Pomegranate: Nutraceutical with Promising Benefits on Human Health
Abstract
1. Introduction
2. Pomegranate Health-Promoting Properties
2.1. Antioxidant Activity
2.2. Anticancer Properties
2.3. Anti-Inflammatory Properties
2.4. Antidiabetic Activity
2.5. Antimicrobial Activities
2.6. Prevention of Cardiovascular Diseases
2.7. Antiviral Activities
2.8. Other Properties
2.8.1. Obesity
2.8.2. Intestinal Regulation
2.8.3. Effects on the Male Reproductive System
2.8.4. Antidiarrheal Effects
2.8.5. Effects on Oral Health
2.8.6. Effects on Pregnant and Breast-Feeding Women
2.8.7. Effects on Nervous System
3. Toxicological Aspects of Pomegranate and Potential Interaction with Drugs
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bellini, E.; Giordani, E.; La Malfa, S. Minor fruit tree species in Italy, a traditional resource for in the innavation of fruitculture: Persimmon and pomegranate as study cases. Italus Hortus 2010, 17, 75–90. [Google Scholar]
- Sepulveda, E.L.G.; Sàenz, C.; Tapia, M. Minimal processing of pomegranate var. wonderful. In Proceedings of the Production, Processing and Marketing of Pomegranate in the Mediterranean Region: Advances in Research and Technology, Universidad Miguel Hernández (EPSO-UMH), Orihuela, Spain, 15–17 October 1998; pp. 237–242. [Google Scholar]
- Da Silva, J.A.T.; Rana, T.S.; Narzary, D.; Verma, N.; Meshram, D.T.; Ranade, S.A. Pomegranate biology and biotechnology: A review. Sci. Hortic. 2013, 160, 85–107. [Google Scholar] [CrossRef]
- Heber, D.; Schulman, R.N.; Seeram, N.P. Pomegranates: Ancient Roots to Modern Medicine; CRC Press: Boca Raton, FL, USA, 2006; p. 262. [Google Scholar]
- Viuda-Martos, M.; Fernandez-Lopez, J.; Perez-Alvarez, J.A. Pomegranate and its Many Functional Components as Related to Human Health: A Review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 635–654. [Google Scholar] [CrossRef]
- Andreu-Sevilla, A.J.; Signes-Pastor, A.J.; Carbonell-Barrachina, A.A. La Granada Y Su Zumo. Producción, Composición Y Propiedades Beneficiosas Para La Salud. Inf. Univ. Miguel Hernández Elchedigital 2008, 234, 36–39. [Google Scholar]
- Jurenka, J.S. Therapeutic applications of pomegranate (Punica granatum L.): A review. Altern. Med. Rev. 2008, 13, 128–144. [Google Scholar]
- Ismail, T.; Sestili, P.; Akhtar, S. Pomegranate peel and fruit extracts: A review of potential anti-inflammatory and anti-infective effects. J. Ethnopharmacol. 2012, 143, 397–405. [Google Scholar] [CrossRef]
- Cano, A.; Arnao, M.B. Hydrophilic and lipophilic antioxidant activity in different leaves of three lettuce varieties. Int. J. Food Prop. 2005, 8, 521–528. [Google Scholar] [CrossRef]
- Plastina, P.; Apriantini, A.; Meijerink, J.; Witkamp, R.; Gabriele, B.; Fazio, A. In Vitro Anti-Inflammatory and Radical Scavenging Properties of Chinotto (Citrus myrtifolia Raf.) Essential Oils. Nutrients 2018, 10, 783. [Google Scholar] [CrossRef]
- Chen, M.L.; Vigneault, C.; Raghavan, G.S.V.; Kubow, S. Importance of phytochemical content of fruits and vegetables to human health. Stewart Postharvest Rev. 2007, 3, 20–32. [Google Scholar]
- Badria, F.A.; Zidan, O.A. Natural products for dental caries prevention. J. Med. Food 2004, 7, 381–384. [Google Scholar] [CrossRef]
- Parisi, O.I.; Casaburi, I.; Sinicropi, M.S.; Avena, P.; Caruso, A.; Givigliano, F.; Pezzi, V.; Puoci, F. Most Relevant Polyphenols Present in the Mediterranean Diet and Their Incidence in Cancer Diseases. In Polyphenols in Human Health and Disease; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 2, pp. 1341–1351. [Google Scholar]
- Plastina, P.; Fazio, A.; Gabriele, B. Comparison of fatty acid profile and antioxidant potential of extracts of seven Citrus rootstock seeds. Nat. Prod. Res. 2012, 26, 2182–2187. [Google Scholar] [CrossRef] [PubMed]
- Leporini, M.; Loizzo, M.R.; Tundis, R.; La Torre, C.; Fazio, A.; Plastina, P. Non-Pungent n-3 Polyunsaturated Fatty Acid (PUFA)-Derived Capsaicin Analogues as Potential Functional Ingredients with Antioxidant and Carbohydrate-Hydrolysing Enzyme Inhibitory Activities. Antioxidants 2019, 8, 162. [Google Scholar] [CrossRef] [PubMed]
- Benincasa, C.; La Torre, C.; Plastina, P.; Fazio, A.; Perri, E.; Caroleo, M.C.; Gallelli, L.; Cannataro, R.; Cione, E. Hydroxytyrosyl Oleate: Improved Extraction Procedure from Olive Oil and By-Products, and In Vitro Antioxidant and Skin Regenerative Properties. Antioxidants 2019, 8, 233. [Google Scholar] [CrossRef] [PubMed]
- Gabriele, B.; Fazio, A.; Carchedi, M.; Plastina, P. In vitro antioxidant activity of extracts of Sybaris liquorice roots from Southern Italy. Nat. Prod. Res. 2012, 26, 2176–2181. [Google Scholar] [CrossRef]
- Cannataro, R.; Caroleo, M.C.; Fazio, A.; La Torre, C.; Plastina, P.; Gallelli, L.; Lauria, G.; Cione, E. Ketogenic Diet and microRNAs Linked to Antioxidant Biochemical Homeostasis. Antioxidants 2019, 8, 269. [Google Scholar] [CrossRef]
- Espin, J.C.; Garcia-Conesa, M.T.; Tomas-Barberan, F.A. Nutraceuticals: Facts and fiction. Phytochemistry 2007, 68, 2986–3008. [Google Scholar] [CrossRef]
- Hounsome, N.; Hounsome, B.; Tomos, D.; Edwards-Jones, G. Plant metabolites and nutritional quality of vegetables. J. Food Sci. 2008, 73, R48–R65. [Google Scholar] [CrossRef]
- Dorais, M.; Ehret, D.L.; Papadopoulos, A.P. Tomato (Solanum lycopersicum) health components: From the seed to the consumer. Phytochem. Rev. 2008, 7, 231–250. [Google Scholar] [CrossRef]
- Amarowicz, R.; Pegg, R.B.; Rahimi-Moghaddam, P.; Barl, B.; Weil, J.A. Free-radical scavenging capacity and antioxidant activity of selected plant species from the Canadian prairies. Food Chem. 2004, 84, 551–562. [Google Scholar] [CrossRef]
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef]
- Basiri, S. Evaluation of antioxidant and antiradical properties of Pomegranate (Punica granatum L.) seed and defatted seed extracts. J. Food Sci. Technol. 2015, 52, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Derakhshan, Z.; Ferrante, M.; Tadi, M.; Ansari, F.; Heydari, A.; Hosseini, M.S.; Conti, G.O.; Sadrabad, E.K. Antioxidant activity and total phenolic content of ethanolic extract of pomegranate peels, juice and seeds. Food Chem. Toxicol. 2018, 114, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Pitonzo, R.; Novara, M.E.; Bongiorno, D.; Indelicato, S.; Gentile, C.; Avellone, G.; Bognanni, R.; Scandurra, S.; Melilli, M.G. Antioxidant activity and phenolic composition in pomegranate (Punica granatum L.) genotypes from south Italy by UHPLC-Orbitrap-MS approach. J. Sci. Food Agric. 2019, 99, 1038–1045. [Google Scholar] [CrossRef]
- Mastrogiovanni, F.; Bernini, R.; Basirico, L.; Bernabucci, U.; Campo, M.; Romani, A.; Santi, L.; Lacetera, N. Antioxidant and anti-inflammatory effects of pomegranate peel extracts on bovine mammary epithelial cells BME-UV1. Nat. Prod. Res. 2020, 34, 1465–1469. [Google Scholar] [CrossRef] [PubMed]
- Nur Hanani, Z.A.; Cheng Yee, F.; Nor-Khaizura, M.A.R. Effect of pomegranate (Punica granatum L.) peel powder on the antioxidant and antimicrobial properties of fish gelatin films as active packaging. Food Hydrocoll. 2019, 89, 253–259. [Google Scholar] [CrossRef]
- Bertolo, M.R.V.; Martins, V.C.A.; Horn, M.M.; Brenelli, L.B.; Plepis, A.M.G. Rheological and antioxidant properties of chitosan/gelatin-based materials functionalized by pomegranate peel extract. Carbohydr. Polym. 2020, 228, 115386. [Google Scholar] [CrossRef]
- Jalal, H.; Pal, M.A.; Ahmad, S.R.; Rather, M.; Andrabi, M.; Hamdani, S. Physico-chemical and functional properties of pomegranate peel and seed powder. Pharm. Innov. J. 2018, 7, 1127–1131. [Google Scholar]
- Surek, E.; Nilufer-Erdil, D. Phenolic contents, antioxidant activities and potential bioaccessibilities of industrial pomegranate nectar processing wastes. Int. J. Food Sci. Technol. 2016, 51, 231–239. [Google Scholar] [CrossRef]
- Pinilla, M.; Iglesias-Moya, J.; Jesús Campos, M.; Corpas, F.J.; Palma, J.M. Pomegranate (Punica granatum L.) Fruits: Characterization of the Main Enzymatic Antioxidants (Peroxisomal Catalase and SOD Isozymes) and the NADPH-Regenerating System. Agronomy 2019, 9, 338. [Google Scholar] [CrossRef]
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef]
- Saturnino, C.; Caruso, A.; Iacopetta, D.; Rosano, C.; Ceramella, J.; Muia, N.; Mariconda, A.; Bonomo, M.G.; Ponassi, M.; Rosace, G.; et al. Inhibition of Human Topoisomerase II by N,N,N-Trimethylethanammonium Iodide Alkylcarbazole Derivatives. Chem. Med. Chem. 2018, 13, 2635–2643. [Google Scholar] [CrossRef] [PubMed]
- Saturnino, C.; Caruso, A.; Longo, P.; Capasso, A.; Pingitore, A.; Caroleo, M.C.; Cione, E.; Perri, M.; Nicolo, F.; Nardo, V.M.; et al. Crystallographic study and biological evaluation of 1,4-dimethyl-N-alkylcarbazoles. Curr. Top. Med. Chem. 2015, 15, 973–979. [Google Scholar] [CrossRef]
- Chimento, A.; Sala, M.; Gomez-Monterrey, I.M.; Musella, S.; Bertamino, A.; Caruso, A.; Sinicropi, M.S.; Sirianni, R.; Puoci, F.; Parisi, O.I.; et al. Biological activity of 3-chloro-azetidin-2-one derivatives having interesting antiproliferative activity on human breast cancer cell lines. Bioorganic Med. Chem. Lett. 2013, 23, 6401–6405. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, S.; Hiraku, Y.; Pinlaor, S.; Ma, N. Oxidative and nitrative DNA damage in animals and patients with inflammatory diseases in relation to inflammation-related carcinogenesis. Biol. Chem. 2006, 387, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K.; Takenaga, K.; Akimoto, M.; Koshikawa, N.; Yamaguchi, A.; Imanishi, H.; Nakada, K.; Honma, Y.; Hayashi, J. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 2008, 320, 661–664. [Google Scholar] [CrossRef]
- Caruso, A.; Iacopetta, D.; Puoci, F.; Cappello, A.R.; Saturnino, C.; Sinicropi, M.S. Carbazole derivatives: A promising scenario for breast cancer treatment. Mini Rev. Med. Chem. 2016, 16, 630–643. [Google Scholar] [CrossRef]
- Ceramella, J.; Caruso, A.; Occhiuzzi, M.A.; Iacopetta, D.; Barbarossa, A.; Rizzuti, B.; Dallemagne, P.; Rault, S.; El-Kashef, H.; Saturnino, C.; et al. Benzothienoquinazolinones as new multi-target scaffolds: Dual inhibition of human Topoisomerase I and tubulin polymerization. Eur. J. Med. Chem. 2019, 181, 111583. [Google Scholar] [CrossRef]
- Sinicropi, M.S.; Iacopetta, D.; Rosano, C.; Randino, R.; Caruso, A.; Saturnino, C.; Muia, N.; Ceramella, J.; Puoci, F.; Rodriquez, M.; et al. N-thioalkylcarbazoles derivatives as new anti-proliferative agents: Synthesis, characterisation and molecular mechanism evaluation. J. Enzyme Inhib. Med. Chem. 2018, 33, 434–444. [Google Scholar] [CrossRef]
- Faienza, M.F.; Corbo, F.; Carocci, A.; Catalano, A.; Clodoveo, M.L.; Grano, M.; Wang, D.Q.-H.; D’Amato, G.; Muraglia, M.; Franchini, C.; et al. Novel insights in health-promoting properties of sweet cherries. J. Funct. Foods 2020, 69, 103945. [Google Scholar] [CrossRef]
- Karaaslan, M.; Vardin, H.; Varliklioz, S.; Yilmaz, F.M. Antiproliferative and antioxidant activities of Turkish pomegranate (Punica granatum L.) accessions. Int. J. Food Sci. Technol. 2014, 49, 82–90. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Vadhanam, M.V.; Kausar, H.; Jeyabalan, J.; Schultz, D.J.; Gupta, R.C. Anti-proliferative activity and protection against oxidative DNA damage by punicalagin isolated from pomegranate husk. Food Res. Int. 2012, 49, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Dikmen, M.; Ozturk, N.; Ozturk, Y. The antioxidant potency of Punica granatum L. Fruit peel reduces cell proliferation and induces apoptosis on breast cancer. J. Med. Food 2011, 14, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.P.; Jiang, W.; Mo, H.; Bravo, L.; Froom, P.; Yu, W.; Harris, N.M.; Neeman, I.; Campbell, M.J. Possible synergistic prostate cancer suppression by anatomically discrete pomegranate fractions. Investig. New Drugs 2005, 23, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Fazio, A.; Iacopetta, D.; La Torre, C.; Ceramella, J.; Muia, N.; Catalano, A.; Carocci, A.; Sinicropi, M.S. Finding solutions for agricultural wastes: Antioxidant and antitumor properties of pomegranate Akko peel extracts and beta-glucan recovery. Food Funct. 2018, 9, 6618–6631. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Seeram, N.P.; Heber, D. Pomegranate polyphenols down-regulate expression of androgen-synthesizing genes in human prostate cancer cells overexpressing the androgen receptor. J. Nutr. Biochem. 2008, 19, 848–855. [Google Scholar] [CrossRef]
- Di Carlo, G.; Mascolo, N.; Izzo, A.A.; Capasso, F. Flavonoids: Old and new aspects of a class of natural therapeutic drugs. Life Sci. 1999, 65, 337–353. [Google Scholar] [CrossRef]
- Noda, Y.; Kaneyuki, T.; Mori, A.; Packer, L. Antioxidant activities of pomegranate fruit extract and its anthocyanidins: Delphinidin, cyanidin, and pelargonidin. J. Agric. Food Chem. 2002, 50, 166–171. [Google Scholar] [CrossRef]
- Iacopetta, D.; Grande, F.; Caruso, A.; Mordocco, R.A.; Plutino, M.R.; Scrivano, L.; Ceramella, J.; Muia, N.; Saturnino, C.; Puoci, F.; et al. New insights for the use of quercetin analogs in cancer treatment. Future Med. Chem. 2017, 9, 2011–2028. [Google Scholar] [CrossRef]
- Levin, G.M. Pomegranate Roads: A Soviet Botanist’s Exile from Eden; Floreant Press: Forestville, CA, USA, 2006. [Google Scholar]
- Grande, F.; Parisi, O.I.; Mordocco, R.A.; Rocca, C.; Puoci, F.; Scrivano, L.; Quintieri, A.M.; Cantafio, P.; Ferla, S.; Brancale, A.; et al. Quercetin derivatives as novel antihypertensive agents: Synthesis and physiological characterization. Eur. J. Pharm. Sci. 2016, 82, 161–170. [Google Scholar] [CrossRef]
- Van Elswijk, D.A.; Schobel, U.P.; Lansky, E.P.; Irth, H.; van der Greef, J. Rapid dereplication of estrogenic compounds in pomegranate (Punica granatum) using on-line biochemical detection coupled to mass spectrometry. Phytochemistry 2004, 65, 233–241. [Google Scholar] [CrossRef]
- Khatib, M. Bioactive Compounds into Edible Syrian Plants: Pomegranate and Capper; University of Florence: Florence, Italy, 2015. [Google Scholar]
- Gonzalez-Sarrias, A.; Espin, J.C.; Tomas-Barberan, F.A.; Garcia-Conesa, M.T. Gene expression, cell cycle arrest and MAPK signalling regulation in Caco-2 cells exposed to ellagic acid and its metabolites, urolithins. Mol. Nutr. Food Res. 2009, 53, 686–698. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.S.; Seeram, N.P.; Aggarwal, B.B.; Takada, Y.; Sand, D.; Heber, D. Pomegranate juice, total pomegranate ellagitannins, and punicalagin suppress inflammatory cell signaling in colon cancer cells. J. Agric. Food Chem. 2006, 54, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Pantuck, A.J.; Leppert, J.T.; Zomorodian, N.; Aronson, W.; Hong, J.; Barnard, R.J.; Seeram, N.; Liker, H.; Wang, H.; Elashoff, R.; et al. Phase II study of pomegranate juice for men with rising prostate-specific antigen following surgery or radiation for prostate cancer. Clin. Cancer Res. 2006, 12, 4018–4026. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S.; Cobb, L.J.; Mehta, H.H.; Seeram, N.P.; Heber, D.; Pantuck, A.J.; Cohen, P. Pomegranate extract induces apoptosis in human prostate cancer cells by modulation of the IGF-IGFBP axis. Growth Horm. IGF Res. 2010, 20, 55–62. [Google Scholar] [CrossRef]
- Khan, G.N.; Gorin, M.A.; Rosenthal, D.; Pan, Q.; Bao, L.W.; Wu, Z.F.; Newman, R.A.; Pawlus, A.D.; Yang, P.; Lansky, E.P.; et al. Pomegranate fruit extract impairs invasion and motility in human breast cancer. Integr. Cancer Ther. 2009, 8, 242–253. [Google Scholar] [CrossRef]
- Aiyer, H.S.; Bouker, K.B.; Cook, K.L.; Facey, C.O.; Hu, R.; Schwartz, J.L.; Shajahan, A.N.; Hilakivi-Clarke, L.; Clarke, R. Interaction of dietary polyphenols with molecular signaling pathways of antiestrogen resistance: Possible role in breast cancer recurrence. Horm. Mol. Biol. Clin. Investig. 2012, 9, 127–141. [Google Scholar] [CrossRef]
- Peng, S.Y.; Hsiao, C.C.; Lan, T.H.; Yen, C.Y.; Farooqi, A.A.; Cheng, C.M.; Tang, J.Y.; Yu, T.J.; Yeh, Y.C.; Chuang, Y.T.; et al. Pomegranate extract inhibits migration and invasion of oral cancer cells by downregulating matrix metalloproteinase-2/9 and epithelial-mesenchymal transition. Environ. Toxicol. 2020, 35, 673–682. [Google Scholar] [CrossRef]
- Kasai, K.; Yoshimura, M.; Koga, T.; Arii, M.; Kawasaki, S. Effects of oral administration of ellagic acid-rich pomegranate extract on ultraviolet-induced pigmentation in the human skin. J. Nutr. Sci. Vitaminol. 2006, 52, 383–388. [Google Scholar] [CrossRef]
- Manasathien, J.; Kupittayanant, S.; Indrapichate, K. Protective Efficacy of Pomegranate (Punica Granatum Linn., Punicaceae) Peel Ethanolic Extract on UVB-Irradiated Rat Skin. Am. Eurasian J. Toxicol. Sci. 2011, 3, 250–258. [Google Scholar]
- Dai, Z.; Nair, V.; Khan, M.; Ciolino, H.P. Pomegranate extract inhibits the proliferation and viability of MMTV-Wnt-1 mouse mammary cancer stem cells in vitro. Oncol. Rep. 2010, 24, 1087–1091. [Google Scholar]
- Nair, V.; Dai, Z.; Khan, M.; Ciolino, H.P. Pomegranate extract induces cell cycle arrest and alters cellular phenotype of human pancreatic cancer cells. Anticancer. Res. 2011, 31, 2699–2704. [Google Scholar]
- Motaal, A.A.; Shaker, S. Anticancer and Antioxidant Activities of Standardized Whole Fruit, Pulp and Peel Extracts of Egyptian Pomegranate. Open Conf. Proc. J. 2011, 2, 41–45. [Google Scholar] [CrossRef]
- El-Awady, M.A.; Awad, N.S.; El-Tarras, A.E. Evaluation of the anticancer activities of pomegranate (Punica granatum) and harmal (Rhazya stricta) plants grown in Saudi Arabia. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 1158–1167. [Google Scholar]
- Modaeinama, S.; Abasi, M.; Abbasi, M.M.; Jahanban-Esfahlan, R. Anti-Tumoral Properties of Punica Granatum (Pomegranate) Peel Extract on Different Human Cancer Cells. Asian Pac. J. Cancer Prev. 2015, 16, 5697–5701. [Google Scholar] [CrossRef] [PubMed]
- Asmaa, M.J.; Ali, A.J.; Farid, J.M.; Azman, S. Growth inhibitory effects of crude pomegranate peel extract on chronic myeloid leukemia, K562 cells. Int. J. Appl. Basic Med. Res. 2015, 5, 100–105. [Google Scholar] [PubMed]
- Rostami, Z.; Khorashadizadeh, M.; Ghoncheh, M.; Naseri, M. Effect of Pomegranate Extract in Mesenchymal Stem Cells by Modulation of MicroRNA-155, MicroRNA-21, MicroRNA-23b, MicroRNA-126a, and PI3K\AKT1\NF-B Expression. DNA Cell Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Saturnino, C.; Barone, I.; Iacopetta, D.; Mariconda, A.; Sinicropi, M.S.; Rosano, C.; Campana, A.; Catalano, S.; Longo, P.; Ando, S. N-heterocyclic carbene complexes of silver and gold as novel tools against breast cancer progression. Futur. Med. Chem. 2016, 8, 2213–2229. [Google Scholar] [CrossRef]
- Chimento, A.; De Amicis, F.; Sirianni, R.; Sinicropi, M.S.; Puoci, F.; Casaburi, I.; Saturnino, C.; Pezzi, V. Progress to Improve Oral Bioavailability and Beneficial Effects of Resveratrol. Int. J. Mol. Sci. 2019, 20, 1381. [Google Scholar] [CrossRef]
- Parisi, O.I.; Morelli, C.; Puoci, F.; Saturnino, C.; Caruso, A.; Sisci, D.; Trombino, G.E.; Picci, N.; Sinicropi, M.S. Magnetic molecularly imprinted polymers (MMIPs) for carbazole derivative release in targeted cancer therapy. J. Mater. Chem. B 2014, 2, 6619–6625. [Google Scholar] [CrossRef]
- Sarkar, S.; Kotteeswaran, V. Green synthesis of silver nanoparticles from aqueous leaf extract of Pomegranate (Punica granatum) and their anticancer activity on human cervical cancer cells. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 025014. [Google Scholar] [CrossRef]
- Yusefi, M.; Shameli, K.; Ali, R.R.; Pang, S.W.; Teow, S.Y. Evaluating Anticancer Activity of Plant-Mediated Synthesized Iron Oxide Nanoparticles Using Punica Granatum Fruit Peel Extract. J. Mol. Struct. 2020, 1204, 127539. [Google Scholar] [CrossRef]
- Sinicropi, M.S.; Caruso, A.; Conforti, F.; Marrelli, M.; El Kashef, H.; Lancelot, J.C.; Rault, S.; Statti, G.A.; Menichini, F. Synthesis, inhibition of NO production and antiproliferative activities of some indole derivatives. J. Enzyme Inhib. Med. Chem. 2009, 24, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Saturnino, C.; Popolo, A.; Ramunno, A.; Adesso, S.; Pecoraro, M.; Plutino, M.R.; Rizzato, S.; Albinati, A.; Marzocco, S.; Sala, M.; et al. Anti-Inflammatory, Antioxidant and Crystallographic Studies of N-Palmitoyl-ethanol Amine (PEA) Derivatives. Molecules 2017, 22, 616. [Google Scholar] [CrossRef] [PubMed]
- Plastina, P.; Benincasa, C.; Perri, E.; Fazio, A.; Augimeri, G.; Poland, M.; Witkamp, R.; Meijerink, J. Identification of hydroxytyrosyl oleate, a derivative of hydroxytyrosol with anti-inflammatory properties, in olive oil by-products. Food Chem. 2019, 279, 105–113. [Google Scholar] [CrossRef]
- Lee, C.J.; Chen, L.G.; Liang, W.L.; Wang, C.C. Anti-inflammatory effects of Punica granatum Linne in vitro and in vivo. Food Chem. 2010, 118, 315–322. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A. Granada y salud: Aspectos farmacologicos y terapéuticos de la Granada. In El Granado—I Jornadas Nacionales Sobre el Granado: Produccion, Economia, Indrustrializacion, Alimentacion y Salud; Moreno, P.M., Garcia, F.H., Murcia, P.L., Eds.; Documentos Poscosecha: Valencia, Spain, 2010; pp. 7–22. [Google Scholar]
- Negro, C.; Longo, L.; Vasapollo, G.; De Bellis, L.; Miceli, A. Biochemical, Antioxidant and Anti-Inflammatory Properties of Pomegranate Fruits Growing in Southern Italy (Salento, Apulia). Acta Aliment. 2012, 41, 190–199. [Google Scholar] [CrossRef]
- Boussetta, T.; Raad, H.; Letteron, P.; Gougerot-Pocidalo, M.A.; Marie, J.C.; Driss, F.; El-Benna, J. Punicic acid a conjugated linolenic acid inhibits TNFalpha-induced neutrophil hyperactivation and protects from experimental colon inflammation in rats. PLoS ONE 2009, 4, e6458. [Google Scholar] [CrossRef]
- Jung, K.H.; Kim, M.J.; Ha, E.; Uhm, Y.K.; Kim, H.K.; Chung, J.H.; Yim, S.V. Suppressive effect of Punica granatum on the production of tumor necrosis factor (Tnf) in BV2 microglial cells. Biol. Pharm. Bull. 2006, 29, 1258–1261. [Google Scholar] [CrossRef]
- De Nigris, F.; Balestrieri, M.L.; Williams-Ignarro, S.; D’Armiento, F.P.; Fiorito, C.; Ignarro, L.J.; Napoli, C. The influence of pomegranate fruit extract in comparison to regular pomegranate juice and seed oil on nitric oxide and arterial function in obese Zucker rats. Nitric Oxide Biol. Chem. 2007, 17, 50–54. [Google Scholar] [CrossRef]
- Larrosa, M.; Gonzalez-Sarrias, A.; Yanez-Gascon, M.J.; Selma, M.V.; Azorin-Ortuno, M.; Toti, S.; Tomas-Barberan, F.; Dolara, P.; Espin, J.C. Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolic metabolism. J. Nutr. Biochem. 2010, 21, 717–725. [Google Scholar] [CrossRef]
- Ouachrif, A.; Khalki, H.; Chaib, S.; Mountassir, M.; Aboufatima, R.; Farouk, L.; Benharraf, A.; Chait, A. Comparative study of the anti-inflammatory and antinociceptive effects of two varieties of Punica granatum. Pharm. Biol. 2012, 50, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Seok, J.K.; Kwak, J.Y.; Suh, H.J.; Kim, Y.M.; Boo, Y.C. Anti-Inflammatory Effects of Pomegranate Peel Extract in THP-1 Cells Exposed to Particulate Matter PM10. Evid. Based Complement. Altern. Med. 2016, 2016, 1–11. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, Y.; Aisa, H.A. Anti-inflammatory effect of pomegranate flower in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages. Pharm. Biol. 2017, 55, 2095–2101. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Banerjee, N.; Ivanov, I.; Pfent, C.M.; Prudhomme, K.R.; Bisson, W.H.; Dashwood, R.H.; Talcott, S.T.; Mertens-Talcott, S.U. Comparison of anti-inflammatory mechanisms of mango (Mangifera indica L.) and pomegranate (Punica granatum L.) in a preclinical model of colitis. Mol. Nutr. Food Res. 2016, 60, 1912–1923. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Trujano, M.E.; Pellicer, F.; Mena, P.; Moreno, D.A.; Garcia-Viguera, C. Antinociceptive and anti-inflammatory activities of a pomegranate (Punica granatum L.) extract rich in ellagitannins. Int. J. Food Sci. Nutr. 2015, 66, 395–399. [Google Scholar] [CrossRef]
- Mo, J.; Panichayupakaranant, P.; Kaewnopparat, N.; Songkro, S.; Reanmongkol, W. Topical anti-inflammatory potential of standardized pomegranate rind extract and ellagic acid in contact dermatitis. Phytother. Res. 2014, 28, 629–632. [Google Scholar] [CrossRef]
- Ruffo, M.; Parisi, O.I.; Scrivano, L.; Restuccia, D.; Amone, F.; Sinicropi, M.S.; Malivindi, R.; Aiello, G.; Puoci, F. Role of Calabrian Black Rice in Metabolic Syndrome: In vitro Evaluation of Oryza sativa L. Indica Biological Properties. Curr. Nutr. Food Sci. 2018, 14, 121–127. [Google Scholar] [CrossRef]
- Cheng, J.T.; Liu, I.M. Stimulatory effect of caffeic acid on alpha1A-adrenoceptors to increase glucose uptake into cultured C2C12 cells. Naunyn Schmiedebergs Arch. Pharmacol. 2000, 362, 122–127. [Google Scholar] [CrossRef]
- Li, Y.; Wen, S.; Kota, B.P.; Peng, G.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D. Punica granatum flower extract, a potent alpha-glucosidase inhibitor, improves postprandial hyperglycemia in Zucker diabetic fatty rats. J. Ethnopharmacol. 2005, 99, 239–244. [Google Scholar] [CrossRef]
- Huang, T.H.W.; Peng, G.; Kota, B.P.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y.H. Anti-diabetic action of Punica granatum flower extract: Activation of PPAR-gamma and identification of an active component. Toxicol. Appl. Pharmacol. 2005, 207, 160–169. [Google Scholar] [CrossRef]
- McFarlin, B.K.; Strohacker, K.A.; Kueht, M.L. Pomegranate seed oil consumption during a period of high-fat feeding reduces weight gain and reduces type 2 diabetes risk in CD-1 mice. Br. J. Nutr. 2009, 102, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Parmar, H.S.; Kar, A. Medicinal values of fruit peels from Citrus sinensis, Punica granatum, and Musa paradisiaca with respect to alterations in tissue lipid peroxidation and serum concentration of glucose, insulin, and thyroid hormones. J. Med. Food 2008, 11, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Makino-Wakagi, Y.; Yoshimura, Y.; Uzawa, Y.; Zaima, N.; Moriyama, T.; Kawamura, Y. Ellagic acid in pomegranate suppresses resistin secretion by a novel regulatory mechanism involving the degradation of intracellular resistin protein in adipocytes. Biochem. Biophys. Res. Commun. 2012, 417, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Cambay, Z.; Baydas, G.; Tuzcu, M.; Bal, R. Pomegranate (Punica granatum L.) flower improves learning and memory performances impaired by diabetes mellitus in rats. Acta Physiol. Hung. 2011, 98, 409–420. [Google Scholar] [CrossRef]
- Faghihimani, Z.; Mirmiran, P.; Sohrab, G.; Iraj, B.; Faghihimani, E. Effects of Pomegranate Seed Oil on Metabolic State of Patients with Type 2 Diabetes Mellitus. Int. J. Prev. Med. 2016, 7, 124. [Google Scholar]
- Tang, D.; Liu, L.; Ajiakber, D.; Ye, J.; Xu, J.; Xin, X.; Aisa, H.A. Anti-diabetic Effect of Punica granatum Flower Polyphenols Extract in Type 2 Diabetic Rats: Activation of Akt/GSK-3beta and Inhibition of IRE1alpha-XBP1 Pathways. Front. Endocrinol. 2018, 9, 586. [Google Scholar] [CrossRef]
- Gharib, E.; Montasser Kouhsari, S. Study of the Antidiabetic Activity of Punica granatum L. Fruits Aqueous Extract on the Alloxan-Diabetic Wistar Rats. Iran. J. Pharm. Res. 2019, 18, 358–368. [Google Scholar]
- Mustafa, S.M.; Chua, L.S.; El-Enshasy, H.A.; Abd Majid, F.A.; Hanapia, S.Z. Kinetic profile and anti-diabetic potential of fermented Punica granatum juice using Lactobacillus casei. Process. Biochem. 2020, 92, 224–231. [Google Scholar] [CrossRef]
- Banihani, S.A.; Shuaibu, S.M.; Al-Husein, B.A.; Makahleh, S.S. Fresh Pomegranate Juice Decreases Fasting Serum Erythropoietin in Patients with Type 2 Diabetes. Int. J. Food Sci. 2019, 2019, 1269341. [Google Scholar] [CrossRef]
- Davidson, P.M.; Branen, A.L. Food Antimicrobials—An Introduction. In Antimicrobials in Food; Davidson, P.M., Sofos, J.M., Branen, A.L., Eds.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Fawole, O.A.; Makunga, N.P.; Opara, U.L. Antibacterial, antioxidant and tyrosinase-inhibition activities of pomegranate fruit peel methanolic extract. BMC Complement. Altern. Med. 2012, 12, 200. [Google Scholar] [CrossRef]
- Naz, S.; Siddiqi, R.; Ahmad, S.; Rasool, S.A.; Sayeed, S.A. Antibacterial activity directed isolation of compounds from Punica granatum. J. Food Sci. 2007, 72, M341–M345. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, L.C.; Sampaio, M.C.; Sampaio, F.C.; Higino, J.S. Use of Punica granatum as an antifungal agent against candidosis associated with denture stomatitis. Mycoses 2003, 46, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Al-Zoreky, N.S. Antimicrobial activity of pomegranate (Punica granatum L.) fruit peels. Int. J. Food Microbiol. 2009, 134, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Wafa, B.A.; Makni, M.; Ammar, S.; Khannous, L.; Hassana, A.B.; Bouaziz, M.; Es-Safi, N.E.; Gdoura, R. Antimicrobial effect of the Tunisian Nana variety Punica granatum L. extracts against Salmonella enterica (serovars Kentucky and Enteritidis) isolated from chicken meat and phenolic composition of its peel extract. Int. J. Food Microbiol. 2017, 241, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Visnjevec, A.M.; Ota, A.; Skrt, M.; Butinar, B.; Mozina, S.S.; Cimerman, N.G.; Necemer, M.; Arbeiter, A.B.; Hladnik, M.; Krapac, M.; et al. Genetic, Biochemical, Nutritional and Antimicrobial Characteristics of Pomegranate (Punica granatum L.)Grown in Istria. Food Technol. Biotechnol. 2017, 55, 151–163. [Google Scholar]
- Devanesan, S.; AlSalhi, M.S.; Balaji, R.V.; Ranjitsingh, A.J.A.; Ahamed, A.; Alfuraydi, A.A.; AlQahtani, F.Y.; Aleanizy, F.S.; Othman, A.H. Antimicrobial and Cytotoxicity Effects of Synthesized Silver Nanoparticles from Punica granatum Peel Extract. Nanoscale Res. Lett. 2018, 13, 315. [Google Scholar] [CrossRef]
- Esmaillzadeh, A.; Azadbakht, L. Food intake patterns may explain the high prevalence of cardiovascular risk factors among Iranian women. J. Nutr. 2008, 138, 1469–1475. [Google Scholar] [CrossRef]
- Zirpoli, H.; Caputo, M.; Carraturo, A.; Torino, G.; Fazio, A.; Attya, M.; Rastrelli, L.; Tecce, M.F. Selective action of human sera differing in fatty acids and cholesterol content on in vitro gene expression. J. Cell Biochem. 2012, 113, 815–823. [Google Scholar] [CrossRef]
- Heinecke, J.W. Lipoprotein oxidation in cardiovascular disease: Chief culprit or innocent bystander? J. Exp. Med. 2006, 203, 813–816. [Google Scholar] [CrossRef]
- Khateeb, J.; Gantman, A.; Kreitenberg, A.J.; Aviram, M.; Fuhrman, B. Paraoxonase 1 (PON1) expression in hepatocytes is upregulated by pomegranate polyphenols: A role for PPAR-gamma pathway. Atherosclerosis 2010, 208, 119–125. [Google Scholar] [CrossRef]
- Riaz, A.; Khan, R.A. Anticoagulant, antiplatelet and antianemic effects of Punica granatum (pomegranate) juice in rabbits. Blood Coagul. Fibrinolysis 2016, 27, 287–293. [Google Scholar] [CrossRef]
- Razani, Z.; Dastani, M.; Kazerani, H.R. Cardioprotective Effects of Pomegranate (Punica granatum) Juice in Patients with Ischemic Heart Disease. Phytother. Res. 2017, 31, 1731–1738. [Google Scholar] [CrossRef]
- Patel, I.B.; Atar, M.A.; Ali, S.A. Punica granatum Peel Extract Ameliorates Doxorubicin Induced Cardiotoxicity. Anal. Chem. Lett. 2019, 9, 835–844. [Google Scholar] [CrossRef]
- Boldaji, R.B.; Akhlaghi, M.; Sagheb, M.M.; Esmaeilinezhad, Z. Pomegranate juice improves cardiometabolic risk factors, biomarkers of oxidative stress and inflammation in hemodialysis patients: A randomized crossover trial. J. Sci. Food Agric. 2019. [Google Scholar] [CrossRef] [PubMed]
- Sohrab, G.; Roshan, H.; Ebrahimof, S.; Nikpayam, O.; Sotoudeh, G.; Siasi, F. Effects of pomegranate juice consumption on blood pressure and lipid profile in patients with type 2 diabetes: A single-blind randomized clinical trial. Clin. Nutr. Espen. 2019, 29, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Caruso, A.; Ceramella, J.; Iacopetta, D.; Saturnino, C.; Mauro, M.V.; Bruno, R.; Aquaro, S.; Sinicropi, M.S. Carbazole Derivatives as Antiviral Agents: An Overview. Molecules 2019, 24, 1912. [Google Scholar] [CrossRef]
- Sundararajan, A.; Ganapathy, R.; Huan, L.; Dunlap, J.R.; Webby, R.J.; Kotwal, G.J.; Sangster, M.Y. Influenza virus variation in susceptibility to inactivation by pomegranate polyphenols is determined by envelope glycoproteins. Antivir. Res. 2010, 88, 1–9. [Google Scholar] [CrossRef]
- Haidari, M.; Ali, M.; Ward Casscells, S.; Madjid, M. Pomegranate (Punica granatum) purified polyphenol extract inhibits influenza virus and has a synergistic effect with oseltamivir. Phytomedicine 2009, 16, 1127–1136. [Google Scholar] [CrossRef]
- Yang, Y.; Xiu, J.; Zhang, L.; Qin, C.; Liu, J. Antiviral activity of punicalagin toward human enterovirus 71 in vitro and in vivo. Phytomedicine 2012, 20, 67–70. [Google Scholar] [CrossRef]
- Reddy, B.U.; Mullick, R.; Kumar, A.; Sudha, G.; Srinivasan, N.; Das, S. Small molecule inhibitors of HCV replication from pomegranate. Sci. Rep. 2014, 4, 5411. [Google Scholar] [CrossRef]
- Moradi, M.; Karimi, A.; Alidadi, S.; Saedi-Marghmaleki, M.; Salehian, M. In vitro Anti-adenovirus activity of pomegranate (Punica granatum L.) peel extract. Adv. Herb. Med. 2016, 2, 1–8. [Google Scholar]
- Houston, D.M.J.; Bugert, J.J.; Denyer, S.P.; Heard, C.M. Correction: Potentiated virucidal activity of pomegranate rind extract (PRE) and punicalagin against Herpes simplex virus (HSV) when co-administered with zinc (II) ions, and antiviral activity of PRE against HSV and aciclovir-resistant HSV. PLoS ONE 2017, 12, e0188609. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, J.; Rajarajan, S. Study on antiviral activities, drug-likeness and molecular docking of bioactive compounds of Punica granatum L. to Herpes simplex virus—2 (HSV-2). Microb. Pathog. 2018, 118, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.T.; AlAjmi, M.F.; Hussain, A. Natural Compounds as Inhibitors of SARS-CoV-2 Main Protease (3CLpro): A Molecular Docking and Simulation Approach to Combat COVID-19. Chem. Rxiv. 2020. [Google Scholar] [CrossRef]
- Vardhan, S.; Sahoo, S.K. Searching inhibitors for three important proteins of COVID-19 through molecular docking studies. arXiv 2020, arXiv:2006.1667. [Google Scholar]
- Khalifa, I.; Zhu, W.; Nafie, M.S.; Dutta, K.; Li, C. Anti-COVID-19 Effects of Ten Structurally Different Hydrolysable Tannins through Binding with the Catalytic-Closed Sites of COVID-19 Main Protease: An In-Silico Approach. Preprints 2020. [Google Scholar] [CrossRef]
- Nettore, I.C.; Rocca, C.; Mancino, G.; Albano, L.; Amelio, D.; Grande, F.; Puoci, F.; Pasqua, T.; Desiderio, S.; Mazza, R.; et al. Quercetin and its derivative Q2 modulate chromatin dynamics in adipogenesis and Q2 prevents obesity and metabolic disorders in rats. J. Nutr. Biochem. 2019, 69, 151–162. [Google Scholar] [CrossRef]
- Mackay, J.; Mensah, G.; Mendis, S.; Greenland, K. The Atlas of Heart Disease and Stroke; World Health Organization, Ed.; World Health Organization in collaboration with the USA Centers for Disease Control and Prevention; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Cerda, B.; Ceron, J.J.; Tomas-Barberan, F.A.; Espin, J.C. Repeated oral administration of high doses of the pomegranate ellagitannin punicalagin to rats for 37 days is not toxic. J. Agric. Food. Chem. 2003, 51, 3493–3501. [Google Scholar] [CrossRef]
- Lei, F.; Zhang, X.N.; Wang, W.; Xing, D.M.; Xie, W.D.; Su, H.; Du, L.J. Evidence of anti-obesity effects of the pomegranate leaf extract in high-fat diet induced obese mice. Int. J. Obes. 2007, 31, 1023–1029. [Google Scholar] [CrossRef]
- Trichur Khabeer, S.; Prashant, A.; Haravey Krishnan, M. Dietary fatty acids from pomegranate seeds (Punica granatum) inhibit adipogenesis and impact the expression of the obesity-associated mRNA transcripts in human adipose-derived mesenchymal stem cells. J. Food Biochem. 2019, 43, e12739. [Google Scholar] [CrossRef]
- Seeram, N.P.; Henning, S.M.; Zhang, Y.; Suchard, M.; Li, Z.; Heber, D. Pomegranate juice ellagitannin metabolites are present in human plasma and some persist in urine for up to 48 hours. J. Nutr. 2006, 136, 2481–2485. [Google Scholar] [CrossRef] [PubMed]
- Bialonska, D.; Kasimsetty, S.G.; Schrader, K.K.; Ferreira, D. The effect of pomegranate (Punica granatum L.) byproducts and ellagitannins on the growth of human gut bacteria. J. Agric. Food. Chem. 2009, 57, 8344–8349. [Google Scholar] [CrossRef]
- Chaves, F.M.; Baptista, I.L.; Simabuco, F.M.; Quaresma, P.G.F.; Pena, F.L.; Bezerra, R.M.N.; Pauli, J.R.; da Cunha, D.T.; Campos-Ferraz, P.L.; Antunes, A.E.C. High-intensity-exercise-induced intestinal damage is protected by fermented milk supplemented with whey protein, probiotic and pomegranate (Punica granatum L.). Br. J. Nutr. 2018, 119, 896–909. [Google Scholar] [CrossRef] [PubMed]
- George, N.S.; Cheung, L.M.; Luthria, D.L.; Santin, M.; Dawson, H.D.; Bhagwat, A.A.; Smith, A.D. Pomegranate peel extract alters the microbiome in mice and dysbiosis caused by Citrobacter rodentium infection. Food Sci. Nutr. 2019, 7, 2565–2576. [Google Scholar] [PubMed]
- Smith, A.D.; George, N.S.; Cheung, L.; Bhagavathy, G.V.; Luthria, D.L.; John, K.M.; Bhagwat, A.A. Pomegranate peel extract reduced colonic damage and bacterial translocation in a mouse model of infectious colitis induced by Citrobacter rodentium. Nutr. Res. 2020, 73, 27–37. [Google Scholar] [CrossRef]
- Turk, G.; Sonmez, M.; Aydin, M.; Yuce, A.; Gur, S.; Yuksel, M.; Aksu, E.H.; Aksoy, H. Effects of pomegranate juice consumption on sperm quality, spermatogenic cell density, antioxidant activity and testosterone level in male rats. Clin. Nutr. 2008, 27, 289–296. [Google Scholar] [CrossRef]
- Turk, G.; Sonmez, M.; Ceribasi, A.O.; Yuce, A.; Atessahin, A. Attenuation of cyclosporine A-induced testicular and spermatozoal damages associated with oxidative stress by ellagic acid. Int. Immunopharmacol. 2010, 10, 177–182. [Google Scholar] [CrossRef]
- Forest, C.P.; Padma-Nathan, H.; Liker, H.R. Efficacy and safety of pomegranate juice on improvement of erectile dysfunction in male patients with mild to moderate erectile dysfunction: A randomized, placebo-controlled, double-blind, crossover study. Int. J. Impot. Res. 2007, 19, 564–567. [Google Scholar] [CrossRef][Green Version]
- Qnais, E.Y.; Elokda, A.S.; Abu Ghalyun, Y.Y.; Abdulla, F.A. Antidiarrheal activity of the aqueous extract of Punica granatum (pomegranate) peels. Pharm. Biol. 2007, 45, 715–720. [Google Scholar] [CrossRef]
- Olapour, S.; Mousavi, E.; Sheikhzade, M.; Hoseininezhad, O.; Najafzadeh, H. Evaluation antidiarrheal effects of pomegranate peel extract. J. Iran. Chem. Soc. 2009, 6, 115–143. [Google Scholar]
- Zhao, S.S.; Ma, D.X.; Zhu, Y.; Zhao, J.H.; Zhang, Y.; Chen, J.Q.; Sheng, Z.L. Antidiarrheal effect of bioactivity-guided fractions and bioactive components of pomegranate (Punica granatum L.) peels. Neurogastroenterol. Motil. 2018, 30, e13364. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, A.L. Influence of periodontal disease on cardiovascular diseases. Rom. J. Intern. Med. 2005, 43, 9–21. [Google Scholar] [PubMed]
- DiSilvestro, R.A.; DiSilvestro, D.J.; DiSilvestro, D.J. Pomegranate Extract Mouth Rinsing Effects on Saliva Measures Relevant to Gingivitis Risk. Phytother. Res. 2009, 23, 1123–1127. [Google Scholar] [CrossRef]
- Sastravaha, G.; Gassmann, G.; Sangtherapitikul, P.; Grimm, W.D. Adjunctive periodontal treatment with Centella asiatica and Punica granatum extracts in supportive periodontal therapy. J. Int. Acad. Periodontol. 2005, 7, 70–79. [Google Scholar] [PubMed]
- Al-Wazni, W.S.; Hadi, B.S. Antivirulence effects of pomegranate peel extracts on most common urinary tract infection pathogens in pregnant women. J. Contemp. Med. Sci. 2015, 1, 7–12. [Google Scholar] [CrossRef]
- Manouchehrian, M.; Shakiba, M.; Shariat, M.; Lotfi, M.H.; Kamalinejad, M.; Babaeian, M. The Effect of Pomegranate Paste on Neonatal Jaundice Incidence: A Clinical Trial in Women during Pregnancy. Int. J. Clin. Med. 2017, 8, 144–151. [Google Scholar] [CrossRef]
- Finn-Sell, S.L.; Cottrell, E.C.; Greenwood, S.L.; Dilworth, M.R.; Cowley, E.J.; Sibley, C.P.; Wareing, M. Pomegranate Juice Supplementation Alters Utero-Placental Vascular Function and Fetal Growth in the eNOS-/- Mouse Model of Fetal Growth Restriction. Front. Physiol. 2018, 9, 1145. [Google Scholar] [CrossRef]
- Henning, S.M.; Wallenstein, M.B.; Weigel, N.; Johnson, C.; Yang, J.; Lee, R.; Korn, M.; Scala, M.; Stevenson, K.D.; Ben-Nissan, D.; et al. Appearance of Ellagic Acid Metabolites from Pomegranate Juice in Breast Milk: A Case Report. Ann. Clin. Case Rep. 2019, 4, 1738. [Google Scholar]
- Heneka, M.T.; Carson, M.J.; El Khoury, J.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L.; Jacobs, A.H.; Wyss-Coray, T.; Vitorica, J.; Ransohoff, R.M.; et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef]
- DaSilva, N.A.; Nahar, P.P.; Ma, H.; Eid, A.; Wei, Z.; Meschwitz, S.; Zawia, N.H.; Slitt, A.L.; Seeram, N.P. Pomegranate ellagitannin-gut microbial-derived metabolites, urolithins, inhibit neuroinflammation in vitro. Nutr. Neurosci. 2019, 22, 185–195. [Google Scholar] [CrossRef]
- Velagapudi, R.; Lepiarz, I.; El-Bakoush, A.; Katola, F.O.; Bhatia, H.; Fiebich, B.L.; Olajide, O.A. Induction of Autophagy and Activation of SIRT-1 Deacetylation Mechanisms Mediate Neuroprotection by the Pomegranate Metabolite Urolithin A in BV2 Microglia and Differentiated 3D Human Neural Progenitor Cells. Mol. Nutr. Food Res. 2019, 63, e1801237. [Google Scholar] [CrossRef] [PubMed]
- Casedas, G.; Les, F.; Choya-Foces, C.; Hugo, M.; Lopez, V. The Metabolite Urolithin-A Ameliorates Oxidative Stress in Neuro-2a Cells, Becoming a Potential Neuroprotective Agent. Antioxidants 2020, 9, 177. [Google Scholar] [CrossRef] [PubMed]
- Kujawska, M.; Jourdes, M.; Kurpik, M.; Szulc, M.; Szaefer, H.; Chmielarz, P.; Kreiner, G.; Krajka-Kuzniak, V.; Mikolajczak, P.L.; Teissedre, P.L.; et al. Neuroprotective Effects of Pomegranate Juice against Parkinson’s Disease and Presence of Ellagitannins-Derived Metabolite-Urolithin A-In the Brain. Int. J. Mol. Sci. 2020, 21, 202. [Google Scholar] [CrossRef] [PubMed]
- Siddarth, P.; Li, Z.P.; Miller, K.J.; Ercoli, L.M.; Merril, D.A.; Henning, S.M.; Heber, D.; Small, G.W. Randomized placebo-controlled study of the memory effects of pomegranate juice in middle-aged and older adults. Am. J. Clin. Nutr. 2020, 111, 170–177. [Google Scholar] [CrossRef]
- Vidal, A.; Fallarero, A.; Pena, B.R.; Medina, M.E.; Gra, B.; Rivera, F.; Gutierrez, Y.; Vuorela, P.M. Studies on the toxicity of Punica granatum L. (Punicaceae) whole fruit extracts. J. Ethnopharmacol. 2003, 89, 295–300. [Google Scholar] [CrossRef]
- Heber, D.; Seeram, N.P.; Wyatt, H.; Henning, S.M.; Zhang, Y.; Ogden, L.G.; Dreher, M.; Hill, J.O. Safety and antioxidant activity of a pomegranate ellagitannin-enriched polyphenol dietary supplement in overweight individuals with increased waist size. J. Agric. Food Chem. 2007, 55, 10050–10054. [Google Scholar] [CrossRef] [PubMed]
- Aviram, M.; Rosenblat, M.; Gaitini, D.; Nitecki, S.; Hoffman, A.; Dornfeld, L.; Volkova, N.; Presser, D.; Attias, J.; Liker, H.; et al. Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clin. Nutr. 2004, 23, 423–433. [Google Scholar] [CrossRef]
- Chimento, A.; Sirianni, R.; Saturnino, C.; Caruso, A.; Sinicropi, M.S.; Pezzi, V. Resveratrol and Its Analogs as Antitumoral Agents for Breast Cancer Treatment. Mini Rev. Med. Chem. 2016, 16, 699–709. [Google Scholar] [CrossRef]
- Iacopetta, D.; Rosano, C.; Puoci, F.; Parisi, O.I.; Saturnino, C.; Caruso, A.; Longo, P.; Ceramella, J.; Malzert-Freon, A.; Dallemagne, P.; et al. Multifaceted properties of 1,4-dimethylcarbazoles: Focus on trimethoxybenzamide and trimethoxyphenylurea derivatives as novel human topoisomerase II inhibitors. Eur. J. Pharm. Sci. 2017, 96, 263–272. [Google Scholar] [CrossRef]
- Sala, M.; Chimento, A.; Saturnino, C.; Gomez-Monterrey, I.M.; Musella, S.; Bertamino, A.; Milite, C.; Sinicropi, M.S.; Caruso, A.; Sirianni, R.; et al. Synthesis and cytotoxic activity evaluation of 2,3-thiazolidin-4-one derivatives on human breast cancer cell lines. Bioorg. Med. Chem. Lett. 2013, 23, 4990–4995. [Google Scholar] [CrossRef]
- Tundis, R.; Iacopetta, D.; Sinicropi, M.S.; Bonesi, M.; Leporini, M.; Passalacqua, N.G.; Ceramella, J.; Menichini, F.; Loizzo, M.R. Assessment of antioxidant, antitumor and pro-apoptotic effects of Salvia fruticosa Mill. subsp. thomasii (Lacaita) Brullo, Guglielmo, Pavone & Terrasi (Lamiaceae). Food Chem. Toxicol. 2017, 106, 155–164. [Google Scholar] [PubMed]
- Ceramella, J.; Loizzo, M.R.; Iacopetta, D.; Bonesi, M.; Sicari, V.; Pellicano, T.M.; Saturnino, C.; Malzert-Freon, A.; Tundis, R.; Sinicropi, M.S. Anchusa azurea Mill. (Boraginaceae) aerial parts methanol extract interfering with cytoskeleton organization induces programmed cancer cells death. Food Funct. 2019, 10, 4280–4290. [Google Scholar] [CrossRef] [PubMed]
- Francomano, F.; Caruso, A.; Barbarossa, A.; Fazio, A.; La Torre, C.; Ceramella, J.; Mallamaci, R.; Saturnino, C.; Iacopetta, D.; Sinicropi, M.S. β-Caryophyllene: A Sesquiterpene with Countless Biological Properties. Appl. Sci. 2019, 9, 5420. [Google Scholar] [CrossRef]
- Khomich, L.M.; Perova, I.B.; Eller, K.I. Pomegranate juice nutritional profile. Vopr. Pitan. 2019, 88, 80–92. [Google Scholar] [PubMed]
- Bar-Ya’akov, I.; Tian, L.; Amir, R.; Holland, D. Primary Metabolites, Anthocyanins, and Hydrolyzable Tannins in the Pomegranate Fruit. Front. Plant Sci. 2019, 10, 620. [Google Scholar] [CrossRef] [PubMed]
- Vucic, V.; Grabez, M.; Trchounian, A.; Arsic, A. Composition and Potential Health Benefits of Pomegranate: A Review. Curr. Pharm. Des. 2019, 25, 1817–1827. [Google Scholar] [CrossRef] [PubMed]
- Viswanath, M.; Sridevi, P.; Bhagavan, B.V.K.; Ravindra Kumar, K.; Subbaramamma, P. Toxicological, Pharmacological and Cellular properties of Pomegranate (Punica granatum L.): A Review. J. Pharmacogn. Phytochem. 2019, 8, 172–176. [Google Scholar]
- Natalello, A.; Hervas, G.; Toral, P.G.; Luciano, G.; Valenti, B.; Mendoza, A.G.; Pauselli, M.; Priolo, A.; Frutos, P. Bioactive compounds from pomegranate by-products increase the in vitro ruminal accumulation of potentially health promoting fatty acids. Anim. Feed Sci. Technol. 2020, 259, 114355. [Google Scholar] [CrossRef]
- Prakash, C.V.S.; Prakash, I. Bioactive Chemical Constituents from Pomegranate (Punica Granatum) Juice, Seed and Peel-A Review. Int. J. Res. Chem. Environ. 2011, 1, 1–18. [Google Scholar]
- Sateriale, D.; Facchiano, S.; Colicchio, R.; Pagliuca, C.; Varricchio, E.; Paolucci, M.; Volpe, M.G.; Salvatore, P.; Pagliarulo, C. In vitro Synergy of Polyphenolic Extracts from Honey, Myrtle and Pomegranate Against Oral Pathogens, S. mutans and R. dentocariosa. Front. Microbiol. 2020, 11, 1465. [Google Scholar] [CrossRef]
| Structure | Activity | Bibliographic Reference |
|---|---|---|
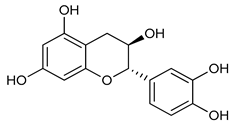 (+)-(2R,3S)-Catechin (1) | Antioxidant | [48] |
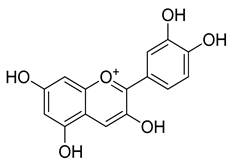 Cyanidin (2) | Antioxidant | [5] |
| Antitumoral Anti-inflammatory | ||
 Delphinidin (3) | Antitumoral | [5] |
| Antioxidant Anti-inflammatory | ||
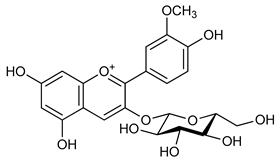 Pelargonidin-3-O-glucoside (4) | Antitumoral | [5] |
| Antioxidant Anti-inflammatory | ||
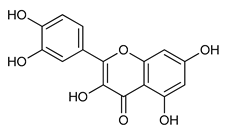 Quercetin (5) | Antiviral | [52] |
| Antioxidant Cardioprotective Antitumoral | ||
 Kaempferol (6) | Anti-cancerogenic | [54] |
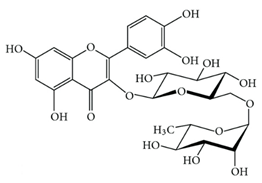 Rutin (7) | Antitumoral Antimicrobial | [54] |
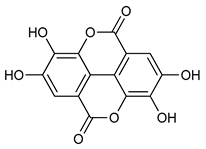 Ellagic Acid (8) | Antitumoral | [56] |
| Antiviral Antimicrobial | ||
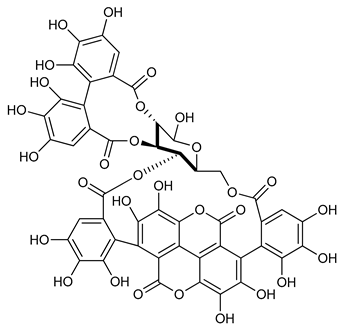 Punicalagin (9) | Anti-inflammatory | [57] |
| Antiproliferative | ||
| Pro-apoptotics Antigenotoxic | ||
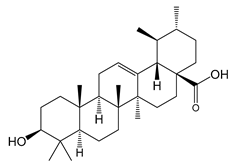 Ursolic acid (10) | Antitumoral | [65] |
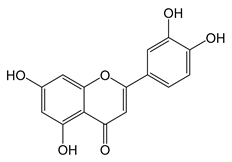 Luteolin (11) | Antitumoral | [65] |
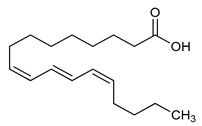 Punicic Acid (12) | Anti-inflammatory | [83] |
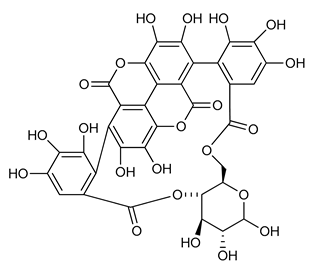 Punicalin (13) | Anti-inflammatory | [80] |
 Vitamin A or retinol (14) | Antioxidant | [20] |
 Vitamin C or ascorbic acid (15) | Antioxidant | [20] |
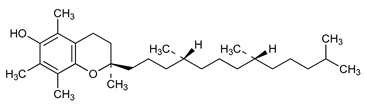 Vitamin E or tocopherol (16) | Antioxidant Anti-inflammatory | [20] |
 Caffeic acid (17) | Antidiabetics | [5] |
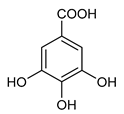 Gallic acid (18) | Antimicrobial Prevention of cardiovascular diseases | [110] |
| Pomegranate Extracts | Properties | References |
|---|---|---|
| Antioxidant activity | ||
| Juice |  ROS ROS | [23,25,26,32] |
| Peel (PPE) |  ROS ROS | [25,27,28,29,30,31] |
| Seed | Radical scavenging | [24,25,32] |
| Anticancer properties | ||
| Fruit (PoMx) | Reverse tamoxifen resistant | [61] |
| Inhibition of cell proliferation and metastasis (HSC-3) | [62] | |
| Husk | Inhibition of cell proliferation (HepG2 and CACO) | [68] |
| Juice | Inhibition of cell proliferation (HT-29) | [5] |
| Leaf | Inhibition of cell proliferation (HeLa) | [75] |
| Necrosis | ||
| Apoptosis | ||
| Peel | Inhibition of cell proliferation (MCF-7 and HCT-116) | [67] |
| Inhibition of cell proliferation (MCF-7, SKOV3, PC3 and A549) | [69] | |
| Inhibition of cell proliferation (K562) | [70] | |
| Cell cycle G2/M | ||
| Apoptosis | ||
| Inhibition of cell proliferation (HONE1) | [76] | |
| Seed | Inhibition of cell proliferation (HepG2 and CACO) | [68] |
| Seedless aril | Inhibition of cell proliferation (LAPC4) | [59] |
| Skin (PoPx) | Inhibition of cell proliferation (MCF-7 and LAPC4) | [59] |
| Apoptosis | ||
| Inhibition of cell proliferation (MCF-7) | [60] | |
| Antiangiogenic | ||
| Inhibition of cell invasion and motility | ||
| Inhibition of cell proliferation (melanocyte) | [63,64] | |
| Free radicals block in UVA and UVB irradiated human skin | ||
| Whole extract (PE) | Inhibition of cell proliferation (WA-4) | [65] |
| Cell cycle arrest G0/G1 | ||
| Apoptosis | ||
| Inhibition of cell proliferation (PANC-1 and AsPC-1) | [66] | |
| Cell cycle arrest G2 | ||
| Immunomodulatory effects in stem cells | [71] | |
| Anti-inflammatory properties | ||
| Flower (PFE) |  NO, iNOS, COX-2, PGE-2, TNF-α, IL-6, IL-1β NO, iNOS, COX-2, PGE-2, TNF-α, IL-6, IL-1β | [89] |
| Inhibition of NF-kB activation and MAPKs phosphorylation | ||
| Fruit (PoMx) |  Thrombospondin, TGF-β1 Thrombospondin, TGF-β1 | [85] |
| Peel (PPE) | 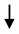 TNF-α, IL-1β, MCP-1, ICAM TNF-α, IL-1β, MCP-1, ICAM | [88] |
| Seed | 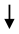 COX, LOX COX, LOX | [81,82] |
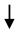 Lipid peroxidation (LPO) Lipid peroxidation (LPO) | [83] | |
| Whole extract (PE) | 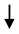 NO NO | [80,84] |
 COX-2, PTGES COX-2, PTGES | [87,91] | |
| Antidiabetic activity | ||
| Flower (PFE) | Suppression of α-glucosidase activity | [95] |
| Activation of PPAR-γ | [5] | |
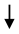 LPO LPO | [100] | |
| Improvement of insulin sensitivity, blood lipid profile, and liver glycogen content | [102] | |
| Fruit (PoMx) | 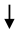 Serum resistin Serum resistin | [99] |
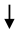 FBG FBG | [103] | |
 IRS-1, Akt, GLUT-2, GLUT-4 IRS-1, Akt, GLUT-2, GLUT-4 | ||
| Juice | Inhibition of DPP-4 | [104] |
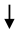 Erythropoietin Erythropoietin | [105] | |
| Peel (PPE) | Inhibition of LPO | [98] |
 Serum glucose concentration Serum glucose concentration | ||
| Seed | Improvement of insulin sensitivity | [97] |
| Antimicrobial activities | ||
| Peel (PPE) | S. Enteritidis, S. Kentucky | [111] |
| C. albicans, C. parapsilosis, R. mucillaginosa, E. dermatitidis, S. aureus | [112] | |
| E.coli, P. aeruginisa, P. vulgaris, S. typhi, S. aureus, S. epidermidis, K. pneumonia | [113] | |
| Skin (PoPx) | S. aureus, E. coli, Y. enterocolitica | [110] |
| Prevention of cardiovascular diseases | ||
| Juice |  Erythrocyte count, bleeding time, thrombin time Erythrocyte count, bleeding time, thrombin time | [118] |
| Activation of thromboplastin time | ||
| Inhibition of platelets aggregation induced by ADP | ||
| Positive effects on angina pectoris | [119] | |
| Improvement of lipid profile, oxidative, and inflammatory biomarkers | [121] | |
 Blood pressure Blood pressure | [121,122] | |
| Peel (PPE) | Reduction of doxorubicin cardiotoxicity | [120] |
| Antiviral activities | ||
| Peel (PPE) | Hepatitis C virus | [127] |
| HSV-2 | [130] | |
| Rind | HSV-1 | [129] |
| Whole extract (PE) | Inhibition of H1N1, H3N2, H5N1 virus replication | [124,125] |
| Inhibition of enterovirus 71 replication | [126] | |
| Adenovirus | [128] | |
| Obesity | ||
| Leaf | Inhibition of pancreatic lipase | [137] |
| Seed | Inhibition of adipogenesis | [138] |
| Intestinal regulation | ||
| Juice |  Intestinal motility Intestinal motility | [141] |
| Rebalancing of intestinal microbiome | ||
| Peel (PPE) |  Pathogenicity of C. rodentium Pathogenicity of C. rodentium | [142,143] |
| Effects on the male reproductive system | ||
| Juice |  Sperm concentration, mobility, and density Sperm concentration, mobility, and density | [145] |
 Erectile activity Erectile activity | [146] | |
| Antidiarrheal effects | ||
| Peel (PPE) |  Gastrointestinal transit Gastrointestinal transit | [149] |
| Skin | Inhibition of spontaneous ileum movements | [147] |
| Effects on oral health | ||
| Whole extract (PE) |  Microorganism from the dental plaque Microorganism from the dental plaque | [151] |
 Microorganism of gingivitis Microorganism of gingivitis | [152] | |
| Effects on pregnant and breastfeeding women | ||
| Juice | Improvement of uterine and umbilical artery function | [155] |
| Presence of ellagic acid in breast milk | [156] | |
| Peel (PPE) | Antibacterial effects against S. aureus and E.coli | [153] |
| Whole extract (PE) |  bilirubin bilirubin | [154] |
| Effects on nervous system | ||
| Juice | Impediment of oxidative damage and α-synuclein aggregationImprovement of mitochondrial ALDH activity | [161] |
| Stabilization of the ability to learn visual information | [162] | |
| Whole extract (PE) |  NO, IL-6, PGE-2, TNF-α in BV-2 cells NO, IL-6, PGE-2, TNF-α in BV-2 cells | [158,159] |
| Protection of SH-SY5Y and BV-2 cell viability | ||
| Activation of Sirt-1 in BV-2 cells | [159] | |
| Improvement of mitochondrial activity, CAT, SOD, GR, and GPx activities and redox state | [160] | |
 Peroxiredoxins 1 and 3 Peroxiredoxins 1 and 3 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caruso, A.; Barbarossa, A.; Tassone, A.; Ceramella, J.; Carocci, A.; Catalano, A.; Basile, G.; Fazio, A.; Iacopetta, D.; Franchini, C.; et al. Pomegranate: Nutraceutical with Promising Benefits on Human Health. Appl. Sci. 2020, 10, 6915. https://doi.org/10.3390/app10196915
Caruso A, Barbarossa A, Tassone A, Ceramella J, Carocci A, Catalano A, Basile G, Fazio A, Iacopetta D, Franchini C, et al. Pomegranate: Nutraceutical with Promising Benefits on Human Health. Applied Sciences. 2020; 10(19):6915. https://doi.org/10.3390/app10196915
Chicago/Turabian StyleCaruso, Anna, Alexia Barbarossa, Antonio Tassone, Jessica Ceramella, Alessia Carocci, Alessia Catalano, Giovanna Basile, Alessia Fazio, Domenico Iacopetta, Carlo Franchini, and et al. 2020. "Pomegranate: Nutraceutical with Promising Benefits on Human Health" Applied Sciences 10, no. 19: 6915. https://doi.org/10.3390/app10196915
APA StyleCaruso, A., Barbarossa, A., Tassone, A., Ceramella, J., Carocci, A., Catalano, A., Basile, G., Fazio, A., Iacopetta, D., Franchini, C., & Sinicropi, M. S. (2020). Pomegranate: Nutraceutical with Promising Benefits on Human Health. Applied Sciences, 10(19), 6915. https://doi.org/10.3390/app10196915









