Field Test of Mini Photoionization Detector-Based Sensors—Monitoring of Volatile Organic Pollutants in Ambient Air
Abstract
:1. Introduction
2. Materials and Methods
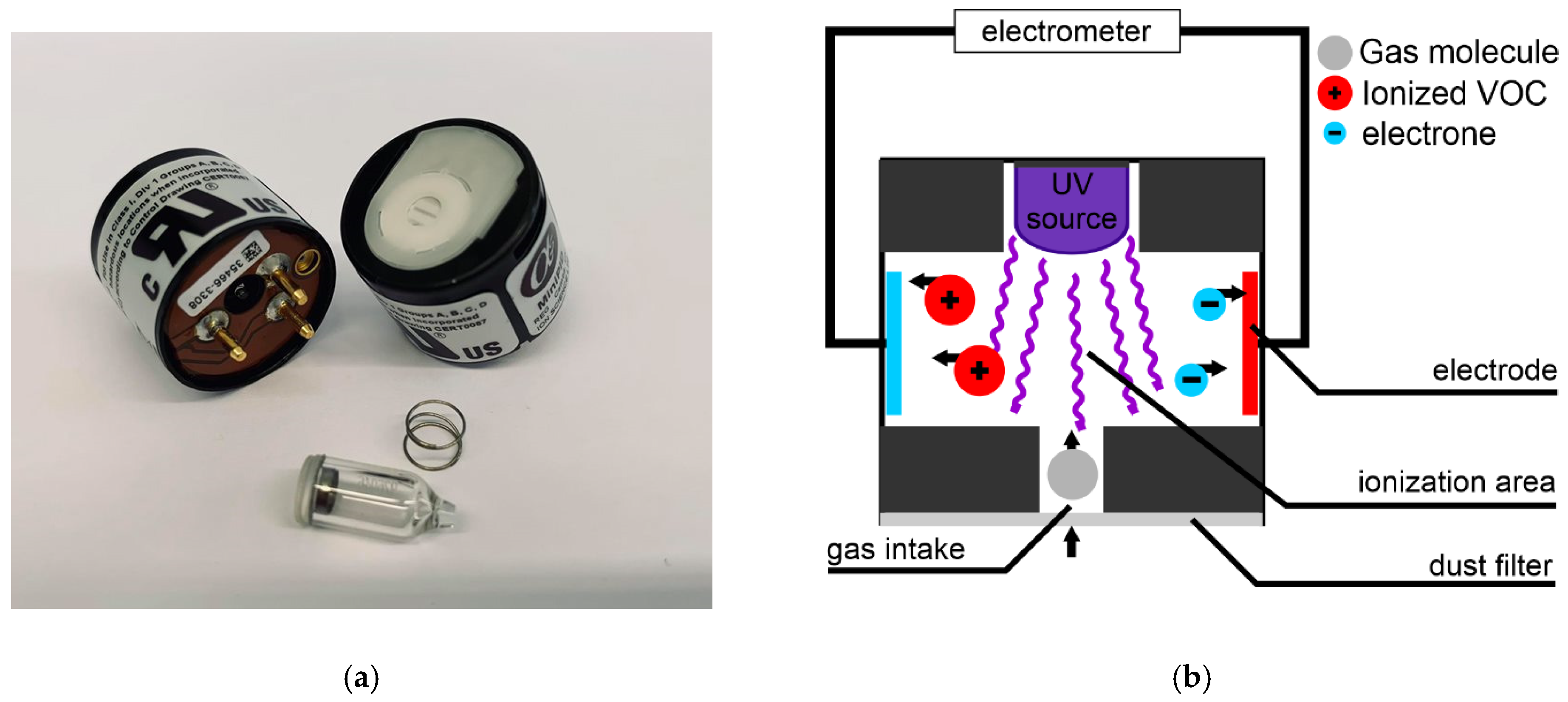
2.1. Mini-PID Overview
2.2. Analyzed Compounds
2.3. Sensor Parameter Check
2.4. Outdoor Measurements—Field Test
2.5. Statistics
3. Results and Discussion
3.1. Linearity, Measurement Range and Concentration Calibration
3.1.1. Blank
3.1.2. Concentration and Voltage Range
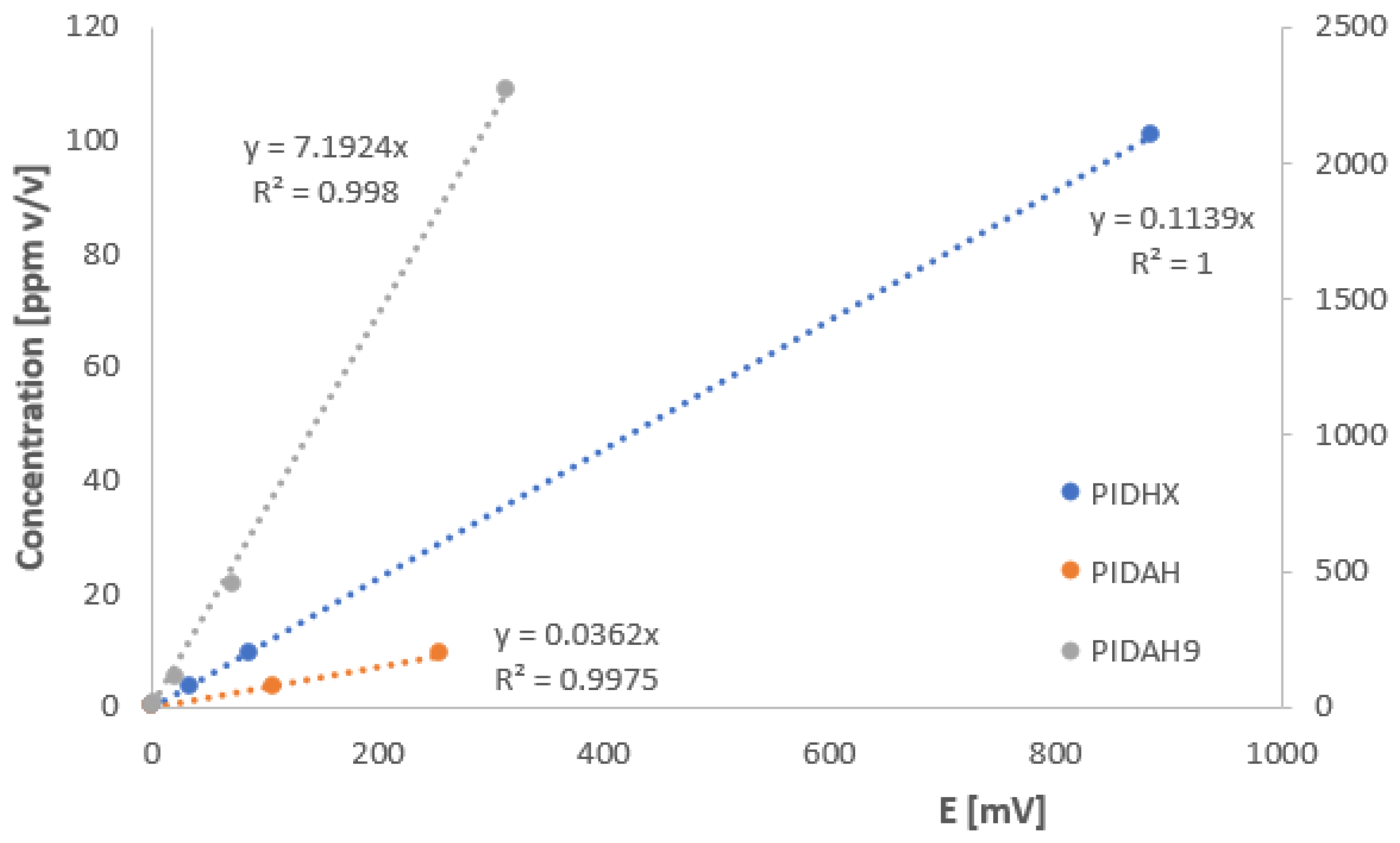
3.1.3. Linearity of the Output, Concentration Calibration
- Sensor AH is usable in the range from 0.5 to 100 ppm
- Sensor HX is usable in the range from 2 to 300 ppm
- Sensor H9 is usable in the range from 100 to 10,000 ppm
3.2. Relative Sensitivity of the Sensors
3.3. Response to Mixtures of Sensed Compounds
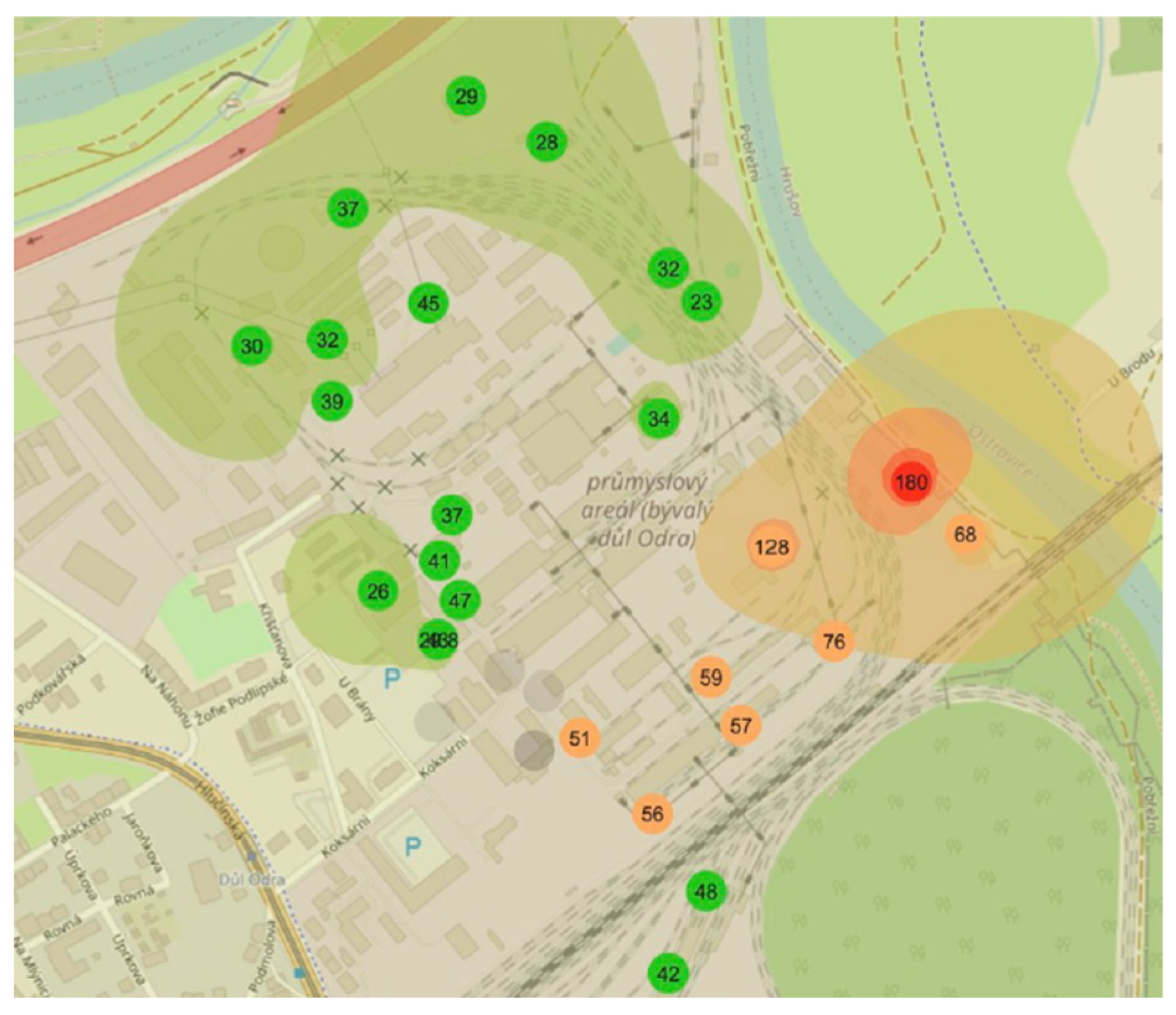
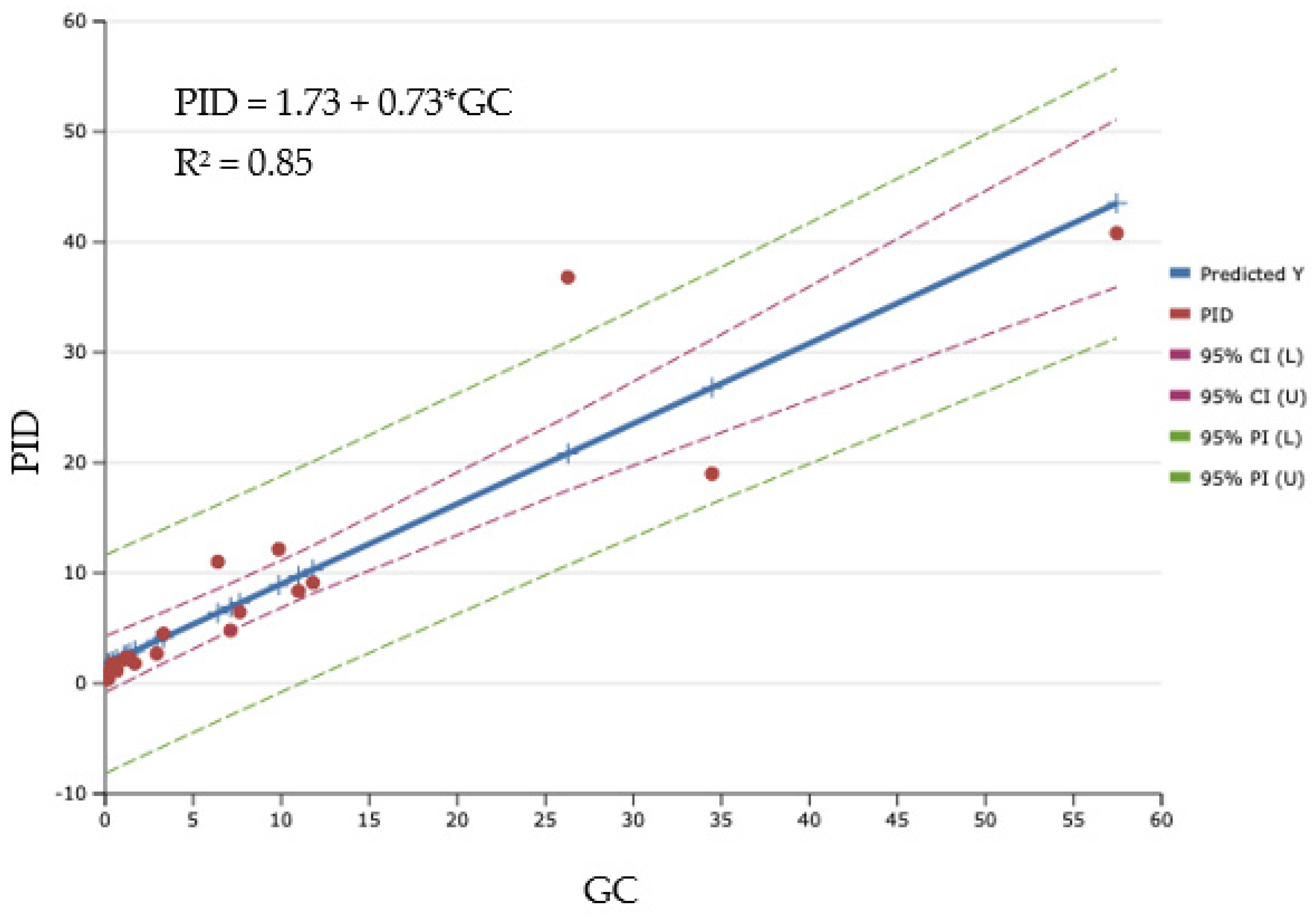
3.4. Field Measurements
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spinelle, L.; Gerboles, M.; Kok, G.; Persijn, S.; Sauerwald, T. Review of Portable and Low-Cost Sensors for the Ambient Air Monitoring of Benzene and Other Volatile Organic Compounds. Sensors 2017, 17, 1520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agbroko, S.O.; Covington, J. A Novel, Low-Cost, Portable PID Sensor for the Detection of Volatile Organic Compounds. Sens. Actuators B Chem. 2018, 275, 10–15. [Google Scholar] [CrossRef] [Green Version]
- Lewis, A.C.; Lee, J.D.; Edwards, P.M.; Shaw, M.D.; Evans, M.J.; Moller, S.J.; Smith, K.R.; Buckley, J.W.; Ellis, M.; Gillot, S.R.; et al. Evaluating the Performance of Low Cost Chemical Sensors for Air Pollution Research. Faraday Discuss. 2016, 189, 85–103. [Google Scholar] [CrossRef] [PubMed]
- Saito, N.; Haneda, H.; Watanabe, K.; Shimanoe, K.; Sakaguchi, I. Highly Sensitive Isoprene Gas Sensor Using Au-Loaded Pyramid-Shaped ZnO Particles. Sens. Actuators B Chem. 2021, 326, 128999. [Google Scholar] [CrossRef]
- Bílek, J.; Bílek, O.; Maršolek, P.; Buček, P. Ambient Air Quality Measurement with Low-Cost Optical and Electrochemical Sensors: An Evaluation of Continuous Year-Long Operation. Environments 2021, 8, 114. [Google Scholar] [CrossRef]
- Spinelle, L.; Gerboles, M.; Villani, M.G.; Aleixandre, M.; Bonavitacola, F. Calibration of a Cluster of Low-Cost Sensors for the Measurement of Air Pollution in Ambient Air. In Proceedings of the 2014 IEEE SENSORS, Valencia, Spain, 2–5 November 2014; pp. 21–24. [Google Scholar]
- Batterman, S.; Chambliss, S.; Isakov, V. Spatial Resolution Requirements for Traffic-Related Air Pollutant Exposure Evaluations. Atmos. Environ. 2014, 94, 518–528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerboles, M.; Spinelle, L.; Borowiak, A. Measuring Air Pollution with Low-Cost Sensors. Available online: https://core.ac.uk/reader/132627234 (accessed on 28 May 2021).
- Lewis, A.C.; von Schneidemesser, E.; Peltier, R.E. (Eds.) Low-Cost Sensors for the Measurement of Atmospheric Composition: Overview of Topic and Future Applications; WMO-No.1215; World Meteorological Organization: Geneva, Switzerland, 2018; ISBN 978-92-63-11215-6. [Google Scholar]
- Bauerová, P.; Šindelářová, A.; Rychlík, Š.; Novák, Z.; Keder, J. Low-Cost Air Quality Sensors: One-Year Field Comparative Measurement of Different Gas Sensors and Particle Counters with Reference Monitors at Tušimice Observatory. Atmosphere 2020, 11, 492. [Google Scholar] [CrossRef]
- Driscoll, J.N. Identification of Hydrocarbons in Complex Mixtures Using a Variable Energy PID and Capillary Column Gas Chromatography. J. Chromatogr. Sci. 1982, 20, 91–94. [Google Scholar] [CrossRef]
- Xu, W.; Cai, Y.; Gao, S.; Hou, S.; Yang, Y.; Duan, Y.; Fu, Q.; Chen, F.; Wu, J. New Understanding of Miniaturized VOCs Monitoring Device: PID-Type Sensors Performance Evaluations in Ambient Air. Sens. Actuators B Chem. 2021, 330, 129285. [Google Scholar] [CrossRef]
- Alphasense Ltd. Technical Specifications: PID-AH2. Available online: https://www.alphasense.com/wp-content/uploads/2019/08/PID-AH2.pdf (accessed on 21 February 2020).
- Alphasense Ltd. Application Note 305-06: VOC Correction Factor. Available online: https://www.alphasense.com/wp-content/uploads/2017/05/AAN-305-06.pdf (accessed on 1 December 2020).
- Alphasense Ltd. Application Note 301-04: Introduction to PhotoIonisation Detection. Available online: http://www.alphasense.com/WEB1213/wp-content/uploads/2013/07/AAN_301-04.pdf (accessed on 21 February 2020).
- Clements, A.L.; Reece, S.; Conner, T.; Williams, R. Observed Data Quality Concerns Involving Low-Cost Air Sensors. Atmos. Environ. X 2019, 3, 100034. [Google Scholar] [CrossRef] [PubMed]
- The European Parliament and The Council of the European Union. Directive 2008/50/EC of the European Parliament and of the Council of 21 May 2008 on Ambient Air Quality and Cleaner Air for Europe. Off. J. Eur. Union 2008, 152, 1. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32008L0050 (accessed on 12 August 2021).
- Directive 361/2007, Sb. of the Government of the Czech Republic on the Occupational Health Protection Conditions. Available online: https://www.zakonyprolidi.cz/cs/2007-361 (accessed on 3 December 2021).
- Alphasense Ltd. Technical Specifications: PID-A12. Available online: http://www.alphasense.com/WEB1213/wp-content/uploads/2019/08/PID-A12-1.pdf (accessed on 16 May 2020).
- CHMI Air Quality Department—Annual Tabelar Overview. Available online: https://www.chmi.cz/files/portal/docs/uoco/isko/tab_roc/tab_roc_CZ.html (accessed on 24 May 2021).
- Pang, X.; Nan, H.; Zhong, J.; Ye, D.; Shaw, M.D.; Lewis, A.C. Low-Cost Photoionization Sensors as Detectors in GC × GC Systems Designed for Ambient VOC Measurements. Sci. Total Environ. 2019, 664, 771–779. [Google Scholar] [CrossRef] [PubMed]
| Sensor Marking | Filling Gas | Ionization Energy (eV) | Measurement Range (ppm Isobutylene) |
|---|---|---|---|
| PID—H9 | Xenon | 9.6 | 0–8000 |
| PID—HX | Krypton 1 | 10.0 | 0–100 |
| PID—AH | Krypton | 10.6 | 0–40 |
| Compound | IE (eV) 1 | Purity | Producer |
|---|---|---|---|
| Benzene | 9.24 | p.a. | Lach-Ner, Neratovice, Czech Republic |
| Ethylbenzene | 8.76 | p.a. | Lach-Ner, Neratovice, Czech Republic |
| Isooctane | 9.86 | p.a. | Penta Chemicals, Praha, Czech Republic |
| 2-propanol | 10.17 | LC-MS | Biosolve, Dienze, France |
| Toluene | 8.82 | p.a. | Mach Chemikalie, Ostrava, Czech Republic |
| Trichloroethylene | 9.45 | Pure | Lach-Ner, Neratovice, Czech Republic |
| PIDHX [mV] | PIDAH [mV] | PIDAH9 [mV] |
|---|---|---|
| 39.1 ± 0.3 | 47.2 ± 2.3 | 35.8 ± 0.1 |
| Benzene | c [ppm] | 0 | 3.6 | 9.4 | 101 | 456 | 2279 |
|---|---|---|---|---|---|---|---|
| PIDHX | E [mv] 1 | 0 | 32 | 86 | 886 | 2358 | --- |
| PIDAH | 0 | 108 | 256 | 2330 | 2328 | --- | |
| PIDAH9 | 0 | 0.6 | 1.7 | 21.5 | 72.7 | 314.2 | |
| Toluene | c [ppm] | 0 | 5.0 | 49 | 202 | 476 | --- |
| PIDHX | E [mv] | 0 | 83 | 353 | 1074 | 2339 | --- |
| PIDAH | 0 | 293 | 1165 | 2330 | 2330 | --- | |
| PIDAH9 | 0 | 1 | 7 | 34 | 82 | --- | |
| Ethylbenzene | c [ppm] | 0 | 3.6 | 13.5 | 35.3 | --- | --- |
| PIDHX | E [mv] | 0 | 33.7 | 81.7 | 198 | --- | --- |
| PIDAH | 0 | 111 | 268 | 669 | --- | --- | |
| PIDAH9 | 0 | 6.1 | 11.9 | 35.4 | --- | --- | |
| Trichloro- ethylene | c [ppm] | 0 | 9.8 | 27.8 | 52 | 174 | --- |
| PIDHX | E [mv] | 0 | 62.8 | 151 | 247 | 817 | --- |
| PIDAH | 0 | 229 | 564 | 1005 | 2329 | --- | |
| PIDAH9 | 0 | 1.9 | 4.4 | 7.6 | 27.8 | --- | |
| 2-propanol | c [ppm] | 0 | 37.7 | 154 | 329 | --- | --- |
| PIDHX | E [mv] | 0 | 17.3 | 36 | --- | --- | --- |
| PIDAH | 0 | 345 | 947 | 1739 | --- | --- | |
| PIDAH9 | 0 | 48.0 | 148 | 330 | --- | --- | |
| Isooctane | c [ppm] | 0 | 22.8 | 54.8 | 249 | 418 | --- |
| PIDHX | E [mv] | 0 | 69.2 | 155 | 521 | 792 | --- |
| PIDAH | 0 | 459 | 997 | 2332 | 2332 | --- | |
| PIDAH9 | 0 | 1.9 | 4.4 | 14.9 | 25.2 | --- |
| Sensor | PIDHX | PIDAH | PIDH9 | |||
|---|---|---|---|---|---|---|
| Compound | Slope | R2 | Slope | R2 | Slope | R2 |
| Benzene | 0.114 | 1 | 0.036 | 0.997 | 7.19 | 0.999 |
| Toluene | 0.183 | 0.986 | 0.041 | 0.97 | 5.83 | 0.999 |
| Ethylbenzene | 0.174 | 0.992 | 0.052 | 0.993 | 9.13 | 0.988 |
| Trichloroethylene | 0.212 | 0.998 | 0.051 | 0.996 | 6.29 | 0.999 |
| 2-propanol | 3.87 | 0.92 | 0.181 | 0.983 | 44.7 | 0.993 |
| Isooctane | 0.507 | 0.99 | 0.051 | 0.997 | 16.5 | 0.997 |
| PIDAH-10.6 eV | PIDHX-10.0 eV | PIDAH9-9.6 eV | |||||||
|---|---|---|---|---|---|---|---|---|---|
| P-IB 1 | P-Ben 2 | Meas 3 | P-IB 1 | P-Ben 2 | Meas 3 | P-IB 1 | P-Ben 2 | Meas 3 | |
| Benzene | 0.5 | 1.00 | 1 | 0.54 | 0.87 | 1 | N/D 4 | N/D | 1 |
| Toluene | 0.56 | 0.85 | 0.88 | 0.6 | 0.83 | 0.62 | N/D | N/D | 1.21 |
| Trichloro ethylene | 0.6 | 0.83 | 0.71 | 0.8 | 0.63 | 0.54 | N/D | N/D | 1.14 |
| Ethyl benzene | 0.56 | 0.85 | 0.7 | 0.6 | 0.83 | 0.65 | N/D | N/D | 0.79 |
| 2-propanol | 4 | 0.13 | 0.2 | 25 | 0.02 | 0.03 | N/D | N/D | 0.16 |
| Isooctane | 1.1 | 0.45 | 0.67 | 3.2 | 0.16 | 0.24 | N/D | N/D | 0.43 |
| GC-FID [ppm v/v] | PIDAH-10.6 eV | PIDHX-10.0 eV | PIDAH9-9.6 eV | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Compounds | TCE 1 | EtB 2 | IPA 3 | Meas 4 | Calc 5 | Meas 4 | Calc 5 | Meas 4 | Calc 5 |
| TCE + EtB | 76.6 | 36.8 | --- | 2047.1 | 2046.8 | 489.8 | 479.4 | 13.5 | 15.8 |
| TCE + IPA | --- | 15.4 | 79.1 | 721.9 | 857.6 | 96.0 | 126.8 | 3.2 | 3.6 |
| TCE + IPA | --- | 16.3 | 143.0 | 1743.2 | 1551.7 | 34.4 | 38.2 | 7.2 | 5.2 |
| Sample No. | GC [µg/m3] | PIDHX [µg/m3] |
|---|---|---|
| 1 | 7.6 | 6.5 |
| 2 | 0.7 | 1.1 |
| 3 | 1.7 | 1.8 |
| 4 | 1.2 | 2.3 |
| 5 | 34.5 | 19 |
| 6 | 0.2 | 1.1 |
| 8 | 0.4 | 1.8 |
| 9 | 2.9 | 2.7 |
| 10 | 9.9 | 12.2 |
| 11 | 11.8 | 9.1 |
| 12 | 6.4 | 11 |
| 13 | 26.3 | 36.8 |
| 14 | 0.2 | 0.4 |
| 15 | 3.3 | 4.5 |
| 16 | 1.4 | 2.3 |
| 17 | 11 | 8.4 |
| 18 | 1.1 | 2.2 |
| 19 | 0.1 | 0.4 |
| 20 | 57.5 | 40.8 |
| 1 | 7.6 | 6.5 |
| PIDHX vs. | R2 |
|---|---|
| Benzene (B) | 0.85 |
| Benzene + Toluene (T) | 0.61 |
| B + T + Ethylbenzene (E) | 0.39 |
| B + T + E + Xylene (X) | 0.31 |
| B + T + E + X + Styrene (S) | 0.29 |
| Benzene vs. | R2 |
|---|---|
| Toluene (T) | 0.35 |
| Ethylbenzene (E) | 0.15 |
| Xylene (X) | 0.24 |
| Styrene (S) | 0.16 |
| Sum of T + E + X + S | 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bílek, J.; Maršolek, P.; Bílek, O.; Buček, P. Field Test of Mini Photoionization Detector-Based Sensors—Monitoring of Volatile Organic Pollutants in Ambient Air. Environments 2022, 9, 49. https://doi.org/10.3390/environments9040049
Bílek J, Maršolek P, Bílek O, Buček P. Field Test of Mini Photoionization Detector-Based Sensors—Monitoring of Volatile Organic Pollutants in Ambient Air. Environments. 2022; 9(4):49. https://doi.org/10.3390/environments9040049
Chicago/Turabian StyleBílek, Jiří, Petr Maršolek, Ondřej Bílek, and Pavel Buček. 2022. "Field Test of Mini Photoionization Detector-Based Sensors—Monitoring of Volatile Organic Pollutants in Ambient Air" Environments 9, no. 4: 49. https://doi.org/10.3390/environments9040049
APA StyleBílek, J., Maršolek, P., Bílek, O., & Buček, P. (2022). Field Test of Mini Photoionization Detector-Based Sensors—Monitoring of Volatile Organic Pollutants in Ambient Air. Environments, 9(4), 49. https://doi.org/10.3390/environments9040049






