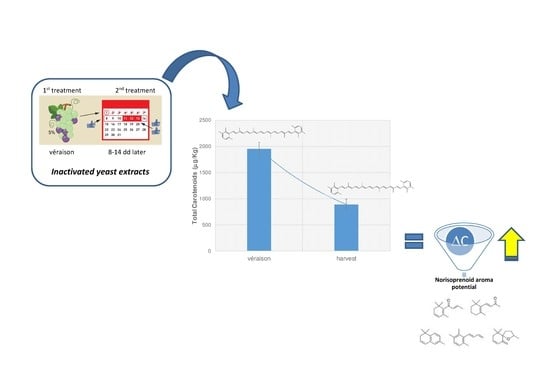
How Pre-Harvest Inactivated Yeast Treatment May Influence the Norisoprenoid Aroma Potential in Wine Grapes
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Chemicals
2.3. Maturation Indexes of Grapes
2.4. Extraction of Carotenoids from Grapes
2.5. HPLC-DAD-MS Analyses of Carotenoids
2.6. Statistical Analysis
3. Results and Discussion
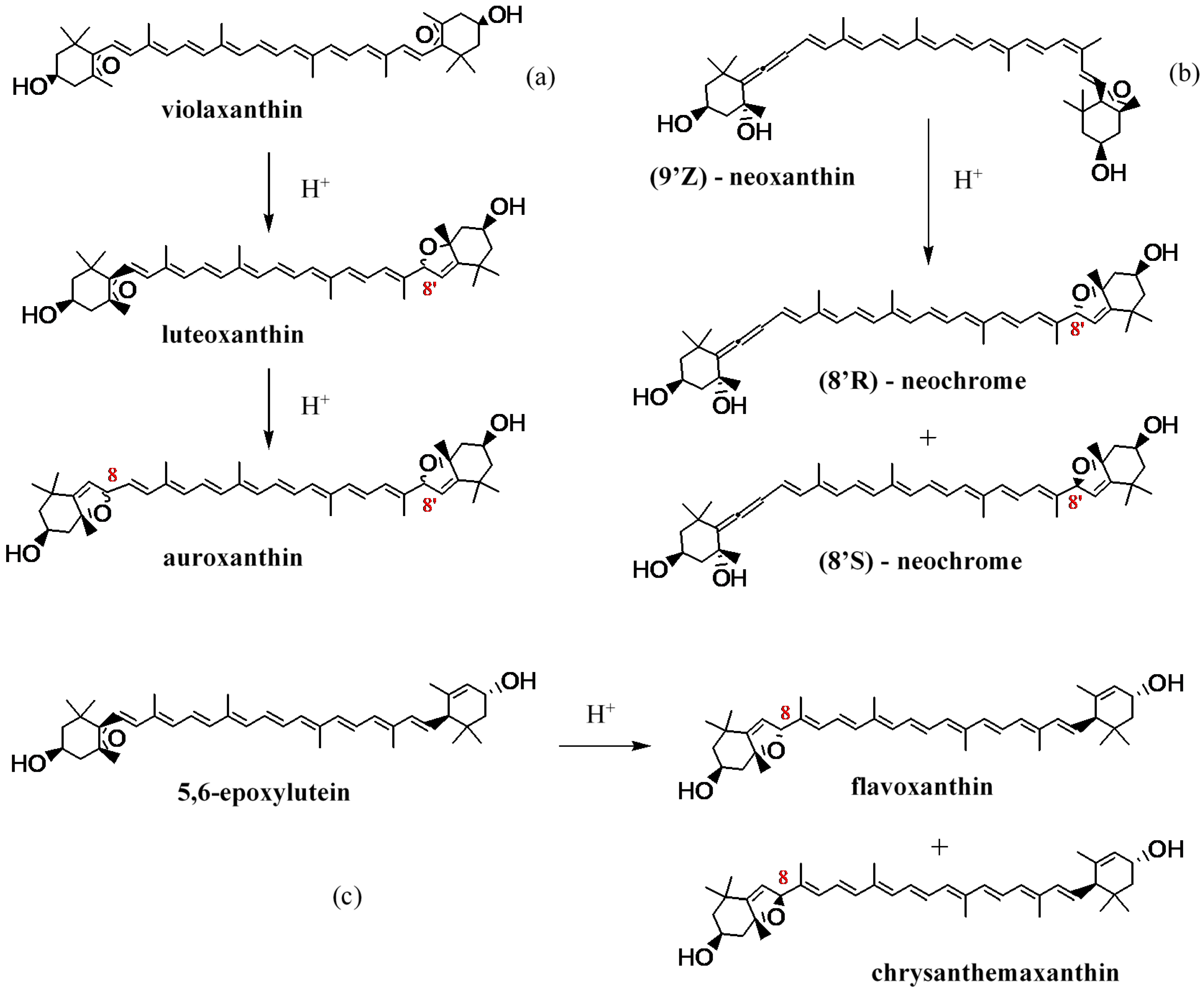
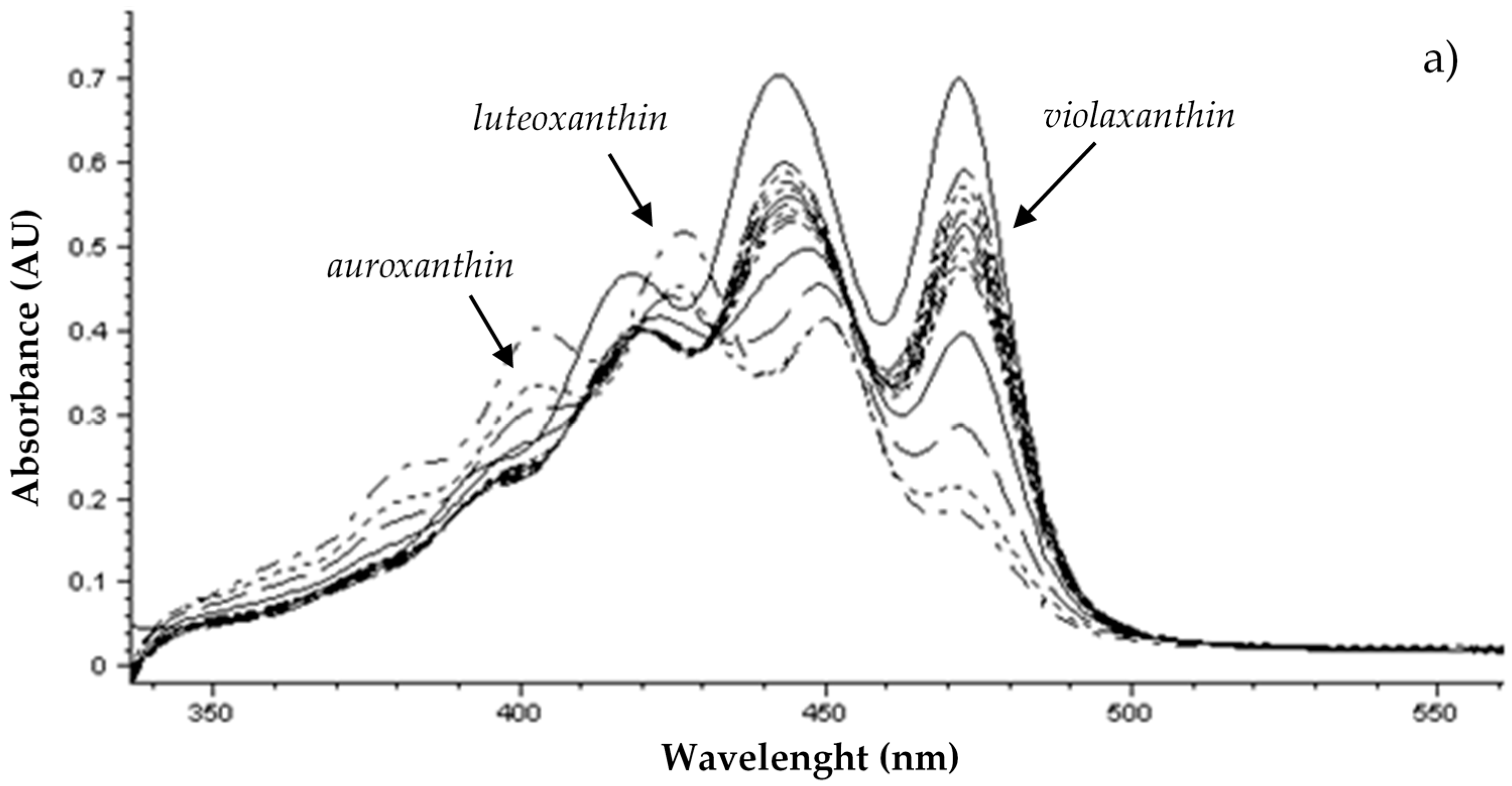
3.1. Carotenoid Composition of the Wine Grape Varieties
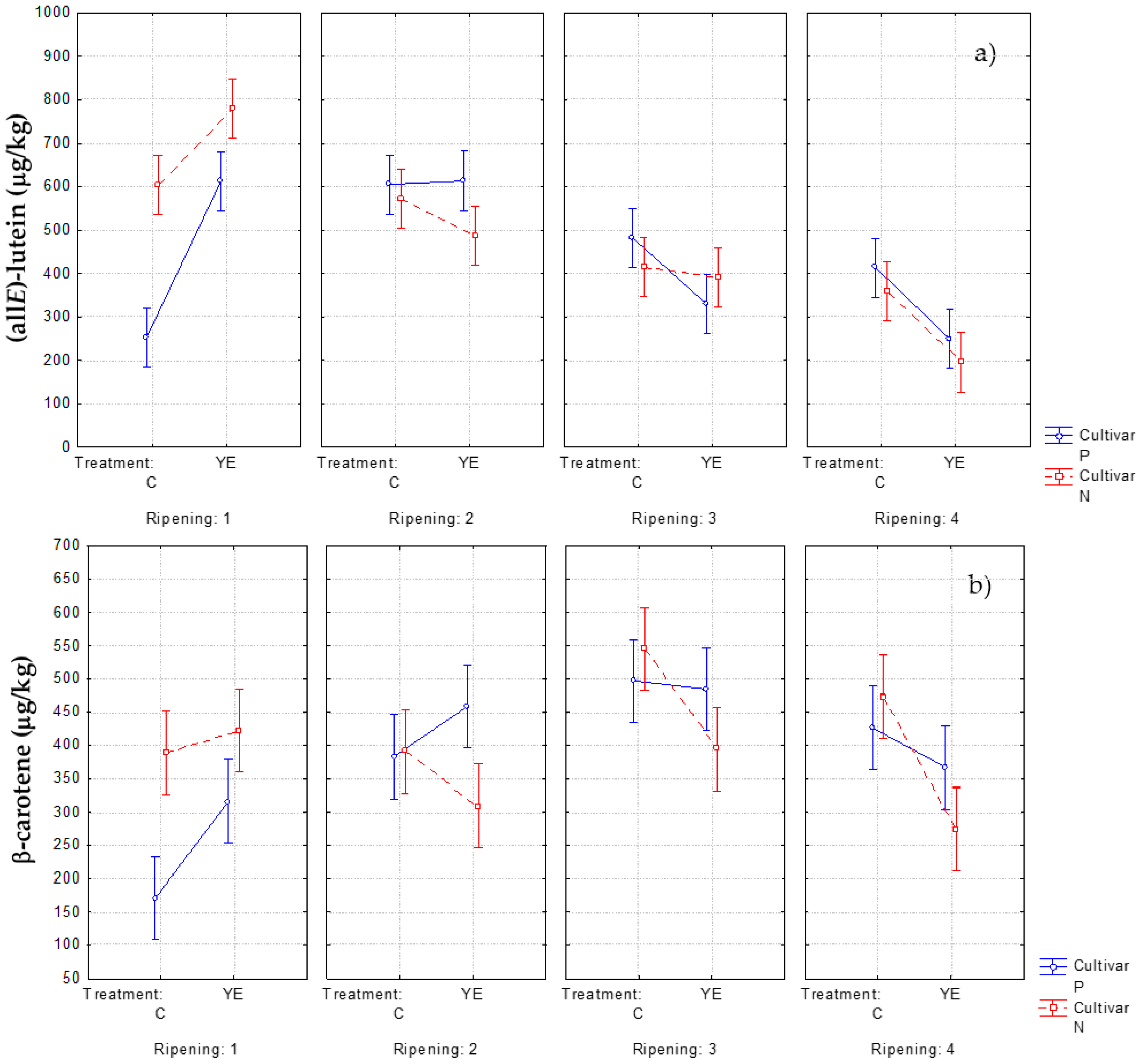
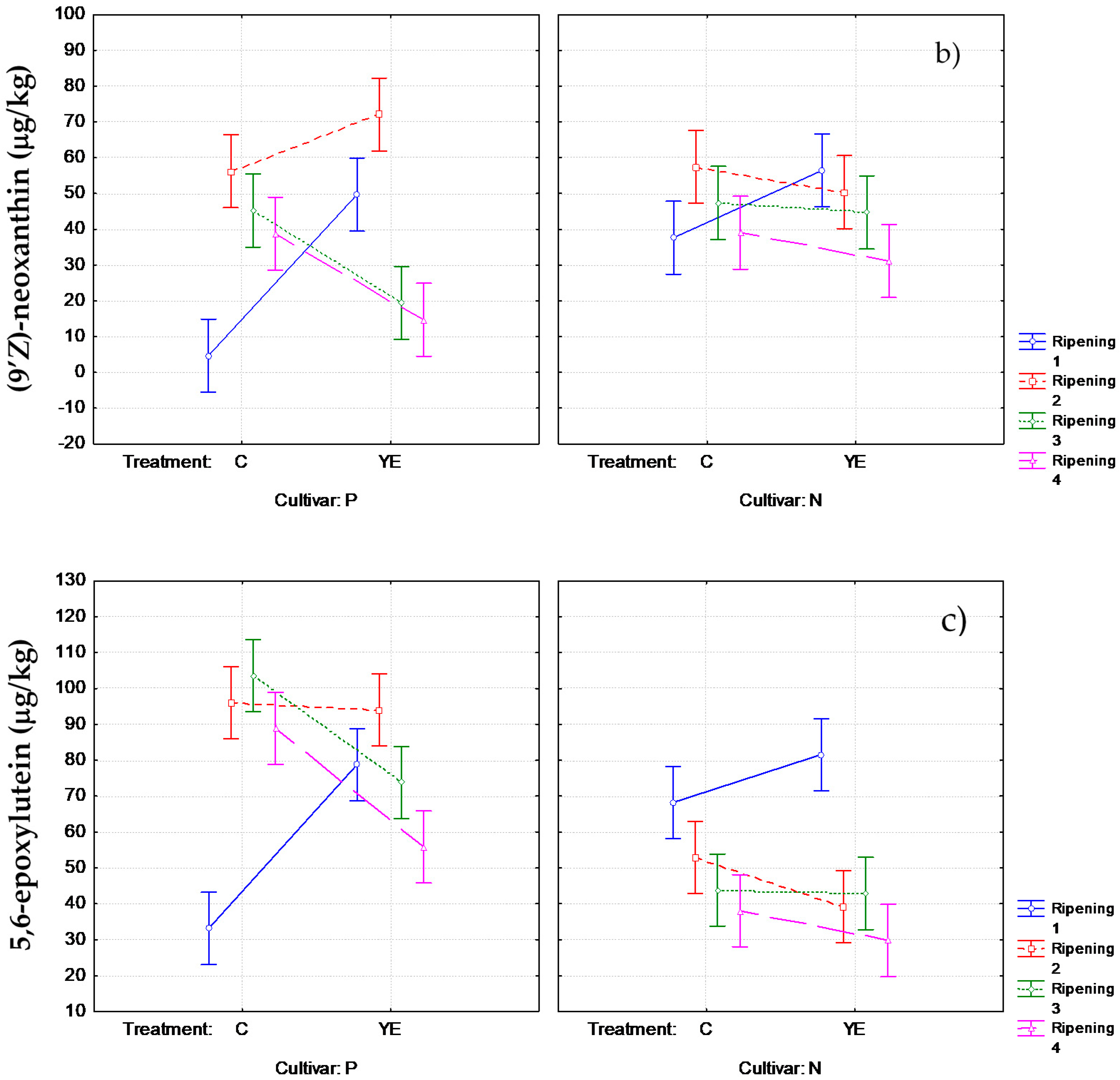
3.2. Effect of Pre-Harvest YE Application on Carotenoid Content
3.3. Influence of YE Treatment on the Norisoprenoid Aroma Potential (ΔC)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Razungles, A.; Babic, I.; Sapis, J.C.; Bayonove, C.L. Particolar behavior of epoxy xanthophylls during veraison and maturation of grape. J. Agric. Food Chem. 1996, 44, 3821–3825. [Google Scholar] [CrossRef]
- Razungles, A.; Bayonove, C.L.; Cordonnier, R.E.; Baumes, R.L. Etude des Carotenoides du Raisin à Maturitè. Vitis 1987, 26, 183–191. [Google Scholar]
- Razungles, A.; Bayonove, C.L.; Cordonnier, R.E.; Sapis, J.C. Grape carotenoides: Changes during the maturation period and localization in mature berries. Am. J. Enol. Vitic. 1988, 39, 44–48. [Google Scholar]
- Crupi, P.; Milella, R.A.; Antonacci, D. Simultaneous HPLC-DAD-MS (ESI+) determination of structural and geometrical isomers of carotenoids in mature grapes. J. Mass Spectrom. 2010, 45, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Mendes-Pinto, M.M.; Silva-Ferreira, A.C.; Caris-Veyrat, C.; Guedes de Pinho, P. Carotenoid, chlorophyll, and chlorophyll-derived compounds in grapes and port wines. J. Agric. Food Chem. 2005, 53, 10034–10041. [Google Scholar] [CrossRef] [PubMed]
- Silva-Ferreira, A.C.; Monteiro, J.; Oliveira, C.; Guedes de Pinho, P. Study of major aromatic compounds in port wines from carotenoid degradation. Food Chem. 2008, 110, 83–87. [Google Scholar] [CrossRef]
- Crupi, P.; Coletta, A.; Milella, R.A.; Palmisano, G.; Baiano, A.; La Notte, E.; Antonacci, D. Carotenoid and Chlorophyll derived compounds in some wine grapes grown in Apulian region. J. Food Sci. 2010, 75, S191–S198. [Google Scholar] [CrossRef]
- Azevedo-Meleiro, C.H.; Rodriguez-Amaya, D.B. Confirmation of the identity of the carotenoids of tropical fruits by HPLC-DAD and HPLC-MS. J. Food Compos. Anal. 2004, 17, 385–396. [Google Scholar] [CrossRef]
- Crupi, P.; Alba, V.; Masi, G.; Caputo, A.R.; Tarricone, L. Effect of two exogenous plant growth regulators on the color and quality parameters of seedless table grape berries. Food Res. Int. 2019, 126, 108667. [Google Scholar] [CrossRef]
- Baumes, R. Wine aroma precursors. In Wine chemistry and biochemistry; Moreno-Arribas, M.V., Polo, M.C., Eds.; Springer: New York, NY, USA, 2009; pp. 251–274. [Google Scholar]
- Crupi, P.; Coletta, A.; Antonacci, D. Analysis of carotenoids in grapes to predict norisoprenoid varietal aroma of wines from Apulia. J. Agric. Food Chem. 2010, 58, 9647–9656. [Google Scholar] [CrossRef]
- Baumes, R.; Wirth, J.; Bureau, S.; Gunata, Y.; Razungles, A. Biogeneration of C13-norisoprenoid compounds: Experiments supportive for an apo-carotenoid pathway in grapevines. Anal. Chim Acta 2002, 458, 3–14. [Google Scholar] [CrossRef]
- Oliveira, C.; Silva Ferreira, A.C.; Mendes Pinto, M.; Hogg, T.; Alves, F. Carotenoid compounds in grapes and their relationship to plant water status. J. Agric. Food Chem. 2003, 51, 5967–5971. [Google Scholar] [CrossRef] [PubMed]
- van Breemen, R.B. Liquid chromatography/mass spectrometry of carotenoids. Pure App. Chem. 1997, 69, 2061–2066. [Google Scholar] [CrossRef]
- Emenhiser, C.; Simunovic, N.; Sander, L.C.; Schwartz, S.J. Separation of geometrical carotenoid isomers in biological extracts using a polymeric C30 column in reversed-phase liquid chromatography. J. Agric. Food Chem. 1996, 44, 3887–3893. [Google Scholar] [CrossRef]
- Ferrari, S. Biological elicitors of plant secondary metabolites: Mode of action and use in the production of nutraceutic. In Bio-Farms for Nutraceuticals: Functional Food and Safety Control by Biosensors; Giardi, M.T., Rea, G., Berra, B., Eds.; Landes Bioscience and Springer Science + Business Media LLC: New York, NY, USA, 2010; pp. 152–166. [Google Scholar]
- Bektas, Y.; Eulgem, T. Synthetic plant defense elicitors. Front. Plant Sci. 2015, 5, 1–17. [Google Scholar] [CrossRef]
- Portu, J.; López, R.; Baroja, E.; Santamaría, P.; Garde-Cerdán, T. Improvement of grape and wine phenolic content by foliar application to grapevine of three different elicitors: Methyl jasmonate, chitosan, and yeast extract. Food Chem. 2016, 201, 213–221. [Google Scholar] [CrossRef]
- Šuklje, K.; Antalick, G.; Buica, A.; Coetzee, Z.A.; Brand, J.; Schmidtke, L.M.; Vivier, M.A. Inactive dry yeast application on grapes modify Sauvignon Blanc wine aroma. Food Chem. 2016, 197, 1073–1084. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Marín-San Román, S.; Jofré, V.; Rubio-Bretón, P.; Pérez-Álvarez, E.P.; Garde-Cerdán, T. Effects on chlorophyll and carotenoid contents in different grape varieties (Vitis vinifera L.) after nitrogen and elicitor foliar applications to the vineyard. Food Chem. 2018, 269, 380–386. [Google Scholar] [CrossRef]
- Betz, M.; Schindler, C.; Schwender, J.; Lichtenthaler, H.K. Jasmonic acid induced changes in carotenoid levels and zeaxanthin cycle performance. In Plant Lipid Metabolism; Kader, J.C., Mazliak, P., Eds.; Springer: Dordrecht, The Netherlands, 1995; pp. 353–355. [Google Scholar]
- Lu, Y.; Jiang, P.; Liu, S.; Gan, Q.; Cui, H.; Quin, S. Methyl jasmonate- or gibberellins A3-induced astaxanthin accumulation is associated with up-regulation of transcription of β-carotene ketolase genes (bkts) in microalga Haematococcus pluvialis. Bioresour. Technol. 2010, 101, 6468–6474. [Google Scholar] [CrossRef]
- Office International de la Vigne et du Vin (OIV). Recueil des Methodes Internationales d’Analyse des Vins et des Mouts; Office International de la Vigne et du Vin: Paris, France, 1990. [Google Scholar]
- Melendez-Martinez, A.J.; Britton, G.; Vicario, I.M.; Heredia, F.J. HPLC analysis of geometrical isomers of lutein epoxide isolated from dandelion (Taraxacum officinale F.Weber exWiggers). Phytochemistry 2006, 67, 771–777. [Google Scholar] [CrossRef]
- Martì, M.P.; Busto, O.; Guash, J. Application of a headspace mass spectrometry system to the differentiation and classification of wines according to their origin, variety and ageing. J. Chromatogr. A 2004, 1057, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.; Palou, A. Chromatographic determination of carotenoids in foods. J. Chromatogr. A 2000, 881, 543–555. [Google Scholar] [CrossRef]
- Bureau, S.M.; Razungles, A.J.; Baumes, R.L.; Bayonove, C.L. Effect of vine or bunch shading on the carotenoid composition in Vitis Vinifera L. berries. I. Syrah grapes. Vitic. Enol. Sci. 1998, 53, 64–71. [Google Scholar]
- González-Gómez, D.; Lozano, M.; Fernández-León, M.F.; Bernalte, M.J.; Ayuso, M.C.; Rodríguez, A.B. Sweet cherry phytochemicals: Identification and characterization by HPLC-DAD/ESI-MS in six sweet cherry cultivars grown in Valle del Jerte (Spain). J. Food Compos. Anal. 2010, 23, 533–539. [Google Scholar] [CrossRef]
- Oliveira, C.; Barbosa, A.; Silva Ferreira, A.C.; Guerra, J.; Guedes De Pinho, P. Carotenoid profile in grapes related to aromatic compounds in wines from Douro region. J. Food Sci. 2006, 71, S1–S7. [Google Scholar] [CrossRef]
- Gómez-Plaza, E.; Mestre-Ortuño, L.; Ruiz García, Y.; Fernández-Fernández, J.I.; López-Roca, J.M. Effect of benzothiadiazole and methyl jasmonate on the volatile compound composition of Vitis vinifera L. Monastrell grapes and wines. Am. J. Enol. Vitic. 2012, 63, 394–401. [Google Scholar] [CrossRef]
- Lucini, L.; Baccolo, G.; Rouphael, Y.; Colla, G.; Bavaresco, L.; Trevisan, M. Chitosan treatment elicited defence mechanisms, pentacyclic triterpenoids and stilbene accumulation in grape (Vitis vinifera L.) bunches. Phytochemistry 2018, 156, 1–8. [Google Scholar] [CrossRef]
- Kogkou, C.; Chorti, E.; Kyraleou, M.; Kallithraka, S.; Koundouras, S.; Logan, G.; Kanakis, I.; Kotseridis, Y. Effects of foliar application of inactivated yeast on the phenolic composition of Vitis vinifera L. cv. Agiorgitiko grapes under different irrigation level. Int. J. Wine Res. 2017, 9, 23–33. [Google Scholar] [CrossRef]
- Paladines-Quezada, D.F.; Fernández-Fernández, J.I.; Bautista-Ortín, A.B.; Gómez-Plaza, E.; Bleda-Sánchez, J.A.; Gil-Muñoz, R. Influence of the use of elicitors over the composition of cell wall grapes. In Proceedings of the In Vino Analytica Scientia 2017, Salamanca, Spain, 17–20 July 2017. [Google Scholar]
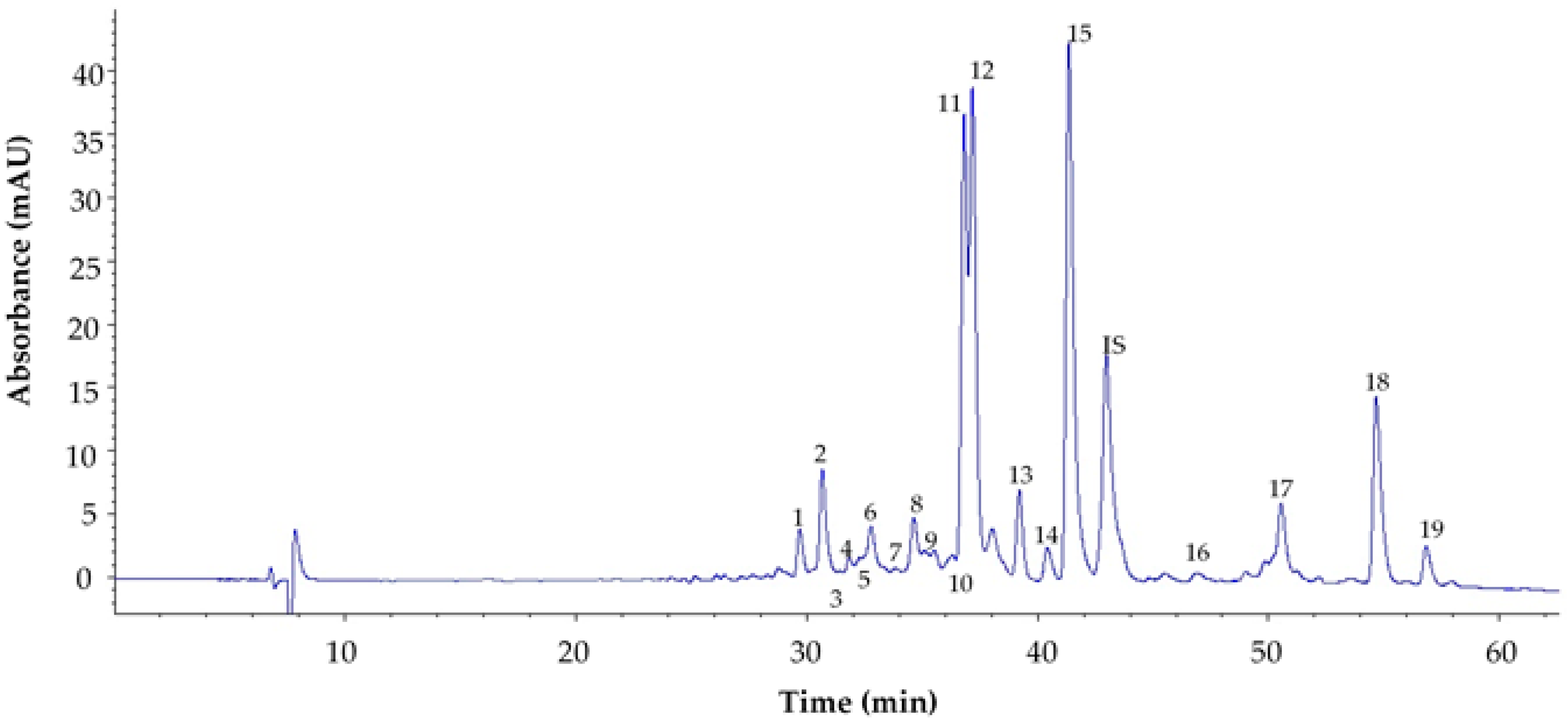




| Peak | Compound | k’ | λmax (nm) | % (III/II)a | DB/DIIb | [M+H]+(m/z) | [M]·+(m/z) | MS2 Product Ions m/z |
|---|---|---|---|---|---|---|---|---|
| 1 | violaxanthin like structure | 2.78 | 418;440;470 | 84 | 601.5 | 600.1 | 583.5, 565.5, 509.5, 491.5, 221.1 | |
| 2 | violaxanthin | 2.92 | 416; 440; 468 | 86 | 601.5 | 600.1 | 583.5, 565.5, 509.5, 491.5, 221.1 | |
| 3 | (8′R)-neochrome | 3.01 | 400; 422; 450 | 88 | 601.5 | 600.1 | 583.2, 565.3, 509.5, 221.1 | |
| 4 | (9′Z)-neoxanthin | 3.06 | 414; 436; 464 | 86 | 601.5 | 600.1 | 583.2, 565.3, 509.5, 221.1 | |
| 5 | (8′S)-neochrome | 3.12 | 400; 422; 450 | 88 | 601.5 | 600.1 | 583.2, 565.3, 509.5, 221.1 | |
| 6 | 5,6-epoxylutein | 3.19 | 416; 440; 468 | 90 | 585.4 | 584.2 | 567.1, 493.1, 221.1 | |
| 7 | luteoxanthin | 3.33 | 399; 422; 448 | 94 | 601.5 | 600.1 | 583.2, 221.1 | |
| 8 | lutein like structure | 3.42 | (425); 446; 474 | 568.9 | 567.9 | 550.9, 532.9, 476.4, 429.4 | ||
| 9 | Z lutein like structure | 3.47 | 328; (412); 436; 464 | 568.9 | 567.9 | 550.9, 532.9, 476.4, 429.4 | ||
| 10 | (8′S)-auroxanthin | 3.64 | 380; 402; 426 | 98 | 601.5 | 600.1 | 583.5, 565.5, 509.5, 491.5, 221.1 | |
| 11 | chlorophyll b | 3.70 | 258; 314; 342; 466; 600; 650 | |||||
| 12 | (allE)-lutein | 3.75 | (422); 446; 472 | 40 | 568.9 | 567.9 | 550.9, 532.9, 476.4, 429.4 | |
| 13 | zeaxanthin | 4.00 | (425); 452; 476 | 22 | 568.9 | 567.9 | 550.9, 532.9, 476.4, 429.4 | |
| 14 | (9Z) or (9’Z)-lutein | 4.16 | 330; (422); 440; 468 | 50 | 0.075 | 568.9 | 567.9 | 550.9, 532.9, 476.4, 429.4 |
| 15 | chlorophyll a | 4.31 | 336; (385); (417); 432; 618; 665 | |||||
| IS | β-apo-8′-carotenal | 4.50 | 460 | |||||
| 16 | pheophytin b | 5.46 | (417); 436; 527; 600; 654 | 885 | ||||
| 17 | pheophytin a | 5.59 | 410; 506; 536; 666 | 871 | ||||
| 18 | β-carotene | 5.98 | (430); 452; 478 | 25 | 536.9 | 535.9 | 444.2, 430.3, 399.3 | |
| 19 | (9Z)- β-carotene | 6.25 | 342; (424); 446; 474 | 17 | 0.03 | 536.9 | 535.9 | 444.2, 430.3, 399.3 |
| Sampling Date | 08/13/19* | 08/26/19 | 09/02/19 | 09/17/19 | ΔCa | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C | YE | C | YE | C | YE | C | YE | C | YE | ||
| TSSb | 153.0c ± 1.7d | 142.3 ± 1.2 | 165 ± 2 | 163 ± 2 | 171.4 ± 1.4 | 189 ± 1.0 | 212 ± 3 | 220 ± 3 | |||
| pH | 3.11 ± 0.01 | 3.17 ± 0.01 | 3.10 ± 0.02 | 3.26 ± 0.01 | 3.31 ± 0.06 | 3.38 ± 0.04 | 3.36 ± 0.04 | 3.43 ± 0.06 | |||
| TAe | 8.47 ± 0.15 | 7.78 ± 0.16 | 6.25 ± 0.18 | 5.82 ± 0.10 | 5.63 ± 0.07 | 6.6 ± 0.4 | 5.8 ± 0.3 | 5.4 ± 0.4 | |||
| TSS/TAf | 18.1 ± 0.4 | 18.3 ± 0.5 | 26.4 ± 1.1 | 27.9 ± 0.4 | 30.4 ± 0.2 | 29 ± 3 | 37 ± 2 | 40 ± 3 | |||
| Compounds | |||||||||||
| violaxanthin like structureg | 30 ± 5ch | 64 ± 15a | 49 ± 5ab | 34 ± 5bc | 39 ± 6bc | 42 ± 7bc | 34 ± 5bc | 29 ± 4c | |||
| violaxanthing | 96 ± 10b | 126 ± 14a | 80 ± 12bc | 67 ± 10c | 91 ± 14bc | 97 ± 15b | 79 ± 12bc | 68 ± 9c | |||
| (8′R)-neochromei | 8.2 ± 0.6a | 9.5 ± 0.3a | 8.5 ± 0.8a | 4.8 ± 1.0b | 3.2 ± 0.6c | 4.2 ± 0.4bc | 2.8 ± 0.5c | 2.95 ± 0.17c | |||
| (9′Z)-neoxanthini | 38 ± 10ab | 56 ± 10a | 58 ± 12a | 50 ± 8ab | 47 ± 13ab | 45 ± 6ab | 39 ± 7ab | 31 ± 3b | |||
| (8′S)-neochromei | 6.0 ± 0.9b | 9 ± 2a | 7.3 ± 1.3ab | 6.2 ± 1.1bc | 4.6 ± 0.7c | 5.5 ± 0.8bc | 4.0 ± 0.6c | 3.8 ± 0.5c | |||
| 5,6-epoxy-luteinl | 68 ± 5a | 82 ± 8a | 53 ± 9b | 39 ± 6bc | 44 ± 7bc | 43 ± 6bc | 38 ± 6bc | 30 ± 3c | |||
| luteoxanthing | 8.8 ± 1.2b | 14 ± 2a | 8.2 ± 1.0bc | 7.6 ± 1.5bc | 9.0 ± 1.0b | 5.8 ± 0.8c | 7.8 ± 0.9bc | tr | |||
| lutein like structure m | 44 ± 7b | 62 ± 5a | 34 ± 5bc | 32 ± 5bcd | 29 ± 6cd | 29 ± 7cd | 25 ± 6cd | 20 ± 4d | |||
| Z lutein like structure m | 9.8 ± 0.8b | 14.9 ± 1.8a | tr | tr | 5.9 ± 1.2c | 4.1 ± 1.8cd | 5.1 ± 1.1cd | 2.8 ± 1.2d | |||
| (8′S)-auroxanthing | 54 ± 7a | 68 ± 15a | 29 ± 5b | 10 ± 3c | 19 ± 8bc | 18 ± 3bc | 15 ± 5bc | 12 ± 2bc | |||
| (all-E)-luteinm | 600 ± 90b | 780 ± 60a | 570 ± 90bc | 490 ± 90bcd | 410 ± 60cd | 390 ± 70d | 360 ± 60de | 200 ± 20e | |||
| zeaxanthinn | 88 ± 4 | 101 ± 5 | 65 ± 9 | 65 ± 15 | 30 ± 5 | 28 ± 4 | 26 ± 5 | 20 ± 2 | |||
| (9Z)-luteinm | 17 ± 5c | 33 ± 5a | 30 ± 6ab | 20 ± 5bc | 10 ± 3d | 11 ± 3cd | 9 ± 2d | 7 ± 2d | |||
| β-caroteneo | 390 ± 60bc | 420 ± 30abc | 390 ± 60bc | 310 ± 60c | 550 ± 90a | 390 ± 60bc | 470 ± 80ab | 270 ± 40c | |||
| (9Z)-β-caroteneo | 130 ± 50 | 110 ± 20 | 180 ± 50 | 150 ± 40 | 230 ± 20 | 167 ± 19 | 200 ± 20 | 116 ± 8 | |||
| Totalp | 1600 ± 170ab | 1950 ± 90a | 1600 ± 200ab | 1280 ± 180b | 1500 ± 200b | 1280 ± 180b | 1320 ± 170b | 820 ± 30c | 270 ± 300 | 1130 ± 120 | |
| Sampling Date | 08/13/19 | 08/26/19* | 09/04/19 | 09/19/19 | ΔCa | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C | YE | C | YE | C | YE | C | YE | C | YE | ||
| TSSb | 175c ± 1.0d | 170 ± 2 | 187 ± 1.5 | 200 ± 2 | 226 ± 4 | 227 ± 14 | 236 ± 5 | 239 ± 15 | |||
| pH | 2.98 ± 0.02 | 2.97 ± 0.03 | 3.19 ± 0.02 | 3.22 ± 0.02 | 3.40 ± 0.01 | 3.35 ± 0.08 | 3.45 ± 0.01 | 3.40 ± 0.07 | |||
| TAe | 11.2 ± 0.4 | 13.1 ± 0.6 | 6.8 ± 0.3 | 7.03 ± 0.15 | 6.2 ± 0.4 | 6.7 ± 0.3 | 5.7 ± 0.4 | 6.1 ± 0.4 | |||
| TSS/TAf | 15.7 ± 0.5 | 12.8 ± 0.4 | 27.8 ± 1.3 | 28. 8 ± 0.7 | 36.3 ± 1.9 | 34 ± 3 | 41 ± 2 | 39 ± 3 | |||
| Compounds | |||||||||||
| violaxanthin like structureg | 5 ± 2dh | 21 ± 6bc | 39 ± 9a | 25 ± 7abc | 35 ± 7ab | 22 ± 6bc | 30 ± 6abc | 17 ± 5cd | |||
| violaxanthing | 31 ± 19d | 144 ± 6ab | 160 ± 30a | 129 ± 9ab | 154 ± 18ab | 115 ± 17bc | 132 ± 15ab | 87 ± 12c | |||
| (8′R)-neochromei | tr | tr | tr | 3.2 ± 0.5 | tr | tr | tr | tr | |||
| (9′Z)-neoxanthini | 4.7 ± 1.4d | 50 ± 15ab | 56 ± 15ab | 72 ± 17a | 45 ± 11b | 19 ± 5cd | 39 ± 9bc | 15 ± 4cd | |||
| (8′S)-neochromei | 10 ± 3a | 3.4 ± 1.6b | 2.3 ± 0.6b | 5.3 ± 1.3b | 3.5 ± 1.2b | 3.3 ± 0.7b | 3.0 ± 1.0b | 2.5 ± 0.5b | |||
| 5,6-epoxy-luteinl | 33 ± 13d | 79 ± 9abc | 100 ± 20ab | 94 ± 14ab | 104 ± 10a | 74 ± 7bc | 89 ± 8ab | 56 ± 5cd | |||
| luteoxanthing | 6 ± 2 | tr | tr | tr | tr | tr | tr | tr | |||
| lutein like structure m | 18 ± 12d | 86 ± 12a | 74 ± 13ab | 69 ± 8ab | 61 ± 10b | 36 ± 4cd | 52 ± 9bc | 28 ± 3d | |||
| Z lutein like structure m | tr | 19 ± 2ab | 25 ± 2a | 18 ± 4ab | 24 ± 5a | 18 ± 4ab | 21 ± 4ab | 14 ± 3b | |||
| (8′S)-auroxanthing | 16 ± 3a | 11 ± 2abc | 10 ± 5abc | 15 ± 5ab | 12.3 ± 1.3abc | 8.1 ± 1.5bc | 10.5 ± 1.1abc | 6.1 ± 1.1c | |||
| (all-E)-luteinm | 250 ± 110d | 610 ± 90a | 610 ± 80a | 610 ± 70a | 480 ± 40ab | 329 ± 19cd | 410 ± 30bc | 249 ± 14d | |||
| zeaxanthinn | 30 ± 20c | 110 ± 20a | 68 ± 16b | 64 ± 6b | 39 ± 12bc | 18 ± 3c | 33 ± 10c | 13 ± 2c | |||
| (9Z)-luteinm | 9 ± 7bc | 27 ± 4a | 26 ± 6a | 20 ± 3ab | 15 ± 6bc | 7 ± 5c | 13 ± 5bc | 5 ± 4c | |||
| β-caroteneo | 170 ± 110c | 320 ± 40b | 380 ± 80ab | 460 ± 70ab | 497 ± 19a | 480 ± 40ab | 426 ± 17ab | 370 ± 30ab | |||
| (9Z)-β-caroteneo | 69 ± 4 | 51 ± 6 | 57 ± 10 | 52 ± 11 | 48 ± 6 | 51 ± 11 | 41 ± 5 | 39 ± 9 | |||
| Totalp | 700 ± 300d | 1500 ± 180ab | 1600 ± 200a | 1640 ± 120a | 1520 ± 80ab | 1190 ± 80bc | 1310 ± 70ab | 900 ± 60cd | 290 ± 300 | 740 ± 180 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crupi, P.; Santamaria, M.; Vallejo, F.; Tomás-Barberán, F.A.; Masi, G.; Caputo, A.R.; Battista, F.; Tarricone, L. How Pre-Harvest Inactivated Yeast Treatment May Influence the Norisoprenoid Aroma Potential in Wine Grapes. Appl. Sci. 2020, 10, 3369. https://doi.org/10.3390/app10103369
Crupi P, Santamaria M, Vallejo F, Tomás-Barberán FA, Masi G, Caputo AR, Battista F, Tarricone L. How Pre-Harvest Inactivated Yeast Treatment May Influence the Norisoprenoid Aroma Potential in Wine Grapes. Applied Sciences. 2020; 10(10):3369. https://doi.org/10.3390/app10103369
Chicago/Turabian StyleCrupi, Pasquale, Marika Santamaria, Fernando Vallejo, Francisco A. Tomás-Barberán, Gianvito Masi, Angelo Raffaele Caputo, Fabrizio Battista, and Luigi Tarricone. 2020. "How Pre-Harvest Inactivated Yeast Treatment May Influence the Norisoprenoid Aroma Potential in Wine Grapes" Applied Sciences 10, no. 10: 3369. https://doi.org/10.3390/app10103369
APA StyleCrupi, P., Santamaria, M., Vallejo, F., Tomás-Barberán, F. A., Masi, G., Caputo, A. R., Battista, F., & Tarricone, L. (2020). How Pre-Harvest Inactivated Yeast Treatment May Influence the Norisoprenoid Aroma Potential in Wine Grapes. Applied Sciences, 10(10), 3369. https://doi.org/10.3390/app10103369