The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome
Abstract
1. Introduction
2. Symptomatology and Etiopathology of Tourette Syndrome
3. Evidence from Animal Models of Tourette Syndrome
4. Structural and Functional Changes in Tourette Patients
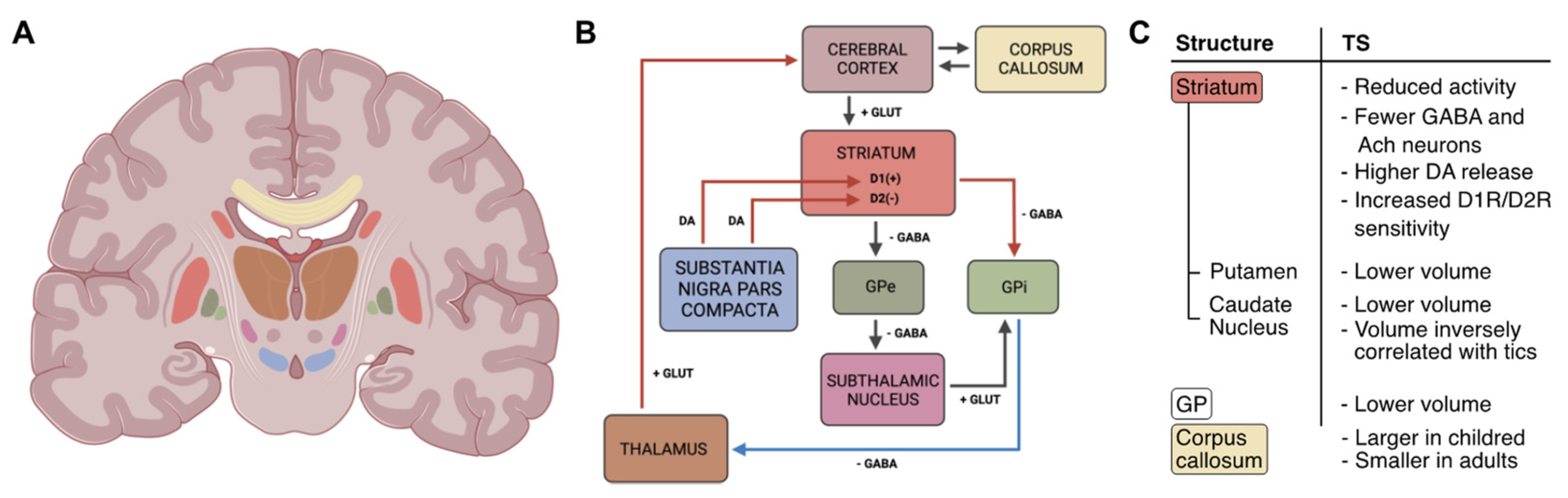
4.1. Cortical-Striatal-Thalamus-Cortical Circuit and Basal Ganglia
4.2. Dopamine System
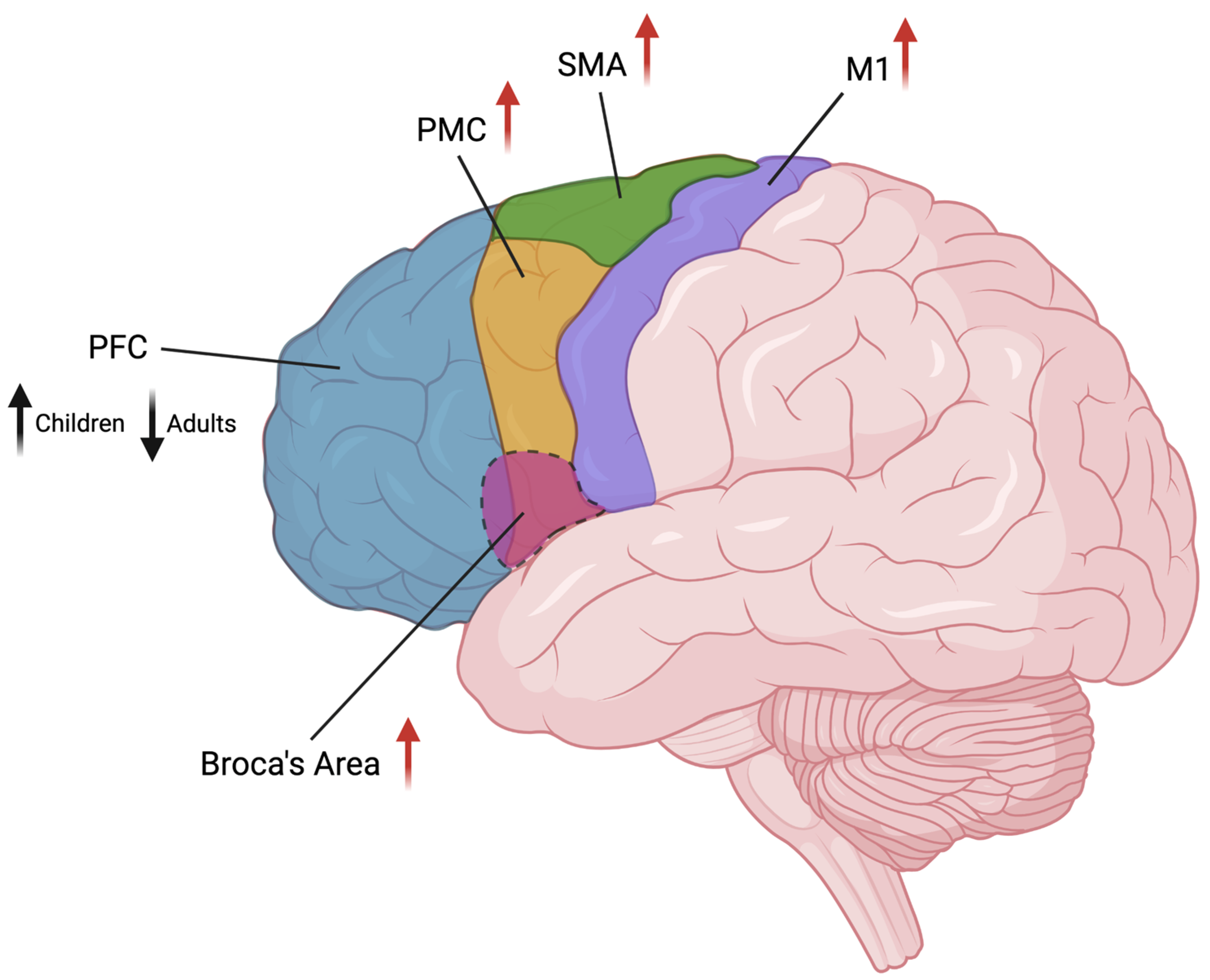
4.3. Cortical and Subcortical Motor Areas
5. Functional Plasticity Alterations in Patients with Tourette Syndrome
6. Striatal Synaptic Plasticity as a Possible Pathophysiological Mechanism
Immunological Bases of Tourette Syndrome and Striatal Plasticity
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Como, P.G. Neuropsychological Function in Tourette Syndrome. Adv. Neurol. 2001, 85, 103–111. [Google Scholar] [CrossRef]
- Robertson, M.M. Tourette Syndrome, Associated Conditions and the Complexities of Treatment. Brain 2000, 123, 425–462. [Google Scholar] [CrossRef]
- Robertson, M.M. The Prevalence and Epidemiology of Gilles de La Tourette Syndrome. Part 1: The Epidemiological and Prevalence Studies. J. Psychosom. Res. 2008, 65, 461–472. [Google Scholar] [CrossRef]
- Robertson, M.M. Tourette Syndrome in Children and Adolescents: Aetiology, Presentation and Treatment. BJPsych Adv. 2016, 22, 165–175. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5), 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013; Volume 280. [Google Scholar] [CrossRef]
- Khalifa, N.; Knorring, A.-L. Tourette Syndrome and Other Tic Disorders in a Total Population of Children: Clinical Assessment and Background. Acta Paediatr. 2005, 94, 1608–1614. [Google Scholar] [CrossRef]
- Robertson, M.M. Mood Disorders and Gilles de La Tourette’s Syndrome: An Update on Prevalence, Etiology, Comorbidity, Clinical Associations, and Implications. J. Psychosom. Res. 2006, 61, 349–358. [Google Scholar] [CrossRef]
- Leckman, J.F. Phenomenology of Tics and Natural History of Tic Disorders. Brain Dev. 2003, 25 (Suppl. 1), S24–S28. [Google Scholar] [CrossRef]
- Cavanna, A.E.; Servo, S.; Monaco, F.; Robertson, M.M. The Behavioral Spectrum of Gilles de La Tourette Syndrome. J. Neuropsychiatry Clin. Neurosci. 2009, 21, 13–23. [Google Scholar] [CrossRef]
- Jankovic, J. Tourette Syndrome. Phenomenology and Classification of Tics. Neurol. Clin. 1997, 15, 267–275. [Google Scholar] [CrossRef]
- Sambrani, T.; Jakubovski, E.; Müller-Vahl, K.R. New Insights into Clinical Characteristics of Gilles de La Tourette Syndrome: Findings in 1032 Patients from a Single German Center. Front. Neurosci. 2016, 10, 415. [Google Scholar] [CrossRef]
- Leckman, J.F.; Walker, D.E.; Cohen, D.J. Premonitory Urges in Tourette’s Syndrome. Am. J. Psychiatry 1993, 150, 98–102. [Google Scholar] [CrossRef]
- Robertson, M.M. A Personal 35 Year Perspective on Gilles de La Tourette Syndrome: Prevalence, Phenomenology, Comorbidities, and Coexistent Psychopathologies. Lancet Psychiatry 2015, 2, 68–87. [Google Scholar] [CrossRef]
- Frick, L.; Pittenger, C. Microglial Dysregulation in OCD, Tourette Syndrome, and PANDAS. J. Immunol. Res. 2016, 2016, 8606057. [Google Scholar] [CrossRef]
- Hickman, S.; Izzy, S.; Sen, P.; Morsett, L.; El Khoury, J. Microglia in Neurodegeneration. Nat. Neurosci. 2018, 21, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Ohbayashi, M. The Roles of the Cortical Motor Areas in Sequential Movements. Front. Behav. Neurosci. 2021, 15, 97. [Google Scholar] [CrossRef] [PubMed]
- Lang, A. Patient Perception of Tics and Other Movement Disorders. Neurology 1991, 41, 223–228. [Google Scholar] [CrossRef]
- Frey, K.A.; Albin, R.L. Neuroimaging of Tourette Syndrome. J. Child Neurol. 2006, 21, 672–677. [Google Scholar] [CrossRef]
- Macrì, S.; Onori, M.P.; Roessner, V.; Laviola, G. Animal Models Recapitulating the Multifactorial Origin of Tourette Syndrome, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 112, ISBN 9780124115460. [Google Scholar]
- Hornig, M.; Lipkin, W.I. Immune-Mediated Animal Models of Tourette Syndrome. Neurosci. Biobehav. Rev. 2013, 37, 1120–1138. [Google Scholar] [CrossRef]
- Bronfeld, M.; Israelashvili, M.; Bar-Gad, I. Pharmacological Animal Models of Tourette Syndrome. Neurosci. Biobehav. Rev. 2013, 37, 1101–1119. [Google Scholar] [CrossRef]
- Worbe, Y.; Malherbe, C.; Hartmann, A.; Pélégrini-Issac, M.; Messé, A.; Vidailhet, M.; Lehéricy, S.; Benali, H. Functional Immaturity of Cortico-Basal Ganglia Networks in Gilles de La Tourette Syndrome. Brain 2012, 135, 1937–1946. [Google Scholar] [CrossRef]
- McCairn, K.W.; Nagai, Y.; Hori, Y.; Ninomiya, T.; Kikuchi, E.; Lee, J.Y.; Suhara, T.; Iriki, A.; Minamimoto, T.; Takada, M.; et al. A Primary Role for Nucleus Accumbens and Related Limbic Network in Vocal Tics. Neuron 2016, 89, 300–307. [Google Scholar] [CrossRef] [PubMed]
- McCairn, K.W.; Bronfeld, M.; Belelovsky, K.; Bar-Gad, I. The Neurophysiological Correlates of Motor Tics Following Focal Striatal Disinhibition. Brain 2009, 132, 2125–2138. [Google Scholar] [CrossRef]
- Worbe, Y.; Sgambato-Faure, V.; Epinat, J.; Chaigneau, M.; Tandé, D.; François, C.; Féger, J.; Tremblay, L. Towards a Primate Model of Gilles de La Tourette Syndrome: Anatomo-Behavioural Correlation of Disorders Induced by Striatal Dysfunction. Cortex 2013, 49, 1126–1140. [Google Scholar] [CrossRef] [PubMed]
- Bronfeld, M.; Yael, D.; Belelovsky, K.; Bar-Gad, I. Motor Tics Evoked by Striatal Disinhibition in the Rat. Front. Syst. Neurosci. 2013, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- Israelashvili, M.; Bar-Gad, I. Corticostriatal Divergent Function in Determining the Temporal and Spatial Properties of Motor Tics. J. Neurosci. 2015, 35, 16340–16351. [Google Scholar] [CrossRef] [PubMed]
- Pogorelov, V.; Xu, M.; Smith, H.R.; Buchanan, G.F.; Pittenger, C. Corticostriatal Interactions in the Generation of Tic-like Behaviors after Local Striatal Disinhibition. Exp. Neurol. 2015, 265, 122–128. [Google Scholar] [CrossRef]
- Taylor, J.L.; Rajbhandari, A.K.; Berridge, K.C.; Aldridge, J.W. Dopamine Receptor Modulation of Repetitive Grooming Actions in the Rat: Potential Relevance for Tourette Syndrome. Brain Res. 2010, 1322, 92–101. [Google Scholar] [CrossRef]
- Nespoli, E.; Rizzo, F.; Boeckers, T.; Schulze, U.; Hengerer, B. Altered Dopaminergic Regulation of the Dorsal Striatum Is Able to Induce Tic-like Movements in Juvenile Rats. PLoS ONE 2018, 13, e0196515. [Google Scholar] [CrossRef]
- Godar, S.C.; Mosher, L.J.; Strathman, H.J.; Gochi, A.M.; Jones, C.M.; Fowler, S.C.; Bortolato, M. The D1CT-7 Mouse Model of Tourette Syndrome Displays Sensorimotor Gating Deficits in Response to Spatial Confinement. Br. J. Pharmacol. 2016, 173, 2111–2121. [Google Scholar] [CrossRef]
- Nordstrom, E.J.; Burton, F.H. A Transgenic Model of Comorbid Tourette’s Syndrome and Obsessive-Compulsive Disorder Circuitry. Mol. Psychiatry 2002, 7, 617–625. [Google Scholar] [CrossRef]
- Campbell, K.M.; De Lecea, L.; Severynse, D.M.; Caron, M.G.; McGrath, M.J.; Sparber, S.B.; Sun, L.Y.; Burton, F.H. OCD-like Behaviors Caused by a Neuropotentiating Transgene Targeted to Cortical and Limbic D1+ Neurons. J. Neurosci. 1999, 19, 5044–5053. [Google Scholar] [CrossRef]
- Berridge, K.C.; Aldridge, J.W.; Houchard, K.R.; Zhuang, X. Sequential Super-Stereotypy of an Instinctive Fixed Action Pattern in Hyper-Dopaminergic Mutant Mice: A Model of Obsessive Compulsive Disorder and Tourette’s. BMC Biol. 2005, 3, 4. [Google Scholar] [CrossRef]
- Lin, L.; Lan, Y.; Zhu, H.; Yu, L.; Wu, S.; Wan, W.; Shu, Y.; Xiang, H.; Hou, T.; Zhang, H.; et al. Effects of Chemogenetic Inhibition of D1 or D2 Receptor-Containing Neurons of the Substantia Nigra and Striatum in Mice With Tourette Syndrome. Front. Mol. Neurosci. 2021, 14, 779436. [Google Scholar] [CrossRef]
- Rapanelli, M.; Frick, L.; Bito, H.; Pittenger, C. Histamine Modulation of the Basal Ganglia Circuitry in the Development of Pathological Grooming. Proc. Natl. Acad. Sci. USA 2017, 114, 6599–6604. [Google Scholar] [CrossRef]
- Ahmari, S.E.; Spellman, T.; Douglass, N.L.; Kheirbek, M.A.; Simpson, H.B.; Deisseroth, K.; Gordon, J.A.; Hen, R. Repeated Cortico-Striatal Stimulation Generates Persistent OCD-like Behavior. Science 2013, 340, 1234–1239. [Google Scholar] [CrossRef]
- Bouchekioua, Y.; Tsutsui-Kimura, I.; Sano, H.; Koizumi, M.; Tanaka, K.F.; Yoshida, K.; Kosaki, Y.; Watanabe, S.; Mimura, M. Striatonigral Direct Pathway Activation Is Sufficient to Induce Repetitive Behaviors. Neurosci. Res. 2017, 132, 53–57. [Google Scholar] [CrossRef]
- Burguière, E.; Monteiro, P.; Feng, G.; Graybiel, A.M. Optogenetic Stimulation of Lateral Orbitofronto-Striatal Pathway Suppresses Compulsive Behaviors. Science 2013, 340, 1243–1246. [Google Scholar] [CrossRef]
- Lennington, J.B.; Coppola, G.; Kataoka-Sasaki, Y.; Fernandez, T.V.; Palejev, D.; Li, Y.; Huttner, A.; Pletikos, M.; Sestan, N.; Leckman, J.F.; et al. Transcriptome Analysis of the Human Striatum in Tourette Syndrome. Biol. Psychiatry 2016, 79, 372–382. [Google Scholar] [CrossRef]
- Liao, C.; Vuokila, V.; Catoire, H.; Akçimen, F.; Ross, J.P.; Bourassa, C.V.; Dion, P.A.; Meijer, I.A.; Rouleau, G.A. Transcriptome-Wide Association Study Reveals Increased Neuronal FLT3 Expression Is Associated with Tourette’s Syndrome. Commun. Biol. 2022, 5, 289. [Google Scholar] [CrossRef]
- Bhikram, T.; Arnold, P.; Crawley, A.; Abi-jaoude, E.; Sandor, P. The Functional Connectivity Profile of Tics and Obsessive-Compulsive Symptoms in Tourette Syndrome. J. Psychiatr. Res. 2020, 123, 128–135. [Google Scholar] [CrossRef]
- Leckman, J.F. Tourette’s Syndrome. Lancet 2002, 360, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Naro, A.; Billeri, L.; Colucci, V.P.; Le Cause, M.; De Domenico, C.; Ciatto, L.; Bramanti, P.; Bramanti, A.; Calabrò, R.S. Brain Functional Connectivity in Chronic Tic Disorders and Gilles de La Tourette Syndrome. Prog. Neurobiol. 2020, 194, 101884. [Google Scholar] [CrossRef] [PubMed]
- Graybiel, A.M.; Aosaki, T.; Flaherty, A.W.; Kimura, M. The Basal Ganglia and Adaptive Motor Control. Science 1994, 265, 1826–1831. [Google Scholar] [CrossRef]
- Alexander, G.E.; DeLong, M.R.; Strick, P.L. Parallel Organization of Functionally Segregated Circuits Linking Basal Ganglia and Cortex. Annu. Rev. Neurosci. 1986, 9, 357–381. [Google Scholar] [CrossRef] [PubMed]
- Eidelberg, D.; Moeller, J.R.; Antonini, A.; Kazumata, K.; Dhawan, V.; Budman, C.; Feigin, A. The Metabolic Anatomy of Tourette’s Syndrome. Neurology 1997, 48, 927–934. [Google Scholar] [CrossRef]
- Bar-Gad, I.; Bergman, H. Stepping out of the Box: Information Processing in the Neural Networks of the Basal Ganglia. Curr. Opin. Neurobiol. 2001, 11, 689–695. [Google Scholar] [CrossRef]
- Aouizerate, B.; Guehl, D.; Cuny, E.; Rougier, A.; Bioulac, B.; Tignol, J.; Burbaud, P. Pathophysiology of Obsessive-Compulsive Disorder: A Necessary Link between Phenomenology, Neuropsychology, Imagery and Physiology. Prog. Neurobiol. 2004, 72, 195–221. [Google Scholar] [CrossRef]
- Felling, R.J.; Singer, H.S. Neurobiology of Tourette Syndrome: Current Status and Need for Further Investigation. J. Neurosci. 2011, 31, 12387–12395. [Google Scholar] [CrossRef]
- Peterson, B.S.; Thomas, P.; Kane, M.J.; Scahill, L.; Zhang, H.; Bronen, R.; King, R.A.; Leckman, J.F.; Staib, L. Basal Ganglia Volumes in Patients with Gilles de La Tourette Syndrome. Arch. Gen. Psychiatry 2003, 60, 415–424. [Google Scholar] [CrossRef]
- Bloch, M.H.; Leckman, J.F.; Zhu, H.; Peterson, B.S. Caudate Volumes in Childhood Predict Symptom Severity in Adults with Tourette Syndrome. Neurology 2005, 65, 1253–1258. [Google Scholar] [CrossRef]
- Hyde, T.M.; Stacey, M.E.; Coppola, R.; Handel, S.F.; Rickler, K.C.; Weinberger, D.R. Cerebral Morphometric Abnormalities in Tourette’s Syndrome: A Quantitative MRI Study of Monozygotic Twins. Neurology 1995, 45, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, R.M.; Cummings, J.L. Frontal-Subcortical Circuitry and Behavior. Dialogues Clin. Neurosci. 2007, 9, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Albin, R.L.; Mink, J.W. Recent Advances in Tourette Syndrome Research. Trends Neurosci. 2006, 29, 175–182. [Google Scholar] [CrossRef]
- Leckman, J.F.; Michael, M.H.; Smith, M.E.; Larabi, D.; Hampson, M. Neurobiological Substrates of Tourette’s Disorder. J. Child Adolesc. Psychopharmacol. 2010, 20, 237–247. [Google Scholar] [CrossRef]
- Gerard, E.; Peterson, B.S. Developmental Processes and Brain Imaging Studies in Tourette Syndrome. J. Psychosom. Res. 2003, 55, 13–22. [Google Scholar] [CrossRef]
- Peterson, B.S.; Staib, L.; Scahill, L.; Zhang, H.; Anderson, C.; Leckman, J.F.; Cohen, D.J.; Gore, J.C.; Albert, J.; Webster, R. Regional Brain and Ventricular Volumes in Tourette Syndrome. Arch. Gen. Psychiatry 2001, 58, 427–440. [Google Scholar] [CrossRef]
- Denys, D.; de Vries, F.; Cath, D.; Figee, M.; Vulink, N.; Veltman, D.J.; van der Doef, T.F.; Boellaard, R.; Westenberg, H.; van Balkom, A.; et al. Dopaminergic Activity in Tourette Syndrome and Obsessive-Compulsive Disorder. Eur. Neuropsychopharmacol. 2013, 23, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Keeler, J.F.; Pretsell, D.O.; Robbins, T.W. Functional Implications of Dopamine D1 vs. D2 Receptors: A “prepare and Select” Model of the Striatal Direct vs. Indirect Pathways. Neuroscience 2014, 282, 156–175. [Google Scholar] [CrossRef]
- Lanciego, J.L.; Luquin, N.; Obeso, J.A. Functional Neuroanatomy of the Basal Ganglia. Cold Spring Harb. Perspect. Med. 2012, 2, 21. [Google Scholar] [CrossRef]
- DeLong, M.R.; Wichmann, T. Circuits and Circuit Disorders of the Basal Ganglia. Arch. Neurol. 2007, 64, 20–24. [Google Scholar] [CrossRef]
- Masellis, M.; Collinson, S.; Freeman, N.; Tampakeras, M.; Levy, J.; Tchelet, A.; Eyal, E.; Berkovich, E.; Eliaz, R.E.; Abler, V.; et al. Dopamine D2 Receptor Gene Variants and Response to Rasagiline in Early Parkinson’s Disease: A Pharmacogenetic Study. Brain 2016, 139, 2050–2062. [Google Scholar] [CrossRef] [PubMed]
- Sanwald, S.; Montag, C.; Kiefer, M. Cumulative Genetic Score of DRD2 Polymorphisms Is Associated with Impulsivity and Masked Semantic Priming. J. Mol. Neurosci. 2022, 72, 1682–1694. [Google Scholar] [CrossRef]
- Yip, S.W.; Potenza, M.N. Application of Research Domain Criteria to Childhood and Adolescent Impulsive and Addictive Disorders: Implications for Treatment. Clin. Psychol. Rev. 2018, 64, 41–56. [Google Scholar] [CrossRef]
- Pruessner, J.C.; Champagne, F.; Meaney, M.J.; Dagher, A. Dopamine Release in Response to a Psychological Stress in Humans and Its Relationship to Early Life Maternal Care: A Positron Emission Tomography Study Using [11C]Raclopride. J. Neurosci. 2004, 24, 2825. [Google Scholar] [CrossRef]
- Lamanna, J.; Isotti, F.; Ferro, M.; Racchetti, G.; Anchora, L.; Rucco, D.; Malgaroli, A. Facilitation of Dopamine-Dependent Long-Term Potentiation in the Medial Prefrontal Cortex of Male Rats Follows the Behavioral Effects of Stress. J. Neurosci. Res. 2021, 99, 662–678. [Google Scholar] [CrossRef] [PubMed]
- Pani, L.; Porcella, A.; Gessa, G.L. The Role of Stress in the Pathophysiology of the Dopaminergic System. Mol. Psychiatry 2000, 5, 14–21. [Google Scholar] [CrossRef]
- Lataster, J.; Collip, D.; Ceccarini, J.; Haas, D.; Booij, L.; van Os, J.; Pruessner, J.; Van Laere, K.; Myin-Germeys, I. Psychosocial Stress Is Associated with in Vivo Dopamine Release in Human Ventromedial Prefrontal Cortex: A Positron Emission Tomography Study Using [18F]Fallypride. Neuroimage 2011, 58, 1081–1089. [Google Scholar] [CrossRef]
- Rice, M.E.; Patel, J.C.; Cragg, S.J. Dopamine Release in the Basal Ganglia. Neuroscience 2011, 198, 112–137. [Google Scholar] [CrossRef] [PubMed]
- Roessner, V.; Plessen, K.J.; Rothenberger, A.; Ludolph, A.G.; Rizzo, R.; Skov, L.; Strand, G.; Stern, J.S.; Termine, C.; Hoekstra, P.J.; et al. European Clinical Guidelines for Tourette Syndrome and Other Tic Disorders. Part II: Pharmacological Treatment. Eur. Child Adolesc. Psychiatry 2011, 20, 173–196. [Google Scholar] [CrossRef]
- Müller-Vahl, K.R.; Grosskreutz, J.; Prell, T.; Kaufmann, J.; Bodammer, N.; Peschel, T. Tics Are Caused by Alterations in Prefrontal Areas, Thalamus and Putamen, While Changes in the Cingulate Gyrus Reflect Secondary Compensatory Mechanisms. BMC Neurosci. 2014, 15, 6. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, K.J.; Schooler, C.; Schoenbach, C.; Herscovitch, P.; Chase, T.N.; Braun, A.R. The Functional Neuroanatomy of Tourette’s Syndrome: An FDG PET Study III: Functional Coupling of Regional Cerebral Metabolic Rates. Neuropsychopharmacology 2002, 27, 92–104. [Google Scholar] [CrossRef]
- Polyanska, L.; Critchley, H.D.; Rae, C.L. Centrality of Prefrontal and Motor Preparation Cortices to Tourette Syndrome Revealed by Meta-Analysis of Task-Based Neuroimaging Studies. NeuroImage Clin. 2017, 16, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Prado, H.D.S.; Do Rosário, M.C.; Lee, J.; Hounie, A.G.; Shavitt, R.G.; Miguel, E.C. Sensory Phenomena in Obsessive-Compulsive Disorder and Tic Disorders: A Review of the Literature. CNS Spectr. 2008, 13, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Bohlhalter, S.; Goldfine, A.; Matteson, S.; Garraux, G.; Hanakawa, T.; Kansaku, K.; Wurzman, R.; Hallett, M. Neural Correlates of Tic Generation in Tourette Syndrome: An Event-Related Functional MRI Study. Brain 2006, 129, 2029–2037. [Google Scholar] [CrossRef]
- Heise, K.F.; Steven, B.; Liuzzi, G.; Thomalla, G.; Jonas, M.; Müller-Vahl, K.; Sauseng, P.; Münchau, A.; Gerloff, C.; Hummel, F.C. Altered Modulation of Intracortical Excitability during Movement Preparation in Gilles de La Tourette Syndrome. Brain 2010, 133, 580–590. [Google Scholar] [CrossRef]
- Zapparoli, L.; Tettamanti, M.; Porta, M.; Zerbi, A.; Servello, D.; Banfi, G.; Paulesu, E. A Tug of War: Antagonistic Effective Connectivity Patterns over the Motor Cortex and the Severity of Motor Symptoms in Gilles de La Tourette Syndrome. Eur. J. Neurosci. 2017, 46, 2203–2213. [Google Scholar] [CrossRef]
- Chappell, P.; Riddle, M.; Anderson, G.; Scahill, L.; Hardin, M.; Walker, D.; Cohen, D.; Leckman, J. Enhanced Stress Responsivity of Tourette Syndrome Patients Undergoing Lumbar Puncture. Biol. Psychiatry 1994, 36, 35–43. [Google Scholar] [CrossRef]
- Pourfar, M.; Feigin, A.; Tang, C.C.; Carbon-Correll, M.; Bussa, M.; Budman, C.; Dhawan, V.; Eidelberg, D. Abnormal Metabolic Brain Networks in Tourette Syndrome. Neurology 2011, 76, 944–952. [Google Scholar] [CrossRef]
- Rotge, J.Y.; Guehl, D.; Dilharreguy, B.; Cuny, E.; Tignol, J.; Bioulac, B.; Allard, M.; Burbaud, P.; Aouizerate, B. Provocation of Obsessive–Compulsive Symptoms: A Quantitative Voxel-Based Meta-Analysis of Functional Neuroimaging Studies. J. Psychiatry Neurosci. 2008, 33, 405. [Google Scholar]
- Wang, Z.; Maia, T.V.; Marsh, R.; Colibazzi, T.; Gerber, A.; Peterson, B.S. The Neural Circuits That Generate Tics in Tourette’s Syndrome. Am. J. Psychiatry 2011, 168, 1326–1337. [Google Scholar] [CrossRef] [PubMed]
- Sigurdsson, H.P.; Jackson, S.R.; Jolley, L.; Mitchell, E.; Jackson, G.M. Alterations in Cerebellar Grey Matter Structure and Covariance Networks in Young People with Tourette Syndrome. Cortex 2020, 126, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bostan, A.C.; Strick, P.L. The Cerebellum and Basal Ganglia Are Interconnected. Neuropsychol. Rev. 2010, 20, 261–270. [Google Scholar] [CrossRef]
- Jackson, S.R.; Parkinson, A.; Kim, S.Y.; Schüermann, M.; Eickhoff, S.B. On the Functional Anatomy of the Urge-for-Action. Cogn. Neurosci. 2011, 2, 227–243. [Google Scholar] [CrossRef] [PubMed]
- Berry, K.P.; Nedivi, E. Spine Dynamics: Are They All the Same? Neuron 2017, 96, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Suppa, A.; Marsili, L.; Di Stasio, F.; Berardelli, I.; Roselli, V.; Pasquini, M.; Cardona, F.; Berardelli, A. Cortical and Brainstem Plasticity in Tourette Syndrome and Obsessive-Compulsive Disorder. Mov. Disord. 2014, 29, 1523–1531. [Google Scholar] [CrossRef]
- Tübing, J.; Gigla, B.; Brandt, V.C.; Verrel, J.; Weissbach, A.; Beste, C.; Münchau, A.; Bäumer, T. Associative Plasticity in Supplementary Motor Area—Motor Cortex Pathways in Tourette Syndrome. Sci. Rep. 2018, 8, 11984. [Google Scholar] [CrossRef]
- Ferro, M.; Lamanna, J.; Spadini, S.; Nespoli, A.; Sulpizio, S.; Malgaroli, A. Synaptic Plasticity Mechanisms behind TMS Efficacy: Insights from Its Application to Animal Models. J. Neural Transm. 2022, 129, 25–36. [Google Scholar] [CrossRef]
- Suppa, A.; Huang, Y.Z.; Funke, K.; Ridding, M.C.; Cheeran, B.; Di Lazzaro, V.; Ziemann, U.; Rothwell, J.C. Ten Years of Theta Burst Stimulation in Humans: Established Knowledge, Unknowns and Prospects. Brain Stimul. 2016, 9, 323–335. [Google Scholar] [CrossRef]
- Wu, S.W.; Gilbert, D.L. Altered Neurophysiologic Response to Intermittent Theta Burst Stimulation in Tourette Syndrome. Brain Stimul. 2012, 5, 315–319. [Google Scholar] [CrossRef]
- Brandt, V.C.; Niessen, E.; Ganos, C.; Kahl, U.; Bäumer, T.; Münchau, A. Altered Synaptic Plasticity in Tourette’s Syndrome and Its Relationship to Motor Skill Learning. PLoS ONE 2014, 9, e98417. [Google Scholar] [CrossRef]
- Suppa, A.; Belvisi, D.; Bologna, M.; Marsili, L.; Berardelli, I.; Moretti, G.; Pasquini, M.; Fabbrini, G.; Berardelli, A. Abnormal Cortical and Brain Stem Plasticity in Gilles de La Tourette Syndrome. Mov. Disord. 2011, 26, 1703–1710. [Google Scholar] [CrossRef]
- Martín-Rodríguez, J.F.; Ruiz-Rodríguez, M.A.; Palomar, F.J.; Cáceres-Redondo, M.T.; Vargas, L.; Porcacchia, P.; Gómez-Crespo, M.; Huertas-Fernández, I.; Carrillo, F.; Madruga-Garrido, M.; et al. Aberrant Cortical Associative Plasticity Associated with Severe Adult Tourette Syndrome. Mov. Disord. 2015, 30, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Stefan, K. Induction of Plasticity in the Human Motor Cortex by Paired Associative Stimulation. Brain 2000, 123, 572–584. [Google Scholar] [CrossRef]
- Piacentini, J.; Woods, D.W.; Scahill, L.; Wilhelm, S.; Peterson, A.L.; Chang, S.; Ginsburg, G.S.; Deckersbach, T.; Dziura, J.; Levi-Pearl, S.; et al. Behavior Therapy for Children With Tourette Disorder. JAMA 2010, 303, 1929. [Google Scholar] [CrossRef] [PubMed]
- Eichele, H.; Plessen, K.J. Neural Plasticity in Functional and Anatomical MRI Studies of Children with Tourette Syndrome. Behav. Neurol. 2013, 27, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Marsili, L.; Suppa, A.; Di Stasio, F.; Belvisi, D.; Upadhyay, N.; Berardelli, I.; Pasquini, M.; Petrucci, S.; Ginevrino, M.; Fabbrini, G.; et al. BDNF and LTP-/LTD-like Plasticity of the Primary Motor Cortex in Gilles de La Tourette Syndrome. Exp. Brain Res. 2017, 235, 841–850. [Google Scholar] [CrossRef]
- Galea, J.M.; Jayaram, G.; Ajagbe, L.; Celnik, P. Modulation of Cerebellar Excitability by Polarity-Specific Noninvasive Direct Current Stimulation. J. Neurosci. 2009, 29, 9115–9122. [Google Scholar] [CrossRef]
- Cooper, I.S.; Upton, A.R.M.; Amin, I. Chronic Cerebellar Stimulation (CCS) and Deep Brain Stimulation (DBS) in Involuntary Movement Disorders. Appl. Neurophysiol. 1982, 45, 209–217. [Google Scholar] [CrossRef]
- Cooper, I.S.; Upton, A.R.M.; Amin, I. Reversibility of Chronic Neurologic Deficits. Some Effects of Electrical Stimulation of the Thalamus and Internal Capsule in Man. Appl. Neurophysiol. 1980, 43, 244–258. [Google Scholar] [CrossRef]
- Cannon, E.; Silburn, P.; Coyne, T.; O’Maley, K.; Crawford, J.D.; Sachdev, P.S. Deep Brain Stimulation of Anteromedial Globus Pallidus Interna for Severe Tourette’s Syndrome. Am. J. Psychiatry 2012, 169, 860–866. [Google Scholar] [CrossRef]
- Houeto, J.L.; Karachi, C.; Mallet, L.; Pillon, B.; Yelnik, J.; Mesnage, V.; Welter, M.L.; Navarro, S.; Pelissolo, A.; Damier, P.; et al. Tourette’s Syndrome and Deep Brain Stimulation. J. Neurol. Neurosurg. Psychiatry 2005, 76, 992–995. [Google Scholar] [CrossRef]
- Dow-Edwards, D.; MacMaster, F.P.; Peterson, B.S.; Niesink, R.; Andersen, S.; Braams, B.R. Experience during Adolescence Shapes Brain Development: From Synapses and Networks to Normal and Pathological Behavior. Neurotoxicol. Teratol. 2019, 76, 106834. [Google Scholar] [CrossRef]
- Johnson, K.A.; Duffley, G.; Foltynie, T.; Hariz, M.; Zrinzo, L.; Joyce, E.M.; Akram, H.; Servello, D.; Galbiati, T.F.; Bona, A.; et al. Basal Ganglia Pathways Associated With Therapeutic Pallidal Deep Brain Stimulation for Tourette Syndrome. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, 6, 961–972. [Google Scholar] [CrossRef]
- McNaught, K.S.P.; Mink, J.W. Advances in Understanding and Treatment of Tourette Syndrome. Nat. Rev. Neurol. 2011, 7, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Kalanithi, P.S.A.; Zheng, W.; Kataoka, Y.; DiFiglia, M.; Grantz, H.; Saper, C.B.; Schwartz, M.L.; Leckman, J.F.; Vaccarino, F.M. Altered Parvalbumin-Positive Neuron Distribution in Basal Ganglia of Individuals with Tourette Syndrome. Proc. Natl. Acad. Sci. USA 2005, 102, 13307–13312. [Google Scholar] [CrossRef]
- Kataoka, Y.; Kalanithi, P.S.A.; Grantz, H.; Schwartz, M.L.; Saper, C.; Leckman, J.F.; Vaccarino, F.M. Decreased Number of Parvalbumin and Cholinergic Interneurons in the Striatum of Individuals with Tourette Syndrome. J. Comp. Neurol. 2010, 518, 277–291. [Google Scholar] [CrossRef]
- Owen, S.F.; Berke, J.D.; Kreitzer, A.C. Fast-Spiking Interneurons Supply Feedforward Control of Bursting, Calcium, and Plasticity for Efficient Learning. Cell 2018, 172, 683–695.e15. [Google Scholar] [CrossRef]
- Kreitzer, A.C.; Malenka, R.C. Striatal Plasticity and Basal Ganglia Circuit Function. Neuron 2008, 60, 543–554. [Google Scholar]
- Calabresi, P.; Saiardi, A.; Pisani, A.; Baik, J.H.; Centonze, D.; Mercuri, N.B.; Bernardi, G.; Borrelli, E.; Vergata, T.; Sanita, D.; et al. Abnormal Synaptic Plasticity in the Striatum of Mice Lacking Dopamine D2 Receptors. J. Neurosci. 1997, 17, 4536–4544. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Flajolet, M.; Greengard, P.; Surmeier, D.J. Dichotomous Control Dopaminergic of Striatal Synaptic Plasticity. Science 2008, 321, 848–851. [Google Scholar] [CrossRef] [PubMed]
- Kreitzer, A.C.; Malenka, R.C. Endocannabinoid-Mediated Rescue of Striatal LTD and Motor Deficits in Parkinson’s Disease Models. Nature 2007, 445, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Augustin, S.M.; Chancey, J.H.; Lovinger, D.M. Dual Dopaminergic Regulation of Corticostriatal Plasticity by Cholinergic Interneurons and Indirect Pathway Medium Spiny Neurons. Cell Rep. 2018, 24, 2883–2893. [Google Scholar] [CrossRef]
- Tang, K.-C. Dopamine-Dependent Synaptic Plasticity in Striatum during in Vivo Development. Proc. Natl. Acad. Sci. USA 2001, 98, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Partridge, J.G.; Tang, K.C.; Lovinger, D.M. Regional and Postnatal Heterogeneity of Activity-Dependent Long-Term Changes in Synaptic Efficacy in the Dorsal Striatum. J. Neurophysiol. 2000, 84, 1422–1429. [Google Scholar] [CrossRef]
- Shindou, T.; Shindou, M.; Watanabe, S.; Wickens, J. A Silent Eligibility Trace Enables Dopamine-Dependent Synaptic Plasticity for Reinforcement Learning in the Mouse Striatum. Eur. J. Neurosci. 2018, 49, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Giordano, N.; Iemolo, A.; Mancini, M.; Cacace, F.; De Risi, M.; Latagliata, E.C.; Ghiglieri, V.; Bellenchi, G.C.; Puglisi-Allegra, S.; Calabresi, P.; et al. Motor Learning and Metaplasticity in Striatal Neurons: Relevance for Parkinson’s Disease. Brain 2018, 141, 505–520. [Google Scholar] [CrossRef]
- Gremel, C.M.; Chancey, J.H.; Atwood, B.K.; Luo, G.; Neve, R.; Ramakrishnan, C.; Deisseroth, K.; Lovinger, D.M.; Costa, R.M. Endocannabinoid Modulation of Orbitostriatal Circuits Gates Habit Formation. Neuron 2016, 90, 1312–1324. [Google Scholar] [CrossRef]
- Lim, K.; See, Y.M.; Lee, J. A Systematic Review of the Effectiveness of Medical Cannabis for Psychiatric, Movement and Neurodegenerative Disorders. Clin. Psychopharmacol. Neurosci. 2017, 15, 301. [Google Scholar] [CrossRef]
- Scholl, C.; Baladron, J.; Vitay, J.; Hamker, F.H. Enhanced Habit Formation in Tourette Patients Explained by Shortcut Modulation in a Hierarchical Cortico-Basal Ganglia Model. Brain Struct. Funct. 2022, 227, 1031–1050. [Google Scholar] [CrossRef]
- Kozorovitskiy, Y.; Saunders, A.; Johnson, C.A.; Lowell, B.B.; Sabatini, B.L. Recurrent Network Activity Drives Striatal Synaptogenesis. Nature 2012, 485, 646–650. [Google Scholar] [CrossRef]
- Martino, D.; Dale, R.C.; Gilbert, D.L.; Giovannoni, G.; Leckamn, J.F. Immunopathogenic Mechanismsin Tourette Syndrome: A Critical Review. Mov. Disord. 2009, 24, 1267–1279. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Yang, H.; Li, Y.; Gui, J.; Cui, Y. Profiles of Proinflammatory Cytokines and T Cells in Patients With Tourette Syndrome: A Meta-Analysis. Front. Immunol. 2022, 13, 843247. [Google Scholar] [CrossRef]
- Hoffman, K.L.; Hornig, M.; Yaddanapudi, K.; Jabado, O.; Lipkin, W.I. A Murine Model for Neuropsychiatric Disorders Associated with Group A β-Hemolytic Streptococcal Infection. J. Neurosci. 2004, 24, 1780–1791. [Google Scholar] [CrossRef]
- Zhang, D.; Patel, A.; Zhu, Y.; Siegel, A.; Zalcman, S.S. Anti-Streptococcus IgM Antibodies Induce Repetitive Stereotyped Movements: Cell Activation and Co-Localization with Fcα/μ Receptors in the Striatum and Motor Cortex. Brain Behav. Immun. 2012, 26, 521–533. [Google Scholar] [CrossRef][Green Version]
- Hallett, J.J.; Harling-Berg, C.J.; Knopf, P.M.; Stopa, E.G.; Kiessling, L.S. Anti-Striatal Antibodies in Tourette Syndrome Cause Neuronal Dysfunction. J. Neuroimmunol. 2000, 111, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Y.; Li, D.; Ju, X. Transplantation of Rat Neural Stem Cells Reduces Stereotypic Behaviors in Rats after Intrastriatal Microinfusion of Tourette Syndrome Sera. Behav. Brain Res. 2008, 186, 84–90. [Google Scholar] [CrossRef]
- Loiselle, C.R.; Lee, O.; Moran, T.H.; Singer, H.S. Striatal Microinfusion of Tourette Syndrome and PANDAS Sera: Failure to Induce Behavioral Changes. Mov. Disord. 2004, 19, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Yaddanapudi, K.; Hornig, M.; Serge, R.; De Miranda, J.; Baghban, A.; Villar, G.; Lipkin, W.I. Passive Transfer of Streptococcus-Induced Antibodies Reproduces Behavioral Disturbances in a Mouse Model of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infection. Mol. Psychiatry 2010, 15, 712–726. [Google Scholar] [CrossRef]
- Xu, J.; Liu, R.J.; Fahey, S.; Frick, L.; Leckman, J.; Vaccarino, F.; Duman, R.S.; Williams, K.; Swedo, S.; Pittenger, C. Antibodies from Children with PANDAS Bind Specifically to Striatal Cholinergic Interneurons and Alter Their Activity. Am. J. Psychiatry 2021, 178, 48–64. [Google Scholar] [CrossRef]
- Yeh, C.B.; Shui, H.A.; Chu, T.H.; Chen, Y.A.; Tsung, H.C.; Shyu, J.F. Hyperpolarisation-Activated Cyclic Nucleotide Channel 4 (HCN4) Involvement in Tourette’s Syndrome Autoimmunity. J. Neuroimmunol. 2012, 250, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.J.; Ma, T.C.; Ding, Y.; Cheung, T.; Joshi, N.; Sulzer, D.; Mosharov, E.V.; Kang, U.J. Alterations in the Intrinsic Properties of Striatal Cholinergic Interneurons after Dopamine Lesion and Chronic L-Dopa. eLife 2020, 9, e56920. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, K.; Liu, X.; Yan, H.; Ma, X.; Zhang, S.; Zheng, J.; Wang, L.; Wei, X. Involvement of HCN Channel in Muscarinic Inhibitory Action on Tonic Firing of Dorsolateral Striatal Cholinergic Interneurons. Front. Cell. Neurosci. 2016, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Dalmau, J.; Gleichman, A.J.; Hughes, E.G.; Rossi, J.E.; Peng, X.; Lai, M.; Dessain, S.K.; Rosenfeld, M.R.; Balice-Gordon, R.; Lynch, D.R. Anti-NMDA-Receptor Encephalitis: Case Series and Analysis of the Effects of Antibodies. Lancet Neurol. 2008, 7, 1091–1098. [Google Scholar] [CrossRef]
- Masdeu, J.C.; Dalmau, J.; Berman, K.F. NMDA Receptor Internalization by Autoantibodies: A Reversible Mechanism Underlying Psychosis? Trends Neurosci. 2016, 39, 300–310. [Google Scholar] [CrossRef]
- Boulanger, L.M.; Shatz, C.J. Immune Signalling in Neural Development, Synaptic Plasticity and Disease. Nat. Rev. Neurosci. 2004, 5, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Dissing-Olesen, L.; MacVicar, B.A.; Stevens, B. Microglia: Dynamic Mediators of Synapse Development and Plasticity. Trends Immunol. 2015, 36, 605–613. [Google Scholar] [CrossRef]
- Mancini, A.; Ghiglieri, V.; Parnetti, L.; Calabresi, P.; Di Filippo, M. Neuro-Immune Cross-Talk in the Striatum: From Basal Ganglia Physiology to Circuit Dysfunction. Front. Immunol. 2021, 12, 644294. [Google Scholar] [CrossRef]
- Baym, C.L.; Corbett, B.A.; Wright, S.B.; Bunge, S.A. Neural Correlates of Tic Severity and Cognitive Control in Children with Tourette Syndrome. Brain 2008, 131, 165–179. [Google Scholar] [CrossRef]
- Tikoo, S.; Cardona, F.; Tommasin, S.; Giannì, C.; Conte, G.; Upadhyay, N.; Mirabella, G.; Suppa, A.; Pantano, P. Resting-State Functional Connectivity in Drug-Naive Pediatric Patients with Tourette Syndrome and Obsessive-Compulsive Disorder. J. Psychiatr. Res. 2020, 129, 129–140. [Google Scholar] [CrossRef]
- Openneer, T.J.C.; Marsman, J.B.C.; van der Meer, D.; Forde, N.J.; Akkermans, S.E.A.; Naaijen, J.; Buitelaar, J.K.; Dietrich, A.; Hoekstra, P.J. A Graph Theory Study of Resting-State Functional Connectivity in Children with Tourette Syndrome. Cortex 2020, 126, 63–72. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamanna, J.; Ferro, M.; Spadini, S.; Racchetti, G.; Malgaroli, A. The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome. Behav. Sci. 2023, 13, 668. https://doi.org/10.3390/bs13080668
Lamanna J, Ferro M, Spadini S, Racchetti G, Malgaroli A. The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome. Behavioral Sciences. 2023; 13(8):668. https://doi.org/10.3390/bs13080668
Chicago/Turabian StyleLamanna, Jacopo, Mattia Ferro, Sara Spadini, Gabriella Racchetti, and Antonio Malgaroli. 2023. "The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome" Behavioral Sciences 13, no. 8: 668. https://doi.org/10.3390/bs13080668
APA StyleLamanna, J., Ferro, M., Spadini, S., Racchetti, G., & Malgaroli, A. (2023). The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome. Behavioral Sciences, 13(8), 668. https://doi.org/10.3390/bs13080668





