Characterization and Production of Extracellular Polysaccharides (EPS) by Bacillus Pseudomycoides U10
Abstract
:1. Introduction
- (i)
- Although the bacterial EPS has produced by various microorganisms, to the best of our knowledge, no study has focused on EPS production by Bacillus pseudomycoides. In this study, the ability of EPS production of B. pseudomycoides U10 and the total amount of carbohydrate, protein, and uronic acid in EPS were also analyzed.
- (ii)
- One of the limiting factors in the commercial success of biopolymer production is the cost of the sugar substrate used. Therefore, cheaper substrates such as whey and glycerol were tested to detect their effect on EPS production.
- (iii)
- Moreover, SAXS, X-ray diffraction (XRD), scanning electron microscopy (SEM) and thermogravimetric (TGA) experiments were conducted in order to determine the structure of lyophilized EPS produced by B. pseudomycoides U10.
2. Methodology
2.1. Bacterial Strain and Culture Media
2.2. Isolation of Extracellular Polysaccharides (EPS)
2.3. Purification and Analysis of EPS
2.4. X-ray Diffraction (XRD)
2.5. Thermogravimetric Analyses (TGA)
2.6. Small-Angle X-ray Scattering (SAXS)
2.7. Scanning Electron Microscopy (SEM)
3. Results and Discussion
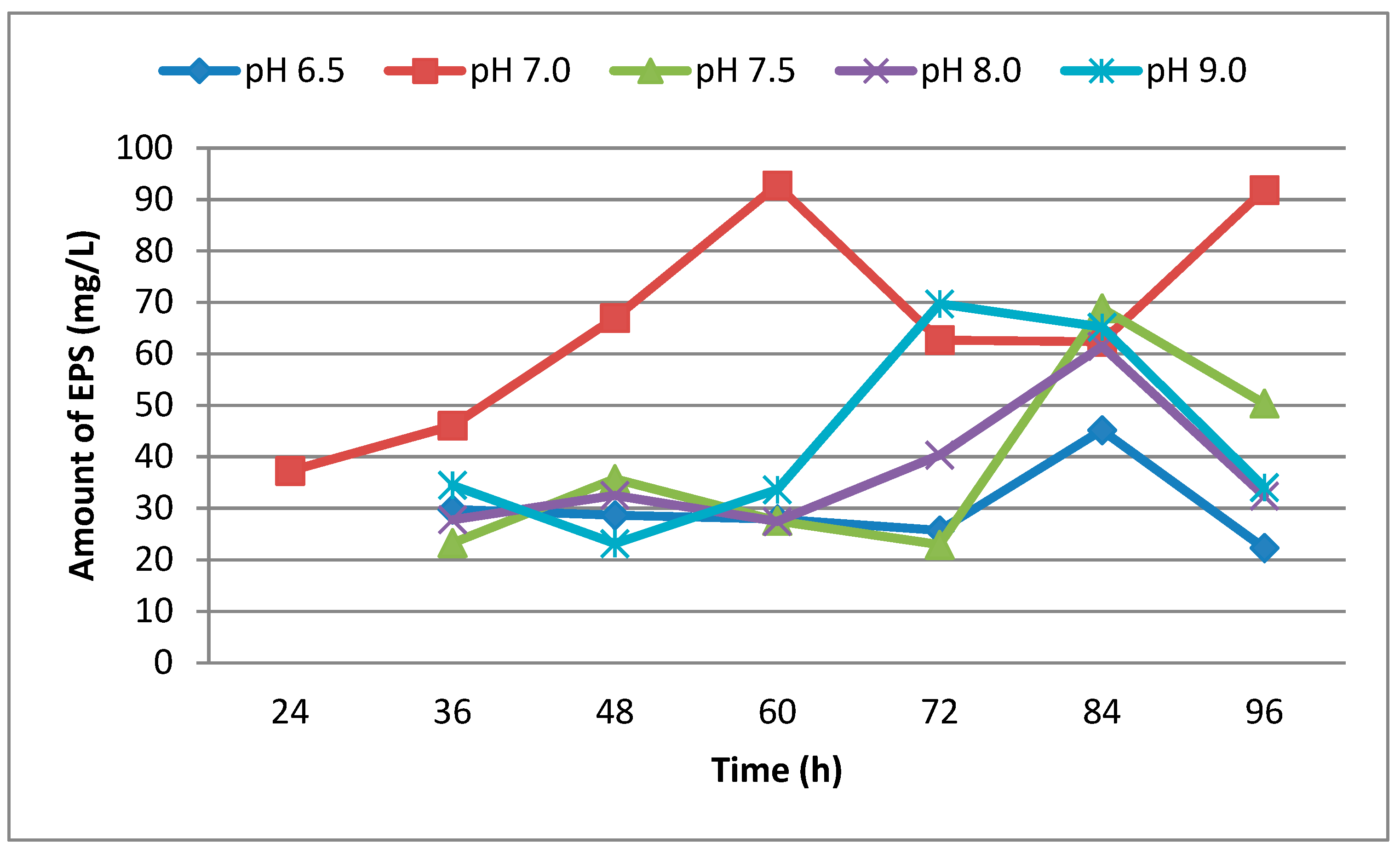
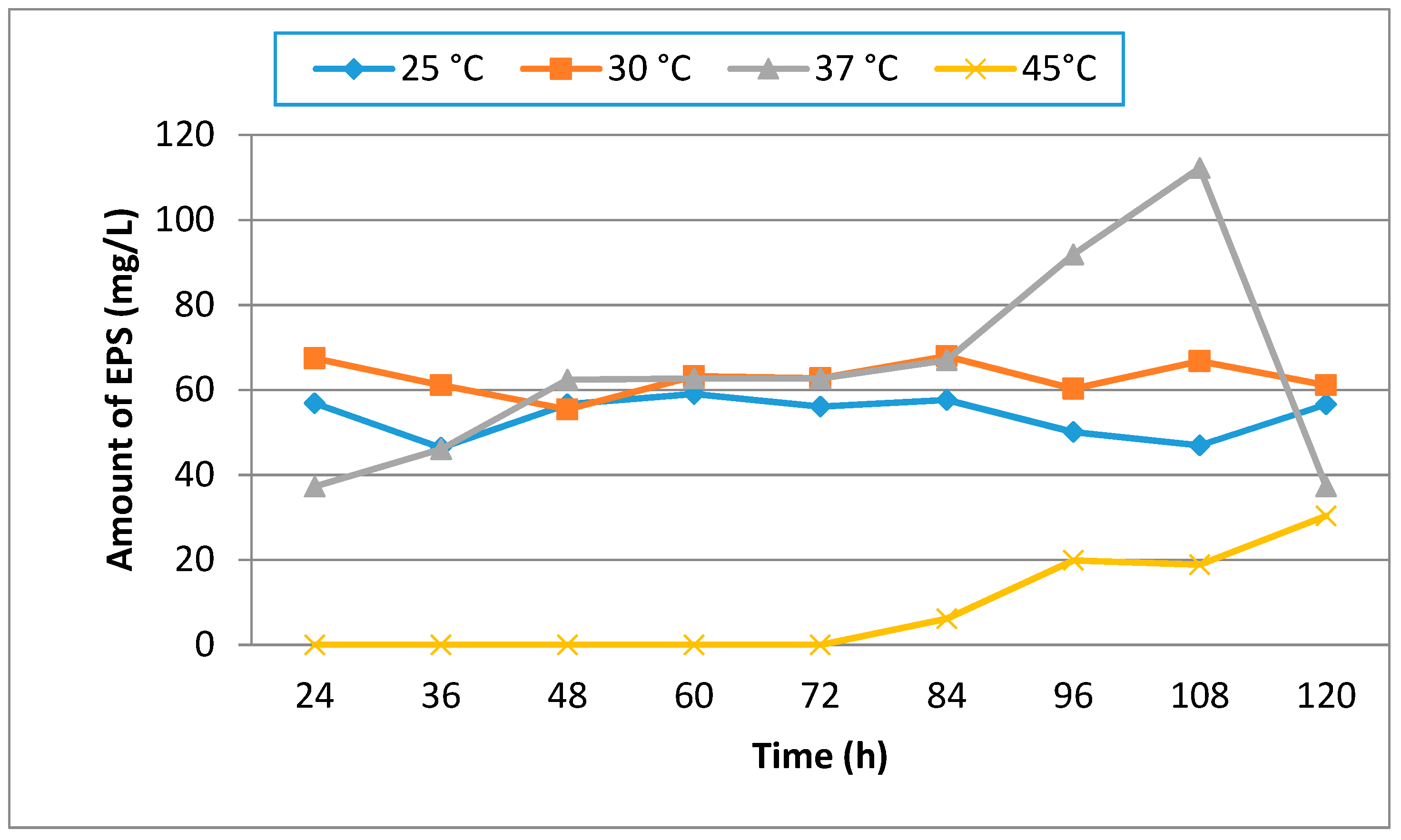
3.1. Effect of Different Media, Initial pH, Temperature and Incubation Time on Production of EPS
3.2. The Effect of Glucose, Whey and Glycerol on the Production of EPS
3.3. Biochemical Composition of EPS
3.4. X-ray Diffraction (XRD) Analysis of EPS
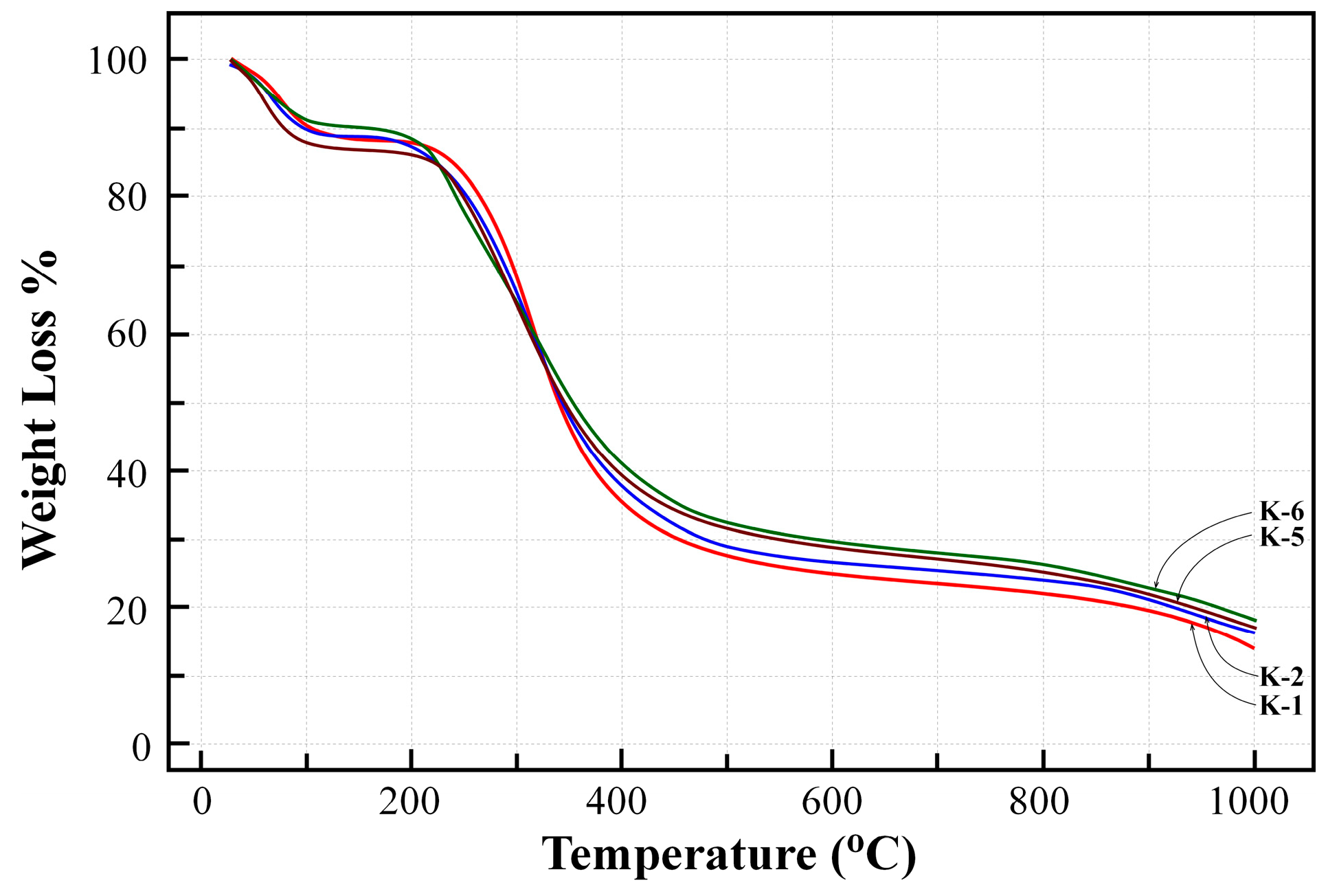
3.5. Thermogravimetric Analysis (TGA) of EPS
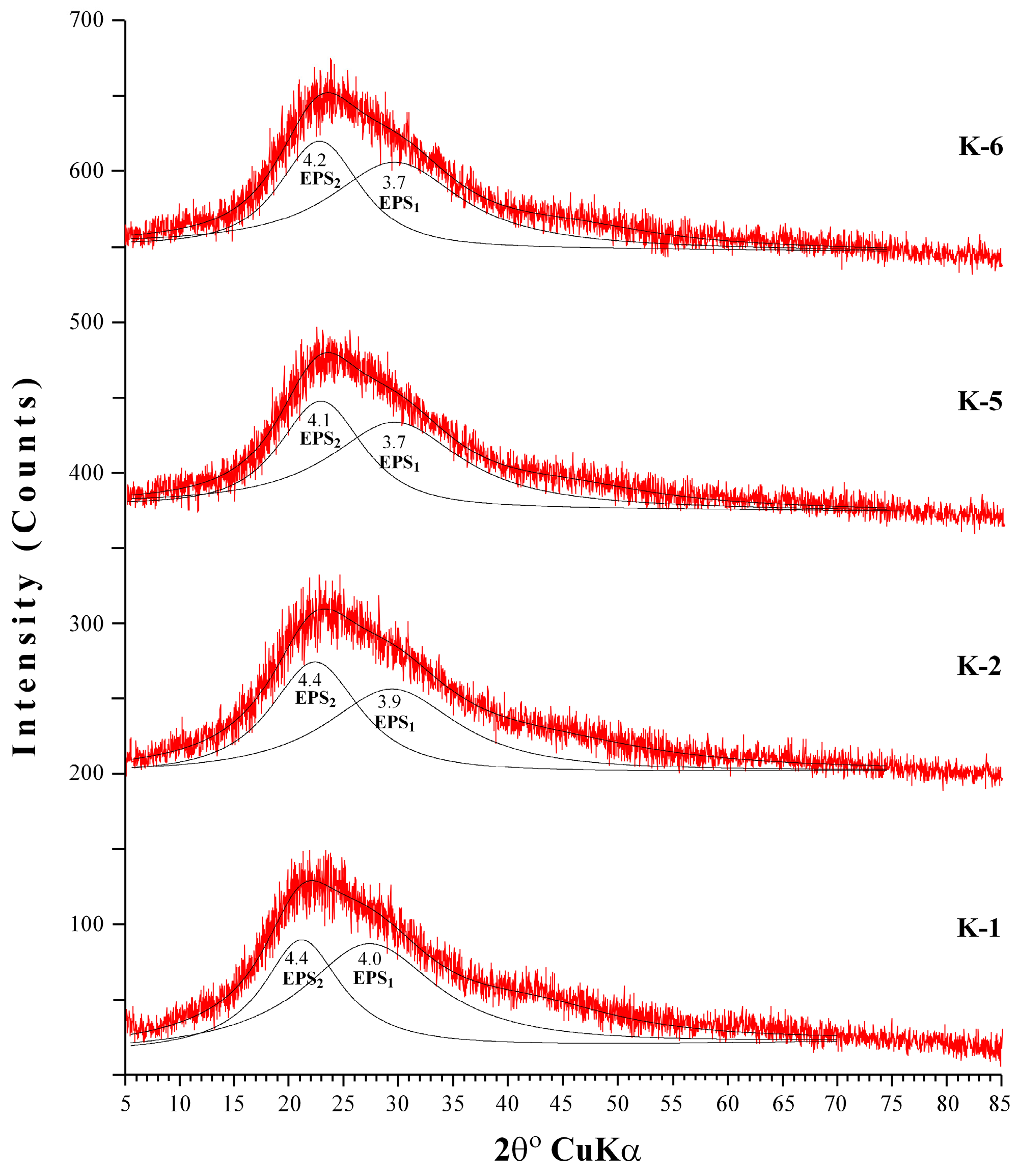
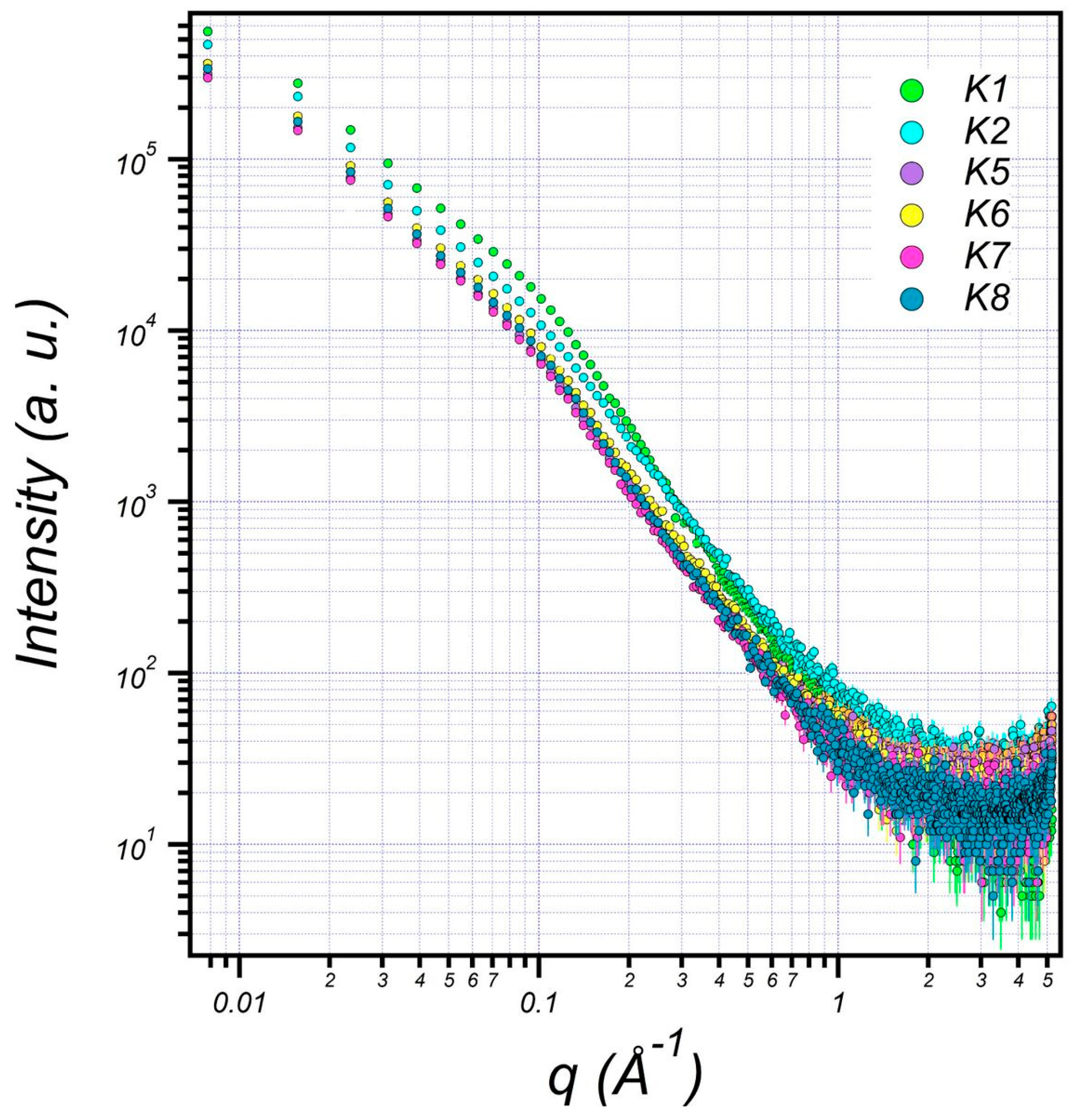
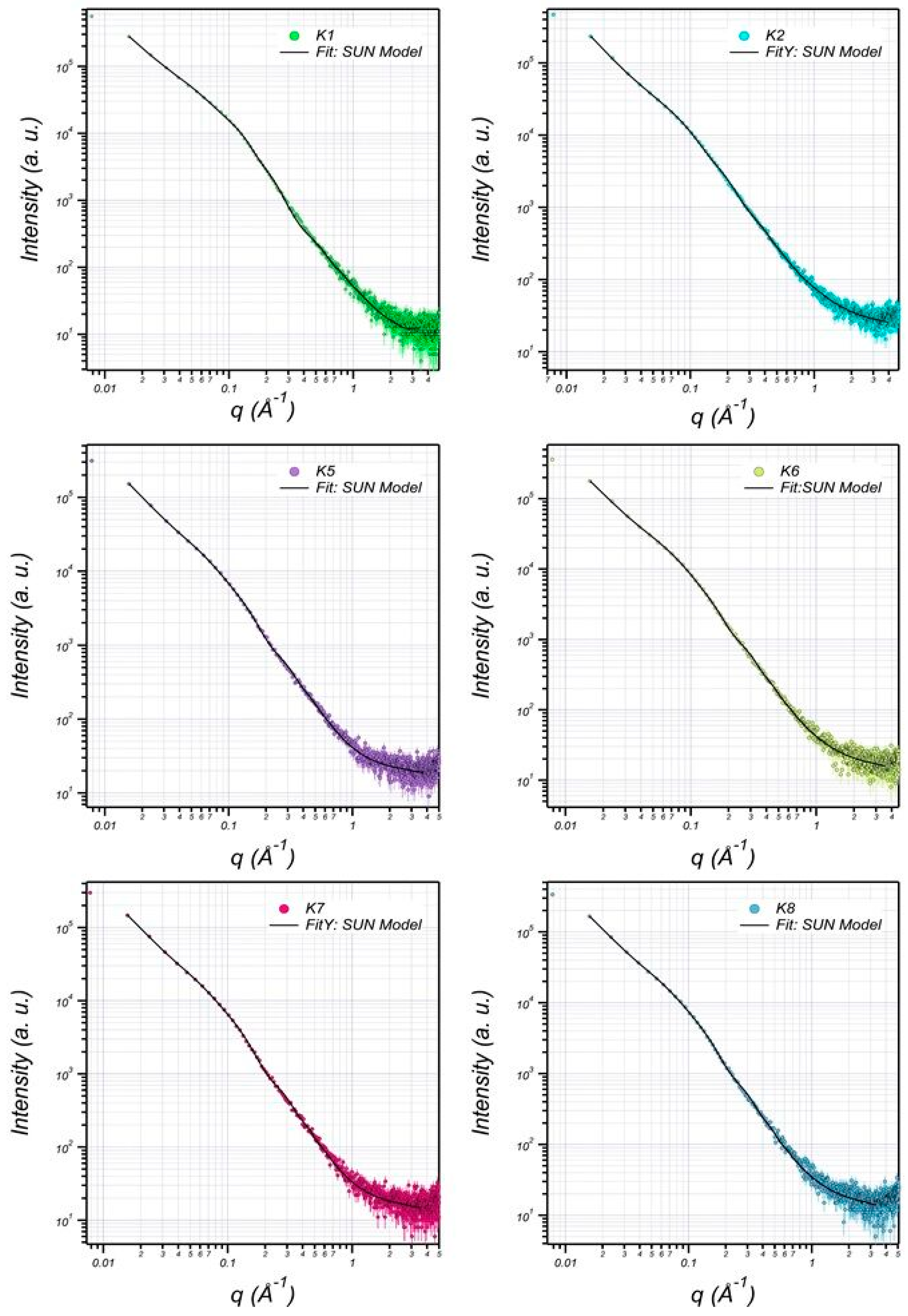
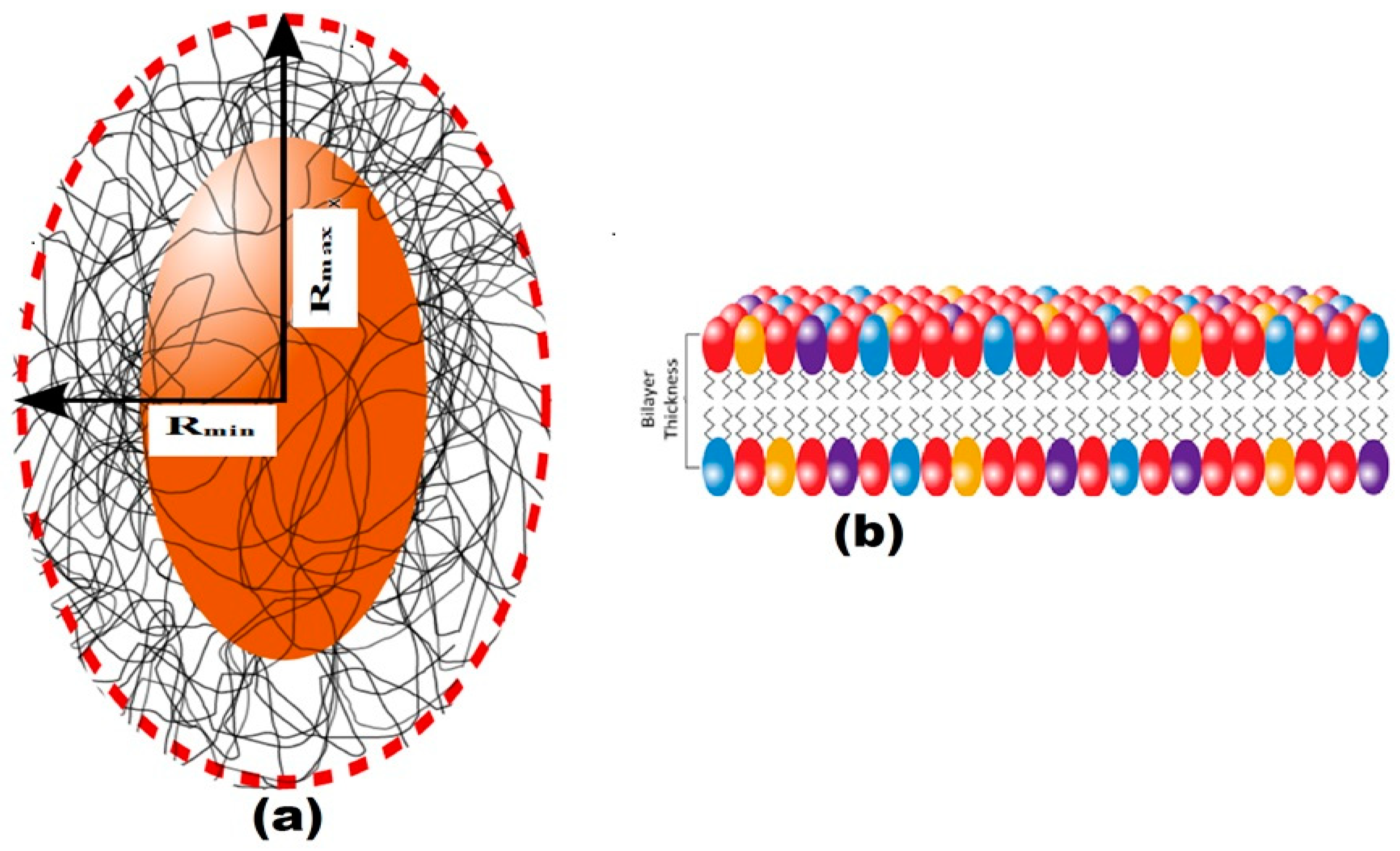
3.6. Structural Characterization of EPS Using Small-Angle X-ray Scattering (SAXS)
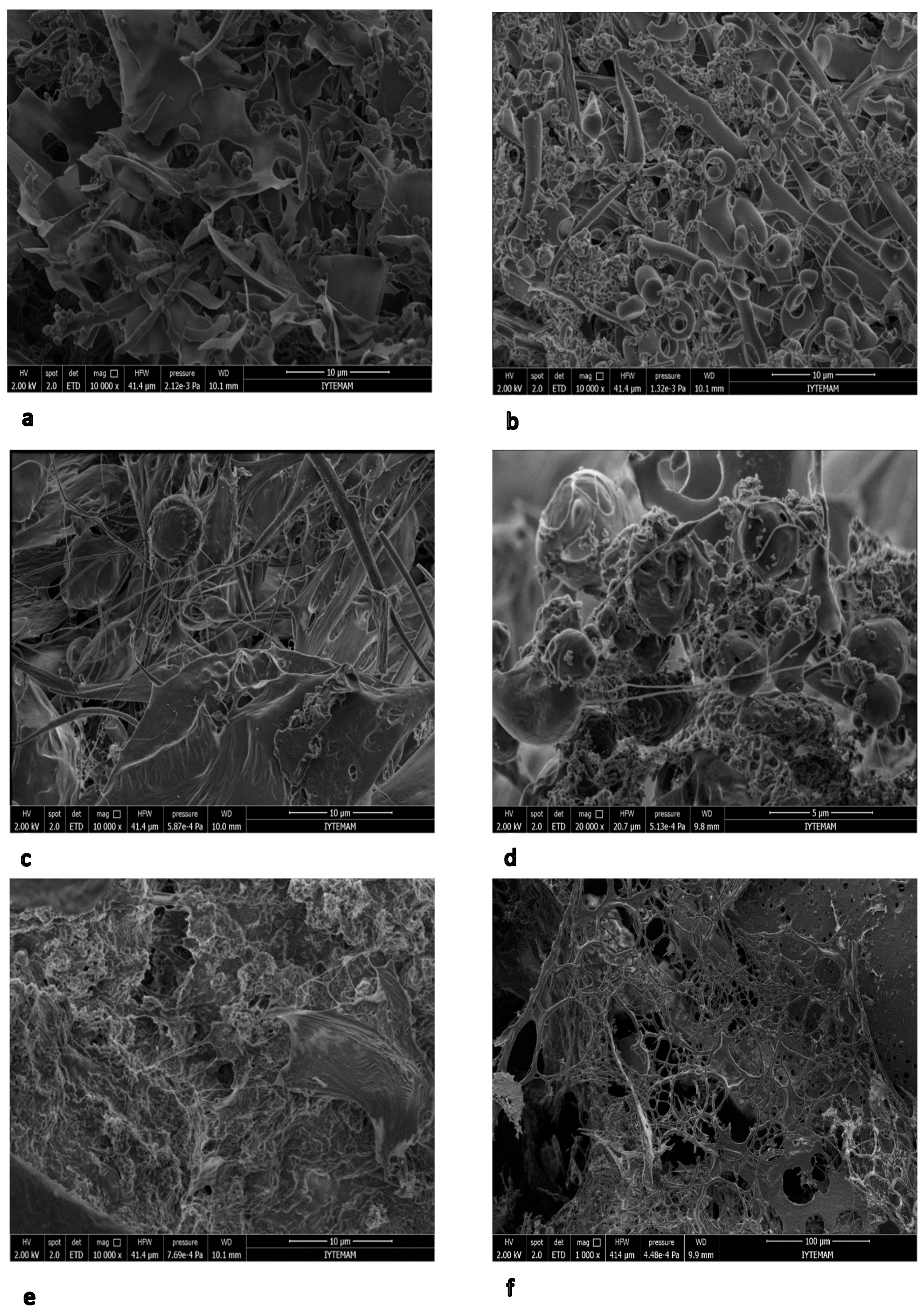
3.7. SEM Analysis of EPS
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Sheng, G.P.; Yu, H.Q.; Li, X.Y. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Frolund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Dignac, M.F.; Urbain, V.; Rybacki, D.; Bruchet, A.; Snidaro, D.; Scribe, P. Chemical description of extracellular polymeric substances: Implication on activated sludge floc structure. Water Sci. Technol. 1998, 38, 45–53. [Google Scholar] [CrossRef]
- Wingender, J.; Neu, T.R.; Flemming, H.C. Microbial Extracellular Polymeric Substances: Characterization, Structure and Function; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1999. [Google Scholar]
- Tian, Y. Behaviour of bacterial extracellular polymeric substances from activated sludge: A review. Int. J. Environ. Pollut. 2008, 32, 78–89. [Google Scholar] [CrossRef]
- Zhang, X.; Bishop, P.L. Biodegradability of biofilm extracellular polymeric substances. Chemosphere 2003, 50, 63–69. [Google Scholar] [CrossRef]
- Rasa, M.; Lefebvre, D.; Derlon, N.; Hamelin, J.; Bernet, N.; Paul, E.; Girbal-Neuhauser, E. Distribution and hydrophobic properties of Extracellular Polymeric Substances in biofilms in relation towards cohesion. J. Biotechnol. 2013, 165, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Li, Y.; Noguera, D.R.; Zhao, N.; Song, Y.; Ding, J.; Zhao, Q.; Cui, F. Adsorption of Cu2+ and Zn2+ by extracellular polymeric substances(EPS) in different sludges: Effect of EPS fractional polarity on binding mechanism. J. Hazard. Mater. 2017, 321, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.D.; Zhang, C.K.; Zhou, Z.; Gong, Z.; Zhou, J.J.; Tao, J.F.; Paterson, D.M.; Feng, Q. Stabilizing Effects of Bacterial Biofilms: EPS Penetration and Redistribution of Bed Stability Down the Sediment Profile. J. Geophys. Res. Biogeosci. 2017, 122, 3113–3125. [Google Scholar] [CrossRef]
- Flemming, H.C.; Leis, A. Sorption Properties of Biofilms, Encyclopedia of Environmental Microbiology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003. [Google Scholar]
- Czaczyk, K.; Myszk, K. Biosynthesis of extracellular polymeric substances (EPS) and its role in microbial biofilm formation. Pol. J. Environ. Stud. 2007, 16, 799–806. [Google Scholar]
- Sun, C.; Wang, J.W.; Fang, L.; Gao, X.D.; Tan, R.X. Free radical scavenging and antioxidant activities of EPS2, an exopolysaccharide produced by a marine filamentous fungus Keissleriella sp. YS 4108. Life Sci. 2004, 75, 1063–1073. [Google Scholar] [CrossRef] [PubMed]
- Iyer, A.; Mody, K.; Jha, B. Emulsifying properties of a marine bacterial exopolysaccharide. Enzym. Microb. Technol. 2006, 38, 220–222. [Google Scholar] [CrossRef]
- Kantar, C.; Demiray, H.; Dogan, N.M. Role of microbial exopolymeric substances (EPS) on chromium sorption and transport in heterogeneous subsurface soils: II. Binding of Cr(III) in EPS/soil system. Chemosphere 2011, 82, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, C.; Rajesh, R.; Kaviarasan, T.; Muthukumar, K.; Kavitake, D.; Shetty, P.H. Synthesis of silver nanoparticles using bacterial exopolysaccharide and its application for degradation of azo-dyes. Biotechnol. Rep. 2017, 15, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, P.H.P.; Grandison, A.S.; Charalampopoulos, D. Microbiological, chemical and rheological properties of low fat set yoghurt produced with exopolysaccharide (EPS) producing Bifidobacterium strains. Food Res. Int. 2013, 51, 15–22. [Google Scholar] [CrossRef]
- Laurienzo, P. Marine Polysaccharides in Pharmaceutical Applications: An Overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, S.; Minami, T.; Nakamura, Y.; Isuda, H.; Kobayashi, H.; Mimura, M.; Urakawa, H.; Kajiwara, K.; Ohno, S. Chain dimensions and scattering function of (1→3)-β-d-glucan simulated by the Monte Carlo method. J. Mol. Struct. THEOCHEM 1997, 395–396, 425–435. [Google Scholar] [CrossRef]
- Jaud, S.; Tobias, D.J.; Brant, D.A. Molecular Dynamics Simulations of Aqueous Pullulan Oligomers. Biomacromolecules 2005, 6, 1239–1251. [Google Scholar] [CrossRef] [PubMed]
- Brant, A.D. Novel approaches to the analysis of polysaccharide structures. Curr. Opin. Struct. Biol. 1996, 9, 556–562. [Google Scholar] [CrossRef]
- Gamini, A.; Paoletti, S.; Toffanin, R.; Micali, F.; Michielin, L.; Bevilacqua, C. Structural investigations of cross-linked hyaluronan. Biomaterials 2002, 23, 1161–1167. [Google Scholar] [CrossRef]
- Hirata, Y.; Sano, Y.; Aoki, M.; Shohji, H.; Katoh, S.; Abe, J.; Hitsukuri, S.; Yamamoto, H. Small angle x-ray scattering studies of moderately concentrated dextran solution. Carbohydr. Polym. 2003, 53, 331–335. [Google Scholar] [CrossRef]
- Mischenko, N.; Denef, B.; Koch, M.H.J.; Reynaers, H. Influence of ionic effects on the ordering and association phenomena in dilute and semidilute carrageenan solutions. Int. J. Biol. Macromol. 1996, 19, 185–194. [Google Scholar] [CrossRef]
- Yuguchi, Y.; Urakawa, H.; Kajiwara, K. The effect of potassium salt on the structural characteristics of gellan gum gel. Food Hydrocoll. 2002, 16, 191–195. [Google Scholar] [CrossRef]
- Dogsa, I.; Kriechbaum, M.; Stopar, D.; Laggner, P. Structure of Bacterial Extracellular Polymeric Substances at Different pH Values as Determined by SAXS. Biophys. J. 2005, 89, 2711–2720. [Google Scholar] [CrossRef] [PubMed]
- Frengova, G.I.; Simova, E.D.; Beshkova, D.M.; Simov, Z.I. Production and monomer composition of exopolysaccharides by yogurt starter cultures. Can. J. Microbiol. 2000, 46, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Roberts, P.A.; Smith, F. Colorimetric Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Hung, C.C.; Santschi, P.H.; Gillow, J.B. Isolation and characterization of extracellular polysaccharides produced by Pseudomonas fluorescens Biovar II. Carbohydr. Polym. 2005, 61, 141–147. [Google Scholar] [CrossRef]
- Hartree, E.F. Determination of Protein: A Modification of the Lowry Method That Gives a Linear Photometric Response; Agricultural Research Council, Unit of Reproductive Physiology and Biochemistry: Cambridge, UK, 2004; p. 307. [Google Scholar]
- Hung, C.C.; Santschi, P.H. Spectrophotometric determination of total uronic acids in seawater using cation-exchange separation and pre-concentration by lyophilization. Anal. Chim. Acta 2001, 427, 111–117. [Google Scholar] [CrossRef]
- Larpin, S.; Sauvageot, N.; Pichereau, V.; Laplace, J.M.; Auffray, Y. Biosynthesis of exopolysaccharide by a Bacillus licheniformis strain isolated from ropy cider. Int. J. Food Microbiol. 2002, 77, 1–9. [Google Scholar] [CrossRef]
- Binupriya, A.R.; Sathishkumar, M.; Ku, C.S.; Yun, S. Sequestration of Reactive Blue 4 by free and immobilized Bacillus subtilis cells and its extracellular polysaccharides. Colloids Surf. B Biointerfaces 2010, 76, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Lu, J.; Lu, L.; Liu, Y.; Wanga, F.; Xiao, M. Isolation, structural characterization and immunological activity of an exopolysaccharide produced by Bacillus licheniformis 8-37-0-1. Bioresour. Technol. 2010, 101, 5528–5533. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.R.; Basak, R.K.; Sen, R.; Adhikari, B. Production of extracellular polysaccharide by Bacillus megaterium RB-05 using jute as substrate. Bioresour. Technol. 2011, 102, 6629–6632. [Google Scholar] [CrossRef] [PubMed]
- Dogan, N.M.; Doganli, G.A.; Dogan, G.; Bozkaya, Ö. Characterization of Extracellular Polysaccharides (EPS) Produced by Thermal Bacillus and Determination of Environmental Conditions Affecting Exopolysaccharide Production. Int. J. Environ. Res. 2015, 9, 1107–1116. [Google Scholar]
- Gandhi, H.P.; Ray, R.M.; Patel, R.M. Exopolymer production by Bacillus species. Carbohydr. Polym. 1997, 34, 323–327. [Google Scholar] [CrossRef]
- Barker, D.J.; Stuckey, D.C. A review of soluble microbial products (SMP) in wastewater treatment systems. Water Res. 1999, 33, 3063–3082. [Google Scholar] [CrossRef]
- Freitas, F.; Alves, V.D.; Pais, J.; Costa, N.; Oliveira, C.; Mafra, L.; Hilliou, L.; Oliveira, R.; Reis, M.A.M. Characterization of an extracellular polysaccharide produced by a Pseudomonas strain grown on glycerol. Bioresour. Technol. 2009, 100, 859–865. [Google Scholar]
- Celik, G.Y.; Aslim, B.; Beyatli, Y. Characterization and production of the exopolysaccharide (EPS) from Pseudomonas aeruginosa G1 and Pseudomonas putida G12 strains. Carbohydr. Polym. 2008, 73, 178–182. [Google Scholar] [CrossRef]
- Guibaud, G.; Comte, S.; Bordas, F.; Dupuy, S.; Baudu, M. Comparison of the complexation potential of extracellular polymeric substances (EPS), extracted from activated sludges and produced by pure bacteria strains, for cadmium, lead and nickel. Chemosphere 2005, 59, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Castro, L.; Zhang, R.; Muñoz, J.A.; González, F.; Blázquez, M.L.; Sand, W.; Ballester, A. Characterization of exopolymeric substances (EPS) produced by Aeromonas hydrophila under reducing conditions. Biofouling 2014, 30, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, B.; Lonngren, J.; Svensson, S. Specific degradation of polysaccharides. Adv. Carbohydr. Chem. Biochem. 1975, 31, 185–239. [Google Scholar]
- Fazio, S.A.; Uhlinger, D.J.; Parker, J.H.; White, D.C. Estimations of Uronic Acids as Quantitative Measures of Extracellular and Cell Wall Polysaccharide Polymers from Environmental Samples. Appl. Environ. Microbiol. 1982, 43, 1151–1159. [Google Scholar] [PubMed]
- Kantar, C.; Honeyman, B.D. Plutonium(IV) complexation with citric and alginic acids at low PuT concentrations. Radiochim. Acta 2005, 93, 757–766. [Google Scholar] [CrossRef]
- Guibaud, G.; Tixier, N.; Bouju, A.; Baudu, M. Use of a polarographic method to determine copper, nickel, and zinc constants of complexation by extracellular polymers extracted from activated sludge. Process Biochem. 2004, 39, 833–839. [Google Scholar] [CrossRef]
- Kantar, C.; Demiray, H.; Mercan Dogan, N.; Dodge, C.J. Role of microbial exopolymeric substances (EPS) on chromium sorption and transport in heterogeneous subsurface soils: I. Cr(III) complexation with EPS in aqueous solution. Chemosphere 2011, 82, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Krumm, S. WINFIT 1.2: Version of November 1996 (The Erlangen geological and mineralogical software collection) of WINFIT 1.0: A public domain program for interactive profile-analysis under WINDOWS. XIII Conference on Clay mineralogy and Petrology, Praha, 1994. Acta Univ. Carol. Geol. 1996, 38, 253–261. [Google Scholar]
- Kumar, C.G.; John, H.S.; Choi, J.W.; Koo, Y.M.; Chang, C.S. Purification and characterization of an extracellular polysaccharide from haloalkalophilic Bacillus sp. I-450. Enzym. Microb. Technol. 2004, 34, 673–681. [Google Scholar] [CrossRef]
- Hamley, I.W. The Physics of Blok Copolymers; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Hamley, I.W.; Castelletto, V. Small-Angle Scattering of Block Copolymers in the Melt, Solution and Crystal States. Prog. Polym. Sci. 2004, 29, 909–948. [Google Scholar]
- Guinier, A. La Diffraction des Rayons X aux Très Faibles Angles: Applications à l’Etude des Phénomènes Ultra-microscopiques. Ann. Phys. 1939, 12, 161–236. [Google Scholar] [CrossRef]
- Guinier, A.; Fournet, G. Small Angle Scattering of X-rays; Wiley: New York, NY, USA, 1955. [Google Scholar]
- Glatter, O.; Kratky, O. Small Angle X-ray Scattering; Academic Press: London, UK, 1982. [Google Scholar]
| Time (h) | Luria-Bertani (LB) | Tryptic Soy Broth (TSB) | Nutrient Broth (NB) |
|---|---|---|---|
| 24 | 37.26 | 8.41 | 19.31 |
| 36 | 46.06 | 13.7 | 34.83 |
| 48 | 66.98 | 31.64 | 45.29 |
| 60 | 92.74 | 46.61 | 78.54 |
| 72 | 62.69 | 19.09 | 32.74 |
| Hours | 6.5 | 7.0 | 7.5 | 8.0 | 9.0 |
|---|---|---|---|---|---|
| 24 | 0.30 | 0.42 | 0.36 | 0.30 | 0.20 |
| 36 | 0.50 | 0.65 | 0.58 | 0.34 | 0.39 |
| 48 | 0.65 | 0.90 | 0.69 | 0.45 | 0.45 |
| 60 | 0.62 | 0.97 | 0.81 | 0.52 | 0.59 |
| 72 | 0.73 | 1.00 | 0.92 | 0.55 | 0.72 |
| 84 | 0.83 | 1.05 | 0.96 | 0.55 | 0.85 |
| 96 | 0.86 | 1.50 | 1.00 | 1.00 | 0.86 |
| Hours | 25 | 30 | 37 | 45 |
|---|---|---|---|---|
| 24 | 0.35 | 0.40 | 0.48 | 0.16 |
| 36 | 0.42 | 0.48 | 0.65 | 0.20 |
| 48 | 0.45 | 0.48 | 0.90 | 0.22 |
| 60 | 0.50 | 0.58 | 0.97 | 0.30 |
| 72 | 0.66 | 0.67 | 1.00 | 0.35 |
| 84 | 0.76 | 0.79 | 1.05 | 0.50 |
| 96 | 0.85 | 0.99 | 1.50 | 0.52 |
| 108 | 0.92 | 1.00 | 1.68 | 0.55 |
| 120 | 1.20 | 1.25 | 1.92 | 0.62 |
| Time (h) | Whey | Glucose | LB (No Glucose and Whey) |
|---|---|---|---|
| 24 | 116.85 | 97.15 | 37.26 |
| 36 | 101.99 | 158.80 | 46.06 |
| 48 | 152.30 | 93.93 | 66.98 |
| 60 | 199.65 | 69.95 | 92.74 |
| 72 | 316.46 | 113.73 | 62.69 |
| 84 | 115.42 | 98.28 | 62.36 |
| 96 | 51.02 | 61.15 | 91.86 |
| Growth Medium | EPSs | Total Carbohydrate | Total Protein | Uronic Acid |
|---|---|---|---|---|
| LB | Dissolved | 380 | 211 | 12 |
| Particulate | 206 | 256 | 9 | |
| LB with whey | Dissolved | 150 | 182 | 16 |
| Particulate | 106 | 308 | 3 | |
| LB with glucose | Dissolved | 464 | 122 | 5 |
| Particulate | 176 | 340 | 5 |
| K1 | K2 | K5 | K6 | K7 | K8 | |
|---|---|---|---|---|---|---|
| Bilayer Thick (delta) (Å) | 14.07 | 14.07 | 12.04 | 12.74 | 12.02 | 12.54 |
| major core (Å) | 1001.52 | 905.85 | 845.75 | 851.85 | 845.65 | 849.85 |
| minor core (Å) | 301.89 | 230.42 | 196.22 | 199.42 | 196.10 | 199.52 |
| major shell (Å) | 1201.46 | 1100.34 | 998.94 | 1001.34 | 998.04 | 1001.94 |
| minor shell (Å) | 405.11 | 301.32 | 242.90 | 249.30 | 242.90 | 249.00 |
| SLD core (Å−2) | 1.65 × 10−6 | 1.15 × 10−6 | 1.21 × 10−6 | 1.11 × 10−6 | 1.21 × 10−6 | 1.11 × 10−6 |
| SLD shell (Å−2) | 1.44 × 10−6 | 1.204 × 10−6 | 1.10 × 10−6 | 1.15 × 10−6 | 1.10 × 10−6 | 1.11 × 10−6 |
| SLD solvent (Å−2) | 9.56 × 10−6 | 9.44 × 10−6 | 9.41 × 10−6 | 9.43 × 10−6 | 9.42 × 10−6 | 9.44 × 10−6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solmaz, K.B.; Ozcan, Y.; Mercan Dogan, N.; Bozkaya, O.; Ide, S. Characterization and Production of Extracellular Polysaccharides (EPS) by Bacillus Pseudomycoides U10. Environments 2018, 5, 63. https://doi.org/10.3390/environments5060063
Solmaz KB, Ozcan Y, Mercan Dogan N, Bozkaya O, Ide S. Characterization and Production of Extracellular Polysaccharides (EPS) by Bacillus Pseudomycoides U10. Environments. 2018; 5(6):63. https://doi.org/10.3390/environments5060063
Chicago/Turabian StyleSolmaz, Kubra Betul, Yusuf Ozcan, Nazime Mercan Dogan, Omer Bozkaya, and Semra Ide. 2018. "Characterization and Production of Extracellular Polysaccharides (EPS) by Bacillus Pseudomycoides U10" Environments 5, no. 6: 63. https://doi.org/10.3390/environments5060063
APA StyleSolmaz, K. B., Ozcan, Y., Mercan Dogan, N., Bozkaya, O., & Ide, S. (2018). Characterization and Production of Extracellular Polysaccharides (EPS) by Bacillus Pseudomycoides U10. Environments, 5(6), 63. https://doi.org/10.3390/environments5060063





