Nickel and Chromium Origin in Fluvisols of the Petruševec Well Field, Zagreb Aquifer
Abstract
1. Introduction
2. Materials and Methods
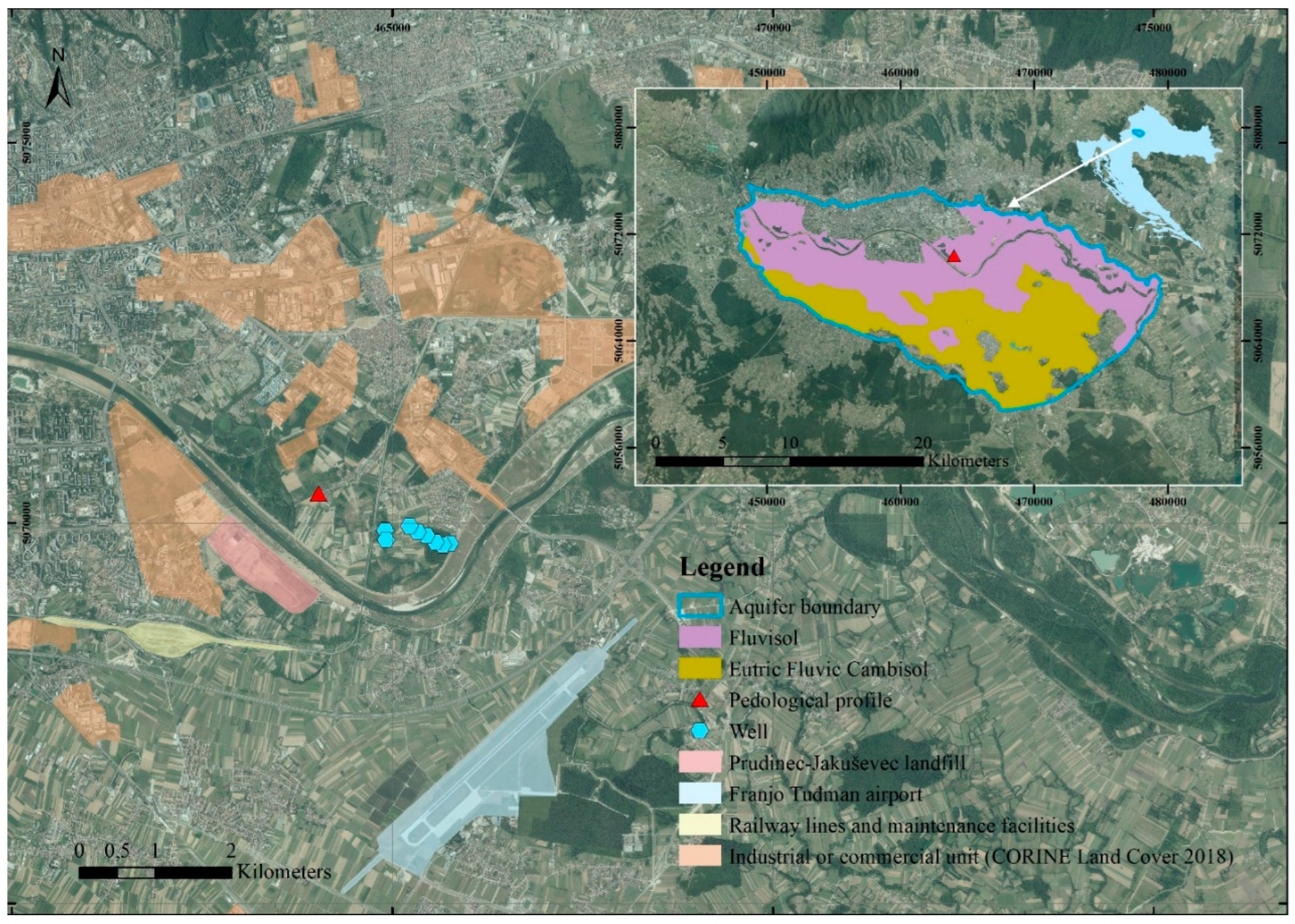
2.1. Study Area Description
2.2. Field and Laboratory Work
2.3. Enrichment Factor and Index of Geoaccumulation
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Palumbo, B.; Angelone, M.; Bellanca, A.; Dazi, C.; Hauser, S.; Neri, R.; Wilson, J. Influence of inheritance and pedogenesis on heavy metal distribution in soils of Sicily, Italy. Geoderma 2000, 95, 247–266. [Google Scholar] [CrossRef]
- Salonen, V.; Korkka-Niemi, K. Influence of parent sediments on the concentration of heavy metals in urban and suburban soils in Turku, Finland. Appl. Geochem. 2007, 22, 906–918. [Google Scholar] [CrossRef]
- Alloway, B.J. Heavy Metals in Soils, 2nd ed.; Blackie Academic & Professional: London, UK, 1995; 368p, ISBN 0-7514-0198-6. [Google Scholar]
- Kierczak, J.; Pietranik, A.; Pędziwiatr, A. Ultramafic geoecosystems as a natural source of Ni, Cr, and Co to the environment: A review. Sci. Total Environ. 2021, 755, 142620. [Google Scholar] [CrossRef]
- Sollitto, D.; Romić, M.; Castrignano, A.; Romić, D.; Bakić, H. Assessing heavy metal contamination in soils of the Zagreb region (Northwest Croatia) using multivariate geostatistics. Catena 2010, 80, 182–194. [Google Scholar] [CrossRef]
- Kasprzak, K.S.; Sunderman, W.F.; Salnikow, K. Nickel carcinogenesis. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2003, 533, 67–97. [Google Scholar] [CrossRef] [PubMed]
- Sedman, R.M.; Beaumont, J.; McDonald, T.A.; Reynolds, S.; Krowech, G.; Howd, R. Review of the evidence regarding the carcinogencity of hexavalent chromium in drinking water. J. Environ. Sci. Health Part C 2006, 24, 155–182. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, J.J.; Sedman, R.M.; Reynolds, S.D.; Sherman, C.D.; Li, L.H.; Howd, R.A.; Sandy, M.S.; Zeise, L.; Alexeeff, G.V. Cancer mortality in a Chinese population exposed to hexavalent chromium in drinking water. Epidemiology 2008, 19, 12–23. [Google Scholar] [CrossRef]
- Goyer, R.A. Toxic effects of metals. In Casarett and Doull’s Toxicology: The Basic Science of Poisons; Klassen, C.D., Ed.; McGraw-Hill: New York, NY, USA, 1996; pp. 691–736. [Google Scholar]
- Kortenkamp, A.; Casadevall, M.; Faux, S.P.; Jenner, A.; Shayer, R.O.J.; Woodbridge, N.; O’Brien, P. A role for molecular oxygen in the formation of DNA damage during the reduction of the carcinogen chromium (VI) by glutathione. Arch. Biochem. Biophys. 1996, 329, 199–207. [Google Scholar] [CrossRef]
- Fantoni, D.; Brozzo, G.; Canepa, M.; Cipolli, F.; Marini, L.; Ottonello, G.; Vetuschi Zuccolini, M. Natural hexavalent chromium in groundwaters interacting with ophiolitic rocks. Environ. Geol. 2022, 42, 871–882. [Google Scholar] [CrossRef]
- Ball, J.W.; Izbicki, J.A. Occurrence of hexavalent chromium in ground water in the western Mojave Desert, California. Appl. Geochem. 2004, 19, 1123–1135. [Google Scholar] [CrossRef]
- Izbicki, J.A.; Ball, J.W.; Bullen, T.D.; Sutley, S.J. Chromium, chromium isotopes and selected traced elements, western Mojave Desert, USA. Appl. Geochem. 2008, 23, 1325–1352. [Google Scholar] [CrossRef]
- Wood, W.W.; Clark, D.; Imes, J.L.; Councell, T.B. Eolian transport of geogenic hexavalent chromium to ground water. Ground Water 2010, 48, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Morrison, J.M.; Goldhaber, M.B.; Lee, L.; Holloway, J.M.; Wanty, R.B.; Wolf, R.E.; Ranville, J.F. A regional-scale study of chromium and nickel in soils of northern California, USA. Appl. Geochem. 2009, 24, 1500–1511. [Google Scholar] [CrossRef]
- Morrison, J.M.; Goldhaber, M.B.; Mills, C.T.; Breit, G.N.; Hooper, R.L.; Holloway, J.M.; Diehl, S.F.; Ranville, J.F. Weathering and transport of chromium and nickel from serpentinite in the Coast Range ophiolite to the Sacramento Valley, California, USA. Appl. Geochem. 2015, 61, 72–86. [Google Scholar] [CrossRef]
- Mills, C.T.; Morrison, J.M.; Goldhaber, M.B.; Ellefsen, K.J. Chromium (VI) generation in vadose zone soils and alluvial sediments of the southwestern Sacramento Valley, California: A potential source of geogenic Cr (VI) to groundwater. Appl. Geochem. 2011, 26, 1488–1501. [Google Scholar] [CrossRef]
- Kabata-Pendias, A. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Rinklebe, J.; Shaheen, S.M. Redox chemistry of nickel in soils and sediments: A review. Chemosphere 2017, 179, 265–278. [Google Scholar] [CrossRef]
- Starr, M.; Lindroos, A.-J.; Ukonmaanaho, L.; Tarvainen, T.; Tanskanen, H. Weathering release of heavy metals from soil in comparison to deposition, litterfall and leaching fluxes in a remote, boreal coniferous forest. Appl. Geochem. 2003, 18, 607–613. [Google Scholar] [CrossRef]
- Cheng, C.-H.; Jien, S.-H.; Iizuka, Y.; Tsai, H.; Chang, Y.-H.; Hseu, Z.-Y. Pedogenic chromium and nickel partitioning in serpentine soils along a toposequence. Soil Sci. Soc. Am. J. 2011, 75, 659–668. [Google Scholar] [CrossRef]
- Abdelwaheb, M.; Jebali, K.; Dhaouadi, H.; Dridi-Dhaouadi, S. Adsorption of nitrate, phosphate, nickel and lead on soils: Risk of groundwater contamination. Ecotoxicol. Environ. Saf. 2019, 179, 182–187. [Google Scholar] [CrossRef]
- Barešić, J.; Parlov, J.; Kovač, Z.; Sironić, A. Use of nuclear power plant released tritium as groundwater 224 tracer. Rud. -Geološko-Naft. Zb. 2020, 35, 25–34. [Google Scholar] [CrossRef]
- Posavec, K.; Vukojević, P.; Ratkaj, M.; Bedeniković, T. Cross-correlation Modelling of Surface Water—Groundwater Interaction Using the Excel Spreadsheet Application. Rud.-Geološko-Naft. Zb. 2017, 32, 25. [Google Scholar] [CrossRef]
- Parlov, J.; Kovač, Z.; Nakić, Z.; Barešić, J. Using water stable isotopes for identifying groundwater recharge 250 sources of the unconfined alluvial Zagreb aquifer (Croatia). Water 2019, 11, 2177. [Google Scholar] [CrossRef]
- Caren, M.; Pavlić, K. Autocorrelation and Cross-Correlation Flow Analysis Along the Confluence of the Kupa and Sava Rivers. Rud.-Geološko-Naft. Zb. 2021, 36, 67–77. [Google Scholar] [CrossRef]
- Kovač, Z.; Krevh, V.; Filipović, L.; Defterdarović, J.; Buškulić, P.; Han, L.; Filipović, V. Utilizing stable water isotopes (δ2H and δ18O) to study soil-water origin in sloped vineyard: First results. Rud.-Geološko-Naft. Zb. 2022, 37, 1–14. [Google Scholar] [CrossRef]
- Kovač, Z.; Ružičić, S.; Rubinić, V.; Nakić, Z.; Sertić, M. Sorption of cadmium, zinc and copper in dominant soils of the Zagreb aquifer system, Croatia. Geol. Croat. 2022, 75, 177–188. [Google Scholar] [CrossRef]
- Miko, S.; Halamić, J.; Peh, Z.; Galović, L. Geochemical Baseline Mapping of Soils Developed on Diverse Bedrock from Two Regions in Croatia. Geol. Croat. 2001, 54, 53–118. [Google Scholar] [CrossRef]
- Romić, M.; Romić, D. Heavy metals distribution in agricultural topsoils in urban area. Environ. Geol. 2003, 43, 795–805. [Google Scholar] [CrossRef]
- Halamić, J.; Miko, S. Geochemical Atlas of the Republic of Croatia; Croatian Geological Survey: Zagreb, Croatia, 2009; p. 87. [Google Scholar]
- Nakić, Z.; Ružičić, S.; Posavec, K.; Mileusnić, M.; Parlov, J.; Bačani, A.; Durn, G. Conceptual model for groundwater status and risk assessment—Ase study of the Zagreb aquifer system. Geol. Croat. 2013, 66, 55. [Google Scholar] [CrossRef]
- Basch, O. Osnovna geološka karta SFRJ 1: 100000. Tumač za list Ivanić-Grad [Basic Geological Map of SFRY 1: 100000, Geology of the Ivanić-Grad sheet− in Croatian]; Geološki Zavod: Zagreb, Croatia; Savezni Geološki Zavod: Beograd, Serbia, 1983; pp. 38–40. [Google Scholar]
- Velić, J.; Saftić, B.; Malvić, T. Lithologic Composition and Stratigraphy of Quaternary Sediments in the Area of the “Jakusevec” Waste Depository (Zagreb, Northern Croatia). Geol. Croat. 1999, 52, 119–130. [Google Scholar]
- Velić, J.; Saftić, B. Subsurface spreading and facies characteristics of middle Pleistocene deposits between Zapresic and Samobor. Geološki Vjesn. 1999, 44, 69–82. [Google Scholar]
- Velić, J.; Durn, G. Alternating Lacustrine-Marsh Sedimentation and Subaerial Exposure Phases during Quaternary: Prečko, Zagreb, Croatia. Geol. Croat. 1993, 46, 71. [Google Scholar]
- Vujević, M.; Posavec, K. Identification of Groundwater Level Decline in the Zagreb and Samobor-Zapresic 284 Aquifers since the Sixties of the Twentieth Century. Rud.-Geološko-Naft. Zb. 2018, 33, 55–64. [Google Scholar] [CrossRef]
- Ružičić, S.; Mileusnić, M.; Posavec, K. Building Conceptual and Mathematical Model for Water Flow and Solute Transport in the Unsaturated zone at Kosnica Site. Rud.-Geološko-Naft. Zb. 2012, 25, 21. [Google Scholar]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014: International Soil Classification System for Naming Soil and Creating Legends for Soil Maps; World Soil Resources Report; FAO: Rome, Italy, 2014; p. 106. [Google Scholar]
- Ružičić, S.; Kovač, Z.; Perković, D.; Bačani, L.; Majhen, L. The Relationship between the Physicochemical Properties and Permeability of the Fluvisols and Eutric Cambisols in the Zagreb Aquifer, Croatia. Geosciences 2019, 9, 416. [Google Scholar] [CrossRef]
- Jahn, R.; Blume, H.P.; Asio, V.B.; Spaargaren, O.; Schad, P. Guidelines for Soil Description, 4th ed.; FAO: Rome, Italy, 2006; p. 97. [Google Scholar]
- ISO 13536; Soil Quality-Determination of pH. International Organisation for Standardisation: Geneva, Switzerland, 2005.
- ISO 10693; Soil Quality-Determination of Carbonate Content—Volumetric Method. International Organisation for Standardisation: Geneva, Switzerland, 1995.
- ISO 13536; Soil Quality-Determination of the Potential Cation Exchange Capacity and Exchangeable Cations Using Barium Chloride Solution Buffered at pH = 8.1. International Organisation for Standardisation: Geneva, Switzerland, 1995.
- Moore, D.M.; Reynolds, R.C. X-ray Diffraction and the Identification and Analysis of Clay Minerals; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Jain, C. Metal fractionation study on bed sediments of River Yamuna, India. Water. Res. 2004, 38, 569–578. [Google Scholar] [CrossRef]
- Thuong, N.T.; Yoneda, M.; Shimada, Y.; Matsui, Y. Assessment of trace metal contamination and exchange between water and sediment systems in the To Lich River in inner Hanoi, Vietnam. Environ. Earth. Sci. 2015, 73, 3925–3936. [Google Scholar] [CrossRef]
- Githaiga, K.B.; Njuguna, S.M.; Makokha, V.A.; Wang, J.; Gituru, R.W.; Yan, X. Assessment of Cu, Zn, Mn, and Fe enrichment in Mt. Kenya soils: Evidence for atmospheric deposition and contamination. Environ. Monit. Assess. 2020, 192, 167. [Google Scholar] [CrossRef]
- Reimann, C.; Caritat, P. Intrinsic flaws of element enrichment factors (EFS) in environmental geochemistry. Environ. Sci. Technol. 2000, 34, 5084–5091. [Google Scholar] [CrossRef]
- Gowd, S.S.; Reddy, M.R.; Govil, P. Assessment of heavy metal contamination in soils at Jajmau (Kanpur) and unnao industrial areas of the ganga plain, uttar pradesh, India. J. Hazard. Mater. 2010, 174, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Uduma, A.U.; Awagu, E.F. Manganese as a Reference Element for the Assessment of Zinc Enrichment and Depletion in Selected Farming Soils of Nigeria. Res. J. Environ. Earth Sci. 2013, 5, 497–504. [Google Scholar] [CrossRef]
- Bu, J.; Sun, Z.; Zhou, A.; Xu, Y.; Ma, R.; Wei, W.; Liu, M. Heavy Metals in Surface Soils in the Upper Reaches of the Heihe River, Northeastern Tibetan Plateau, China. Int. J. Environ. Res. Public Health 2016, 13, 247. [Google Scholar] [CrossRef] [PubMed]
- Githaiga, K.B.; Njuguna, S.M.; Yan, X. Local Geochemical Baselines Reduce Variation Caused by the Use of Different Conservative Elements in Predicting Cu and Zn Enrichment in Agricultural Soils, Kenya. Chem. Afr. 2021, 4, 869–880. [Google Scholar] [CrossRef]
- Birch, G. A Scheme for Assessing Human Impacts on Coastal Aquatic Environments Using Sediments; Woodcoffe, C.D., Furness, R.A., Eds.; Coastal GIS 2003; Wollongong University Papers in Center for Maritime Policy: Wollongong, Australia, 2003. [Google Scholar]
- Rajmohan, N.; Prathapar, S.A.; Jayaprakash, M.; Nagarajan, R. Vertical distribution of heavy metals in soil profile in a seasonally waterlogging agriculture field in Eastern Ganges Basin. Env. Monit. Assess. 2014, 186, 5411–5427. [Google Scholar] [CrossRef] [PubMed]
- Muller, G. Schwermetalle in den sediments des RheinsVeranderungen seitt 1971. Umschan 1979, 79, 778–783. [Google Scholar]
- Förstner, U.; Müller, G. Concentrations of heavy metals and polycyclic aromatic hydrocarbons in river sediments: Geochemical background, man’s influence and environmental impact. GeoJournal 1981, 5, 417. [Google Scholar] [CrossRef]
- Praveena, S.M.; Ahmed, A.; Radojevic, M.; Abdullah, M.H.; Aris, A.Z. Multivariate and geoaccumulation index evaluation in mangrove surface sediment of Mengkabong Lagoon, Sabah. Bull. Environ. Contam. Toxicol. 2008, 81, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Mohammad Ali, B.N.; Lin, C.Y.; Cleophas, F.; Abdullah, M.H.; Musta, B. Assessment of heavy metals contamination in Mamut river sediments using sediment quality guidelines and geochemical indices. Environ. Monit. Assess. 2015, 187, 4190. [Google Scholar] [CrossRef]
- Müller, G. The Heavy Metal Pollution of the Sediments of Neckars and its Tributary: A Stocktaking. Chem. Ztg. 1981, 105, 157–164. [Google Scholar]
- Banat, K.M.; Howari, F.M.; Al-Hamad, A.A. Heavy metals in urban soils of central Jordan: Should we worry about their environmental risks? Environ. Res. 2005, 97, 258–273. [Google Scholar] [CrossRef]
- Sainz, A.; Ruiz, F. Influence of the very polluted inputs of the Tinto-Odiel system on the adjacent littoral sediments of southwestern Spain: A statistical approach. Chemosphere 2006, 62, 1612–1622. [Google Scholar] [CrossRef]
- Ružičić, S.; Mileusnić, M.; Posavec, K.; Nakić, Z.; Durn, G.; Filipović, V. Water flow and solute transport model of potentially toxic elements through unsaturated zone at regional wellfield Kosnica. Hydrol. Processes 2016, 30, 4113–4124. [Google Scholar] [CrossRef]
- Chaudhari, P.R.; Ahire, D.V.; Chkravarty, M.; Maity, S. Electrical conductivity as a tool for tetermining the physical properties of Indian soils. Int. J. Sci. Res. Publ. 2014, 4, 1–4. [Google Scholar]
- Officer, S.J.; Kravchenko, A.; Bollero, G.A.; Sudduth, K.A.; Kitchen, N.R.; Wiebold, W.J.; Palm, H.L.; Bullock, D.G. Relationships between soil bulk electrical conductivity and the principal component analysis of topography and soil fertility values. Plant Soil 2004, 258, 269–280. [Google Scholar] [CrossRef]
- Ružičić, S.; Jašaragić-Rako, T. Multielement sorption of cadmium, zinc, copper and lead onto a Fluvisol profile at the Stara Loza site, Croatia. Int. J. Environ. Pollut. 2017, 62, 63. [Google Scholar] [CrossRef]
- Meegoda, J.N.; Martin, L. In-situ determination of specific surface area of clays. Geotech. Geol. Eng. 2019, 37, 465–474. [Google Scholar] [CrossRef]
- Sherene, T. Mobility and transport of heavy metals in polluted soil environment. Biol. Forum—Int. J. 2010, 2, 112–121. [Google Scholar]
- McGrath, S.P.; Loveland, P.J. The Soil Geochemical Atlas of England and Wales; Blackie & Sons: London, UK, 1992. [Google Scholar]
- Pils, J.R.V.; Karathanasis, A.D.; Mueller, T.G. Concentration and Distribution of Six Trace Metals in Northern Kentucky Soils. Soil Sediment Contam. 2004, 13, 37–51. [Google Scholar] [CrossRef]
- Kierczak, J.; Néel, C.; Aleksander-Kwaterczak, U.; Helios-Rybicka, E.; Bril, H.; Puziewicz, J. Solid speciation and mobility of potentially toxic elements from natural and contaminated soils: A combined approach. Chemosphere 2008, 73, 776–784. [Google Scholar] [CrossRef]
- Bani, A.; Echevarria, G.; Montarges-Pelletier, E.; Gjoka, F.; Sulce, S.; Morel, J.L. Pedogenesis and nickel biogeochemistry in a typical Albanian ultramafic toposequence. Environ. Monit. Assess. 2014, 186, 4431–4442. [Google Scholar] [CrossRef]
- Schulin, R.; Curchod, F.; Mondeshka, M.; Daskalova, A.; Keller, A. Heavy metal contamination along a soil transect in the vicinity of the iron smelter of Kremikovtzi (Bulgaria). Geoderma 2007, 140, 52–61. [Google Scholar] [CrossRef]
- Ružičić, S.; Kovač, Z.; Nakić, Z.; Kireta, D. Fluvisol permeability estimation using soil water content variability. Geofizika 2017, 34, 141–155. [Google Scholar] [CrossRef]
- He, G.; Zhang, Z.; Wu, X.; Cui, M.; Zhang, J.; Huang, X. Adsorption of Heavy Metals on Soil Collected from Lixisol of Typical Karst Areas in the Presence of CaCO3 and Soil Clay and Their Competition Behavior. Sustainability 2020, 12, 7315. [Google Scholar] [CrossRef]
- Elbana, T.A.; Selim, H.M. Modeling of cadmium and nickel release from different soils. Geoderma 2019, 338, 78–87. [Google Scholar] [CrossRef]
- Dennis, J.K.; Such, T.E. Nickel and Chromium Plating, 3rd ed.; Woodhead Publishing Ltd.: Cambridge, UK, 1993. [Google Scholar]
- Tyle, H.; Scott-Fordsmand, J. Nickel and nickel compounds. European Union Risk Assessment Report 2008, Denmark. [Google Scholar]
- Stopić, D.; Mandarić, A.; Prce, M.; Begić, D.; Malenica, M.; Fratrić, T.; Mirić, S. Report on the State of the Environment in Zagreb County 2017–2020. 2021. Available online: https://www.zagrebacka-zupanija.hr/media/filer_public/17/70/1770fb57-e25f-4c47bd02-1615bca2e620/izvjesce_o_stanju_okolisa_zagrebacke_zupanije_2017_-_2020.pdf (accessed on 11 October 2022). (In Croatian).
- Ngole, V.M.; Ekosse, G.I.E. Copper, nickel and zinc contamination in soils within the precincts of mining and landfilling environments. Int. J. Environ. Sci. Technol. 2012, 9, 485–494. [Google Scholar] [CrossRef]
- Filipović, L.; Romić, M.; Romić, D.; Filipović, V.; Ondrašek, G. Organic matter and salinity modify cadmium soil (phyto)availability. Ecotoxicol Environ. Saf. 2018, 147, 824–831. [Google Scholar] [CrossRef]
- Sipos, P.; Choi, C.; Németh, T.; Szalai, Z.; Póka, T. Relationship between iron and trace metal fractionation in soils. Chem. Speciat. Bioavailab. 2014, 26, 21–30. [Google Scholar] [CrossRef]
- Antoniadis, V.; Shaheen, S.M.; Tsadilas, C.D.; Selim, M.H. Zinc sorption by different soils as affected by selective removal of carbonates and hydrous oxides. Appl. Geochem. 2018, 88, 49–58. [Google Scholar] [CrossRef]
- Flem, B.; Reimann, C.; Fabian, K. Excess Cr and Ni in top soil: Comparing the effect of geology, diffuse contamination, and biogenic influence. Sci. Total Environ. 2022, 843, 157059. [Google Scholar] [CrossRef]
- Mengel, K.; Kirkby, E.A.; Kosegarten, H.; Appel, T. Principles of Plant Nutrition; Springer: Dordrecht, The Netherlands, 2001. [Google Scholar] [CrossRef]
- Bednářová, Z.; Komprdová, K.; Kalábová, T.; Sáňka, M. Impact of floods and their frequency on content and distribution of risk elements in alluvial soils. Water Air Soil Pollut. 2015, 226, 15. [Google Scholar] [CrossRef]
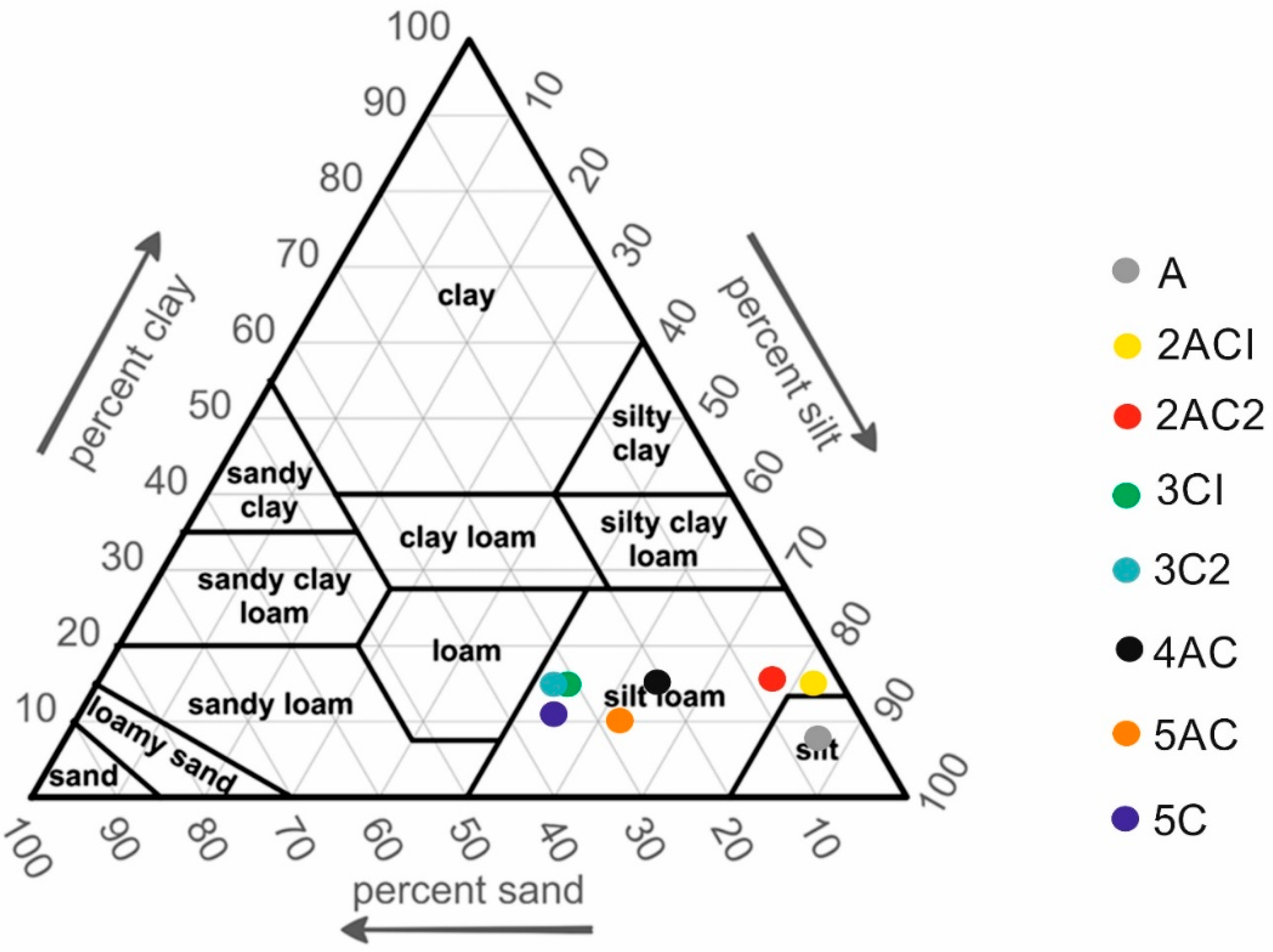
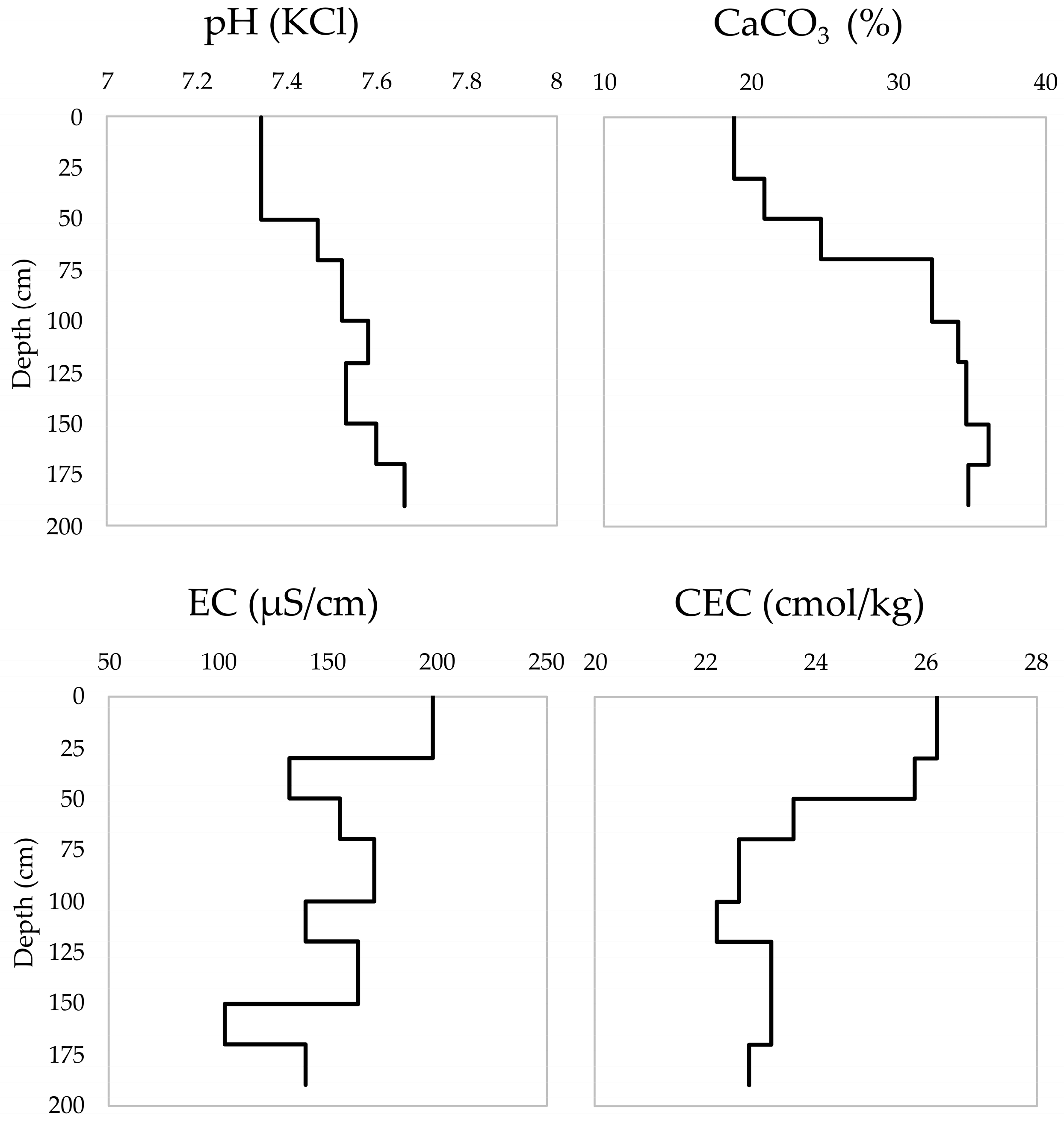
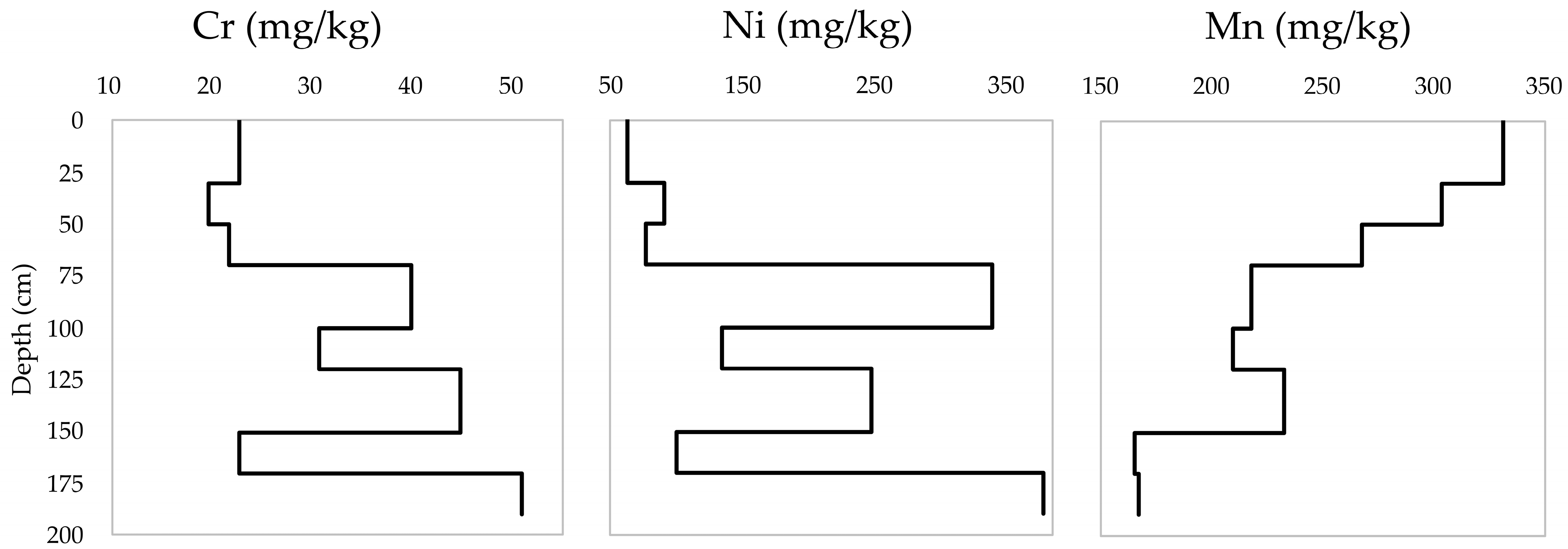
| Soil Depth (cm) | Soil Horizons | Soil Color | pH (KCl) | EC (μS/cm) | CaCO3 (%) | CEC (cmol/kg) | Soil Texture |
|---|---|---|---|---|---|---|---|
| 0–30 | A | 10YR/5/3 | 7.34 | 198 | 18.8 | 26.2 | silt |
| 30–50 | 2ACI | 10YR/5/3 | 7.34 | 132 | 20.8 | 25.8 | silt loam |
| 50–70 | 2AC2 | 10YR/5/3 | 7.47 | 155 | 24.7 | 23.6 | silt loam |
| 70–100 | 3CI | 10YR/4/3 | 7.52 | 171 | 32.3 | 22.6 | silt loam |
| 100–120 | 3C2 | 10YR/4/3 | 7.58 | 140 | 34.1 | 22.2 | silt loam |
| 120–150 | 4AC | 10YR/4/3 | 7.53 | 164 | 34.6 | 23.2 | silt loam |
| 150–170 | 5AC | 10YR/4/4 | 7.60 | 103 | 36.1 | 23.2 | silt loam |
| 170–190 | 5C | 10YR/4/4 | 7.66 | 140 | 34.8 | 22.8 | silt loam |
| Soil Depths (cm) | Soil Horizons | Qtz | Pl | Kfs | Gt | Hem | M | Kln | Chl | MLCM |
|---|---|---|---|---|---|---|---|---|---|---|
| 0–30 | A | 23 | 7 | x | x | xxx | x | xxx | ? | x |
| 30–50 | 2ACI | 23 | 8 | x | ? | xxx | x | xx | xx | x |
| 50–70 | 2AC2 | 26 | 10 | x | x | xxx | x | xxx | xx | x |
| 70–100 | 3CI | 36 | 9 | x | x | xxx | - | xx | xx | x |
| 100–120 | 3C2 | 37 | 11 | x | - | xxx | - | xx | ? | x |
| 120–150 | 4AC | 32 | 8 | x | ? | xxx | ? | xxx | xx | x |
| 150–170 | 5AC | 36 | 8 | x | ? | xxx | - | xx | xx | x |
| 170–190 | 5C | 37 | 12 | x | x | xxx | x | xx | xx | x |
| Parameter | pH (KCl) | Clay | Silt | Sand | Carbonate | EC | CEC | Cr | Ni |
|---|---|---|---|---|---|---|---|---|---|
| Cr | 0.64 | 0.24 | −0.65 | 0.64 | 0.64 | 0.14 | −0.59 | - | - |
| Ni | 0.60 | 0.22 | −0.67 | 0.67 | 0.59 | 0.08 | −0.57 | 0.94 | - |
| Enrichment Factors | Igeo | |||||||
|---|---|---|---|---|---|---|---|---|
| Croatia | Local | Croatia | Local | |||||
| Soil Horizons | Ni | Cr | Ni | Cr | Ni | Cr | Ni | Cr |
| A | 2.82 | 0.26 | 3.13 | 0.24 | 0.22 | −2.52 | 0.32 | −2.27 |
| 2ACI | 4.50 | 0.23 | 4.99 | 0.21 | 0.34 | −2.72 | 0.88 | −2.47 |
| 2AC2 | 4.27 | 0.25 | 4.73 | 0.23 | 0.08 | −2.58 | 0.62 | −2.33 |
| 3CI | 23.40 | 0.45 | 25.96 | 0.41 | 2.24 | −1.72 | 2.78 | −1.47 |
| 3C2 | 9.64 | 0.35 | 10.71 | 0.32 | 0.90 | −2.09 | 1.44 | −1.84 |
| 4AC | 15.94 | 0.51 | 17.70 | 0.46 | 1.78 | −1.55 | 2.32 | −1.30 |
| 5AC | 9.06 | 0.26 | 10.06 | 0.24 | 0.47 | −2.52 | 1.01 | −2.27 |
| 5C | 33.93 | 0.58 | 37.66 | 0.53 | 2.40 | −1.37 | 2.94 | −1.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ružičić, S.; Balaž, B.-I.; Kovač, Z.; Filipović, L.; Nakić, Z.; Kopić, J. Nickel and Chromium Origin in Fluvisols of the Petruševec Well Field, Zagreb Aquifer. Environments 2022, 9, 154. https://doi.org/10.3390/environments9120154
Ružičić S, Balaž B-I, Kovač Z, Filipović L, Nakić Z, Kopić J. Nickel and Chromium Origin in Fluvisols of the Petruševec Well Field, Zagreb Aquifer. Environments. 2022; 9(12):154. https://doi.org/10.3390/environments9120154
Chicago/Turabian StyleRužičić, Stanko, Borna-Ivan Balaž, Zoran Kovač, Lana Filipović, Zoran Nakić, and Jasna Kopić. 2022. "Nickel and Chromium Origin in Fluvisols of the Petruševec Well Field, Zagreb Aquifer" Environments 9, no. 12: 154. https://doi.org/10.3390/environments9120154
APA StyleRužičić, S., Balaž, B.-I., Kovač, Z., Filipović, L., Nakić, Z., & Kopić, J. (2022). Nickel and Chromium Origin in Fluvisols of the Petruševec Well Field, Zagreb Aquifer. Environments, 9(12), 154. https://doi.org/10.3390/environments9120154










