High-Frequency Deep Sclerotomy, A Minimal Invasive Ab Interno Glaucoma Procedure Combined with Cataract Surgery: Physical Properties and Clinical Outcome
Abstract
1. Introduction
2. Materials and Methods
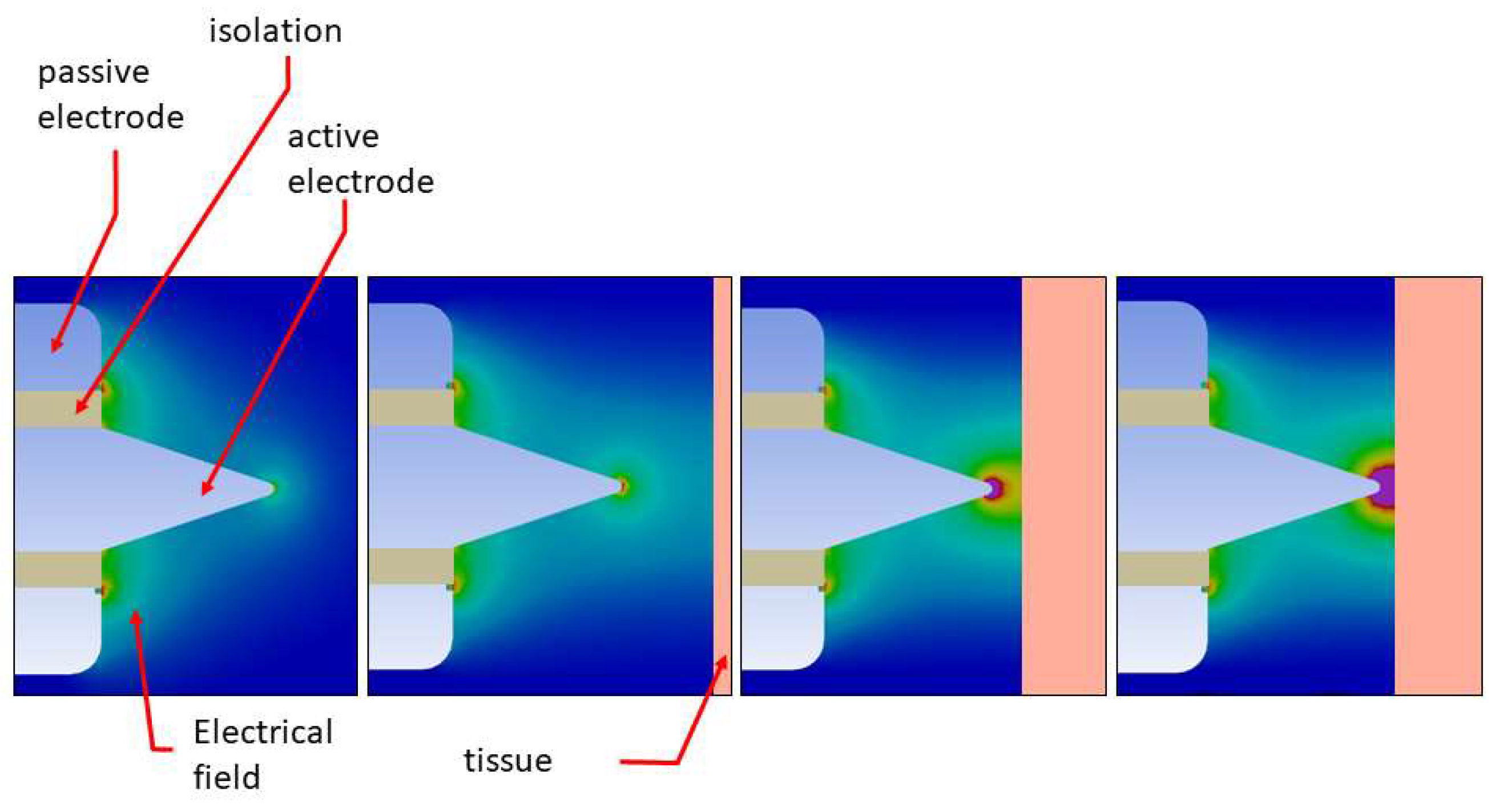
High-Frequency Diathermic Probe
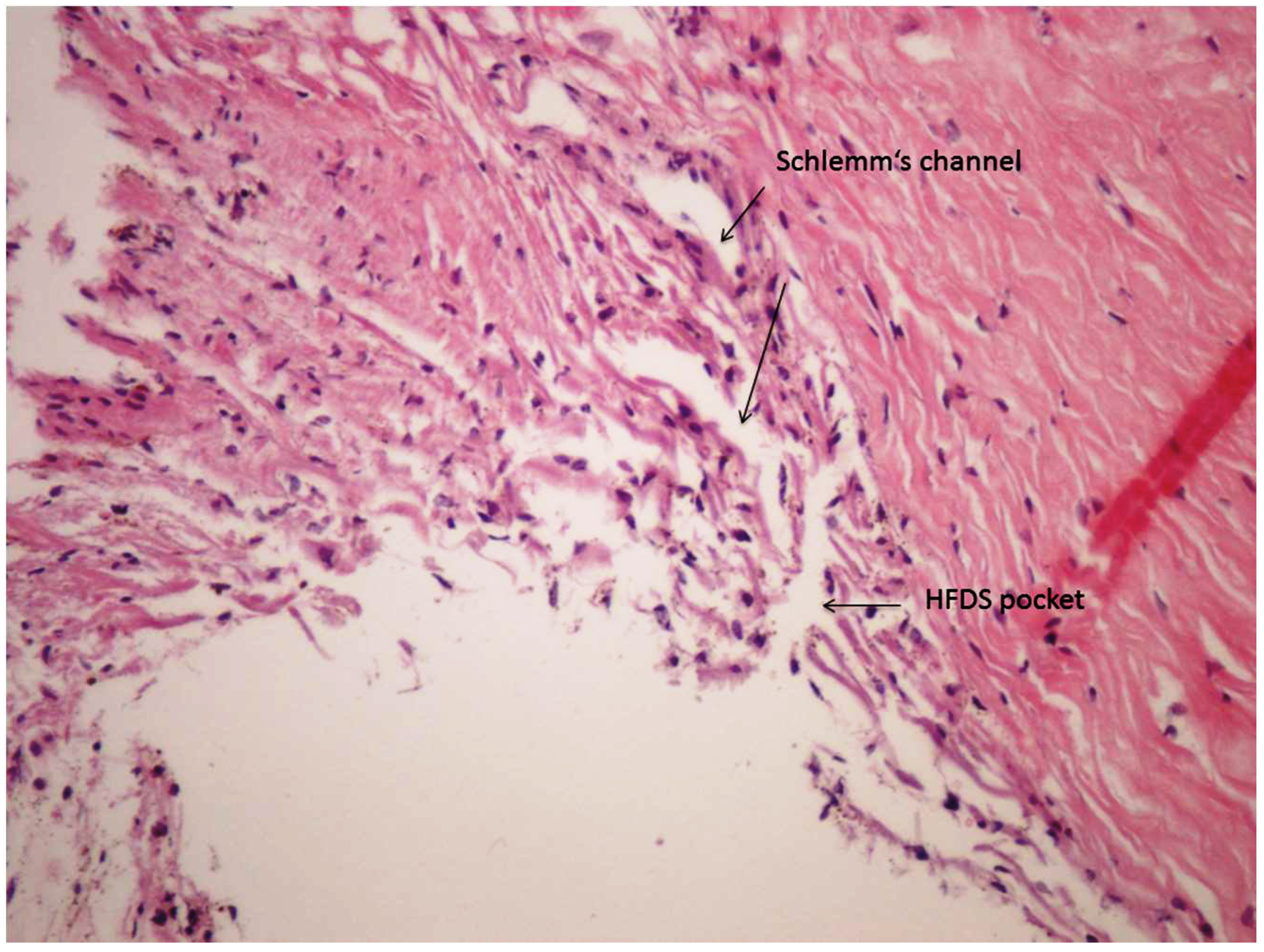
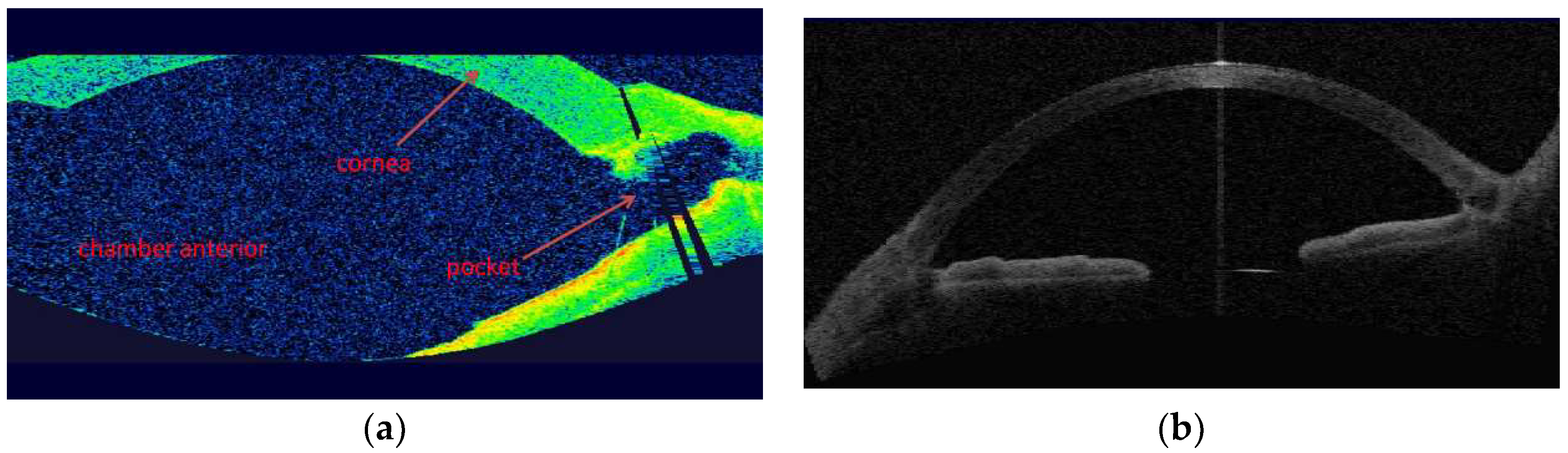
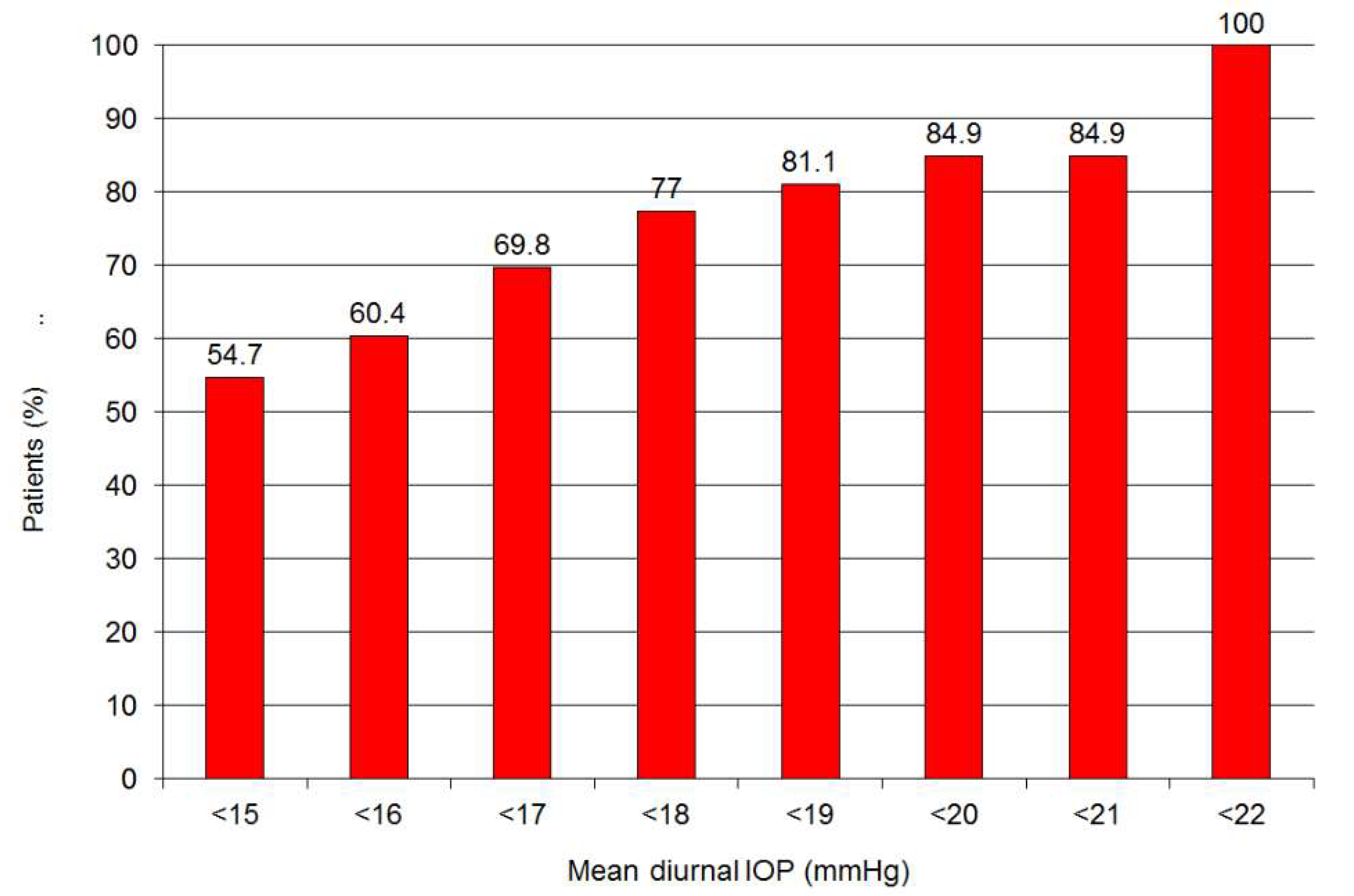
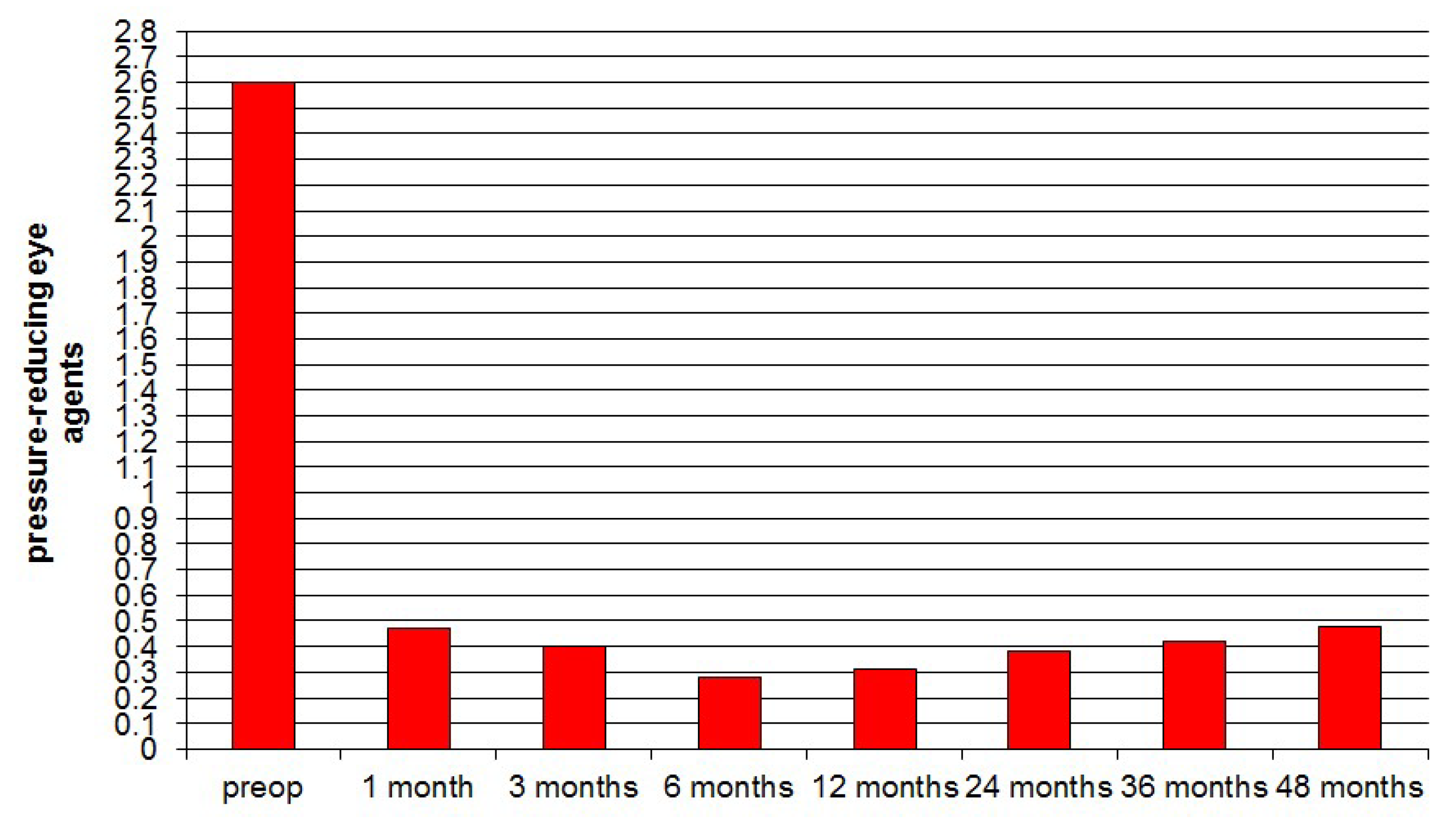
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Flaxman, S.R.; Bourne, R.R.A.; Resnikoff, S.; Ackland, P.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, 1221–1234. [Google Scholar] [CrossRef]
- Goh, Y.W.; Ang, G.S.; Azuara-Blanco, A. Lifetime visual prognosis of patients with glaucoma. Clin. Exp. Ophthalmol. 2011, 39, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Razeghinejad, M.R.; Fudemberg, S.J.; Spaeth, G.L. The changing conceptual basis of trabeculectomy: A review of past and current surgical techniques. Surv. Ophthalmol. 2012, 57, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Vinod, K.; Gedde, S.J.; Feuer, W.J.; Panarelli, J.F.; Chang, T.C.; Chen, P.P.; Parrish, R.K. Practice preferences for glaucoma surgery: A survey of the American Glaucoma Society. J. Glaucoma 2017, 26, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Gedde, S.J.; Chen, P.P.; Heuer, D.K.; Singh, K.; Wright, M.M.; Feuer, W.J.; Schiffman, J.C.; Shi, W.; Primary Tube Versus Trabeculectomy Study Group. The Primary Tube Versus Trabeculectomy Study: Methodology of a Multicenter Randomized Clinical Trial Comparing Tube Shunt Surgery and Trabeculectomy with Mitomycin, C. Ophthalmology 2018, 125, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, K.; Schwartz, S.G.; Kishor, K.S.; Fortun, J.A.; Moshfeghi, D.M.; Moshfeghi, A.A.; Flynn, H.W., Jr. Incidence of postoperative suprachoroidal hemorrhage after glaucoma filtration surgeries in the United States. Clin. Ophthalmol. 2014, 9, 579–584. [Google Scholar]
- Takihara, Y.; Inatani, M.; Ogata-Iwao, M.; Kawai, M.; Inoue, T.; Iwao, K.; Tanihara, H. Trabeculectomy for open-angle glaucoma in phakic eyes vs in pseudophakic eyes after phacoemulsification: A prospective clinical cohort study. JAMA Ophthalmol. 2014, 132, 69–76. [Google Scholar] [CrossRef]
- Jeganathan, V.S.; Ghosh, S.; Ruddle, J.B.; Gupta, V.; Coote, M.A.; Crowston, J.G. Risk factors for delayed suprachoroidal haemorrhage following glaucoma surgery. Br. J. Ophthalmol. 2008, 92, 1393–1396. [Google Scholar] [CrossRef]
- Gedde, S.J.; Herndon, L.W.; Brandt, J.D.; Budenz, D.L.; Feuer, W.J.; Schiffman, J.C. Surgical complications in the Tube Versus Trabeculectomy Study during the first year of follow-up. Am. J. Ophthalmol. 2007, 143, 23–31. [Google Scholar] [CrossRef]
- Johnson, D.H.; Johnson, M. Glaucoma surgery and aqueous outflow: How does nonpenetrating glaucoma surgery work? Arch. Ophthalmol. 2002, 120, 67–70. [Google Scholar] [CrossRef]
- Schuman, J.S.; Chang, W.; Wang, N.; de Kater, A.W.; Allingham, R.R. Excimer laser effects on outflow facility and outflow pathway morphology. Investig. Ophthalmol. Vis. Sci. 1999, 40, 1676–1680. [Google Scholar]
- Shaarawy, T. Glaucoma surgery: Taking the sub-conjunctival route. Middle East Afr. J. Ophthalmol. 2015, 22, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Watanabe, M.; Ichikawa, K. Evaluation of trabectome in open-angle glaucoma. J. Glaucoma 2013, 22, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Minckler, D.S.; Baerveldt, G.; Alfaro, M.R.; Francis, B.A. Clinical results with the Trabectome for treatment of open-angle glaucoma. Ophthalmology 2005, 112, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Resende, A.F.; Patel, N.S.; Waisbourd, M.; Katz, L.J. iStent® Trabecular Microbypass Stent: An Update. J. Ophthalmol. 2016, 2016, 2731856. [Google Scholar] [CrossRef]
- Pfeiffer, N.; Garcia-Feijoo, J.; Martinez-de-la-Casa, J.M.; Larrosa, J.M.; Fea, A.; Lemij, H.; Gandolfi, S.; Schwenn, O.; Lorenz, K.; Samuelson, T.W. A Randomized Trial of a Schlemm’s Canal Microstent with Phacoemulsification for Reducing Intraocular Pressure in Open-Angle Glaucoma. Ophthalmology 2015, 122, 1283–1293. [Google Scholar] [CrossRef]
- Pajic, B.; Pajic-Eggspuehler, B.; Haefliger, I. New minimally invasive, deep sclerotomy ab interno surgical procedure for glaucoma, six years of follow-up. J. Glaucoma 2011, 20, 109–114. [Google Scholar] [CrossRef]
- Abushanab, M.M.I.; El-Shiaty, A.; El-Beltagi, T.; Hassan Salah, S. The Efficacy and Safety of High-Frequency Deep Sclerotomy in Treatment of Chronic Open-Angle Glaucoma Patients. BioMed Res. Int. 2019, 2019, 1850141. [Google Scholar] [CrossRef]
- Pillunat, L.E.; Erb, C.; Jünemann, A.G.; Kimmich, F. Micro-invasive glaucoma surgery (MIGS): A review of surgical procedures using stents. Clin. Ophthalmol. 2017, 11, 1583–1600. [Google Scholar] [CrossRef]
- Edmunds, B.; Thompson, J.R.; Salmon, J.F.; Wormald, R.P. The National Survey of Trabeculectomy. III. Early and late complications. Eye 2002, 16, 297–303. [Google Scholar] [CrossRef]
- Lindemann, F.; Plange, N.; Kuerten, D.; Schimitzek, H.; Koutsonas, A. Three-Year Follow-Up of Trabeculectomy with 5-Fluorouracil. Ophthalmic Res. 2017, 58, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Una, I.; Rotchford, A.P.; King, A.J. Outcome of repeat trabeculectomies: Long-term follow-up. Br. J. Ophthalmol. 2017, 101, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Harju, M.; Suominen, S.; Allinen, P.; Vesti, E. Long-term results of deep sclerectomy in normal-tension glaucoma. Acta Ophthalmol. 2018, 96, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Bettin, P.; Di Matteo, F.; Rabiolo, A.; Fiori, M.; Ciampi, C.; Bandello, F. Deep Sclerectomy with Mitomycin C and Injectable Cross-linked Hyaluronic Acid Implant: Long-term Results. J. Glaucoma 2016, 25, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Pajic, B.; Pallas, G.; Gerding, H.; Böhnke, M. A novel technique of ab interno glaucoma surgery: Follow-up results after 24 months. Graefes Arch. Clin. Exp. Ophthalmol. 2006, 244, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Seibold, L.K.; Soohoo, J.R.; Ammar, D.A.; Kahook, M.Y. Preclinical investigation of ab interno trabeculectomy using a novel dual-blade device. Am. J. Ophthalmol. 2013, 155, 524–529. [Google Scholar] [CrossRef]
- SooHoo, J.R.; Seibold, L.K.; Kahook, M.Y. Ab interno trabeculectomy in the adult patient. Middle East Afr. J. Ophthalmol. 2015, 22, 25–29. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pajic, B.; Cvejic, Z.; Mansouri, K.; Resan, M.; Allemann, R. High-Frequency Deep Sclerotomy, A Minimal Invasive Ab Interno Glaucoma Procedure Combined with Cataract Surgery: Physical Properties and Clinical Outcome. Appl. Sci. 2020, 10, 218. https://doi.org/10.3390/app10010218
Pajic B, Cvejic Z, Mansouri K, Resan M, Allemann R. High-Frequency Deep Sclerotomy, A Minimal Invasive Ab Interno Glaucoma Procedure Combined with Cataract Surgery: Physical Properties and Clinical Outcome. Applied Sciences. 2020; 10(1):218. https://doi.org/10.3390/app10010218
Chicago/Turabian StylePajic, Bojan, Zeljka Cvejic, Kaweh Mansouri, Mirko Resan, and Reto Allemann. 2020. "High-Frequency Deep Sclerotomy, A Minimal Invasive Ab Interno Glaucoma Procedure Combined with Cataract Surgery: Physical Properties and Clinical Outcome" Applied Sciences 10, no. 1: 218. https://doi.org/10.3390/app10010218
APA StylePajic, B., Cvejic, Z., Mansouri, K., Resan, M., & Allemann, R. (2020). High-Frequency Deep Sclerotomy, A Minimal Invasive Ab Interno Glaucoma Procedure Combined with Cataract Surgery: Physical Properties and Clinical Outcome. Applied Sciences, 10(1), 218. https://doi.org/10.3390/app10010218






