Origins and Impact of Psychological Traits in Polycystic Ovary Syndrome
Abstract
1. Introduction
2. Anxiety and Depression in Women with Polycystic Ovary Syndrome
3. Genetics in Polycystic Ovary Syndrome
4. Epigenetics in Polycystic Ovary Syndrome
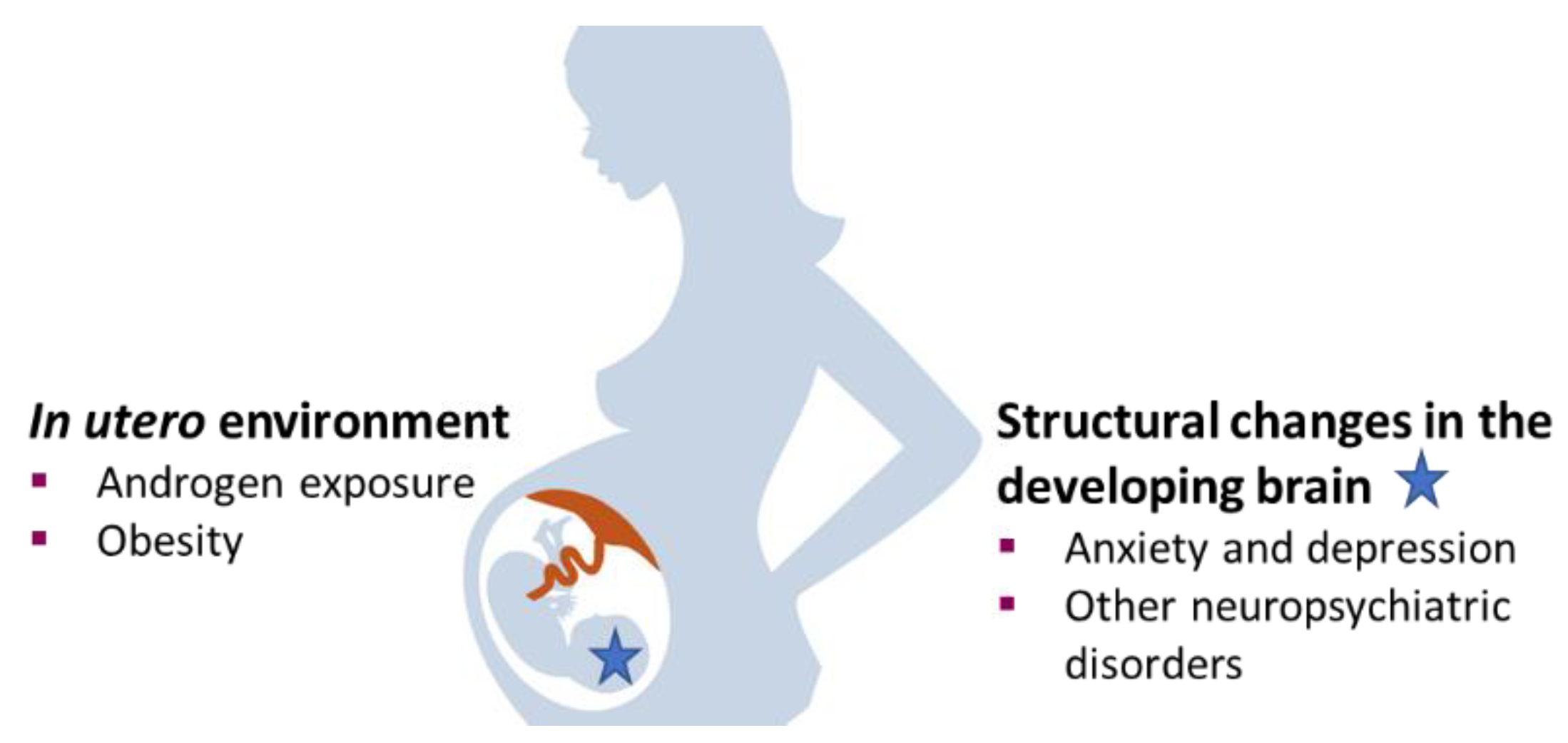
5. In Utero Androgen Exposure and Anxiety-Like Behavior in Polycystic Ovary Syndrome Offspring
Funding
Acknowledgments
Conflicts of Interest
References
- Dumesic, D.A.; Oberfield, S.E.; Stener-Victorin, E.; Marshall, J.C.; Laven, J.S.; Legro, R.S. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr. Rev. 2015, 36, 487–525. [Google Scholar] [CrossRef] [PubMed]
- Cooney, L.G.; Lee, I.; Sammel, M.D.; Dokras, A. High prevalence of moderate and severe depressive and anxiety symptoms in polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. 2017, 32, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Dokras, A.; Stener-Victorin, E.; Yildiz, B.O.; Li, R.; Ottey, S.; Shah, D.; Epperson, N.; Teede, H. Androgen Excess—Polycystic Ovary Syndrome Society: Position statement on depression, anxiety, quality of life, and eating disorders in polycystic ovary syndrome. Fertil. Steril. 2018, 109, 888–899. [Google Scholar] [CrossRef] [PubMed]
- Jedel, E.; Gustafson, D.; Waern, M.; Sverrisdottir, Y.B.; Landen, M.; Janson, P.O.; Labrie, F.; Ohlsson, C.; Stener-Victorin, E. Sex steroids, insulin sensitivity and sympathetic nerve activity in relation to affective symptoms in women with polycystic ovary syndrome. Psychoneuroendocrinology 2011, 36, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.H.; Tarantal, A.F.; Dumesic, D.A. Fetal, infant, adolescent and adult phenotypes of polycystic ovary syndrome in prenatally androgenized female rhesus monkeys. Am. J. Primatol. 2009, 71, 776–784. [Google Scholar] [CrossRef]
- Crisosto, N.; Echiburu, B.; Maliqueo, M.; Luchsinger, M.; Rojas, P.; Recabarren, S.; Sir-Petermann, T. Reproductive and metabolic features during puberty in sons of women with polycystic ovary syndrome. Endocr. Connect. 2017, 6, 607–613. [Google Scholar] [CrossRef]
- Brutocao, C.; Zaiem, F.; Alsawas, M.; Morrow, A.S.; Murad, M.H.; Javed, A. Psychiatric disorders in women with polycystic ovary syndrome: A systematic review and meta-analysis. Endocrine 2018, 62, 318–325. [Google Scholar] [CrossRef]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum. Reprod. 2018, 33, 1602–1618. [Google Scholar] [CrossRef]
- Akdag Cirik, D.; Dilbaz, B.; Aksakal, S.; Kotan, Z.; Ozelci, R.; Akpinar, F.; Mollamahmutoğlu, L. Do anxiety and depression statuses differ in differentpolycystic ovary syndrome phenotypes? Turk. J. Med. Sci. 2016, 46, 1846–1853. [Google Scholar] [CrossRef]
- Asik, M.; Altinbas, K.; Eroglu, M.; Karaahmet, E.; Erbag, G.; Ertekin, H.; Sen, H. Evaluation of affective temperament and anxiety-depression levels of patients with polycystic ovary syndrome. J. Affect. Disord. 2015, 185, 214–218. [Google Scholar] [CrossRef]
- Seyid Ahmet, A.Y.; Baskoy, K.; Deniz, F.; Yonem, A. Relationship Between the Clinical and Biochemical Findings of Polycystic Ovary Syndrome and Depression and Anxiety. Noro Psikiyatr Ars. 2016, 53, 366–367. [Google Scholar]
- Batool, S.; Ul Ain Ahmed, F.; Ambreen, A.; Sheikh, A.; Faryad, N. Depression and anxiety in women with polycystic ovary syndrome and its biochemical associates. J. SAFOG 2016, 8, 44–47. [Google Scholar]
- Cesta, C.E.; Kuja-Halkola, R.; Lehto, K.; Iliadou, A.N.; Landen, M. Polycystic ovary syndrome, personality, and depression: A twin study. Psychoneuroendocrinology 2017, 85, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Cooney, L.G. Polycystic ovary syndrome, obesity, and depression: Identifying the highest-risk group among an already high-risk group. Fertil. Steril. 2019, 111, 54–55. [Google Scholar] [CrossRef] [PubMed]
- Cooney, L.G.; Dokras, A. Depression and Anxiety in Polycystic Ovary Syndrome: Etiology and Treatment. Curr. Psychiatry Rep. 2017, 19, 83. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.; March, W.; Whitrow, M.; Fernandez, R.; Moore, V. Women with polycystic ovary syndrome are at increased risk of postnatal depression. In Human Reproduction; Oxford University Press: Oxford, UK, 2018; Volume 33, p. 322. [Google Scholar]
- Emeksiz, H.C.; Bideci, A.; Nalbantoglu, B.; Nalbantoglu, A.; Celik, C.; Yulaf, Y.; Çamurdan, M.O.; Cinaz, P. Anxiety and depression states of adolescents with polycystic ovary syndrome. Turk. J. Med. Sci. 2018, 48, 531–536. [Google Scholar] [PubMed]
- Enjezab, B.; Eftekhar, M.; Ghadiri-Anari, A. Association between severity of depression and clinico-biochemical markers of polycystic ovary syndrome. Electron. Phys. 2017, 9, 5820–5825. [Google Scholar] [CrossRef] [PubMed]
- Enjezab, B.; Ghadiri anari, A.; Eftekhar, M.; Mohajeri, M. Depression and clinical markers in polycystic ovary syndrome. Iran. J. Reprod. Med. 2015, 13, 1. [Google Scholar]
- Greenwood, E.A.; Pasch, L.; Legro, R.S.; Cedars, M.; Huddleston, H. Quality of life and depression in polycystic ovary syndrome. Fertil. Steril. 2017, 108, e64–e65. [Google Scholar] [CrossRef][Green Version]
- Greenwood, E.A.; Pasch, L.A.; Cedars, M.I.; Legro, R.S.; Eisenberg, E.; Huddleston, H.G.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Reproductive Medicine Network. Insulin resistance is associated with depression risk in polycystic ovary syndrome. Fertil. Steril. 2018, 110, 27–34. [Google Scholar] [CrossRef]
- Greenwood, E.A.; Pasch, L.A.; Cedars, M.I.; Legro, R.S.; Huddleston, H.G.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Reproductive Medicine Network. Association among depression, symptom experience, and quality of life in polycystic ovary syndrome. Am. J. Obstet. Gynecol. 2018, 219, 279. e1–279. e7. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, E.A.; Pasch, L.A.; Shinkai, K.; Cedars, M.I.; Huddleston, H.G. Putative role for insulin resistance in depression risk in polycystic ovary syndrome. Fertil. Steril. 2015, 104, 707–714.e1. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, E.A.; Pasch, L.A.; Shinkai, K.; Cedars, M.I.; Huddleston, H.G. Clinical course of depression symptoms and predictors of enduring depression risk in women with polycystic ovary syndrome: Results of a longitudinal study. Fertil. Steril. 2019, 111, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, E.A.; Yaffe, K.; Wellons, M.F.; Cedars, M.I.; Huddleston, H.G. Depression over the lifespan in a population-based cohort of women with polycystic ovary syndrome: Longitudinal analysis. J. Clin. Endocrinol. Metab. 2019, 15, 2809–2819. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, C.S.; Kimble, L.P.; Hodges, H.F.; Koci, A.F.; Mills, B.B. A mixed-methods study of coping and depression in adolescent girls with polycystic ovary syndrome. J. Am. Assoc. Nurse Pract. 2019, 31, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Klimczak, D.; Szlendak-Sauer, K.; Radowicki, S. Depression in relation to biochemical parameters and age in women with polycystic ovary syndrome. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 184, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Kogure, G.S.; Ribeiro, V.B.; Lopes, I.P.; Furtado, C.L.M.; Kodato, S.; de Sá, M.F.S.; Ferriani, R.A.; da Silva Lara, L.A.; dos Reis, R.M. Body image and its relationships with sexual functioning, anxiety, and depression in women with polycystic ovary syndrome. J. Affect. Disord. 2019, 253, 385–393. [Google Scholar] [CrossRef] [PubMed]
- March, W.A.; Whitrow, M.J.; Davies, M.J.; Fernandez, R.C.; Moore, V.M. Postnatal depression in a community-based study of women with polycystic ovary syndrome. Acta Obstet. Gynecol. Scand. 2018, 97, 838–844. [Google Scholar] [CrossRef]
- Mehrabadi, S.; Jahanian Sadatmahalleh, S.H.; Kazemnejad, A. The effect of depression and anxiety on cognitive performance in women with polycystic ovary syndrome. Int. J. Fertil. Steril. 2016, 10, 95. [Google Scholar]
- Mehrabadi, S.; Sadatmahalleh, S.J.; Kazemnejad, A. Association of depression and anxiety with cognitive function in patients with polycystic ovary syndrome. J. Maz. Univ. Med. Sci. 2017, 27, 159–170. [Google Scholar]
- Naqvi, S.H.; Moore, A.; Bevilacqua, K.; Lathief, S.; Williams, J.; Naqvi, N.; Pal, L. Predictors of depression in women with polycystic ovary syndrome. Arch. Women Ment. Health 2015, 18, 95–101. [Google Scholar] [CrossRef]
- Ozdemir, O.; Kurdoglu, Z.; Yildiz, S.; Ozdemir, P.G.; Yilmaz, E. The relationship between atypical depression and insulin resistance in patients with polycystic ovary syndrome and major depression. Psychiatry Res. 2017, 258, 171–176. [Google Scholar] [CrossRef]
- Salehifar, D.; Lotfi, R.; Ramezani Tehrani, F. The comparative study of depression in women with polycystic ovary syndrome and control group. Iran. J. Endocrinol. Metab. 2016, 18, 180–186. [Google Scholar]
- Stapinska-Syniec, A.; Grabowska, K.; Szpotanska-Sikorska, M.; Pietrzak, B. Depression, sexual satisfaction, and other psychological issues in women with polycystic ovary syndrome. Gynecol. Endocrinol. 2018, 34, 597–600. [Google Scholar] [CrossRef]
- Stefanaki, C.; Bacopoulou, F.; Livadas, S.; Kandaraki, A.; Karachalios, A.; Chrousos, G.P.; Diamanti-Kandarakis, E. Impact of a mindfulness stress management program on stress, anxiety, depression and quality of life in women with polycystic ovary syndrome: A randomized controlled trial. Stress 2015, 18, 57–66. [Google Scholar] [CrossRef]
- Xiang, S.; Wu, Y.; Wang, L.; Hua, F. Vitamin D supplementation for depression in women with polycystic ovary syndrome: Killing two birds with one stone? Arch. Women Ment. Health 2015, 18, 743–744. [Google Scholar] [CrossRef]
- Yu, Q.; Hao, S.; Wang, H.; Song, X.; Shen, Q.; Kang, J. Depression-Like Behavior in a Dehydroepiandrosterone-Induced Mouse Model of Polycystic Ovary Syndrome. Biol. Reprod. 2016, 95, 1–10. [Google Scholar] [CrossRef]
- Carron, R.C. The Effects of Depressive and Anxiety Symptoms, Perceived Stress, Body Mass Index, and Coping Processes on the Health-Related Quality of Life of Adult Women with Polycystic Ovary Syndrome. Ph.D. Thesis, University of Colorado Anschutz Medical Campus, Aurora, CO, USA, 2014. [Google Scholar]
- Macik, D. Loss of attributes of femininity, anxiety and value crisis. Women with polycystic ovary syndrome compared to women after mastectomy and in menopause. Health Psychol. Rep. 2016, 4, 159–169. [Google Scholar] [CrossRef]
- Legro, R.S.; Driscoll, D.; Strauss, J.F., 3rd; Fox, J.; Dunaif, A. Evidence for a genetic basis for hyperandrogenemia in polycystic ovary syndrome. Proc. Natl. Acad. Sci. USA 1998, 95, 14956–14960. [Google Scholar] [CrossRef]
- Day, F.; Karaderi, T.; Jones, M.R.; Meun, C.; He, C.; Drong, A.; Kraft, P.; Lin, N.; Huang, H.; Broer, L.; et al. Large-scale genome-wide meta-analysis of polycystic ovary syndrome suggests shared genetic architecture for different diagnosis criteria. PLoS Genet. 2018, 14, e1007813. [Google Scholar] [CrossRef]
- Azziz, R. PCOS in 2015: New insights into the genetics of polycystic ovary syndrome. Nat. Rev. Endocrinol. 2016, 12, 74–75. [Google Scholar] [CrossRef]
- Xu, N.; Azziz, R.; Goodarzi, M.O. Epigenetics in polycystic ovary syndrome: A pilot study of global DNA methylation. Fertil. Steril. 2010, 94, 781–783. [Google Scholar] [CrossRef]
- Xu, N.; Kwon, S.; Abbott, D.H.; Geller, D.H.; Dumesic, D.A.; Azziz, R.; Guo, X.; Goodarzi, M.O. Epigenetic mechanism underlying the development of polycystic ovary syndrome (PCOS)-like phenotypes in prenatally androgenized rhesus monkeys. PLoS ONE 2011, 6, e27286. [Google Scholar] [CrossRef]
- Kokosar, M.; Benrick, A.; Perfilyev, A.; Fornes, R.; Nilsson, E.; Maliqueo, M.; Behre, C.J.; Sazonova, A.; Ohlsson, C.; Ling, C.; et al. Epigenetic and Transcriptional Alterations in Human Adipose Tissue of Polycystic Ovary Syndrome. Sci. Rep. 2016, 6, 22883. [Google Scholar] [CrossRef]
- Nilsson, E.; Benrick, A.; Kokosar, M.; Krook, A.; Lindgren, E.; Kallman, T.; Martis, M.M.; Højlund, K.; Ling, C.; Stener-Victorin, E. Transcriptional and epigenetic changes influencing skeletal muscle metabolism in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2018, 103, 4465–4477. [Google Scholar] [CrossRef]
- Jones, M.R.; Brower, M.A.; Xu, N.; Cui, J.; Mengesha, E.; Chen, Y.D.; Taylor, K.D.; Azziz, R.; Goodrari, M.O. Systems Genetics Reveals the Functional Context of PCOS Loci and Identifies Genetic and Molecular Mechanisms of Disease Heterogeneity. PLoS Genet. 2015, 11, e1005455. [Google Scholar] [CrossRef]
- Abbott, D.H.; Dumesic, D.A.; Levine, J.E. Hyperandrogenic origins of polycystic ovary syndrome—Implications for pathophysiology and therapy. Expert Rev. Endocrinol. Metab. 2019, 14, 131–143. [Google Scholar] [CrossRef]
- Maliqueo, M.; Sundstrom Poromaa, I.; Vanky, E.; Fornes, R.; Benrick, A.; Akerud, H.; Stridsklev, S.; Labrie, F.; Jansson, T.; Stener-Victorin, E. Placental STAT3 signaling is activated in women with polycystic ovary syndrome. Hum. Reprod. 2015, 30, 692–700. [Google Scholar] [CrossRef]
- Kratimenos, P.; Penn, A.A. Placental programming of neuropsychiatric disease. Pediatric Res. 2019, 86, 157–164. [Google Scholar] [CrossRef]
- Chura, L.R.; Lombardo, M.V.; Ashwin, E.; Auyeung, B.; Chakrabarti, B.; Bullmore, E.T.; Baron-Cohen, S. Organizational effects of fetal testosterone on human corpus callosum size and asymmetry. Psychoneuroendocrinology 2010, 35, 122–132. [Google Scholar] [CrossRef]
- Rees, D.A.; Udiawar, M.; Berlot, R.; Jones, D.K.; O’Sullivan, M.J. White Matter Microstructure and Cognitive Function in Young Women With Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2016, 101, 314–323. [Google Scholar] [CrossRef]
- Lombardo, M.V.; Ashwin, E.; Auyeung, B.; Chakrabarti, B.; Lai, M.C.; Taylor, K.; Hackett, G.; Bullmore, E.T.; Baron-Cohen, S. Fetal programming effects of testosterone on the reward system and behavioral approach tendencies in humans. Biol. Psychiatry 2012, 72, 839–847. [Google Scholar] [CrossRef]
- Berni, T.R.; Morgan, C.L.; Berni, E.R.; Rees, D.A. Polycystic Ovary Syndrome Is Associated With Adverse Mental Health and Neurodevelopmental Outcomes. J. Clin. Endocrinol. Metab. 2018, 103, 2116–2125. [Google Scholar] [CrossRef]
- Cesta, C.E.; Mansson, M.; Palm, C.; Lichtenstein, P.; Iliadou, A.N.; Landen, M. Polycystic ovary syndrome and psychiatric disorders: Co-morbidity and heritability in a nationwide Swedish cohort. Psychoneuroendocrinology 2016, 73, 196–203. [Google Scholar] [CrossRef]
- Cesta, C.E.; Oberg, A.S.; Ibrahimson, A.; Yusuf, I.; Larsson, H.; Almqvist, C.; D’Onofrio, B.M.; Bulik, C.M.; de la Cru, L.F.; Mataix-Cols, D. Maternal polycystic ovary syndrome and risk of neuropsychiatric disorders in offspring: Prenatal androgen exposure or genetic confounding? Psychol. Med. 2019, 1–9. [Google Scholar] [CrossRef]
- Kosidou, K.; Dalman, C.; Widman, L.; Arver, S.; Lee, B.K.; Magnusson, C.; Gardner, R.M. Maternal polycystic ovary syndrome and the risk of autism spectrum disorders in the offspring: A population-based nationwide study in Sweden. Mol. Psychiatry 2016, 21, 1441–1448. [Google Scholar] [CrossRef]
- Kosidou, K.; Dalman, C.; Widman, L.; Arver, S.; Lee, B.K.; Magnusson, C.; Gardner, R.M. Maternal Polycystic Ovary Syndrome and Risk for Attention-Deficit/Hyperactivity Disorder in the Offspring. Biol Psychiatry 2017, 82, 651–659. [Google Scholar] [CrossRef]
- Barrett, E.S.; Hoeger, K.M.; Sathyanarayana, S.; Abbott, D.H.; Redmon, J.B.; Nguyen, R.H.N.; Swan, S.H. Anogenital distance in newborn daughters of women with polycystic ovary syndrome indicates fetal testosterone exposure. J. Dev. Orig. Health Dis. 2018, 9, 307–314. [Google Scholar] [CrossRef]
- Sir-Petermann, T.; Maliqueo, M.; Angel, B.; Lara, H.E.; Perez-Bravo, F.; Recabarren, S.E. Maternal serum androgens in pregnant women with polycystic ovarian syndrome: Possible implications in prenatal androgenization. Hum. Reprod. 2002, 17, 2573–2579. [Google Scholar] [CrossRef]
- Crisosto, N.; Codner, E.; Maliqueo, M.; Echiburu, B.; Sanchez, F.; Cassorla, F.; Sir-Peterman, T. Anti-Müllerian hormone levels in peripubertal daughters of women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2007, 92, 2739–2743. [Google Scholar] [CrossRef]
- Auyeung, B.; Lombardo, M.V.; Baron-Cohen, S. Prenatal and postnatal hormone effects on the human brain and cognition. Pflug. Arch. Eur. J. Physiol. 2013, 465, 557–571. [Google Scholar] [CrossRef]
- Wang, L.J.; Chou, M.C.; Chou, W.J.; Lee, M.J.; Lee, S.Y.; Lin, P.Y.; Lee, L.H.; Yang, Y.H.; Yen, C.F. Potential role of pre- and postnatal testosterone levels in attention-deficit/hyperactivity disorder: Is there a sex difference? Neuropsychiatr. Dis. Treat. 2017, 13, 1331–1339. [Google Scholar] [CrossRef]
- Hu, M.; Richard, J.E.; Maliqueo, M.; Kokosar, M.; Fornes, R.; Benrick, A.; Jansson, T.; Ohlsson, C.; Wu, X.; Skibicka, K.P.; et al. Maternal testosterone exposure increases anxiety-like behavior and impacts the limbic system in the offspring. Proc. Natl. Acad. Sci. USA 2015, 112, 14348–14353. [Google Scholar] [CrossRef]
- Jones, H.N.; Woollett, L.A.; Barbour, N.; Prasad, P.D.; Powell, T.L.; Jansson, T. High-fat diet before and during pregnancy causes marked up-regulation of placental nutrient transport and fetal overgrowth in C57/BL6 mice. FASEB J. 2009, 23, 271–278. [Google Scholar] [CrossRef]
- Sathishkumar, K.; Elkins, R.; Chinnathambi, V.; Gao, H.J.; Hankins, G.D.V.; Yallampalli, C. Prenatal testosterone-induced fetal growth restriction is associated with down-regulation of rat placental amino acid transport. Reprod. Biol. Endocrin. 2011, 9, 110. [Google Scholar] [CrossRef]
- Maliqueo, M.; Lara, H.E.; Sanchez, F.; Echiburu, B.; Crisosto, N.; Sir-Petermann, T. Placental steroidogenesis in pregnant women with polycystic ovary syndrome. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 166, 151–155. [Google Scholar] [CrossRef]
- Saben, J.L.; Boudoures, A.L.; Asghar, Z.; Thompson, A.; Drury, A.; Zhang, W.; Chi, M.; Cusumano, A.; Scheaffer, S.; Moley, K.H. Maternal Metabolic Syndrome Programs Mitochondrial Dysfunction via Germline Changes across Three Generations. Cell Rep. 2016, 16, 1–8. [Google Scholar] [CrossRef]
- Sasson, I.E.; Vitins, A.P.; Mainigi, M.A.; Moley, K.H.; Simmons, R.A. Pre-gestational vs. gestational exposure to maternal obesity differentially programs the offspring in mice. Diabetologia 2015, 58, 615–624. [Google Scholar] [CrossRef]
- Class, Q.A.; Rickert, M.E.; Larsson, H.; Lichtenstein, P.; D’Onofrio, B.M. Fetal growth and psychiatric and socioeconomic problems: Population-based sibling comparison. Br. J. Psychiatry 2014, 205, 355–361. [Google Scholar] [CrossRef]
- Manti, M.; Fornes, R.; Qi, X.; Folmerz, E.; Linden Hirschberg, A.; de Castro Barbosa, T.; Maliqueo, M.; Benrick, A.; Stener-Victorin, E. Maternal androgen excess and obesity induce sexually dimorphic anxiety-like behavior in the offspring. FASEB J. 2018, 32, 4158–4171. [Google Scholar] [CrossRef]
- Fornes, R.; Manti, M.; Qi, X.; Vorontsov, E.; Sihlbom, C.; Nystrom, J.; Jerlhag, E.; Maliqueo, M.; Hirschberg, A.L.; Carlström, M.; et al. Mice exposed to maternal androgen excess and diet-induced obesity have altered phosphorylation of catechol-O-methyltransferase in the placenta and fetal liver. Int J. Obes. 2019. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stener-Victorin, E.; Manti, M.; Fornes, R.; Risal, S.; Lu, H.; Benrick, A. Origins and Impact of Psychological Traits in Polycystic Ovary Syndrome. Med. Sci. 2019, 7, 86. https://doi.org/10.3390/medsci7080086
Stener-Victorin E, Manti M, Fornes R, Risal S, Lu H, Benrick A. Origins and Impact of Psychological Traits in Polycystic Ovary Syndrome. Medical Sciences. 2019; 7(8):86. https://doi.org/10.3390/medsci7080086
Chicago/Turabian StyleStener-Victorin, Elisabet, Maria Manti, Romina Fornes, Sanjiv Risal, Haojiang Lu, and Anna Benrick. 2019. "Origins and Impact of Psychological Traits in Polycystic Ovary Syndrome" Medical Sciences 7, no. 8: 86. https://doi.org/10.3390/medsci7080086
APA StyleStener-Victorin, E., Manti, M., Fornes, R., Risal, S., Lu, H., & Benrick, A. (2019). Origins and Impact of Psychological Traits in Polycystic Ovary Syndrome. Medical Sciences, 7(8), 86. https://doi.org/10.3390/medsci7080086




