amiji, M.M. Preparation and characterization of freeze-dried chitosan-poly (ethylene oxide) hydrogels for site-specific antibiotic delivery in the stomach. Pharm. Res. 1996, 13, 588–593. [Google Scholar] [CrossRef]
- Raafat, A.I. Gelatin based pH-sensitive hydrogels for colon-specific oral drug delivery: Synthesis, characterization, and in vitro release study. J. Appl. Polym. Sci. 2010, 118, 2642–2649. [Google Scholar] [CrossRef]
- Sethuraman, V.A.; Na, K.; Bae, Y.H. pH-responsive sulfonamide/PEI system for tumor specific gene delivery: An in vitro study. Biomacromolecules 2006, 7, 64–70. [Google Scholar] [CrossRef]
- Shantha, K.L.; Harding, D.R.K. Preparation and in-vitro evaluation of poly [N-vinyl-2-pyrrolidone-polyethylene glycol diacrylate]-chitosan interpolymeric pH-responsive hydrogels for oral drug delivery. Int. J. Pharm. 2000, 207, 65–70. [Google Scholar] [CrossRef]
- Webber, G.B.; Wanless, E.J.; Bütün, V.; Armes, S.P.; Biggs, S. Self-organized monolayer films of stimulus-responsive micelles. Nano Lett. 2002, 2, 1307–1313. [Google Scholar] [CrossRef]
- Şen, M.; Uzun, C.; Güven, O. Controlled release of terbinafine hydrochloride from pH sensitive poly (acrylamide/maleic acid) hydrogels. Int. J. Pharm. 2000, 203, 149–157. [Google Scholar]
- Wright, D.B.; Patterson, J.P.; Pitto-Barry, A.; Cotanda, P.; Chassenieux, C.; Colombani, O.; O’Reilly, R.K. Tuning the aggregation behavior of pH-responsive micelles by copolymerization. Polym. Chem. 2015, 6, 2761–2768. [Google Scholar] [CrossRef]
- Battogtokh, G.; Ko, Y.T. Active-targeted pH-responsive albumin--photosensitizer conjugate nanoparticles as theranostic agents. J. Mater. Chem. B 2015, 3, 9349–9359. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.; Hanif, M.; Khan, M.A. pH responsive alginate polymeric rafts for controlled drug release by using box behnken response surface design. Des. Monomers Polym. 2017, 20, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Nie, W.; He, C.; Zhou, X.; Chen, L.; Qiu, K.; Wang, W.; Yin, Z. Effect of pH-responsive alginate/chitosan multilayers coating on delivery efficiency, cellular uptake and biodistribution of mesoporous silica nanoparticles based nanocarriers. ACS Appl. Mater. Interfaces 2014, 6, 8447–8460. [Google Scholar] [CrossRef] [PubMed]
- Serres, A.; Baudyš, M.; Kim, S.W. Temperature and pH-sensitive polymers for human calcitonin delivery. Pharm. Res. 1996, 13, 196–201. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, L.; Ma, J.; Wang, J.; Yu, H.; Xiao, A. Temperature-and pH-responsive star amphiphilic block copolymer prepared by a combining strategy of ring-opening polymerization and reversible addition--fragmentation transfer polymerization. Eur. Polym. J. 2010, 46, 1288–1298. [Google Scholar] [CrossRef]
- Xu, D.; Asai, D.; Chilkoti, A.; Craig, S.L. Rheological properties of cysteine-containing elastin-like polypeptide solutions and hydrogels. Biomacromolecules 2012, 13, 2315–2321. [Google Scholar] [CrossRef][Green Version]
- Yin, R.; Wang, K.; Du, S.; Chen, L.; Nie, J.; Zhang, W. Design of genipin-crosslinked microgels from concanavalin A and glucosyloxyethyl acrylated chitosan for glucose-responsive insulin delivery. Carbohydr. Polym. 2014, 103, 369–376. [Google Scholar] [CrossRef]
- Chen, M.; Huang, C.; He, C.; Zhu, W.; Xu, Y.; Lu, Y. A glucose-responsive controlled release system using glucose oxidase-gated mesoporous silica nanocontainers. Chem. Commun. 2012, 48, 9522–9524. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhang, H.; He, Q.; Li, Y.; Gu, J.; Li, L.; Li, H.; Shi, J. A glucose-responsive controlled release of insulin system based on enzyme multilayers-coated mesoporous silica particles. Chem. Commun. 2011, 47, 9459–9461. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Altunbas, A.; Yucel, T.; Nagarkar, R.P.; Schneider, J.P.; Pochan, D.J. Injectable solid hydrogel: Mechanism of shear-thinning and immediate recovery of injectable β-hairpin peptide hydrogels. Soft Matter 2010, 6, 5143–5156. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, L.; Detamore, M.S.; Berkland, C. Biodegradable colloidal gels as moldable tissue engineering scaffolds. Adv. Mater. 2008, 20, 236–239. [Google Scholar] [CrossRef]
- Żołek-Tryznowska, Z. 6-Rheology of Printing Inks. Print. Polym. William Andrew Publ. 2016, 87–99. [Google Scholar] [CrossRef]
- Azimi, B.; Milazzo, M.; Lazzeri, A.; Berrettini, S.; Uddin, M.J.; Qin, Z.; Buehler, M.J.; Danti, S. Electrospinning Piezoelectric Fibers for Biocompatible Devices. Adv. Healthc. Mater. 2019. [Google Scholar] [CrossRef] [PubMed]
- Fukada, E. Piezoelectricity of biopolymers. Biorheology 1995, 32, 593–609. [Google Scholar] [CrossRef]
- Mota, C.; Labardi, M.; Trombi, L.; Astolfi, L.; D’Acunto, M.; Puppi, D.; Gallone, G.; Chiellini, F.; Berrettini, S.; Bruschini, L.; et al. Design, fabrication and characterization of composite piezoelectric ultrafine fibers for cochlear stimulation. Mater. Des. 2017, 122, 206–219. [Google Scholar] [CrossRef]
- Danti, S.; Azimi, B.; Candito, M.; Fusco, A.; Sorayani Bafqi, M.S.; Ricci, C.; Milazzo, M.; Cristallini, C.; Latifi, M.; Donnarumma, G.; et al. Lithium niobate nanoparticles as biofunctional interface material for inner ear devices. Biointerphases 2020, 15, 31004. [Google Scholar] [CrossRef]
- Azimi, B.; Sorayani Bafqi, M.S.; Fusco, A.; Ricci, C.; Gallone, G.; Bagherzadeh, R.; Donnarumma, G.; Uddin, M.J.; Latifi, M.; Lazzeri, A.; et al. Electrospun ZnO/poly (vinylidene fluoride-trifluoroethylene) scaffolds for lung tissue engineering. Tissue Eng. Part A 2020. [Google Scholar] [CrossRef]
- Chowdhury, A.R.; Abdullah, A.M.; Hussain, I.; Lopez, J.; Cantu, D.; Gupta, S.K.; Mao, Y.; Danti, S.; Uddin, M.J. Lithium doped zinc oxide based flexible piezoelectric-triboelectric hybrid nanogenerator. Nano Energy 2019, 61, 327–336. [Google Scholar] [CrossRef]
- Chowdhury, A.R.; Jaksik, J.; Hussain, I.; Tran, P.; Danti, S.; Uddin, M.J. Surface Modified Nanostructured Piezoelectric Device as Cost-Effective Transducer for Energy and Biomedicine. Energy Technol. 2018. [Google Scholar] [CrossRef]
- Bodkhe, S.; Turcot, G.; Gosselin, F.P.; Therriault, D. One-step solvent evaporation-assisted 3D printing of piezoelectric PVDF nanocomposite structures. ACS Appl. Mater. Interfaces 2017, 9, 20833–20842. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Zhou, C.; Chung, D.D.L. Piezoelectric behavior of three-dimensionally printed acrylate polymer without filler or poling. J. Mater. Sci. 2018, 53, 6819–6830. [Google Scholar] [CrossRef]
- Chen, Z.; Song, X.; Lei, L.; Chen, X.; Fei, C.; Chiu, C.T.; Qian, X.; Ma, T.; Yang, Y.; Shung, K.; et al. 3D printing of piezoelectric element for energy focusing and ultrasonic sensing. Nano Energy 2016, 27, 78–86. [Google Scholar] [CrossRef]
- Egunov, A.I.; Korvink, J.G.; Luchnikov, V.A. Polydimethylsiloxane bilayer films with an embedded spontaneous curvature. Soft Matter 2016, 12, 45–52. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.H.; Ryu, H.; Lee, J.-H.; Khan, U.; Kim, H.; Kwak, S.S.; Kim, S.-W. High-Performance Piezoelectric, Pyroelectric, and Triboelectric Nanogenerators Based on P (VDF-TrFE) with Controlled Crystallinity and Dipole Alignment. Adv. Funct. Mater. 2017, 27, 1700702. [Google Scholar] [CrossRef]
- Kim, K.; Zhu, W.; Qu, X.; Aaronson, C.; McCall, W.R.; Chen, S.; Sirbuly, D.J. 3D optical printing of piezoelectric nanoparticle--polymer composite materials. ACS Nano 2014, 8, 9799–9806. [Google Scholar] [CrossRef]
- Ge, Q.; Dunn, C.K.; Qi, H.J.; Dunn, M.L. Active origami by 4D printing. Smart Mater. Struct. 2014, 23, 94007. [Google Scholar] [CrossRef]
- Ge, Q.; Qi, H.J.; Dunn, M.L. Active materials by four-dimension printing. Appl. Phys. Lett. 2013, 103, 131901. [Google Scholar] [CrossRef]
- Kim, H.; Torres, F.; Villagran, D.; Stewart, C.; Lin, Y.; Tseng, T.-L.B. 3D printing of BaTiO3/PVDF composites with electric in situ poling for pressure sensor applications. Macromol. Mater. Eng. 2017, 302, 1700229. [Google Scholar] [CrossRef]
- Kuang, X.; Roach, D.J.; Wu, J.; Hamel, C.M.; Ding, Z.; Wang, T.; Dunn, M.L.; Qi, H.J. Advances in 4D printing: Materials and applications. Adv. Funct. Mater. 2019, 29, 1805290. [Google Scholar] [CrossRef]
- Joshi, S.; Rawat, K.; Karunakaran, C.; Rajamohan, V.; Mathew, A.T.; Koziol, K.; Thakur, V.K.; Balan, A.S.S. 4D printing of materials for the future: Opportunities and challenges. Appl. Mater. Today 2020, 18, 100490. [Google Scholar] [CrossRef]
- Chu, H.; Yang, W.; Sun, L.; Cai, S.; Yang, R.; Liang, W.; Yu, H.; Liu, L. 4D Printing: A Review on Recent Progresses. Micromachines 2020, 11, 796. [Google Scholar] [CrossRef]
- Wan, X.; Luo, L.; Liu, Y.; Leng, J. Direct Ink Writing Based 4D Printing of Materials and Their Applications. Adv. Sci. 2020, 7, 2001000. [Google Scholar] [CrossRef]
- Wan, Z.; Zhang, P.; Liu, Y.; Lv, L.; Zhou, Y. Four-dimensional bioprinting: Current developments and applications in bone tissue engineering. Acta Biomater. 2020, 101, 26–42. [Google Scholar] [CrossRef]
- Yang, Q.; Gao, B.; Xu, F. Recent advances in 4D bioprinting. Biotechnol. J. 2020, 15, 1900086. [Google Scholar] [CrossRef]
- Lui, Y.S.; Sow, W.T.; Tan, L.P.; Wu, Y.; Lai, Y.; Li, H. 4D printing and stimuli-responsive materials in biomedical aspects. Acta Biomater. 2019, 92, 19–36. [Google Scholar] [CrossRef]
- Wong, K.V.; Hernandez, A. A review of additive manufacturing. ISRN Mech. Eng. 2012, 1, 1–10. [Google Scholar] [CrossRef]
- Apsite, I.; Constante, G.; Dulle, M.; Vogt, L.; Caspari, A.; Boccaccini, A.R.; Synytska, A.; Salehi, S.; Ionov, L. 4D biofabrication of fibrous artificial nerve graft for neuron regeneration. Biofabrication 2020, 12, 035027. [Google Scholar] [CrossRef]
- Apsite, I.; Uribe, J.M.; Posada, A.F.; Rosenfeldt, S.; Salehi, S.; Ionov, L. 4D biofabrication of skeletal muscle microtissues. Biofabrication 2019, 12, 15016. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yang, F.; Zhao, H.; Gao, Q.; Xia, B.; Fu, J. Research on the printability of hydrogels in 3D bioprinting. Sci. Rep. 2016, 6, 29977. [Google Scholar] [CrossRef] [PubMed]
- Cholleti, E.R. A Review on 3D printing of piezoelectric materials. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Istanbul, Turkey, 20–24 June 2018; pp. 455–473. [Google Scholar]
- Cui, H.; Hensleigh, R.; Yao, D.; Maurya, D.; Kumar, P.; Kang, M.G.; Priya, S.; Zheng, X.R. Three-dimensional printing of piezoelectric materials with designed anisotropy and directional response. Nat. Mater. 2019, 18, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ware, H.O.T.; Baker, E.; Chu, W.; Hu, J.; Sun, C. The development of an all-polymer-based piezoelectric photocurable resin for additive manufacturing. Procedia CIRP 2017, 65, 157–162. [Google Scholar] [CrossRef]
- Lee, S.C.; Gillispie, G.; Prim, P.; Lee, S.J. Physical and Chemical Factors Influencing the Printability of Hydrogel-based Extrusion Bioinks. Chem. Rev. 2020, 120, 10834–10886. [Google Scholar] [CrossRef]
- Naghieh, S.; Sarker, M.D.; Sharma, N.K.; Barhoumi, Z.; Chen, X. Printability of 3D printed hydrogel scaffolds: Influence of hydrogel composition and printing parameters. Appl. Sci. 2020, 10, 292. [Google Scholar] [CrossRef]
- Boularaoui, S.; Al Hussein, G.; Khan, K.A.; Christoforou, N.; Stefanini, C. An overview of extrusion-based bioprinting with a focus on induced shear stress and its effect on cell viability. Bioprinting 2020, 20, e00093. [Google Scholar] [CrossRef]
- Zhang, J.; Yun, S.; Karami, A.; Jing, B.; Zannettino, A.; Du, Y.; Zhang, H. 3D printing of a thermosensitive hydrogel for skin tissue engineering: A proof of concept study. Bioprinting 2020, e00089. [Google Scholar] [CrossRef]
- Celikkin, N.; Simó Padial, J.; Costantini, M.; Hendrikse, H.; Cohn, R.; Wilson, C.J.; Rowan, A.E.; Święszkowski, W. 3D printing of thermoresponsive polyisocyanide (PIC) hydrogels as bioink and fugitive material for tissue engineering. Polymers (Basel) 2018, 10, 555. [Google Scholar] [CrossRef]
- Sossou, G.; Demoly, F.; Belkebir, H.; Qi, H.J.; Gomes, S.; Montavon, G. Design for 4D printing: Modeling and computation of smart materials distributions. Mater. Des. 2019, 181, 108074. [Google Scholar] [CrossRef]
- Medeiros, S.F.; Santos, A.M.; Fessi, H.; Elaissari, A. Stimuli-responsive magnetic particles for biomedical applications. Int. J. Pharm. 2011, 403, 139–161. [Google Scholar] [CrossRef] [PubMed]
- Villar, G.; Heron, A.J.; Bayley, H. Formation of droplet networks that function in aqueous environments. Nat. Nanotechnol. 2011, 6, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Kirillova, A.; Maxson, R.; Stoychev, G.; Gomillion, C.T.; Ionov, L. 4D biofabrication using shape-morphing hydrogels. Adv. Mater. 2017, 29, 1703443. [Google Scholar] [CrossRef] [PubMed]
- Apsite, I.; Stoychev, G.; Zhang, W.; Jehnichen, D.; Xie, J.; Ionov, L. Porous stimuli-responsive self-folding electrospun mats for 4D biofabrication. Biomacromolecules 2017, 18, 3178–3184. [Google Scholar] [CrossRef]
- Ye, C.; Nikolov, S.V.; Calabrese, R.; Dindar, A.; Alexeev, A.; Kippelen, B.; Kaplan, D.L.; Tsukruk, V.V. Self-(Un) rolling Biopolymer Microstructures: Rings, Tubules, and Helical Tubules from the Same Material. Angew. Chem. 2015, 127, 8610–8613. [Google Scholar] [CrossRef]
- Huang, Y.; Fitzpatrick, V.; Zheng, N.; Cheng, R.; Huang, H.; Ghezzi, C.; Kaplan, D.L.; Yang, C. Self-Folding 3D Silk Biomaterial Rolls to Facilitate Axon and Bone Regeneration. Adv. Healthc. Mater. 2020, 9, 2000530. [Google Scholar] [CrossRef]
- Kumar, A.; Han, S.S. Emergence of bioprinting in tissue engineering: A mini review. Adv. Tissue Eng. Regen. Med. 2016, 1, 13. [Google Scholar] [CrossRef]
- Dababneh, A.B.; Ozbolat, I.T. Bioprinting technology: A current state-of-the-art review. J. Manuf. Sci. Eng. 2014, 136. [Google Scholar] [CrossRef]
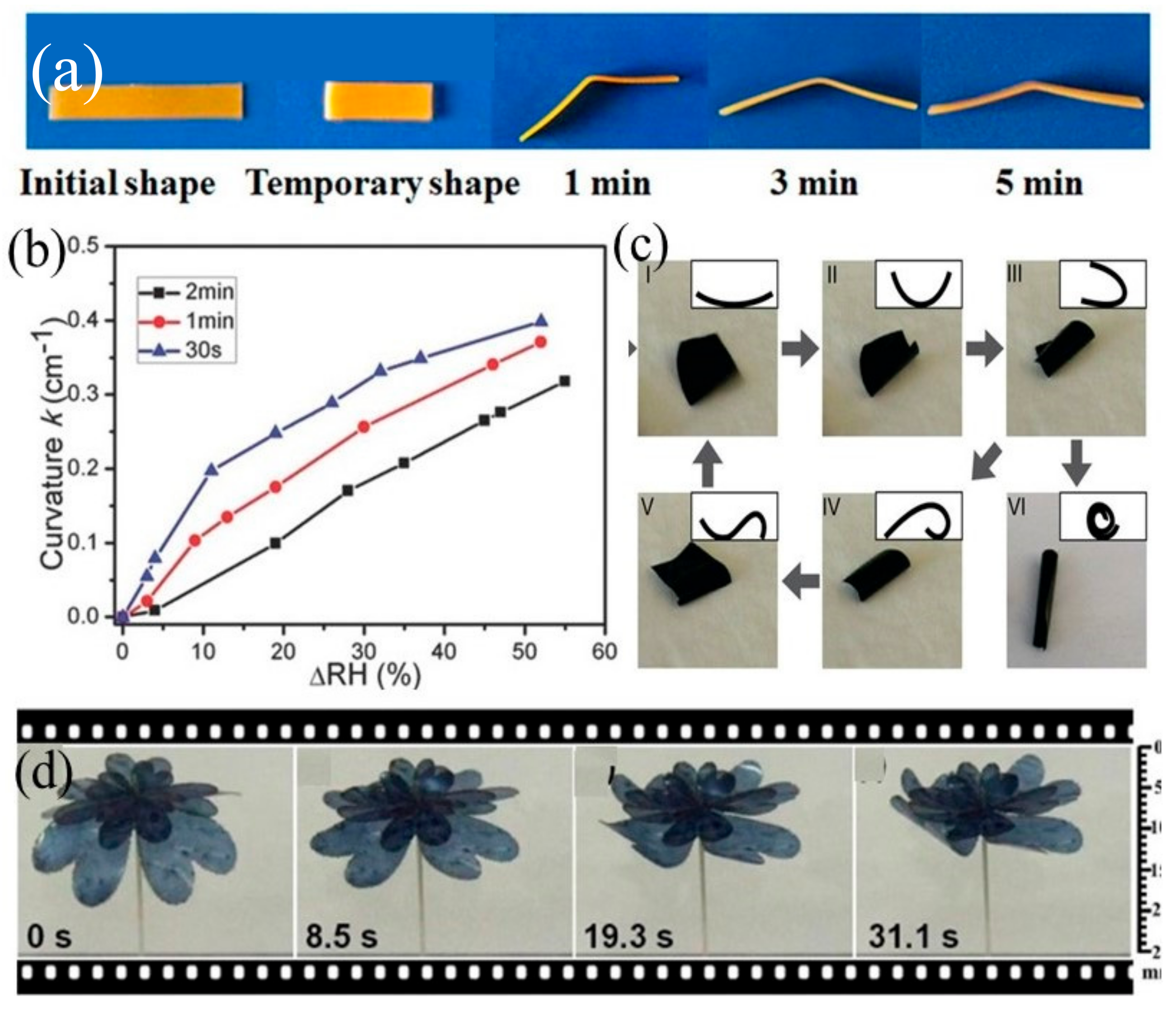


| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| PEG–PCAD@AG | Humidity | For applications that require contactless actuation in response to the day–night rhythm of atmospheric humidity | [48] |
| PEG–PCL–CNC | Humidity | Medical applications such as smart sutures, vascular stents, esophageal stents, tissue engineering scaffolds, and drug delivery | [49] |
| Liquid–crystalline (LC) polymer networks activated with an alkaline solution | Humidity | Humidity-responsive actuators for application in smart textiles | [47,50] |
| Poly(ethylene glycol) diacrylate hydrogel | Humidity | Sensors, actuators, or construction of soft robots, artificial muscles, molecular motors, programmable origami, and energy generators | [51,52] |
| Polyethylene glycol | Humidity | Tissue engineering | [53] |
| Combining polypyrrole and polyol-borate | Humidity | Actuator and generator | [54] |
| Cellulose stearoyl esters (CSEs) | Humidity, temperature | Sensors, actuators, or construction of soft robots | [55] |
| Poly(3,4-ethylenedioxythiophene): polystyrene sulfonate and poly(vinylidene fluoride) PEDOT:PSS/PVDF composite | Humidity | Actuator and generator | [56] |
| Printing a layer of hydrophilic material over a layer of rigid material | Humidity | Self-evolving structure | [57] |
| Hydrogel composite ink composed of stiff cellulose fibrils embedded in a soft acrylamide matrix | Humidity | Tissue engineering, biomedical devices, soft robotics | [58] |
| Poly(ionic liquid)s (PILs), poly(1-methyl azobenzene-3-vinylimidazolium bromide) ([PMAzoVIm]Br) and poly(acrylic acid) (PAA) | Humidity and light | Selective dye adsorption | [59] |
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Polythiophene (PT) | Electricity | Drug delivery, biosensing, implant devices | [94,95] |
| Sulfonated-polystyrene (PSS) | Electricity | Active ingredients in marketed vaginal contraceptive | [96] |
| Poly(2-(acrylamide)-2-methylpropanesulfonic acid) (PAMPS) | Electricity | Actuators or artificial muscles | [97] |
| Poly(2-hydroxyethyl methacrylate) (PHEMA) | Electricity | Drug delivery | [98] |
| Poly(L-lactic acid)/Poly(3,4-ethylenedioxythiophene) (PLLA/PEDOT) | Electricity | Electrically conductive 3D scaffold for tissue engineering | [99] |
| Poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) | Electricity | Smart Micro Electro-Mechanical Systems (MEMS) interface applications, neural tissue engineering | [100,101,102] |
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Cobalt ferrite (CoFe2O4) | Magnetism | Controlled drug release | [29,108,115] |
| Ferromagnetic cobalt nanoparticles | Magnetism | [107] | |
| Iron platinum (FePt) | Magnetism | [29,115] | |
| Iron (III) oxide (Fe2O3) | Magnetism | Drug delivery application | [29,115] |
| Iron (II, III) oxide (Fe3O4) | Magnetism | [29,115] | |
| CoPt | Magnetism | Drug delivery application | [115] |
| (Fe3O4) with PNIPAM, PVA, Poly(acrylamide), alginate | Magnetism | Semiconductor, active scaffolds for on-demand drug and cell delivery | [105,106,109,110,112] |
| Iron nanoparticles/[11-(2-bromo-2-methyl)-propionyloxy]undecyltrichlorosilane (Fenp-PS gel) | Magnetism | Cancer therapy, sensors, switches, various separations, membranes, and drug delivery systems | [113] |
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Polymers containing Azobenzene groups | Light (ultraviolet (UV) light) | Swimming soft robot | [119,120,121] |
| Polymers containing Spiropyran groups | Light | [123] | |
| Polymers containing Nitrobenzyl groups | Light | [124] | |
| Poly (acrylic acid) (PAA) containing Azobenzene groups | Light | [125] | |
| Poly (N-(2-hydroxypropyl) methacrylamide) (PHPMAm) containing Azobenzene groups | Light | Polymeric micelles in anticancer therapy, scaffold for tissue engineering | [126,127] |
| PNIPAAm | Light | [74] | |
| PEO-PPyMA (poly(1-pyrenylmethyl methacrylate)) | Light | Micelles in anticancer therapy | [136,137] |
| PEO-b-PNBMA (poly(2-nitrobenzylmethyl methacrylate)) | Light (UV/NIR) | Micelles in anticancer therapy | [137] |
| PEO-b-P(MA-DEACM) | Light (UV/NIR) | Micelles in anticancer therapy | [135] |
| Material(s) | Mechanism(s) | Application(s) | Ref. |
|---|---|---|---|
| Poly(acrylic acid) (PAA) | pH | Peroral-controlled drug delivery, taste-masking of bitter drugs, and intravascular drug release during elevated blood pH in certain cardiovascular defects | [143] |
| Poly(acrylic acid) (PAA):PEO | pH | Drug delivery | [140] |
| Based on gelatin (G) and acrylic acid (AAc) | pH | Colon-specific oral drug delivery | [146] |
| Poly(methacrylic acid) (PMAA) | pH | [143] | |
| Poly(acrylamide/maleic acid) (P(AAm/MA)) | pH | Drug delivery | [150] |
| Poly(diethylaminoethyl methacrylate) (PDEAEMA) | pH | [143] | |
| Poly(dimethylaminoethyl methacrylate) (PDMAEMA) | pH | Drug delivery, DNA transfer | [143,149] |
| Poly (DMAPMA-co-HEMA) | pH | Anticancer drug delivery | [144] |
| Chitosan-PEO | pH | Drug delivery | [145] |
| Chitosan and polyacrylamide (PAAm) | pH | Biomedical field or in agriculture | [141] |
| Gelatin–PEO | pH | Drug delivery | [139] |
| Poly(2-hydroxyethyl methacrylate) (PHEMA) | pH | Drug delivery | [142] |
| N-vinyl pyrrolidone, polyethylene glycol diacrylate, chitosan | pH | Drug delivery | [148] |
| Sulfonamide/polyethyleneimine (PEI) | pH | Tumor specific gene delivery | [147] |
| Poly(2-(diethylamino)ethyl methacrylate-co-2-(dimethylamino)ethyl methacrylate)-b-poly(2-(dimethylamino)ethyl methacrylate), P(DEAEMA-co-DMAEMA)-b-PDMAEMA | pH | [151] | |
| Albumin | pH | Theragnostic agent | [152] |
| Alginate/Chitosan | pH | Drug delivery | [154] |
| Alginate | pH | Drug delivery | [153] |
| Poly(N-isopropyl acrylamide-co-butyl methacrylate-co-acrylic acid) | pH and temperature | Drug delivery | [155] |
| Star-shaped poly(e-caprolactone)-b-poly(2-(dimethylamino)ethyl methacrylate) (HPs-Star-PCL-b-PDMAEMA) | pH and temperature | Controlled drug delivery | [156] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morouço, P.; Azimi, B.; Milazzo, M.; Mokhtari, F.; Fernandes, C.; Reis, D.; Danti, S. Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability. Appl. Sci. 2020, 10, 9143. https://doi.org/10.3390/app10249143
Morouço P, Azimi B, Milazzo M, Mokhtari F, Fernandes C, Reis D, Danti S. Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability. Applied Sciences. 2020; 10(24):9143. https://doi.org/10.3390/app10249143
Chicago/Turabian StyleMorouço, Pedro, Bahareh Azimi, Mario Milazzo, Fatemeh Mokhtari, Cristiana Fernandes, Diana Reis, and Serena Danti. 2020. "Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability" Applied Sciences 10, no. 24: 9143. https://doi.org/10.3390/app10249143
APA StyleMorouço, P., Azimi, B., Milazzo, M., Mokhtari, F., Fernandes, C., Reis, D., & Danti, S. (2020). Four-Dimensional (Bio-)printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability. Applied Sciences, 10(24), 9143. https://doi.org/10.3390/app10249143










