A Pilot Study to Quantify Volatile Organic Compounds and Their Sources Inside and Outside Homes in Urban India in Summer and Winter during Normal Daily Activities
Abstract
1. Introduction
2. Materials and Methods
2.1. General Description of the Study Area
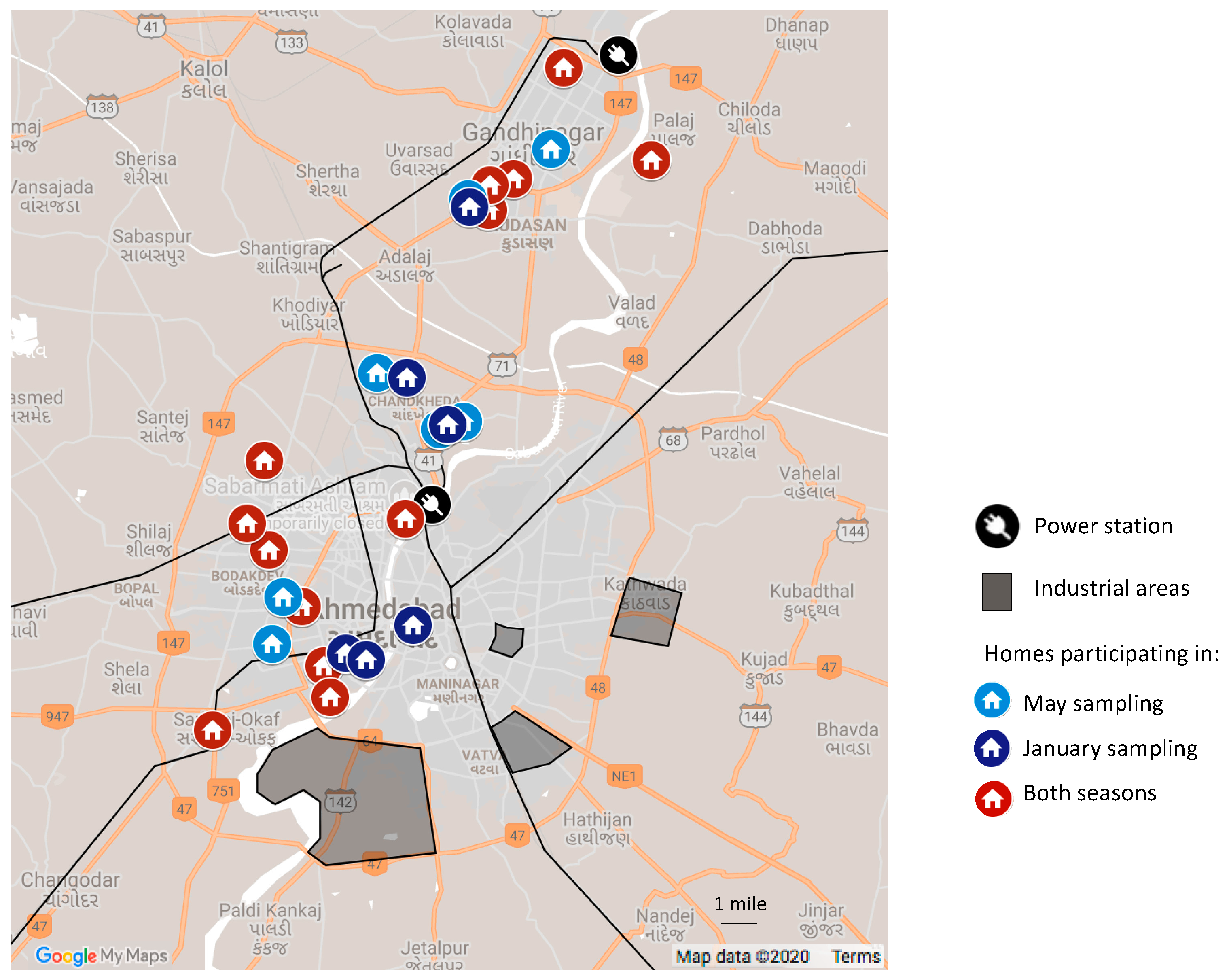
2.2. Recruitment and Description of Homes
2.3. Overview of Volatile Organic Compound Sample Collection and Analysis
2.4. Home Survey
2.5. Statistical Analysis
2.5.1. Overview of Concentrations, Detection Rates, and Differences by Location and Sample Collection Time
2.5.2. Non-Negative Matrix Factorization and Investigation of Pollutant Sources
3. Results
3.1. Overview of Compounds Measured
3.1.1. Total Volatile Organic Compounds
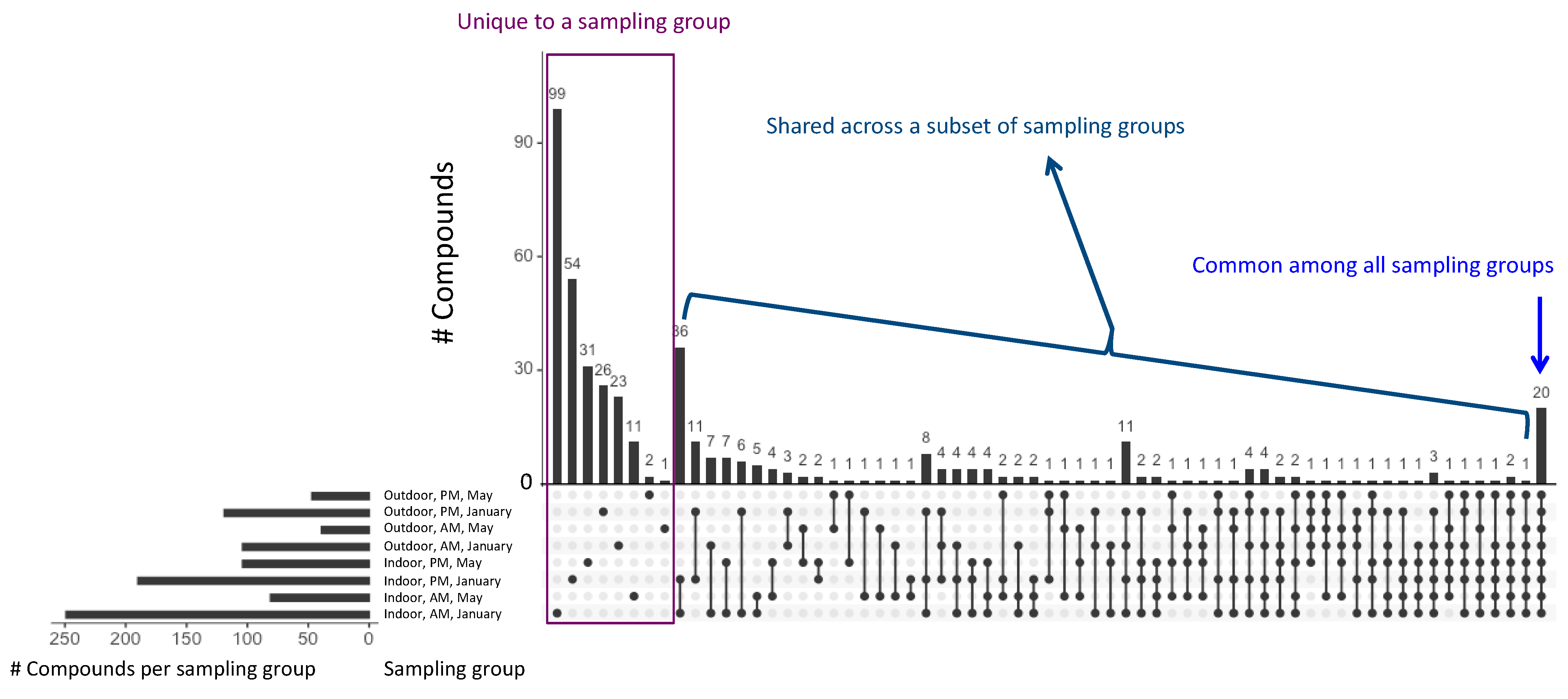
3.1.2. Individual Compounds: Prevalence and Temporal and Spatial Patterns
3.1.3. Hazardous Compounds
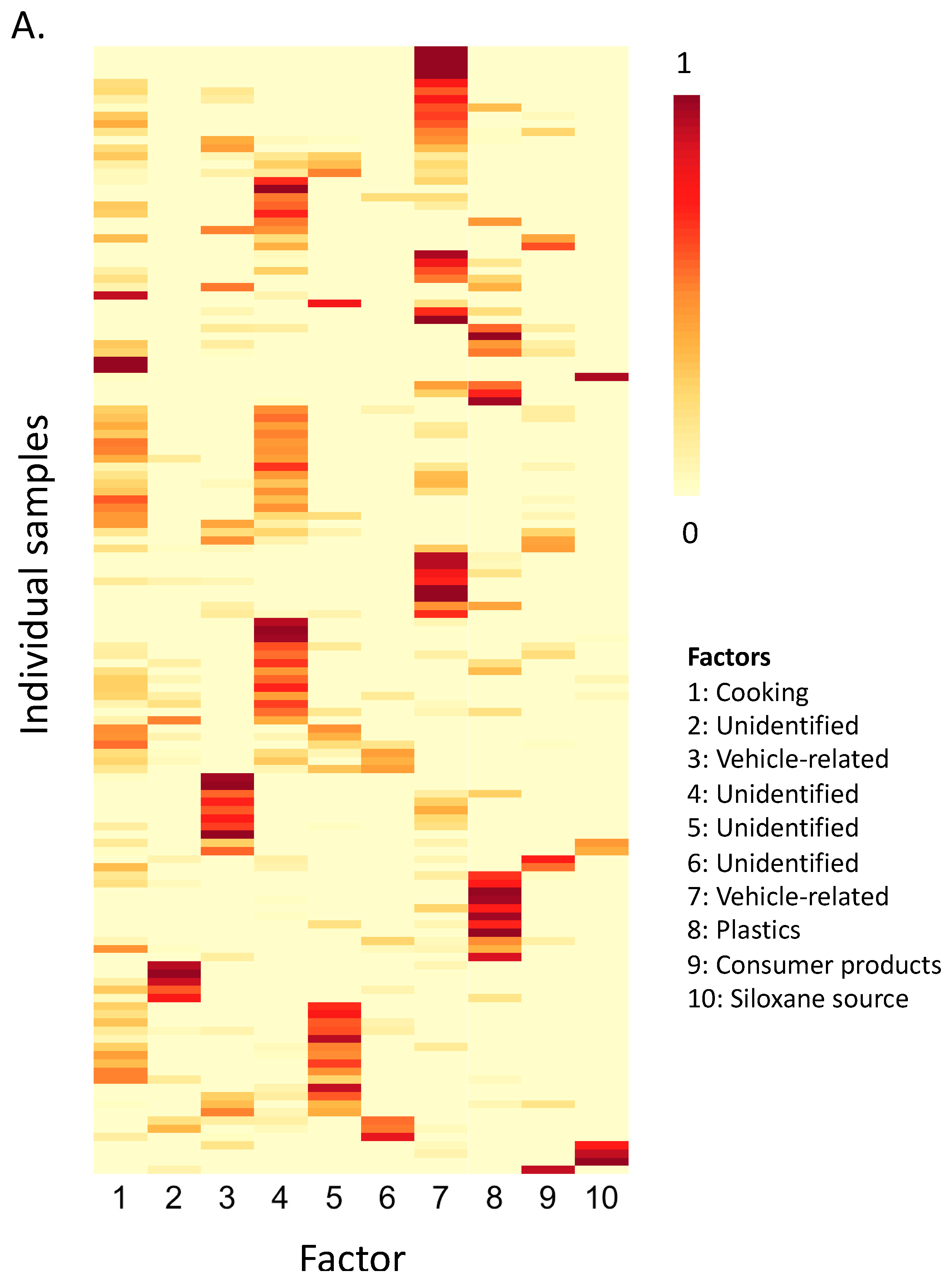
3.2. Source Identification Using Non-Negative Matrix Factorization
3.2.1. Factor 1: Cooking
3.2.2. Factor 3: Vehicle-Related
3.2.3. Factor 7: Vehicle-Related
3.2.4. Factor 8: Plastics
3.2.5. Factor 9: Consumer Products
3.2.6. Factor 10: Siloxane Source
3.2.7. Unidentified Factors
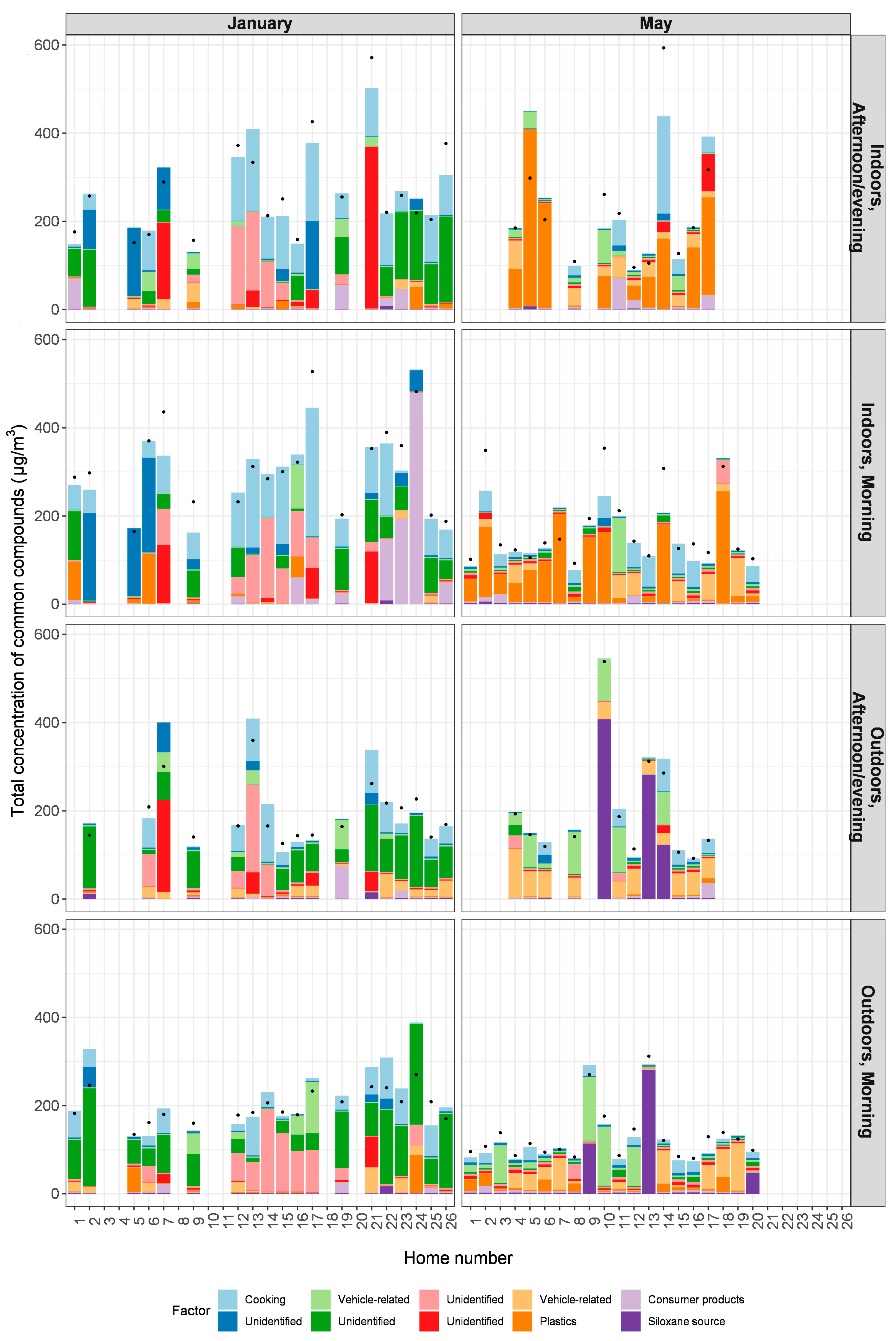
3.3. Factor Contributions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mukhopadhyay, K.; Forssell, O. An empirical investigation of air pollution from fossil fuel combustion and its impact on health in India during 1973–1974 to 1996–1997. Ecol. Econ. 2005, 55, 235–250. [Google Scholar] [CrossRef]
- Pandey, J.; Agrawal, M. Evaluation of air pollution phytotoxicity in a seasonally dry tropical urban environment using three woody perennials. New Phytol. 1994, 126, 53–61. [Google Scholar] [CrossRef]
- Beig, G.; Chate, D.M.; Ghude, S.D.; Mahajan, A.S.; Srinivas, R.; Ali, K.; Sahu, S.K.; Parkhi, N.; Surendran, D.; Trimbake, H.R. Quantifying the effect of air quality control measures during the 2010 Commonwealth Games at Delhi, India. Atmos. Environ. 2013, 80, 455–463. [Google Scholar] [CrossRef]
- Greenstone, M.; Harish, S.; Pande, R.; Sudarshan, A. The Solvable Challenge of Air Pollution in India. In Proceedings of the India Policy Forum, New Delhi, India, 11–12 July 2017. [Google Scholar]
- Balakrishnan, K.; Dey, S.; Gupta, T.; Dhaliwal, R.S.; Brauer, M.; Cohen, A.J.; Stanaway, J.D.; Beig, G.; Joshi, T.K.; Aggarwal, A.N.; et al. The impact of air pollution on deaths, disease burden, and life expectancy across the states of India: The Global Burden of Disease Study 2017. Lancet Planet. Health 2019, 3, e26–e39. [Google Scholar] [CrossRef]
- Sarkar, S. India Launches a National Clean Air Program. Available online: https://www.nrdc.org/experts/anjali-jaiswal/india-launches-national-clean-air-program (accessed on 7 September 2019).
- Gaur, M.; Singh, R.; Shukla, A. Volatile Organic Compounds in India: Concentration and Sources. J. Civ. Environ. Eng. 2016, 6, 23–27. [Google Scholar] [CrossRef]
- Kjærgaard, S.K.; Mølhave, L.; Pedersen, O.F. Human reactions to a mixture of indoor air volatile organic compounds. Atmos. Environ. Part A Gen. Top. 1991, 25, 1417–1426. [Google Scholar] [CrossRef]
- Ware, J.H.; Spengler, J.D.; Neas, L.M.; Samet, J.M.; Wagner, G.R.; Coultas, D.; Ozkaynak, H.; Schwab, M. Respiratory and irritant health effects of ambient volatile organic compounds. The Kanawha County Health Study. Am. J. Epidemiol. 1993, 137, 1287–1301. [Google Scholar] [CrossRef]
- LBNL. VOCs and Cancer. Available online: https://iaqscience.lbl.gov/voc-cancer (accessed on 10 April 2018).
- Huangfu, Y.; Lima, N.M.; O’Keeffe, P.T.; Kirk, W.M.; Lamb, B.K.; Pressley, S.N.; Lin, B.; Cook, D.J.; Walden, V.P.; Jobson, B.T. Diel variation of formaldehyde levels and other VOCs in homes driven by temperature dependent infiltration and emission rates. Build. Environ. 2019, 159, 106153. [Google Scholar] [CrossRef]
- USEPA. Indoor Air Quality—Technical Overview of Volatile Organic Compounds. Available online: https://www.epa.gov/indoor-air-quality-iaq/technical-overview-volatile-organic-compounds#3 (accessed on 10 February 2018).
- Steinemann, A. Volatile emissions from common consumer products. Air Qual. Atmos. Health 2015, 8, 273–281. [Google Scholar] [CrossRef]
- Holøs, S.B.; Yang, A.; Lind, M.; Thunshelle, K.; Schild, P.; Mysen, M. VOC emission rates in newly built and renovated buildings, and the influence of ventilation—A review and meta-analysis. Int. J. Vent. 2018, 18, 153–166. [Google Scholar] [CrossRef]
- Zhong, L.; Goldberg, M.S.; Parent, M.E.; Hanley, J.A. Risk of developing lung cancer in relation to exposure to fumes from Chinese-style cooking. Scand. J. Work. Environ. Health 1999, 25, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Thijsse, T.R.; van Oss, R.F.; Lenschow, P. Determination of Source Contributions to Ambient Volatile Organic Compound Concentrations in Berlin. J. Air Waste Manag. Assoc. 1999, 49, 1394–1404. [Google Scholar] [CrossRef]
- Barletta, B.; Meinardi, S.; Simpson, I.J.; Atlas, E.L.; Beyersdorf, A.J.; Baker, A.K.; Blake, N.J.; Yang, M.; Midyett, J.R.; Novak, B.J.; et al. Characterization of volatile organic compounds (VOCs) in Asian and north American pollution plumes during INTEX-B: Identification of specific Chinese air mass tracers. Atmos. Chem. Phys. 2009, 9, 5371–5388. [Google Scholar] [CrossRef]
- Guenther, A. The contribution of reactive carbon emissions from vegetation to the carbon balance of terrestrial ecosystems. Chemosphere 2002, 49, 837–844. [Google Scholar] [CrossRef]
- Adgate, J.L.; Church, T.R.; Ryan, A.D.; Ramachandran, G.; Fredrickson, A.L.; Stock, T.H.; Morandi, M.T.; Sexton, K. Outdoor, indoor, and personal exposure to VOCs in children. Environ. Health Perspect. 2004, 112, 1386–1392. [Google Scholar] [CrossRef]
- Brown, S.K.; Sim, M.R.; Abramson, M.J.; Gray, C.N. Concentrations of Volatile Organic Compounds in Indoor Air—A Review. Indoor Air 1994, 4, 123–134. [Google Scholar] [CrossRef]
- Edwards, R.D.; Jurvelin, J.; Saarela, K.; Jantunen, M. VOC concentrations measured in personal samples and residential indoor, outdoor and workplace microenvironments in EXPOLIS-Helsinki, Finland. Atmos. Environ. 2001, 35, 4531–4543. [Google Scholar] [CrossRef]
- Su, F.-C.; Mukherjee, B.; Batterman, S. Determinants of personal, indoor and outdoor VOC concentrations: An analysis of the RIOPA data. Environ. Res. 2013, 126, 192–203. [Google Scholar] [CrossRef]
- McDonald, B.C.; de Gouw, J.A.; Gilman, J.B.; Jathar, S.H.; Akherati, A.; Cappa, C.D.; Jimenez, J.L.; Lee-Taylor, J.; Hayes, P.L.; McKeen, S.A.; et al. Volatile chemical products emerging as largest petrochemical source of urban organic emissions. Science 2018, 359, 760–764. [Google Scholar] [CrossRef]
- Goyal, R.; Khare, M.; Kumar, P. Indoor Air Quality: Current Status, Missing Links and Future Road Map for India. J. Civ. Environ. Eng. 2012, 2, 2–4. [Google Scholar] [CrossRef]
- Gordon, T.; Balakrishnan, K.; Dey, S.; Rajagopalan, S.; Thornburg, J.; Thurston, G.; Agrawal, A.; Collman, G.; Guleria, R.; Limaye, S.; et al. Air pollution health research priorities for India: Perspectives of the Indo-U.S. Communities of Researchers. Environ. Int. 2018, 119, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.K.; Chattopadhyay, B.P.; Roy, S.K.; Das, S.; Mazumdar, D.; Roy, M.; Chakraborty, R.; Yadav, A. Work-exposure to PM10 and aromatic volatile organic compounds, excretion of urinary biomarkers and effect on the pulmonary function and heme-metabolism: A study of petrol pump workers and traffic police personnel in Kolkata City, India. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2016, 51, 135–149. [Google Scholar] [CrossRef]
- Rao, P.S.; Ansari, M.F.; Gavane, A.G.; Pandit, V.I.; Nema, P.; Devotta, S. Seasonal variation of toxic benzene emissions in petroleum refinery. Environ. Monit. Assess. 2007, 128, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Singla, V.; Pachauri, T.; Satsangi, A.; Kumari, K.M.; Lakhani, A. Comparison of BTX profiles and their mutagenicity assessment at two sites of Agra, India. Sci. World J. 2012, 2012, 272853. [Google Scholar] [CrossRef]
- Srivastava, A.; Joseph, A.E.; More, A.; Patil, S. Emissions of VOCs at urban petrol retail distribution centres in India (Delhi and Mumbai). Environ. Monit. Assess. 2005, 109, 227–242. [Google Scholar] [CrossRef]
- Srivastava, A.; Majumdar, D. Emission inventory of evaporative emissions of VOCs in four metro cities in India. Environ. Monit. Assess. 2010, 160, 315–322. [Google Scholar] [CrossRef]
- Pandit, G.G.; Srivastava, P.K.; Rao, A.M. Monitoring of indoor volatile organic compounds and polycyclic aromatic hydrocarbons arising from kerosene cooking fuel. Sci. Total Environ. 2001, 279, 159–165. [Google Scholar] [CrossRef]
- Singh, A.; Kamal, R.; Mudiam, M.K.; Gupta, M.K.; Satyanarayana, G.N.; Bihari, V.; Shukla, N.; Khan, A.H.; Kesavachandran, C.N. Heat and PAHs Emissions in Indoor Kitchen Air and Its Impact on Kidney Dysfunctions among Kitchen Workers in Lucknow, North India. PLoS ONE 2016, 11, e0148641. [Google Scholar] [CrossRef]
- Singh, A.; Kesavachandran, C.N.; Kamal, R.; Bihari, V.; Ansari, A.; Azeez, P.A.; Saxena, P.N.; Ks, A.K.; Khan, A.H. Indoor air pollution and its association with poor lung function, microalbuminuria and variations in blood pressure among kitchen workers in India: A cross-sectional study. Environ. Health 2017, 16, 33. [Google Scholar] [CrossRef]
- Srivastava, P.K.; Pandit, G.G.; Sharma, S.; Mohan Rao, A.M. Volatile organic compounds in indoor environments in Mumbai, India. Sci. Total Environ. 2000, 255, 161–168. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, B.P.; Punia, M.; Singh, D.; Kumar, K.; Jain, V.K. Assessment of indoor air concentrations of VOCs and their associated health risks in the library of Jawaharlal Nehru University, New Delhi. Environ. Sci. Pollut. Res. Int. 2014, 21, 2240–2248. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, B.P.; Punia, M.; Singh, D.; Kumar, K.; Jain, V.K. Determination of volatile organic compounds and associated health risk assessment in residential homes and hostels within an academic institute, New Delhi. Indoor Air 2014, 24, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Devotta, S. Indoor air quality of public places in Mumbai, India in terms of volatile organic compounds. Environ. Monit. Assess. 2007, 133, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Pervez, S.; Dewangan, S.; Chakrabarty, R.; Zielinska, B. Indoor VOCs from Religious and Ritual Burning Practices in India. Aerosol Air Qual. Res. 2014, 14, 1418–1430. [Google Scholar] [CrossRef]
- Dewangan, S.; Chakrabarty, R.; Zielinska, B.; Pervez, S. Emission of volatile organic compounds from religious and ritual activities in India. Environ. Monit. Assess. 2013, 185, 9279–9286. [Google Scholar] [CrossRef]
- Bhatt, J.G.; Jani, O.K. Smart Development of Ahmedabad-Gandhinagar Twin City Metropolitan Region, Gujarat, India. In Smart Metropolitan Regional Development; Vinod Kumar, T.M., Ed.; Springer: Singapore, 2019; pp. 313–356. [Google Scholar]
- Tripathi, L.; Mishra, A.K.; Dubey, A.K.; Tripathi, C.B.; Baredar, P. Renewable energy: An overview on its contribution in current energy scenario of India. Renew. Sustain. Energy Rev. 2016, 60, 226–233. [Google Scholar] [CrossRef]
- Guttikunda, S.K.; Jawahar, P. Application of SIM-air modeling tools to assess air quality in Indian cities. Atmos. Environ. 2012, 62, 551–561. [Google Scholar] [CrossRef]
- Nagpure, A.S.; Ramaswami, A.; Russell, A. Characterizing the Spatial and Temporal Patterns of Open Burning of Municipal Solid Waste (MSW) in Indian Cities. Environ. Sci. Technol. 2015, 49, 12904–12912. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.Y.; Zhang, Q.; Wong, J.P.S.; Weber, R.J.; Black, M.S. Characterization of volatile organic compound emissions from consumer level material extrusion 3D printers. Build. Environ. 2019, 160, 106209. [Google Scholar]
- Norris, C.; Fang, L.; Barkjohn, K.K.; Carlson, D.; Zhang, Y.; Mo, J.; Li, Z.; Zhang, J.; Cui, X.; Schauer, J.J.; et al. Sources of volatile organic compounds in suburban homes in Shanghai, China, and the impact of air filtration on compound concentrations. Chemosphere 2019, 231, 256–268. [Google Scholar] [CrossRef]
- Udesky, J.O.; Dodson, R.E.; Perovich, L.J.; Rudel, R.A. Wrangling environmental exposure data: Guidance for getting the best information from your laboratory measurements. Environ. Health 2019, 18, 99. [Google Scholar] [CrossRef] [PubMed]
- Bari, M.A.; Kindzierski, W.B.; Wheeler, A.J.; Héroux, M.-È.; Wallace, L.A. Source apportionment of indoor and outdoor volatile organic compounds at homes in Edmonton, Canada. Build. Environ. 2015, 90, 114–124. [Google Scholar] [CrossRef]
- Balakrishnan, K.; Mehta, S.; Kumar, P.; Ramaswamy, P.; Sambandam, S.; Kumar, K.S.; Smith, K.R. Indoor Air Pollution Associated with Household Fuel Use in INDIA: An Exposure Assessment and Modeling Exercise in Rural Districts of Andhra Pradesh (English); World Bank: Washington, DC, USA, 2004. [Google Scholar]
- Lee, D.D.; Seung, H.S. Learning the parts of objects by non-negative matrix factorization. Nature 1999, 401, 788–791. [Google Scholar] [CrossRef] [PubMed]
- Sahu, L.K.; Saxena, P. High time and mass resolved PTR-TOF-MS measurements of VOCs at an urban site of India during winter: Role of anthropogenic, biomass burning, biogenic and photochemical sources. Atmos. Res. 2015, 164–165, 84–94. [Google Scholar] [CrossRef]
- Badarinath, K.V.S.; Sharma, A.R.; Kharol, S.K.; Prasad, V.K. Variations in CO, O3 and black carbon aerosol mass concentrations associated with planetary boundary layer (PBL) over tropical urban environment in India. J. Atmos. Chem. 2009, 62, 73–86. [Google Scholar] [CrossRef]
- Sarkar, C.; Chatterjee, A.; Majumdar, D.; Ghosh, S.K.; Srivastava, A.; Raha, S. Volatile organic compounds over Eastern Himalaya, India: Temporal variation and source characterization using Positive Matrix Factorization. Atmos. Chem. Phys. Discuss. 2014, 2014, 32133–32175. [Google Scholar] [CrossRef]
- Yang, D.S.; Shewfelt, R.L.; Lee, K.S.; Kays, S.J. Comparison of odor-active compounds from six distinctly different rice flavor types. J. Agric. Food Chem. 2008, 56, 2780–2787. [Google Scholar] [CrossRef]
- Sahu, L.K.; Yadav, R.; Pal, D. Source identification of VOCs at an urban site of western India: Effect of marathon events and anthropogenic emissions. J. Geophys. Res. Atmos. 2016, 121, 2416–2433. [Google Scholar] [CrossRef]
- California Office of Environmental Health Hazard Assessment. Chemicals. Available online: https://oehha.ca.gov/chemicals (accessed on 9 September 2019).
- World Health Organization International Agency for Research on Cancer. IARC Monographs on the Identification of Carcinogenic Hazards to Humans. Available online: https://monographs.iarc.fr/agents-classified-by-the-iarc/ (accessed on 9 September 2019).
- USEPA. IRIS Assessments. Available online: https://cfpub.epa.gov/ncea/iris_drafts/atoz.cfm?list_type=alpha (accessed on 9 September 2019).
- Sax, S.N.; Bennett, D.H.; Chillrud, S.N.; Kinney, P.L.; Spengler, J.D. Differences in source emission rates of volatile organic compounds in inner-city residences of New York City and Los Angeles. J. Expo. Anal. Environ. Epidemiol. 2004, 14 (Suppl. 1), S95–S109. [Google Scholar] [CrossRef]
- Marchand, C.; Bulliot, B.; Le Calvé, S.; Mirabel, P. Aldehyde measurements in indoor environments in Strasbourg (France). Atmos. Environ. 2006, 40, 1336–1345. [Google Scholar] [CrossRef]
- Salthammer, T.; Mentese, S.; Marutzky, R. Formaldehyde in the indoor environment. Chem. Rev. 2010, 110, 2536–2572. [Google Scholar] [CrossRef] [PubMed]
- Smedje, G.; NorbÄCk, D.; Edling, C. Asthma among secondary schoolchildren in relation to the school environment. Clin. Exp. Allergy 1997, 27, 1270–1278. [Google Scholar] [CrossRef] [PubMed]
- Rumchev, K.B.; Spickett, J.T.; Bulsara, M.K.; Phillips, M.R.; Stick, S.M. Domestic exposure to formaldehyde significantly increases the risk of asthma in young children. Eur. Respir. J. 2002, 20, 403–408. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guidelines for Indoor Air Quality: Selected Pollutants; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2010. [Google Scholar]
- Belford, M.; Mac Namee, B.; Greene, D. Stability of topic modeling via matrix factorization. Expert Syst. Appl. 2018, 91, 159–169. [Google Scholar] [CrossRef]
- Maga, J.A. Rice product volatiles: A review. J. Agric. Food Chem. 2002, 32, 964–970. [Google Scholar] [CrossRef]
- Tsai, J.H.; Liu, Y.Y.; Yang, C.Y.; Chiang, H.L.; Chang, L.P. Volatile organic profiles and photochemical potentials from motorcycle engine exhaust. J. Air Waste Manag. Assoc. 2003, 53, 516–522. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lagoudi, A.; Loizidou, M.; Asimakopoulos, D. Volatile Organic Compounds in Office Buildings. Indoor Built Environ. 2016, 5, 348–354. [Google Scholar] [CrossRef]
- Krishan, S. Benzene Levels High Again: Need Action; Right to Clean Air Campaign; Centre for Science and Environment: New Delhi, India, 2012. [Google Scholar]
- Majumdar, D.; Dutta, C.; Mukherjee, A.K.; Sen, S. Source apportionment of VOCs at the petrol pumps in Kolkata, India; exposure of workers and assessment of associated health risk. Transp. Res. Part D Transp. Environ. 2008, 13, 524–530. [Google Scholar] [CrossRef]
- Subedi, B.; Sullivan, K.D.; Dhungana, B. Phthalate and non-phthalate plasticizers in indoor dust from childcare facilities, salons, and homes across the USA. Environ. Pollut. 2017, 230, 701–708. [Google Scholar] [CrossRef]
- Nalli, S.; Horn, O.J.; Grochowalski, A.R.; Cooper, D.G.; Nicell, J.A. Origin of 2-ethylhexanol as a VOC. Environ. Pollut. 2006, 140, 181–185. [Google Scholar] [CrossRef]
- Billings, A.; Jones, K.C.; Pereira, M.G.; Spurgeon, D.J. Plasticisers in the terrestrial environment: Sources, occurrence and fate. Environ. Chem. 2021, 18, 111–130. [Google Scholar] [CrossRef]
- Lattuati-Derieux, A.; Egasse, C.; Thao-Heu, S.; Balcar, N.; Barabant, G.; Lavédrine, B. What do plastics emit? HS-SPME-GC/MS analyses of new standard plastics and plastic objects in museum collections. J. Cult. Herit. 2013, 14, 238–247. [Google Scholar] [CrossRef]
- Steinemann, A.C.; MacGregor, I.C.; Gordon, S.M.; Gallagher, L.G.; Davis, A.L.; Ribeiro, D.S.; Wallace, L.A. Fragranced consumer products: Chemicals emitted, ingredients unlisted. Environ. Impact Assess. Rev. 2011, 31, 328–333. [Google Scholar] [CrossRef]
- Lin, J.; Aoll, J.; Niclass, Y.; Velazco, M.I.; Wunsche, L.; Pika, J.; Starkenmann, C. Qualitative and quantitative analysis of volatile constituents from latrines. Environ. Sci. Technol. 2013, 47, 7876–7882. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, V.; Anandan, R.; Nair, S. Fatty acid composition of Sargassum wightii and Amphiroa anceps collected from the Mandapam coast Tamil Nadu, India. J. Chem. Pharm. Res. 2011, 3, 210–216. [Google Scholar]
- Tran, T.M.; Abualnaja, K.O.; Asimakopoulos, A.G.; Covaci, A.; Gevao, B.; Johnson-Restrepo, B.; Kumosani, T.A.; Malarvannan, G.; Minh, T.B.; Moon, H.B.; et al. A survey of cyclic and linear siloxanes in indoor dust and their implications for human exposures in twelve countries. Environ. Int. 2015, 78, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Horii, Y.; Kannan, K. Survey of organosilicone compounds, including cyclic and linear siloxanes, in personal-care and household products. Arch. Environ. Contam. Toxicol. 2008, 55, 701–710. [Google Scholar] [CrossRef]
- Rauert, C.; Shoieb, M.; Schuster, J.K.; Eng, A.; Harner, T. Atmospheric concentrations and trends of poly- and perfluoroalkyl substances (PFAS) and volatile methyl siloxanes (VMS) over 7 years of sampling in the Global Atmospheric Passive Sampling (GAPS) network. Environ. Pollut. 2018, 238, 94–102. [Google Scholar] [CrossRef]
- Kwon, K.D.; Jo, W.K.; Lim, H.J.; Jeong, W.S. Characterization of emissions composition for selected household products available in Korea. J. Hazard. Mater. 2007, 148, 192–198. [Google Scholar] [CrossRef]
- Lasekan, O.; Juhari, N.H.; Pattiram, P.D. Headspace Solid-phase Microextraction Analysis of the Volatile Flavour Compounds of Roasted Chickpea (Cicer arietinum L.). J. Food Processing Technol. 2011, 2, 1000112. [Google Scholar] [CrossRef]
- Rembold, H.; Wallner, P.; Nitz, S.; Kollmannsberger, H.; Drawert, F. Volatile components of chickpea (Cicer arietinum L.) seed. J. Agric. Food Chem. 2002, 37, 659–662. [Google Scholar] [CrossRef]
- Sharma, N.; Sharma, A.R.; Patel, B.D.; Shrestha, K. Investigation on phytochemical, antimicrobial activity and essential oil constituents of Nardostachys jatamansi DC. in different regions of Nepal. J. Coast. Life Med. 2016, 4, 56–60. [Google Scholar] [CrossRef]
- Schauer, J.J.; Kleeman, M.J.; Cass, G.R.; Simoneit, B.R. Measurement of emissions from air pollution sources. 4. C1-C27 organic compounds from cooking with seed oils. Environ. Sci. Technol. 2002, 36, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Corsi, R.L. Texanol® ester alcohol emissions from latex paints: Temporal variations and multi-component recoveries. Atmos. Environ. 2007, 41, 3225–3234. [Google Scholar] [CrossRef]
- Fu, P.Q.; Kawamura, K.; Pavuluri, C.M.; Swaminathan, T.; Chen, J. Molecular characterization of urban organic aerosol in tropical India: Contributions of primary emissions and secondary photooxidation. Atmos. Chem. Phys. 2010, 10, 2663–2689. [Google Scholar] [CrossRef]
- Sahu, L.K.; Tripathi, N.; Yadav, R. Contribution of biogenic and photochemical sources to ambient VOCs during winter to summer transition at a semi-arid urban site in India. Environ. Pollut. 2017, 229, 595–606. [Google Scholar] [CrossRef]
- Majumdar, D.; Srivastava, A. Volatile organic compound emissions from municipal solid waste disposal sites: A case study of Mumbai, India. J. Air Waste Manag. Assoc. 2012, 62, 398–407. [Google Scholar] [CrossRef]


| Sampling Group | Variable for Comparison | Compounds in Both Groups | Compounds with Statistically Significant Differences (p < 0.05) |
|---|---|---|---|
| Indoors, AM | Season 1 | 52 | Toluene; Decanal; Butanal; Nonanal; 2,2,4,6,6-pentamethylheptane; Trimethylbenzene *; Hexanal; Benzene; Diethyl phthalate; Naphthalene; Xylene (para/meta); Undecane; Benzoic acid; Hexanoic acid; Ethylbenzene; Xylene (ortho) |
| Indoors, PM | Season 1 | 45 | Butanal; 2,2,4,6,6-pentamethylheptane; Benzene; Benzoic acid; Trimethylbenzene *; Acetone |
| Outdoors, AM | Season 1 | 27 | Benzaldehyde; Toluene; Hexane; 2,2,4,6,6-pentamethylheptane; Nonanal; Benzoic acid; Xylene (para/meta); Decanal; Trimethylbenzene *; Naphthalene |
| Outdoors, PM | Season 1 | 32 | 2,2,4,6,6-pentamethylheptane; 2,2,4,4-Tetramethyloctane; Benzoic acid; Acetaldehyde; Benzaldehyde; Methylcyclopentane |
| May, indoors | Time 2 | 49 | Nonanal; Decanal; Benzoic acid |
| May, outdoors | Time 2 | 31 | Hexane; Benzoic acid; Trimethylbenzene *; Benzaldehyde; Decanal; Butanal |
| January, indoors | Time 2 | 114 | Toluene; Formaldehyde; Benzene; Acetaldehyde; Naphthalene; Benzoic acid; Hexanoic acid; Heptane |
| January, outdoors | Time 2 | 60 | Benzoic acid; Benzene |
| May, AM | Location 3 | 32 | Toluene; Nonanal; Formaldehyde; Xylene (para/meta); Decanal; Trimethylbenzene *; Naphthalene |
| May, PM | Location 3 | 34 | Toluene; Hexane; Decanal; Nonanal; Formaldehyde; Benzoic acid; Acetone; Benzaldehyde; Naphthalene |
| January, AM | Location 3 | 67 | Toluene; Decanal; Butanal; Nonanal; Decamethylcyclopentasiloxane; Formaldehyde; Hexanal; Acetaldehyde; Diethyl phthalate; Naphthalene; Xylene (para/meta); Hexane; Benzoic acid; Ethylbenzene; Xylene (ortho) |
| January, PM | Location 3 | 78 | Butanal; Nonanal; Formaldehyde; Acetaldehyde; Diethyl phthalate; Decanal; Benzoic acid; Naphthalene |
| Factor | Compounds with Strongest Associations | Strongest Representation in Samples |
|---|---|---|
| 1: Cooking | Butanal; ethylbenzene; naphthalene; diethyl phthalate; hexanal | Stronger indoors but present outdoors; January |
| 2: Unidentified | Tridecane; dodecane; undecane; tetradecane; 3-methyldodecane; 2-methyldecane; 1-butanol; 2-butyl-1-octanol | Indoors; January |
| 3: Vehicle-Related | 2-methylpentane; 3-methylpentane; methylcyclopentane; hexane; acetone; 2-ethyloxetane | Outdoors; afternoon/evening; May |
| 4: Unidentified | 2,2,3,3-tetramethylpentane; 2,6,6-trimethyldecane; 2,5-diphenyl-2,5-cyclohexadiene-1,4-dione; 2,2,4,4-tetramethyloctane; phenylmaleic anhydride; Medium loadings: benzene; benzoic acid; 2,2,4,6,6-pentamethylheptane; 4-(4′-hydroxyphenoxy)biphenyl; 2-ethyl-1-hexanol | Higher outdoors than indoors in May; January |
| 5: Unidentified | Octane; 1.4-methano-1H-cyclopenta[d]pyridazine.4.4a.5.7a-tetrahydro; 2,5,6-trimethyloctane; and 3-(chloromethyl)heptane | Indoors and outdoors; morning; much stronger in January than May |
| 6: Unidentified | 3a.4.5.6.7.7a.hexahydro.4.7.methanoindene; propyl benzene; indane; benzene, 1-ethyl-2-methyl (aka 2-ethyltoluene); benzene, 1-methylethyl (aka Cumene); propanoic acid, 2-methyl-, 3-hydroxy-2,2,4-trimethylpentyl ester (aka Texanol) | January |
| 7: Vehicle-Related | Unresolved hydrocarbons; propanal; benzaldehyde | Outdoors, but indoors as well in May |
| 8: Plastics | Di-isobutyl phthalate; diethyl phthalate; 2-ethyl-1-hexanol; (1R)-1,7,7-trimethylbicyclo [2.2.1]heptan-2-one (aka D-camphor); pentadecane; octanal | Indoors; May |
| 9: Consumer products | Octane; camphor; hexanoic acid; 1,4-dichlorobenzene; methanol. oxo-. benzoate; D-limonene | Indoors; morning; January |
| 10: Siloxane source | Hexadecamethyl-cyclooctasiloxane [D8]; dodecamethyl cyclohexasiloxane [D6]; tetradecamethyl-cycloheptasiloxane [D7] | Outdoors; afternoon/evening; May |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norris, C.L.; Edwards, R.; Ghoroi, C.; Schauer, J.J.; Black, M.; Bergin, M.H. A Pilot Study to Quantify Volatile Organic Compounds and Their Sources Inside and Outside Homes in Urban India in Summer and Winter during Normal Daily Activities. Environments 2022, 9, 75. https://doi.org/10.3390/environments9070075
Norris CL, Edwards R, Ghoroi C, Schauer JJ, Black M, Bergin MH. A Pilot Study to Quantify Volatile Organic Compounds and Their Sources Inside and Outside Homes in Urban India in Summer and Winter during Normal Daily Activities. Environments. 2022; 9(7):75. https://doi.org/10.3390/environments9070075
Chicago/Turabian StyleNorris, Christina L., Ross Edwards, Chinmay Ghoroi, James J. Schauer, Marilyn Black, and Michael H. Bergin. 2022. "A Pilot Study to Quantify Volatile Organic Compounds and Their Sources Inside and Outside Homes in Urban India in Summer and Winter during Normal Daily Activities" Environments 9, no. 7: 75. https://doi.org/10.3390/environments9070075
APA StyleNorris, C. L., Edwards, R., Ghoroi, C., Schauer, J. J., Black, M., & Bergin, M. H. (2022). A Pilot Study to Quantify Volatile Organic Compounds and Their Sources Inside and Outside Homes in Urban India in Summer and Winter during Normal Daily Activities. Environments, 9(7), 75. https://doi.org/10.3390/environments9070075






