Filial Imprinting: Behaviour and Neurobiology
Abstract
1. Introduction
2. Learning the Features of an Imprinting Stimulus
3. The Preference for a Familiar Object as a Measure of Imprinting
4. Inference of Learning from Preference Score
5. The Correlation Between a Neurobiological Measurement and Preference Score
- = variance of trained chicks;
- = correlation coefficient and is the proportion of variance attributable to the correlation;
- = residual variance of trained chicks about the regression line.
- Each component of the above model can be estimated experimentally.
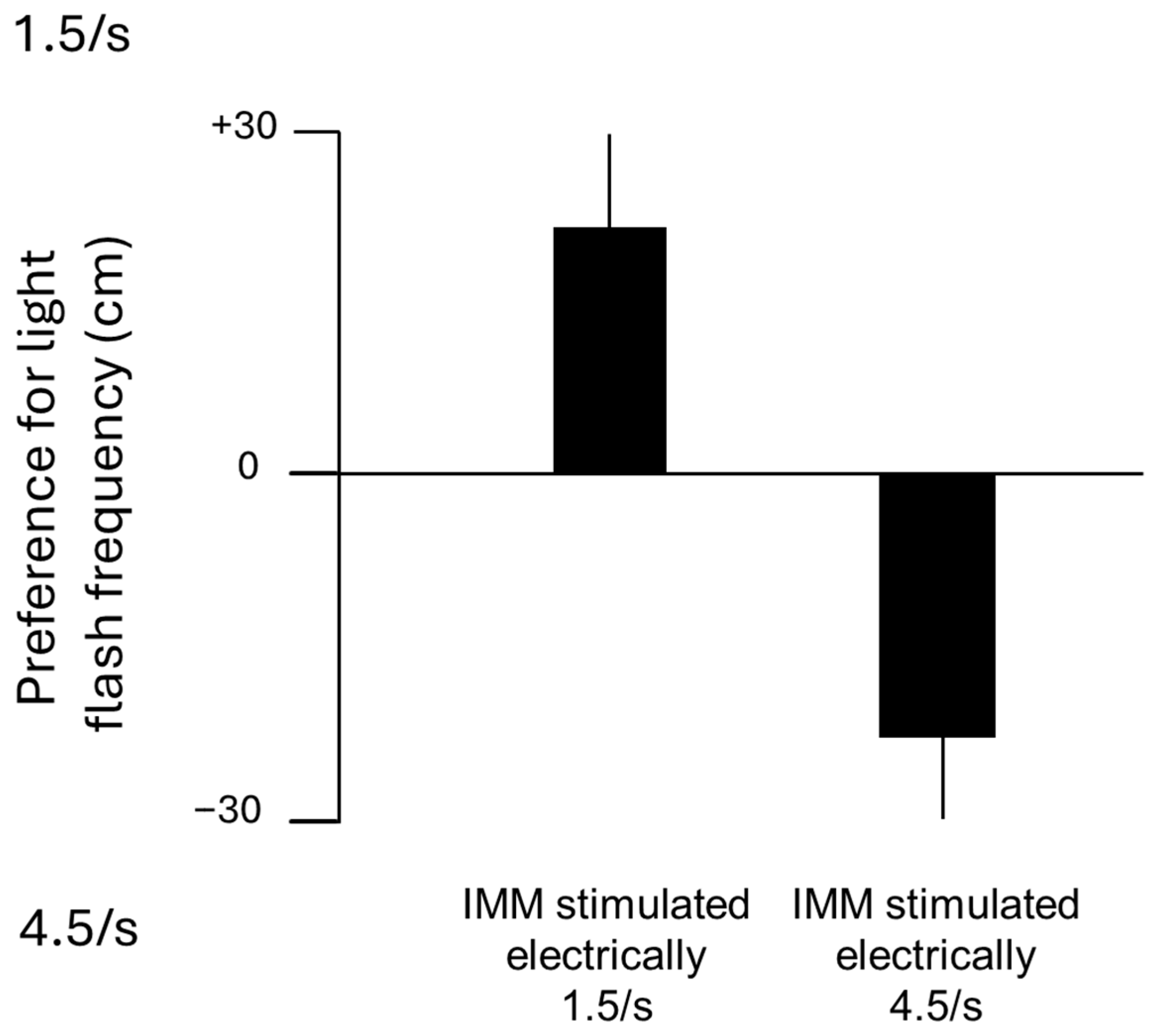
6. The IMM (Previously Named IMHV)
7. Memory Processing in the IMM
8. Electrophysiological Studies of the IMM
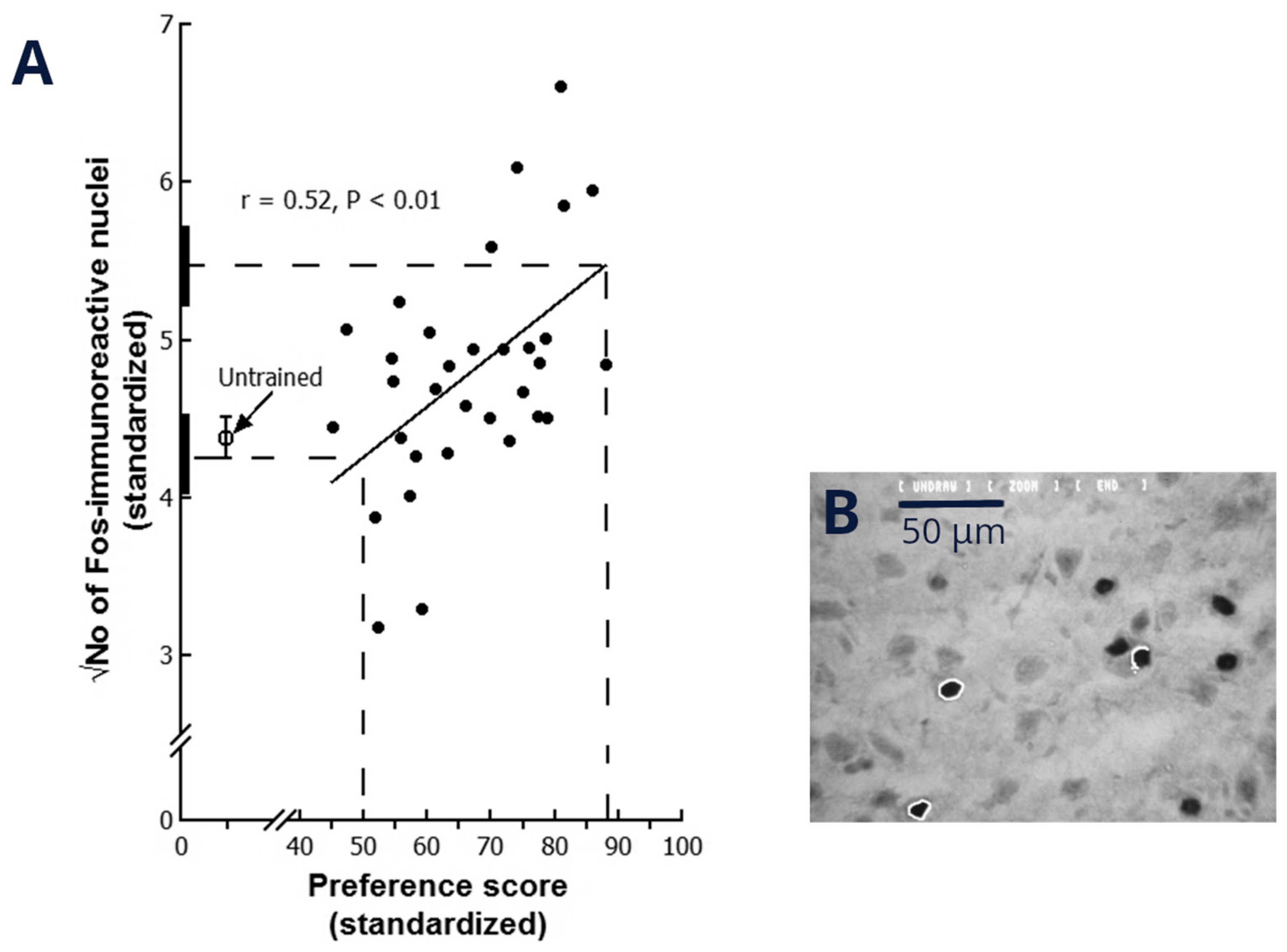
9. Learning-Related Synaptic Changes in the IMM Following Imprinting Training
10. Implications for Human Development: Predispositions
11. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SEM | Standard error of the mean |
| RNA | Ribonucleic acid |
References
- Ambalavanar, R., McCabe, B. J., Potter, K. N., & Horn, G. (1999). Learning-related Fos-like immunoreactivity in the chick brain: Time-course and co-localization with GABA and parvalbumin. Neuroscience, 93(4), 1515–1524. [Google Scholar] [CrossRef]
- Ambalavanar, R., Van der Zee, E. A., Bolhuis, J. J., McCabe, B. J., & Horn, G. (1993). Co-expression of Fos immunoreactivity in protein kinase (PKCγ)-positive neurones: Quantitative analysis of a brain region involved in learning. Brain Research, 606(2), 315–318. [Google Scholar] [CrossRef]
- Aoki, N., Yamaguchi, S., Fujita, T., Mori, C., Fujita, E., Matsushima, T., & Homma, K. J. (2018). GABA-A and GABA-B receptors in filial imprinting linked with opening and closing of the sensitive period in domestic chicks (Gallus gallus domesticus). Frontiers in Physiology, 9, 1837. [Google Scholar] [CrossRef]
- Bateson, P. (1964). An effect of imprinting on the perceptual development of domestic chicks. Nature, 202(4930), 421–422. [Google Scholar] [CrossRef] [PubMed]
- Bateson, P. (1966). The characteristics and context of imprinting. Biological Reviews, 41, 177–220. [Google Scholar] [CrossRef]
- Bateson, P. (1973). Preferences for familiarity and novelty: A model for the simultaneous development of both. Journal of Theoretical Biology, 41(2), 249–259, (Correction in 1974, Journal of Theoretical Biology, 45(1), 293). [Google Scholar] [CrossRef]
- Bateson, P. (1978). Sexual imprinting and optimal outbreeding. Nature, 273(5664), 659–660. [Google Scholar] [CrossRef] [PubMed]
- Bateson, P. (1982). Preferences for cousins in japanese quail. Nature, 295(5846), 236–237. [Google Scholar] [CrossRef]
- Bateson, P. (1983). Optimal outbreeding. In Mate choice (pp. 257–277). Cambridge University Press. [Google Scholar]
- Bateson, P., & Chantrey, D. F. (1972). Retardation of discrimination learning in monkeys and chicks previously exposed to both stimuli. Nature, 237(5351), 173–174. [Google Scholar] [CrossRef]
- Bateson, P., & Jaeckel, J. B. (1976). Chicks’ preferences for familiar and novel conspicuous objects after different periods of exposure. Animal Behaviour, 24(2), 386–390. [Google Scholar] [CrossRef]
- Bateson, P., & Wainwright, A. A. (1972). Effects of prior exposure to light on imprinting process in domestic chicks. Behaviour, 43, 279–290. [Google Scholar] [CrossRef]
- Behroozi, M., Lorenzi, E., Tabrik, S., Tegenthoff, M., Gozzi, A., Güntürkün, O., & Vallortigara, G. (2022). Functional MRI reveals memories of mother in newborn chicks [Preprint]. Neuroscience. [Google Scholar] [CrossRef]
- Bolhuis, J. J. (1991). Mechanisms of avian imprinting: A review. Biological Reviews, 66, 303–345. [Google Scholar] [CrossRef]
- Bolhuis, J. J., Cook, S., & Horn, G. (2000). Getting better all the time: Improving preference scores reflect increases in the strength of filial imprinting. Animal Behaviour, 59(6), 1153–1159. [Google Scholar] [CrossRef][Green Version]
- Bolhuis, J. J., Johnson, M., Horn, G., & Bateson, P. (1989). Long-lasting effects of IMHV lesions on social preferences in domestic fowl. Behavioral Neuroscience, 103(2), 438–441. [Google Scholar] [CrossRef]
- Bolhuis, J. J., Johnson, M. H., & Horn, G. (1985). Effects of early experience on the development of filial preferences in the domestic chick. Developmental Psychobiology, 18(4), 299–308. [Google Scholar] [CrossRef]
- Bolhuis, J. J., McCabe, B., & Horn, G. (1986). Androgens and imprinting—Differential-effects of testosterone on filial preference in the domestic chick. Behavioral Neuroscience, 100(1), 51–56. [Google Scholar] [CrossRef]
- Bradley, P., Horn, G., & Bateson, P. (1981). Imprinting: An electron microscopic study of chick hyperstriatum ventrale. Experimental Brain Research, 41, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Brown, M., & Horn, G. (1994). Learning-related alterations in the visual responsiveness of neurons in a memory system of the chick brain. European Journal of Neuroscience, 6(9), 1479–1490. [Google Scholar] [CrossRef] [PubMed]
- Carter, C. S., & Marr, J. N. (1970). Olfactory imprinting and age variables in the guinea-pig, Cavia porcellus. Animal Behaviour, 18, 238–244. [Google Scholar] [CrossRef]
- Chantrey, D. F. (1974). Stimulus preexposure and discrimination-learning by domestic chicks—Effect of varying interstimulus time. Journal of Comparative and Physiological Psychology, 87(3), 517–525. [Google Scholar] [CrossRef] [PubMed]
- Chitadze, L., Margvelani, G., Meparishvili, M., Tsverava, L., Mikautadze, E., Kikvidze, M., Lagani, V., McCabe, B. J., & Solomonia, R. (2020). Chicken cognin interactome and the memory of filial imprinting. NeuroReport, 31, 399–405. [Google Scholar] [CrossRef]
- Chitadze, L., Meparishvili, M., Lagani, V., Khuchua, Z., McCabe, B. J., & Solomonia, R. (2024). Src-NADH dehydrogenase subunit 2 complex and recognition memory of imprinting in domestic chicks. PLoS ONE, 19(1), e0297166. [Google Scholar] [CrossRef]
- Chitadze, L., Tevdoradze, E., Kiguradze, T., McCabe, B., & Solomonia, R. (2023). Filial imprinting in domestic chicks; cytoplasmic polyadenylation element binding protein 3, predispositions and learning. NeuroReport, 34(3), 144–149. [Google Scholar] [CrossRef]
- Cipolla-Neto, J., Horn, G., & McCabe, B. J. (1982). Hemispheric asymmetry and imprinting: The effect of sequential lesions to the hyperstriatum ventrale. Experimental Brain Research, 48, 22–27. [Google Scholar] [CrossRef]
- Davies, D. C., Horn, G., & McCabe, B. J. (1985). Noradrenaline and learning—Effects of the noradrenergic neurotoxin DSP4 on imprinting in the domestic chick. Behavioral Neuroscience, 99(4), 652–660. [Google Scholar] [CrossRef] [PubMed]
- Davies, D. C., Johnson, M. H., & Horn, G. (1992). The effect of the neurotoxin DSP4 on the development of a predisposition. Developmental Psychobiology, 25(4), 251–259. [Google Scholar] [CrossRef] [PubMed]
- Derkach, V., Barria, A., & Soderling, T. (1999). Ca2+/calmodulin-kinase II enhances channel conductance of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate type glutamate receptors. Proceedings of the National Academy of Sciences of the United States of America, 96(6), 3269–3274. [Google Scholar] [CrossRef]
- Diekelmann, S., & Born, J. (2010). The memory function of sleep. Nature Reviews Neuroscience, 11(2), 114–126. [Google Scholar] [CrossRef]
- Di Giorgio, E., Loveland, J. L., Mayer, U., Rosa-Salva, O., Versace, E., & Vallortigara, G. (2017). Filial responses as predisposed and learned preferences: Early attachment in chicks and babies. Behavioural Brain Research, 325, 90–104. [Google Scholar] [CrossRef]
- Ferry, A. L., Hespos, S. J., & Gentner, D. (2015). Prelinguistic relational concepts: Investigating analogical processing in infants. Child Development, 86(5), 1386–1405. [Google Scholar] [CrossRef]
- Hebb, D. O. (1949). The organization of behavior: A neuropsychological theory. Wiley. [Google Scholar]
- Honey, R. C., & Bateson, P. (1996). Stimulus comparison and perceptual learning: Further evidence and evaluation from an imprinting procedure. Quarterly Journal of Experimental Psychology Section B-Comparative and Physiological Psychology, 49(3), 259–269. [Google Scholar]
- Honey, R. C., Bateson, P., & Horn, G. (1994). The role of stimulus comparison in perceptual-learning—An investigation with the domestic chick. Quarterly Journal of Experimental Psychology Section B-Comparative and Physiological Psychology, 47, 83–103. [Google Scholar]
- Honey, R. C., Horn, G., & Bateson, P. (1993). Perceptual-learning during filial imprinting—Evidence from transfer of training studies. Quarterly Journal of Experimental Psychology Section B-Comparative and Physiological Psychology, 46(3), 253–269. [Google Scholar] [CrossRef]
- Honey, R. C., Horn, G., Bateson, P., & Walpole, M. (1995). Functionally distinct memories for imprinting stimuli—Behavioral and neural dissociations. Behavioral Neuroscience, 109, 689–698. [Google Scholar] [CrossRef]
- Horn, G. (1985). Memory, imprinting, and the brain. Oxford University Press. Available online: http://www.oxfordscholarship.com/view/10.1093/acprof:oso/9780198521563.001.0001/acprof-9780198521563 (accessed on 31 July 2014).
- Horn, G. (1998). Visual imprinting and the neural mechanisms of recognition memory. Trends in Neurosciences, 21(7), 300–305. [Google Scholar] [CrossRef] [PubMed]
- Horn, G. (2004). Pathways of the past: The imprint of memory. Nature Reviews Neuroscience, 5, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Horn, G., Bradley, P., & McCabe, B. (1985). Changes in the structure of synapses associated with learning. Journal of Neuroscience, 5(12), 3161–3168. [Google Scholar] [CrossRef] [PubMed]
- Horn, G., & McCabe, B. J. (1984). Predispositions and preferences—Effects on imprinting of lesions to the chick brain. Animal Behaviour, 32(FEB), 288–292. [Google Scholar] [CrossRef]
- Horn, G., McCabe, B. J., & Bateson, P. (1979). Autoradiographic study of the chick brain after imprinting. Brain Research, 168(2), 361–373. [Google Scholar] [CrossRef]
- Horn, G., McCabe, B. J., & Cipolla-Neto, J. (1983). Imprinting in the domestic chick: The role of each side of the hyperstriatum ventrale in acquisition and retention. Experimental Brain Research, 53(1), 91–98. [Google Scholar] [CrossRef] [PubMed]
- Horn, G., Nicol, A. U., & Brown, M. W. (2001). Tracking memory’s trace. Proceedings of the National Academy of Sciences of the United States of America, 98(9), 5282–5287. [Google Scholar] [CrossRef]
- Hu, A., Wang, Z., Lan, D., Fu, Y., Zhu, Y., Dong, Y., & Zheng, P. (2007). Inhibition of evoked glutamate release by neurosteroid allopregnanolone via inhibition of L-type calcium channels in rat medial prefrontal cortex. Neuropsychopharmacology, 32(7), 1477–1489. [Google Scholar] [CrossRef][Green Version]
- Jackson, C., McCabe, B. J., Nicol, A. U., Grout, A. S., Brown, M. W., & Horn, G. (2008). Dynamics of a memory trace: Effects of sleep on consolidation. Current Biology, 18(6), 393–400. [Google Scholar] [CrossRef]
- Jackson, P. S., & Bateson, P. P. G. (1974). Imprinting and exploration of slight novelty in chicks. Nature, 251(5476), 609–610. [Google Scholar] [CrossRef]
- Janus, C. (1993). Stability of preference for odors after short-term exposure in young spiny mice. Developmental Psychobiology, 26(1), 65–79. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M., Bolhuis, J., & Horn, G. (1985). Interaction between acquired preferences and developing predispositions during imprinting. Animal Behaviour, 33(AUG), 1000–1006. [Google Scholar] [CrossRef]
- Johnson, M., Davies, D., & Horn, G. (1989). A sensitive period for the development of a predisposition in dark-reared chicks. Animal Behaviour, 37, 1044–1046. [Google Scholar] [CrossRef]
- Johnson, M., Dziurawiec, S., Ellis, H., & Morton, J. (1991). Newborns preferential tracking of face-like stimuli and its subsequent decline. Cognition, 40(1–2), 1–19. [Google Scholar] [CrossRef]
- Johnson, M., & Horn, G. (1987). The role of a restricted region of the chick forebrain in the recognition of individual conspecifics. Behavioural Brain Research, 23(3), 269–275. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M., & Horn, G. (1988). Development of filial preferences in dark-reared chicks. Animal Behaviour, 36, 675–683. [Google Scholar] [CrossRef]
- Johnson, M., Senju, A., & Tomalski, P. (2015). The two-process theory of face processing: Modifications based on two decades of data from infants and adults. Neuroscience and Biobehavioral Reviews, 50, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Katugampola, N., Pinfield, N. C. R., Reeve, R. J., Ridgeway, M., & McCabe, B. J. (2004). Imprinting: Susceptibility to disruption by novel stimuli. In Proceedings, 7th International Congress of Neuroethology. International Society for Neuroethology. [Google Scholar]
- Klin, A., Shultz, S., & Jones, W. (2015). Social visual engagement in infants and toddlers with autism: Early developmental transitions and a model of pathogenesis. Neuroscience & Biobehavioral Reviews, 50, 189–203. [Google Scholar] [CrossRef]
- Kristensen, A., Jenkins, M., Banke, T., Schousboe, A., Makino, Y., Johnson, R., Huganir, R., & Traynelis, S. (2011). Mechanism of Ca2+/calmodulin-dependent kinase II regulation of AMPA receptor gating. Nature Neuroscience, 14(6), 727-U327. [Google Scholar] [CrossRef]
- Lagani, V., Chitadze, L., Gonzalez Alvarez, A. C., Bokuchava, T., Sansone, G., Bokuchava, V., Tsverava, L., Jones, A., Lisgo, S., Martinez De Morentin, X., Lehmann, R., Tegner, J., Ibrahim, L. A., McCabe, B. J., Khuchua, Z., Cabrero, D. G., & Solomonia, R. (2025). Charting the single cell transcriptional landscape governing visual imprinting. bioRxiv, 2025.06.23.660422. [Google Scholar] [CrossRef]
- Lemaire, B. S., Rucco, D., Josserand, M., Vallortigara, G., & Versace, E. (2021). Stability and individual variability of social attachment in imprinting. Scientific Reports, 11(1), 7914. [Google Scholar] [CrossRef]
- Lutz, N. D., Harkotte, M., & Born, J. (2026). Sleep’s contribution to memory formation. Physiological Reviews, 106(1), 363–483. [Google Scholar] [CrossRef]
- Margvelani, G., Meparishvili, M., Kiguradze, T., McCabe, B. J., & Solomonia, R. (2018a). Micro-RNAs, their target proteins, predispositions and the memory of filial imprinting. Scientific Reports, 8(1). [Google Scholar] [CrossRef] [PubMed]
- Margvelani, G., Meparishvili, M., Tevdoradze, E., McCabe, B. J., & Solomonia, R. (2018b). Mitochondrial fusion and fission proteins and the recognition memory of imprinting in domestic chicks. NeuroReport, 29(2), 128–133. [Google Scholar] [CrossRef]
- Martinho, A., & Kacelnik, A. (2016). Ducklings imprint on the relational concept of “same or different”. Science, 353(6296), 286–288. [Google Scholar] [CrossRef]
- Matsushima, T., Izumi, T., & Vallortigara, G. (2024). The domestic chick as an animal model of autism spectrum disorder: Building adaptive social perceptions through prenatally formed predispositions. Frontiers in Neuroscience, 18, 1279947. [Google Scholar] [CrossRef]
- McCabe, B. J. (2019). Visual imprinting in birds: Behavior, models, and neural mechanisms. Frontiers in Physiology, 10, 658. [Google Scholar] [CrossRef]
- McCabe, B. J., Davey, J. E., & Horn, G. (1992). Impairment of learning by localized injection of an N-methyl-D-aspartate receptor antagonist into the hyperstriatum-ventrale of the domestic chick. Behavioral Neuroscience, 106(6), 947–953. [Google Scholar] [CrossRef] [PubMed]
- McCabe, B. J., & Horn, G. (1988). Learning and memory: Regional changes in N-methyl-D-aspartate receptors in the chick brain after imprinting. Proceedings of the National Academy of Sciences of the United States of America, 85, 2849–2853. [Google Scholar] [CrossRef] [PubMed]
- McCabe, B. J., & Horn, G. (1991). Synaptic transmission and recognition memory—Time course of changes in N-methyl-D-aspartate receptors after imprinting. Behavioral Neuroscience, 105(2), 289–294. [Google Scholar] [CrossRef]
- McCabe, B. J., & Horn, G. (1994). Learning-related changes in Fos-like immunoreactivity in the chick forebrain after imprinting. Proceedings of the National Academy of Sciences of the United States of America, 91, 11417–11421. [Google Scholar] [CrossRef] [PubMed]
- McCabe, B. J., Horn, G., & Bateson, P. P. G. (1979). Effects of rhythmic hyperstriatal stimulation on chicks’ preferences for visual flicker. Physiology & Behavior, 23(1), 137–140. [Google Scholar] [CrossRef]
- McCabe, B. J., Horn, G., & Bateson, P. P. G. (1981). Effects of restricted lesions of the chick forebrain on the acquisition of filial preferences during imprinting. Brain Research, 205(1), 29–37. [Google Scholar] [CrossRef]
- McCabe, B. J., Kendrick, K. M., & Horn, G. (2001). Gamma-aminobutyric acid, taurine and learning: Release of amino acids from slices of chick brain following filial imprinting. Neuroscience, 105(2), 317–324. [Google Scholar] [CrossRef]
- Meparishvili, M., Chitadze, L., Lagani, V., McCabe, B. J., & Solomonia, R. O. (2021). Src and memory: A study of filial imprinting and predispositions in the domestic chick. Frontiers in Physiology, 12, 1517. [Google Scholar] [CrossRef]
- Meparishvili, M., Nozadze, M., Margvelani, G., McCabe, B. J., & Solomonia, R. O. (2015). A proteomic study of memory after imprinting in the domestic chick. Frontiers in Behavioral Neuroscience, 9, 319. [Google Scholar] [CrossRef]
- Meredith, R. M., McCabe, B. J., Kendrick, K. M., & Horn, G. (2004). Amino acid neurotransmitter release and learning: A study of visual imprinting. Neuroscience, 126, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Montuori, L. M., & Honey, R. C. (2015). Representation in development: From a model system to some general processes. Brain, Memory and Development: The Imprint of Gabriel Horn, 50, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Moorman, S., & Nicol, A. U. (2015). Memory-related brain lateralisation in birds and humans. Neuroscience and Biobehavioral Reviews, 50, 86–102. [Google Scholar] [CrossRef] [PubMed]
- Morton, J., & Johnson, M. H. (1991). CONSPEC and CONLERN: A two-process theory of infant face recognition. Psychological Review, 98(2), 164–181. [Google Scholar] [CrossRef]
- Nicol, A. U., Brown, M. W., & Horn, G. (1995). Neurophysiological investigations of a recognition memory system for imprinting in the domestic chick. European Journal of Neuroscience, 7(4), 766–776. [Google Scholar] [CrossRef]
- Nicol, A. U., & McCabe, B. (2014). Transcranial stimulation promotes consolidation of imprinted memory in domestic chicks (Gallus gallus). Physiology, 31, C24. [Google Scholar]
- Nicol, A. U., Oakeshott, S. M., Brown, M. W., & Horn, G. (2003). Sixth IBRO world Cong (p. 4146). Neuroscience Abstracts. [Google Scholar]
- Parsons, C. H., & Rogers, L. J. (1997). Pharmacological extension of the sensitive period for imprinting in gallus domesticus. Physiology & Behavior, 62(6), 1303–1310. [Google Scholar] [CrossRef]
- Parsons, C. H., & Rogers, L. J. (2000). NMDA receptor antagonists extend the sensitive period for imprinting. Physiology & Behavior, 68(5), 749–753. [Google Scholar] [CrossRef]
- Porter, R., & Etscorn, F. (1974). Olfactory imprinting resulting from brief exposure in acomys-cahirinus. Nature, 250(5469), 732–733. [Google Scholar] [CrossRef]
- Reiner, A., Perkel, D. J., Bruce, L. L., Butler, A. B., Csillag, A., Kuenzel, W., Medina, L., Paxinos, G., Shimizu, T., Striedter, G., Wild, M., Ball, G. F., Durand, S., Gunturkun, O., Lee, D. W., Mello, C. V., Powers, A., White, S. A., Hough, G., … Jarvis, E. D. (2004). Revised nomenclature for avian telencephalon and some related brainstem nuclei. Journal of Comparative Neurology, 473(3), 377–414. [Google Scholar] [CrossRef]
- Rigato, S., Stets, M., Dvergsdal, H., & Holmboe, K. (2025). Infant neural processing of mother’s face is associated with falling reactivity in the first year of life. Developmental Cognitive Neuroscience, 71, 101502. [Google Scholar] [CrossRef] [PubMed]
- Robledo, J.-P., Cross, I., Boada-Bayona, L., & Demogeot, N. (2022). Back to basics: A re-evaluation of the relevance of imprinting in the genesis of Bowlby’s attachment theory. Frontiers in Psychology, 13, 1033746. [Google Scholar] [CrossRef]
- Rosa-Salva, O., Mayer, U., & Vallortigara, G. (2019). Unlearned visual preferences for the head region in domestic chicks. PLoS ONE, 14(9), e0222079. [Google Scholar] [CrossRef]
- Rosa-Salva, O., Regolin, L., & Vallortigara, G. (2009). Faces are special for newly hatched chicks: Evidence for inborn domain-specific mechanisms underlying spontaneous preferences for face-like stimuli: Newly hatched chicks’ preferences for faces. Developmental Science, 13(4), 565–577. [Google Scholar] [CrossRef]
- Salzen, E., & Meyer, C. (1968). Reversibility of imprinting. Journal of Comparative and Physiological Psychology, 66(2), 269–275. [Google Scholar] [CrossRef]
- Sluckin, W. (1972). Imprinting and early learning (2nd ed.). Methuen. [Google Scholar]
- Smith, F. V., & Bird, M. W. (1963). Group factors in the response of the domestic chick to a distant visual stimulus. Animal Behaviour, 11(2), 397–399. [Google Scholar] [CrossRef]
- Solomonia, R. O., Apkhazava, D., Nozadze, M., Jackson, A. P., McCabe, B. J., & Horn, G. (2008). Different forms of MARCKS protein are involved in memory formation in the learning process of imprinting. Experimental Brain Research, 188, 323–330. [Google Scholar] [CrossRef]
- Solomonia, R. O., Kiguradze, T., McCabe, B. J., & Horn, G. (2000). Neural cell adhesion molecules, CaM kinase II and long-term memory in the chick. NeuroReport, 11(14), 3139–3143. [Google Scholar] [CrossRef] [PubMed]
- Solomonia, R. O., Kotorashvili, A., Kiguradze, T., McCabe, B. J., & Horn, G. (2005). Ca2+/calmodulin protein kinase II and memory: Learning-related changes in a localized region of the domestic chick brain. Journal of Physiology-London, 569(2), 643–653. [Google Scholar] [CrossRef] [PubMed]
- Solomonia, R. O., Kunelauri, N., Mikautadze, E., Apkhazava, D., McCabe, B. J., & Horn, G. (2011). Mitochondrial proteins, learning and memory: Biochemical specialization of a memory system. Neuroscience, 194, 112–123. [Google Scholar] [CrossRef]
- Solomonia, R. O., & McCabe, B. J. (2015). Molecular mechanisms of memory in imprinting. Neuroscience & Biobehavioral Reviews, 50, 56–69. [Google Scholar] [CrossRef]
- Solomonia, R. O., McCabe, B. J., & Horn, G. (1998). Neural cell adhesion molecules, learning and memory. Behavioral Neuroscience, 112, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Solomonia, R. O., McCabe, B. J., Jackson, A. P., & Horn, G. (1997). Clathrin proteins and recognition memory. Neuroscience, 80, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Solomonia, R. O., Meparishvili, M., Mikautadze, E., Kunelauri, N., Apkhazava, D., & McCabe, B. J. (2013). AMPA receptor phosphorylation and recognition memory: Learning-related, time-dependent changes in the chick brain following filial imprinting. Experimental Brain Research, 226(2), 297–308. [Google Scholar] [CrossRef]
- Solomonia, R. O., Morgan, K., Kotorashvili, A., McCabe, B. J., Jackson, A. P., & Horn, G. (2003). Analysis of differential gene expression supports a role for amyloid precursor protein and a protein kinase C substrate (MARCKS) in long-term memory. European Journal of Neuroscience, 17(5), 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Spalding, D. A. (1873). Instinct; with original observations on young animals. Macmillan’s Magazine, 27, 282–293. [Google Scholar]
- Suge, R., Kato, H., & McCabe, B. J. (2010). Rapid induction of the immediate early gene c-fos in a chick forebrain system involved in memory. Experimental Brain Research, 200(2), 183–188. [Google Scholar] [CrossRef]
- Suge, R., & McCabe, B. J. (2004). Early stages of memory formation in filial imprinting: Fos-like immunoreactivity and behavior in the domestic chick. Neuroscience, 123(4), 847–856. [Google Scholar] [CrossRef]
- Tiunova, A., Komissarova, N., & Anokhin, K. (2019). Mapping the neural substrates of recent and remote visual imprinting memory in the chick brain. Frontiers in Physiology, 10, 351. [Google Scholar] [CrossRef]
- Town, S. M. (2011a). Preliminary evidence of a neurophysiological basis for individual discrimination in filial imprinting. Behavioural Brain Research, 225(2), 651–654. [Google Scholar] [CrossRef] [PubMed]
- Town, S. M. (2011b). The effects of social rearing on preferences formed during filial imprinting and their neural correlates. Experimental Brain Research, 212(4), 575–581. [Google Scholar] [CrossRef] [PubMed]
- Vallortigara, G. (2021). Born knowing: Imprinting and the origins of knowledge. The MIT Press. [Google Scholar]
- Vallortigara, G., Regolin, L., & Marconato, F. (2005). Visually inexperienced chicks exhibit spontaneous preference for biological motion patterns. PLoS Biology, 3(7), e208. [Google Scholar] [CrossRef]
- Wu, J.-Y., & Prentice, H. (2010). Role of taurine in the central nervous system. Journal of Biomedical Science, 17(1), S1. [Google Scholar] [CrossRef]
- Yamaguchi, S., Aoki, N., Kitajima, T., Iikubo, E., Katagiri, S., Matsushima, T., & Homma, K. J. (2012). Thyroid hormone determines the start of the sensitive period of imprinting and primes later learning. Nature Communications, 3, 1081. [Google Scholar] [CrossRef] [PubMed]




| Molecule | Ascribed to Learning During Training or to Predisposition to Learn Well | Side of IMM | Reference |
|---|---|---|---|
| Clathrin heavy chain protein | Learning | Left | Solomonia et al. (1997) |
| Neural cell adhesion molecule proteins | Learning | Left | Solomonia et al. (1998) |
| MARCKS protein, Amyloid precursor protein | Learning | Left | Solomonia et al. (2003) |
| Membrane-bound (unphosphorylated) MARCKS protein | Learning | Data from left and right IMM were pooled in this analysis | Solomonia et al. (2008) |
| Cytochrome c oxidase protein subunits I and II, α-fodrin protein | Learning | Left | Solomonia et al. (2011) |
| Membrane-bound cognin protein, Protein resembling P32 subunit of splicing factor SF2 (membrane fraction), Dynamin-1 protein, P38 protein in membrane-mitochondrial fraction, Voltage-dependent anionic channel 1 protein, heterogeneous nuclear ribonucleoprotein A2/B1, Mitochondrial transcription factor A protein, Nuclear respiratory factor 1 protein | Learning | Left | Meparishvili et al. (2015) |
| Mitofusin-1 protein, Dynamin-related protein-1 | Learning | Left | Margvelani et al. (2018b) |
| Micro-RNA gga-miR-130b-3p, Membrane-associated cytoplasmic polyadenylation element binding protein 3 (CPEB-3) | Predisposition | Left | Margvelani et al. (2018a) |
| Mitochondrial ATP synthase (b5 subunit) protein, Na/K ATPase protein (α2 subunit) | Learning | Left | Chitadze et al. (2020) |
| Src tyrosine kinase protein, Tyrosine 527-phosphorylated Src (inhibited form) | Learning | Left | Meparishvili et al. (2021) |
| Tyrosine 416-phosphorylated Src (activated form) as % of total Src | Predisposition (negative correlation) | Left | “ |
| CPEB-3 protein, aggregated form | Predisposition | Left | Chitadze et al. (2023) |
| Src-NADH2 complex | Learning (negative correlation) | Right | Chitadze et al. (2024) |
| Long non-coding RNA ENSGALG00010007489, protein LUC7L, protein Retinoid-related orphan receptor-513-α (RORA), Forkhead box protein P2 (FOXP2) | Learning | Left | Lagani et al. (2025) |
| Long-non-coding RNA ENSGALG00010026609, Roundabout guidance receptor 1 (ROBO-1) | Predisposition | Left | “ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
McCabe, B.J. Filial Imprinting: Behaviour and Neurobiology. Behav. Sci. 2026, 16, 741. https://doi.org/10.3390/bs16050741
McCabe BJ. Filial Imprinting: Behaviour and Neurobiology. Behavioral Sciences. 2026; 16(5):741. https://doi.org/10.3390/bs16050741
Chicago/Turabian StyleMcCabe, Brian J. 2026. "Filial Imprinting: Behaviour and Neurobiology" Behavioral Sciences 16, no. 5: 741. https://doi.org/10.3390/bs16050741
APA StyleMcCabe, B. J. (2026). Filial Imprinting: Behaviour and Neurobiology. Behavioral Sciences, 16(5), 741. https://doi.org/10.3390/bs16050741






