Freshwater Diatoms as Indicators of Combined Long-Term Mining and Urban Stressors in Junction Creek (Ontario, Canada)
Abstract
1. Introduction
2. Materials and Methods
2.1. A Brief History of Mining Around Sudbury and the Resulting Ecological Damages
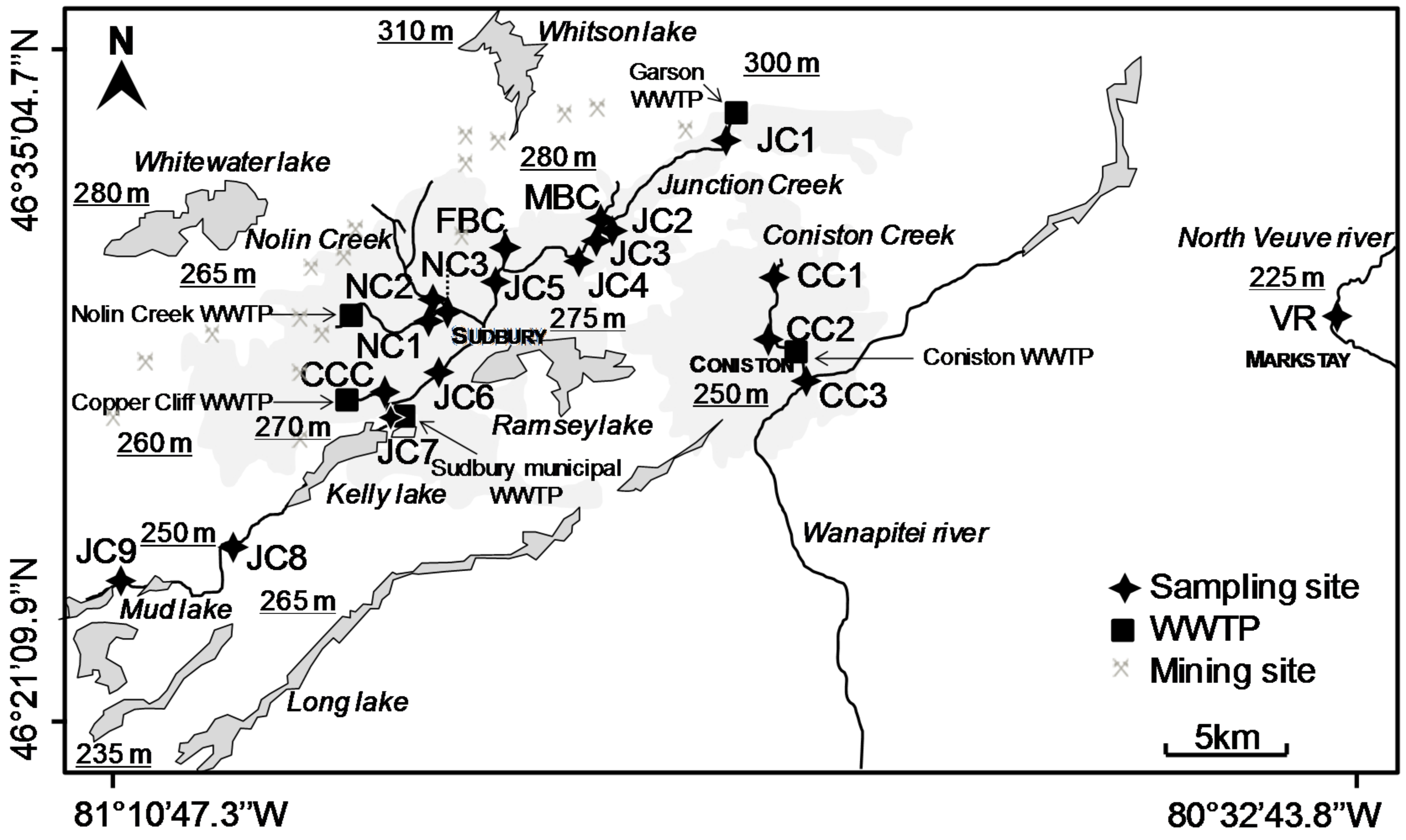
2.2. Study Area
2.3. Water and Biofilm Collection
2.4. Water Chemistry and Diatom Assemblage Analyses
2.5. Toxicity Criteria and CCU Calculation
3. Results and Discussion
3.1. General Water Chemistry
3.2. Metal Concentrations and CCU
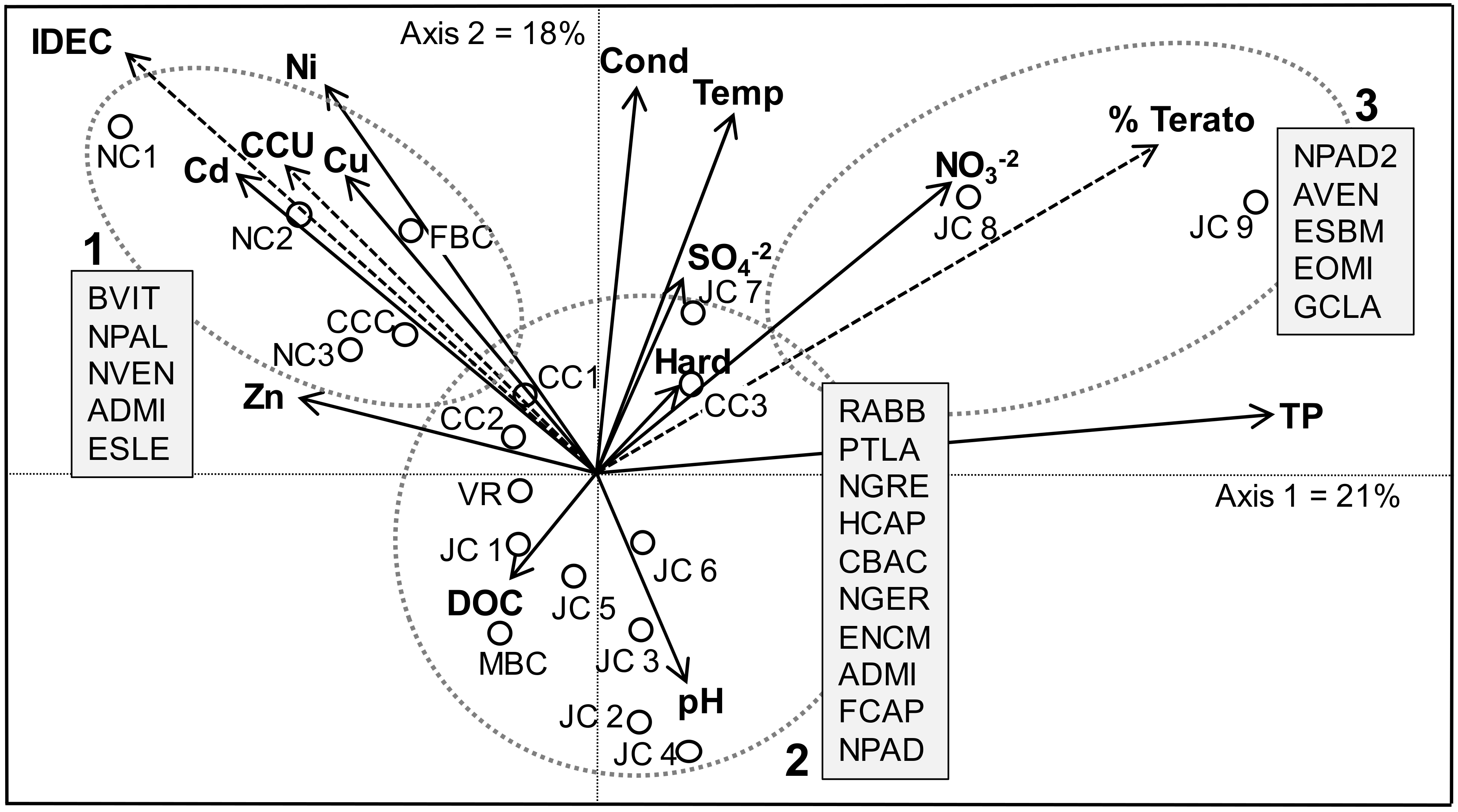
3.3. Relationships between Environmental Factors and Biological Indicators
3.3.1. Biotypology, IDEC Scores and Metal-Tolerant Taxa
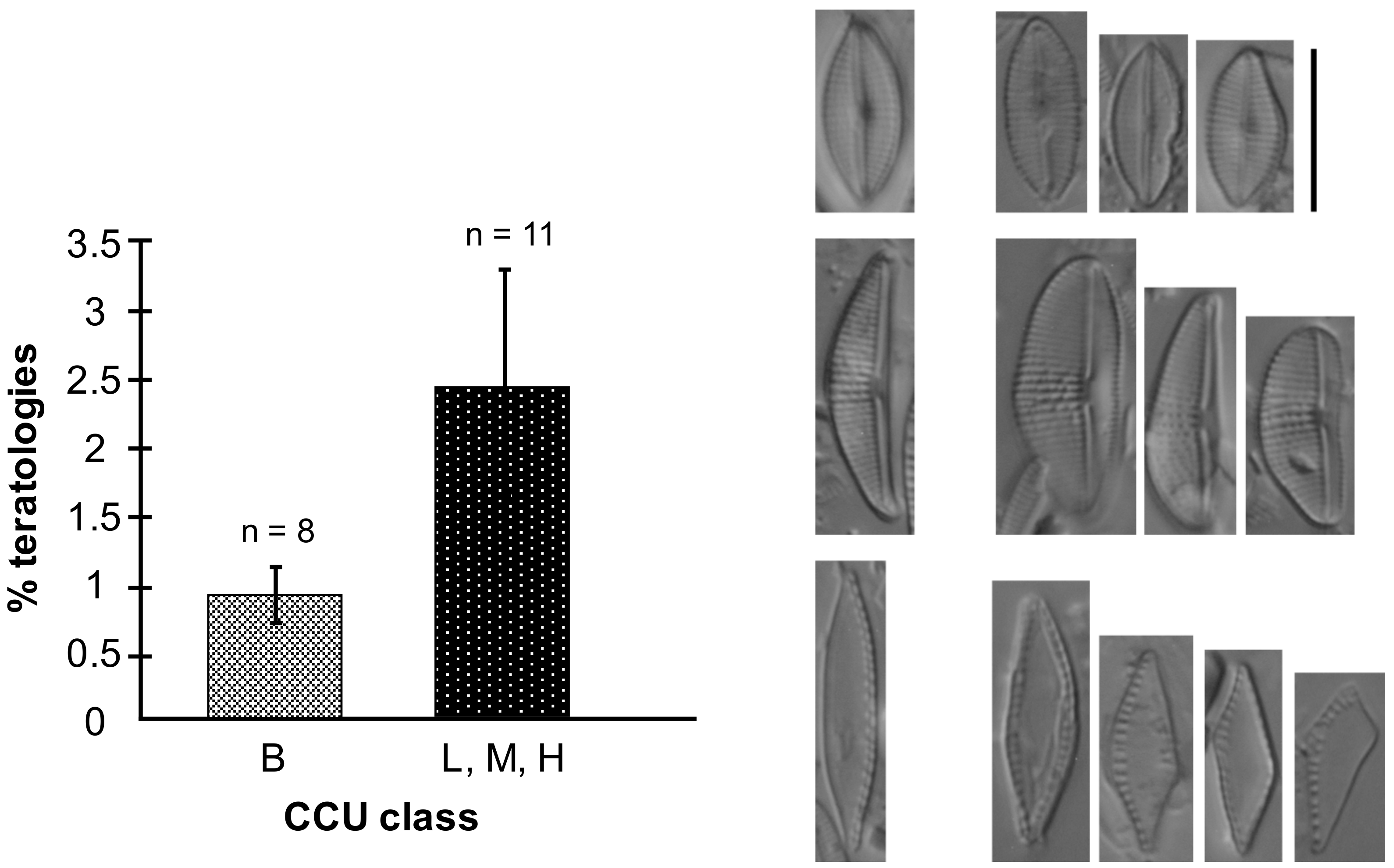
3.3.2. Diatom Teratologies as a Response to Stress
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jaagumagi, R.; Bédard, D. Junction Creek System (Sudbury) Environmental Monitoring Study, September 1999; Ontario Ministry of the Environment Northern Region, Sudbury District Office: Sudbury, ON, Canada, 2002; p. 90. [Google Scholar]
- Gunn, J.; Sarrazin-Delay, C.; Wesolek, B.; Stasko, A.; Szkokan-Emilson, E. Delayed recovery of benthic macroinvertebrate communities in Junction Creek, Sudbury, Ontario, after the diversion of acid mine drainage. Hum. Ecol. Risk Assess. 2010, 16, 901–912. [Google Scholar] [CrossRef]
- Weber, L.P.; Dubé, M.G.; Rickwood, C.J.; Driedger, K.; Portt, C.; Brereton, C.; Janz, D.M. Effects of multiple effluents on resident fish from Junction Creek, Sudbury, Ontario. Ecotoxicol. Environ. Saf. 2008, 70, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Keller, W.; Yan, N.D.; Somers, K.M.; Heneberry, J.H. Crustacean zooplankton communities in lakes recovering from acidification. Can. J. Fish. Aquat. Sci. 2002, 59, 726–735. [Google Scholar] [CrossRef]
- Yan, N.D.; Girard, R.; Heneberry, J.H.; Keller, W.B.; Gunn, J.M.; Dillon, P.J. Recovery of copepod, but not cladoceran, zooplankton from severe and chronic effects of multiple stressors. Ecol. Lett. 2004, 7, 452–460. [Google Scholar] [CrossRef]
- Yan, N.D.; Keller, W.; Somers, K.M.; Pawson, T.W.; Girard, R.E. Recovery of crustacean zooplankton communities from acid and metal contamination: Comparing manipulated and reference lakes. Can. J. Fish. Aquat. Sci. 1996, 53, 1301–1327. [Google Scholar] [CrossRef]
- Scheibener, S.A.; Rivera, N.A.; Hesterberg, D.; Duckworth, O.W.; Buchwalter, D.B. Periphyton uptake and trophic transfer of coal fly-ash-derived trace elements. Environ. Toxicol. Chem. 2017, 36, 2991–2996. [Google Scholar] [CrossRef] [PubMed]
- Fadhlaoui, M.; Pierron, F.; Couture, P. Temperature and metal exposure affect membrane fatty acid composition and transcription of desaturases and elongases in fathead minnow muscle and brain. Ecotoxicol. Environ. Saf. 2018, 148, 632–643. [Google Scholar] [CrossRef] [PubMed]
- Scott, G.R.; Sloman, K.A. The effects of environmental pollutants on complex fish behaviour: Integrating behavioural and physiological indicators of toxicity. Aquat. Toxicol. 2004, 68, 369–392. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, I.; Lavoie, M.; Fortin, C. A mine of information: Benthic algal communities as biomonitors of metal contamination from abandoned tailings. Sci. Total Environ. 2012, 425, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Leguay, S.; Lavoie, I.; Levy, J.L.; Fortin, C. Using biofilms for monitoring metal contamination in lotic ecosystems: The protective effects of hardness and pH on metal bioaccumulation. Environ. Toxicol. Chem. 2016, 35, 1489–1501. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, I.; Campeau, S.; Zugic-Drakulic, N.; Winter, J.G.; Fortin, C. Using diatoms to monitor stream biological integrity in Eastern Canada: An overview of 10 years of index development and ongoing challenges. Sci. Total Environ. 2014, 475, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.G.; Whitton, B.A. The Trophic Diatom Index: A new index for monitoring eutrophication in rivers. J. Appl. Phycol. 1995, 7, 433–444. [Google Scholar] [CrossRef]
- Prygiel, J.; Coste, M.; Bukowska, J. Review of the major diatom-based techniques for the quality assessment of rivers—State of the art in Europe. In Use of Algae for Monitoring Rivers III; Prygiel, J., Whitton, B.A., Bukowska, J., Eds.; Agence de l’Eau Artois Picardie: Douai, France, 1999; pp. 224–238. [Google Scholar]
- Morin, S.; Cordonier, A.; Lavoie, I.; Arini, A.; Blanco, S.; Duong, T.T.; Tornés, E.; Bonet, B.; Corcoll, N.; Faggiano, L.; et al. Consistency in diatom response to metal-contaminated environments. In Handbook of Environmental Chemistry; Guasch, H., Ginebreda, A., Geiszinger, A., Eds.; Springer: Heidelberg, Germany, 2012; Volume 19, pp. 117–146. [Google Scholar]
- Debenest, T.; Silvestre, J.; Coste, M.; Delmas, F.; Pinelli, E. Herbicide effects on freshwater benthic diatoms: Induction of nucleus alterations and silica cell wall abnormalities. Aquat. Toxicol. 2008, 88, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, I.; Hamilton, P.B.; Morin, S.; Kim Tiam, S.; Kahlert, M.; Gonçalves, S.; Falasco, E.; Fortin, C.; Gontero, B.; Heudre, D.; et al. Diatom teratologies as biomarkers of contamination: Are all deformities ecologically meaningful? Ecol. Indic. 2017, 82, 539–550. [Google Scholar] [CrossRef]
- Morin, S.; Corcoll, N.; Bonet, B.; Tlili, A.; Guasch, H. Diatom responses to zinc contamination along a Mediterranean river. Plant Ecol. Evol. 2014, 147, 325–332. [Google Scholar] [CrossRef]
- Morin, S.; Bonet, B.; Corcoll, N.; Guasch, H.; Bottin, M.; Coste, M. Cumulative stressors trigger increased vulnerability of diatom communities to additional disturbances. Microb. Ecol. 2015, 70, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Guasch, H.; Navarro, E.; Serra, A.; Sabater, S. Phosphate limitation influences the sensitivity to copper in periphytic algae. Freshw. Biol. 2004, 49, 463–473. [Google Scholar] [CrossRef]
- Tlili, A.; Bérard, A.; Roulier, J.-L.; Volat, B.; Montuelle, B. PO43− dependence of the tolerance of autotrophic and heterotrophic biofilm communities to copper and diuron. Aquat. Toxicol. 2010, 98, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Nriagu, J.O.; Wong, H.K.T.; Lawson, G.; Daniel, P. Saturation of ecosystems with toxic metals in Sudbury basin, Ontario, Canada. Sci. Total Environ. 1998, 223, 99–117. [Google Scholar] [CrossRef]
- Gunn, J.; Keller, W.; Negusanti, J.; Potvin, R.; Beckett, P.; Winterhalder, K. Ecosystem recovery after emission reductions: Sudbury, Canada. Water Air Soil Pollut. 1995, 85, 1783–1788. [Google Scholar] [CrossRef]
- Keller, W.; Yan, N.D.; Gunn, J.M.; Heneberry, J. Recovery of acidified lakes: Lessons from Sudbury, Ontario, Canada. In Acid Rain—Deposition to Recovery; Brimblecombe, P., Hara, H., Houle, D., Novak, M., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 317–322. [Google Scholar]
- Potvin, R.R.; Negusanti, J.J. Declining industrial emissions, improving air quality, and reduced damage to vegetation. In Restoration and Recovery of an Industrial Region: Progress in Restoring the Smelter-Damaged Landscape near Sudbury, Canada; Gunn, J.M., Ed.; Springer: New York, NY, USA, 1995; pp. 51–65. [Google Scholar]
- Keller, W.; Gunn, J.M.; Yan, N.D. Evidence of biological recovery in acid-stressed lakes near Sudbury, Canada. Environ. Pollut. 1992, 78, 79–85. [Google Scholar] [CrossRef]
- Pearson, D.A.B.; Gunn, J.M.; Keller, W. The past, present and future of Sudbury’s Lakes. In The Physical Environment of the City of Greater Sudbury; Rousell, D.H., Jansons, K.J., Eds.; Special volume 6, Ontario Geological Survey: Sudbury, ON, Canada, 2002; pp. 195–215. [Google Scholar]
- Neary, B.P.; Dillion, P.J.; Munro, J.R.; Clark, B.J. The Acidification of Ontario Lakes: An Assessment of Their Sensitivity and Current Status with Respect to Biological Damage; Technical Report; Ontario Ministry of Environment: Dorset, ON, Canada, 1990; p. 170. [Google Scholar]
- Keller, W. Limnology in northeastern Ontario: From acidification to multiple stressors. Can. J. Fish. Aquat. Sci. 2009, 66, 1189–1198. [Google Scholar] [CrossRef]
- Lento, J.; Dillon, P.J.; Somers, K.M.; Reid, R.A. Changes in littoral benthic macroinvertebrate communities in relation to water chemistry in 17 Precambrian Shield lakes. Can. J. Fish. Aquat. Sci. 2008, 65, 906–918. [Google Scholar] [CrossRef]
- Association Française de Normalisation (AFNOR). Water Quality—Sampling, Treatment and Analysis of Benthic Diatoms from Streams and Canals (Standard NF T90-354); AFNOR: La Plaine Saint-Denis, France, 2016; p. 119. [Google Scholar]
- Lemieux, E.S.; Gunn, J.M.; Sheardown, J. Fish Community Assessment of Junction Creek 2004; Coop Freshwater Unit, Junction Creek Stewardship Committee: Sudbury, ON, Canada, 2004; p. 68. [Google Scholar]
- Souter, L.E.; Watmough, S.A. Geochemistry and toxicity of a large slag pile and its drainage complex in Sudbury, Ontario. Sci. Total Environ. 2017, 15, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, I.; Hamilton, P.B.; Campeau, S.; Grenier, M.; Dillon, P.J. Guide d’Identification des Diatomées des Rivières de l’Est du Canada; Presses de l’Université du Québec: Quebec City, QC, Canada, 2008. [Google Scholar]
- Falasco, E.; Bona, F.; Badino, G.; Hoffmann, L.; Ector, L. Diatom teratological forms and environmental alterations: A review. Hydrobiologia 2009, 623, 1–35. [Google Scholar] [CrossRef]
- Lavoie, I.; Campeau, S.; Grenier, M.; Dillon, P.J. A diatom-based index for the biological assessment of eastern Canadian rivers: An application of correspondence analysis (CA). Can. J. Fish. Aquat. Sci. 2006, 63, 1793–1811. [Google Scholar] [CrossRef]
- Grenier, M.; Campeau, S.; Lavoie, I.; Park, Y.S.; Lek, S. Diatom reference communities in Quebec (Canada) streams based on Kohonen self-organizing maps and multivariate analyses. Can. J. Fish. Aquat. Sci. 2006, 63, 2087–2106. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Biometris: Wageningen, The Netherlands, 2002. [Google Scholar]
- Dufrêne, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- McCune, B.; Mefford, M.J. Multivariate Analysis of Ecological Data, Version 4.01; MJM Software: Gleneden Beach, OR, USA, 1999; p. 237. [Google Scholar]
- Clements, W.H.; Carlisle, D.M.; Lazorchak, J.M.; Johnson, P.C. Heavy metals structure benthic communities in Colorado mountain streams. Ecol. Appl. 2000, 10, 626–638. [Google Scholar] [CrossRef]
- Canadian Council of Ministers of the Environment (CCME). Canadian Environmental Quality Guidelines; CCME: Winnipeg, MB, Canada, 1999. [Google Scholar]
- US EPA. Draft Aquatic Life Criteria for Aluminum in Freshwater. Available online: https://www.epa.gov/wqc/2017-draft-aquatic-life-criteria-aluminum-freshwater-documents (accessed on 21 November 2017).
- Guasch, H.; Leira, M.; Montuelle, B.; Geiszinger, A.; Roulier, J.-L.; Tornés, E.; Serra, A. Use of multivariate analyses to investigate the contribution of metal pollution to diatom species composition: Search for the most appropriate cases and explanatory variables. Hydrobiologia 2009, 627, 143–158. [Google Scholar] [CrossRef]
- Rozon-Ramilo, L.D.; Dubé, M.G.; Rickwood, C.J.; Niyogi, S. Examining the effects of metal mining mixtures on fathead minnow (Pimephales promelas) using field-based multi-trophic artificial streams. Ecotoxicol. Environ. Saf. 2011, 74, 1536–1547. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J. Applying the Reference Condition Approach to Monitor Invertebrates in Streams of the Sudbury Mining Area. Master’s Thesis, Laurentian University, Sudbury, ON, Canada, December 2002. [Google Scholar]
- Driedger, K.L.F. Effects of Metal Mine and Municipal Wastewater on Growth and Energy Stores in Juvenile Fishes. Master’s Thesis, University of Saskatchewan, Saskatoon, SK, Canada, October 2009. [Google Scholar]
- Conservation Sudbury. Vermillion River Watershed. Surface Water Quality Report on Current Conditions; Conservation Sudbury: Sudbury, ON, Canada, 2017; p. 43. [Google Scholar]
- CWWF. Canada-Ontario Clean Water and Wastewater Fund. Available online: https://www.canada.ca/en/office-infrastructure/news/2017/05/backgrounder_canadaandontarioannouncefundingforcleanwaterandwast2.html?=undefined&wbdisable=true (accessed on 28 November 2017).
- The Sudbury Star. Available online: http://www.thesudburystar.com/2015/08/05/fish-return-to-sudburys-nolin-creek (accessed on 12 November 2017).
- Cattaneo, A.; Couillard, Y.; Wunsam, S.; Courcelles, M. Diatom taxonomic and morphological changes as indicators of metal pollution and recovery in Lac Dufault (Québec, Canada). J. Paleolimnol. 2004, 32, 163–175. [Google Scholar] [CrossRef]
- Cattaneo, A.; Couillard, Y.; Wunsam, S.; Fortin, C. Littoral diatoms as indicators of recent water and sediment contamination by metals in lakes. J. Environ. Monit. 2011, 13, 572–582. [Google Scholar] [CrossRef] [PubMed]
- Duong, T.T.; Morin, S.; Herlory, O.; Feurtet-Mazel, A.; Coste, M.; Boudou, A. Seasonal effects of cadmium accumulation in periphytic diatom communities of freshwater biofilms. Aquat. Toxicol. 2008, 90, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Luís, A.T.; Teixeira, P.; Almeida, S.F.P.; Ector, L.; Matos, J.X.; Ferreira da Silva, E.A. Impact of acid mine drainage (AMD) on water quality, stream sediments and periphytic diatom communities in the surrounding streams of Aljustrel mining area (Portugal). Water Air Soil Pollut. 2009, 200, 147–167. [Google Scholar] [CrossRef]
- Martin, G.; Fernandez, M.D.L.R. Diatoms as indicators of water quality and ecological status: Sampling, analysis and some ecological remarks, Ecological Water Quality. In Water Treatment and Reuse; Voudouris, K., Ed.; InTech: London, UK, 2012; pp. 183–204. [Google Scholar]
- Morin, S.; Vivas-Nogues, M.; Duong, T.T.; Boudou, A.; Coste, M.; Delmas, F. Dynamics of benthic diatom colonization in a cadmium/zinc-polluted river (Riou-Mort, France). Fundam. Appl. Limnol. Arch. Hydrobiol. 2007, 168, 179–187. [Google Scholar] [CrossRef]
- Coste, M.; Boutry, S.; Tison-Rosebery, J.; Delmas, F. Improvements of the Biological Diatom Index (BDI): Description and efficiency of the new version (BDI-2006). Ecol. Indic. 2009, 9, 621–650. [Google Scholar] [CrossRef]
- Kelly, M.; Urbanic, G.; Acs, E.; Bennion, H.; Bertrin, V.; Burgess, A.; Denys, L.; Gottschalk, S.; Kahlert, M.; Karjalainen, S.; et al. Comparing aspirations: Intercalibration of ecological status concepts across European lakes for littoral diatoms. Hydrobiologia 2014, 734, 125–141. [Google Scholar] [CrossRef]
- Van Dam, H.; Mertens, A.; Sinkeldam, J. A coded checklist and ecological indicator values of freshwater diatoms from The Netherlands. Neth. J. Aquat. Ecol. 1994, 28, 117–133. [Google Scholar]
- Trobajo Pujadas, R. Ecological Analysis of Periphytic Diatoms in Mediterranean Coastal Wetlands (Empordà Wetlands, NE Spain); Koeltz Scientific Books: Koenigstein, Germany, 2007. [Google Scholar]
- Della Bella, V.; Puccinelli, C.; Marcheggiani, S.; Mancini, L. Benthic diatom communities and their relationship to water chemistry in wetlands of central Italy. Int. J. Limnol. 2007, 43, 89–99. [Google Scholar] [CrossRef][Green Version]
- Rimet, F. Diatoms: An Ecoregional Indicator of Nutrients, Organic Matter and Micropollutants Pollution. Ph.D. Thesis, Université de Grenoble, France, 2012. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 4. Teil: Achnanthaceae. Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema. : , 1991; Volume Band 2/4. G. Fischer Verlag.: Stuttgart, Germany, 1999; Volume Band 2/4. [Google Scholar]
- Morin, S.; Duong, T.T.; Dabrin, A.; Coynel, A.; Herlory, O.; Baudrimont, M.; Delmas, F.; Durrieu, G.; Schäfer, J.; Winterton, P.; et al. Long-term survey of heavy metal pollution, biofilm contamination and diatom community structure in the Riou-Mort watershed, South West France. Environ. Pollut. 2008, 151, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Arini, A.; Feurtet-Mazel, A.; Maury-Brachet, R.; Pokrovsky, O.; Coste, M.; Delmas, F. Recovery potential of periphytic biofilms translocated in artificial streams after industrial contamination (Cd and Zn). Ecotoxicology 2012, 21, 1403–1414. [Google Scholar] [CrossRef] [PubMed]
- Bayona, Y.; Roucaute, M.; Cailleaud, K.; Lagadic, L.; Bassères, A.; Caquet, T. Structural and biological trait responses of diatom assemblages to organic chemicals in outdoor flow-through mesocosms. Environ. Pollut. 2014, 192, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Lainé, M.; Morin, S.; Tison-Rosebery, J. A multicompartment approach—Diatoms, macrophytes, benthic macroinvertebrates and fish—To assess the impact of toxic industrial releases on a small French river. PLoS ONE 2014, 9, e102358. [Google Scholar] [CrossRef] [PubMed]
- Rimet, F.; Ector, L.; Dohet, A.; Cauchie, H.M. Impacts of fluoranthene on diatom assemblages and frustule morphology in indoor microcosms. Vie et Milieu 2004, 54, 145–156. [Google Scholar]
- Larras, F.; Coulaud, R.; Gautreau, E.; Billoir, E.; Rosebery, J.; Usseglio-Polatera, P. Assessing anthropogenic pressures on streams: A random forest approach based on benthic diatom communities. Sci. Total Environ. 2017, 586, 1101–1112. [Google Scholar] [CrossRef] [PubMed]
- Greater Sudbury Climate Change Consortium. Available online: http://www.sudburyclimateaction.ca/en/ (accessed on 19 October 2017).
- Morin, S.; Lambert, A.S.; Planes Rodriguez, E.; Dabrin, A.; Coquery, M.; Pesce, S. Changes in copper toxicity towards diatom communities with experimental warming. J. Hazard. Mater. 2017, 334, 223–232. [Google Scholar] [CrossRef] [PubMed]
| Sampling Sites | MBC | VR | CC1 | CC2 | CC3 | JC 1 | JC 2 | JC 3 | JC 4 | JC 5 | JC 6 | JC 7 | JC 8 | JC 9 | FBC | NC 1 | NC 2 | NC 3 | CCC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | 13.6 | 14.3 | 14.4 | 16.0 | 16.7 | 16.6 | 14.4 | 13.4 | 13.2 | 14.0 | 14.9 | 17.8 | 16.9 | 15.5 | 15.6 | 13.4 | 12.1 | 15.6 | 17.7 |
| Conductivity (mS/cm) | 0.501 | 0.0963 | 1.12 | 1.13 | 1.08 | NA | NA | NA | 1.33 | NA | 1.51 | 3.51 | 2.56 | 1.62 | 1.35 | 1.43 | 1.24 | 1.46 | 4.70 |
| pH | 7.5 | 6.6 | 6.5 | 6.5 | 6.7 | 7.2 | 7.7 | 7.2 | 7.3 | 7.1 | 8.0 | 7.8 | 7.0 | 7.0 | 7.7 | 6.2 | 7.4 | 7.9 | 5.7 |
| Hardness (mg/L) | 177 ± 0 | 37.6 ± 0.2 | 331 ± 2 | 338 ± 2 | 320 ± 1 | 1010 ± 13 | 602 ± 1 | 506 ± 1 | 430 ± 2 | 303 ± 1 | 256 ± 1 | 1018 ± 8 | 637 ± 2 | 536 ± 2 | 260 ± 0 | 336 ± 1 | 154 ± 1 | 218 ± 0 | 1620 ± 8 |
| DOC (mg/L) | 5.34 ± 0.00 | 15.8 ± 0.3 | 4.06 ± 0.00 | 4.04 ± 0.07 | 4.20 ± 0.00 | 1.68 ± 0.03 | 3.46 ± 0.07 | 3.78 ± 0.00 | 6.28 ± 0.02 | 5.78 ± 0.07 | 4.49 ± 0.00 | 2.21 ± 0.00 | 4.13 ± 0.00 | 3.54 ± 0.00 | 5.49 ± 0.07 | 1.84 ± 0.04 | 3.96 ± 0.12 | 5.26 ± 0.00 | 5.28 ± 0.14 |
| Mg (mg/L) | 14.3 ± 0.1 | 3.30 ± 0.01 | 12.5 ± 0.1 | 12.7 ± 0.1 | 12.4 ± 0.0 | 31.9 ± 0.1 | 23.4 ± 0.1 | 20.7 ± 0.0 | 18.9 ± 0.1 | 15.6 ± 0.0 | 14.2 ± 0.1 | 26.9 ± 0.2 | 20.2 ± 0.0 | 17.8 ± 0.1 | 26.5 ± 0.0 | 12.8 ± 0.0 | 13.4 ± 0.1 | 12.8 ± 0.0 | 39.3 ± 0.5 |
| Ca (mg/L) | 47.3 ± 0.1 | 9.64 ± 0.06 | 112 ± 1 | 115 ± 1 | 108 ± 0 | 352 ± 6 | 202 ± 0 | 169 ± 0 | 141 ± 1 | 95.5 ± 0.5 | 79.0 ± 0.2 | 364 ± 3 | 222 ± 1 | 185 ± 1 | 60.4 ± 0.1 | 113 ± 0 | 39.4 ± 0.3 | 66.5 ± 0.0 | 584 ± 4 |
| SO4 (mg/L) | 34.0 ± 0.0 | 5.00 ± 0.00 | 241 ± 2 | 245 ± 1 | 230 ± 1 | 942 ± 4 | 453 ± 3 | 363 ± 3 | 166 ± 1 | 194 ± 1 | 254 ± 2 | 1202 ± 33 | 701 ± 3 | 561 ± 3 | 194 ± 1 | 268 ± 4 | 137 ± 1 | 172 ± 2 | 1923 ± 7 |
| NO3 (mg/L) | 0.94 ± 0.06 | 0.78 ± 0.00 | 7.64 ± 0.58 | 6.24 ± 0.58 | 5.0 ± 1.6 | 19.7 ± 0.3 | 7.63 ± 0.15 | 5.98 ± 0.06 | 0.95 ± 0.03 | 3.20 ± 0.00 | 7.23 ± 0.58 | 28.6 ± 2.1 | 20.6 ± 1.2 | 16.4 ± 0.4 | 3.20 ± 0.00 | 6.6 ± 1.2 | 3.7 ± 1.1 | 5.30 ± 0.58 | 3.20 ± 0.00 |
| TP (μg/L) | 22.6 ± 0.4 | 29.9 ± 1.1 | 9.00 ± 0.20 | 8.87 ± 0.12 | 38.5 ± 0.4 | 12.7 ± 0.5 | 47.3 ± 0.4 | 46.1 ± 0.7 | 41.5 ± 1.3 | 29.1 ± 0.7 | 36.0 ± 0.8 | 48.1 ± 2.2 | 70.2 ± 3.0 | 137 ± 1 | 15.7 ± 0.3 | 11.5 ± 0.9 | 9.63 ± 0.06 | 16.9 ± 0.1 | 6.57 ± 0.15 |
| Al (μg/L) | 13.1 ± 0.8 | 41 ± 16 | 19.6 ± 6.1 | 16.3 ± 3.8 | 14.6 ± 0.3 | 15.1 ± 1.7 | 14.0 ± 1.5 | 16.5 ± 0.1 | 16.1 ± 0.4 | 15.0 ± 1.0 | 18.1 ± 2.5 | 15.9 ± 1.2 | 15.5 ± 1.3 | 13.5 ± 1.4 | 10.9 ± 0.5 | 13.0 ± 3.1 | 14.2 ± 1.5 | 15.5 ± 0.5 | 17.9 ± 2.5 |
| Ni (μg/L) | 18.1 ± 0.2 | 7.22 ± 0.22 | 25.1 ± 0.1 | 25.7 ± 0.0 | 197 ± 1 | 226 ± 1 | 199 ± 1 | 152 ± 0 | 113 ± 0 | 41.9 ± 0.4 | 89.6 ± 0.4 | 290 ± 1 | 211 ± 1 | 185 ± 0 | 788 ± 3 | 1037 ± 3 | 804 ± 6 | 689 ± 2 | 32.9 ± 1.0 |
| Cu (μg/L) | 1.72 ± 0.05 | 1.97 ± 0.07 | 2.47 ± 0.04 | 2.72 ± 0.06 | 5.4 ± 2.1 | 3.11 ± 0.06 | 2.09 ± 0.02 | 2.33 ± 0.06 | 3.48 ± 0.04 | 2.62 ± 0.09 | 7.40 ± 0.24 | 5.91 ± 0.10 | 4.32 ± 0.11 | 3.90 ± 0.05 | 5.72 ± 0.17 | 10.2 ± 0.2 | 38.0 ± 2.7 | 19.3 ± 1.4 | 5.96 ± 0.18 |
| Zn (μg/L) | NA | 1.8 ± 3.0 | NA | NA | 1.6 ± 2.7 | 3.3 ± 0.5 | 7.43 ± 0.03 | 6.2 ± 1.0 | 6.1 ± 2.0 | 1.5 ± 1.1 | 5.06 ± 0.49 | 5.79 ± 0.13 | NA | NA | 2.02 ± 0.08 | 23.8 ± 0.2 | 23.2 ± 0.1 | 11.4 ± 0.6 | 0.38 ± 0.08 |
| Cd (μg/L) | 0.029 ± 0.000 | 0.006 ± 0.001 | 0.039 ± 0.002 | 0.038 ± 0.002 | 0.041 ± 0.002 | 0.101 ± 0.001 | 0.050 ± 0.003 | 0.045 ± 0.003 | 0.047 ± 0.001 | 0.016 ± 0.001 | 0.043 ± 0.002 | 0.161 ± 0.001 | 0.025 ± 0.004 | 0.021 ± 0.001 | 0.080 ± 0.003 | 0.298 ± 0.002 | 0.476 ± 0.009 | 0.278 ± 0.008 | 0.096 ± 0.002 |
| Pb (μg/L) | 0.016 ± 0.002 | 0.20 ± 0.06 | 0.006 ± 0.001 | 0.005 ± 0.000 | 0.007 ± 0.004 | 0.155 ± 0.002 | 0.041 ± 0.002 | 0.042 ± 0.003 | 0.036 ± 0.002 | 0.012 ± 0.003 | 0.04 ± 0.02 | 0.038 ± 0.001 | 0.042 ± 0.001 | 0.011 ± 0.002 | 0.006 ± 0.002 | NA | NA | NA | 0.012 ± 0.002 |
| CCU | 0.7 (B) | 2.6 (M) | 0.6 (B) | 0.6 (B) | 1.8 (L) | 0.7 (B) | 0.9 (B) | 0.8 (B) | 1 (L) | 0.7 (B) | 2.1 (M) | 1.1 (L) | 1 (L) | 1 (L) | 5.3 (M) | 6.7 (M) | 19.9 (H) | 9.4 (M) | 0.4 (B) |
| % teratologies | 0.7 | 0.0 | 0.0 | 0.2 | 0.5 | 1.0 | 1.0 | 1.7 | 1.2 | 1.0 | 1.0 | 1.2 | 6.1 | 8.7 | 1.5 | 1.2 | 1.0 | 4.5 | 1.5 |
| IDEC score/Class | 24/C | 42/C | 64/B | 62/B | 31/C | 83/A | 6/D | 14/D | 1/D | 26/C | 27/C | 36/C | 6/D | 21/C | 85/A | 100/A | 100/A | 78/A | 43/C |
| Species | Group on the CCA | Indicator Value | Mean | SD | p-Value |
|---|---|---|---|---|---|
| Brachysira vitrea (BVIT) | 1 | 96.4 | 54.3 | 14.70 | 0.002 |
| Navicula gregaria (NGRE) | 2 | 75.0 | 41.0 | 15.77 | 0.05 |
| Nitzschia palea var. debilis (NPAD) | 2 | 70.8 | 42.8 | 12.43 | 0.02 |
| Eolimna minima (EOMI) | 3 | 70.1 | 44.9 | 12.25 | 0.043 |
| Eolimna subminuscula (ESBM) | 3 | 99.9 | 25.5 | 14.35 | 0.004 |
| Nitzschia palea aff. debilis form 2 (NPAD2) | 3 | 99.7 | 25.1 | 14.22 | 0.004 |
| Amphora veneta (AVEN) | 3 | 99.7 | 29.0 | 14.57 | 0.003 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavoie, I.; Morin, S.; Laderriere, V.; Fortin, C. Freshwater Diatoms as Indicators of Combined Long-Term Mining and Urban Stressors in Junction Creek (Ontario, Canada). Environments 2018, 5, 30. https://doi.org/10.3390/environments5020030
Lavoie I, Morin S, Laderriere V, Fortin C. Freshwater Diatoms as Indicators of Combined Long-Term Mining and Urban Stressors in Junction Creek (Ontario, Canada). Environments. 2018; 5(2):30. https://doi.org/10.3390/environments5020030
Chicago/Turabian StyleLavoie, Isabelle, Soizic Morin, Vincent Laderriere, and Claude Fortin. 2018. "Freshwater Diatoms as Indicators of Combined Long-Term Mining and Urban Stressors in Junction Creek (Ontario, Canada)" Environments 5, no. 2: 30. https://doi.org/10.3390/environments5020030
APA StyleLavoie, I., Morin, S., Laderriere, V., & Fortin, C. (2018). Freshwater Diatoms as Indicators of Combined Long-Term Mining and Urban Stressors in Junction Creek (Ontario, Canada). Environments, 5(2), 30. https://doi.org/10.3390/environments5020030







