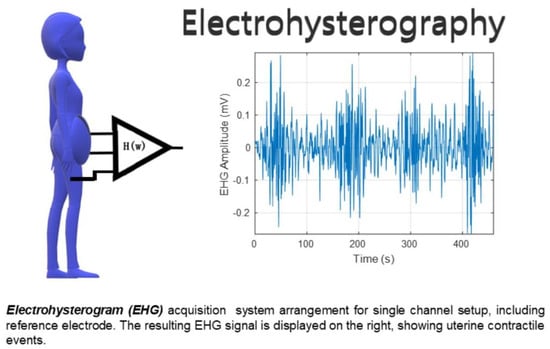
Automatic Contraction Detection Using Uterine Electromyography
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
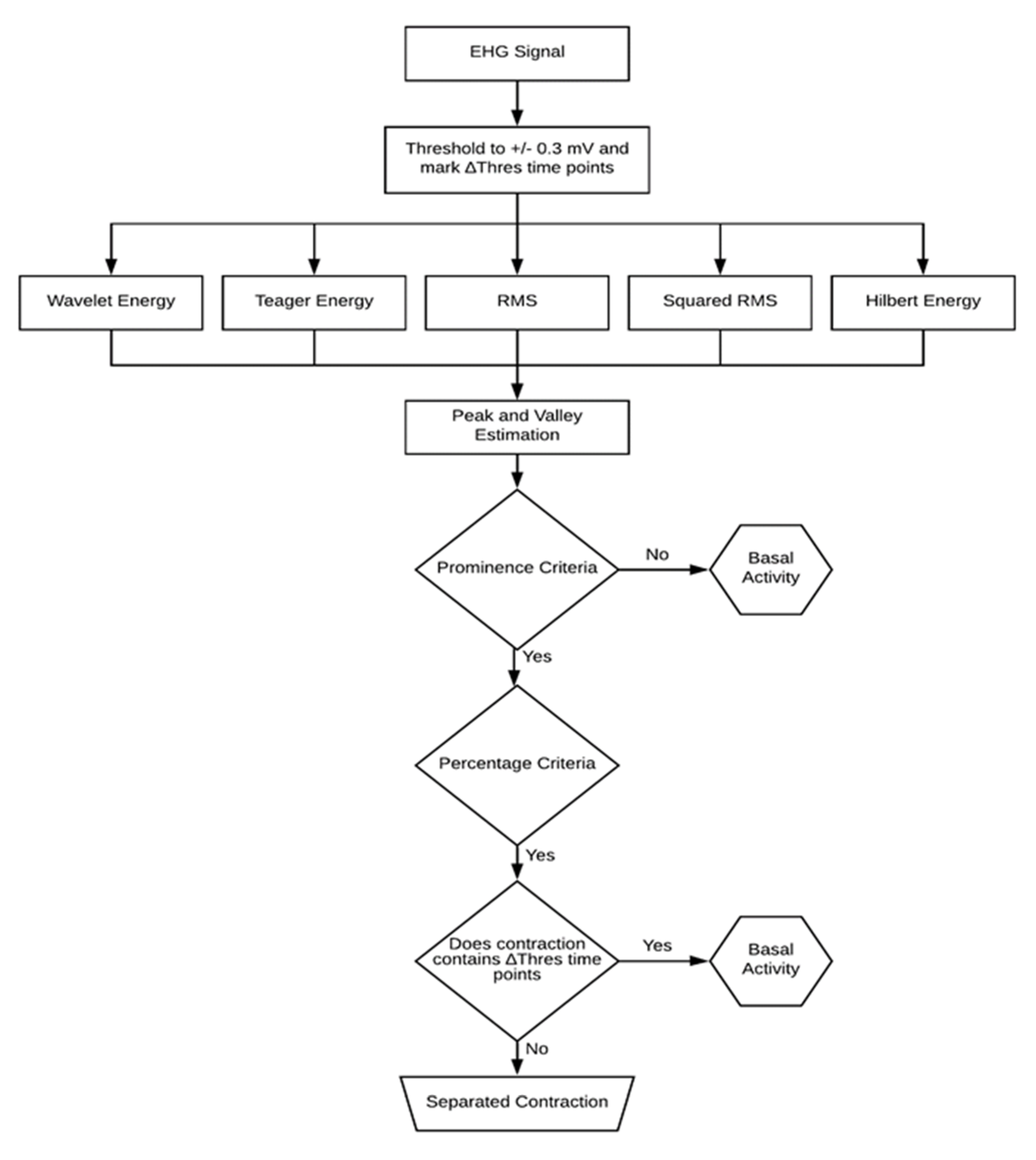
2.2. Data Contraction Detection Methods
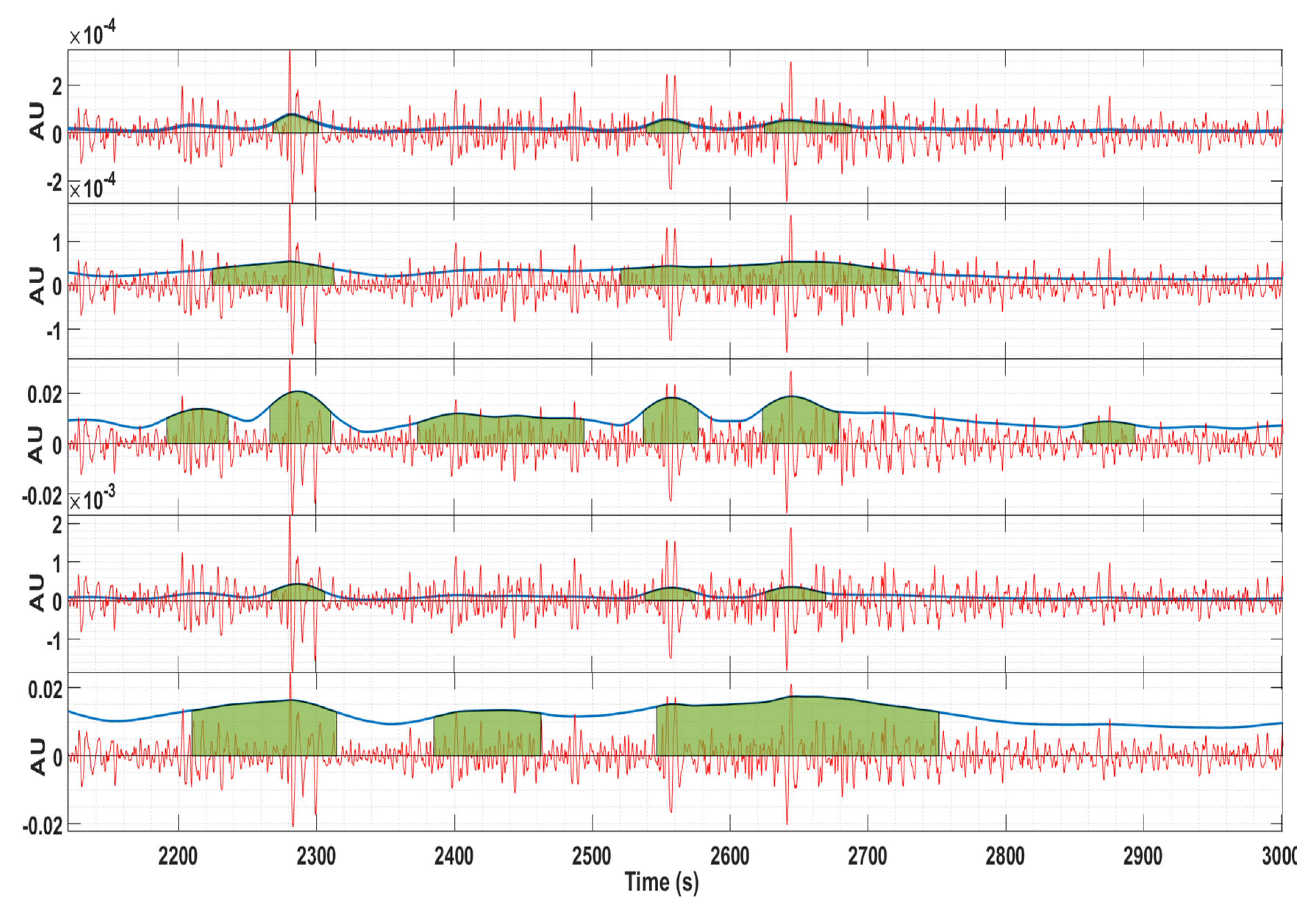
2.2.1. Wavelet Energy
2.2.2. Teager Energy Operator
2.2.3. Root Mean Square
2.2.4. Squared RMS
2.2.5. Hilbert Envelope
2.3. Contraction Detection Algorithm
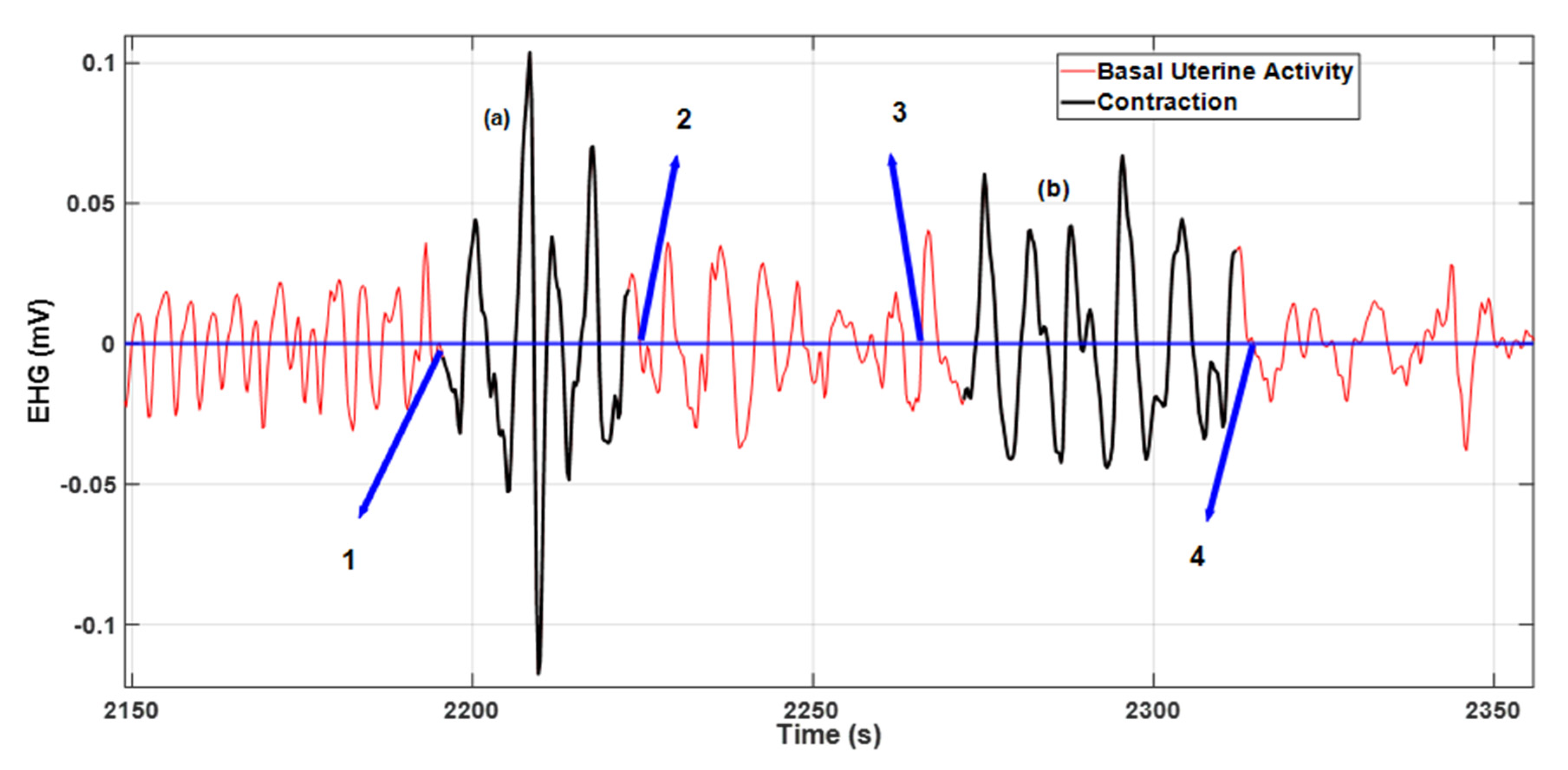
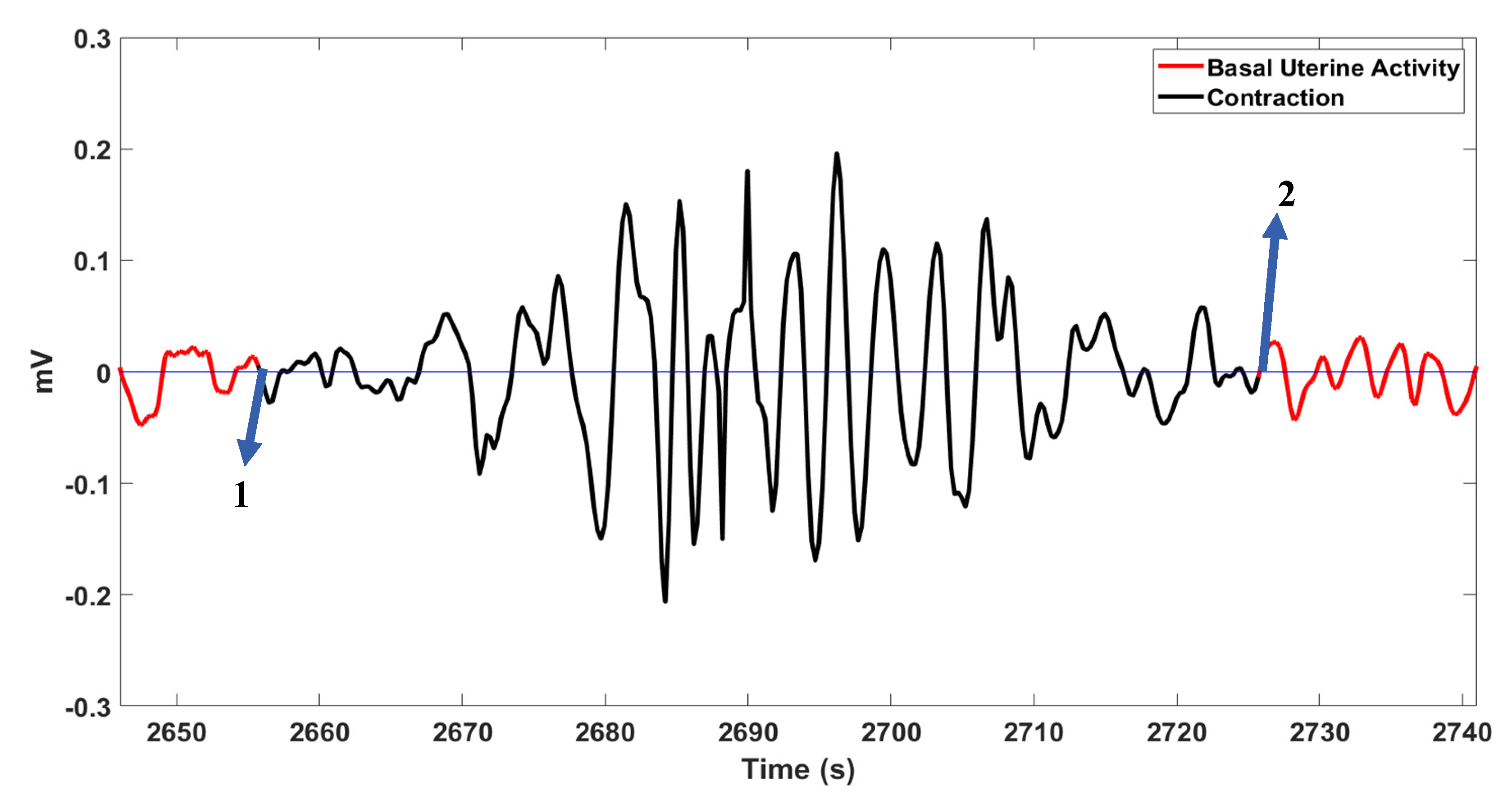
2.4. Visualization and Scoring Methodology
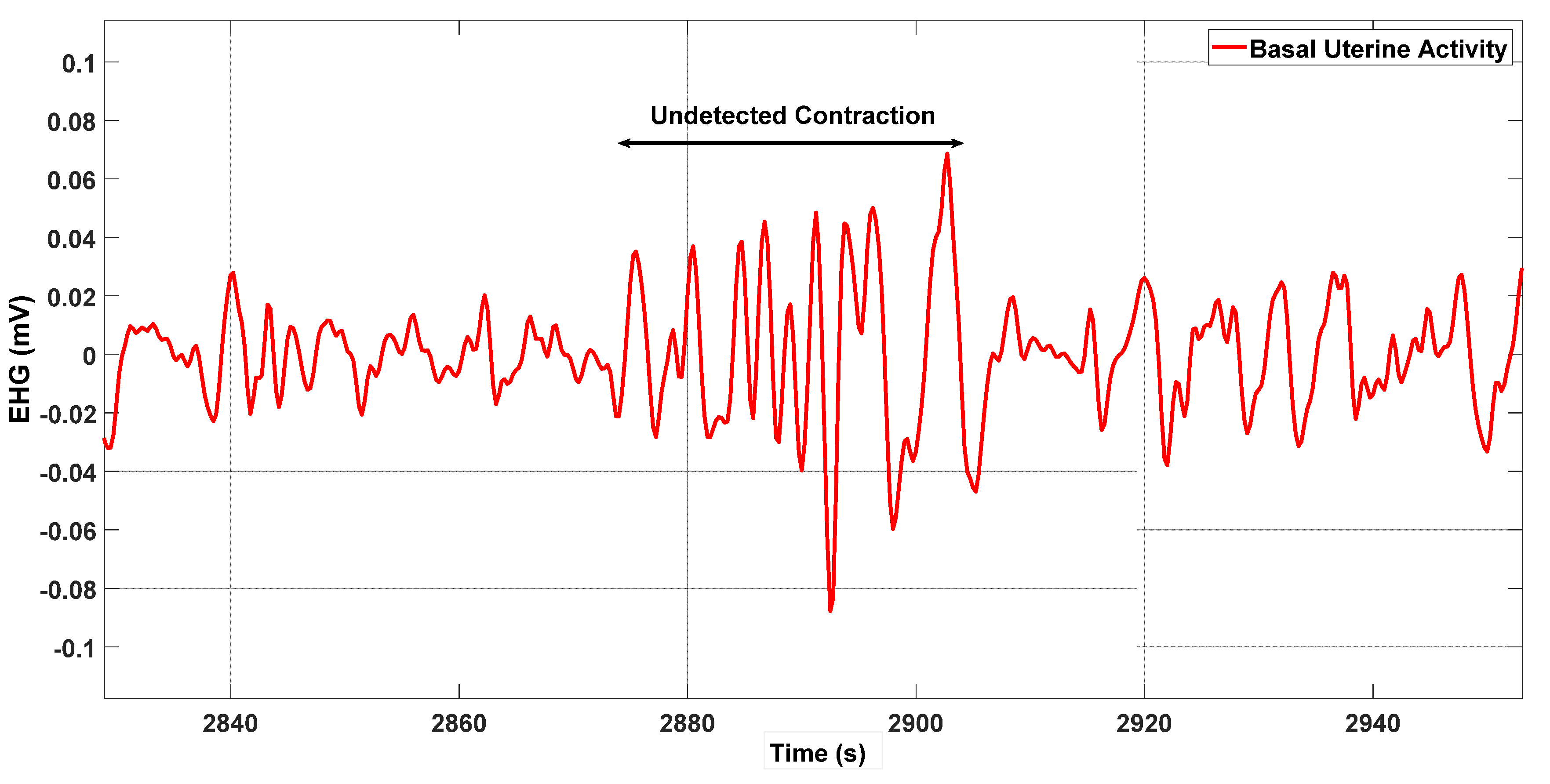
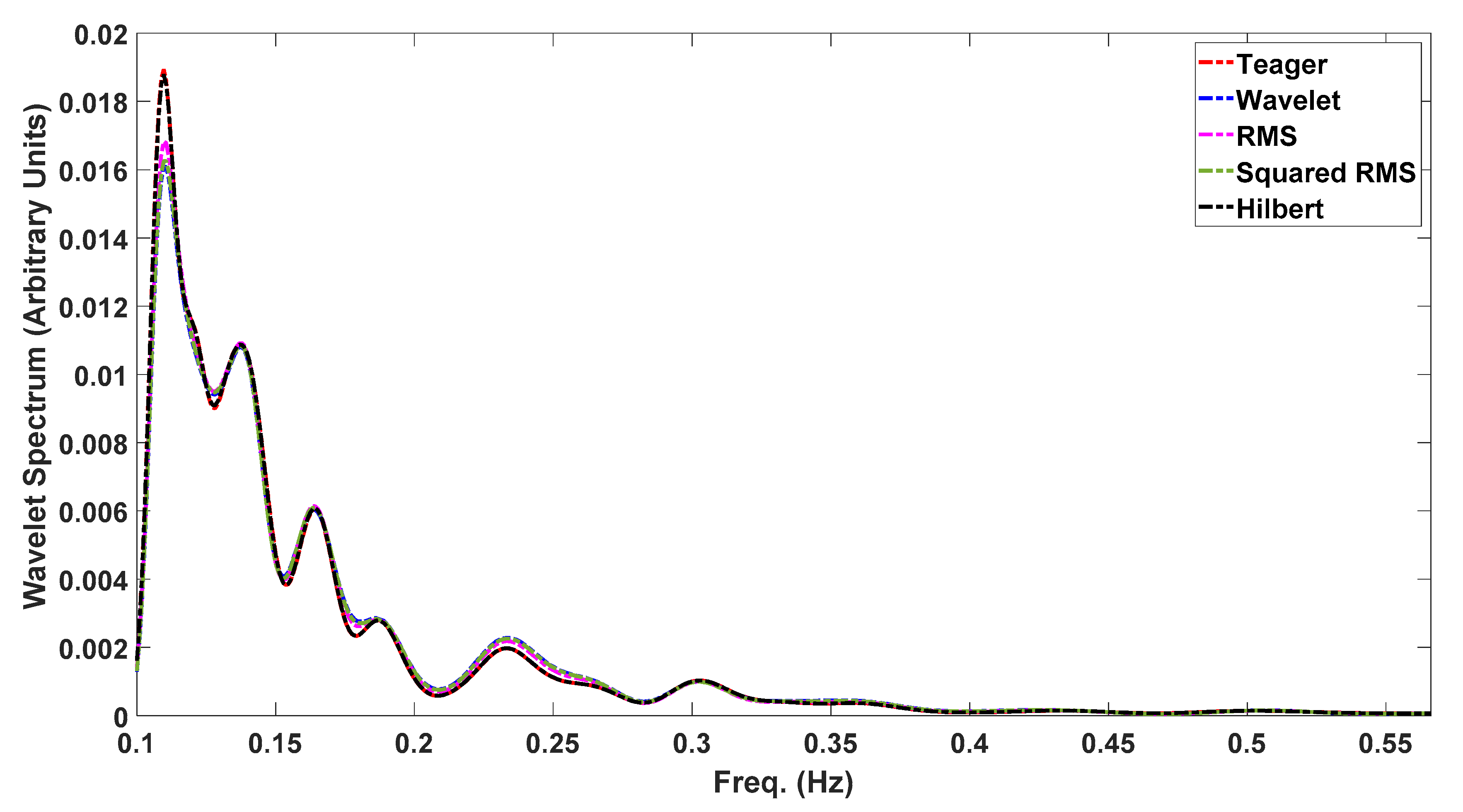
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Alberola-Rubio, J.; Prats-Boluda, G.; Ye-Lin, Y.; Valero, J.; Perales, A.; Garcia-Casado, J. Comparison of non-invasive electrohysterographic recording techniques for monitoring uterine dynamics. Med. Eng. Phys. 2013, 35, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Hadar, E.; Biron-Shental, T.; Gavish, O.; Raban, O.; Yogev, Y. A comparison between electrical uterine monitor, tocodynamometer and intra uterine pressure catheter for uterine activity in labor. J. Matern. Neonatal Med. 2015, 28, 1367–1374. [Google Scholar] [CrossRef]
- Horoba, K.; Wrobel, J.; Jezewski, J.; Kupka, T.; Roj, D.; Jezewski, M. Automated detection of uterine contractions in tocography signals–Comparison of algorithms. Biocybern. Biomed. Eng. 2016, 36, 610–618. [Google Scholar] [CrossRef]
- Alvarez, H.; Caldeyro-Barcia, R. The normal and abnormal contractile waves of the uterus during labour. Gynaecologia 1954, 138, 190–212. [Google Scholar] [CrossRef] [PubMed]
- Auger, F.; Flandrin, P.; Gonçalves, P.; Lemoine, O. Time-Frequency Toolbox Reference Guide; Rice University: Houston, TX, USA, 1995. [Google Scholar]
- Macones, G.A.; Cahill, A.; Stamilio, D.M.; Odibo, A. A new method for assessing uterine activity: Haran et al. Am. J. Obstet. Gynecol. 2012, 206, 449. [Google Scholar] [CrossRef]
- Chendeb, M.; Khalil, M.; Hewson, D.; Duchêne, J. Classification of non stationary signals using multiscale decomposition. J. Biomed. Sci. Eng. 2010, 3, 193–199. [Google Scholar] [CrossRef][Green Version]
- Devedeux, D.; Marque, C.; Mansour, S.; Germain, G.; Duchêne, J. Uterine electromyography: A critical review. Am. J. Obstet. Gynecol. 1993, 169, 1636–1653. [Google Scholar] [CrossRef]
- Fele-Žorž, G.; Kavšek, G.; Novak-Antolič, Ž.; Jager, F. A comparison of various linear and non-linear signal processing techniques to separate uterine EMG records of term and pre-term delivery groups. Med. Biol. Eng. Comput. 2008, 46, 911–922. [Google Scholar] [CrossRef]
- Vlemminx, M.W.C.; Thijssen, K.M.J.; Bajlekov, G.I.; Dieleman, J.P.; der Jagt, M.B.v.; Oei, S.G. Could electrohysterography be the solution for external uterine monitoring in obese women? J. Perinatol. 2018, 38, 580–586. [Google Scholar] [CrossRef]
- Lucovnik, M.; Chambliss, L.R.; Blumrick, R.; Balducci, J.; Gersak, K.; Garfield, R.E. Effect of obesity on preterm delivery prediction by transabdominal recording of uterine electromyography. Taiwan. J. Obstet. Gynecol. 2016, 55, 692–696. [Google Scholar] [CrossRef][Green Version]
- Diab, A. Study of the Nonlinear Properties and Propagation Characteristics of the Uterine Electrical Activity during Pregnancy and Labor. Ph.D. Thesis, Université de Technologie de Compiègne, Compiègne, France, 2015. [Google Scholar]
- Zaylaa, A.; Diab, A.; Khalil, M.; Marque, C. Multichannel EHG segmentation for automatically identifying contractions and motion artifacts. In Proceedings of the 2017 Fourth International Conference on Advances in Biomedical Engineering (ICABME), Beirut, Lebanon, 19–21 October 2017; pp. 1–4. [Google Scholar] [CrossRef]
- Rabotti, C.; Mischi, M.; van Laar, J.O.E.H.; Oei, S.G.; Bergmans, J.W.M. Myometrium electromechanical modeling for internal uterine pressure estimation by electrohysterography. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 6259–6262. [Google Scholar] [CrossRef]
- Bregar, A.T.; Lucovnik, M.; Verdenik, I.; Jager, F.; Gersak, K.; Garfield, R.E. Uterine electromyography during active phase compared with latent phase of labor at term. Acta Obstet. Gynecol. Scand. 2016, 95, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Sims, S.M.; Daniel, E.E.; Garfield, R.E. Improved electrical coupling in uterine smooth muscle is associated with increased numbers of gap junctions at parturition. J. Gen. Physiol. 1982, 80, 353–375. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.M.; Garfield, R.E.; Daniel, E.E. Improved propagation in myometrium associated with gap junctions during parturition. Am. J. Physiol. Physiol. 1989, 256, C130–C141. [Google Scholar] [CrossRef] [PubMed]
- Verhoeff, A. Myometrial Contractility and Gap junCtions: An Experimental Study in Chronically Instrumented Ewes; Erasmus University Rotterdam: Rotterdam, The Netherlands, 1985. [Google Scholar]
- Miyoshi, H.; Boyle, M.B.; MacKay, L.B.; Garfield, R.E. Gap junction currents in cultured muscle cells from human myometrium. Am. J. Obstet. Gynecol. 1998, 178, 588–593. [Google Scholar] [CrossRef]
- Khalil, M.; Duchene, J. Uterine EMG analysis: A dynamic approach for change detection and classification. IEEE Trans. Biomed. Eng. 2000, 47, 748–756. [Google Scholar] [CrossRef]
- Marque, C.; Gondry, J.; Rossi, J.; Baaklini, N.; Duchêne, J. Surveillance des grossesses à risque par électromyographie utérine. RBM-News 1995, 17, 25–31. [Google Scholar] [CrossRef]
- Horoba, K.; Jezewski, J.; Wrobel, J.; Graczyk, S. Algorithm for detection of uterine contractions from electrohysterogram. In Proceedings of the 23rd Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Istanbul, Turkey, 25–28 October 2001. [Google Scholar] [CrossRef]
- Jezewski, J.; Horoba, K.; Matonia, A.; Wrobel, J. Quantitative analysis of contraction patterns in electrical activity signal of pregnant uterus as an alternative to mechanical approach. Physiol. Meas. 2005, 26, 753–767. [Google Scholar] [CrossRef]
- Chendeb, M. Détection et Classification des Signaux non Stationnaires par Utilisation des Ondelettes. Application aux Signaux Électromyographiques Utérins. Ph.D. Thesis, Université de Technologie de Troyes, Troyes, France, 2006. [Google Scholar]
- Muszynski, C.; Happillon, T.; Azudin, K.; Tylcz, J.B.; Istrate, D.; Marque, C. Automated electrohysterographic detection of uterine contractions for monitoring of pregnancy: Feasibility and prospects. BMC Pregnancy Childbirth 2018, 18, 1–8. [Google Scholar] [CrossRef]
- Rooijakkers, M.J.; Song, S.; Rabotti, C.; Oei, S.G.; Bergmans, J.W.; Cantatore, E.; Mischi, M. Influence of electrode placement on signal quality for ambulatory pregnancy monitoring. Comput. Math. Methods Med. 2014, 2014. [Google Scholar] [CrossRef]
- Furdea, A.; Eswaran, H.; Wilson, J.D.; Preissl, H.; Lowery, C.L.; Govindan, R.B. Magnetomyographic recording and identification of uterine contractions using Hilbert-wavelet transforms. Physiol. Meas. 2009, 30, 1051–1060. [Google Scholar] [CrossRef]
- Peng, J.; Hao, D.; Liu, H.; Liu, J.; Zhou, X.; Zheng, D. Preliminary Study on the Efficient Electrohysterogram Segments for Recognizing Uterine Contractions with Convolutional Neural Networks. Biomed Res. Int. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Hao, D.; Zhang, L.; Liu, J.; Zhou, X.; Yang, L.; Yang, Y.; Li, X.; Zhang, S.; Zheng, D. Comparison of electrohysterogram characteristics during uterine contraction and non-contraction during labor. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju Island, Korea, 11–15 July 2017; pp. 2924–2927. [Google Scholar] [CrossRef]
- Esgalhado, F.; Batista, A.G.; Mouriño, H.; Russo, S.; Dos Reis, C.R.; Serrano, F.; Vassilenko, V.; Ortigueira, M. Uterine contractions clustering based on electrohysterography. Comput. Biol. Med. 2020, 123, 103897. [Google Scholar] [CrossRef] [PubMed]
- Alexandersson, A.; Steingrimsdottir, T.; Terrien, J.; Marque, C.; Karlsson, B. The Icelandic 16-electrode electrohysterogram database. Sci. Data 2015, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sousa, C. Electrohysterogram Signal Component Cataloging with Spectral and Time-Frequency Methods. Master’s Thesis, Universidade Nova de Lisboa, Lisbon, Portugal, 2015. [Google Scholar]
- Esgalhado, F. Uterine Contractions Clustering Based on Surface Electromyography: An Input for Pregnancy Monitoring. Master’s Thesis, Lisbon University, Lisbon, Portugal, 2018. [Google Scholar]
- Batista, A.G.; Najdi, S.; Godinho, D.M.; Martins, C.; Serrano, F.C.; Ortigueira, M.D.; Rato, R.T. A multichannel time–frequency and multi-wavelet toolbox for uterine electromyography processing and visualisation. Comput. Biol. Med. 2016, 76, 178–191. [Google Scholar] [CrossRef]
- Rooijakkers, M.J.; Rabotti, C.; Oei, S.G.; Aarts, R.M.; Mischi, M. Low-complexity intrauterine pressure monitoring by Teager energy estimation. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 7424–7427. [Google Scholar] [CrossRef]
- Rabotti, C.; Mischi, M.; van Laar, J.O.E.H.; Oei, G.S.; Bergmans, J.W.M. Estimation of internal uterine pressure by joint amplitude and frequency analysis of electrohysterographic signals. Physiol. Meas. 2008, 29, 829–841. [Google Scholar] [CrossRef]
- Kaiser, J.F. On a Simple Algorithm to Calculate the ‘energy’ of a Signal. In Proceedings of the International Conference on Acoustics, Speech, and Signal Processing, Albuquerque, NM, USA, 3–6 April 1990. [Google Scholar]
- Chen, L.; Hao, Y. Feature Extraction and Classification of EHG between Pregnancy and Labour Group Using Hilbert-Huang Transform and Extreme Learning Machine. Comput. Math. Methods Med. 2017, 2017. [Google Scholar] [CrossRef]
- Jager, F.; Libenšek, S.; Geršak, K. Characterization and Automatic Classification of Preterm and Term Uterine records. PLoS ONE 2018, 13, e0202125. [Google Scholar] [CrossRef]
- Hassan, M. Analysis of the Propagation of Uterine Electrical Activity Applied To Predict Preterm Labor. Ph.D. Thesis, Université de Technologie de Compiègne, Compiègne, France, 2015. [Google Scholar]


| Detection Method in the Respective EHG Channel | |
|---|---|
| Wavelet_cont | Number of contractions detected with wavelet energy method |
| Teager_cont | Number of contractions detected with Teager method |
| RMS_cont | Number of contractions detected with RMS method |
| RMS_Squared_cont | Number of contractions detected with squared RMS method |
| Hilbert_Envelope_cont | Number of contractions detected with Hilbert envelope method |
| Classification (%) | Definition | |
|---|---|---|
| Contraction Accuracy | 0 | Contraction was detected |
| 50 | Contraction was partially detected | |
| 100 | Contraction was not detected | |
| Delineation Accuracy | 100 | Contraction delineation is correct |
| 50 | Contraction delineation is partially correct |
| Detection Method | Contraction Accuracy (%) | Delineation Accuracy (%) | False Negative Rate (%) |
|---|---|---|---|
| Wavelet Energy | 92.28 ± 6.66 | 79.19 ± 13.60 | 1.93 |
| Teager Energy | 65.57 ± 25.05 | 71.12 ± 10.61 | 4.74 |
| RMS | 93.64 ± 12.08 | 83.99 ± 12.67 | 1.51 |
| Squared RMS | 97.15 ± 4.66 | 89.43 ± 8.10 | 0.63 |
| Hilbert Envelope | 73.00 ± 19.85 | 71.99 ± 13.59 | 4.41 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esgalhado, F.; Batista, A.G.; Mouriño, H.; Russo, S.; dos Reis, C.R.P.; Serrano, F.; Vassilenko, V.; Duarte Ortigueira, M. Automatic Contraction Detection Using Uterine Electromyography. Appl. Sci. 2020, 10, 7014. https://doi.org/10.3390/app10207014
Esgalhado F, Batista AG, Mouriño H, Russo S, dos Reis CRP, Serrano F, Vassilenko V, Duarte Ortigueira M. Automatic Contraction Detection Using Uterine Electromyography. Applied Sciences. 2020; 10(20):7014. https://doi.org/10.3390/app10207014
Chicago/Turabian StyleEsgalhado, Filipa, Arnaldo G. Batista, Helena Mouriño, Sara Russo, Catarina R. Palma dos Reis, Fátima Serrano, Valentina Vassilenko, and Manuel Duarte Ortigueira. 2020. "Automatic Contraction Detection Using Uterine Electromyography" Applied Sciences 10, no. 20: 7014. https://doi.org/10.3390/app10207014
APA StyleEsgalhado, F., Batista, A. G., Mouriño, H., Russo, S., dos Reis, C. R. P., Serrano, F., Vassilenko, V., & Duarte Ortigueira, M. (2020). Automatic Contraction Detection Using Uterine Electromyography. Applied Sciences, 10(20), 7014. https://doi.org/10.3390/app10207014