Poultry Litter, Biochar, and Fertilizer Effect on Corn Yield, Nutrient Uptake, N2O and CO2 Emissions
Abstract
1. Introduction
2. Material and Methods
3. Results and Discussion
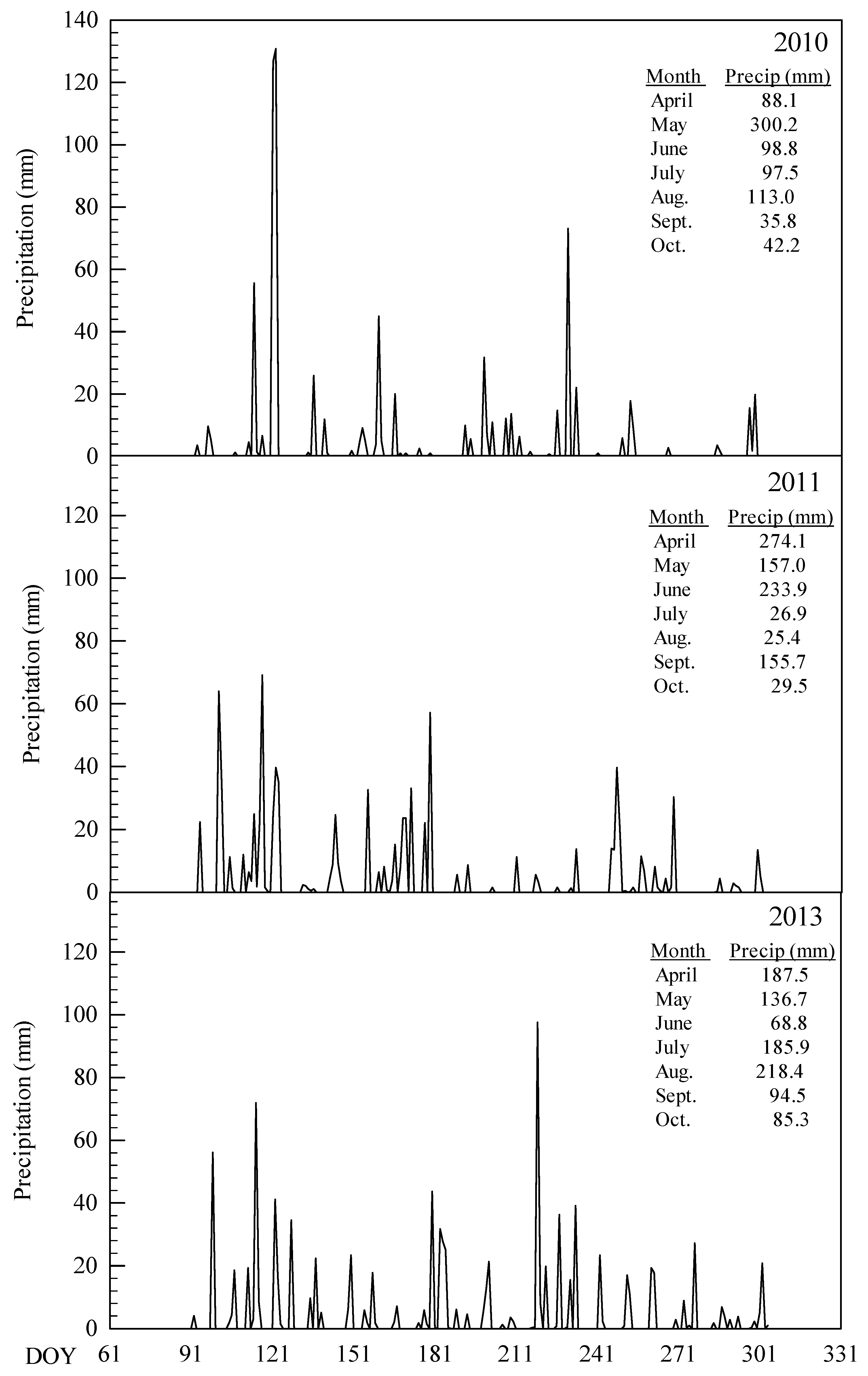
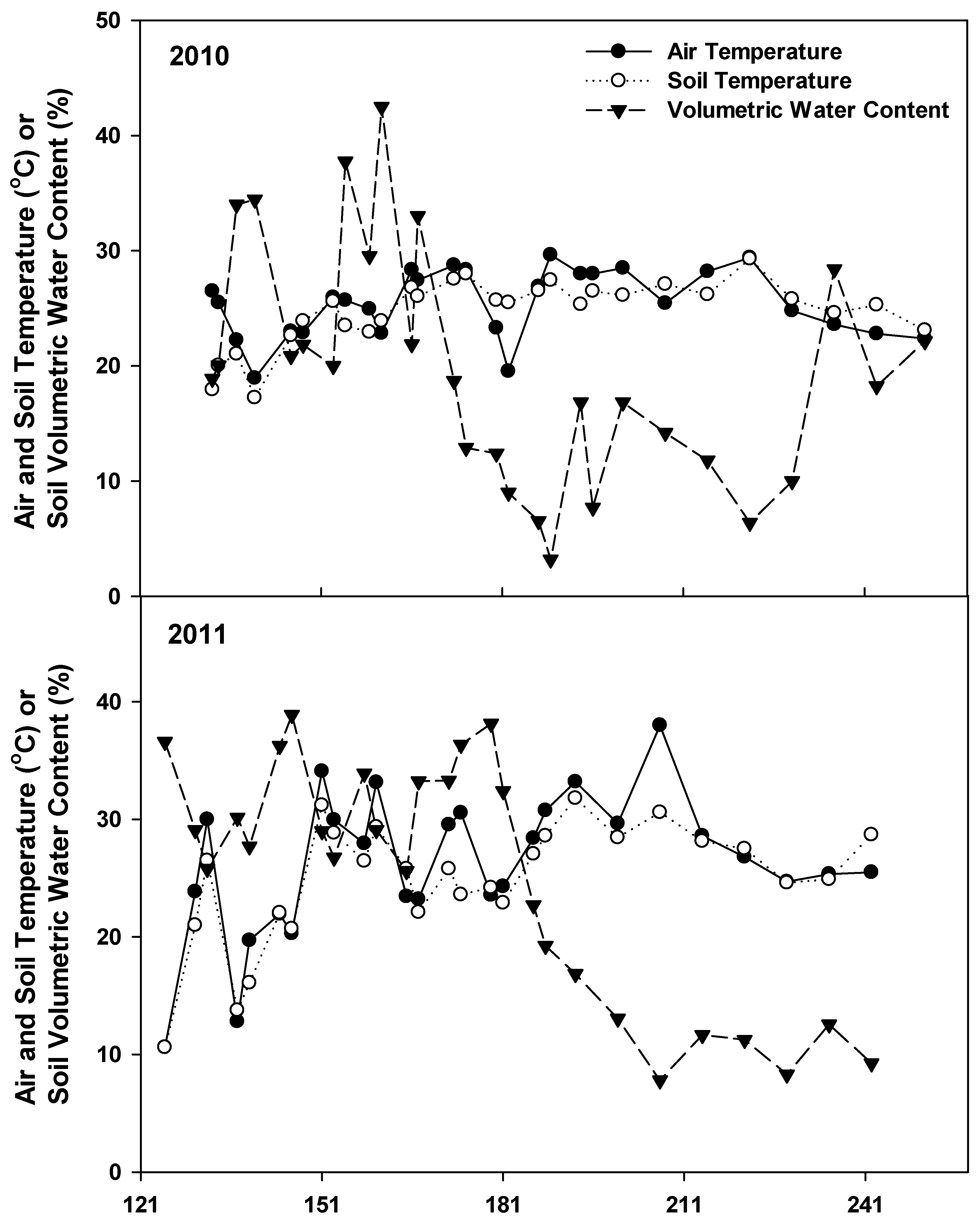
3.1. Environmental Condition
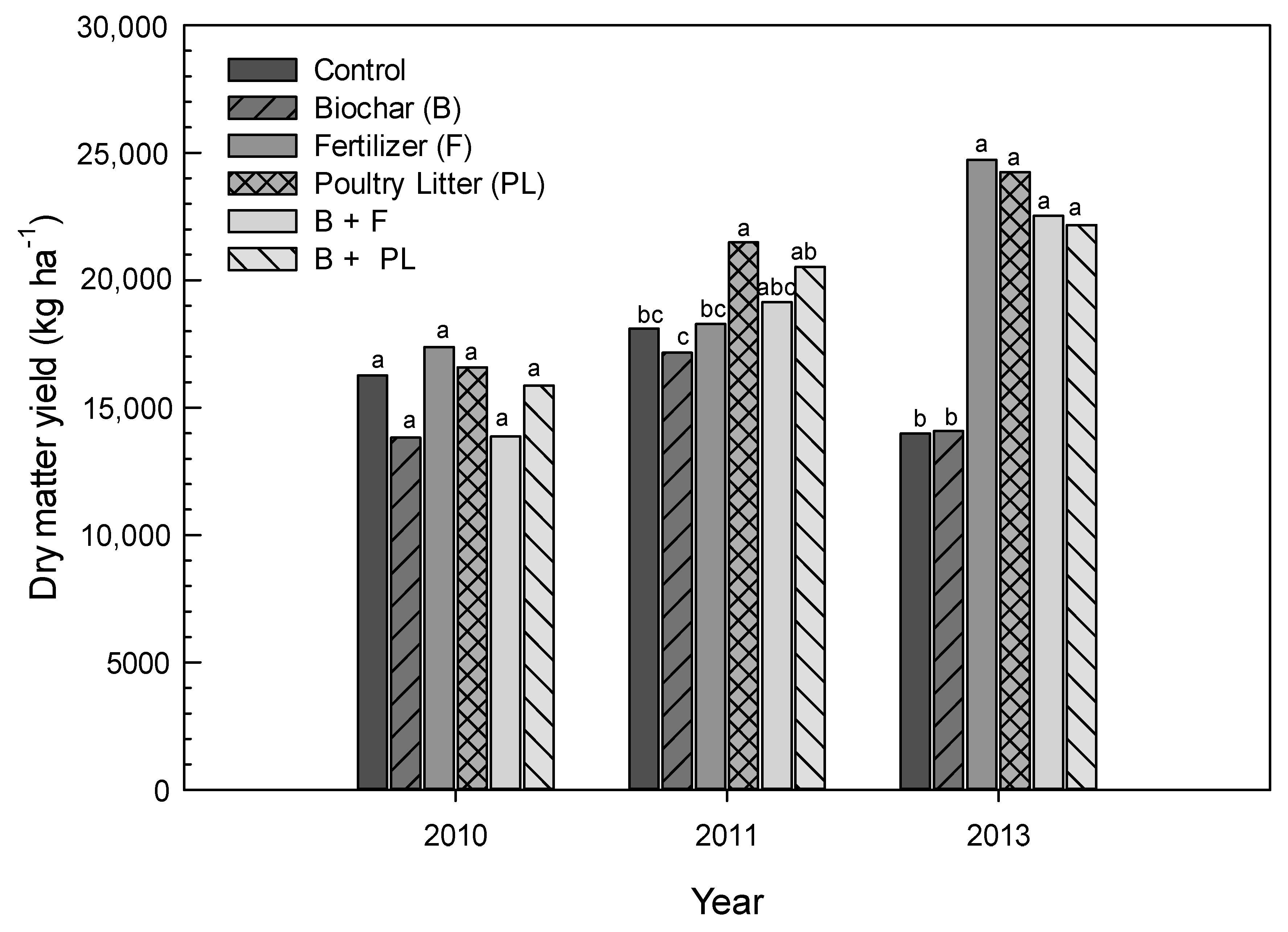
3.2. Corn Dry Matter Yield
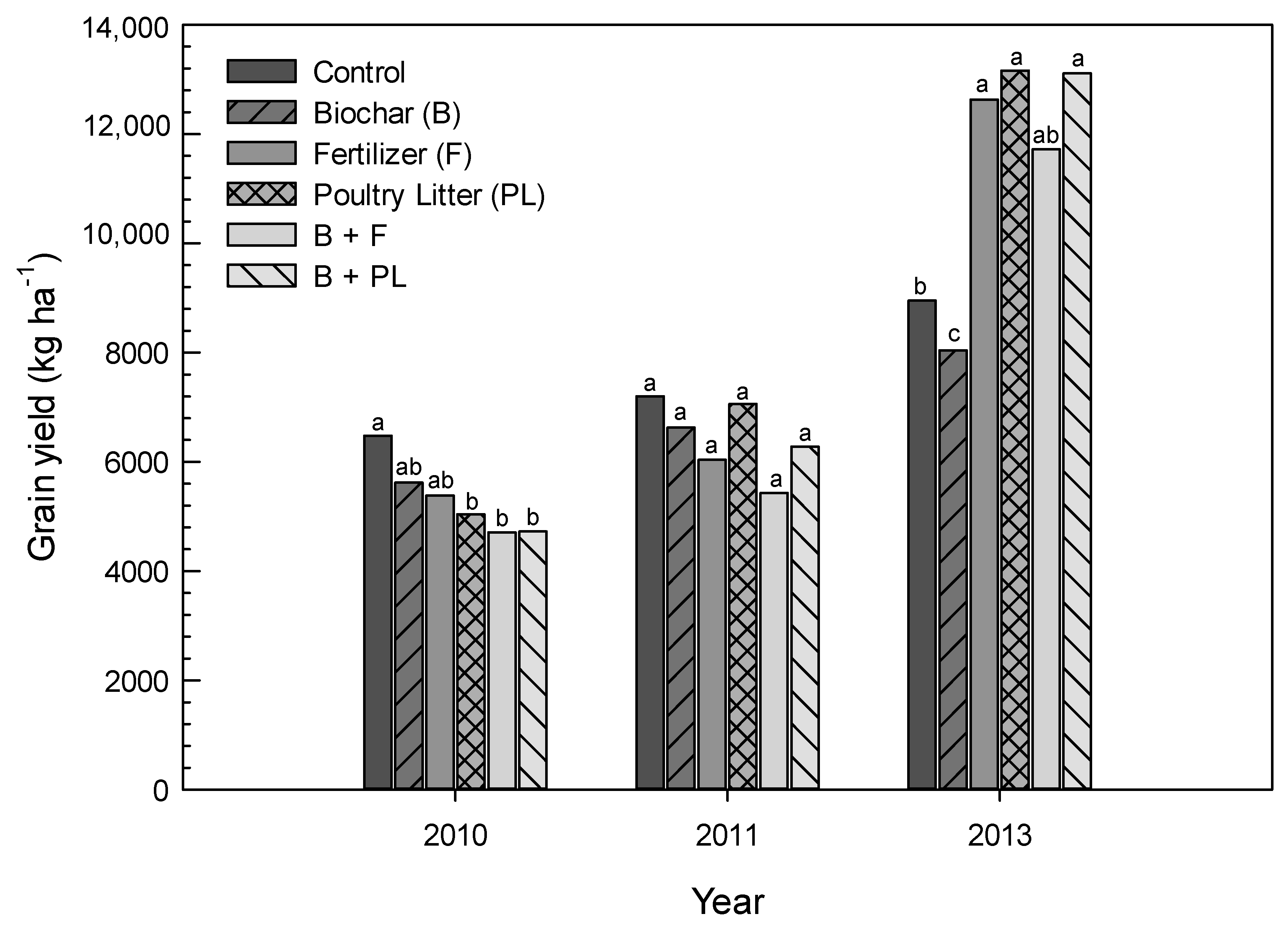
3.3. Corn Grain Yield
3.4. Corn biomass N, P, and K Concentration and Uptake
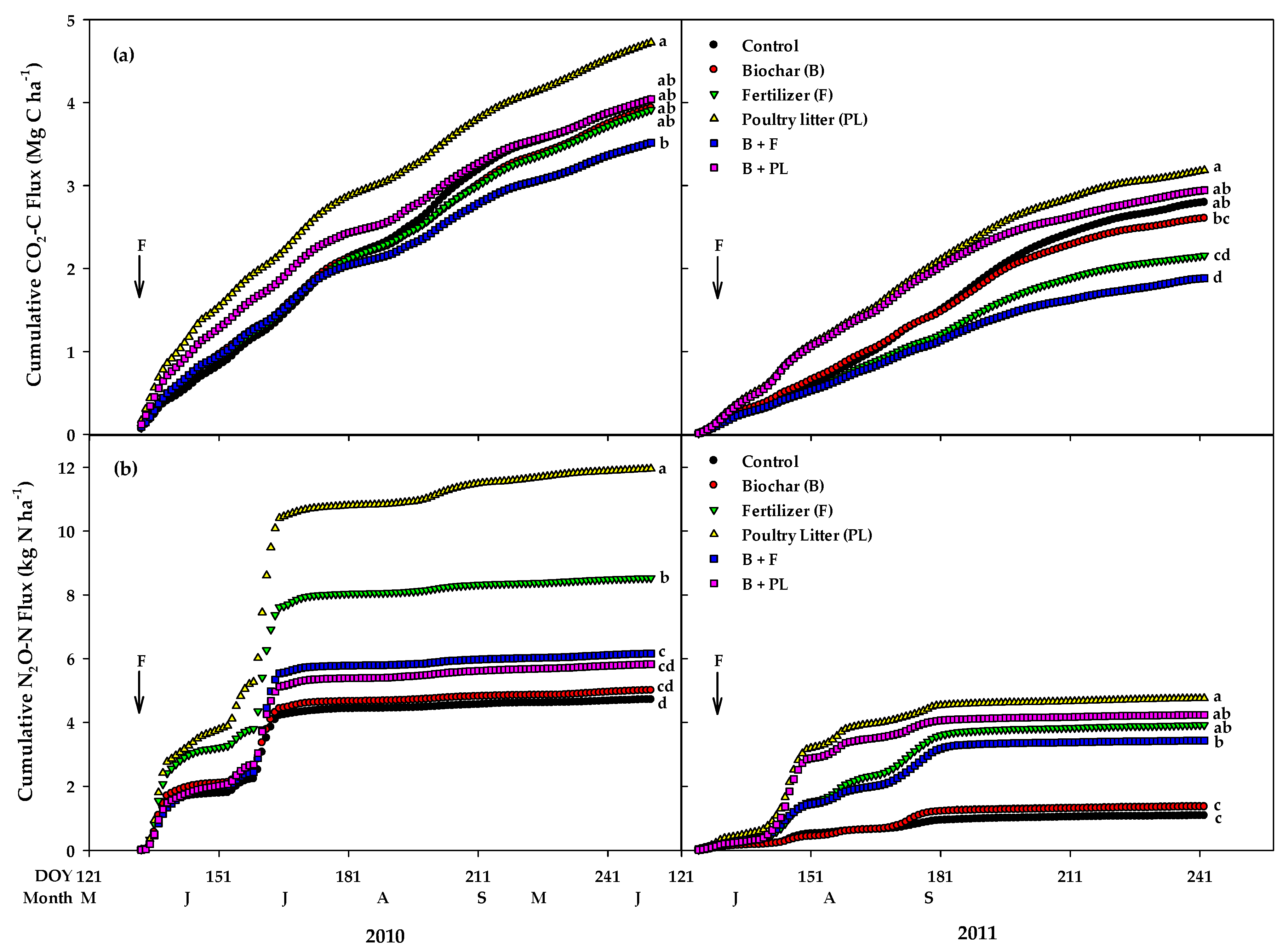
3.5. Quantification of N2O and CO2
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lehmann, J. Bio-Energy in the black. Front. Ecol. Environ. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Spokas, K.A.; Cantrell, K.B.; Novak, J.M.; Archer, D.A.; Ippolito, J.A.; Collins, H.P.; Boateng, A.A.; Lima, I.M.; Lamb, M.C.; McAloon, A.J.; et al. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J. Environ. Qual. 2012, 41, 973–989. [Google Scholar] [CrossRef]
- Lehmann, J.; Abiven, S.; Kleber, M.; Pan, G.; Singh, B.P.; Sohi, S.P.; Zimmerman, A.R. Persistence of biochar in soil. In Biochar for Environmental Management: Science, Technology and Implementation, 2nd ed.; Lehmann, J.L., Joseph, S., Eds.; Earthscan: London, UK, 2015; pp. 235–282. [Google Scholar]
- Fowles, M. Black carbon sequestration as an alternative to bioenergy. Biomass Bioenergy 2007, 31, 426–432. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal-a review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Theis, J.; Luizao, R.J.; Peterson, J.; et al. Black carbon increases cation exchange capacity in soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic values of green-waste biochar as soil amendment. Aust. J. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Biederman, L.A.; Harpole, W.S. Biochar and its effects on plant productivity and nutrient cycling: A meta-analysis. GCB Bioenergy 2013, 5, 202–214. [Google Scholar] [CrossRef]
- Clough, T.J.; Condron, L.M. Biochar and the nitrogen cycle: Introduction. J. Environ. Qual. 2010, 39, 1218–1223. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, C.J.; Fitzgerald, J.D.; Hipps, N.A. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 2010, 337, 1–18. [Google Scholar] [CrossRef]
- Lehmann, J.; Rilling, M.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar effects on soil biota: A review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- Novak, J.M.; Spokas, K.A.; Cantrell, K.B.; Ro, K.S.; Watts, D.W.; Glaz, B.; Busscher, W.J.; Hunt, P.G. Effects of biochar and hydrochars produced from lignocellulosic and animal manure on fertility of a Mollisol and Entisol. Soil Use Manag. 2014, 30, 175–181. [Google Scholar] [CrossRef]
- Spokas, K.A.; Reicosky, D. Impacts of sixteen different biochars on soil greenhouse gas production. Ann. Environ. Sci. 2009, 3, 179–193. [Google Scholar]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joesph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 2010, 327, 235–246. [Google Scholar] [CrossRef]
- Novak, J.M.; Lima, I.M.; Xing, B.; Gaskin, J.W.; Steiner, C.; Das, K.C.; Ahmedna, M.; Rehrah, D.; Watts, D.W.; Busscher, W.J.; et al. Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Soil Sci. 2009, 3, 195–206. [Google Scholar]
- Novak, J.M.; Ro, K.; Ok, Y.S.; Sigua, G.; Spokas, K.; Uchimiya, S.; Bolan, N. Biochars multifunctional role as a novel technology in the agricultural, environmental, and industrial sectors. Chemosphere 2016, 142, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, S.; Verheijen, F.G.; Van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Crane-Droesch, A.; Abiven, S.; Jeffery, S.; Torn, M.S. Heterogeneous global crop yield response to biochar: A meta-regression analysis. Environ. Res. Lett. 2013, 8, 1–8. [Google Scholar] [CrossRef]
- Ronsee, F.; Van Hecke, S.; Dickinson, D.; Prins, W. Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. GCB Bioenergy 2013, 5, 104–115. [Google Scholar] [CrossRef]
- Hass, A.; Gonzalez, J.M.; Lima, I.M.; Godwin, H.W.; Halvorson, J.J.; Boyer, D.G. Chicken manure biochar as liming and nutrient source for acid Appalachian soil. J. Environ. Qual. 2012, 41, 1096–1106. [Google Scholar] [CrossRef]
- Wang, L.; Butterly, C.R.; Wang, Y.; Herath, H.M.; Xi, Y.G.; Xiao, X.J. Effect of crop residue biochar on soil acidity amelioration in strongly acidic tea garden soils. Soil Use Manag. 2014, 40, 119–128. [Google Scholar] [CrossRef]
- Keeney, D.R.; Nelson, D.W. Nitrogen-inorganic forms. In Methods of Soil Analysis, Part 2, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA and SSSA: Madison, WI, USA, 1982; pp. 643–698. [Google Scholar]
- Mehlich, A. Mehlich 3 soil extractant: A modification of Mehlich-2 extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 1409–1416. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Laird, D.L.; Ahmedna, M.; Watts, D.W.; Niandou, M.A. Impact of biochar on fertility of a southeastern Coastal Plain soil. Soil Sci. 2009, 74, 105–112. [Google Scholar] [CrossRef]
- Miller, R.O. High-temperature oxidation: Dry ashing. In Referenced Methods for Plant Analysis; Kalra, V.P., Ed.; Handbook of. CRC Press: Boca Raton, FL, USA, 1998; pp. 53–56. [Google Scholar]
- Livingston, G.P.; Hutchinson, G.L. Enclosure-based measurement of trace gas exchange: Applications and sources of error. In Biogenic Trace Gases: Measuring Emissions from Soil and Water; Matson, P.A., Harriss, R.C., Eds.; Blackwell Sci. Ltd.: London, UK, 1995. [Google Scholar]
- Mosier, A.R.; Halvorson, A.D.; Reule, C.A.; Liu, X.J. Net global warming potential and greenhouse gas intensity in irrigated cropping systems in northeastern Colorado. J. Environ. Qual. 2006, 35, 1584–1598. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, G.L.; Mosier, A.R. Improved soil cover method for field measurements of nitrous oxide fluxes. Soil Sci. Soc. Am. J. 1981, 45, 311–316. [Google Scholar] [CrossRef]
- Yamato, M.; Okimori, Y.; Wibowo, I.F.; Anshori, S.; Ogawa, M. Effects of the application of charred bark of Acaciamangium on the yield of maize, cowpea and peanut and soil chemical properties in south Sumatra, Indonesia. Soil Sci. Plant Nut. 2006, 52, 489–495. [Google Scholar] [CrossRef]
- Steiner, C.; Teixeira, W.G.; Lehmann, J.; Nehls, T.; Macedo, J.; Blum, W.H.; Zech, W. Long term effect of manure, charcoal and mineral fertilization on crop production and fertility on a highly weathered Central Amazonian upland soil. Plant Soil 2007, 291, 275–290. [Google Scholar] [CrossRef]
- Mau, A.E.; Utami, S.R. Effects of biochar amendment and arbuscular mycorrhizal fungi inoculation on availability of soil phosphorus and growth of maize. J. Degr. Min. Lands Manag. 2014, 1, 69–74. [Google Scholar]
- Ahmad, M.A.; Asghar, H.N.; Saleem, M.; Khan, M.Y.; Zahir, Z.A. Synergistic effect of rhizobia and biochar on growth and physiology of maize. Agron. J. 2015, 107, 2327–2334. [Google Scholar] [CrossRef]
- Adejumo, S.A.; Owolabi, M.O.; Odesola, I.F. Agro-physiologic effects of compost and biochar produced at different temperatures on growth, photosynthetic pigment and micronutrients uptake of maize crop. Afr. J. Agric. Res. 2015, 11, 661–673. [Google Scholar] [CrossRef]
- Eghball, B.; Wienhold, B.J.; Gilley, J.E.; Eigenberg, R.A. Mineralization of manure nutrients. J. Soil Water Conserv. 2002, 57, 470–478. [Google Scholar]
- Motavalli, P.P.; Kelling, K.A.; Converse, J.C. First-year nutrient availability from injected dairy manure. J. Environ. Qual. 1998, 18, 180–185. [Google Scholar] [CrossRef]
- Lentz, R.D.; Ippolito, J.A.; Spokas, K.A. Biochar and Manure Effects on Net Nitrogen Mineralization and Greenhouse Gas Emissions from Calcareous Soil under Corn. Soil Sci. Soc. Am. J. 2014, 78, 1641–1655. [Google Scholar] [CrossRef]
- Laird, D.A.; Novak, J.M.; Collins, H.P.; Ippolito, J.A.; Karlen, D.L.; Lentz, R.D.; Sistani, K.R.; Spokas, K.; van Pelt, R.S. Multi-year and multi-location soil quality and crop biomass yield responses to hardwood fast pyrolysis biochar. Geoderma 2017, 289, 46–53. [Google Scholar] [CrossRef]
| Property | Mean | SD |
|---|---|---|
| %H2O | 4.6 | 0.61 |
| %ash | 14.16 | 13.21 |
| %Fixed C | 46 | 8.49 |
| %Volatile C | 54 | 26 |
| %C | 68.29 | 6.86 |
| %H | 2.67 | 0.65 |
| %N | 0.25 | 0.09 |
| %O* | 13.38 | 3.86 |
| O/C | 0.16 | 0.02 |
| H/C | 0.51 | 0.04 |
| %Ca | 0.49 | 0.06 |
| %K | 0.65 | 0.05 |
| %P | 0.03 | na |
| pH (H2O) | 5.59 | 0.61 |
| SSA (m2/g) † | 1.29 | na |
| Total acidity (cmol/100g) | 120 | na |
| Whole Plant Dry Matter Yield | Grain Yield | |||||
|---|---|---|---|---|---|---|
| Treatment (T) | 2010 | 2011 | 2013 | 2010 | 2011 | 2013 |
| kg ha−1 | ||||||
| Control | 16,262a † | 18,100bc | 13,995b | 6480a | 7201a | 8952bc |
| Biochar (B) | 13,828a | 17,168c | 14,090b | 5619ab | 6629a | 8042c |
| Fertilizer (F) | 17,379a | 18,291bc | 24,721a | 5382ab | 6036a | 12,632a |
| Poultry Litter (PL) | 16,581a | 21,495a | 24,245a | 5037b | 7062a | 13,163a |
| B + F | 13,875a | 19,145abc | 22,526a | 4706b | 5430a | 11,723ab |
| B + PL | 15,874a | 20,524ab | 22,170a | 4721b | 6281a | 13,114a |
| LSD(0.05) | 4454 | 2920 | 6276 | 1232 | 1825 | 3557 |
| Whole Plant N, P, and K Concentration | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| N | P | K | |||||||
| Treatment (T) | 2010 | 2011 | 2013 | 2010 | 2011 | 2013 | 2010 | 2011 | 2013 |
| g kg−1 | |||||||||
| Control | 11.1c † | 7.9d | 6.8b | 1.3c | 1.0d | 1.3c | 8.9b | 13.8b | 8.2b |
| Biochar (B) | 12.9b | 8.5d | 7.3b | 1.5bc | 1.2cd | 1.4c | 10.6b | 14.1b | 8.7b |
| Fertilizer (F) | 13.2ab | 12.5ab | 9.6a | 1.5bc | 1.4bc | 1.6b | 9.7b | 15.5ab | 8.6b |
| Poultry Litter (PL) | 13.9ab | 11.6bc | 9.9a | 2.0a | 1.8ab | 2.0a | 11.8ab | 18.5a | 12.7a |
| B + F | 14.0a | 13.3a | 9.2a | 1.7b | 1.5b | 1.6b | 11.4ab | 17.6a | 8.7b |
| B + PL | 14.2a | 10.7c | 9.7a | 2.1a | 1.9a | 2.0a | 13.8a | 18.9a | 13.3a |
| LSD(0.05) | 1.09 | 1.35 | 0.99 | 0.26 | 0.35 | 0.21 | 2.99 | 3.40 | 3.29 |
| Whole Plant N, P, and K Uptake | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| N | P | K | |||||||
| Treatment (T) | 2010 | 2011 | 2013 | 2010 | 2011 | 2013 | 2010 | 2011 | 2013 |
| kg ha−1 | |||||||||
| Control | 182a † | 144b | 95b | 21.0b | 16.5d | 17.6b | 150b | 250c | 116c |
| Biochar (B) | 178a | 146b | 103b | 20.6b | 20.2cd | 19.1b | 145b | 243c | 123c |
| Fertilizer (F) | 230a | 228a | 239a | 26.5ab | 25.8bc | 40.1a | 170ab | 283bc | 212b |
| Poultry Litter (PL) | 231a | 249a | 240a | 33.9a | 37.8a | 48.5a | 195ab | 398a | 306a |
| B + F | 194a | 254a | 207a | 23.0b | 29.5b | 36.0a | 159ab | 337ab | 195b |
| B + PL | 225a | 220a | 215a | 32.5a | 39.0a | 44.8a | 217a | 390a | 287a |
| LSD(0.05) | 62.6 | 37.5 | 68.0 | 9.40 | 6.86 | 14.08 | 66.7 | 75.0 | 53.9 |
| Treatment | CO2-C Emissions | N2O-N Emissions | N2O-N Emission Factor ‡ | Yield Scaled N2O-N Emissions | ||||
|---|---|---|---|---|---|---|---|---|
| 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | |
| Mg ha−1 | kg ha−1 | % | kg N2O-N Mg grain yield−1 | |||||
| Control | 4.04ab † | 2.80ab | 4.72d | 1.09c | - | - | 0.73e | 0.15c |
| Biochar (B) | 3.94ab | 2.61bc | 5.01cd | 1.36c | - | - | 0.91de | 0.21c |
| Fertilizer (F) | 3.92ab | 2.16cd | 8.53b | 3.92ab | 1.70a | 1.26a | 1.82b | 0.63b |
| Poultry litter (PL) | 4.73a | 3.19a | 11.95a | 4.76a | 1.61a | 0.82a | 2.54a | 0.88a |
| B + F | 3.52b | 1.89d | 6.16c | 3.43b | 0.64b | 1.05a | 1.22c | 0.51b |
| B + PL | 4.04ab | 2.94ab | 5.83cd | 4.24ab | 0.25b | 0.70a | 1.09cd | 0.71ab |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sistani, K.R.; Simmons, J.R.; Jn-Baptiste, M.; Novak, J.M. Poultry Litter, Biochar, and Fertilizer Effect on Corn Yield, Nutrient Uptake, N2O and CO2 Emissions. Environments 2019, 6, 55. https://doi.org/10.3390/environments6050055
Sistani KR, Simmons JR, Jn-Baptiste M, Novak JM. Poultry Litter, Biochar, and Fertilizer Effect on Corn Yield, Nutrient Uptake, N2O and CO2 Emissions. Environments. 2019; 6(5):55. https://doi.org/10.3390/environments6050055
Chicago/Turabian StyleSistani, Karamat R., Jason R. Simmons, Marcia Jn-Baptiste, and Jeff M. Novak. 2019. "Poultry Litter, Biochar, and Fertilizer Effect on Corn Yield, Nutrient Uptake, N2O and CO2 Emissions" Environments 6, no. 5: 55. https://doi.org/10.3390/environments6050055
APA StyleSistani, K. R., Simmons, J. R., Jn-Baptiste, M., & Novak, J. M. (2019). Poultry Litter, Biochar, and Fertilizer Effect on Corn Yield, Nutrient Uptake, N2O and CO2 Emissions. Environments, 6(5), 55. https://doi.org/10.3390/environments6050055




