Batch Fermentative Biohydrogen Production Process Using Immobilized Anaerobic Sludge from Organic Solid Waste
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pretreatment of Substrate
2.2. Inoculum Development
2.3. Immobilization of Bacteria with Calcium Alginate
2.4. Batch Fermentative Biohydrogen Production Process
2.5. Analytical Methods
3. Results and Discussion
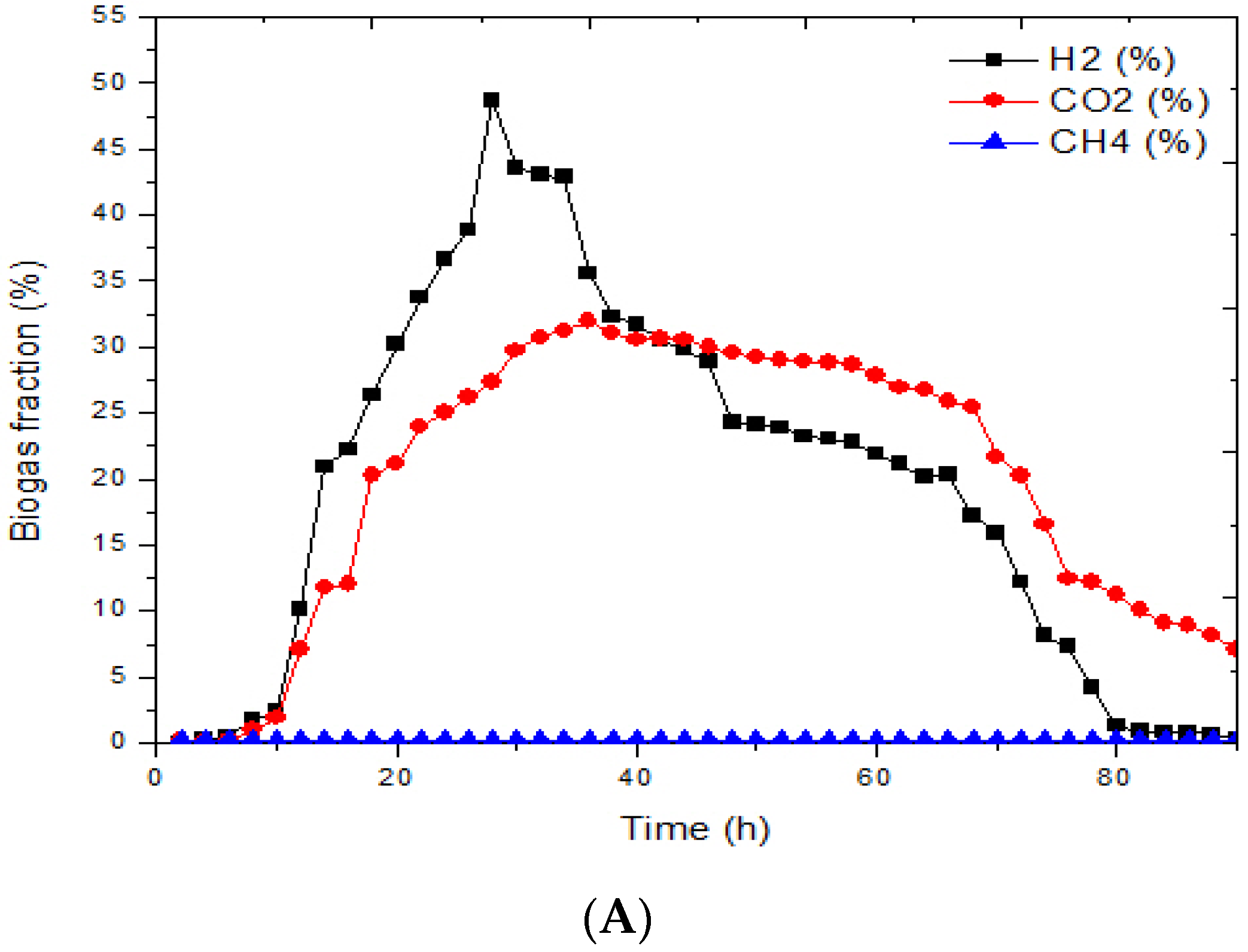
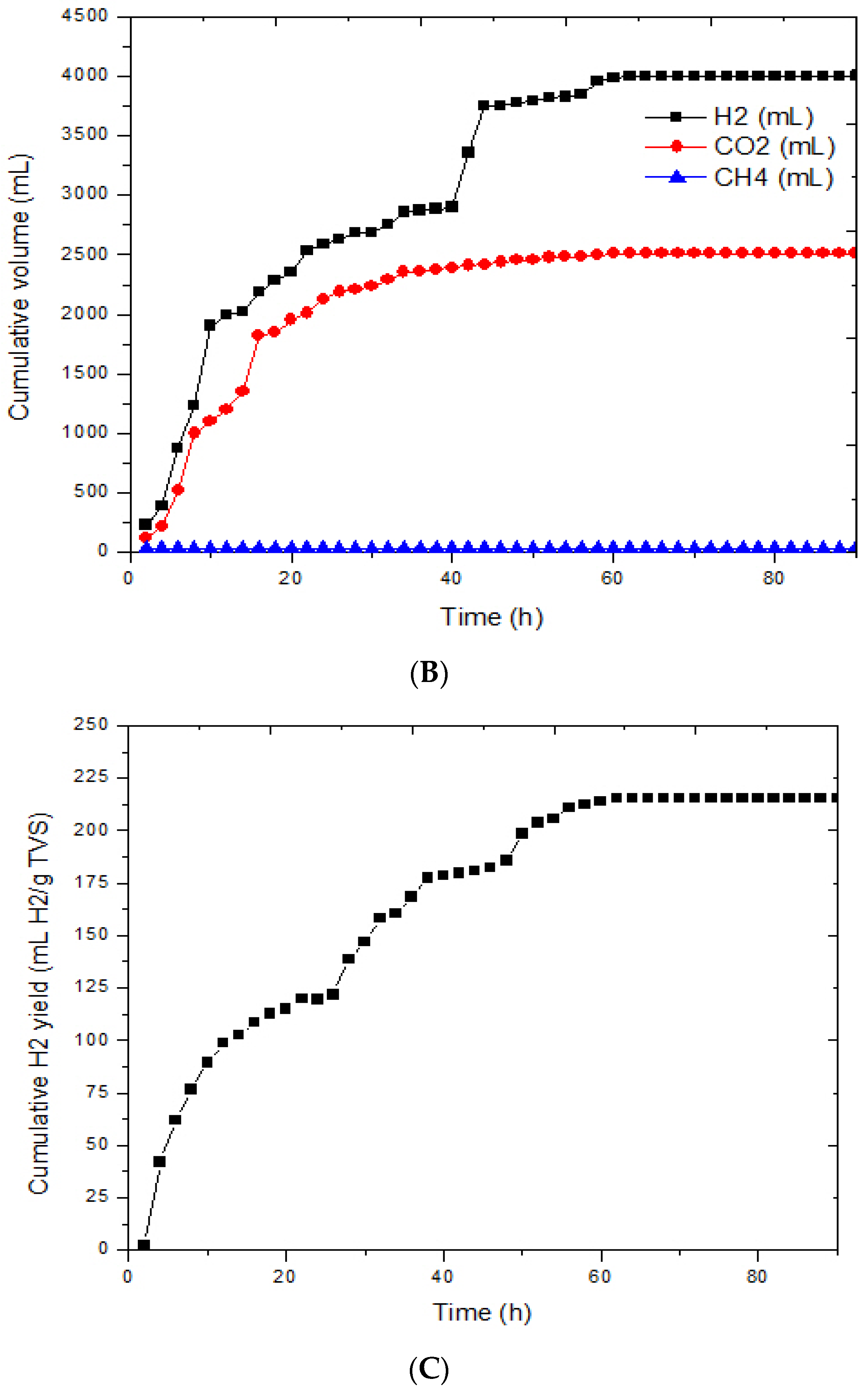
3.1. Effects of Immobilized Bacteria on Biohydrogen Production
3.2. Volatile Fatty Acids Production during Biohydrogen Fermentation
3.3. Strategies Used to Improve the Mechanical Stability of Calcium Alginate Matrix
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wu, K.; Chang, J.; Chang, C. Biohydrogen production using suspended and immobilized mixed microflora. J. Chin. Inst. Chem. Eng. 2006, 37, 545–550. [Google Scholar]
- Faloye, F.D.; Gueguim Kana, E.B. Optimization of biohydrogen inoculum development via a hybrid pH and microwave treatment technique—Semi pilot scale production assessment. Int. J. Hydrogen Energy 2014, 39, 5607–5616. [Google Scholar] [CrossRef]
- Ghimire, A.; Frunzo, L.; Pirozzi, F.; Trably, E.; Escudie, R.; Lens, P.N.L.; Esposito, G. A review on dark fermentative biohydrogen production from organic biomass: Process parameters and use of by-products. Appl. Energy 2015, 144, 73–95. [Google Scholar] [CrossRef]
- Chen, C.; Lin, C.; Chang, J. Kinetics of hydrogen production with continuous anaerobic cultures utilizing sucrose as the limiting substrate. Appl. Microbiol. Biotechnol. 2002, 57, 56–64. [Google Scholar]
- Wu, S.Y.; Lin, C.N.; Chang, J.S. Biohydrogen production with anaerobic sludge immobilized by ethylene-vinyl acetate copolymer. Int. J. Hydrogen Energy 2005, 30, 1375–1381. [Google Scholar] [CrossRef]
- Chang, J.S.; Lee, K.S.; Lin, P.J. Biohydrogen production with fixed-bed bioreactors. Int. J. Hydrogen Energy 2002, 27, 1167–1174. [Google Scholar] [CrossRef]
- Kumar, N.; Das, D. Continuous hydrogen production by immobilized Enterobacter cloacae IIT-BT 08 using lignocellulosic materials as solid matrices. Enzym. Microb. Technol. 2001, 29, 280–287. [Google Scholar] [CrossRef]
- Leino, H.; Kosourov, S.N.; Saari, L.; Sivonen, K.; Tsygankov, A.A.; Aro, E.M.; Allahverdiyeva, Y. Extended H2 photoproduction by N2-fixing cyanobacteria immobilized in thin alginate films. Int. J. Hydrogen Energy 2011, 37, 151–161. [Google Scholar] [CrossRef]
- Lin, C.N.; Wu, S.Y.; Chang, J.S.; Chang, J.S. Biohydrogen production in a three-phase fluidized bed bioreactor using sewage sludge immobilized by ethylene-vinyl acetate copolymer. Bioresour. Technol. 2009, 100, 3298–3301. [Google Scholar] [CrossRef] [PubMed]
- Seelert, T.; Ghosh, D.; Yargeau, V. Improving biohydrogen production using Clostridium beijerinckii immobilized with magnetite nanoparticles. Appl. Microbiol. Biotechnol. 2015, 9, 4107–4116. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.R.; Silva, E.L. Hydrogen and ethanol production in anaerobic fluidized bed reactors: Performance evaluation for three support materials under different operating conditions. Biochem. Eng. J. 2012, 61, 59–65. [Google Scholar] [CrossRef]
- Krasaekoopt, W.; Bhandari, B.; Deeth, H. The influence of coating materials on some properties of alginate beads and survivability of microencapsulated probiotic bacteria. Int. Dairy J. 2004, 14, 737–743. [Google Scholar] [CrossRef]
- Wongthanate, J.; Polprasert, C. Immobilized Biofilm in Thermophilic Biohydrogen Production using Synthetic versus Biological Materials. Braz. Arch. Biol. Technol. 2015, 58, 124–130. [Google Scholar] [CrossRef]
- Lin, S.Y.; Lin, C.N.; Chang, J.S.; Lee, K.S.; Lin, P.J. Microbial hydrogen production with immobilized sewage sludge. Biotechnol. Prog. 2002, 18, 921–926. [Google Scholar]
- Lin, C.Y.; Lee, C.Y.; Tseng, I.; Shiao, I. Biohydrogen production from sucrose using base-enriched anaerobic mixed microflora. Process Biochem. 2006, 41, 915–919. [Google Scholar] [CrossRef]
- Zhang, Z.P.; Show, K.Y.; Tay, J.H.; Liang, D.T.; Lee, D.J. Enhanced Continuous Biohydrogen Production by Immobilized Anaerobic Microflora. Energy Fuels 2007, 22, 87–92. [Google Scholar] [CrossRef]
- Chang, F.Y.; Lin, C.Y. Biohydrogen production using an up-flow anaerobic sludge blanket reactor. Int. J. Hydrogen Energy 2004, 29, 33–39. [Google Scholar] [CrossRef]
- Lee, K.S.; Lin, P.J.; Chang, J.S. Temperature effects on biohydrogen production in a granular sludge bed induced by activated carbon carriers. Int. J. Hydrogen Energy 2006, 31, 465–472. [Google Scholar] [CrossRef]
- Wu, S.Y.; Hung, C.H.; Lin, C.N.; Chen, H.W.; Lee, A.S.; Chang, J.S. Fermentative hydrogen production and bacterial community structure in high-rate anaerobic bioreactors containing silicone-immobilized and self-flocculated sludge. Biotechnol. Bioeng. 2006, 93, 934–946. [Google Scholar] [CrossRef] [PubMed]
- Seol, E.; Manimaran, A.; Jang, Y.; Kim, S.; Oh, Y.K.; Park, S. Sustained hydrogen production from formate using immobilized recombinant Escherichia coli SH5. Int. J. Hydrogen Energy 2011, 36, 8681–8686. [Google Scholar] [CrossRef]
- Smidsrod, O. Alginate as immobilization matrix for cells. Trends Biotechnol. 1990, 8, 71–78. [Google Scholar] [CrossRef]
- Tan, T.W.; Jin, X.H.; Hu, B.; Zhang, M.J. Release behavior of ketoprofen from chitosan/alginate microcapsules. Bioact. Compat. Polym. 2003, 18, 207–218. [Google Scholar] [CrossRef]
- Klein, J.; Stock, J.; Vorlop, K.D. Pore size and properties of spherical Ca-alginate biocatalysts. Eur. J. Appl. Microbiol. Biotechnol. 1983, 18, 86–91. [Google Scholar] [CrossRef]
- Singh, L.; Wahid, Z.A.; Siddiqui, M.F.; Ahmad, A.; Ab Rahim, M.H.; Sakinah, M. Application of immobilized upflow anaerobic sludge blanket reactor using Clostridium LS2 for enhanced biohydrogen production and treatment efficiency of palm oil mill effluent. Int. J. Hydrogen Energy 2012, 38, 2221–2229. [Google Scholar] [CrossRef]
- Department of Environmental Affairs. National Waste Information Baseline Report; Department of Environmental Affairs: Pretoria, South Africa, 2014.
- Broomfield, M.; Davies, J.; Harrison, R. Review of Environmental and Health Effects of Management of Municipal Solid Waste; Department for Environment, Food and Rural Affairs: London, UK, 2004.
- Tadesse, T. Solid and Hazardous Waste Management; Ethiopia Public Health Training Initiative: Addis Ababa, Ethiopia, 2004. [Google Scholar]
- Devesa-Rey, R.; Vecino, X.; Varela-Alende, J.L.; Barral, M.T.; Cruz, J.M.; Moldes, A.B. Valorization of winery waste vs. the costs of not recycling. Waste Manag. 2011, 31, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Gomez, X.; Moran, A.; Cuetos, M.; Sanchez, M. The production of hydrogen by dark fermentation of municipal solid wastes and slaughterhouse waste. J. Power Sources 2006, 157, 727–732. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC,USA, 1998. [Google Scholar]
- Sekoai, P.T.; Gueguim Kana, E.B. Semi-pilot scale production of hydrogen from Organic Fraction of Solid Municipal Waste and electricity generation from process effluents. Biomass Bioenergy 2014, 60, 156–163. [Google Scholar] [CrossRef]
- Akinbomi, J.; Wikandari, R.; Taherzadeh, M.J. Enhanced Fermentative Hydrogen and Methane Production from an Inhibitory Fruit-Flavored Medium with Membrane-Encapsulated Cells. Membranes 2015, 5, 616–631. [Google Scholar] [CrossRef] [PubMed]
- Keskin, T.; Giusti, L.; Azbar, N. Continuous biohydrogen production in immobilized biofilm system versus suspended cell culture. Int. J. Hydrogen Energy 2012, 37, 1418–1424. [Google Scholar] [CrossRef]
- Cai, G.; Jin, B.; Monis, P.; Saint, C. Metabolic flux network and analysis of fermentative hydrogen production. Biotechnol. Adv. 2011, 29, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Junior, A.D.N.Z.; Zaiat, M.; Gupta, M.; Elbeshbishy, E.; Hafez, H.; Nakhla, G. Impact of organic loading rate on biohydrogen production in an up-flow anaerobic packed bed reactor (UAnPBR). Bioresour. Technol. 2014, 164, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.Y.; Liu, C.Z. Hydrogen production via thermophilic fermentation of cornstalk by Clostridium thermocellum. Energy Fuels 2011, 25, 1714–1720. [Google Scholar] [CrossRef]
- Wang, X.; Jin, B. Process optimization of biological hydrogen production from molasses by a newly isolated Clostridium butyricum W5. Biosci. Bioeng. 2009, 107, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, H.; Tokushige, T.; Hirose, J.; Hayashi, S.; Takasaki, Y. Hydrogen production by immobilized cells of aciduric Enterobacter aerogenes strain HO-39. J. Ferment. Bioeng. 1997, 83, 481–484. [Google Scholar] [CrossRef]
- Leenen, E.J.; Dos Santos, V.A.; Grolle, K.C.; Tramper, J.; Wijffels, R. Characteristics of and selection criteria for support materials for cell immobilization in wastewater treatment. Water Res. 1996, 30, 2985–2996. [Google Scholar] [CrossRef]
- Moreno-Garrido, I. Microalgae immobilization: Current techniques and uses. Bioresour. Technol. 2008, 99, 3949–3964. [Google Scholar] [CrossRef] [PubMed]
- Pekguzel, E.A.; Gundogdu, T.K.; Azbar, N. Enhancement of Biohydrogen Production via Thermophilic Cell Culture Immobilized on Glass Beads and Raschig Rings of Different Sizes in a Packed Bed Reactor. Chem. Biochem. Eng. Q. 2015, 24, 541–547. [Google Scholar] [CrossRef]
| Parameter | Value |
|---|---|
| pH | 7.2 ± 0.2 |
| Total COD (mg/L) | 2010 ± 1630 |
| VSS (mg/L) | 1015 ± 428 |
| TS (mg/L) | 1308 ± 1820 |
| TVS (mg/L) | 335 ± 720 |
| Parameter | Value |
|---|---|
| Total COD (mg/L) | 2210 ± 1852 |
| Soluble COD (mg/L) | 736 ± 35 |
| BOD (mg/L) | 1623 ± 1520 |
| TKN (mg/L) | 198 ± 121 |
| TS (mg/L) | 1123 ± 1310 |
| TVS (mg/L) | 835 ± 456 |
| pH | 6.8 ± 0.2 |
| Elemental composition (dry weight) | |
| C (%) | 42.58 |
| H (%) | 6.32 |
| N (%) | 0.36 |
| S (%) | 0.85 |
| Time (h) | Volatile Fatty Acids | mg/L | Molar % |
|---|---|---|---|
| 4 | Acetate | 482.1 | 56.37 |
| Butyrate | 525.3 | 41.86 | |
| Propionate | 18.7 | 1.77 | |
| 12 | Acetate | 589.7 | 57.19 |
| Butyrate | 624.4 | 41.27 | |
| Propionate | 19.7 | 1.55 | |
| 20 | Acetate | 896.0 | 68.09 |
| Butyrate | 575.7 | 29.82 | |
| Propionate | 33.9 | 2.09 | |
| 32 | Acetate | 1767.3 | 66.59 |
| Butyrate | 1258.7 | 32.32 | |
| Propionate | 35.6 | 1.09 | |
| 40 | Acetate | 1249.5 | 53.07 |
| Butyrate | 1582.3 | 45.8 | |
| Propionate | 32.7 | 1.13 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekoai, P.T.; Yoro, K.O.; Daramola, M.O. Batch Fermentative Biohydrogen Production Process Using Immobilized Anaerobic Sludge from Organic Solid Waste. Environments 2016, 3, 38. https://doi.org/10.3390/environments3040038
Sekoai PT, Yoro KO, Daramola MO. Batch Fermentative Biohydrogen Production Process Using Immobilized Anaerobic Sludge from Organic Solid Waste. Environments. 2016; 3(4):38. https://doi.org/10.3390/environments3040038
Chicago/Turabian StyleSekoai, Patrick T., Kelvin O. Yoro, and Michael O. Daramola. 2016. "Batch Fermentative Biohydrogen Production Process Using Immobilized Anaerobic Sludge from Organic Solid Waste" Environments 3, no. 4: 38. https://doi.org/10.3390/environments3040038
APA StyleSekoai, P. T., Yoro, K. O., & Daramola, M. O. (2016). Batch Fermentative Biohydrogen Production Process Using Immobilized Anaerobic Sludge from Organic Solid Waste. Environments, 3(4), 38. https://doi.org/10.3390/environments3040038






