Comparison of Optical Imaging Techniques to Quantitatively Assess the Perfusion of the Gastric Conduit during Oesophagectomy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study
2.2. Procedure
2.3. Imaging Modalities and Software
2.4. Outcomes
2.5. Quantitative Parameters
- 1.
- The percentage of speckle contrast pixels (speckle %) indicative of the flow obtained in the M-mode of the scans;
- 2.
- Total vessel density (TVD): ratio between the total vessel length and the area of ROI (mm/mm2);
- 3.
- Perfused vessel density (PVD): ratio between the perfused scored vessel length and the area of ROI (mm/mm2);
- 4.
- Microvascular flow index (MFI): sum of the qualitative determination (0 no, 1 intermittent, 2 sluggish, 3 continuous) of the predominate flow in four quadrants of the SDF image, divided by four;
- 5.
- De Backer Score (DBS): number of vessels crossing a grid overlay divided by the total length of the vessels;
- 6.
- Proportion of perfused vessels (PPV): perfused vessel length divided by the total vessel length of the ROI in %;
- 7.
- Velocity (μm/s): assessed per selected vessels by a space–time diagram;
- 8.
- Flux in laser speckle perfusion units (LSPU);
- 9.
- ∆%Flux defined as the percentage difference in Flux relative to Loc1 (positive number when there is a decrease in Flux);
- 10.
- Influx time point (τ): time in seconds between the occurrence of the peak fluorescence in the right gastroepiploic artery and the time point at which the fluorescence intensity in the ROI was statistically significantly larger than the background;
- 11.
- Fmax: maximal intensity in arbitrary units (AU) within 50 s after the influx time point. Fmax was corrected by subtracting the median background value measured from the start of imaging to the influx time point (τ);
- 12.
- Tmax: time in seconds after the influx time point (τ) at which the background-corrected fluorescence intensity reached Fmax, also known as the time-to-peak;
- 13.
- T1/2: time in seconds after the influx time point (τ) at which the background-corrected fluorescence intensity was half of the Fmax;
- 14.
- Mean slope: rate at which the fluorescence intensity increased (AU/s).
2.6. Statistics
3. Results
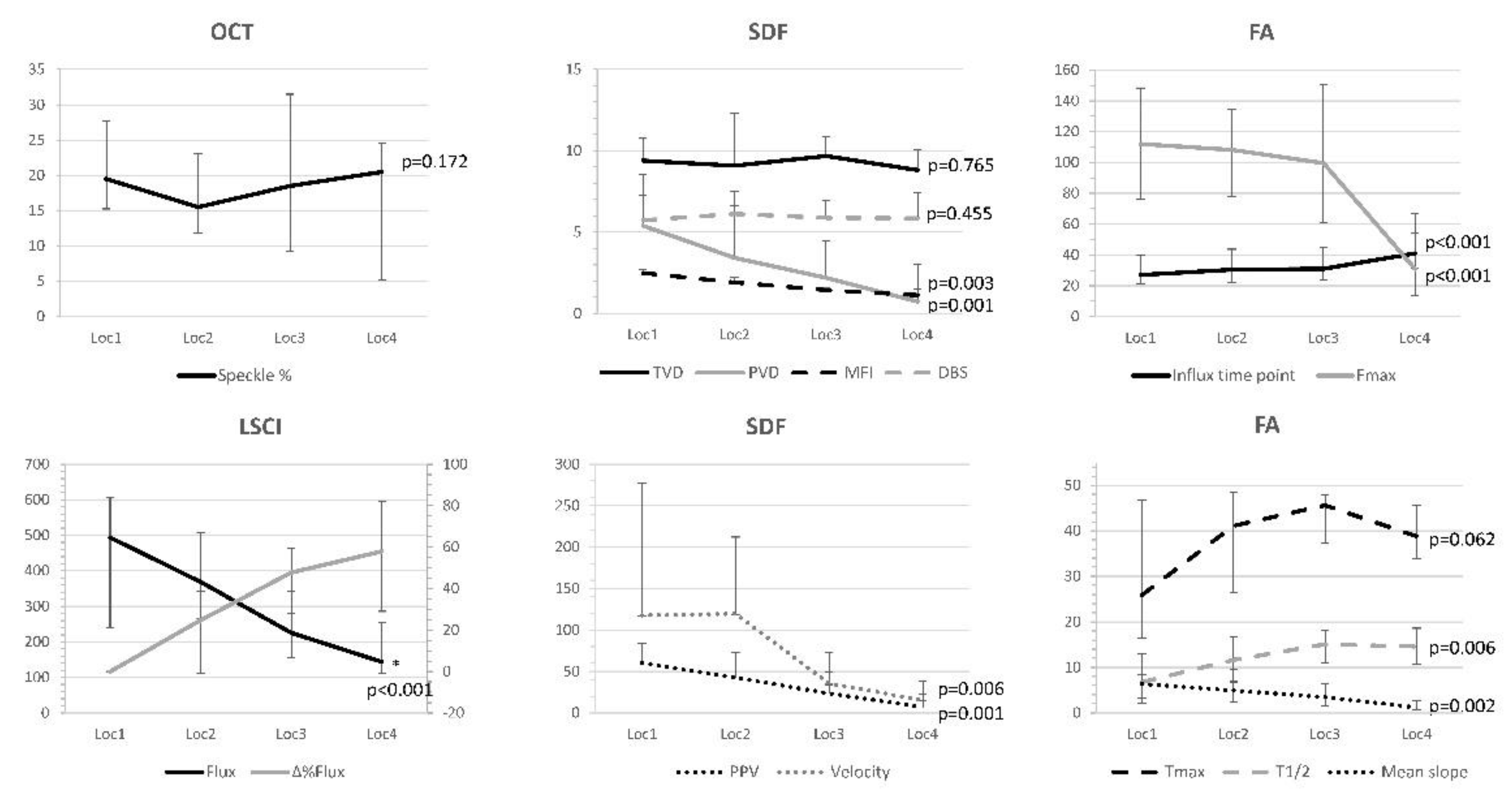
3.1. Imaging Characteristics
3.2. Quantitative Parameters
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Van Hagen, P.; Hulshof, M.C.; van Lanschot, J.J.; Steyerberg, E.W.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.; Richel, D.J.; Nieuwenhuijzen, G.A.; Hospers, G.A.; Bonenkamp, J.J.; et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N. Engl. J. Med. 2012, 366, 2074–2084. [Google Scholar] [CrossRef] [PubMed]
- Fransen, L.F.C.; Berkelmans, G.H.K.; Asti, E.; van Berge Henegouwen, M.I.; Berlth, F.; Bonavina, L.; Brown, A.; Bruns, C.; van Daele, E.; Gisbertz, S.S.; et al. The Effect of Postoperative Complications After Minimally Invasive Esophagectomy on Long-term Survival: An International Multicenter Cohort Study. Ann. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Findlay, J.M.; Gillies, R.S.; Millo, J.; Sgromo, B.; Marshall, R.E.; Maynard, N.D. Enhanced recovery for esophagectomy: A systematic review and evidence-based guidelines. Ann. Surg. 2014, 259, 413–431. [Google Scholar] [CrossRef]
- Fischer, C.; Lingsma, H.; Klazinga, N.; Hardwick, R.; Cromwell, D.; Steyerberg, E.; Groene, O. Volume-outcome revisited: The effect of hospital and surgeon volumes on multiple outcome measures in oesophago-gastric cancer surgery. PLoS ONE 2017, 12, E0183955. [Google Scholar] [CrossRef] [PubMed]
- Jansen, S.M.; de Bruin, D.M.; van Berge Henegouwen, M.I.; Strackee, S.D.; Veelo, D.P.; van Leeuwen, T.G.; Gisbertz, S.S. Optical techniques for perfusion monitoring of the gastric tube after esophagectomy: A review of technologies and thresholds. Dis. Esophagus Off. J. Int. Soc. Dis. Esophagus 2018, 31. [Google Scholar] [CrossRef]
- Van Manen, L.; Handgraaf, H.J.M.; Diana, M.; Dijkstra, J.; Ishizawa, T.; Vahrmeijer, A.L.; Mieog, J.S.D. A practical guide for the use of indocyanine green and methylene blue in fluorescence-guided abdominal surgery. J. Surg. Oncol. 2018, 118, 283–300. [Google Scholar] [CrossRef]
- Jansen, S.M.; de Bruin, D.M.; Faber, D.J.; Dobbe, I.; Heeg, E.; Milstein, D.M.J.; Strackee, S.D.; van Leeuwen, T.G. Applicability of quantitative optical imaging techniques for intraoperative perfusion diagnostics: A comparison of laser speckle contrast imaging, sidestream dark-field microscopy, and optical coherence tomography. J. Biomed. Opt. 2017, 22, 086004. [Google Scholar] [CrossRef]
- Prasetya, H.; Jansen, S.M.; Marquering, H.A.; van Leeuwen, T.G.; Gisbertz, S.S.; de Bruin, D.M.; van Bavel, E. Estimation of microvascular perfusion after esophagectomy: A quantitative model of dynamic fluorescence imaging. Med. Biol. Eng. Comput. 2019, 57, 1889–1990. [Google Scholar] [CrossRef]
- AV, D.S.; Lin, H.; Henderson, E.R.; Samkoe, K.S.; Pogue, B.W. Review of fluorescence guided surgery systems: Identification of key performance capabilities beyond indocyanine green imaging. J. Biomed. Opt. 2016, 21, 80901. [Google Scholar] [CrossRef]
- Bossuyt, P.M.; Reitsma, J.B.; Bruns, D.E.; Gatsonis, C.A.; Glasziou, P.P.; Irwig, L.M.; Moher, D.; Rennie, D.; de Vet, H.C.; Lijmer, J.G. The STARD statement for reporting studies of diagnostic accuracy: Explanation and elaboration. Clin. Chem. 2003, 49, 7–18. [Google Scholar] [CrossRef]
- Vandenbroucke, J.P.; von Elm, E.; Altman, D.G.; Gotzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Int. J. Surg. (Lond. Engl.) 2014, 12, 1500–1524. [Google Scholar] [CrossRef] [PubMed]
- Jansen, S.M.; de Bruin, D.M.; van Berge Henegouwen, M.I.; Strackee, S.D.; Veelo, D.P.; van Leeuwen, T.G.; Gisbertz, S.S. Can we predict necrosis intra-operatively? Real-time optical quantitative perfusion imaging in surgery: Study protocol for a prospective, observational, in vivo pilot study. Pilot Feasibility Stud. 2017, 3, 65. [Google Scholar] [CrossRef] [PubMed]
- Hagens, E.R.C.; Künzli, H.T.; van Rijswijk, A.S.; Meijer, S.L.; Mijnals, R.C.D.; Weusten, B.; Geijsen, E.D.; van Laarhoven, H.W.M.; van Berge Henegouwen, M.I.; Gisbertz, S.S. Distribution of lymph node metastases in esophageal adenocarcinoma after neoadjuvant chemoradiation therapy: A prospective study. Surg. Endosc. 2019. [Google Scholar] [CrossRef]
- Jansen, S.M.; Almasian, M.; Wilk, L.S.; de Bruin, D.M.; van Berge Henegouwen, M.I.; Strackee, S.D.; Bloemen, P.R.; Meijer, S.L.; Gisbertz, S.S.; van Leeuwen, T.G. Feasibility of Optical Coherence Tomography (OCT) for Intra-Operative Detection of Blood Flow during Gastric Tube Reconstruction. Sensors 2018, 18, 1331. [Google Scholar] [CrossRef] [PubMed]
- De Backer, D.; Hollenberg, S.; Boerma, C.; Goedhart, P.; Büchele, G.; Ospina-Tascon, G.; Dobbe, I.; Ince, C. How to evaluate the microcirculation: Report of a round table conference. Crit. Care (Lond. Engl.) 2007, 11, R101. [Google Scholar] [CrossRef] [PubMed]
- Balestra, G.M.; Bezemer, R.; Boerma, E.C.; Yong, Z.Y.; Sjauw, K.D.; Engstrom, A.E.; Koopmans, M.; Ince, C. Improvement of sidestream dark field imaging with an image acquisition stabilizer. BMC Med. Imaging 2010, 10, 15. [Google Scholar] [CrossRef]
- Jansen, S.M.; de Bruin, D.M.; van Berge Henegouwen, M.I.; Bloemen, P.R.; Strackee, S.D.; Veelo, D.P.; van Leeuwen, T.G.; Gisbertz, S.S. Effect of ephedrine on gastric conduit perfusion measured by laser speckle contrast imaging after esophagectomy: A prospective in vivo cohort study. Dis. Esophagus Off. J. Int. Soc. Dis. Esophagus 2018, 31, doy031. [Google Scholar] [CrossRef]
- Low, D.E.; Alderson, D.; Cecconello, I.; Chang, A.C.; Darling, G.E.; D’Journo, X.B.; Griffin, S.M.; Holscher, A.H.; Hofstetter, W.L.; Jobe, B.A.; et al. International Consensus on Standardization of Data Collection for Complications Associated With Esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann. Surg. 2015, 262, 286–294. [Google Scholar] [CrossRef]
- Slooter, M.D.; Mansvelders, M.S.E.; Bloemen, P.R.; Gisbertz, S.S.; Bemelman, W.A.; Tanis, P.J.; Hompes, R.; van Berge Henegouwen, M.I.; de Bruin, D.M. Defining indocyanine green fluorescence to assess anastomotic perfusion during gastrointestinal surgery: Systematic review. BJS Open 2020, in press. [Google Scholar]
- Milstein, D.M.; Ince, C.; Gisbertz, S.S.; Boateng, K.B.; Geerts, B.F.; Hollmann, M.W.; van Berge Henegouwen, M.I.; Veelo, D.P. Laser speckle contrast imaging identifies ischemic areas on gastric tube reconstructions following esophagectomy. Medicine 2016, 95, e3875. [Google Scholar] [CrossRef]
- Kamiya, K.; Unno, N.; Miyazaki, S.; Sano, M.; Kikuchi, H.; Hiramatsu, Y.; Ohta, M.; Yamatodani, T.; Mineta, H.; Konno, H. Quantitative assessment of the free jejunal graft perfusion. J. Surg. Res. 2015, 194, 394–399. [Google Scholar] [CrossRef]
- Kumagai, Y.; Hatano, S.; Sobajima, J.; Ishiguro, T.; Fukuchi, M.; Ishibashi, K.I.; Mochiki, E.; Nakajima, Y.; Ishida, H. Indocyanine green fluorescence angiography of the reconstructed gastric tube during esophagectomy: Efficacy of the 90-second rule. Dis. Esophagus Off. J. Int. Soc. Dis. Esophagus 2018, 31. [Google Scholar] [CrossRef]
- Koyanagi, K.; Ozawa, S.; Oguma, J.; Kazuno, A.; Yamazaki, Y.; Ninomiya, Y.; Ochiai, H.; Tachimori, Y. Blood flow speed of the gastric conduit assessed by indocyanine green fluorescence: New predictive evaluation of anastomotic leakage after esophagectomy. Medicine 2016, 95, e4386. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Sugiyama, A.; Ikegami, T.; Ishida, K.; Maruta, F.; Shimizu, F.; Ikeno, T.; Kawasaki, S. Revascularization using the short gastric vessels of the gastric tube after subtotal esophagectomy for intrathoracic esophageal carcinoma. J. Am. Coll. Surg. 2000, 190, 71–77. [Google Scholar] [CrossRef]
- Buise, M.P.; Ince, C.; Tilanus, H.W.; Klein, J.; Gommers, D.; van Bommel, J. The effect of nitroglycerin on microvascular perfusion and oxygenation during gastric tube reconstruction. Anesth. Analg. 2005, 100, 1107–1111. [Google Scholar] [CrossRef] [PubMed]
- Mcmillan, D.E. Development of vascular complications in diabetes. Vasc. Med. 1997, 2, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Strain, W.D.; Paldánius, P.M. Diabetes, cardiovascular disease and the microcirculation. Cardiovasc. Diabetol. 2018, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Gooszen, J.A.H.; Goense, L.; Gisbertz, S.S.; Ruurda, J.P.; van Hillegersberg, R.; van Berge Henegouwen, M.I. Intrathoracic versus cervical anastomosis and predictors of anastomotic leakage after oesophagectomy for cancer. Br. J. Surg. 2018, 105, 552–560. [Google Scholar] [CrossRef]
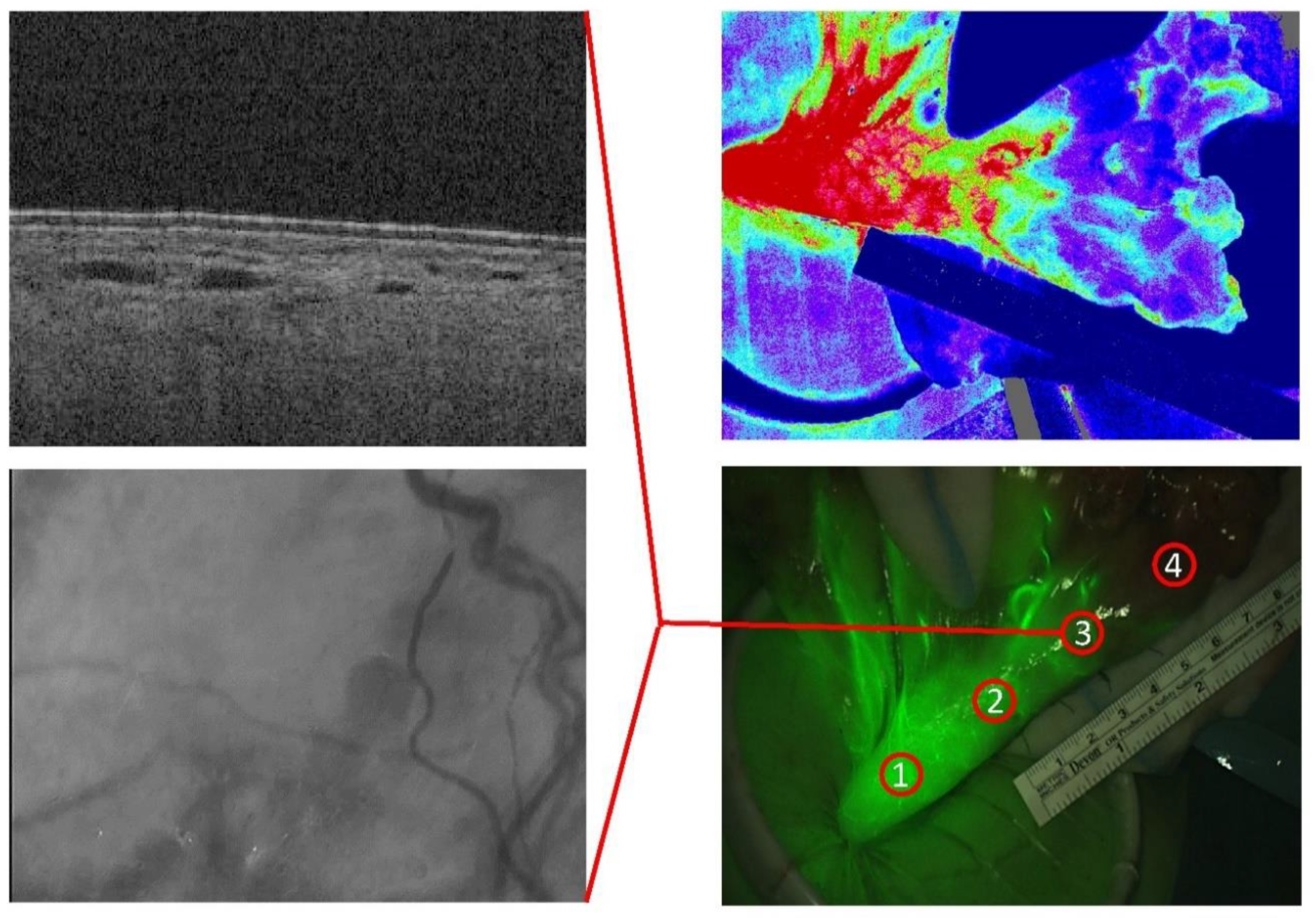
| Cohort (n = 22) | |
|---|---|
| Age (years) median total range | 62 (37–79) |
| Gender (male), n (%) | 19 (86.4) |
| Body mass index (kg/m2), median total range | 25.9 (17.0–34.2) |
| Surgical procedures, n (%) | |
| Ivor Lewis | 20 (91) |
| McKeown | 2 (9) |
| Cardiovascular disease, n (%) | 5 (23) |
| Diabetes Mellitus *, n (%) | 2 (9) |
| COPD, n (%) | 2 (9) |
| Field of View | Depth-Resolving | Ease of Use | Real-Time | Practical Limitations | |
|---|---|---|---|---|---|
| OCT | 10 mm × 10 mm × 2.5 mm | + | Sterile sheet, probe handling | + | Motion artefacts, shadowing |
| SDF | 1 mm × 1 mm | - | Sterile sheet, tissue-contact | + | Motion artefacts, focus |
| LSCI | Wide-field ROI: ø 1 cm | - | Non-contact | + | OR lights off |
| FA | Wide-field ROI: ø 1 cm | - | Non-contact | + | Invasive (ICG injection), OR lights off * |
| Location 4 No Anastomotic Leakage (n = 18) | Location 4 Anastomotic Leakage (n = 4) | p-Value | ||
|---|---|---|---|---|
| OCT | Speckle (%) | 21 (14–31) | 17 (17) | 0.600 |
| SDF | TVD (mm/mm2) | 8.7 (5.1–15.9) | 9.8 (9.5–17.4) | 0.088 |
| PVD (mm/mm2) | 0.5 (0.0–7.0) | 2.0 (0.9–5.4) | 0.155 | |
| MFI | 1.1 (0.3–2.0) | 1.3 (1–1.5) | 0.646 | |
| DBS | 5.4 (3.0–10.3) | 7.2 (6.9–11.00) | 0.056 | |
| PPV (%) | 5.7 (0.0–86.4) | 21.3 (9.3–31.1) | 0.155 | |
| Velocity (μm/s) | 15.2 (4.3–186.0) | 23.8 (14.8–60.3) | 0.560 | |
| LSCI | Flux (LSPU) | 128.7 (41.1–480.5) | 167.1 (114.1–335.2) | 0.422 |
| ∆%Flux (%) | 54 (−31–87) | 82 (52–87) | 0.072 | |
| FA | Influx time point (s) | 39.5 (21–73) | 44.0 (42–46) | 0.673 |
| Fmax (AU) | 39.8 (1.8–163.2) | 12.3 (10.9–13.7) | 0.131 | |
| Tmax (s) | 38.8 (34.2–44.6) | 39.8 (31.1–48.58) | 0.888 | |
| T1/2 (s) | 13.0 (6.3–21.3) | 29.3 (27.8–30.8) | 0.025 | |
| Mean slope (AU/s) | 1.3 (0.1–11.0) | 0.2 (0.1–0.2) | 0.041 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slooter, M.D.; Jansen, S.M.A.; Bloemen, P.R.; van den Elzen, R.M.; Wilk, L.S.; van Leeuwen, T.G.; van Berge Henegouwen, M.I.; de Bruin, D.M.; Gisbertz, S.S. Comparison of Optical Imaging Techniques to Quantitatively Assess the Perfusion of the Gastric Conduit during Oesophagectomy. Appl. Sci. 2020, 10, 5522. https://doi.org/10.3390/app10165522
Slooter MD, Jansen SMA, Bloemen PR, van den Elzen RM, Wilk LS, van Leeuwen TG, van Berge Henegouwen MI, de Bruin DM, Gisbertz SS. Comparison of Optical Imaging Techniques to Quantitatively Assess the Perfusion of the Gastric Conduit during Oesophagectomy. Applied Sciences. 2020; 10(16):5522. https://doi.org/10.3390/app10165522
Chicago/Turabian StyleSlooter, Maxime D., Sanne M. A. Jansen, Paul R. Bloemen, Richard M. van den Elzen, Leah S. Wilk, Ton G. van Leeuwen, Mark I. van Berge Henegouwen, Daniel M. de Bruin, and Suzanne S. Gisbertz. 2020. "Comparison of Optical Imaging Techniques to Quantitatively Assess the Perfusion of the Gastric Conduit during Oesophagectomy" Applied Sciences 10, no. 16: 5522. https://doi.org/10.3390/app10165522
APA StyleSlooter, M. D., Jansen, S. M. A., Bloemen, P. R., van den Elzen, R. M., Wilk, L. S., van Leeuwen, T. G., van Berge Henegouwen, M. I., de Bruin, D. M., & Gisbertz, S. S. (2020). Comparison of Optical Imaging Techniques to Quantitatively Assess the Perfusion of the Gastric Conduit during Oesophagectomy. Applied Sciences, 10(16), 5522. https://doi.org/10.3390/app10165522






