Performance of Pd-Based Membranes and Effects of Various Gas Mixtures on H2 Permeation
Abstract
1. Introduction
2. Materials and Methods
2.1. Pd-Based Membrane
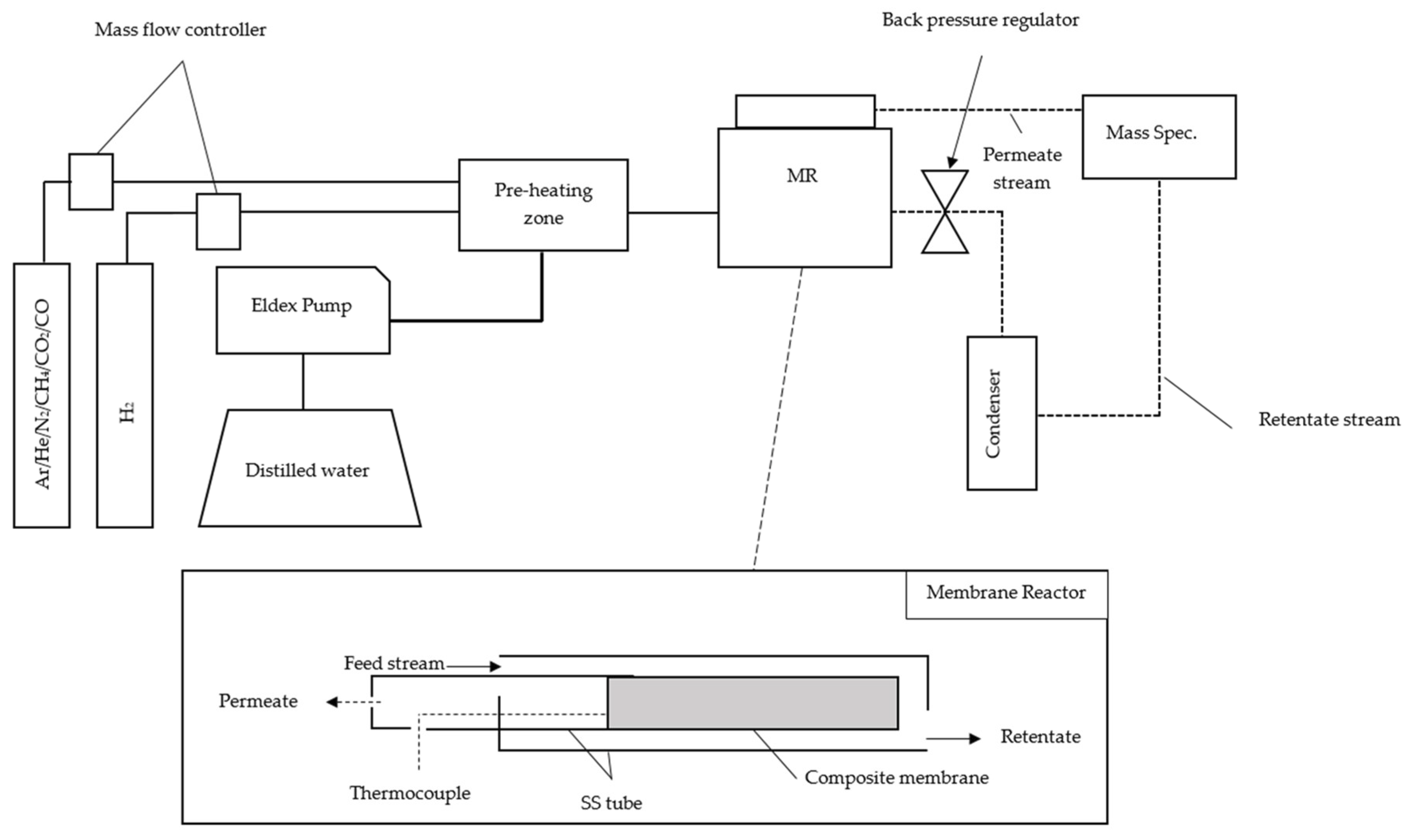
2.2. Procedure
2.3. Materials
3. Results and Discussion
3.1. Membrane Permeation Tests
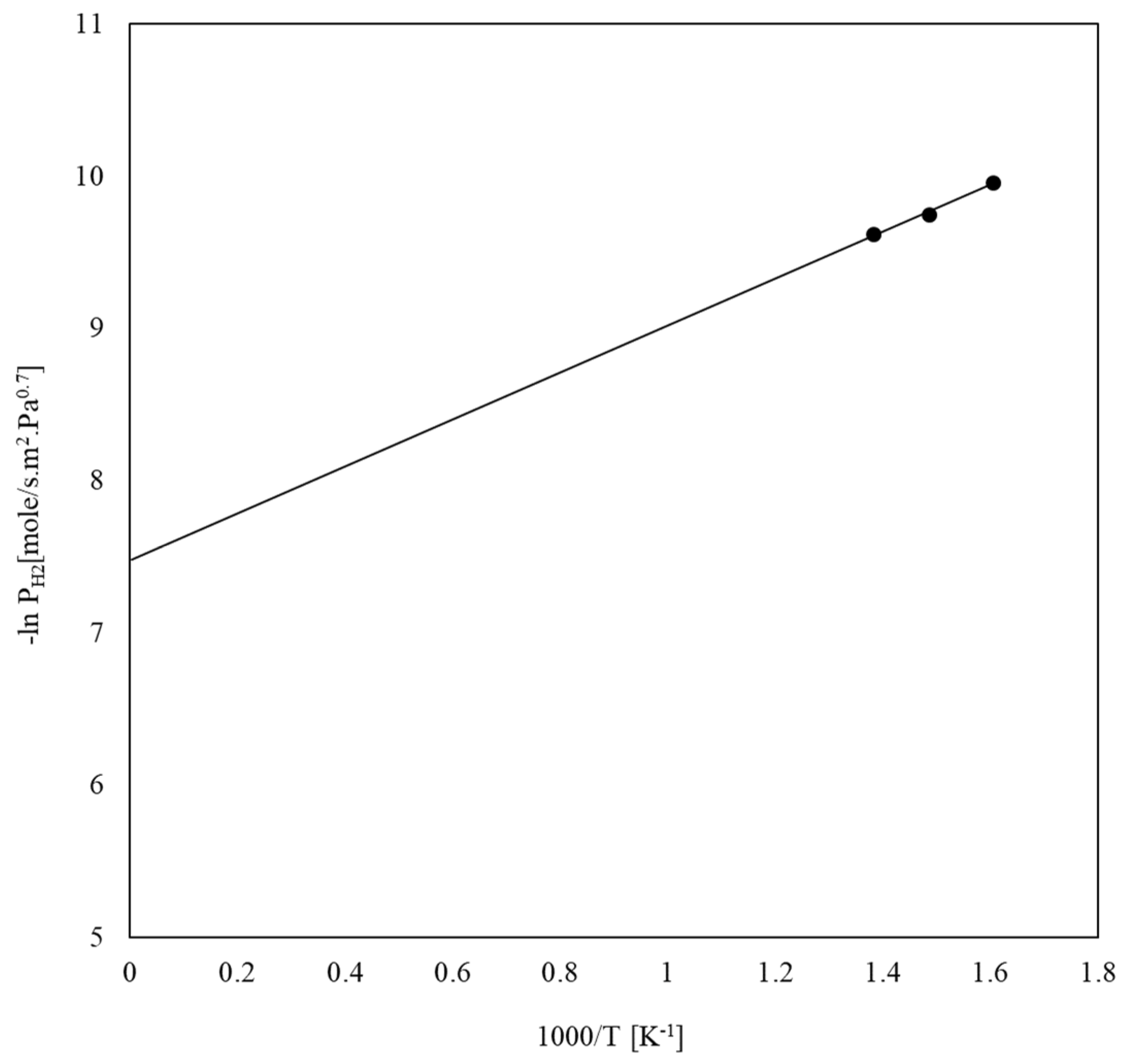
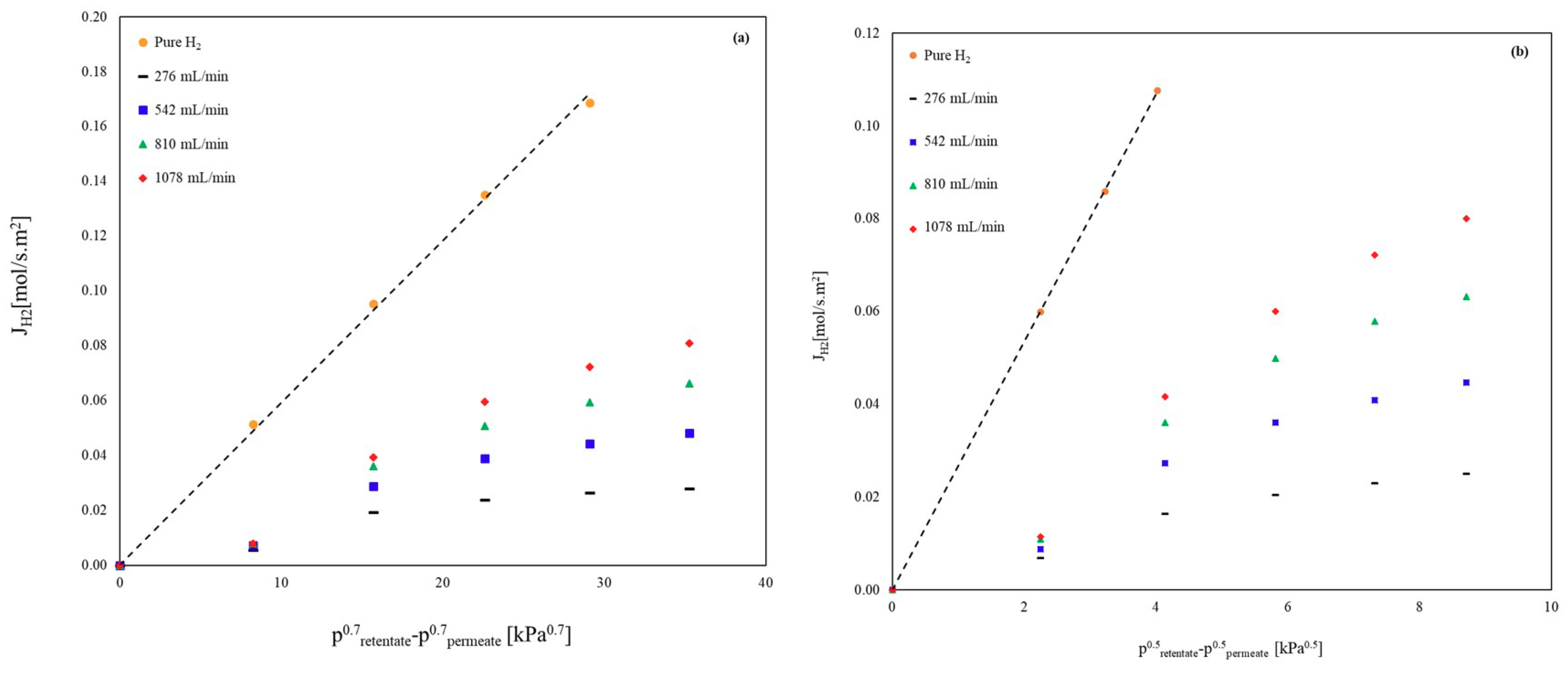
3.1.1. Pure Components
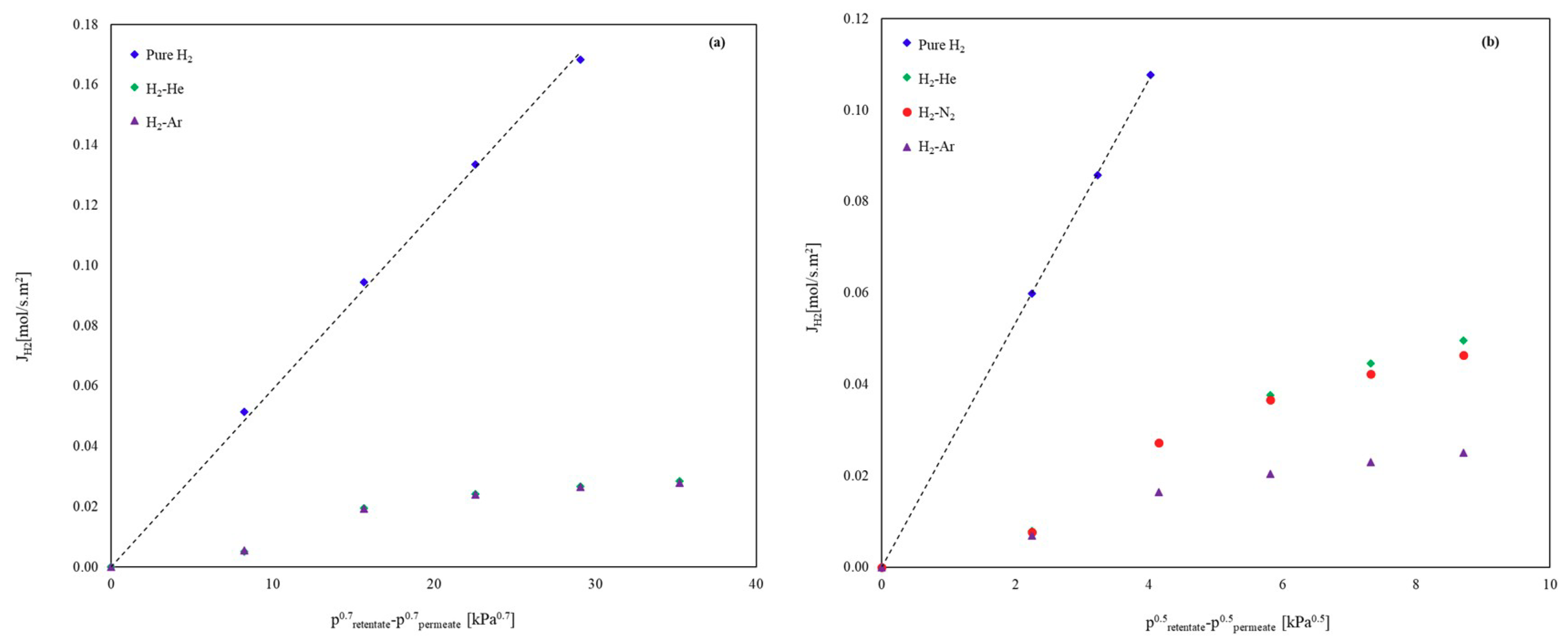
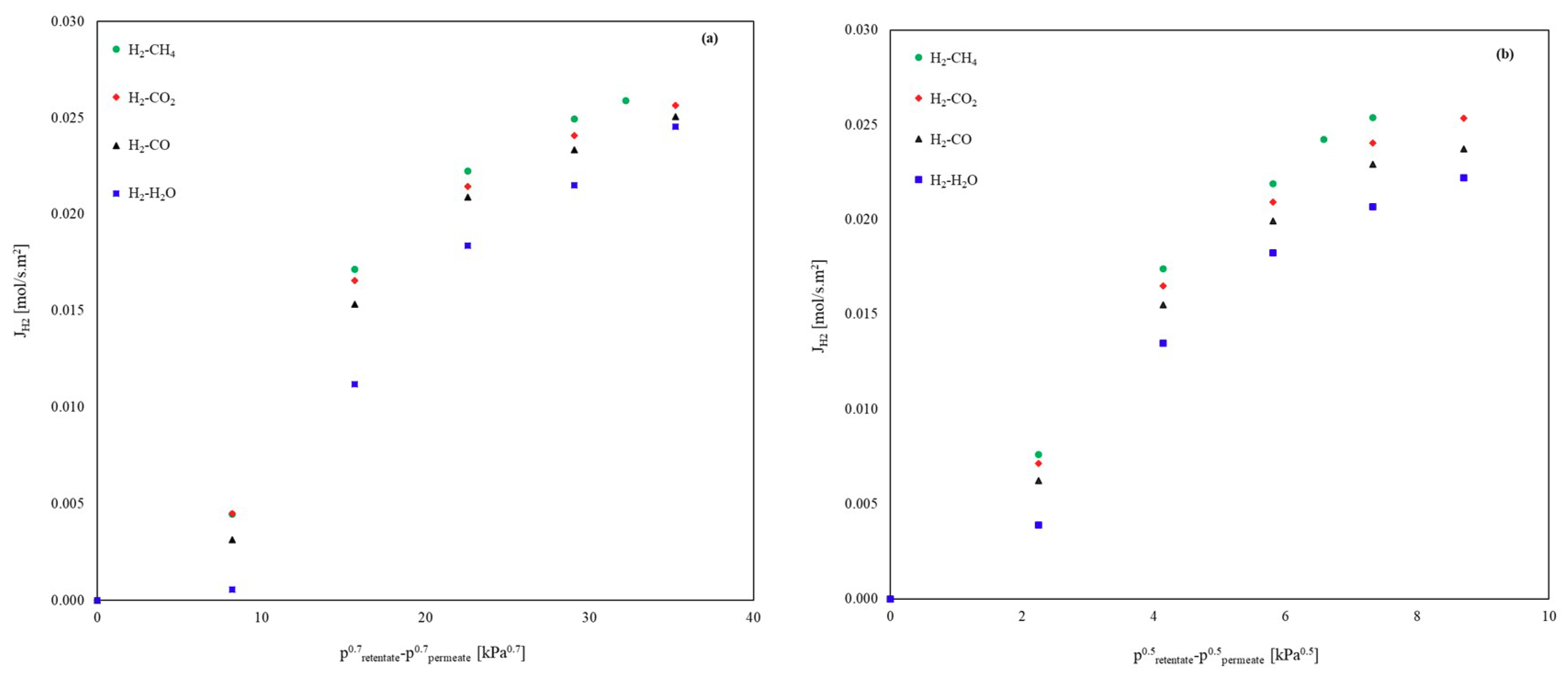
3.1.2. Binary Mixtures
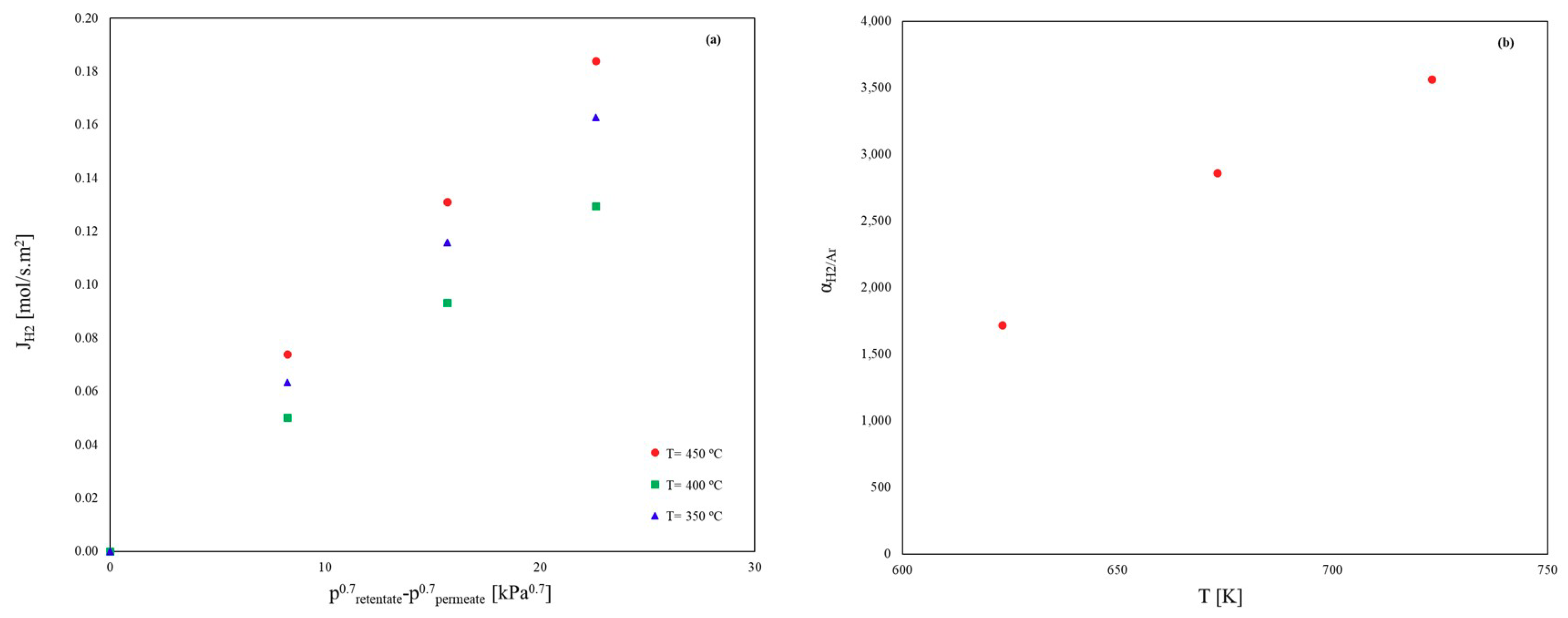
Influence of Inert Gases on the H2 Permeation
Influence of CH4, CO2, CO and H2O on the H2 Permeation
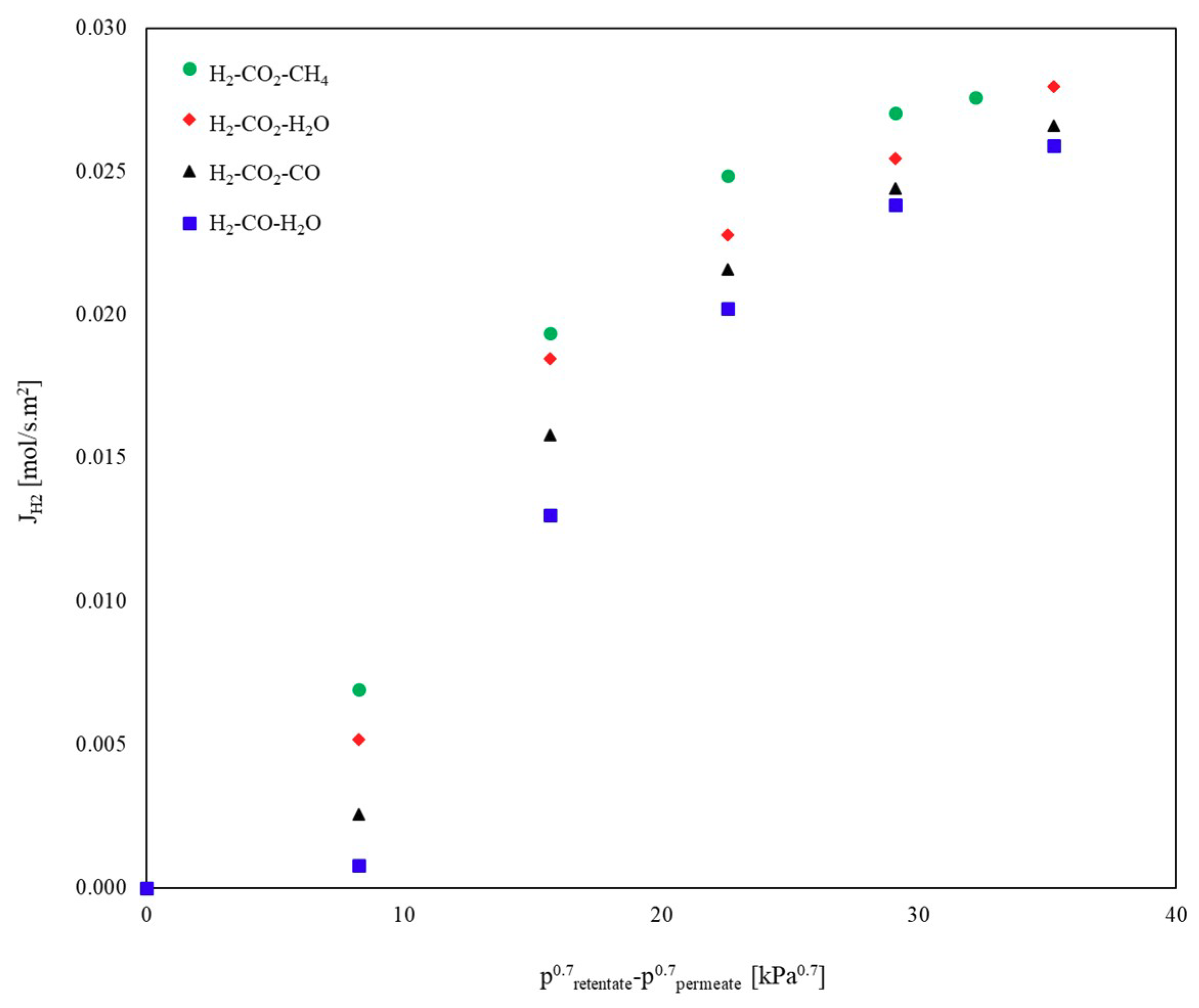
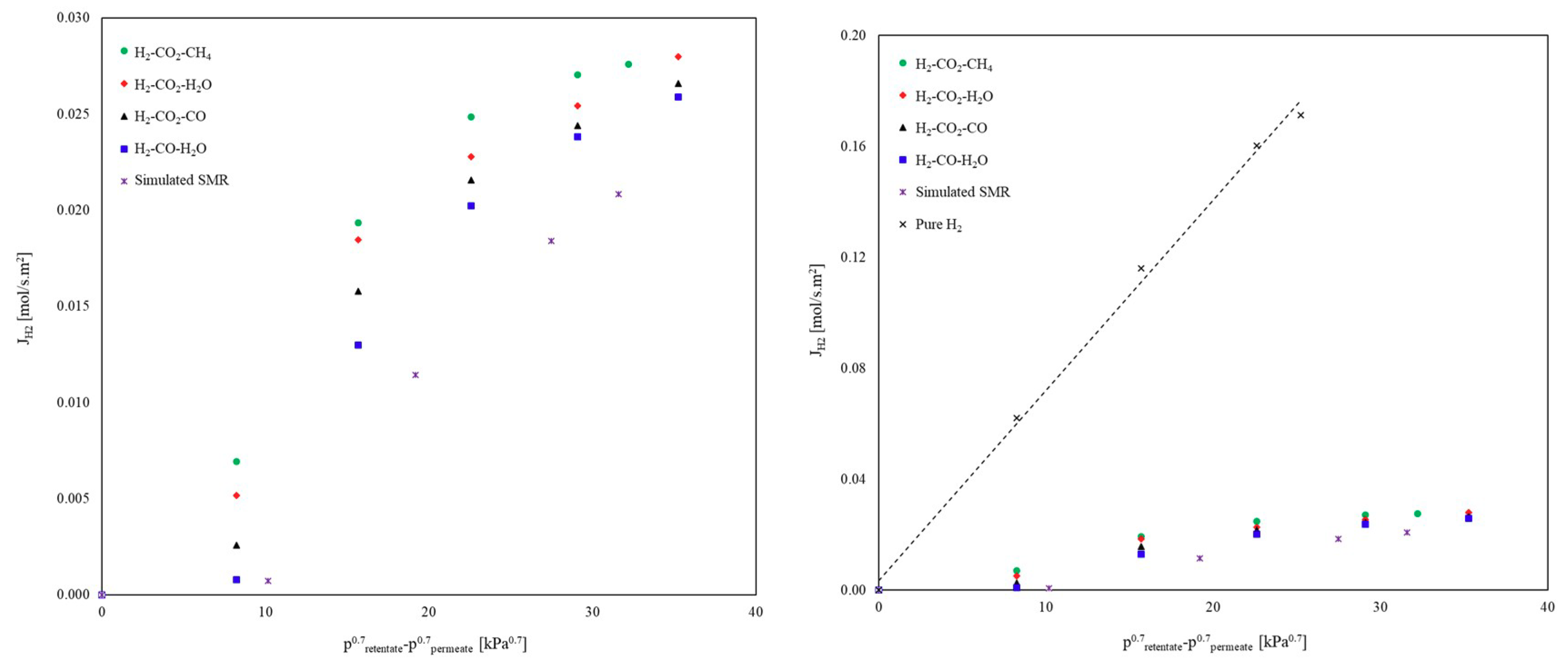
3.1.3. Ternary Mixtures
3.1.4. Simulated SMR Stream
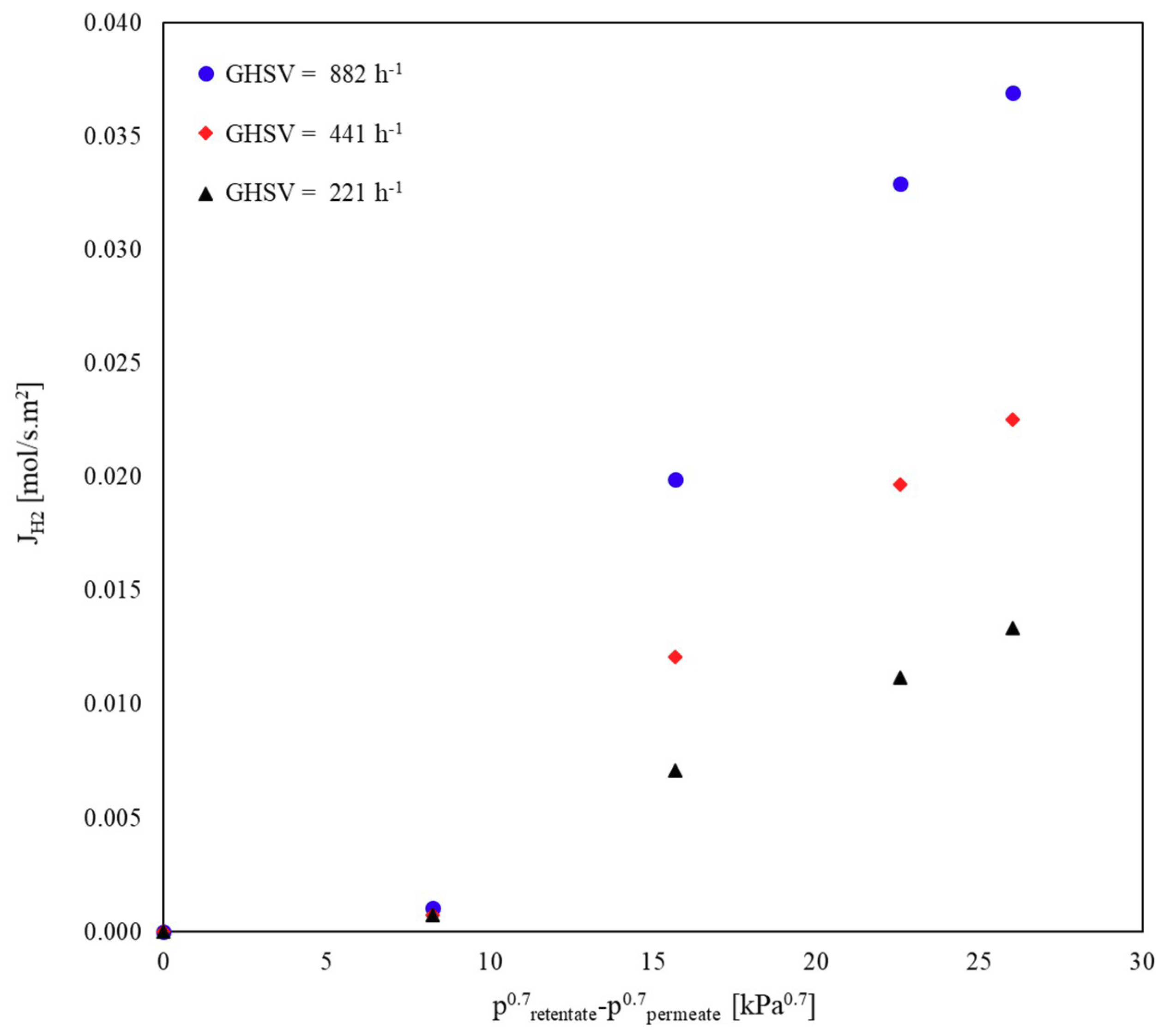
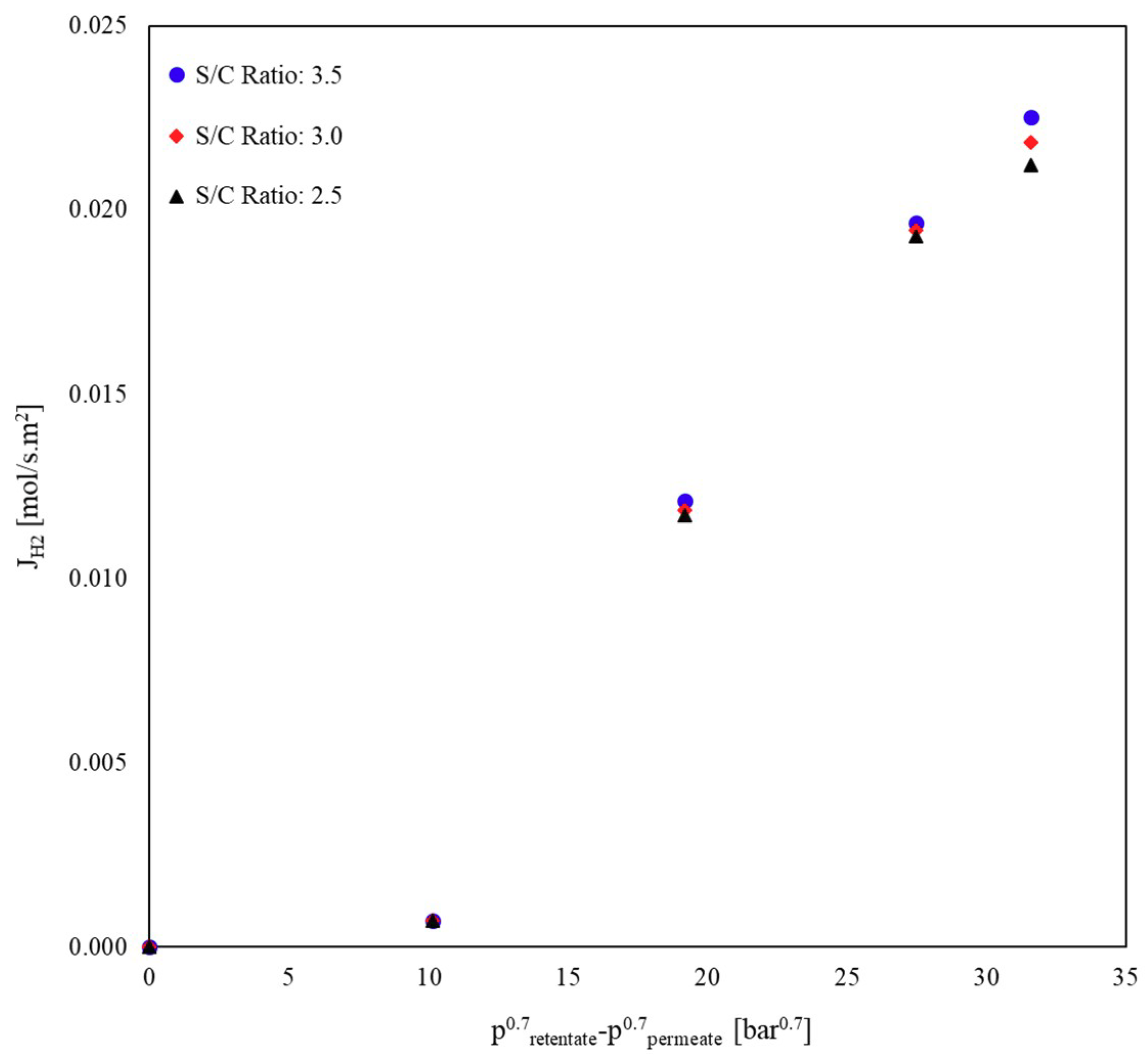
Influence of GHSV and S/C Ratio on the H2 Permeation
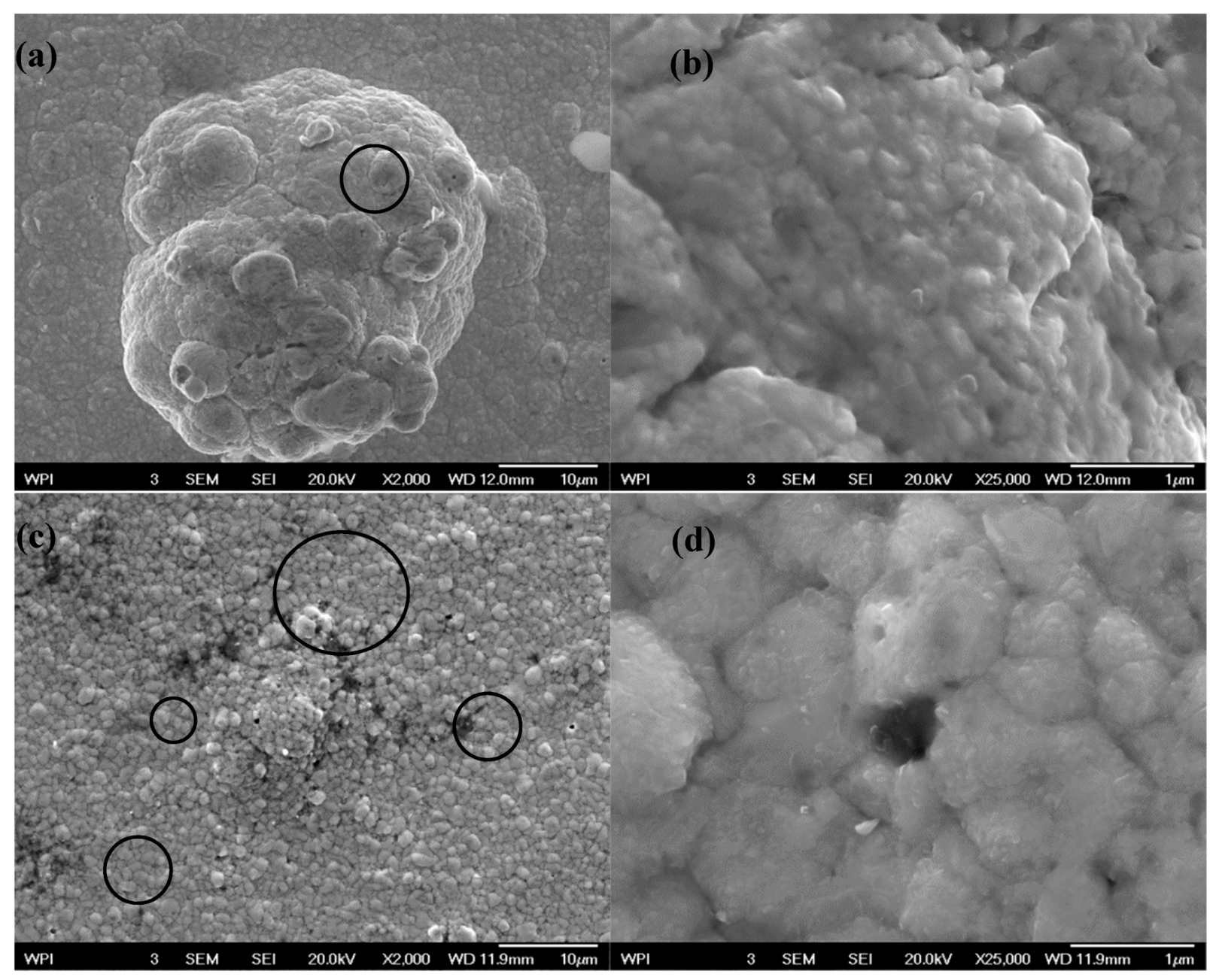
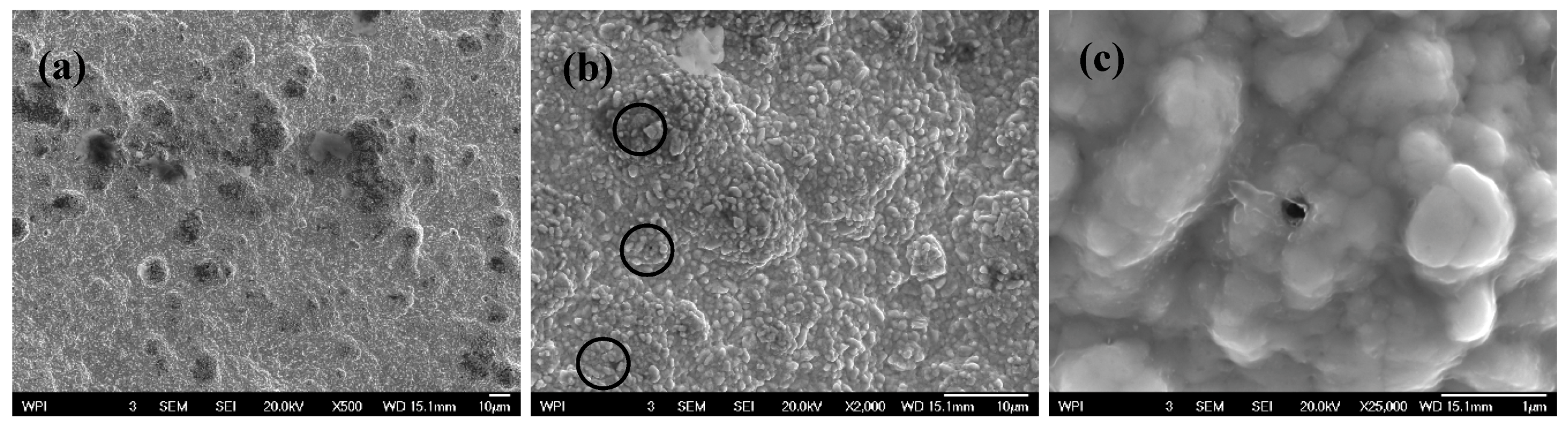
3.2. Scanning Electron Microscope (SEM) Tests
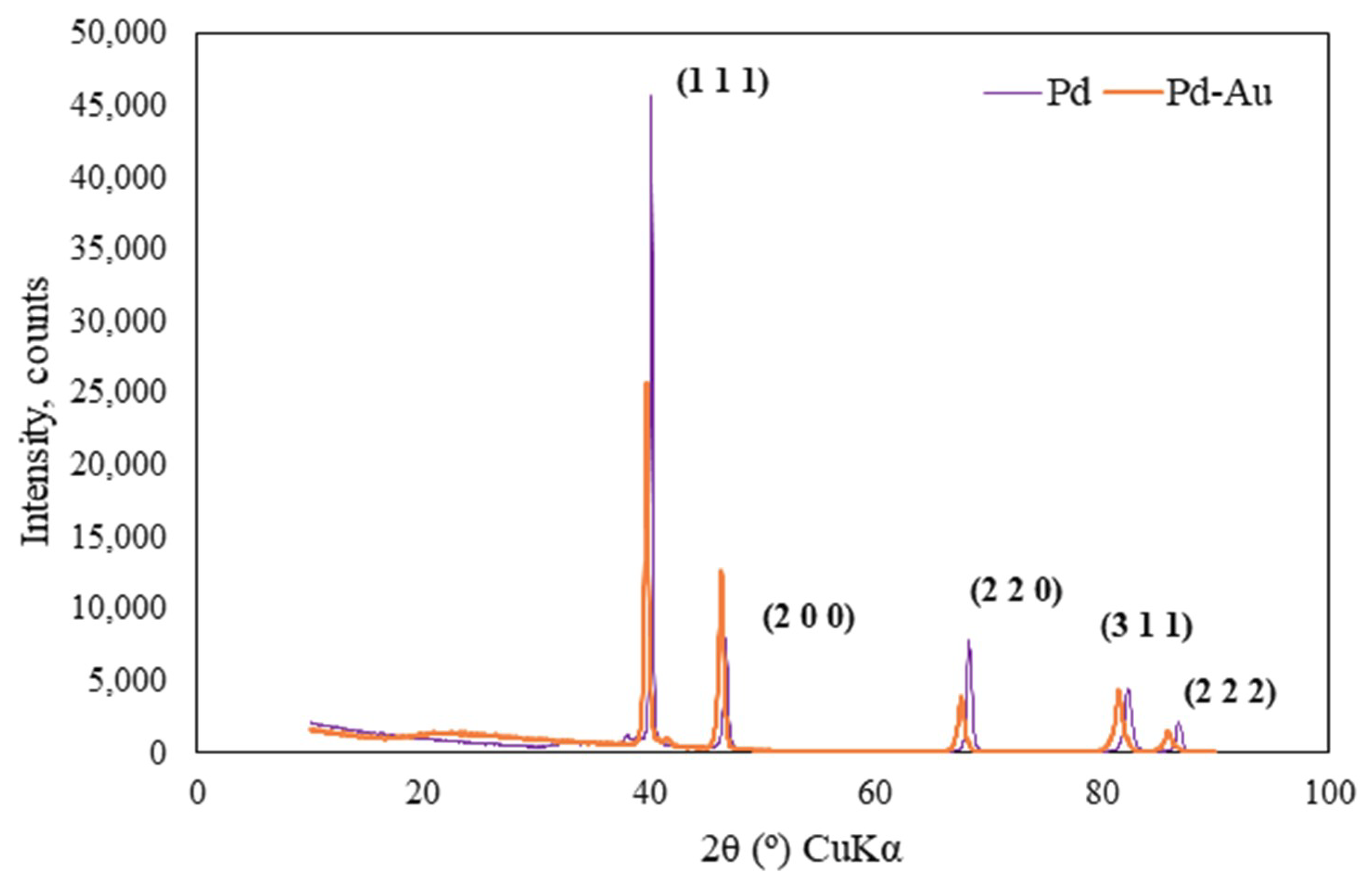
3.3. X-ray Diffraction (XRD) Tests
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EIA Annual Energy Outlook, Energy Information Administration (EIA); U.S. Department of Energy: Washington, DC, USA, 2014.
- U.S. Energy Information Administration. Monthly Energy Review; EIA: Washington, DC, USA, 2018.
- Holladay, J.D.; Hu, J.; King, D.L.; Wang, Y. An overview of hydrogen production technologies. Catal. Today 2009, 139, 244–260. [Google Scholar] [CrossRef]
- Plasynski, S.I.; Litynski, J.T.; McIlvried, H.G.; Srivastava, R.D. Progress and new developments in carbon capture and storage. CRC. Crit. Rev. Plant Sci. 2009, 28, 123–138. [Google Scholar] [CrossRef]
- Lattin, W.C.; Utgikar, V.P. Transition to hydrogen economy in the United States: A 2006 status report. Int. J. Hydrogen Energy 2007, 32, 3230–3237. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Hydrogen, Fuel Cells and Infrastructure Technologies Program, Multi-Year Research, Development and Demonstration Plan: Planned program activities for 2005–2015; US Department Of Energy: Washington, DC, USA, 2007.
- Iulianelli, A.; Liguori, S.; Wilcox, J.; Basile, A. Advances on methane steam reforming to produce hydrogen through membrane reactors technology: A review. Catal. Rev. Sci. Eng. 2016, 58, 1–35. [Google Scholar] [CrossRef]
- Liguori, S.; Pinacci, K.P.; Seelam, R.; Keiski, F.; Drago, V.; Calabrò, V.; Basile, A.; Iulianelli, A. Performance of a Pd/PSS membrane reactor to produce high purity hydrogen via WGS reaction. Catal. Today 2012, 193, 87–94. [Google Scholar] [CrossRef]
- Ghenciu, A.F. Review of fuel processing catalysts for hydrogen production in PEM fuel cell systems. Curr. Opin. Solid State Mater. Sci. 2002, 6, 389–399. [Google Scholar] [CrossRef]
- Al-Mufachi, N.A.; Rees, N.V.; Steinberger-Wilkens, R. Hydrogen selective membranes: A review of palladium-based dense metal membranes. Renew. Sustain. Energy Rev. 2015, 47, 540–551. [Google Scholar] [CrossRef]
- Ohi, J.M.; Vanderborgh, N.; Voecks, G. Hydrogen Fuel Quality Specifications for Polymer Electrolyte Fuel Cells in Road Vehicles; US DOE EERE: Washington, DC, USA, 2016.
- De Falco, M.D.; Marrelli, L.; Iaquaniello, G. Membrane Reactors for Hydrogen Production Processes; Springer: London, UK, 2011; ISBN 9780857291509. [Google Scholar]
- Burkhanov, G.S.; Gorina, N.B.; Kolchugina, N.B.; Roshan, N.R.; Slovetsky, D.I.; Chistov, E.M. Palladium-based alloy membranes for separation of high purity hydrogen from hydrogen-containing gas mixtures. Platin. Met. Rev. 2011, 55, 3–12. [Google Scholar] [CrossRef]
- Li, A.; Grace, J.R.; Lim, C.J. Preparation of thin Pd-based composite membrane on planar metallic substrate. Part II. Preparation of membranes by electroless plating and characterization. J. Memb. Sci. 2007, 306, 159–165. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, D.; He, M.; Xu, H.; Li, W. Experimental and simulation studies on concentration polarization in H 2 enrichment by highly permeable and selective Pd membranes. J. Memb. Sci. 2006, 274, 83–91. [Google Scholar] [CrossRef]
- Zhang, D.; Fan, Y.; Xu, N.; He, Y. Effect of intermetallic diffusion between Pd and Ti-Al alloy on the performance of Pd/Ti-Al alloy composite membranes. J. Memb. Sci. 2011, 377, 221–230. [Google Scholar] [CrossRef]
- Yan, S.; Maeda, H.; Kusakabe, K.; Morooka, S. Thin Palladium Membrane Formed in Support Pores by Metal-Organic Chemical Vapor Deposition Method and Application to Hydrogen Separation. Ind. Eng. Chem. Res. 1994, 33, 616–622. [Google Scholar] [CrossRef]
- Tong, J.; Kashima, Y.; Shirai, R.; Suda, H.; Matsumura, Y. Thin defect-free Pd membrane deposited on asymmetric porous stainless steel substrate. Ind. Eng. Chem. Res. 2005, 44, 8025–8032. [Google Scholar] [CrossRef]
- Li, A.; Grace, J.R.; Lim, C.J. Preparation of thin Pd-based composite membrane on planar metallic substrate: Part I: Pre-treatment of porous stainless steel substrate. J. Memb. Sci. 2007, 298, 175–181. [Google Scholar] [CrossRef]
- Mardilovich, I.P.; Engwall, E.; Ma, Y.H. Dependence of hydrogen flux on the pore size and plating surface topology of asymmetric Pd-porous stainless steel membranes. Desalination 2002, 144, 85–89. [Google Scholar] [CrossRef]
- Yepes, D.; Cornaglia, L.M.; Irusta, S.; Lombardo, E.A. Different oxides used as diffusion barriers in composite hydrogen permeable membranes. J. Memb. Sci. 2006, 274, 92–101. [Google Scholar] [CrossRef]
- Wang, J.; Luo, J.; Feng, S.; Li, H.; Wan, Y.; Zhang, X. Recent development of ionic liquid membranes. Green Energy Environ. 2016, 1, 43–61. [Google Scholar] [CrossRef]
- Shu, J.; Adnot, A.; Grandjean, B.P.A.; Kaliaguine, S. Structurally stable composite Pd-Ag alloy membranes: Introduction of a diffusion barrier. Thin Solid Films 1996, 286, 72–79. [Google Scholar] [CrossRef]
- Liguori, S.; Iulianelli, A.; Dalena, F.; Pinacci, P.; Drago, F.; Broglia, M.; Huang, Y.; Basile, A. Performance and long-term stability of Pd/PSS and Pd/Al2O3 membranes for hydrogen separation. Membranes 2014, 4, 143–162. [Google Scholar] [CrossRef]
- Dolan, M.D. Non-Pd BCC alloy membranes for industrial hydrogen separation. J. Memb. Sci. 2010, 362, 12–28. [Google Scholar] [CrossRef]
- Borgognoni, F.; Tosti, S.; Vadrucci, M.; Santucci, A. Pure hydrogen production in a Pd-Ag multi-membranes module by methane steam reforming. Int. J. Hydrogen Energy 2011, 36, 7550–7558. [Google Scholar] [CrossRef]
- Brunetti, A.; Caravella, A.; Drioli, E.; Barbieri, G. Process Intensification by Membrane Reactors: High-Temperature Water Gas Shift Reaction as Single Stage for Syngas Upgrading. Chem. Eng. Technol. 2012, 35, 1238–1248. [Google Scholar] [CrossRef]
- Peters, T.A.; Stange, M.; Klette, H.; Bredesen, R. High pressure performance of thin Pd-23%Ag/stainless steel composite membranes in water gas shift gas mixtures; influence of dilution, mass transfer and surface effects on the hydrogen flux. J. Memb. Sci. 2008, 316, 119–127. [Google Scholar] [CrossRef]
- Dolan, M.D.; Donelson, R.; Dave, N.C. Performance and economics of a Pd-based planar WGS membrane reactor for coal gasification. Int. J. Hydrogen Energy 2010, 35, 10994–11003. [Google Scholar] [CrossRef]
- Schramm, O.; Seidel-Morgenstern, A. Comparing porous and dense membranes for the application in membrane reactors. Chem. Eng. Sci. 1999, 54, 1447–1453. [Google Scholar] [CrossRef]
- Gallucci, F.; Paturzo, L.; Famà, A.; Basile, A. Experimental Study of the Methane Steam Reforming Reaction in a Dense Pd/Ag Membrane Reactor. Ind. Eng. Chem. Res. 2004, 43, 928–933. [Google Scholar] [CrossRef]
- Jørgensen, S.L.; Nielsen, P.E.H.; Lehrmann, P.E. Steam reforming of methane in a membrane reactor. Catal. Today 1995, 25, 303–307. [Google Scholar] [CrossRef]
- Chen, W.H.; Syu, W.Z.; Hung, C.I. Numerical characterization on concentration polarization of hydrogen permeation in a Pd-based membrane tube. Int. J. Hydrogen Energy 2011, 36, 14734–14744. [Google Scholar] [CrossRef]
- Mori, N.; Nakamura, T.; Noda, K.-I.I.; Sakai, O.; Takahashi, A.; Ogawa, N.; Sakai, H.; Iwamoto, Y.; Hattori, T. Reactor Configuration and Concentration Polarization in Methane Steam Reforming by a Membrane Reactor with a Highly Hydrogen-Permeable Membrane. Ind. Eng. Chem. Res. 2007, 46, 1952–1958. [Google Scholar] [CrossRef]
- Caravella, A.; Barbieri, G.; Drioli, E. Concentration polarization analysis in self-supported Pd-based membranes. Sep. Purif. Technol. 2009, 66, 613–624. [Google Scholar] [CrossRef]
- Caravella, A.; Sun, Y. Correct evaluation of the effective concentration polarization influence in membrane-assisted devices. Case study: H2 production by Water Gas Shift in Pd-membrane reactors. Int. J. Hydrogen Energy 2016, 41, 11653–11659. [Google Scholar] [CrossRef]
- Hara, S.; Sakaki, K.; Itoh, N. Decline in hydrogen permeation due to concentration polarization and CO hindrance in a palladium membrane reactor. Ind. Eng. Chem. Res. 1999, 38, 4913–4918. [Google Scholar] [CrossRef]
- Thoen, P.M.; Roa, F.; Way, J.D. High flux palladium-copper composite membranes for hydrogen separations. Desalination 2006, 193, 224–229. [Google Scholar] [CrossRef]
- Coroneo, M.; Montante, G.; Catalano, J.; Paglianti, A. Modelling the effect of operating conditions on hydrodynamics and mass transfer in a Pd-Ag membrane module for H2 purification. J. Memb. Sci. 2009, 343, 34–41. [Google Scholar] [CrossRef]
- He, G.; Mi, Y.; Lock Yue, P.; Chen, G. Theoretical study on concentration polarization in gas separation membrane processes. J. Memb. Sci. 1999, 153, 243–258. [Google Scholar] [CrossRef]
- Mourgues, A.; Sanchez, J. Theoretical analysis of concentration polarization in membrane modules for gas separation with feed inside the hollow-fibers. J. Memb. Sci. 2005, 252, 133–144. [Google Scholar] [CrossRef]
- Takaba, H.; Nakao, S. Computational fluid dynamics study on concentration polarization in H2/CO separation membranes. J. Memb. Sci. 2005, 249, 83–88. [Google Scholar] [CrossRef]
- Ma, Y.H.; Mardilovich, I.P. Composite Structures with Porous Anodic Oxide Layers and Methods of Fabricationn. U.S. Patent No 8,366,805, 5 February 2013. [Google Scholar]
- Ma, Y.H.; Mardilovich, I.P.; Engwall, E.E. Composite Gas Separation Modules Having High Tamman Temperature Intermediate Layersn. U.S. Patent No 7,255,726, 14 August 2007. [Google Scholar]
- Ma, Y.H.; Mardilovich, P.P.; She, Y. Hydrogen Gas-Extraction Module and Method of Fabricationn. U.S. Patent No 6,152,987, 28 November 2000. [Google Scholar]
- Collins, J.P.; Way, J.D. Preparation and Characterization of a Composite Palladium-Ceramic Membrane. Ind. Eng. Chem. Res. 1993, 32, 3006–3013. [Google Scholar] [CrossRef]
- Anzelmo, B. On-Board Hydrogen Production from Natural Gas via a Metallic Pd-Based Membrane Reactor. Ph.D. Thesis, Stanford University, Stanford, CA, USA, 2016. [Google Scholar]
- Helmi, A.; Fernandez, E.; Melendez, J.; Tanaka, D.A.P.; Gallucci, F.; Van Sint Annaland, M. Fluidized bed membrane reactors for ultra pure H2 production—A step forward towards commercialization. Molecules 2016, 21, 376. [Google Scholar] [CrossRef]
- Iulianelli, A.; Liguori, S.; Huang, Y.; Basile, A. Model biogas steam reforming in a thin Pd-supported membrane reactor to generate clean hydrogen for fuel cells. J. Power Sources 2015, 273, 25–32. [Google Scholar] [CrossRef]
- Pacheco Tanaka, D.A.; Llosa Tanco, M.A.; Nagase, T.; Okazaki, J.; Wakui, Y.; Mizukami, F.; Suzuki, T.M. Fabrication of hydrogen-permeable composite membranes packed with palladium nanoparticles. Adv. Mater. 2006, 18, 630–632. [Google Scholar] [CrossRef]
- Gade, S.K.; Thoen, P.M.; Way, J.D. Unsupported palladium alloy foil membranes fabricated by electroless plating. J. Memb. Sci. 2008, 316, 112–118. [Google Scholar] [CrossRef]
- Peters, T.A.; Stange, M.; Bredesen, R. On the high pressure performance of thin supported Pd–23%Ag membranes—Evidence of ultrahigh hydrogen flux after air treatment. J. Memb. Sci. 2011, 378, 28–34. [Google Scholar] [CrossRef]
- Mardilovich, P.P.; She, Y.; Ma, Y.H.; Rei, M.H. Defect-Free Palladium Membranes on Porous Stainless-Steel Support. AIChE J. 1998, 44, 310–322. [Google Scholar] [CrossRef]
- Mejdell, A.L.; Jøndahl, M.; Peters, T.A.; Bredesen, R.; Venvik, H.J. Effects of CO and CO2 on hydrogen permeation through a ~3 μm Pd/Ag 23 wt.% membrane employed in a microchannel membrane configuration. Sep. Purif. Technol. 2009, 68, 178–184. [Google Scholar] [CrossRef]
- Amandusson, H.; Ekedahl, L.G.; Dannetun, H. Hydrogen permeation through surface modified Pd and PdAg membranes. J. Memb. Sci. 2001, 193, 35–47. [Google Scholar] [CrossRef]
- Li, A.; Liang, W.; Hughes, R. The effect of carbon monoxide and steam on the hydrogen permeability of a Pd/stainless steel membrane. J. Memb. Sci. 2000, 165, 135–141. [Google Scholar] [CrossRef]
- Amano, M.; Nishimura, C.; Komaki, M. Effects of High Concentration CO and CO2 on Hydrogen Permeation through the Palladium Membrane. Mater. Trans. JIM 1990, 31, 404–408. [Google Scholar] [CrossRef]
- Eriksson, M.; Ekedahl, L.-G.G. Real time measurements of hydrogen desorption and absorption during CO exposures of Pd: Hydrogen sticking and dissolution. Appl. Surf. Sci. 1998, 133, 89–97. [Google Scholar] [CrossRef]
- Gallucci, F.; Chiaravalloti, F.; Tosti, S.; Drioli, E.; Basile, A. The effect of mixture gas on hydrogen permeation through a palladium membrane: Experimental study and theoretical approach. Int. J. Hydrogen Energy 2007, 32, 1837–1845. [Google Scholar] [CrossRef]
- Yamazaki, O.; Tomishige, K.; Fujimoto, K. Development of highly stable nickel catalyst for methane-steam reaction under low steam to carbon ratio. Appl. Catal. A Gen. 1996, 136, 49–56. [Google Scholar] [CrossRef]
- Rostrup-Nielsen, J.; Trimm, D.L. Mechanisms of carbon formation on nickel-containing catalysts. J. Catal. 1977, 48, 155–165. [Google Scholar] [CrossRef]
- Fernandez, E.; Coenen, K.; Helmi, A.; Melendez, J.; Zuñiga, J.; Pacheco Tanaka, D.A.; Van Sint Annaland, M.; Gallucci, F. Preparation and characterization of thin-film Pd-Ag supported membranes for high-temperature applications. Int. J. Hydrogen Energy 2015, 40, 13463–13478. [Google Scholar] [CrossRef]
- Suleiman, M.; Jisrawi, N.M.; Dankert, O.; Reetz, M.T.; Bähtz, C.; Kirchheim, R.; Pundt, A. Phase transition and lattice expansion during hydrogen loading of nanometer sized palladium clusters. J. Alloys Compd. 2003, 356–357, 644–648. [Google Scholar] [CrossRef]
- Baloyi, L.N.; North, B.C.; Langmi, H.W.; Bladergroen, B.J.; Ojumu, T.V. The production of hydrogen through the use of a 77 wt% Pd 23 wt% Ag membrane water gas shift reactor. S. Afr. J. Chem. Eng. 2016, 22, 44–54. [Google Scholar] [CrossRef]
- Guazzone, F.; Ma, Y.H. Leak growth mechanism in composite Pd membranes prepared by the electroless deposition method. AIChE J. 2008, 54, 487–494. [Google Scholar] [CrossRef]
- Goldbach, A.; Yuan, L.; Xu, H. Impact of the fcc/bcc phase transition on the homogeneity and behavior of PdCu membranes. Sep. Purif. Technol. 2010, 73, 65–70. [Google Scholar] [CrossRef]
- Hou, K.; Hughes, R. The effect of external mass transfer, competitive adsorption and coking on hydrogen permeation through thin Pd/Ag membranes. J. Memb. Sci. 2002, 206, 119–130. [Google Scholar] [CrossRef]
- King, H.W.; Manchester, F.D. A low-temperature X-ray diffraction study of Pd and some Pd-H alloys. J. Phys. F Met. Phys. 1978, 8, 15–26. [Google Scholar] [CrossRef]
- Baba, K.; Miyagawa, U.; Watanabe, K.; Sakamoto, Y.; Flanagan, T.B. Electrical resistivity changes due to interstitial hydrogen in palladium-rich substitutional alloys. J. Mater. Sci. 1990, 25, 3910–3916. [Google Scholar] [CrossRef]
- Ungár, T. Microstructural parameters from X-ray diffraction peak broadening. Scr. Mater. 2004, 51, 777–781. [Google Scholar] [CrossRef]


| Gas Mixture Type | Gas Mixture Feed | Gas Mixture Composition | Total Flow Rate (mL/min) |
|---|---|---|---|
| Pd Membrane | |||
| Binary | H2/Ar | 50/50 | 276 |
| Binary | H2/Ar | 50/50 | 542 |
| Binary | H2/Ar | 50/50 | 810 |
| Binary | H2/Ar | 50/50 | 1078 |
| Binary | H2/He | 50/50 | 276 |
| Binary | H2/CH4 | 50/50 | 276 |
| Binary | H2/H2O | 50/50 | 276 |
| Binary | H2/CO2 | 50/50 | 276 |
| Ternary | H2/CO/He | 50/5/45 | 276 |
| Ternary | H2/CO2/CH4 | 50/25/25 | 276 |
| Ternary | H2/CO2/H2O | 50/25/25 | 276 |
| Quaternary | H2/CO2/CO/He | 50/25/2.5/22.5 | 276 |
| Quaternary | H2/H2O/CO/He | 50/25/2.5/22.5 | 276 |
| Senary | H2/CO2/H2O/CH4/CO/He | 40/20/23/7/1/9 | 276 |
| Pd-Au Membrane | |||
| Binary | H2/Ar | 50/50 | 276 |
| Binary | H2/He | 50/50 | 276 |
| Binary | H2/N2 | 50/50 | 276 |
| Binary | H2/H2O | 50/50 | 276 |
| Membrane | ∆p (kPa) | Pd Thickness (µm) | T (°C) | Ea (kJ/mol) | P (mol/m2·s·Pa0.5) | n | Reference |
|---|---|---|---|---|---|---|---|
| Pd/Al2O3 | 400 | 5 | 300 | NA | * 10−6 | 0.5 | [50] |
| Pd/SS disk | 220 | 7.2 | 400 | NA | * 15.6 × 10−7 | 0.595 | [51] |
| Pd/SS disk | 220 | 9.6 | 400 | NA | * 10.6 × 10−4 | 0.595 | [51] |
| Pd/SS disk | 220 | 12.1 | 400 | NA | * 9.3 × 10−4 | 0.595 | [51] |
| Pd/SS disk | 220 | 14.6 | 400 | NA | * 5.5 × 10−4 | 0.595 | [51] |
| Pd-Ag23%/PSS | 2500 | 2.8 | 400 | NA | 6.48 × 10−3 | 0.5 | [52] |
| Pd-Ag23%/PSS | 1900 | 2.8 | 400 | NA | 6.20 × 10−3 | 0.5 | [52] |
| Pd-Ag23%/PSS | 1400 | 2.8 | 400 | NA | 5.96 × 10−3 | 0.5 | [52] |
| Pd-Ag23%/PSS | 900 | 2.8 | 400 | NA | 5.25 × 10−3 | 0.5 | [52] |
| Pd-Ag23%/PSS | 500 | 2.8 | 400 | NA | 4.66 × 10−3 | 0.5 | [52] |
| Pd-Ag23%/PSS | 100 | 2.8 | 400 | NA | 3.67 × 10−3 | 0.5 | [52] |
| Pd/PSS | 100 | 20 | 350 | 16.4 | * 5 × 10−7 | 0.5 | [53] |
| Pd/PSS | 50 | 10 | 400 | 14.7 | * 8.7 × 10−7 | 0.55 | [24] |
| Pd/Al2O3 | 200 | 7 | 400 | 11.7 | * 2.3 × 10−6 | 0.54 | [24] |
| Pd/YSZ | 50 | 11 | 400 | 12.8 | ** 2.54 × 10−5 | 0.7 | This work |
| Pd-Au/Al2O3 | 50 | 8 | 400 | NA | 8.42 × 10−4 | 0.5 | This work |
| ∆p (kPa) | αH2/He | αH2/Ar |
|---|---|---|
| Pd Membrane | ||
| 50 | 700 | 5900 |
| 100 | 660 | 4000 |
| 150 | 650 | 3800 |
| 200 | 640 | 3700 |
| Pd-Au Membrane | ||
| 50 | ∞ | ∞ |
| 100 | 6700 | 18,200 |
| 150 | 4600 | 12,400 |
| 2θ (°) | d (°A) | dlit (°A) | Rel. Int. (%) | h | k | l |
|---|---|---|---|---|---|---|
| Pd Membrane | ||||||
| 40.1307 | 2.24703 | 2.24439 | 100 | 1 | 1 | 1 |
| 46.7137 | 1.94457 | 1.94370 | 16.81 | 2 | 0 | 0 |
| 68.1775 | 1.37436 | 1.37440 | 16.55 | 2 | 2 | 0 |
| 82.2012 | 1.17177 | 1.17210 | 9.46 | 3 | 1 | 1 |
| 86.6651 | 1.12251 | 1.12220 | 4.27 | 2 | 2 | 2 |
| Pd-Au Membrane | ||||||
| 39.8528 | 2.26018 | 2.25802 | 100.00 | 1 | 1 | 1 |
| 46.4581 | 1.95305 | 1.95550 | 47.66 | 0 | 0 | 2 |
| 67.7534 | 1.38193 | 1.38275 | 15.56 | 0 | 2 | 2 |
| 81.7610 | 1.17696 | 1.17921 | 15.33 | 1 | 1 | 3 |
| 86.1299 | 1.12810 | 1.12901 | 4.26 | 2 | 2 | 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kian, K.; Woodall, C.M.; Wilcox, J.; Liguori, S. Performance of Pd-Based Membranes and Effects of Various Gas Mixtures on H2 Permeation. Environments 2018, 5, 128. https://doi.org/10.3390/environments5120128
Kian K, Woodall CM, Wilcox J, Liguori S. Performance of Pd-Based Membranes and Effects of Various Gas Mixtures on H2 Permeation. Environments. 2018; 5(12):128. https://doi.org/10.3390/environments5120128
Chicago/Turabian StyleKian, Kourosh, Caleb M Woodall, Jennifer Wilcox, and Simona Liguori. 2018. "Performance of Pd-Based Membranes and Effects of Various Gas Mixtures on H2 Permeation" Environments 5, no. 12: 128. https://doi.org/10.3390/environments5120128
APA StyleKian, K., Woodall, C. M., Wilcox, J., & Liguori, S. (2018). Performance of Pd-Based Membranes and Effects of Various Gas Mixtures on H2 Permeation. Environments, 5(12), 128. https://doi.org/10.3390/environments5120128






