Assessment of Prevalence and Heterogeneity of Meso- and Microplastic Pollution in Icelandic Waters
Abstract
1. Introduction
2. Materials and Methods
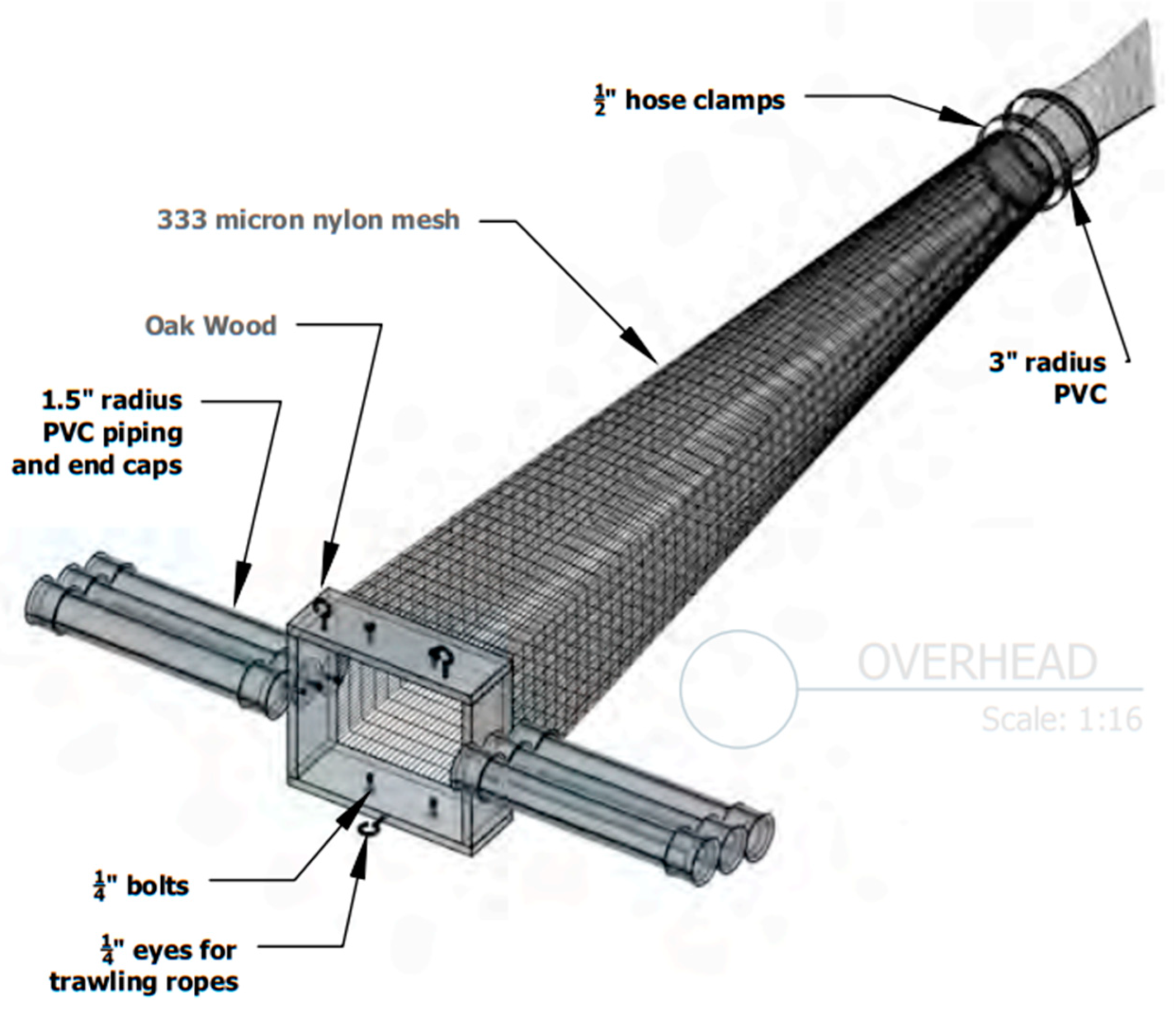
2.1. Low-Tech Aquatic Debris Instrument (LADI)
2.2. Sample Collection
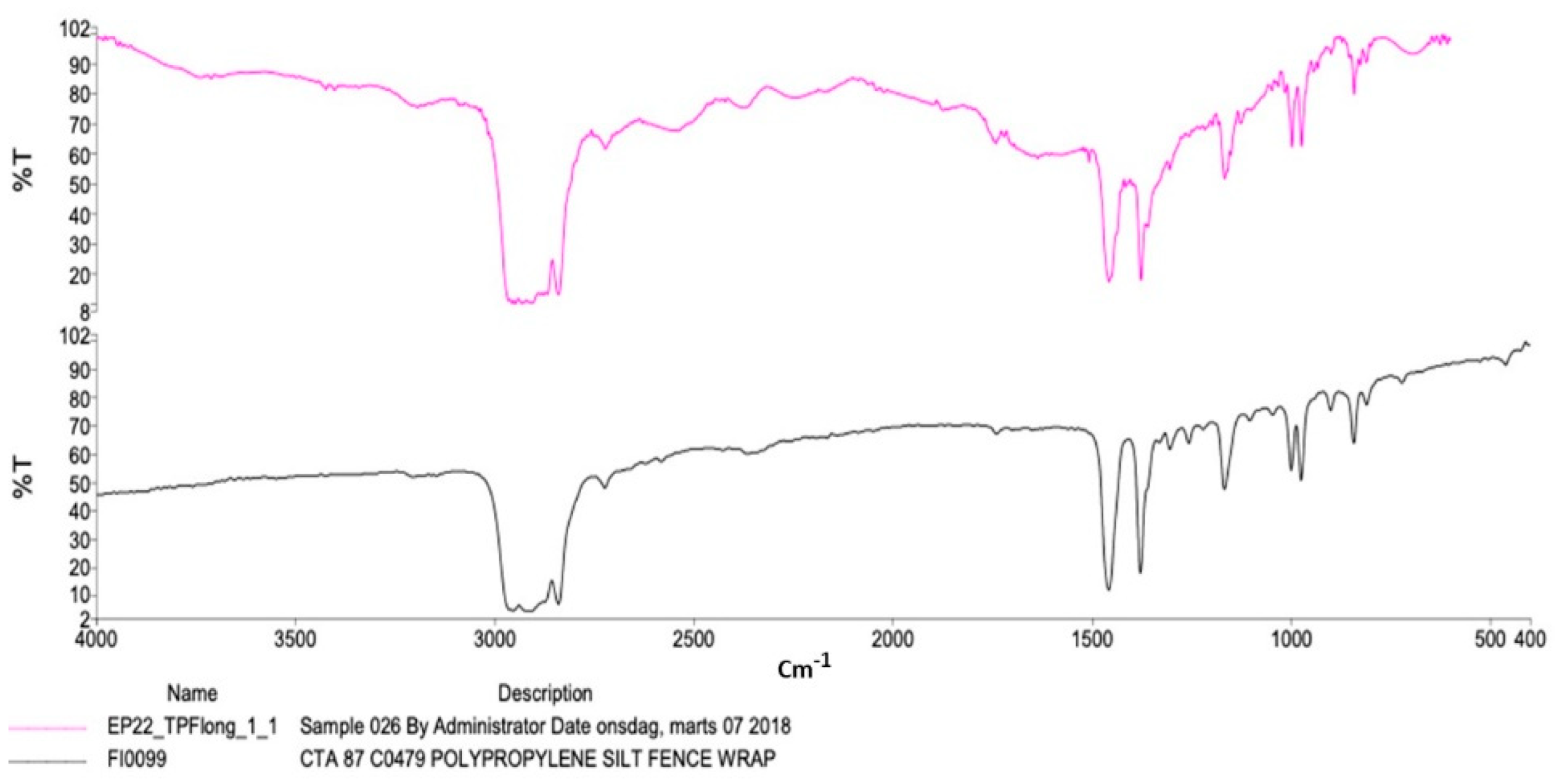
2.3. Characterization of Polymer Composition
3. Results
3.1. Abundance
3.2. Characteristics of Plastic Samples
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.M.; Brennholt, N.; Cole, M.; et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef]
- Carpenter, E.J.; Smith, K.L., Jr. Plastic on the Sargasso Sea surface. Science 1972, 175, 1240–1241. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, X.; Li, Y.; Powell, T.; Wang, X.; Wang, G.; Zhang, P. Microplastics as Contaminants in the Soil Environment: A Mini-Review. Sci. Total Environ. 2019, 691, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, R.; Azzoni, R.S.; Pittino, F.; Diolaiuti, G.; Franzetti, A.; Parolini, M. First Evidence of Microplastic Contamination in the Supraglacial Debris of an Alpine Glacier. Environ. Pollut. 2019, 253, 297–301. [Google Scholar] [CrossRef]
- Eerkes-Medrano, D.; Thompson, R.C.; Aldridge, D.C. Microplastics in Freshwater Systems: A Review of the Emerging Threats, Identification of Knowledge Gaps and Prioritization of Research Needs. Water Res. 2015, 75, 63–82. [Google Scholar] [CrossRef]
- Fogašová, K.; Manko, P.; Oboňa, J. The first evidence of microplastics in plant-formed fresh-water micro-ecosystems: Dipsacus teasel phytotelmata in Slovakia contaminated with MPs. BioRisk 2022, 18, 133. [Google Scholar] [CrossRef]
- Susanti, N.K.Y.; Mardiastuti, A.; Wardiatno, Y. Microplastics and the Impact of Plastic on Wildlife: A Literature Review. IOP Conf. Ser. Earth Environ. Sci. 2020, 528, 012013. [Google Scholar] [CrossRef]
- Kannan, K.; Vimalkumar, K. A review of human exposure to microplastics and insights into microplastics as obesogens. Front. Endocrinol. 2021, 12, 978. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.; Lebreton, L.C.M.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Galgani, F.; Ryan, P.G.; Reisser, J. Plastic pollution in the world’s oceans: More than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS ONE 2014, 9, e111913. [Google Scholar] [CrossRef] [PubMed]
- Peeken, I.; Primpke, S.; Beyer, B.; Gütermann, J.; Katlein, C.; Krumpen, T.; Bergmann, M.; Hehemann, L.; Gerdts, G. Arctic Sea ice is an important temporal sink and means of transport for microplastic. Nat. Commun. 2018, 9, 1505. [Google Scholar] [CrossRef]
- Karlsson, T.M.; Kärrman, A.; Rotander, A.; Hassellöv, M. Comparison between manta trawl and in situ pump filtration methods, and guidance for visual identification of microplastics in surface waters. Environ. Sci. Pollut. Res. 2020, 27, 5559–5571. [Google Scholar] [CrossRef] [PubMed]
- Linnebjerg, J.F.; Baak, J.E.; Barry, T.; Gavrilo, M.V.; Mallory, M.L.; Merkel, F.R.; Price, C.; Strand, J.; Walker, T.R.; Provencher, J.F. Review of plastic pollution policies of Arctic countries in relation to seabirds. FACETS 2021, 6, 1–25. [Google Scholar] [CrossRef]
- SeaIceland. Available online: https://seaiceland.is/what/gastropods/sea-shells/mussels (accessed on 13 April 2022).
- Vernet, R.G.; Borrell, A.; Víkingsson, G.; Halldórsson, S.D.; Aguilar, A. Ecological niche partitioning between baleen whales inhabiting Icelandic waters. Prog. Oceanogr. 2021, 199, 102690. [Google Scholar] [CrossRef]
- McQuilkin, J.V.; Böhme, L.; Alinaghizadeh, B.I.; Proietti, N.M. Microplastic in drinking water sources and distribution systems in Iceland. Resour. Int. Ehf. 2021. Available online: https://www.resource.is/wp-content/uploads/2020/01/Microplastics-in-drinking-water-sources-and-distribution-systems-in-Iceland.pdf (accessed on 1 April 2022).
- Loughlin, C.; Mendes, A.R.M.; Morrison, L.; Morley, A. The role of oceanographic processes and sedimentological settings on the deposition of microplastics in marine sediment: Icelandic waters. Mar. Pollut. Bull. 2021, 164, 111976. [Google Scholar] [CrossRef] [PubMed]
- AMAP. Litter and Microplastics Monitoring Guidelines, Version 1.0; Arctic Monitoring and Assessment Programme (AMAP): Tromsø, Norway, 2021; p. 257. [Google Scholar]
- Umhverfisstofnun. Niðurstöður Vöktunar Stranda 2004–2019. 2020. Available online: https://www.ust.is/library/sida/haf-og-vatn/V%c3%b6ktun%20stranda%202016-2019.pdf (accessed on 12 May 2022).
- Monteiro, R.C.; Sul, J.A.I.D.; Costa, M.F. Plastic pollution in islands of the Atlantic Ocean. Mar. Pollut. Bull. 2018, 238, 103–110. [Google Scholar] [CrossRef]
- Kienitz, A.T. Marine Debris in the Coastal Environment of Iceland’s Nature Reserve, Hornstrandir: Sources, Consequences and Prevention Measures. Master’s Thesis, University of Akureyri, Akureyri, Iceland, 2013. Available online: https://skemman.is/bitstream/1946/15898/4/Anna-Theresa%20Kienitz%20(3).pdf (accessed on 11 February 2022).
- Hafrannsóknarstofnun. Plast í Hafinu við Ísland. 2019. Available online: https://www.hafogvatn.is/is/moya/news/rannsoknir-hafrannsoknastofnunar-a-plasti-i-hafi (accessed on 15 December 2021).
- O’Rourke, A. Occurrence, Prevalence, and Classification of Fishing Related Marine Debris in Iceland’s Westfjords. Master’s Thesis, University of Akureyri, Akureyri, Iceland, 2020. Available online: https://skemman.is/handle/1946/34986 (accessed on 13 March 2022).
- Ovide, B.G.; Basran, C.; Guimard, E. Ocean Missions Impact report 2020–2021. 2021. Available online: https://oceanmissions.org/wp-content/uploads/final-impact-report-2020-2021.pdf (accessed on 25 May 2022).
- Ólafsson, H.; Furger, M.; Brümmer, B. The weather and climate of Iceland. Meteorol. Z. 2007, 16, 5–8. [Google Scholar] [CrossRef]
- Van Sebille, E.; Aliani, S.; Law, K.L.; Maximenko, N.; Alsina, J.M.; Bagaev, A.; Bergmann, M.; Chapron, B.; Chubarenko, I.; Cózar, A.; et al. The physical oceanography of the transport of floating marine debris. Environ. Res. Lett. 2020, 15, 023003. [Google Scholar] [CrossRef]
- Galloway, T.; Cole, M.; Lewis, C. Interactions of microplastic debris throughout the marine ecosystem. Nat. Ecol. Evol. 2017, 1, 0116. [Google Scholar] [CrossRef]
- Bråte, I.L.N.; Hurley, R.; Lusher, A.; Buenaventura, N.; Hultman, M.; Halsband, C.; Green, N. Microplastics in Marine Bivalves from the Nordic Environment; Nordic Council of Ministers: Copenhagen, Denmark, 2020. [Google Scholar]
- Garcia-Garin, O.; Aguilar, A.; Vighi, M.; Víkingsson, G.A.; Chosson, V.; Borrell, A. Ingestion of synthetic particles by fin whales feeding off western Iceland in summer. Sciencedirect 2021, 279, 130564. [Google Scholar] [CrossRef]
- Roch, S.; Friedrich, C.; Brinker, A. Uptake routes of microplastics in fishes: Practical and theoretical approaches to test existing theories. Sci. Rep. 2020, 10, 3896. [Google Scholar] [CrossRef] [PubMed]
- Procter, J.; Hopkins, F.E.; Fileman, E.S.; Lindeque, P.K. Smells good enough to eat: Dimethyl sulfide (DMS) enhances copepod ingestion of microplastics. Mar. Pollut. Bull. 2019, 238, 103–110. [Google Scholar] [CrossRef]
- Zantis, L.J.; Bosker, T.; Lawler, F.; Nelms, S.E.; Rorke, R.O.; Constantine, R.; Sewell, M.; Carroll, E.L. Assessing microplastic exposure of large marine filter-feeders. Sci. Total Environ. 2022, 818, 151815. [Google Scholar] [CrossRef]
- Bashir, A.; Hashmi, I. Detection in influx sources and estimation of microplastics abundance in surface waters of Rawal Lake, Pakistan. Heliyon 2022, 8, e09166. [Google Scholar] [CrossRef] [PubMed]
- Coyle, C.; Novaceski, M.; Wells, E.; Liboiron, M. LADI and the trawl. Civ. Lab. Environ. Action Res. 2016, 1, 15–43. [Google Scholar]
- Viršek, M.K.; Palatinus, A.; Koren, Š.; Peterlin, M.; Horvat, P.; Kržan, A. Protocol for Microplastics Sampling on the Sea Surface and Sample Analysis. J. Vis. Exp. 2016, 118, e55161. [Google Scholar] [CrossRef]
- Cózar, A.; Echevarría, F.; González-Gordillo, J.I.; Irigoien, X.; Úbeda, B.; Hernández-León, S.; Palma, Á.T.; Navarro, S.; García-De-Lomas, J.; Ruiz, A.; et al. Plastic debris in the open ocean. Proc. Natl. Acad. Sci. USA 2014, 111, 10239–10244. [Google Scholar] [CrossRef] [PubMed]
- Gesamp. Guidelines or the Monitoring and Assessment of Plastic Litter and Microplastics in the Ocean; Kershaw, P.J., Turra, A., Galgani, F., Eds.; GESAMP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection: London, UK, 2019; Volume 99, p. 130. [Google Scholar]
- Gunnlaugsson, S.B.; Valtysson, H. Sustainability and wealth creation, but no consensus: Recent decades in Iceland’s ITQ-managed fisheries. Policy 2022, 135, 104836. [Google Scholar] [CrossRef]
- Iceland Responsible Fisheries. A Nation with Fishing in Its Genes. 2020. Available online: https://www.responsiblefisheries.is/seafood-industry/a-nation-with-fishing-in-its-genes (accessed on 1 April 2022).
- Logemann, K.; Ólafsson, J.; Snorrason, Á.; Valdimarsson, H.; Marteinsdóttir, G. The circulation of Icelandic waters—A modelling study. Ocean Sci. 2013, 9, 931–955. [Google Scholar] [CrossRef]
- Syberg, K.; Knudsen, C.M.; Tairova, Z.; Khan, F.R.; Shashoua, Y.; Geertz, T.; Pedersen, H.B.; Sick, C.; Mortensen, J.; Strand, J.; et al. Sorption of PCBs to Environmental Plastic Pollution in the North Atlantic Ocean: Importance of Size and Polymer Type. Case Stud. Chem. Environ. Eng. 2020, 2, 100062. [Google Scholar] [CrossRef]
- Carollo, C.; Astin, I. Extreme Events in Ocean Currents through Depth in the Vicinity of the Iceland-Scotland Ridge. In Oceans ‘04 MTS/IEEE Techno-Ocean ‘04 (IEEE Cat. No.04CH37600); IEEE: New York, NY, USA, 2004; Volume 4, pp. 2331–2337. [Google Scholar] [CrossRef]
- Everaert, G.; Van Cauwenberghe, L.; De Rijcke, M.; Koelmans, A.A.; Mees, J.; Vandegehuchte, M.; Janssen, C.R. Risk assessment of microplastics in the ocean: Modelling approach and first conclusions. Environ. Pollut. 2018, 242 Pt B, 1930–1938. [Google Scholar] [CrossRef]
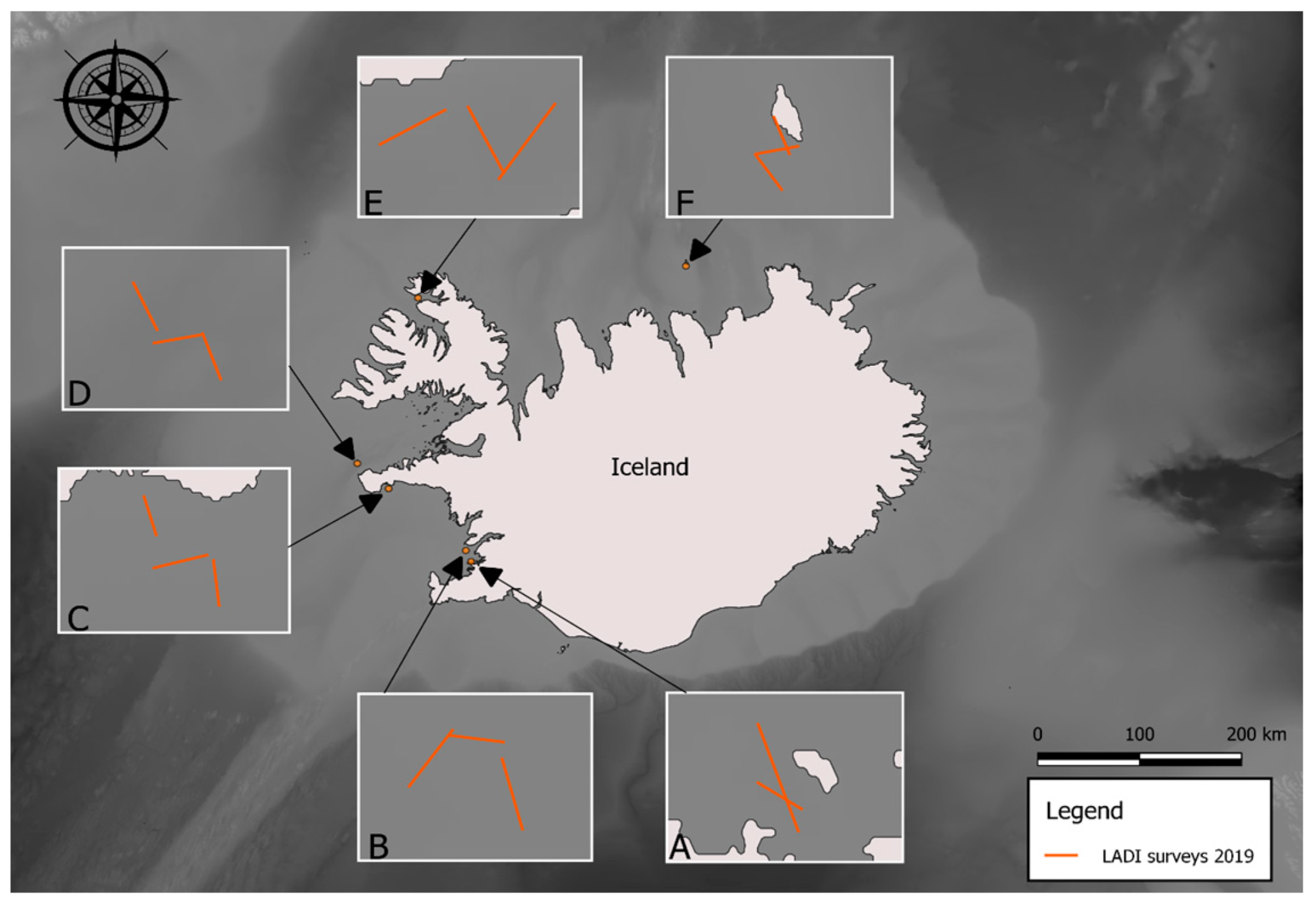
| Location | Start | End | Distance Traveled (m) | Sea State | Total MP | MP/km2 |
|---|---|---|---|---|---|---|
| Reykjavík (A) | 64 09.25 N | 64 10.58 N | 2926 | 2 | 2 | 2010 |
| 21 54.87 W | 21 56.67 W | 2759 | 1 | 1066 | ||
| 2593 | 2 | 0 | 0 | |||
| Mean MPs/km2 | 1025.4 | |||||
| Flaxaflói (B) | 64 13.79 N | 64 14.96 N | 2593 | 3 | 2 | 2269 |
| 22 00.68 W | 22 02.15 W | 2445 | 3 | 0 | 0 | |
| 2611 | 3 | 0 | 0 | |||
| Mean MPs/km2 | 756.3 | |||||
| Arnastapi (C) | 64 44.25 N | 64 45.45 N | 2408 | 1 + swell | 10 | 12,214 |
| 23 25.59 W | 23 26.24 W | 2778 | 1 + swell | 6 | 6352 | |
| 2778 | 1 + swell | 0 | 0 | |||
| Mean MPs/km2 | 6189.3 | |||||
| Snæfellsnes (D) | 64 56.95 N | 64 58.59 N | 3148 | 1 + swell | 4 | 3737 |
| 24 02.47 W | 24 03.52 W | 2222 | 1 + swell | 5 | 6617 | |
| 2593 | 1 + swell | 7 | 7941 | |||
| Mean MPs/km2 | 6098.3 | |||||
| Ísafjörður (E) | 66 16.34 N | 66 16.76 N | 2408 | 3 | 1 | 1221 |
| 22 59.79 W | 22 56.72 W | 2963 | 3 | 0 | 0 | |
| 3148 | 2 | 0 | 0 | |||
| Mean MPs/km2 | 407 | |||||
| Grímsey (F) | 66 31.93 N | 66 30.73 N | 2593 | 3 | 0 | 0 |
| 18 01.28 W | 17 59.54 W | 3019 | 4 | 1 | 974 | |
| 2778 | 4 | 0 | 0 | |||
| Mean MPs/km2 | 324.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ovide, B.G.; Cirino, E.; Basran, C.J.; Geertz, T.; Syberg, K. Assessment of Prevalence and Heterogeneity of Meso- and Microplastic Pollution in Icelandic Waters. Environments 2022, 9, 150. https://doi.org/10.3390/environments9120150
Ovide BG, Cirino E, Basran CJ, Geertz T, Syberg K. Assessment of Prevalence and Heterogeneity of Meso- and Microplastic Pollution in Icelandic Waters. Environments. 2022; 9(12):150. https://doi.org/10.3390/environments9120150
Chicago/Turabian StyleOvide, Belén García, Erica Cirino, Charla Jean Basran, Torsten Geertz, and Kristian Syberg. 2022. "Assessment of Prevalence and Heterogeneity of Meso- and Microplastic Pollution in Icelandic Waters" Environments 9, no. 12: 150. https://doi.org/10.3390/environments9120150
APA StyleOvide, B. G., Cirino, E., Basran, C. J., Geertz, T., & Syberg, K. (2022). Assessment of Prevalence and Heterogeneity of Meso- and Microplastic Pollution in Icelandic Waters. Environments, 9(12), 150. https://doi.org/10.3390/environments9120150










