1. Introduction
Mining activities usually produce a large volume of spoils, waste rocks, and tailings, which are usually deposited at the soil surface. If the spoils contain heavy metals that are soluble, there is a potential of heavy metal pollution contamination and off-site movement. Mined areas near Webb City in Jasper County, Missouri, contained mine waste piles that were removed, but still provide a source of heavy metal contamination, particularly Zn and Cd in the underlying soil. Mining activities can lead to extensive environmental pollution of terrestrial ecosystem due to the deposition of heavy-metal containing waste materials, tailings, and lagoon wastes [
1,
2,
3].
Metal (loid) contaminants such as Cd and Zn are significant issues, not only for the environment, but especially for human health [
4,
5,
6]. These contaminated areas present a health risk and are recognized as areas that need to be remediated to allow for crop phytostabilization to occur [
1]. Often, contaminated sites are not conducive for plant growth due to metal toxicity, lack of soil nutrients, low pH values, poor microbial activity, and unsuitable physical soil properties. Both physical and chemical techniques have been considered in mine spoil remediation, but these methods have flaws, are expensive, and can be disruptive to soils. Remediation of these contaminated and hazardous soils by conventional practices using excavation and landfilling is arguably unfeasible on large scales because these techniques are cost-prohibitive and environmentally disruptive [
7,
8]. Phytostabilization techniques that involve the establishment of plant cover on the surface of contaminated sites could serve as an efficient alternative remediation approach as they provide low-cost and environmentally friendly options [
7,
9]. For this reason, remediation of contaminated sites using phytostabilization techniques require the amendment to improve soil-plant relationships thereby stimulating plant growth.
Remediation of mine spoil can be a complex process due to several chemical and physical factors that can limit plant growth [
10]. Bolan et al. [
11] summarized the different factors affecting phytostabilization. For example, soil, plant, contaminants, and environmental factors determine the successful outcome of phytostabilization technology in relation to both the remediation and revegetation of contaminated sites. Mine spoils may have unfavorable soil chemical characteristics, e.g., very low pH, phytotoxic metals [
12,
13], physical limitations (e.g., high bulk density, low soil moisture retention, poor aggregation [
14]; and unsuitable microbial habitat conditions, e.g., low soil organic matter and poor nutrient turnover [
15]. These aspects can severely limit plant growth. As such, reclamation plans usually involve applying soil amendments (i.e., composts, lime) to neutralize their low pH, and to raise organic matter levels that favors organic binding of metals, along with enhanced microbial enzymatic activity for nutrient cycling [
16].
Phytostabilization can be enhanced by using soil amendments that immobilize metal(loid)s when combined with plant species that are tolerant of high levels of contaminants while simultaneously improving the physical, chemical, and biological properties of mine soils. Some previously used amendments to improve soil conditions include biosolids, lime, green waste, or biochars. Among these amendment types, the use of biochar has recently been investigated for in situ remediation of contaminated lands in association with plants [
10,
17,
18,
19]. The incorporation of organic amendments improves the quality of mine soils and makes it possible for vegetation to be established [
20,
21]. Recent studies have highlighted that biochars are effective soil amendments in that they improve soil conditions to raise the agronomic values of soils [
22,
23,
24,
25].
Numerous studies may have shown that adding organic amendments (e.g., biochars, sewage sludge, manures) to soil promotes the phytoextraction process [
26,
27], but only few studies have evaluated the combined effect of organic amendments and phytostabilization with corn in Cd and Zn contaminated mine soils. There is a lack of agreement over the influence of organic amendments such as biochars on metal immobilization in soil. Moreover, the application of biochars to contaminated soil systems has not been systematically investigated to any great extent. Biochar may be a tool for mine spoil remediation; however, its mechanisms for achieving this goal are still not well understood. The objective of our study was to evaluate the interactive effects of biochar additions with or without the manure-based compost (MBC) on shoots biomass (SBY), roots biomass (RBY), uptake, and bioconcentration factor (BCF) of Zn and Cd in corn (
Zea mays L.) grown in mine soil.
4. Discussion
Overall, our results showed that mine spoil remediation can be potentially enhanced by using soil amendments capable of immobilizing metal(loid)s when combined with plant species that are tolerant of high levels of contaminants (
Table 1). The incorporation of organic amendments improves the quality of mine soils and makes it possible for vegetation to be established [
20,
21]. Hossain et al. [
24] and Dede et al. [
26] have reported that the addition of organic amendments (e.g., biochars, sewage sludge, manures) to soil have promoted the phytoextraction process and improved soil conditions to raise the agronomic values of the soils.
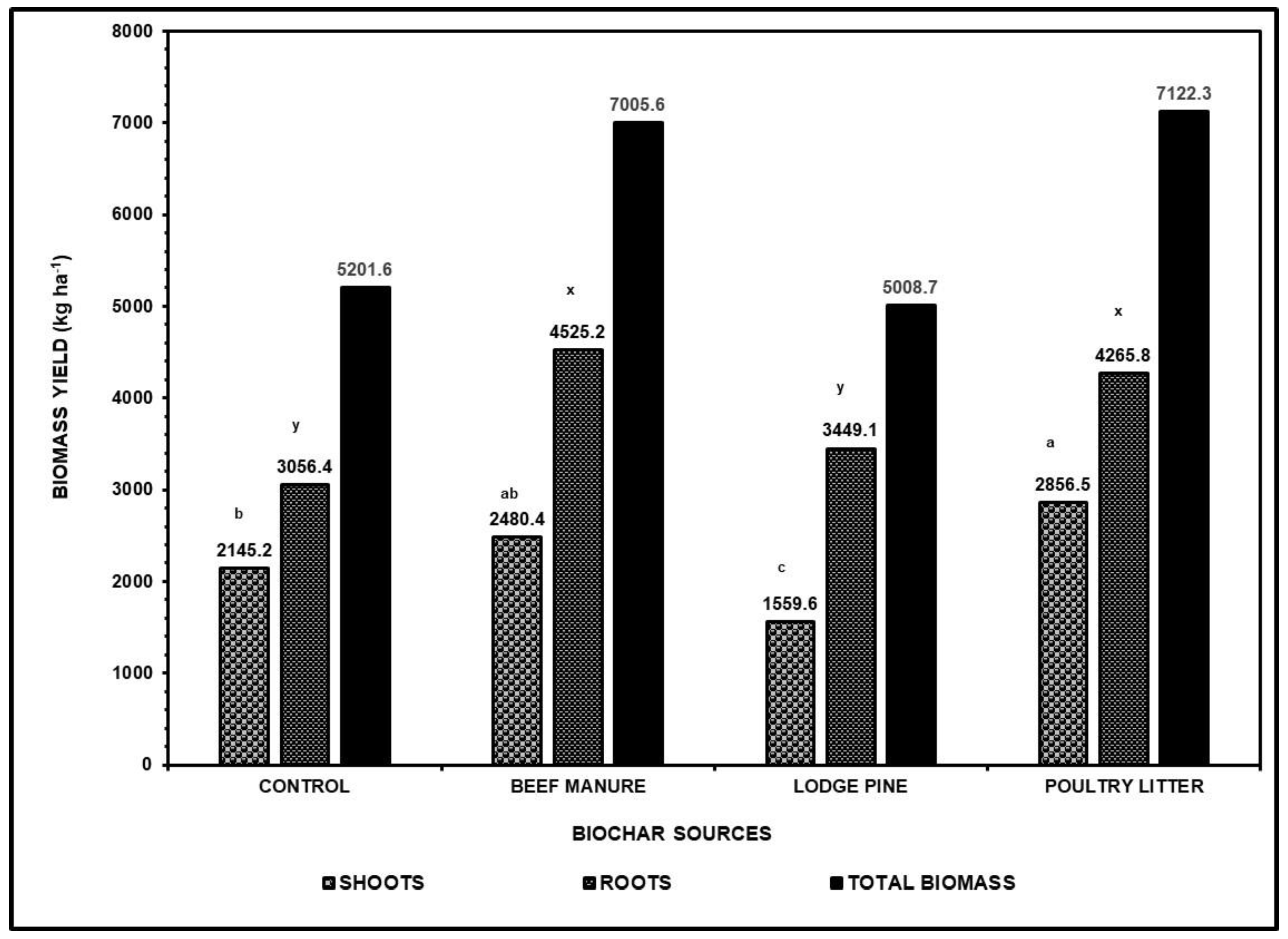
Our results validate the beneficial effects of biochars in combination with the beef cattle manure compost on enhancing the shoot and root biomass and nutritional uptake of corn grown in mine soil with heavy metal contaminations. The greatest total corn biomass was from soils treated with manure-based biochars (PL and BCM) and the least total biomass was from wood-based biochar (LPP) untreated soils. The shoot and root biomass varied significantly with different biochar additions. Results have suggested that biochar applications in mine soils are more likely to influence the biomass, and the effect could be long lasting. Several factors could have had affected the outcome of our study. For instance, differences in the rapidity of decomposition and chemical stability between manure-based and wood-based biochars. In addition, the C:N ratio of the biochars, age of feedstocks, and the degree of disintegration or particle size of the biochars can govern the amount of nutrients released in the soil [
33,
34]. The C:N ratio of the different biochars that were used in the study are as follows: Poultry litter (8.9) < beef cattle manure (13.8) < lodgepole pine (129.3). Lodgepole pine with wide C:N ratio and low nitrogen content (
Table 1) is associated with a slow decay while PL and BCM with narrow C:N ratio and containing higher nitrogen content may undergo rapid mineralization. The profound differences in the C:N ratio of these biochars can explain the striking difference in the decomposition rates, hence faster release of nutrients from these additions to the soils. The rates of mineralization in biochars may have had significant effects on the biomass and nutrient uptake of crop. Our results confirmed the significant effects of different additions of biochars with or without beef cattle manures on biomass productivity and Cd and Zn uptake of corn. As observed in our study, improvements in the corn biomass yield after the biochar addition is often attributed to increased water and nutrient retention, improved biological properties and CEC and improvements in soil pH.
Manure-based biochars, particularly when pyrolyzed at higher temperatures (500 °C and above), have been shown to have strong metal binding capabilities [
35]; results which are supported by this study with concentrations of water-soluble Cd and Zn lowest in soils amended with both manure-based biochars (PL and BCM). Concomitantly, additions of PL and BCM resulted in increased total plant biomass yields as compared with the untreated soils and wood-based biochar amendments (PLL). These results are potentially indicative of reduced plant toxicity, though another possibility is that reductions in the available soil of Zn and Cd resulted in reduced stress on soil rhizosphere communities. Rhizospheric microbial communities provide critical ecosystem services, including nutrient cycling and uptake [
36], which result in increased soil fertility. Ippolito et al. [
37] previously demonstrated that heavy metal concentrations can have a deleterious effect on microbial community diversity, and additional studies have shown reductions in microbial abundance when faced with increased soil heavy metal concentrations, both of which can negatively impact soil health.
The use of biochar has been investigated for in situ remediation of contaminated lands associated with plants [
38,
39]. Our results suggest that the incorporation of biochar enhanced phytostabilization of Cd and Zn with concentrations of water-soluble Cd and Zn lowest in soils amended with both manure-based biochars (PL and BCM) while improving the biomass productivity of corn. The biochar application has been shown to be effective in metal immobilization, thereby reducing the bioavailability and phytotoxicity of heavy metals. They also reported that the addition of biochars improve agronomic properties by increasing nutrient availability and microbial activity. The uptake of heavy metals by most plant species decreases in the presence of biochars [
40,
41,
42]. Further benefits of adding biochars to soil have also been reported; these include the adsorption of dissolved organic carbon [
43], increases in soil pH and key soil macro-elements [
44], and reductions in trace metals in leachates. Our results support the idea that biochar has proven to be effective at reducing the high concentration of soluble Cd and Zn originating from a contaminated soil and we can now more affirmatively say that sorption is one of the mechanisms by which those metals are retained [
45].
The concentrations of water-soluble Cd and Zn in the soil treated with 2.5% and 5% biochars in combination with the increasing beef cattle manure were considerably lower when compared with the control. These results showed effective lowering of Cd and Zn in mine soils after harvesting of corn may well relate to soil pH and phytostabilization of Cd and Zn due to the application of different additions of biochars, especially the manure-based biochar. Sorption of Cd and Zn in biochars can be due to complexation of the heavy metals with different functional groups present in the biochar, such as Ca
+2 and Mg
+2 [
46], K
+, Na
+ and S [
47], or due to physical adsorption [
47]. Some other compounds present in the ash, such as carbonates, phosphates or sulphates [
48,
49] can also help to stabilize heavy metals by precipitation of these compounds with heavy metals [
13].
Overall, the pH of mine soils was significantly affected by the increasing rate (2.5% to 5.0%) of different additions of biochars. The soil pH of mine soil treated with 2.5% and 5.0% BCM was increased from 5.2 to 5.61. Similarly, the pH of soils treated with 2% and 5% LPP was increased slightly from 4.7 to 4.8. A much higher increase in the pH of mine soils with 5% PL (6.5) when compared with the control. The application of biochar in our study increased the soil pH and thus enhanced the phytostabilization of metals and our results agreed with the findings of Park et al. [
49] and Zhang et al. [
50]. The specific mechanism of metal immobilization in the biochar treatments, with increased soil pH, was likely a result in the formation of precipitates such as Cd(OH)
2 and Zn(OH)
2. For Cd and Zn, the speciation of which in soil solution is more dominated by free metal ion. Shuman [
51] reported that at a pH above eight, chemical precipitation took place and therefore retention of Zn in the soil was due to fixation as a solid phase. Singh and Abrol [
52] also concluded that above pH 7.9, pH-pZn curves for different soil systems merged and precipitation reactions were controlling Zn retention.
Metal adsorption in the soil, in addition to pH, organic matter has overriding importance on metal solubility and retention in many soils [
53]. Few reports in the literature about soil amendments, such as lime and compost being used to reduce the bioavailability of heavy metals [
54]. Biochars can also stabilize heavy metals in soils and thus reduce plant uptake [
13]. Addition of soil organic matter in the form of BCM has been recognized as a critical component in the retention of heavy metals in our study. For example, soils treated with 5% BA (PL, BCM, or LPP) when combined with 5% BCM had the lowest concentrations of water-soluble Cd and Zn in the soil. A decreasing trend was noted on the concentrations of water-soluble Cd and Zn in soils with increasing rates of the manure-based compost. The addition of MBC may have enhanced the redistribution of Cd and Zn fractions in the soils and enhanced the phytostabilization and bioavailability of these metals [
55]. Our results showed that heavy metal concentrations of Cd and Zn in the plants could be profoundly affected by the amount of plant available heavy metals in the soil. Additionally, it is possible that the increase in soil pH caused by the biochar application could have had enhanced the adsorption and complexation of Cd and Zn on biochar, which caused a decrease in water-soluble Cd and Zn in the soil at 5% level of biochars in our study. It has been shown that organic materials can strongly bind heavy metals such as Cu, Pb, Cd, Zn, and Ni. The solubility of the metals depends mainly on the metal loading over soil sorbents, pH, and the concentration of dissolved organic matter in the soil solution [
56].
Another important part of this study is on the effect of different additions and application rates of biochars on the bioconcentration factor (BCF) of Cd and Zn in corn shoots and roots. Plant’s ability to accumulate metals from soils can be estimated using BCF, which is defined as the ratio of metal concentration in the shoots or roots to that in the soil. The plant’s ability to translocate metals from the roots to the shoots is measured using the translocation factor (TF), which is defined as the ratio of the metal concentration in the shoots to the roots. As shown in our data (
Table 7), corn has demonstrated a high degree of tolerance factor because we did not see restriction in soil-root and root-shoot transfers. Corn grown in contaminated mine soils can be considered as a hyperaccumulator because it has actively taken up and translocated Cd and Zn into their biomass. Our results showed that BCF of Cd and Zn varied significantly with the different additions and application rates of biochars. Corn applied with 2.5% BCM has the greatest Cd and Zn BCF in the shoots and these results suggest that corn can accumulate large quantities of metal in their shoot tissues when grown in contaminated mine soils. Based on averaged BCF in corn with different additions and rates of biochars, corn can be considered a minor accumulator of Cd and Zn. However, the BCF values of Cd and Zn in corn (
Table 7) were much greater than one, are evident that Cd and Zn in mine soils were highly bio-accumulated and phytostablized. Lu et al. [
57] from their study on the removal of Cd and Zn by water hyacinth suggested that water hyacinth as a moderate accumulator of Cd and Zn with BCF values of 622 and 789, respectively. Another study on the use of biochar and phytostabilization using
Brassica napus L. was conducted to target Cd-polluted soils [
7]. Additionally, the results of Hartley et al. [
58] and Case et al. [
59] showed that biochar can be used in combination with Miscanthus for phytostabilization of Cd and Zn in contaminated soils. Novak et al. [
60] from their most recent study on using blends of compost and biochars concluded that the designer biochar is an important management component in developing successful mine site phytostabilization program.