From Rapid Recommendation to Online Preference-Sensitive Decision Support: The Case of Severe Aortic Stenosis
Abstract
1. Introduction
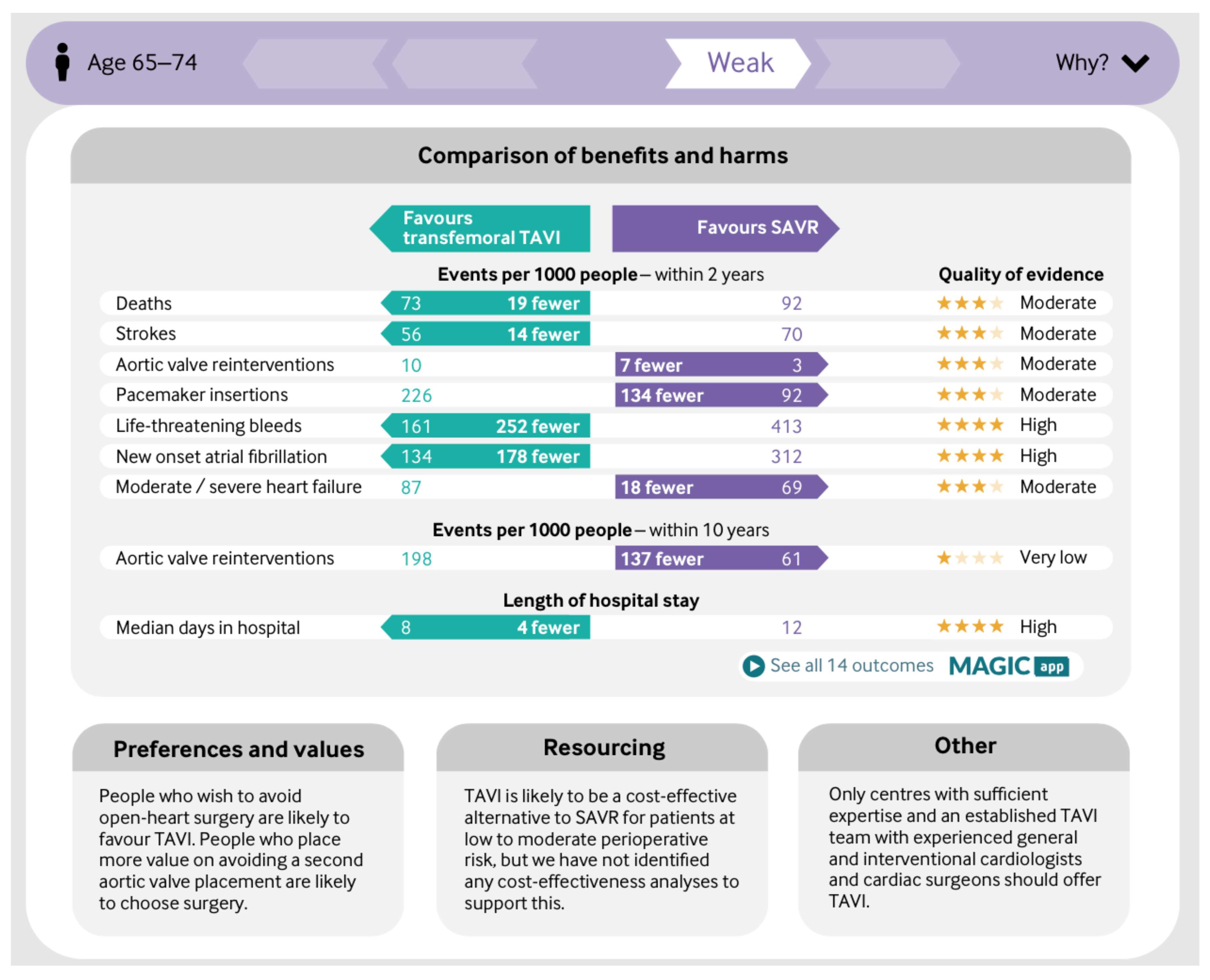
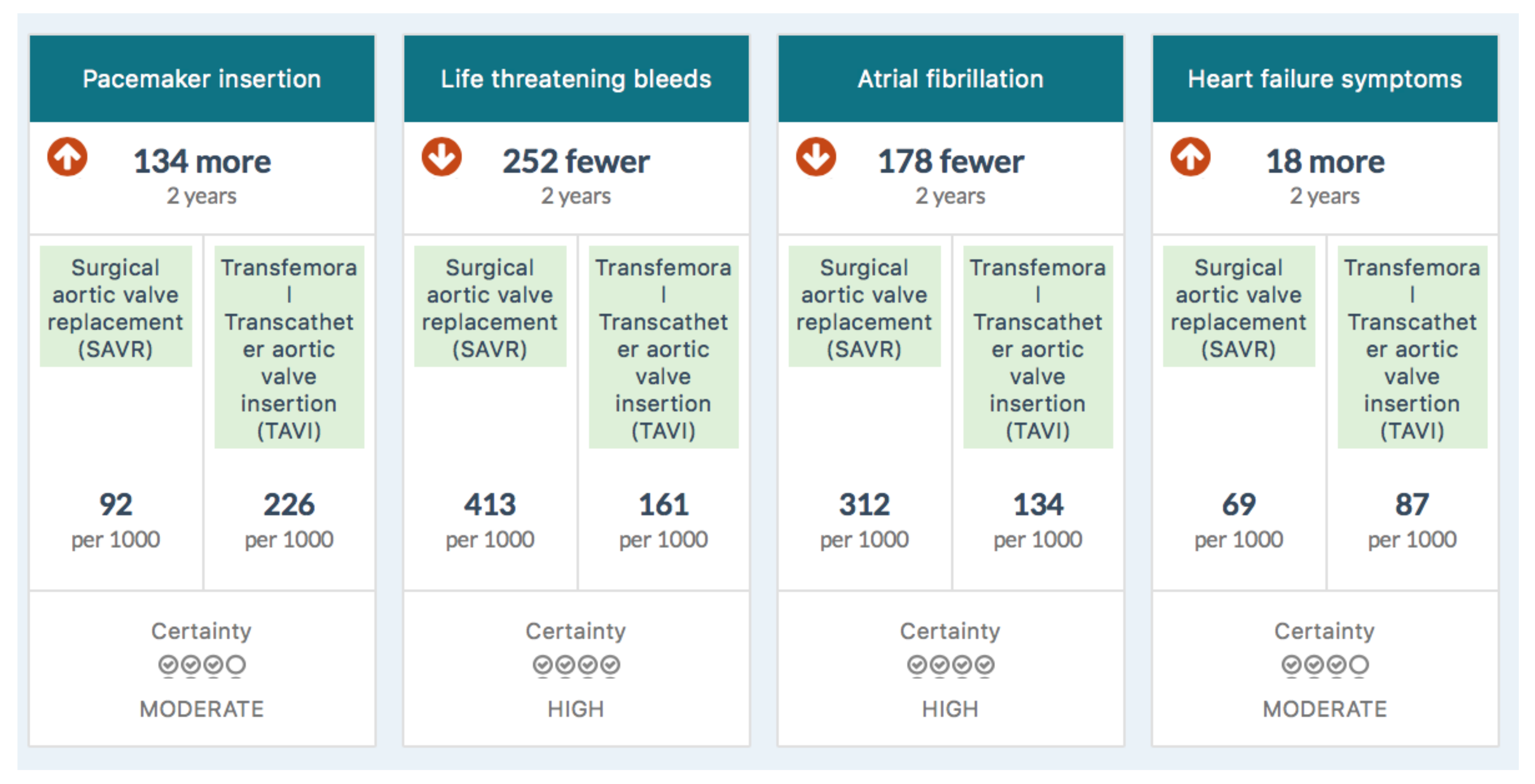
Mortality was reduced with transfemoral TAVI compared with SAVR by about 3%, stroke by 2%, acute kidney injury by 5%, bleeding by 24%, new onset atrial fibrillation by 18%, and duration of index admission by three days. These benefits, however, come with associated harms. TAVI was associated with an increased risk of experiencing symptoms of heart failure by about 6% (2% of which were moderate or severe), permanent pacemaker insertion by about 15%, and aortic valve reintervention over the short term by about 1%.[1] (p. 8)
2. Materials
2.1. The Rapid Recommendations
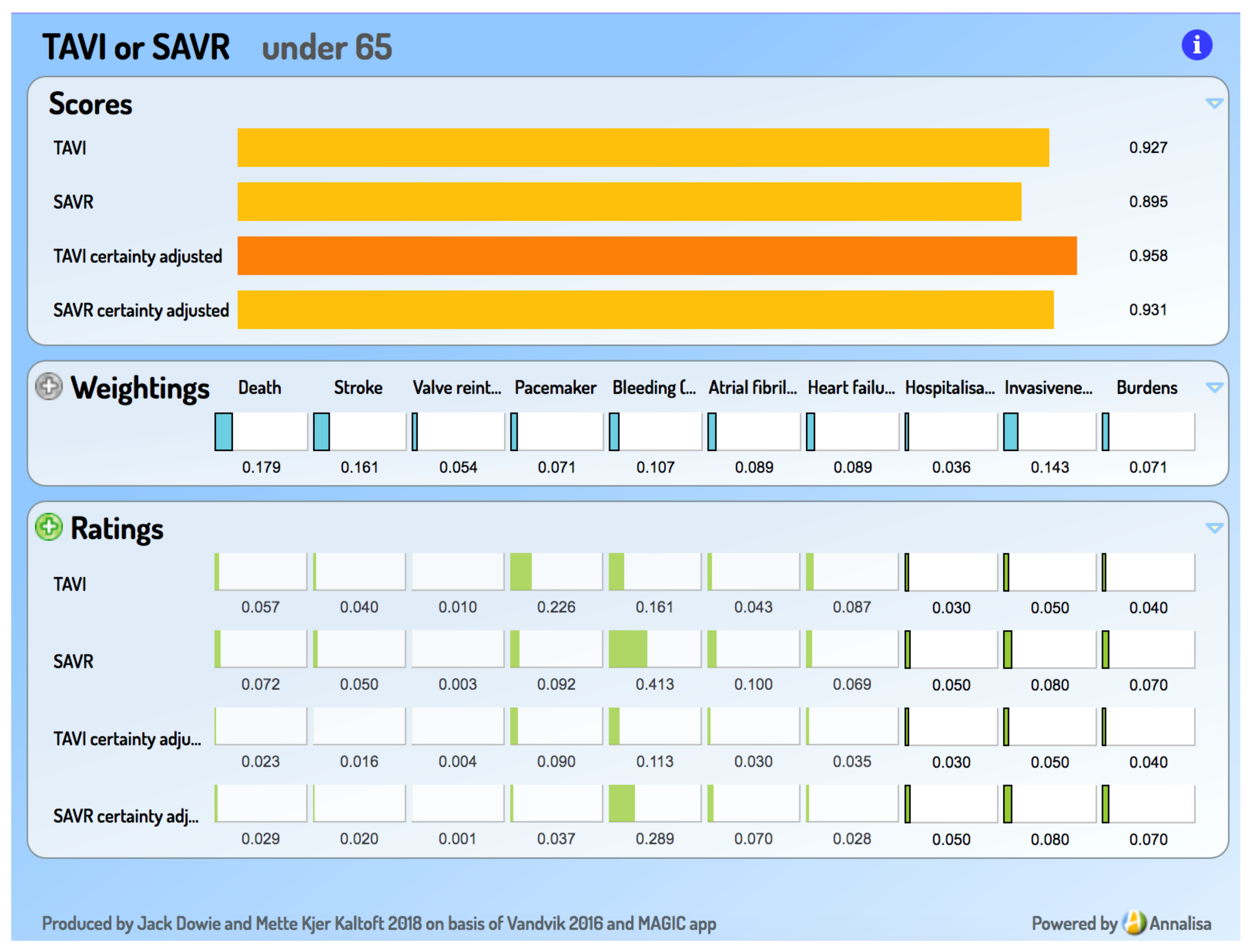
- Under 65: Strong recommendation for SAVR. ‘Since durability of TAVI valves is unknown, younger people may place a high value on avoiding a second aortic valve placement.’
- 65 and under 75: Weak recommendation for SAVR. ‘People who wish to avoid open-heart surgery are likely to favour TAVI. People who place more value on avoiding a second aortic valve placement are likely to choose surgery.’
- 75 and under 85: Weak recommendation for TAVI. Same as statement for previous group.
- 85 and over: Strong recommendation for TAVI. ‘The uncertainty around long-term durability of TAVI valves is not likely to concern those over 85. These older patients are also likely to place a high value on avoiding open heart surgery.’
- We suggest SAVR rather than TAVI [Weak recommendation].
- Benefits outweigh harms for the majority, but not for everyone. The majority of patients would likely want this option.
- Preference and values: Patients are likely to place different value on benefits and harms associated with TAVI. Patients aged 75 or younger—with a life expectancy well beyond 10 years—are likely to place a particularly high value on avoiding need for a second aortic valve replacement and are likely to choose surgery. Patients who place a high value on avoiding initial open heart surgery and are willing to accept an increased risk for aortic valve reintervention are likely to choose TAVI. A systematic review of values and preferences provided limited evidence to inform our judgements.
- Strong recommendations mean that most informed patients would choose the recommended management and that clinicians can structure their interactions with patients accordingly
- Weak recommendations mean that patients’ choices will vary according to their values and preferences, and clinicians must ensure that patients’ care is in keeping with their values and preferences
- Strength of recommendation is determined by the balance between desirable and undesirable consequences of alternative management strategies, quality of evidence, variability in values and preferences, and resource use [4] (p. 1051).
Panels should consider the evidence about the benefits and harms of the options and how certain that evidence is. They also need to consider how much the people affected directly by the decision value the benefits and harms, whether there is important uncertainty about this, and whether there is important variability in how much people value the benefits and harms. They must then consider all these criteria together to make a judgment about the balance between the desirable and undesirable effects of the option.[5] (p. 7, italics supplied)
When moving from evidence to recommendations, the panel integrated information on benefits and harms of treatment alternatives, quality of evidence, and values and preferences of patients as well as acceptability, feasibility, and resources.[2] (p. 2, italics supplied)
Uncertainty about how much those affected (patients or their carers) value the outcomes of interest can be a reason to make a weak (conditional) rather than a strong recommendation. Variability in how patients value the main outcomes (to the extent that individuals with different values would make different decisions) is another reason for a weak recommendation. For example, some patients might place a lower value on avoiding a stroke compared with avoiding serious gastrointestinal bleeding or the burden of warfarin treatment than other patients.[5] (p. 4)
2.2. The MAGIC Decision Aid
- This interactive tool for shared-decision making is designed to help you meet your patients’ needs by:
- Exploring what outcomes they wish to discuss
- Communicating the benefits and harms of each alternative, as well as their (un)certainty
- Discussing practical issues associated with each alternative
- This decision aid does not replace clinical judgment. Adapt it to the context as needed and use your own communication style.
This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial.[2] (Footnote)
For a fully interactive version of this graphical summary of recommendations, please visit: https://doi.org/10.1136/bmj.i5085
Disclaimer: This infographic is not a validated clinical decision aid. This information is provided without any representations, conditions or warranties that it is accurate or up to date. BMJ and its licensors assume no responsibility for any aspect of treatment administered with the aid of this information. Any reliance placed on this information is strictly at the user’s own risk.
3. Method
4. Result: An Add-On Decision Support Tool for the TAVI versus SAVR Choice
5. Discussion
As clinicians become more aware of variability in patients’ values and preferences, they are turning to structured decision aids to facilitate the decision making process. A strong recommendation indicates that use of a decision aid is unnecessary—almost all informed patients will make the same choice. A weak recommendation indicates that a decision aid could be useful.[4] (p. 1049)
… there is the danger of guideline recommendations being applied to people who do not place the same values on those recommendations as their clinician, or indeed those intended by the guideline creators… Surveys have shown that most patients wish either to share decision making with their clinicians or to take the decisions themselves. Guidelines should enable, not subvert, this process… guideline producers need to resist the temptation to tell clinicians and patients what to do. Making recommendations for the population, often based on expert opinion, reinforces the power imbalance between professional expertise and the patient’s values and preferences… The impact of the side effect at the level of the population may be high if the treatment is used widely, and may be considered important enough to withdraw the treatment from general use. This removes a potentially valuable treatment option for someone who has perhaps found the treatment highly effective and has a very low absolute risk of being harmed…It is difficult to personalise recommendations from guidelines, even for those skilled in evidence based medicine… Usable decision aids should [therefore] now be seen as one of the most important end products for evidence based medicine.[16] (pp. 1–2)
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siemieniuk, R.A.; Agoritsas, T.; Manja, V.; Devji, T.; Chang, Y.; Bala, M.M.; Thabane, L.; Guyatt, G.H. Transcatheter versus surgical aortic valve replacement in patients with severe aortic stenosis at low and intermediate risk: Systematic review and meta-analysis. BMJ 2016, 354, i5130. [Google Scholar] [CrossRef] [PubMed]
- Vandvik, P.O.; Otto, C.M.; Siemieniuk, R.A.; Bagur, R.; Guyatt, G.H.; Lytvyn, L.; Whitlock, R.; Vartdal, T.; Brieger, D.; Aergeerts, B.; et al. Transcatheter or surgical aortic valve replacement for patients with severe, symptomatic, aortic stenosis at low to intermediate surgical risk: A clinical practice guideline. BMJ 2016, 354, i5085. [Google Scholar] [CrossRef] [PubMed]
- Lytvyn, L.; Guyatt, G.H.; Manja, V.; Siemieniuk, R.A.; Zhang, Y.; Agoritsas, T.; Vandvik, P.O. Patient values and preferences on transcatheter or surgical aortic valve replacement therapy for aortic stenosis: A systematic review. BMJ Open 2016, 6, e014327. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Falck-Ytter, Y.; Vist, G.E.; Liberati, A.; Schünemann, H.J. GRADE: Going from evidence to recommendations. BMJ 2008, 336, 1049–1051. [Google Scholar] [CrossRef] [PubMed]
- Moberg, J.; Oxman, A.D.; Rosenbaum, S.; Schünemann, H.J.; Guyatt, G.H.; Flottorp, S.; Glenton, C.; Lewin, S.; Morelli, S.; Rada, G.; et al. The GRADE Evidence to Decision (EtD) framework for health system and public health decisions. Health Res. Policy Syst. 2018, 16, 45. [Google Scholar] [CrossRef] [PubMed]
- Kaltoft, M.K.; Nielsen, J.B.; Salkeld, G.; Dowie, J. Health informatics can avoid committing symbolic violence by recognizing and supporting generic decision-making competencies. Stud. Health Technol. Inf. 2015, 218, 172–177. [Google Scholar]
- Dowie, J.; Kaltoft, M.K.; Salkeld, G.; Cunich, M. Towards generic online multicriteria decision support in patient-centred health care. Health Expect 2013, 18, 689–702. [Google Scholar] [CrossRef] [PubMed]
- Kaltoft, M.K.; Nielsen, J.B.; Salkeld, G.; Dowie, J. Can a Discrete Choice Experiment contribute to person-centered healthcare? Eur. J. Pers. Centered Healthcare 2015, 3, 431–437. [Google Scholar] [CrossRef]
- Kaltoft, M.K.; Nielsen, J.B.; Salkeld, G.; Dowie, J. Preferences cannot be treated as epidemiological characteristics in person-centred care: A riposte. Eur. J. Pers. Centered Healthcare 2016, 4, 6–9. [Google Scholar] [CrossRef]
- Cheung, E.; Goodyear, G.; Yoong, W. Medicolegal update on consent: The Montgomery Ruling. Obstet. Gynaecol. 2016, 18, 171–172. [Google Scholar] [CrossRef]
- Williams, A.H.; Cookson, R.A. Equity-efficiency trade-offs in health technology assessment. Int. J. Technol. Assess. Health Care 2006, 22, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dowie, J.; Wildman, M. Choosing the surgical mortality threshold for high risk patients with stage la non-small cell lung cancer: Insights from decision analysis. Thorax 2002, 57, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Kaltoft, M.K.; Nielsen, J.B.; Dowie, J. Separating risk assessment from risk management poses legal and ethical problems in person-centred Care. Stud. Health Technol. Inf. 2018, 251, 23–26. [Google Scholar]
- Kaltoft, M.K.; Nielsen, J.B.; Dowie, J. Risk thresholds and risk classifications pose problems for person-centred care. Stud. Health Technol. Inf. 2018, 251, 19–22. [Google Scholar]
- vand der Weijden, T.; Boivin, A.; Burgers, J.; Schünemann, H.J.; Elwyn, G. Clinical practice guidelines and patient decision aids. An inevitable relationship. J. Clin. Epidemiol. 2012, 65, 584–589. [Google Scholar] [CrossRef] [PubMed]
- McCartney, M.; Treadwell, J.; Maskrey, N.; Lehman, R. Making evidence based medicine work for individual patients. BMJ 2016, 353, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dowie, J.; Kaltoft, M.K. The future of health is self-production and co-creation based on apomediative decision support. Med. Sci. 2018, 6, 66. [Google Scholar] [CrossRef] [PubMed]
- Dowie, J.; Kaltoft, M.K. Preference-sensitive apomediative decision support is key to facilitating self-produced health. Stud. Health Technol. Inf. 2018, 255, 132–136. [Google Scholar]
- Elwyn, G.; O’Connor, A.M.; Bennett, C.; Newcombe, R.G.; Politi, M.; Durand, M.-A.; Drake, E.; Joseph-Williams, N.; Khangura, S.; Saarimaki, A.; et al. Assessing the quality of decision support technologies using the International Patient Decision Aid Standards instrument (IPDASi). PLoS ONE 2009, 4, e4705. [Google Scholar] [CrossRef] [PubMed]
- Elwyn, G.; Scholl, I.; Tietbohl, C.; Mann, M.; Edwards, A.G.; Clay, C.; Légaré, F.; van der Weijden, T.; Lewis, C.L.; Wexler, R.M.; et al. “Many miles to go …”: A systematic review of the implementation of patient decision support interventions into routine clinical practice. BMC Med. Inf. Decis. Mak. 2013, 13 (Suppl. 2), S14. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, K.D.; Branda, M.E.; Anderson, R.T.; Pencille, L.J.; Montori, V.M.; Hess, E.P.; Ting, H.H.; LeBlanc, A. Peering into the black box: A meta-analysis of how clinicians use decision aids during clinical encounters. Implement Sci. 2014, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Lauck, S.B.; Baumbusch, J.; Achtem, L.; Forman, J.M.; Carrol, S.L.; Cheung, A.; Ye, J.; Wood, D.A.; Webb, J.G. Factors influencing the decision of older adults to be assessed for transcatheter aortic valve implantation: An exploratory study. Eur. J. Cardiovasc. Nurs. 2016, 15, 486–494. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dowie, J.; Kaltoft, M.K. From Rapid Recommendation to Online Preference-Sensitive Decision Support: The Case of Severe Aortic Stenosis. Med. Sci. 2018, 6, 109. https://doi.org/10.3390/medsci6040109
Dowie J, Kaltoft MK. From Rapid Recommendation to Online Preference-Sensitive Decision Support: The Case of Severe Aortic Stenosis. Medical Sciences. 2018; 6(4):109. https://doi.org/10.3390/medsci6040109
Chicago/Turabian StyleDowie, Jack, and Mette Kjer Kaltoft. 2018. "From Rapid Recommendation to Online Preference-Sensitive Decision Support: The Case of Severe Aortic Stenosis" Medical Sciences 6, no. 4: 109. https://doi.org/10.3390/medsci6040109
APA StyleDowie, J., & Kaltoft, M. K. (2018). From Rapid Recommendation to Online Preference-Sensitive Decision Support: The Case of Severe Aortic Stenosis. Medical Sciences, 6(4), 109. https://doi.org/10.3390/medsci6040109




