Association between Endocrine Therapy and Weight Gain after Breast Cancer Diagnosis among Japanese Patients: A Retrospective Cohort Study
Abstract
:1. Introduction
2. Patients and Methods
2.1. Study Design and Setting
2.2. Eligibility Criteria
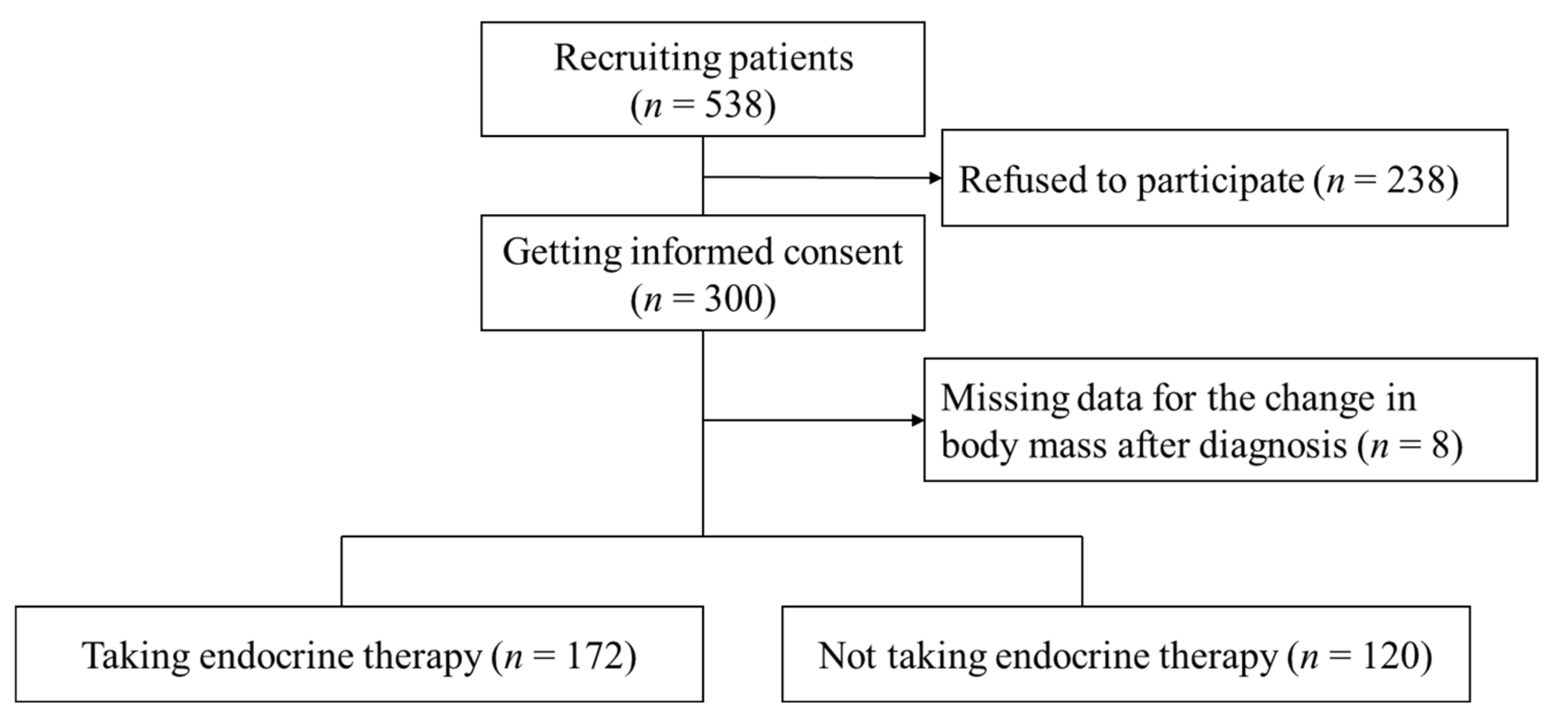
2.3. Study Process
2.4. Measurements
2.4.1. Primary Outcome
2.4.2. Anthropometric and Medical History Measurements
2.4.3. Sociodemographic and Lifestyle Variables
2.4.4. Physical Activity
2.4.5. Dietary Intake
2.4.6. Sample Size
2.4.7. Statistical Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global Cancer Statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Latest Information about Cancer Statistic. National Cancer Center for Cancer Control and Information Services. Available online: https://ganjoho.jp/reg_stat/statistics/stat/summary.html (accessed on 2 November 2020).
- Wong, I.O.; Schooling, C.M.; Cowling, B.J.; Leung, G.M. Breast cancer incidence and mortality in a transitioning Chinese population: Current and future trends. Br. J. Cancer 2015, 112, 167–170. [Google Scholar] [CrossRef] [Green Version]
- Demark-Wahnefried, W.; Campbell, K.L.; Hayes, S.C. Weight management and its role in breast cancer rehabilitation. Cancer 2012, 118 (Suppl. 8), 2277–2287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irwin, M.L.; McTiernan, A.; Baumgartner, R.N.; Baumgartner, K.B.; Bernstein, L.; Gilliland, F.D.; Ballard-Barbash, R. Changes in body fat and weight after a breast cancer diagnosis: Influence of demographic, prognostic, and lifestyle factors. J. Clin. Oncol. 2005, 23, 774–782. [Google Scholar] [CrossRef]
- Rock, C.L.; Demark-Wahnefried, W. Nutrition and survival after the diagnosis of breast cancer: A review of the evidence. J. Clin. Oncol. 2002, 20, 3302–3316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kroenke, C.H.; Chen, W.Y.; Rosner, B.; Holmes, M.D. Weight, weight gain, and survival after breast cancer diagnosis. J. Clin. Oncol. 2005, 23, 1370–1378. [Google Scholar] [CrossRef]
- Gibb, F.W.; Dixon, J.M.; Clarke, C.; Homer, N.Z.; Faqehi, A.M.M.; Andrew, R.; Walker, B.R. Higher Insulin Resistance and Adiposity in Postmenopausal Women with Breast Cancer Treated with Aromatase Inhibitors. J. Clin. Endocrinol. Metab. 2019, 104, 3670–3678. [Google Scholar] [CrossRef]
- Nestoriuc, Y.; von Blanckenburg, P.; Schuricht, F.; Barsky, A.J.; Hadji, P.; Albert, U.S.; Rief, W. Is it best to expect the worst? Influence of patients’ side-effect expectations on endocrine treatment outcome in a 2-year prospective clinical cohort study. Ann. Oncol. 2016, 27, 1909–1915. [Google Scholar] [CrossRef]
- Nyrop, K.A.; Deal, A.M.; Lee, J.T.; Muss, H.B.; Choi, S.K.; Dixon, S.; Wheless, A.; Carey, L.A.; Shachar, S.S. Weight changes in postmenopausal breast cancer survivors over 2 years of endocrine therapy: A retrospective chart review. Breast Cancer Res. Treat. 2017, 162, 375–388. [Google Scholar] [CrossRef]
- Brown, J.C.; Mao, J.J.; Stricker, C.; Hwang, W.T.; Tan, K.S.; Schmitz, K.H. Aromatase inhibitor associated musculoskeletal symptoms are associated with reduced physical activity among breast cancer survivors. Breast J. 2014, 20, 22–28. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Lu, W.; Gu, K.; Chen, Z.; Zheng, Y.; Zheng, W.; Shu, X.O. Weight change and its correlates among breast cancer survivors. Nutr. Cancer 2011, 63, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Shin, W.K.; Song, S.; Jung, S.Y.; Lee, E.; Kim, Z.; Moon, H.G.; Noh, D.Y.; Lee, J.E. The association between physical activity and health-related quality of life among breast cancer survivors. Health Qual. Life Outcomes 2017, 15, 132. [Google Scholar] [CrossRef] [Green Version]
- Irwin, M.L.; Crumley, D.; McTiernan, A.; Bernstein, L.; Baumgartner, R.; Gilliland, F.D.; Kriska, A.; Ballard-Barbash, R. Physical activity levels before and after a diagnosis of breast carcinoma: The Health, Eating, Activity, and Lifestyle (HEAL) study. Cancer 2003, 97, 1746–1757. [Google Scholar] [CrossRef] [Green Version]
- Kang, K.D.; Bae, S.; Kim, H.J.; Hwang, I.G.; Kim, S.M.; Han, D.H. The Relationship between Physical Activity Intensity and Mental Health Status in Patients with Breast Cancer. J. Korean Med. Sci. 2017, 32, 1345–1350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuoka, Y.; Haskell, W.; Vittinghoff, E. New insights into discrepancies between self-reported and accelerometer-measured moderate to vigorous physical activity among women—The mPED trial. BMC Public Health 2016, 16, 761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prince, S.A.; Adamo, K.B.; Hamel, M.E.; Hardt, J.; Connor Gorber, S.; Tremblay, M. A comparison of direct versus self-report measures for assessing physical activity in adults: A systematic review. Int. J. Behav. Nutr. Phys. Acta 2008, 5, 56. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Comparison of relative validity of food group intakes estimated by comprehensive and brief-type self-administered diet history questionnaires against 16 d dietary records in Japanese adults. Public Health Nutr. 2011, 14, 1200–1211. [Google Scholar] [CrossRef]
- National Cancer Institute. Diet History Questionnaire. Available online: https://epi.grants.cancer.gov/dhq3/dhq3-past-month-with-serving-sizes-questionnaire.pdf (accessed on 2 November 2020).
- Velentzis, L.S.; Keshtgar, M.R.; Woodside, J.V.; Leathem, A.J.; Titcomb, A.; Perkins, K.A.; Mazurowska, M.; Anderson, V.; Wardell, K.; Cantwell, M.M. Significant changes in dietary intake and supplement use after breast cancer diagnosis in a UK multicentre study. Breast Cancer Res. Treat. 2011, 128, 473–482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The EPIC-Norfolk Food Frequency Questionnaire and FETA Software. The EPIC-Norfolk Study. Available online: http://www.srl.cam.ac.uk/epic/epicffq/index.shtml (accessed on 2 November 2020).
- Oshima, Y.; Kawaguchi, K.; Tanaka, S.; Ohkawara, K.; Hikihara, Y.; Ishikawa-Takata, K.; Tabata, I. Classifying household and locomotive activities using a triaxial accelerometer. Gait Posture 2010, 31, 370–374. [Google Scholar] [CrossRef]
- Cleland, C.L.; Hunter, R.F.; Kee, F.; Cupples, M.E.; Sallis, J.F.; Tully, M.A. Validity of the global physical activity questionnaire (GPAQ) in assessing levels and change in moderate-vigorous physical activity and sedentary behaviour. BMC Public Health 2014, 14, 1255. [Google Scholar] [CrossRef] [Green Version]
- Ohkawara, K.; Oshima, Y.; Hikihara, Y.; Ishikawa-Takata, K.; Tabata, I.; Tanaka, S. Real-time estimation of daily physical activity intensity by a triaxial accelerometer and a gravity-removal classification algorithm. Br. J. Nutr. 2011, 105, 1681–1691. [Google Scholar] [CrossRef]
- Shiraishi, M.; Haruna, M.; Matsuzaki, M.; Murayama, R.; Sasaki, S. The biomarker-based validity of a brief-type diet history questionnaire for estimating eicosapentaenoic acid and docosahexaenoic acid intakes in pregnant Japanese women. Asia Pac. J. Clin. Nutr. 2015, 24, 316–322. [Google Scholar]
- The National Health and Nutrition Survey in Japan. National Institute of Health and Nutrition. Available online: http://www.mhlw.go.jp/stf/houdou/0000177189.html (accessed on 2 November 2020).
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Heideman, W.H.; Russell, N.S.; Gundy, C.; Rookus, M.A.; Voskuil, D.W. The frequency, magnitude and timing of post-diagnosis body weight gain in Dutch breast cancer survivors. Eur. J. Cancer 2009, 45, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Kurebayashi, J.; Miyoshi, Y.; Ishikawa, T.; Saji, S.; Sugie, T.; Suzuki, T.; Takahashi, S.; Nozaki, M.; Yamashita, H.; Tokuda, Y.; et al. Clinicopathological characteristics of breast cancer and trends in the management of breast cancer patients in Japan: Based on the Breast Cancer Registry of the Japanese Breast Cancer Society between 2004 and 2011. Breast Cancer 2015, 22, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Caan, B.J.; Kwan, M.L.; Hartzell, G.; Castillo, A.; Slattery, M.L.; Sternfeld, B.; Weltzien, E. Pre-diagnosis body mass index, post-diagnosis weight change, and prognosis among women with early stage breast cancer. Cancer Causes Control 2008, 19, 1319–1328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caan, B.J.; Kwan, M.L.; Shu, X.O.; Pierce, J.P.; Patterson, R.E.; Nechuta, S.J.; Poole, E.M.; Kroenke, C.H.; Weltzien, E.K.; Flatt, S.W.; et al. Weight change and survival after breast cancer in the after breast cancer pooling project. Cancer Epidemiol. Biomark. Prev. 2012, 21, 1260–1271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mortimer, J.; Behrendt, C.E. Severe menopausal symptoms are widespread among survivors of breast cancer treatment regardless of time since diagnosis. J. Palliat. Med. 2013, 16, 1130–1134. [Google Scholar] [CrossRef]
- Kim, S.H.; Cho, Y.U.; Kim, S.J. Weight Gain and its Correlates among Breast Cancer Survivors. Asian Nurs. Res. 2013, 7, 161–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, K.; Chen, X.; Zheng, Y.; Chen, Z.; Zheng, W.; Lu, W.; Shu, X.O. Weight change patterns among breast cancer survivors: Results from the Shanghai breast cancer survival study. Cancer Causes Control 2010, 21, 621–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, B.M.; Dunstan, D.W.; Healy, G.N.; Winkler, E.; Eakin, E.; Owen, N. Objectively measured physical activity and sedentary time of breast cancer survivors, and associations with adiposity: Findings from NHANES (2003–2006). Cancer Causes Control 2010, 21, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Yaw, Y.H.; Shariff, Z.M.; Kandiah, M.; Weay, Y.H.; Saibul, N.; Sariman, S.; Hashim, Z. Diet and physical activity in relation to weight change among breast cancer patients. Asian Pac. J. Cancer Prev. 2014, 15, 39–44. [Google Scholar] [CrossRef] [Green Version]
- Rock, C.L.; Flatt, S.W.; Newman, V.; Caan, B.J.; Haan, M.N.; Stefanick, M.L.; Faerber, S.; Pierce, J.P. Factors associated with weight gain in women after diagnosis of breast cancer. Women’s Healthy Eating and Living Study Group. J. Am. Diet. Assoc. 1999, 99, 1212–1221. [Google Scholar] [CrossRef]
- Lei, Y.Y.; Ho, S.C.; Cheng, A.; Kwok, C.; Cheung, K.L.; He, Y.Q.; Lee, C.I.; Lee, R.; Yeo, W. Dietary changes in the first 3 years after breast cancer diagnosis: A prospective Chinese breast cancer cohort study. Cancer Manag. Res. 2018, 10, 4073–4084. [Google Scholar] [CrossRef] [Green Version]
- Naude, C.E.; Schoonees, A.; Senekal, M.; Young, T.; Garner, P.; Volmink, J. Low carbohydrate versus isoenergetic balanced diets for reducing weight and cardiovascular risk: A systematic review and meta-analysis. PLoS ONE 2014, 9, e100652. [Google Scholar] [CrossRef] [PubMed]
- Vergnaud, A.C.; Norat, T.; Romaguera, D.; Mouw, T.; May, A.M.; Travier, N.; Luan, J.; Wareham, N.; Slimani, N.; Rinaldi, S.; et al. Meat consumption and prospective weight change in participants of the EPIC-PANACEA study. Am. J. Clin. Nutr. 2010, 92, 398–407. [Google Scholar] [CrossRef] [Green Version]
- Thorsdottir, I.; Tomasson, H.; Gunnarsdottir, I.; Gisladottir, E.; Kiely, M.; Parra, M.D.; Bandarra, N.M.; Schaafsma, G.; Martinez, J.A. Randomized trial of weight-loss-diets for young adults varying in fish and fish oil content. Int. J. Obes. 2007, 31, 1560–1566. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Bi, L.F.; Fang, J.H.; Su, X.L.; Da, G.L.; Kuwamori, T.; Kagamimori, S. Beneficial effects of taurine on serum lipids in overweight or obese non-diabetic subjects. Amino Acids 2004, 26, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Hao, T.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Changes in diet and lifestyle and long-term weight gain in women and men. N. Engl. J. Med. 2011, 364, 2392–2404. [Google Scholar] [CrossRef] [Green Version]
- Koo, H.Y.; Seo, Y.G.; Cho, M.H.; Kim, M.J.; Choi, H.C. Weight Change and Associated Factors in Long-Term Breast Cancer Survivors. PLoS ONE 2016, 11, e0159098. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.F. Descriptive epidemiology of body weight and weight change in U.S. adults. Ann. Intern. Med. 1993, 119, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Saelens, B.E. Assessment of physical activity by self-report: Status, limitations, and future directions. Res. Q Exerc. Sport 2000, 71 (Suppl. 2), 1–14. [Google Scholar] [CrossRef] [PubMed]
- Average Income per One Household or One Household Member by Income Quintile Level by Year. Ministry of Health Labour and Welfare of Japan. Available online: https://www.mhlw.go.jp/toukei/saikin/hw/k-tyosa/k-tyosa18/dl/03.pdf (accessed on 2 November 2020).
- Annual Statistics. Basic Survey of Schools in Japan. Portal Site of Official Statistics of Japan. Available online: https://www.e-stat.go.jp/dbview?sid=0003147040 (accessed on 2 November 2020).
- Kuba, S.; Maeda, S.; Matsumoto, M.; Yamanouchi, K.; Yano, H.; Morita, M.; Sakimura, C.; Hatachi, T.; Tokai, Y.; Takatsuki, M.; et al. Adherence to Adjuvant Endocrine Therapy in Women With Breast Cancer: A Prospective Observational Study in Japanese Women. Clin. Breast Cancer 2018, 18, 150–156. [Google Scholar] [CrossRef] [PubMed]
| Variable at Diagnosis | Overall n = 292 | Taking Endocrine Therapy n = 172 | Not Taking Endocrine Therapy n = 120 | p-Value |
|---|---|---|---|---|
| Age at diagnosis (years) | 46.6 ± 6.9 | 47.0 ± 6.3 | 46.2 ± 7.6 | 0.37 |
| Body mass (kg) | 53.7 ± 7.8 | 54.4 ± 8.5 | 52.7 ± 6.5 | 0.049 |
| Height at surgery (cm) | 158.6 ± 5.1 | 158.6 ± 5.0 | 158.6 ± 5.1 | 0.998 |
| Body mass index (kg/m2) | 21.3 ± 2.9 | 21.6 ± 3.1 | 20.9 ± 2.6 | 0.057 |
| Underweight (< 18.5) | 41 (14.0%) | 21 (12.2%) | 20 (16.7%) | 0.20 |
| Normal (18.5 to < 25) | 219 (75.0%) | 128 (74.4%) | 91 (75.8%) | |
| Overweight (≥ 25) | 32 (11.0%) | 23 (13.4%) | 9 (7.5%) | |
| Breast cancer stage (number) | ||||
| 0 | 51 (17.5%) | 2 (1.2%) | 49 (40.8%) | < 0.01 |
| I | 99 (33.9%) | 71 (41.3%) | 28 (23.3%) | |
| II | 115 (39.4%) | 84 (48.8%) | 31 (25.8%) | |
| III | 27 (9.2%) | 15 (8.7%) | 12 (10.0%) | |
| Subtype (number) | ||||
| Luminal | 198 (68.3%) | 150 (88.2%) | 48 (40.0%) | < 0.01 |
| Triple positive | 20 (6.9%) | 18 (10.6%) | 2 (1.7%) | |
| Her2 | 28 (9.7%) | 0 (0%) | 28 (23.3%) | |
| Triple negative | 44 (15.1%) | 2 (1.2%) | 42 (35.0%) | |
| Chemotherapy | ||||
| Yes (number) | 139 (47.6%) | 83 (48.3%) | 56 (46.7%) | 0.79 |
| No (number) | 153 (52.4%) | 89 (51.7%) | 64 (53.3%) | |
| Radiation therapy | ||||
| Yes (number) | 194 (66.4%) | 120 (69.8%) | 74 (61.7%) | 0.15 |
| No (number) | 98 (33.6%) | 52 (30.2%) | 46 (38.3%) | |
| Type of endocrine therapy (number) | ||||
| Tamoxifen | 99 (57.6%) | |||
| Aromatase inhibitor | 30 (17.4%) | |||
| Combined | 43 (25.0%) | |||
| Postmenopausal (number) | 73 (25.0%) | 43 (26.2%) | 30 (25.9%) | 0.95 |
| Variable at Recruitment | Overall n = 292 | Taking Endocrine Therapy n = 172 | Not Taking Endocrine Therapy n = 120 | p-Value |
|---|---|---|---|---|
| Time since diagnosis (year) | 3.0 (3.0–5.0) | 3.0 (3.0–5.0) | 3.0 (2.0–5.0) | 0.17 |
| Body mass (kg) | 55.0 ± 9.1 | 55.7 ± 9.5 | 54.0 ± 8.5 | 0.12 |
| Body mass index, mean (standard deviation) | 21.9 ± 3.5 | 22.1 ± 3.5 | 21.5 ± 3.5 | 0.13 |
| Underweight (< 18.5) | 39 (13.4%) | 18 (10.5%) | 21 (17.5%) | 0.19 |
| Normal (18.5 to < 25) | 205 (70.2%) | 123 (71.5%) | 82 (68.3%) | |
| Overweight (≥ 25) | 48 (16.4%) | 31 (18.0%) | 17 (14.2%) | |
| Weight change from diagnosis (kg) | 1.3 ± 3.9 | 1.3 ± 4.0 | 1.4 ± 3.8 | 0.92 |
| Weight gain more than 5% (number) | 90 (30.8%) | 57 (33.1%) | 33 (27.5%) | 0.31 |
| MVPA using IPAQ (min/week) a | 300.0 (180.0–615.0) | 307.5 (185.0–660.0) | 300.0 (150.0–500.0) | 0.26 |
| Objectively measured MVPA (min/week) b | 462 ± 186 | 466 ± 181 | 456 ± 193 | 0.68 |
| Objectively measured sedentary behavior (min/d) b | 503 ± 109 | 502 ± 110 | 505 ± 107 | 0.83 |
| Dietary intake (kcal) c | 1643 ± 479 | 1645 ± 500 | 1641 ± 450 | 0.94 |
| Fish and shellfish (kcal) c | 124.5 ± 88.2 | 118.6 ± 83.7 | 133.0 ± 93.9 | 0.18 |
| Meat (kcal) c | 138.3 ± 80.9 | 139.1 ± 82.8 | 137.2 ± 78.4 | 0.84 |
| Total vegetables (kcal) c | 72.7 ± 42.5 | 71.8 ± 40.0 | 74.4 ± 46.0 | 0.66 |
| Smoking habit (number) c | ||||
| Never | 211 (75.6%) | 122 (74.4%) | 89 (77.4%) | 0.73 |
| Former | 64 (22.9%) | 39 (25.6%) | 25 (22.6%) | |
| Currently | 4 (1.4%) | 3 (1.8%) | 1 (0.9%) | |
| Education (number) c | ||||
| High school | 35 (12.5%) | 24 (14.6%) | 11 (9.6%) | 0.20 |
| Some college | 117 (41.9%) | 72 (43.9%) | 45 (39.1%) | |
| ≥ University | 127 (45.5%) | 68 (41.5%) | 59 (51.3%) | |
| Postmenopausal (number) c | 197 (70.6%) | 128 (78.0%) | 69 (60.0%) | < 0.01 |
| Household income (number) d | ||||
| < 5 million yen | 56 (20.3%) | 34 (20.7%) | 22 (19.6%) | 0.89 |
| ≥ 5 million yen, < 10 million yen | 11 (40.2%) | 64 (39.0%) | 47 (42.0%) | |
| ≥ 10 million yen | 109 (39.5%) | 66 (40.2%) | 43 (38.4%) |
| Variable | Crude Odds Ratio (95% CI) | Adjusted Odds Ratio (95% CI) |
|---|---|---|
| Age at diagnosis (year) | 0.95 (0.92–0.989) | 0.94 (0.89–0.99) |
| Time since diagnosis (year) | 1.04 (0.92–1.19) | 0.997 (0.85–1.17) |
| Body mass index at diagnosis (kg/m2) | ||
| Underweight (< 18.5) | Ref | Ref |
| Normal (18.5 to < 25) | 0.52 (0.26–1.04) | 0.46 (0.20–1.06) |
| Overweight (≥ 25) | 1.10 (0.43–2.80) | 0.88 (0.26–2.96) |
| Breast cancer stage | ||
| 0 | Ref | Ref |
| I | 1.99 (0.91–4.36) | 1.73 (0.59–5.06) |
| II | 1.40 (0.64–3.07) | 1.15 (0.33–4.07) |
| III | 2.91 (1.06–7.99) | 2.03 (0.43–9.61) |
| Chemotherapy | ||
| No | Ref | Ref |
| Yes | 1.39 (0.85–2.29) | 1.43 (0.61–3.35) |
| Endocrine therapy | ||
| No | Ref | Ref |
| Yes | 1.31 (0.78–2.18) | 1.12 (0.55–2.27) |
| Radiation therapy | ||
| No | Ref | Ref |
| Yes | 1.27 (0.74–2.16) | 1.23 (0.63–2.41) |
| Postmenopausal at diagnosis | ||
| Yes | Ref | Ref |
| No | 1.78 (0.95–3.33) | 1.19 (0.46–3.12) |
| Smoking | ||
| Never | Ref | Ref |
| Former & currently | 1.34 (0.75–2.40) | 1.06 (0.54–2.10) |
| Education | ||
| High school | Ref | Ref |
| Some college | 0.75 (0.35–1.63) | 0.69 (0.28–1.67) |
| ≥ College | 0.51 (0.23–1.11) | 0.43 (0.17–1.13) |
| Household income | ||
| < 5 million yen | Ref | Ref |
| ≥ 5 million yen, < 10 million yen | 1.56 (0.76–3.21) | 1.61 (0.73–3.67) |
| ≥ 10 million yen | 1.19 (0.57–2.48) | 1.12 (0.47–2.68) |
| MVPA using IPAQ (min/week) | ||
| Q1 (≤ 180) | Ref | Ref |
| Q2 (181–300) | 1.27 (0.64–2.54) | − |
| Q3 (301–618) | 0.50 (0.24–1.06) | − |
| Q4 (≥ 619) | 0.80 (0.40–1.61) | − |
| Objectively measured MVPA (min/d) | ||
| Q1 (≤ 327) | Ref | Ref |
| Q2 (328–439) | 0.46 (0.22–0.97) | 0.43 (0.18–1.03) |
| Q3 (440–565) | 0.60 (0.29–1.23) | 0.48 (0.21–1.11) |
| Q4 (≥ 566) | 0.85 (0.42–1.70) | 0.78 (0.34–1.81) |
| Dietary intake (kcal/d) | ||
| Q1 (≤1334) | Ref | Ref |
| Q2 (1335–1602) | 1.23 (0.60–2.52) | 1.28 (0.52–3.14) |
| Q3 (1603–1874) | 1.03 (0.50–2.13) | 1.06 (0.40–2.77) |
| Q4 (≥ 1875) | 1.05 (0.51–2.18) | 1.43 (0.49–4.17) |
| Fish and shellfish intake (kcal/d) | ||
| Q1 (≤62) | Ref | Ref |
| Q2 (63–106) | 1.32 (0.65–2.66) | 1.39 (0.60–3.23) |
| Q3 (107–156) | 0.86 (0.42–1.76) | 0.87 (0.37–2.06) |
| Q4 (≥ 157) | 0.71 (0.34–1.50) | 0.78 (0.30–2.04) |
| Meat intake (kcal/d) | ||
| Q1 (≤ 82) | Ref | Ref |
| Q2 (83–131) | 1.14 (0.56–2.31) | 1.06 (0.45–2.46) |
| Q3 (132–173) | 0.77 (0.37–1.61) | 0.68 (0.27–1.73) |
| Q4 (≥ 174) | 0.94 (0.46–1.92) | 0.96 (0.38–2.45) |
| Total vegetable intake (kcal/d) | ||
| Q1 (≤ 43) | Ref | Ref |
| Q2 (44–65) | 0.78 (0.39–1.56) | 0.67 (0.28–1.57) |
| Q3 (66–90) | 0.63 (0.31–1.29) | 0.73 (0.31–1.70) |
| Q4 (≥ 91) | 0.60 (0.29–1.23) | 0.59 (0.23–1.54) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okumatsu, K.; Yamauchi, H.; Kotake, R.; Gosho, M.; Nakata, Y. Association between Endocrine Therapy and Weight Gain after Breast Cancer Diagnosis among Japanese Patients: A Retrospective Cohort Study. Med. Sci. 2021, 9, 50. https://doi.org/10.3390/medsci9030050
Okumatsu K, Yamauchi H, Kotake R, Gosho M, Nakata Y. Association between Endocrine Therapy and Weight Gain after Breast Cancer Diagnosis among Japanese Patients: A Retrospective Cohort Study. Medical Sciences. 2021; 9(3):50. https://doi.org/10.3390/medsci9030050
Chicago/Turabian StyleOkumatsu, Koki, Hideko Yamauchi, Rina Kotake, Masahiko Gosho, and Yoshio Nakata. 2021. "Association between Endocrine Therapy and Weight Gain after Breast Cancer Diagnosis among Japanese Patients: A Retrospective Cohort Study" Medical Sciences 9, no. 3: 50. https://doi.org/10.3390/medsci9030050
APA StyleOkumatsu, K., Yamauchi, H., Kotake, R., Gosho, M., & Nakata, Y. (2021). Association between Endocrine Therapy and Weight Gain after Breast Cancer Diagnosis among Japanese Patients: A Retrospective Cohort Study. Medical Sciences, 9(3), 50. https://doi.org/10.3390/medsci9030050







