Sex-Specific Effects of Childhood Poverty on Neurocircuitry of Processing of Emotional Cues: A Neuroimaging Study
Abstract
:1. Introduction
2. Method
2.1. Participants
2.2. Experimental Tasks
2.2.1. Acquisition of MRI Data
2.2.2. MRI Data Analysis
3. Results
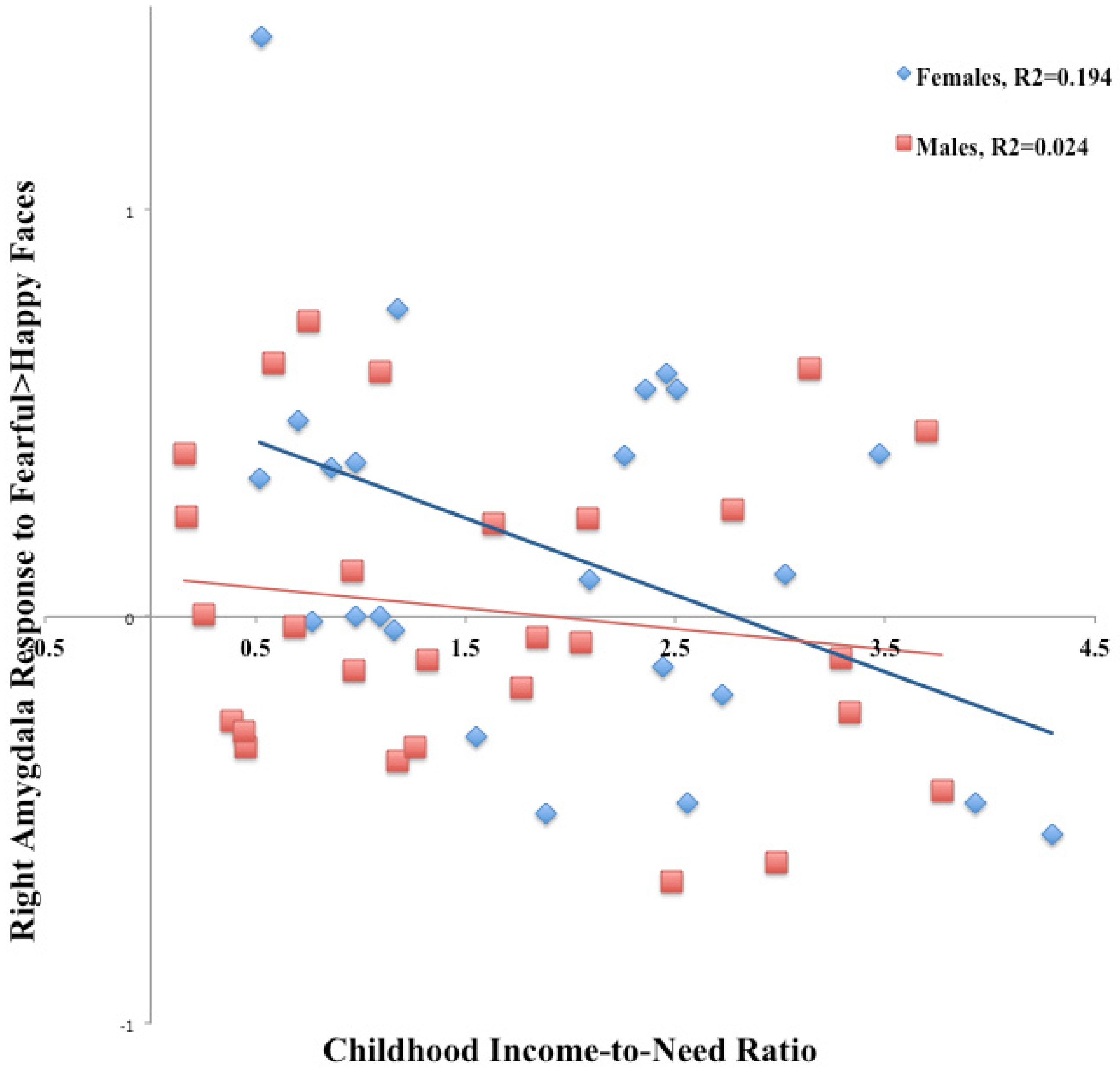
3.1. Emotional Face Assessment Task (EFAT)
3.2. Emotion Regulation Task (ERT)
3.3. Shifted-Attention Emotion Appraisal Task (SEAT)
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AAL | Automated Anatomical Labeling |
| DLPFC | Dorsolateral Prefrontal Cortex |
| DSM | Diagnostic and Statistical Manual |
| EFAT | Emotional Face Assessment Task |
| ERT | Emotion Regulation Task |
| MRI | Magnetic Resonance Imaging |
| SEAT | Shifting Emotion Attention Task |
| VLPFC | ventrolateral Prefrontal Cortex |
References
- Adler, N.E.; Rehkopf, D.H. U.S. disparities in health: Descriptions, causes, and mechanisms. Annu. Rev. Public Health 2008, 29, 235–252. [Google Scholar] [CrossRef] [PubMed]
- Birnie, K.; Cooper, R.; Martin, R.M.; Kuh, D.; Sayer, A.A.; Alvarado, B.E.; Bayer, A.; Christensen, K.; Cho, S.I.; Cooper, C.; et al. Childhood socioeconomic position and objectively measured physical capability levels in adulthood: A systematic review and meta-analysis. PLoS ONE 2011, 6, e15564. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Cohen, P.; West, S.G.; West, L.S. Applied Multiple Regression/Correlation Analysis for the Behavioral Sciences; Erlbaum Press: New York, NY, USA, 2003; pp. 375–388. [Google Scholar]
- Shonkoff, J.P.; Boyce, W.T.; McEwen, B.S. Neuroscience, molecular biology, and the childhood roots of health disparities: Building a new framework for health promotion and disease prevention. JAMA 2009, 301, 2252–2259. [Google Scholar] [CrossRef] [PubMed]
- Wadsworth, M.E.; Evans, G.W.; Grant, K.; Carter, J.S.; Duffy, S. Poverty and the development of psychopathology. In Developmental Psychopathology; Cicchetti, D., Ed.; Wiley: New York, NY, USA, 2016. [Google Scholar]
- Poulton, R.; Caspi, A.; Milne, B.J.; Thomson, W.M.; Taylor, A.; Sears, M.R.; Moffitt, T.E. Association between children’s ex-perience of socioeconomic disadvantage and adult health: A life-course study. Lancet 2002, 360, 1640–1645. [Google Scholar] [CrossRef]
- Mattingly, M.J.; Carson, J.A.; Schaefer, A. 2012 National Child Poverty Rate Stagnates at 22.6 Percent: New Hampshire Child Poverty Jumps 30 Percent Since 2011; National Issue Brief #65; Carsey Institute, University of New Hampshire: Durham, NH, USA, 2013. [Google Scholar]
- Hanson, J.L.; Nacewicz, B.M.; Sutterer, M.J.; Cayo, A.A.; Schaefer, S.M.; Rudolph, K.D.; Shirtcliff, E.A.; Pollak, S.D.; Davidson, R.J. Behavioral Problems After Early Life Stress: Contributions of the Hippocampus and Amygdala. Biol. Psychiatry 2015, 77, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Luby, J.; Belden, A.; Botteron, K.; Marrus, N.; Harms, M.P.; Babb, C.; Nishino, T.; Barch, D. The effects of poverty on childhood brain development: The mediating effect of caregiving and stressful life events. JAMA Pediatr. 2013, 167, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Noble, K.G.; Houston, S.M.; Kan, E.; Sowell, E.R. Neural correlates of socioeconomic status in the developing human brain. Dev. Sci. 2012, 15, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Gianaros, P.J.; Horenstein, J.A.; Hariri, A.R.; Sheu, L.K.; Manuck, S.B.; Matthews, K.A.; Cohen, S. Potential neural embedding of parental social standing. Soc. Cogn. Affect. Neurosci. 2008, 3, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.; Ho, S.S.; Evans, G.W.; Liberzon, I.; Swain, J.E. Childhood social inequalities influences neural processes in young adult caregiving. Dev. Psychobiol. 2015, 57, 948–960. [Google Scholar] [CrossRef] [PubMed]
- Javanbakht, A.; King, A.P.; Evans, G.W.; Swain, J.E.; Angstadt, M.; Phan, K.L.; Liberzon, I. Childhood poverty predicts adult amygdala and frontal activity and connectivity in response to emotional faces. Front. Behav. Neurosci. 2015, 9, 154. [Google Scholar] [CrossRef] [PubMed]
- Gianaros, P.J.; Horenstein, J.A.; Cohen, S.; Matthews, K.A.; Brown, S.M.; Flory, J.D.; Critchley, H.D.; Manuck, S.B.; Hariri, A.R. Perigenual anterior cingulate morphology covaries with perceived social standing. Soc. Cogn. Affect. Neurosci. 2007, 2, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Silverman, M.E.; Muennig, P.; Liu, X.; Rosen, Z.; Goldstein, M.A. The impact of socioeconomic status on the neural substrates associated with pleasure. Open Neuroimage J. 2009, 3, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Liberzon, I.; Ma, S.T.; Okada, G.; Shaun Ho, S.; Swain, J.E.; Evans, G.W. Childhood poverty and recruitment of adult emotion regulatory neurocircuitry. Soc. Cogn. Affect. Neurosci. 2015, 10, 1596–1606. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.; Evans, G.W.; Angstadt, M.; Ho, S.S.; Sripada, C.S.; Swain, J.E.; Liberzon, I.; Phan, K.L. Effects of childhood poverty and chronic stress on emotion regulatory brain function in adulthood. Proc. Natl. Acad. Sci. USA 2013, 110, 18442–18447. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; McGonagle, K.A.; Nelson, C.B.; Hughes, M.; Swartz, M.; Blazer, D.G. Sex and depression in the National Comorbidity Survey. II: Cohort effects. J. Affect. Disord. 1994, 1, 15–26. [Google Scholar] [CrossRef]
- Bangasser, D.A.; Valentino, R.J. Sex differences in stress-related psychiatric disorders: Neurobiological perspectives. Front. Neuroendocrinol. 2014, 35, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Merikangas, K.R.; He, J.P.; Burstein, M.; Swanson, S.A.; Avenevoli, S.; Cui, L.; Benjet, C.; Georgiades, K.; Swendsen, J. Lifetime prevalence of mental disorders in U.S. adolescents: Results from the National Comorbidity Survey Replication--Adolescent Supplement (NCS-A). J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; McGonagle, K.A.; Zhao, S.; Nelson, C.B.; Hughes, M.; Eshleman, S.; Wittchen, H.U.; Kendler, K.S. Lifetime and 12 month prevalence of, D.S.M-111-R psychiatric disorders in the United States. Arch. Gen. Psychiatry 1994, 51, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.; Gunnell, D. Suicide in England and Wales 1861–2007: A time-trends analysis. Int. J. Epidemiol. 2010, 39, 1464–1475. [Google Scholar] [CrossRef] [PubMed]
- Stark, R.; Wolf, O.T.; Tabbert, K.; Kagerer, S.; Zimmermann, M.; Kirsch, P.; Schienle, A.; Vaitl, D. Influence of the stress hormone cortisol on fear conditioning in humans: Evidence for sex differences in the response of the prefrontal cortex. Neuroimage 2006, 32, 1290–1298. [Google Scholar] [CrossRef] [PubMed]
- Merz, C.J.; Wolf, O.T.; Schweckendiek, J.; Klucken, T.; Vaitl, D.; Stark, R. Stress differentially affects fear conditioning in men and women. Psychoneuroendocrinology 2013, 38, 2529–2541. [Google Scholar] [CrossRef] [PubMed]
- Bradley, M.M.; Cuthbert, B.N.; Lang, P.N. Affect and the startle reflex. In Startle Modification: Implications for Neuroscience, Cognitive Science, and Clinical Science; Dawson, M.E., Schell, A.M., Bohmelt, A.H., Eds.; Cambridge University Press: New York, NY, USA, 1999; pp. 157–183. [Google Scholar]
- Canli, T.; Desmond, J.E.; Zho, Z.; Gabrieli, J.D. Sex differences in the neural basis of emotional memories. Proc. Natl. Acad. Sci. USA 2002, 99, 10789–10794. [Google Scholar] [CrossRef] [PubMed]
- Ohrmann, P.; Pedersen, A.; Braun, M.; Bauer, J.; Kugel, H.; Kersting, A.; Domschke, K.; Deckert, J.; Suslow, T. Effect of gender on processing threat-related stimuli in patients with panic disorder: Sex does matter. Depress. Anxiety 2010, 27, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- McClure, E.B.; Monk, C.S.; Nelson, E.E.; Zarahn, E.; Leibenluft, E.; Bilder, R.M.; Charney, D.S.; Ernst, M.; Pine, D.S. A developmental examination of gender differences in brain engagement during evaluation of threat. Biol. Psychiatry 2004, 55, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Brody, L.R.; Hall, J.A. Gender and Emotion. In Handbook of Emotions; Lewis, M., Haviland, J.M., Eds.; Guilford: New York, NY, USA, 1993; pp. 447–460. [Google Scholar]
- Vijayakumar, N.; Whittle, S.; Yücel, M.; Dennison, M.; Simmons, J.; Allen, N.B. Thinning of the lateral prefrontal cortex during adolescence predicts emotion regulation in females. Soc. Cogn. Affect. Neurosci. 2014, 9, 1845–1854. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W. A multimethodological analysis of cumulative risk and allostatic load among rural children. Dev. Psychol. 2003, 39, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Phan, K.L.; Fitzgerald, D.A.; Nathan, P.J.; Moore, G.J.; Uhde, T.W.; Tancer, M.E. Neural substrates for voluntary suppression of negative affect: A functional magnetic resonance imaging study. Biol. Psychiatry 2005, 57, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, M.; Matlina, E.; Maor, G.I.; Rosen, H.; McEwen, B.S. Prenatal stress selectively alters the reactivity of the hypothalamic-pituitary adrenal system in the female rat. Brain Res. 1992, 595, 195–200. [Google Scholar] [CrossRef]
- McCormick, C.M.; Smythe, J.W.; Sharma, S.; Meaney, M.J. Sex-specific effects of prenatal stress on hypothalamic–pituitary–adrenal responses to stress and brain glucocorticoid receptor density in adults rats. Dev. Brain Res. 1995, 84, 55–61. [Google Scholar] [CrossRef]
- Kayser, V.; Berkley, K.J.; Keita, H.; Gautron, M.; Guilbaud, G. Estrous and sex variations in vocalization thresholds to hind paw and tail pressure stimulation in the rat. Brain Res. 1996, 742, 352–354. [Google Scholar] [CrossRef]
- Chaloner, A.; Greenwood-Van Meerveld, B. Sexually dimorphic effects of unpredictable early life adversity on visceral pain behavior in a rodent model. J. Pain 2013, 14, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Myers, B.; Schulkin, J.; Greenwood-Van Meerveld, B. Sex steroids localized to the amygdala increase pain responses to visceral stimulation in rats. J. Pain 2011, 12, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L.; Haier, R.J.; White, N.S.; Fallon, J.; Kilpatrick, L.; Lawrence, C.; Potkin, S.G.; Alkire, M.T. Sex-related difference in amygdala activity during emotionally influenced memory storage. Neurobiol. Learn. Mem. 2001, 75, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L.; Uncapher, M.; Kilpatrick, L.; Alkire, M.T.; Turner, J. Sex-related hemispheric lateralization of amygdala function in emotionally influenced memory: An fMRI investigation. Learn. Mem. 2004, 11, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Suenaga, T.; Yukie, M.; Gao, S.; Nakahara, D. Sex-specific effects of prenatal stress on neuronal development in the medial prefrontal cortex and the hippocampus. Neuroreport 2012, 23, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Blaze, J.; Scheuing, L.; Roth, T.L. Differential methylation of genes in the medial prefrontal cortex of developing and adult rats following exposure to maltreatment or nurturing care during infancy. Dev. Neurosci. 2014, 35, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Hariri, A.R.; Tessitore, A.; Mattay, V.S.; Fera, F.; Weinberger, D.R. The amygdala response to emotional stimuli: A comparison of faces and scenes. Neuroimage 2002, 17, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Britton, J.C.; Taylor, S.F.; Sudheimer, K.D.; Liberzon, I. Facial expressions and complex IAPS pictures: Common and differential networks. Neuroimage 2006, 31, 906–919. [Google Scholar] [CrossRef] [PubMed]
| Study | Number of Participants | M/F | Age |
|---|---|---|---|
| EFAT | 52 | 28/24 | 24.4 ± 1.2 |
| ERT | 49 | 27/22 | 23.6 ± 1.3 |
| SEAT | 49 | 27/22 | 23.7 ± 1.3 |
| Task/Brain Region | Beta Coefficient | T-Ratio | Significance Level |
|---|---|---|---|
| EFAT Right Amygdala Fearful > Happy | |||
| Age | −0.43 | −1.64 | 0.11 |
| CITN | −0.78 | −2.86 | 0.007 |
| AITN | 0.45 | 0.126 | 0.9 |
| Gender | −5.43 | −1.75 | 0.087 |
| Gender by Age | 4.73 | 1.62 | 0.11 |
| Gender by AITN | −0.28 | −0.67 | 0.51 |
| Gender by CITN | 0.80 | 2.25 | 0.03 |
| EFAT Right Amygdala Fearful | |||
| Age | −0.61 | −2.39 | 0.02 |
| CITN | −0.65 | −2.42 | 0.02 |
| AITN | 0.16 | 0.05 | 0.96 |
| Gender | −10.80 | −3.58 | 0.001 |
| Gender by Age | 10.17 | 3.60 | 0.001 |
| Gender by AITN | 0.86 | 2.48 | 0.017 |
| Gender by CITN | −0.14 | −0.34 | 0.74 |
| ERT DLPFC Reappraise > Maintain | |||
| Age | −0.63 | −2.12 | 0.04 |
| CITN | −5.41 | −1.20 | 0.05 |
| AITN | 4.16 | 1.03 | 0.3 |
| Gender | −5.01 | −2.24 | 0.03 |
| Gender by Age | 4.98 | 2.23 | 0.03 |
| Gender by AITN | −4.17 | −1.04 | 0.30 |
| Gender by CITN | 5.71 | 2.18 | 0.03 |
| ERT VLPFC Reappraise > Maintain | |||
| Age | −0.53 | −1.58 | 0.12 |
| CITN | −5.67 | −1.84 | 0.07 |
| AITN | 6.73 | 1.47 | 0.15 |
| Gender | −5.20 | −2.05 | 0.046 |
| Gender by Age | 5.21 | 2.07 | 0.05 |
| Gender by AITN | −6.82 | −1.51 | 0.14 |
| Gender by CITN | 5.78 | 1.95 | 0.05 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Javanbakht, A.; Kim, P.; Swain, J.E.; Evans, G.W.; Phan, K.L.; Liberzon, I. Sex-Specific Effects of Childhood Poverty on Neurocircuitry of Processing of Emotional Cues: A Neuroimaging Study. Behav. Sci. 2016, 6, 28. https://doi.org/10.3390/bs6040028
Javanbakht A, Kim P, Swain JE, Evans GW, Phan KL, Liberzon I. Sex-Specific Effects of Childhood Poverty on Neurocircuitry of Processing of Emotional Cues: A Neuroimaging Study. Behavioral Sciences. 2016; 6(4):28. https://doi.org/10.3390/bs6040028
Chicago/Turabian StyleJavanbakht, Arash, Pilyoung Kim, James E. Swain, Gary W. Evans, K. Luan Phan, and Israel Liberzon. 2016. "Sex-Specific Effects of Childhood Poverty on Neurocircuitry of Processing of Emotional Cues: A Neuroimaging Study" Behavioral Sciences 6, no. 4: 28. https://doi.org/10.3390/bs6040028
APA StyleJavanbakht, A., Kim, P., Swain, J. E., Evans, G. W., Phan, K. L., & Liberzon, I. (2016). Sex-Specific Effects of Childhood Poverty on Neurocircuitry of Processing of Emotional Cues: A Neuroimaging Study. Behavioral Sciences, 6(4), 28. https://doi.org/10.3390/bs6040028







