The Relationship between Executive Functions and Body Weight: Sex as a Moderating Variable
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Materials and Procedure
2.3. Statistical Analyses
3. Results
3.1. Preliminary Data Analysis: Normality Assumptions and Missing Data
3.2. Descriptive Statistics
3.3. Moderation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Polito, R.; Scarinci, A.; Ambrosi, A.; Tartaglia, N.; Tafuri, D.; Monda, M.; Messina, A.; Cimmino, F.; Catapano, A.; Sessa, F.; et al. The Beneficial Effects of Physical Activity and Weight Loss on Human Colorectal Carcinoma Cell Lines. J. Hum. Sport Exerc. 2020, 15, S252–S260. [Google Scholar] [CrossRef]
- The GBD 2015 Obesity Collaborators Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [CrossRef]
- Sarısoy, G.; Kaçar, Ö.F.; Pazvantoğlu, O.; Öztürk, A.; Korkmaz, I.Z.; Kocamanoğlu, B.; Böke, Ö.; Sahin, A.R. Temperament and Character Traits in Patients with Bipolar Disorder and Associations with Attempted Suicide. Compr. Psychiatry 2012, 53, 1096–1102. [Google Scholar] [CrossRef]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. PharmacoEconomics 2015, 33, 673–689. [Google Scholar] [CrossRef]
- Abdelaal, M.; le Roux, C.W.; Docherty, N.G. Morbidity and Mortality Associated with Obesity. Ann. Transl. Med. 2017, 5, 161. [Google Scholar] [CrossRef]
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation. World Health Organ. Tech. Rep. Ser. 2000, 894, 1–253.
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, Regional, and National Prevalence of Overweight and Obesity in Children and Adults during 1980–2013: A Systematic Analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Zhang, H.; Wu, N.; Liu, Y.; Han, P.; Wang, F.; Wang, J.; Xie, F.; Niu, S.; Hu, H.; et al. Relationship between Obesity-Related Anthropometric Indicators and Cognitive Function in Chinese Suburb-Dwelling Older Adults. PLoS ONE 2021, 16, e0258922. [Google Scholar] [CrossRef]
- Gustafson, D.; Rothenberg, E.; Blennow, K.; Steen, B.; Skoog, I. An 18-Year Follow-up of Overweight and Risk of Alzheimer Disease. Arch. Intern. Med. 2003, 163, 1524. [Google Scholar] [CrossRef] [PubMed]
- Rosengren, A. Body Mass Index, Other Cardiovascular Risk Factors, and Hospitalization for Dementia. Arch. Intern. Med. 2005, 165, 321. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; D’Introno, A.; Colacicco, A.M.; Capurso, C.; Pichichero, G.; Capurso, S.A.; Capurso, A.; Solfrizzi, V. Lipid Metabolism in Cognitive Decline and Dementia. Brain Res. Rev. 2006, 51, 275–292. [Google Scholar] [CrossRef]
- Whitmer, R.A.; Gustafson, D.R.; Barrett-Connor, E.; Haan, M.N.; Gunderson, E.P.; Yaffe, K. Central Obesity and Increased Risk of Dementia More than Three Decades Later. Neurology 2008, 71, 1057–1064. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Beydoun, H.A.; Wang, Y. Obesity and Central Obesity as Risk Factors for Incident Dementia and Its Subtypes: A Systematic Review and Meta-Analysis. Obes. Rev. 2008, 9, 204–218. [Google Scholar] [CrossRef]
- Whitmer, R.A.; Gunderson, E.P.; Barrett-Connor, E.; Quesenberry, C.P.; Yaffe, K. Obesity in Middle Age and Future Risk of Dementia: A 27 Year Longitudinal Population Based Study. BMJ 2005, 330, 1360. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.A.; Spencer, S.J. Obesity and Neuroinflammation: A Pathway to Cognitive Impairment. Brain Behav. Immun. 2014, 42, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Bischof, G.N.; Park, D.C. Obesity and Aging: Consequences for Cognition, Brain Structure, and Brain Function. Psychosom. Med. 2015, 77, 697–709. [Google Scholar] [CrossRef]
- Dye, L.; Boyle, N.B.; Champ, C.; Lawton, C. The Relationship between Obesity and Cognitive Health and Decline. Proc. Nutr. Soc. 2017, 76, 443–454. [Google Scholar] [CrossRef]
- Leigh, S.-J.; Morris, M.J. Diet, Inflammation and the Gut Microbiome: Mechanisms for Obesity-Associated Cognitive Impairment. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2020, 1866, 165767. [Google Scholar] [CrossRef]
- Chieffi, S.; Castaldi, C.; Di Maio, G.; La Marra, M.; Messina, A.; Monda, V.; Villano, I. Attentional Bias in the Radial and Vertical Dimensions of Space. Comptes Rendus Biol. 2019, 342, 97–100. [Google Scholar] [CrossRef]
- Ardila, A. On the Evolutionary Origins of Executive Functions. Brain Cogn. 2008, 68, 92–99. [Google Scholar] [CrossRef]
- Burgess, P.W.; Simons, J.S. Theories of Frontal Lobe Executive Function: Clinical Applications. In The Effectiveness of Rehabilitation for Cognitive Deficits; Halligan, P.W., Wade, D.T., Eds.; Oxford University Press: Oxford, UK, 2005; pp. 211–231. ISBN 978-0-19-852654-4. [Google Scholar]
- Gilbert, S.J.; Burgess, P.W. Executive Function. Curr. Biol. 2008, 18, R110–R114. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive Functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Rodrigue, C.; Ouellette, A.-S.; Lemieux, S.; Tchernof, A.; Biertho, L.; Bégin, C. Executive Functioning and Psychological Symptoms in Food Addiction: A Study among Individuals with Severe Obesity. Eat. Weight. Disord. 2018, 23, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Sims, R.C.; Bennett, N.-K.; Mwendwa, D.T.; Ali, M.K.; Levy, S.-A.T.; Callender, C.O.; Campbell, A.L. Executive Function and Negative Eating Behaviors in Severely Obese African Americans. Ethn. Dis. 2014, 24, 328–334. [Google Scholar] [PubMed]
- Riggs, N.; Chou, C.-P.; Spruijt-Metz, D.; Pentz, M.A. Executive Cognitive Function as a Correlate and Predictor of Child Food Intake and Physical Activity. Child Neuropsychol. 2010, 16, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Powell, D.J.H.; McMinn, D.; Allan, J.L. Does Real Time Variability in Inhibitory Control Drive Snacking Behavior? An Intensive Longitudinal Study. Health Psychol. 2017, 36, 356–364. [Google Scholar] [CrossRef]
- Yang, Y.; Shields, G.S.; Guo, C.; Liu, Y. Executive Function Performance in Obesity and Overweight Individuals: A Meta-Analysis and Review. Neurosci. Biobehav. Rev. 2018, 84, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Dassen, F.C.M.; Houben, K.; Allom, V.; Jansen, A. Self-Regulation and Obesity: The Role of Executive Function and Delay Discounting in the Prediction of Weight Loss. J. Behav. Med. 2018, 41, 806–818. [Google Scholar] [CrossRef]
- Polito, R.; Francavilla, V.C.; Ambrosi, A.; Tartaglia, N.; Tafuri, D.; Monda, M.; Messina, A.; Sessa, F.; Di Maio, G.; Ametta, A.; et al. The Orexin-A Serum Levels Are Strongly Modulated by Physical Activity Intervention in Diabetes Mellitus Patients. J. Hum. Sport Exerc. 2020, 15, 244–251. [Google Scholar]
- Ribeiro, O.; Carmo, I.D.; Paiva, T.; Figueira, M.L. Neuropsychological Profile, Cognitive Reserve and Emotional Distress in a Portuguese Sample of Severely Obese Patients. Acta Med. Port. 2020, 33, 38. [Google Scholar] [CrossRef]
- Gluck, M.E.; Viswanath, P.; Stinson, E.J. Obesity, Appetite, and the Prefrontal Cortex. Curr. Obes. Rep. 2017, 6, 380–388. [Google Scholar] [CrossRef]
- Horstmann, A.; Busse, F.P.; Mathar, D.; Müller, K.; Lepsien, J.; Schlögl, H.; Kabisch, S.; Kratzsch, J.; Neumann, J.; Stumvoll, M.; et al. Obesity-Related Differences between Women and Men in Brain Structure and Goal-Directed Behavior. Front. Hum. Neurosci. 2011, 5, 58. [Google Scholar] [CrossRef]
- Alarcón, G.; Ray, S.; Nagel, B.J. Lower Working Memory Performance in Overweight and Obese Adolescents Is Mediated by White Matter Microstructure. J. Int. Neuropsychol. Soc. 2016, 22, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Balleine, B.W.; Delgado, M.R.; Hikosaka, O. The Role of the Dorsal Striatum in Reward and Decision-Making: Figure 1. J. Neurosci. 2007, 27, 8161–8165. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G.E.; Crutcher, M.D. Functional Architecture of Basal Ganglia Circuits: Neural Substrates of Parallel Processing. Trends Neurosci. 1990, 13, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.T.; Moujaes, F.; Schleifer, C.H.; Starc, M.; Ji, J.L.; Santamauro, N.; Adkinson, B.; Kolobaric, A.; Flynn, M.; Krystal, J.H.; et al. Reward and Loss Incentives Improve Spatial Working Memory by Shaping Trial-by-Trial Posterior Frontoparietal Signals. NeuroImage 2022, 254, 119139. [Google Scholar] [CrossRef]
- Hassenstab, J.J.; Sweet, L.H.; Del Parigi, A.; McCaffery, J.M.; Haley, A.P.; Demos, K.E.; Cohen, R.A.; Wing, R.R. Cortical Thickness of the Cognitive Control Network in Obesity and Successful Weight Loss Maintenance: A Preliminary MRI Study. Psychiatry Res. Neuroimaging 2012, 202, 77–79. [Google Scholar] [CrossRef]
- Pannacciulli, N.; Del Parigi, A.; Chen, K.; Le, D.S.N.T.; Reiman, E.M.; Tataranni, P.A. Brain Abnormalities in Human Obesity: A Voxel-Based Morphometric Study. NeuroImage 2006, 31, 1419–1425. [Google Scholar] [CrossRef]
- Shott, M.E.; Cornier, M.-A.; Mittal, V.A.; Pryor, T.L.; Orr, J.M.; Brown, M.S.; Frank, G.K.W. Orbitofrontal Cortex Volume and Brain Reward Response in Obesity. Int. J. Obes. 2015, 39, 214–221. [Google Scholar] [CrossRef]
- Kishinevsky, F.I.; Cox, J.E.; Murdaugh, D.L.; Stoeckel, L.E.; Cook, E.W.; Weller, R.E. fMRI Reactivity on a Delay Discounting Task Predicts Weight Gain in Obese Women. Appetite 2012, 58, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Artham, S.M.; Lavie, C.J.; Milani, R.V.; Ventura, H.O. The Obesity Paradox: Impact of Obesity on the Prevalence and Prognosis of Cardiovascular Diseases. Postgrad. Med. 2008, 120, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Childers, D.K.; Allison, D.B. The ‘Obesity Paradox’: A Parsimonious Explanation for Relations among Obesity, Mortality Rate and Aging? Int. J. Obes. 2010, 34, 1231–1238. [Google Scholar] [CrossRef]
- Flegal, K.M.; Kit, B.K.; Orpana, H.; Graubard, B.I. Association of All-Cause Mortality With Overweight and Obesity Using Standard Body Mass Index Categories: A Systematic Review and Meta-Analysis. JAMA 2013, 309, 71. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, S.; Gilbert, S.; Serpell, L. Systematic Review: Are Overweight and Obese Individuals Impaired on Behavioural Tasks of Executive Functioning? Neuropsychol. Rev. 2013, 23, 138–156. [Google Scholar] [CrossRef]
- Boeka, A.; Lokken, K. Neuropsychological Performance of a Clinical Sample of Extremely Obese Individuals. Arch. Clin. Neuropsychol. 2008, 23, 467–474. [Google Scholar] [CrossRef]
- Gunstad, J.; Lhotsky, A.; Wendell, C.R.; Ferrucci, L.; Zonderman, A.B. Longitudinal Examination of Obesity and Cognitive Function: Results from the Baltimore Longitudinal Study of Aging. Neuroepidemiology 2010, 34, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Hartanto, A.; Yong, J.; Toh, W. Bidirectional Associations between Obesity and Cognitive Function in Midlife Adults: A Longitudinal Study. Nutrients 2019, 11, 2343. [Google Scholar] [CrossRef]
- Skinner, J.S.; Abel, W.M.; McCoy, K.; Wilkins, C.H. Exploring the “Obesity Paradox” as a Correlate of Cognitive and Physical Function in Community-Dwelling Black and White Older Adults. Ethn. Dis. 2017, 27, 387. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Telang, F. Overlapping Neuronal Circuits in Addiction and Obesity: Evidence of Systems Pathology. Phil. Trans. R. Soc. B 2008, 363, 3191–3200. [Google Scholar] [CrossRef]
- Monica, D.; Paulo, M.; Appolinário, J.C.; de Freitas, S.R.; Coutinho, G.; Santos, C.; Coutinho, W. Assessment of Executive Functions in Obese Individuals with Binge Eating Disorder. Rev. Bras. Psiquiatr. 2010, 32, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Favieri, F.; Forte, G.; Casagrande, M. The Executive Functions in Overweight and Obesity: A Systematic Review of Neuropsychological Cross-Sectional and Longitudinal Studies. Front. Psychol. 2019, 10, 2126. [Google Scholar] [CrossRef] [PubMed]
- La Marra, M.; Ilardi, C.R.; Villano, I.; Carosella, M.; Staiano, M.; Iavarone, A.; Chieffi, S.; Messina, G.; Polito, R.; Scarinci, A.; et al. Functional Relationship between Inhibitory Control, Cognitive Flexibility, Psychomotor Speed and Obesity. Brain Sci. 2022, 12, 1080. [Google Scholar] [CrossRef] [PubMed]
- La Marra, M.; Villano, I.; Ilardi, C.R.; Carosella, M.; Staiano, M.; Iavarone, A.; Chieffi, S.; Messina, G.; Polito, R.; Porro, C.; et al. Executive Functions in Overweight and Obese Treatment-Seeking Patients: Cross-Sectional Data and Longitudinal Perspectives. Brain Sci. 2022, 12, 777. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.; McClain, J.E.; Truesdale, K.P. Selection of Measures in Epidemiologic Studies of the Consequences of Obesity. Int. J. Obes. 2008, 32, S60–S66. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Visser, M.; Sepulveda, D.; Pierson, R.N.; Harris, T.; Heymsfield, S.B. How Useful Is Body Mass Index for Comparison of Body Fatness across Age, Sex, and Ethnic Groups? Am. J. Epidemiol. 1996, 143, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J. BMI-Related Errors in the Measurement of Obesity. Int. J. Obes. 2008, 32, S56–S59. [Google Scholar] [CrossRef] [PubMed]
- Alves Junior, C.A.; Mocellin, M.C.; Gonçalves, E.C.A.; Silva, D.A.; Trindade, E.B. Anthropometric Indicators as Body Fat Discriminators in Children and Adolescents: A Systematic Review and Meta-Analysis. Adv. Nutr. 2017, 8, 718–727. [Google Scholar] [CrossRef]
- Villano, I.; La Marra, M.; Messina, A.; Di Maio, G.; Moscatelli, F.; Chieffi, S.; Monda, M.; Messina, G.; Monda, V. Effects of Vegetarian and Vegan Nutrition on Body Composition in Competitive Futsal Athletes. Prog. Nutr. 2021, 23, e2021126. [Google Scholar] [CrossRef]
- Zhu, S.; Heymsfield, S.B.; Toyoshima, H.; Wang, Z.; Pietrobelli, A.; Heshka, S. Race-Ethnicity–Specific Waist Circumference Cutoffs for Identifying Cardiovascular Disease Risk Factors. Am. J. Clin. Nutr. 2005, 81, 409–415. [Google Scholar] [CrossRef]
- Janssen, I.; Katzmarzyk, P.T.; Ross, R. Waist Circumference and Not Body Mass Index Explains Obesity-Related Health Risk. Am. J. Clin. Nutr. 2004, 79, 379–384. [Google Scholar] [CrossRef]
- Price, G.M.; Uauy, R.; Breeze, E.; Bulpitt, C.J.; Fletcher, A.E. Weight, Shape, and Mortality Risk in Older Persons: Elevated Waist-Hip Ratio, Not High Body Mass Index, Is Associated with a Greater Risk of Death. Am. J. Clin. Nutr. 2006, 84, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Stival, C.; Lugo, A.; Odone, A.; van den Brandt, P.A.; Fernandez, E.; Tigova, O.; Soriano, J.B.; José López, M.; Scaglioni, S.; Gallus, S.; et al. Prevalence and Correlates of Overweight and Obesity in 12 European Countries in 2017–2018. Obes. Facts 2022, 15, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, B.J. Obesity Prevalence Among U.S. Adults During the COVID-19 Pandemic. Am. J. Prev. Med. 2022, 63, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Lartey, S.T.; Magnussen, C.G.; Si, L.; Boateng, G.O.; de Graaff, B.; Biritwum, R.B.; Minicuci, N.; Kowal, P.; Blizzard, L.; Palmer, A.J. Rapidly Increasing Prevalence of Overweight and Obesity in Older Ghanaian Adults from 2007–2015: Evidence from WHO-SAGE Waves 1 & 2. PLoS ONE 2019, 14, e0215045. [Google Scholar] [CrossRef]
- Lange, E.H.; Nesvåg, R.; Ringen, P.A.; Hartberg, C.B.; Haukvik, U.K.; Andreassen, O.A.; Melle, I.; Agartz, I. One Year Follow-up of Alcohol and Illicit Substance Use in First-Episode Psychosis: Does Gender Matter? Compr. Psychiatry 2014, 55, 274–282. [Google Scholar] [CrossRef]
- Bezdjian, S.; Baker, L.A.; Lozano, D.I.; Raine, A. Assessing Inattention and Impulsivity in Children during the Go/NoGo Task. Br. J. Dev. Psychol. 2009, 27, 365–383. [Google Scholar] [CrossRef]
- Ilardi, C.R.; Chieffi, S.; Scuotto, C.; Gamboz, N.; Galeone, F.; Sannino, M.; Garofalo, E.; La Marra, M.; Ronga, B.; Iavarone, A. The Frontal Assessment Battery 20 Years Later: Normative Data for a Shortened Version (FAB15). Neurol. Sci. 2022, 43, 1709–1719. [Google Scholar] [CrossRef]
- Giambra, L.M.; Quilter, R.E. Sex Differences in Sustained Attention across the Adult Life Span. J. Appl. Psychol. 1989, 74, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Riley, N.; Lubans, D.R.; Holmes, K.; Morgan, P.J. Findings From the EASY Minds Cluster Randomized Controlled Trial: Evaluation of a Physical Activity Integration Program for Mathematics in Primary Schools. J. Phys. Act. Health 2016, 13, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.H.; Jones, P.B.; Robbins, T.W.; Müller, U. Effects of the Catechol-O-Methyltransferase Val158Met Polymorphism on Executive Function: A Meta-Analysis of the Wisconsin Card Sort Test in Schizophrenia and Healthy Controls. Mol. Psychiatry 2007, 12, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; Verde, L.; Vetrani, C.; Barrea, L.; Savastano, S.; Colao, A. Obesity: A Gender-View. J. Endocrinol. Investig. 2023, 47, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Conners, C.K.; Epstein, J.N.; Angold, A.; Klaric, J. Continuous Performance Test Performance in a Normative Epidemiological Sample. J. Abnorm. Child. Psychol. 2003, 31, 555–562. [Google Scholar] [CrossRef]
- Naglieri, J.A.; Rojahn, J. Gender Differences in Planning, Attention, Simultaneous, and Successive (PASS) Cognitive Processes and Achievement. J. Educ. Psychol. 2001, 93, 430–437. [Google Scholar] [CrossRef]
- Bardos, A.N.; Naglieri, J.A.; Prewett, P.N. Gender Differences on Planning, Attention, Simultaneous, and Successive Cognitive Processing Tasks. J. Sch. Psychol. 1992, 30, 293–305. [Google Scholar] [CrossRef]
- Warrick, P.D.; Naglieri, J.A. Gender Differences in Planning, Attention, Simultaneous, and Successive (PASS) Cognitive Processes. J. Educ. Psychol. 1993, 85, 693–701. [Google Scholar] [CrossRef]
- Overman, W.H.; Pierce, A. Iowa Gambling Task with Non-Clinical Participants: Effects of Using Real + Virtual Cards and Additional Trials. Front. Psychol. 2013, 4, 64997. [Google Scholar] [CrossRef]
- Van Den Bos, R.; Homberg, J.; De Visser, L. A Critical Review of Sex Differences in Decision-Making Tasks: Focus on the Iowa Gambling Task. Behav. Brain Res. 2013, 238, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.L.; Gallagher, N.M.; Sullivan, M.; Callicott, J.H.; Green, A.E. Sex Differences in Verbal Working Memory Performance Emerge at Very High Loads of Common Neuroimaging Tasks. Brain Cogn. 2017, 113, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Rahman, Q.; Abrahams, S.; Jussab, F. Sex Differences in a Human Analogue of the Radial Arm Maze: The “17-Box Maze Test”. Brain Cogn. 2005, 58, 312–317. [Google Scholar] [CrossRef]
- Duff, S.J.; Hampson, E. A Sex Difference on a Novel Spatial Working Memory Task in Humans. Brain Cogn. 2001, 47, 470–493. [Google Scholar] [CrossRef] [PubMed]
- Lejbak, L.; Vrbancic, M.; Crossley, M. The Female Advantage in Object Location Memory Is Robust to Verbalizability and Mode of Presentation of Test Stimuli. Brain Cogn. 2009, 69, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, A.; Fehring, D.J.; Rossell, S.L. A Systematic Review and Meta-analysis of Behavioural Sex Differences in Executive Control. Eur. J. Neurosci. 2021, 53, 519–542. [Google Scholar] [CrossRef] [PubMed]
- Grissom, N.M.; Reyes, T.M. Let’s Call the Whole Thing off: Evaluating Gender and Sex Differences in Executive Function. Neuropsychopharmacology 2019, 44, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Verbruggen, F.; Logan, G.D. Response Inhibition in the Stop-Signal Paradigm. Trends Cogn. Sci. 2008, 12, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L. Why Sex Matters for Neuroscience. Nat. Rev. Neurosci. 2006, 7, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L. Frontal-Subcortical Circuits and Human Behavior. Arch. Neurol. 1993, 50, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.K.; Cohen, J.D. An Integrative Theory of Prefrontal Cortex Function. Annu. Rev. Neurosci. 2001, 24, 167–202. [Google Scholar] [CrossRef] [PubMed]
- Bonté, E.; Flemming, T.; Fagot, J. Executive Control of Perceptual Features and Abstract Relations by Baboons (Papio Papio). Behav. Brain Res. 2011, 222, 176–182. [Google Scholar] [CrossRef]
- Mansouri, F.A.; Tanaka, K.; Buckley, M.J. Conflict-Induced Behavioural Adjustment: A Clue to the Executive Functions of the Prefrontal Cortex. Nat. Rev. Neurosci. 2009, 10, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Ilardi, C.R.; La Marra, M.; Amato, R.; Di Cecca, A.; Di Maio, G.; Ciccarelli, G.; Migliaccio, M.; Cavaliere, C.; Federico, G. The “Little Circles Test” (LCT): A Dusted-off Tool for Assessing Fine Visuomotor Function. Aging Clin. Exp. Res. 2023, 35, 2807–2820. [Google Scholar] [CrossRef] [PubMed]
- Measso, G.; Cavarzeran, F.; Zappalà, G.; Lebowitz, B.D.; Crook, T.H.; Pirozzolo, F.J.; Amaducci, L.A.; Massari, D.; Grigoletto, F. The Mini-mental State Examination: Normative Study of an Italian Random Sample. Dev. Neuropsychol. 1993, 9, 77–85. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. Mini-Mental State. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- La Marra, M.; Ilardi, C.R.; Villano, I.; Polito, R.; Sibillo, M.R.; Franchetti, M.; Caggiano, A.; Strangio, F.; Messina, G.; Monda, V.; et al. Higher General Executive Functions Predicts Lower Body Mass Index by Mitigating Avoidance Behaviors. Front. Endocrinol. 2022, 13, 1048363. [Google Scholar] [CrossRef] [PubMed]
- La Marra, M.; Messina, A.; Ilardi, C.R.; Verde, G.; Amato, R.; Esposito, N.; Troise, S.; Orlando, A.; Messina, G.; Monda, V.; et al. The Neglected Factor in the Relationship between Executive Functioning and Obesity: The Role of Motor Control. Healthcare 2022, 10, 1775. [Google Scholar] [CrossRef] [PubMed]
- Vainik, U.; Dagher, A.; Dubé, L.; Fellows, L.K. Neurobehavioural Correlates of Body Mass Index and Eating Behaviours in Adults: A Systematic Review. Neurosci. Biobehav. Rev. 2013, 37, 279–299. [Google Scholar] [CrossRef]
- Emery, R.L.; Levine, M.D. Questionnaire and Behavioral Task Measures of Impulsivity Are Differentially Associated with Body Mass Index: A Comprehensive Meta-Analysis. Psychol. Bull. 2017, 143, 868–902. [Google Scholar] [CrossRef]
- Gettens, K.M.; Gorin, A.A. Executive Function in Weight Loss and Weight Loss Maintenance: A Conceptual Review and Novel Neuropsychological Model of Weight Control. J. Behav. Med. 2017, 40, 687–701. [Google Scholar] [CrossRef]
- Favieri, F.; Forte, G.; Pazzaglia, M.; Chen, E.Y.; Casagrande, M. High-Level Executive Functions: A Possible Role of Sex and Weight Condition in Planning and Decision-Making Performances. Brain Sci. 2022, 12, 149. [Google Scholar] [CrossRef] [PubMed]
- Jupp, B.; Caprioli, D.; Saigal, N.; Reverte, I.; Shrestha, S.; Cumming, P.; Everitt, B.J.; Robbins, T.W.; Dalley, J.W. Dopaminergic and GABA -ergic Markers of Impulsivity in Rats: Evidence for Anatomical Localisation in Ventral Striatum and Prefrontal Cortex. Eur. J. Neurosci. 2013, 37, 1519–1528. [Google Scholar] [CrossRef]
- Selleck, R.A.; Lake, C.; Estrada, V.; Riederer, J.; Andrzejewski, M.; Sadeghian, K.; Baldo, B.A. Endogenous Opioid Signaling in the Medial Prefrontal Cortex Is Required for the Expression of Hunger-Induced Impulsive Action. Neuropsychopharmacology 2015, 40, 2464–2474. [Google Scholar] [CrossRef]
- Feja, M.; Koch, M. Ventral Medial Prefrontal Cortex Inactivation Impairs Impulse Control but Does Not Affect Delay-Discounting in Rats. Behav. Brain Res. 2014, 264, 230–239. [Google Scholar] [CrossRef]
- Chieffi, S.; Messina, G.; La Marra, M.; Iavarone, A.; Viggiano, A.; De Luca, V.; Monda, M. Distractor Interference in Visual Motor Tasks. In Horizons in Neuroscience Research; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2014; Volume 13, pp. 151–160. [Google Scholar]
- Ilardi, C.R.; Di Maio, G.; Villano, I.; Messina, G.; Monda, V.; Messina, A.; Porro, C.; Panaro, M.A.; Gamboz, N.; Iavarone, A.; et al. The Assessment of Executive Functions to Test the Integrity of the Nigrostriatal Network: A Pilot Study. Front. Psychol. 2023, 14, 1121251. [Google Scholar] [CrossRef] [PubMed]
- Yates, J.R.; Darna, M.; Beckmann, J.S.; Dwoskin, L.P.; Bardo, M.T. Individual Differences in Impulsive Action and Dopamine Transporter Function in Rat Orbitofrontal Cortex. Neuroscience 2016, 313, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Tessitore, A.; Giordano, A.; Russo, A.; Tedeschi, G. Structural Connectivity in Parkinson’s Disease. Park. Relat. Disord. 2016, 22, S56–S59. [Google Scholar] [CrossRef] [PubMed]
- Benn, A.; Robinson, E.S.J. Investigating Glutamatergic Mechanism in Attention and Impulse Control Using Rats in a Modified 5-Choice Serial Reaction Time Task. PLoS ONE 2014, 9, e115374. [Google Scholar] [CrossRef] [PubMed]
- Murphy-Royal, C.; Dupuis, J.; Groc, L.; Oliet, S.H.R. Astroglial Glutamate Transporters in the Brain: Regulating Neurotransmitter Homeostasis and Synaptic Transmission. J. Neurosci. Res. 2017, 95, 2140–2151. [Google Scholar] [CrossRef] [PubMed]
- Di Maio, G.; Villano, I.; Ilardi, C.R.; Messina, A.; Monda, V.; Iodice, A.C.; Porro, C.; Panaro, M.A.; Chieffi, S.; Messina, G.; et al. Mechanisms of Transmission and Processing of Pain: A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20, 3064. [Google Scholar] [CrossRef] [PubMed]
- D’Amour-Horvat, V.; Leyton, M. Impulsive Actions and Choices in Laboratory Animals and Humans: Effects of High vs. Low Dopamine States Produced by Systemic Treatments given to Neurologically Intact Subjects. Front. Behav. Neurosci. 2014, 8, 432. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Roige, S.; Ripley, T.L.; Stephens, D.N. Alleviating Waiting Impulsivity and Perseverative Responding by μ-Opioid Receptor Antagonism in Two Inbred Mouse Strains. Psychopharmacology 2015, 232, 1483–1492. [Google Scholar] [CrossRef]
- Kolisnyk, B.; Al-Onaizi, M.A.; Hirata, P.H.F.; Guzman, M.S.; Nikolova, S.; Barbash, S.; Soreq, H.; Bartha, R.; Prado, M.A.M.; Prado, V.F. Forebrain Deletion of the Vesicular Acetylcholine Transporter Results in Deficits in Executive Function, Metabolic, and RNA Splicing Abnormalities in the Prefrontal Cortex. J. Neurosci. 2013, 33, 14908–14920. [Google Scholar] [CrossRef] [PubMed]
- Staiti, A.M.; Morgane, P.J.; Galler, J.R.; Grivetti, J.Y.; Bass, D.C.; Mokler, D.J. A Microdialysis Study of the Medial Prefrontal Cortex of Adolescent and Adult Rats. Neuropharmacology 2011, 61, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Alves, N.C.; Bailey, C.D.C.; Nashmi, R.; Lambe, E.K. Developmental Sex Differences in Nicotinic Currents of Prefrontal Layer VI Neurons in Mice and Rats. PLoS ONE 2010, 5, e9261. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.L.; Teicher, M.H. Sex Differences in Dopamine Receptors and Their Relevance to ADHD. Neurosci. Biobehav. Rev. 2000, 24, 137–141. [Google Scholar] [CrossRef]
- LaRoche, R.B.; Morgan, R.E. Adolescent Fluoxetine Exposure Produces Enduring, Sex-Specific Alterations of Visual Discrimination and Attention in Rats. Neurotoxicol. Teratol. 2007, 29, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Kolb, B.; Mychasiuk, R.; Muhammad, A.; Li, Y.; Frost, D.O.; Gibb, R. Experience and the Developing Prefrontal Cortex. Proc. Natl. Acad. Sci. USA 2012, 109, 17186–17193. [Google Scholar] [CrossRef]
- McGrath, J.; Saha, S.; Welham, J.; El Saadi, O.; MacCauley, C.; Chant, D. A Systematic Review of the Incidence of Schizophrenia: The Distribution of Rates and the Influence of Sex, Urbanicity, Migrant Status and Methodology. BMC Med. 2004, 2, 13. [Google Scholar] [CrossRef]
- Wu, G.; Qi, F.; Shen, D. Learning-Based Deformable Registration of MR Brain Images. IEEE Trans. Med. Imaging 2006, 25, 1145–1157. [Google Scholar] [CrossRef]
- Villano, I.; La Marra, M.; Di Maio, G.; Monda, V.; Chieffi, S.; Guatteo, E.; Messina, G.; Moscatelli, F.; Monda, M.; Messina, A. Physiological Role of Orexinergic System for Health. Int. J. Environ. Res. Public Health 2022, 19, 8353. [Google Scholar] [CrossRef]
- Stevens, B.; Allen, N.J.; Vazquez, L.E.; Howell, G.R.; Christopherson, K.S.; Nouri, N.; Micheva, K.D.; Mehalow, A.K.; Huberman, A.D.; Stafford, B.; et al. The Classical Complement Cascade Mediates CNS Synapse Elimination. Cell 2007, 131, 1164–1178. [Google Scholar] [CrossRef]
- Bollinger, J.L.; Bergeon Burns, C.M.; Wellman, C.L. Differential Effects of Stress on Microglial Cell Activation in Male and Female Medial Prefrontal Cortex. Brain Behav. Immun. 2016, 52, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, J.L.; Collins, K.E.; Patel, R.; Wellman, C.L. Behavioral Stress Alters Corticolimbic Microglia in a Sex- and Brain Region-Specific Manner. PLoS ONE 2017, 12, e0187631. [Google Scholar] [CrossRef] [PubMed]
- Barton, E.; Baker, C.; Leasure, J. Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise. Brain Sci. 2017, 7, 139. [Google Scholar] [CrossRef] [PubMed]
- Gurvich, C.; Le, J.; Thomas, N.; Thomas, E.H.X.; Kulkarni, J. Sex Hormones and Cognition in Aging. In Vitamins and Hormones; Elsevier: Amsterdam, The Netherlands, 2021; Volume 115, pp. 511–533. ISBN 978-0-323-85548-8. [Google Scholar]
- Sultana, F.; Davis, S.R.; Murray, A.M.; Woods, R.L.; McNeil, J.J.; Islam, R.M. Sex Hormones, SHBG and Cognitive Performance among Older Australian Women: An Observational Study. Climacteric 2023, 26, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.-V.; Lew, J.; Albaugh, M.D.; Botteron, K.N.; Hudziak, J.J.; Fonov, V.S.; Collins, D.L.; Ducharme, S.; McCracken, J.T. Sex-Specific Associations of Testosterone with Prefrontal-Hippocampal Development and Executive Function. Psychoneuroendocrinology 2017, 76, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Sie, J.-H.; Chen, Y.-H.; Shiau, Y.-H.; Chu, W.-C. Gender- and Age-Specific Differences in Resting-State Functional Connectivity of the Central Autonomic Network in Adulthood. Front. Hum. Neurosci. 2019, 13, 369. [Google Scholar] [CrossRef] [PubMed]
- Derntl, B.; Pintzinger, N.; Kryspin-Exner, I.; Schöpf, V. The Impact of Sex Hormone Concentrations on Decision-Making in Females and Males. Front. Neurosci. 2014, 8, 111184. [Google Scholar] [CrossRef] [PubMed]
- Rotge, J.-Y.; Poitou, C.; Fossati, P.; Aron-Wisnewsky, J.; Oppert, J.-M. Decision-making in Obesity without Eating Disorders: A Systematic Review and Meta-analysis of Iowa Gambling Task Performances. Obes. Rev. 2017, 18, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Damasio, A.R. On Some Functions of the Human Prefrontal Cortexa. Ann. N. Y. Acad. Sci. 1995, 769, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Villano, I.; La Marra, M.; Allocca, S.; Ilardi, C.R.; Polito, R.; Porro, C.; Chieffi, S.; Messina, G.; Monda, V.; Di Maio, G.; et al. The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy. Biomolecules 2022, 12, 1118. [Google Scholar] [CrossRef] [PubMed]
- Villano, I.; Ilardi, C.R.; Arena, S.; Scuotto, C.; Gleijeses, M.G.; Messina, G.; Messina, A.; Monda, V.; Monda, M.; Iavarone, A.; et al. Obese Subjects without Eating Disorders Experience Binge Episodes Also Independently of Emotional Eating and Personality Traits among University Students of Southern Italy. Brain Sci. 2021, 11, 1145. [Google Scholar] [CrossRef] [PubMed]
- La Marra, M.; Messina, A.; Ilardi, C.R.; Staiano, M.; Di Maio, G.; Messina, G.; Polito, R.; Valenzano, A.; Cibelli, G.; Monda, V.; et al. Factorial Model of Obese Adolescents: The Role of Body Image Concerns and Selective Depersonalization—A Pilot Study. Int. J. Environ. Res. Public Health 2022, 19, 11501. [Google Scholar] [CrossRef] [PubMed]
- Val-Laillet, D.; Layec, S.; Guérin, S.; Meurice, P.; Malbert, C. Changes in Brain Activity After a Diet-Induced Obesity. Obesity 2011, 19, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Morella, I.M.; Brambilla, R.; Morè, L. Emerging Roles of Brain Metabolism in Cognitive Impairment and Neuropsychiatric Disorders. Neurosci. Biobehav. Rev. 2022, 142, 104892. [Google Scholar] [CrossRef]
- Iozzo, P.; Guzzardi, M.A. Imaging of Brain Glucose Uptake by PET in Obesity and Cognitive Dysfunction: Life-Course Perspective. Endocr. Connect. 2019, 8, R169–R183. [Google Scholar] [CrossRef] [PubMed]
- Rebelos, E.; Hirvonen, J.; Bucci, M.; Pekkarinen, L.; Nyman, M.; Hannukainen, J.C.; Iozzo, P.; Salminen, P.; Nummenmaa, L.; Ferrannini, E.; et al. Brain Free Fatty Acid Uptake Is Elevated in Morbid Obesity, and Is Irreversible 6 Months after Bariatric Surgery: A Positron Emission Tomography Study. Diabetes Obes. Metab. 2020, 22, 1074–1082. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Profant, M.; Wang, C. Metabolic Sex Dimorphism of the Brain at the Gene, Cell, and Tissue Level. J. Immunol. 2022, 208, 212–220. [Google Scholar] [CrossRef]
- Gaignard, P.; Fréchou, M.; Liere, P.; Thérond, P.; Schumacher, M.; Slama, A.; Guennoun, R. Sex Differences in Brain Mitochondrial Metabolism: Influence of Endogenous Steroids and Stroke. J. Neuroendocrinol. 2018, 30, e12497. [Google Scholar] [CrossRef]
- Francavilla, V.C.; Genovesi, F.; Asmundo, A.; Di Nunno, N.R.; Ambrosi, A.; Tartaglia, N.; Tafuri, D.; Monda, V.; Monda, M.; Messina, A.; et al. Fascia and Movement: The Primary Link in the Prevention of Accidents in Soccer. Revision and Models of Intervention. Med. Sport. 2020, 73, 291–301. [Google Scholar] [CrossRef]
- Murray, P.J. Macrophage Polarization. Annu. Rev. Physiol. 2017, 79, 541–566. [Google Scholar] [CrossRef]
- Guillemot-Legris, O.; Muccioli, G.G. Obesity-Induced Neuroinflammation: Beyond the Hypothalamus. Trends Neurosci. 2017, 40, 237–253. [Google Scholar] [CrossRef]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in Inflammation and Metabolic Disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Di Maio, G.; Alessio, N.; Demirsoy, I.H.; Peluso, G.; Perrotta, S.; Monda, M.; Di Bernardo, G. Evaluation of Browning Agents on the White Adipogenesis of Bone Marrow Mesenchymal Stromal Cells: A Contribution to Fighting Obesity. Cells 2021, 10, 403. [Google Scholar] [CrossRef] [PubMed]
- Castanon, N.; Luheshi, G.; Layé, S. Role of Neuroinflammation in the Emotional and Cognitive Alterations Displayed by Animal Models of Obesity. Front. Neurosci. 2015, 9, 146137. [Google Scholar] [CrossRef] [PubMed]
- Tucsek, Z.; Toth, P.; Tarantini, S.; Sosnowska, D.; Gautam, T.; Warrington, J.P.; Giles, C.B.; Wren, J.D.; Koller, A.; Ballabh, P.; et al. Aging Exacerbates Obesity-Induced Cerebromicrovascular Rarefaction, Neurovascular Uncoupling, and Cognitive Decline in Mice. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 1339–1352. [Google Scholar] [CrossRef] [PubMed]
- Mask, L.; Blanchard, C.M. The Protective Role of General Self-Determination against ‘Thin Ideal’ Media Exposure on Women’s Body Image and Eating-Related Concerns. J. Health Psychol. 2011, 16, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Groesz, L.M.; Levine, M.P.; Murnen, S.K. The Effect of Experimental Presentation of Thin Media Images on Body Satisfaction: A Meta-analytic Review. Int. J. Eat. Disord. 2002, 31, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Posavac, S.S.; Posavac, H.D. Predictors of Women’s Concern with Body Weight: The Roles of Perceived Self-Media Ideal Discrepancies and Self-Esteem. Eat. Disord. 2002, 10, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Sands, E.R.; Wardle, J. Internalization of Ideal Body Shapes in 9–12-year-old Girls. Int. J. Eat. Disord. 2003, 33, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Ahern, A.L.; Bennett, K.M.; Kelly, M.; Hetherington, M.M. A Qualitative Exploration of Young Women’s Attitudes towards the Thin Ideal. J. Health Psychol. 2011, 16, 70–79. [Google Scholar] [CrossRef]
- Pradeilles, R.; Holdsworth, M.; Olaitan, O.; Irache, A.; Osei-Kwasi, H.A.; Ngandu, C.B.; Cohen, E. Body Size Preferences for Women and Adolescent Girls Living in Africa: A Mixed-Methods Systematic Review. Public Health Nutr. 2022, 25, 738–759. [Google Scholar] [CrossRef] [PubMed]
- Rguibi, M.; Belahsen, R. Body Size Preferences and Sociocultural Influences on Attitudes towards Obesity among Moroccan Sahraoui Women. Body Image 2006, 3, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Rguibi, M.; Belahsen, R. Fattening Practices among Moroccan Saharawi Women. East. Mediterr. Health J. 2006, 12, 619–624. [Google Scholar] [PubMed]
| Whole Sample (N = 386) | Females (n = 222) | Males (n = 164) | p-Value | |
|---|---|---|---|---|
| Age (years) | 41.80 (18.32) | 42.66 (18.18) | 40.63 (18.50) | ns |
| Education (years) | 15.11 (3.96) | 15.17 (4.03) | 15.02 (3.89) | ns |
| MMSE | 29.11 (1.29) | 29.09 (1.40) | 29.13 (1.12) | ns |
| FAB15 | 13.56 (1.62) | 13.64 (1.72) | 13.46 (1.47) | ns |
| BMI | 25.32 (4.53) | 25.10 (4.81) | 25.62 (4.13) | ns |
| WC | 86.11 (14.83) | 83.26 (15.07) | 90.17 (13.53) | <0.001 |
| b | SE | t | p | LLCI | ULCI | |
|---|---|---|---|---|---|---|
| Constant | 36.213 | 3.159 | 11.462 | <0.001 | 29.998 | 42.428 |
| FAB15 | −0.787 | 0.233 | −3.376 | <0.001 | −1.246 | −0.329 |
| Sex | 5.939 | 3.937 | 1.508 | 0.132 | −1.806 | 13.685 |
| FAB15×Sex | −0.476 | 0.289 | −1.647 | 0.100 | −1.045 | 0.092 |
| b | SE | t | p | LLCI | ULCI | |
|---|---|---|---|---|---|---|
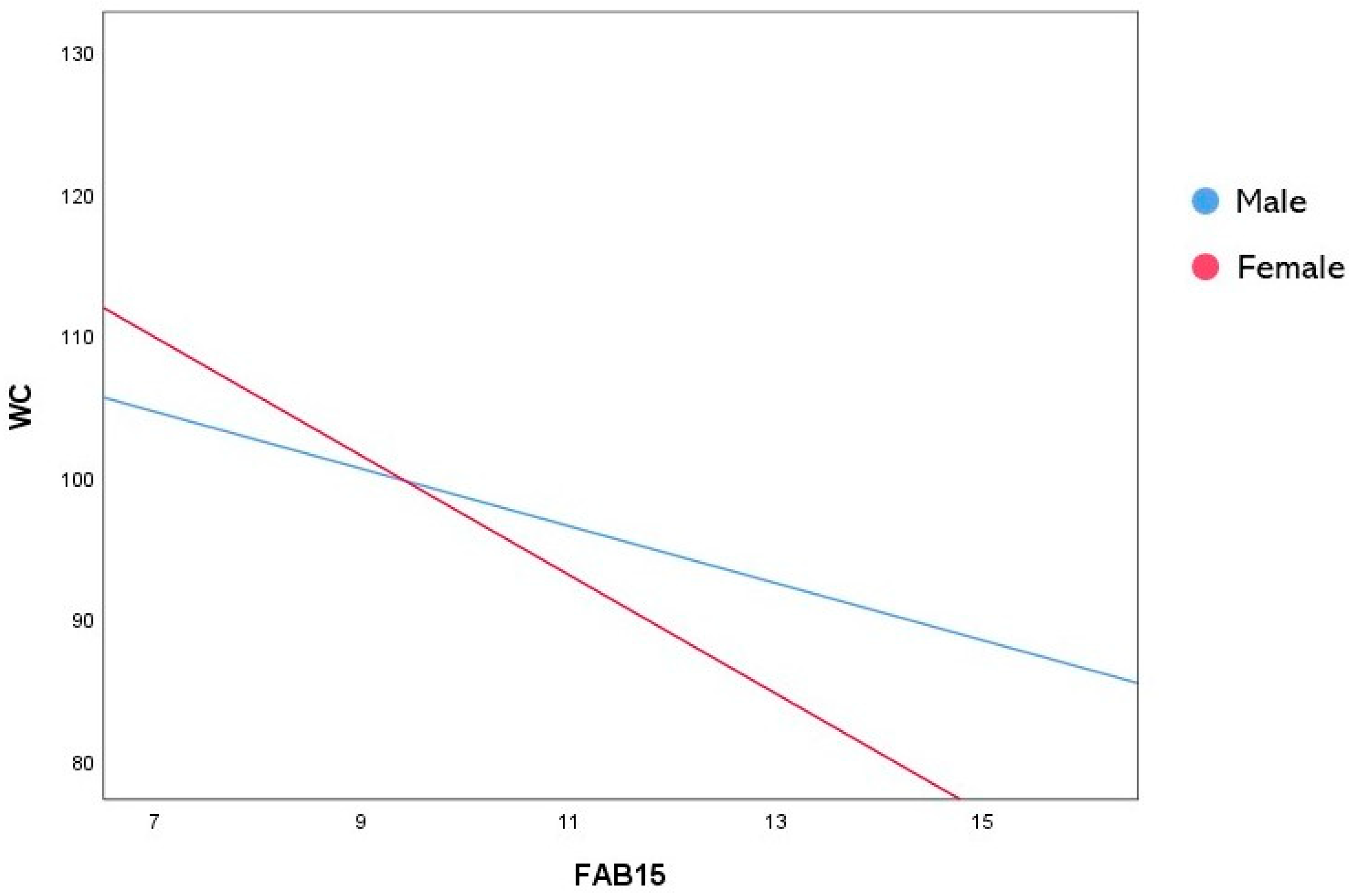
| Constant | 118.653 | 9.702 | 12.229 | <0.001 | 99.567 | 137.739 |
| FAB15 | −2.014 | 0.717 | −2.808 | 0.005 | −3.426 | −0.603 |
| Sex | 20.444 | 12.065 | 1.694 | 0.091 | −3.290 | 44.179 |
| FAB15×Sex | −2.172 | 0.887 | −2.448 | 0.015 | −3.917 | −0.427 |
| b | SE | t | p | LLCI | ULCI | |
|---|---|---|---|---|---|---|
| Male | −2.014 | 0.717 | −2.808 | 0.005 | −3.426 | −0.603 |
| Female | −4.187 | 0.522 | −8.023 | <0.001 | −5.213 | −3.160 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilardi, C.R.; Monda, A.; Iavarone, A.; Chieffi, S.; Casillo, M.; Messina, A.; Villano, I.; Federico, G.; Alfano, V.; Salvatore, M.; et al. The Relationship between Executive Functions and Body Weight: Sex as a Moderating Variable. Behav. Sci. 2024, 14, 258. https://doi.org/10.3390/bs14030258
Ilardi CR, Monda A, Iavarone A, Chieffi S, Casillo M, Messina A, Villano I, Federico G, Alfano V, Salvatore M, et al. The Relationship between Executive Functions and Body Weight: Sex as a Moderating Variable. Behavioral Sciences. 2024; 14(3):258. https://doi.org/10.3390/bs14030258
Chicago/Turabian StyleIlardi, Ciro Rosario, Antonietta Monda, Alessandro Iavarone, Sergio Chieffi, Maria Casillo, Antonietta Messina, Ines Villano, Giovanni Federico, Vincenzo Alfano, Marco Salvatore, and et al. 2024. "The Relationship between Executive Functions and Body Weight: Sex as a Moderating Variable" Behavioral Sciences 14, no. 3: 258. https://doi.org/10.3390/bs14030258
APA StyleIlardi, C. R., Monda, A., Iavarone, A., Chieffi, S., Casillo, M., Messina, A., Villano, I., Federico, G., Alfano, V., Salvatore, M., Sapuppo, W., Monda, V., Monda, M., Di Maio, G., & La Marra, M. (2024). The Relationship between Executive Functions and Body Weight: Sex as a Moderating Variable. Behavioral Sciences, 14(3), 258. https://doi.org/10.3390/bs14030258








