Abstract
Myeloperoxidase (MPO) belongs to the family of heme-containing peroxidases, produced mostly from polymorphonuclear neutrophils. The active enzyme (150 kDa) is the product of the MPO gene located on long arm of chromosome 17. The primary gene product undergoes several modifications, such as the removal of introns and signal peptides, and leads to the formation of enzymatically inactive glycosylated apoproMPO which complexes with chaperons, producing inactive proMPO by the insertion of a heme moiety. The active enzyme is a homodimer of heavy and light chain protomers. This enzyme is released into the extracellular fluid after oxidative stress and different inflammatory responses. Myeloperoxidase is the only type of peroxidase that uses H2O2 to oxidize several halides and pseudohalides to form different hypohalous acids. So, the antibacterial activities of MPO involve the production of reactive oxygen and reactive nitrogen species. Controlled MPO release at the site of infection is of prime importance for its efficient activities. Any uncontrolled degranulation exaggerates the inflammation and can also lead to tissue damage even in absence of inflammation. Several types of tissue injuries and the pathogenesis of several other major chronic diseases such as rheumatoid arthritis, cardiovascular diseases, liver diseases, diabetes, and cancer have been reported to be linked with MPO-derived oxidants. Thus, the enhanced level of MPO activity is one of the best diagnostic tools of inflammatory and oxidative stress biomarkers among these commonly-occurring diseases.
1. Introduction
Myeloperoxidase (MPO) (EC 1.11.1.7) is a member of subfamily of peroxidases. It is most abundantly expressed in immune cells, such as neutrophilic polymorphonuclear leukocytes (neutrophils) and lymphocytes [1,2], monocytes, and macrophages [3], and is also produced in other body cells. Myeloperoxidase is stored in cytoplasmic membrane-bound azurophilic granules and, during stimulation, these granules are secreted out to the extracellular space by degranulation or exocytosis [4,5]. The complete biochemical mechanism of neutrophil degranulation is not yet clear, but oxidative stress plays a key role in the release of MPO from these cells [6,7].
Neutrophils are well known white blood cells (WBCs) playing a pivotal role in innate immunity and frontline defense against microbial attacks [8]. In addition to MPO, several other proteins or enzymes are present in neutrophils which also show antimicrobial properties, e.g., defensins, serine proteases, cathepsin G, alkaline phosphatase, lysozyme, NADPH oxidase, collagenase, lactoferrin, cathepsin, and gelatinase, etc. [9]. Among these antimicrobial agents, MPO is the most abundant and constitutes 5% dry weight of neutrophils and 25% of the azurophilic granular proteins [10].
Normally, neutrophils degranulate at the infection site to combat different types of microbial activities and help to cure diseases. However, any unusual expression and release of MPO from activated neutrophils intensifies the inflammation and tissue damage and may result in several other diseases, even in the absence of infection [8,11].
This review article mainly focuses on the recent advances in the biochemical and the pathological aspects of myeloperoxidase and its significance as a disease biomarker in some commonly occurring chronic diseases.
2. Biochemistry of Myeloperoxidase
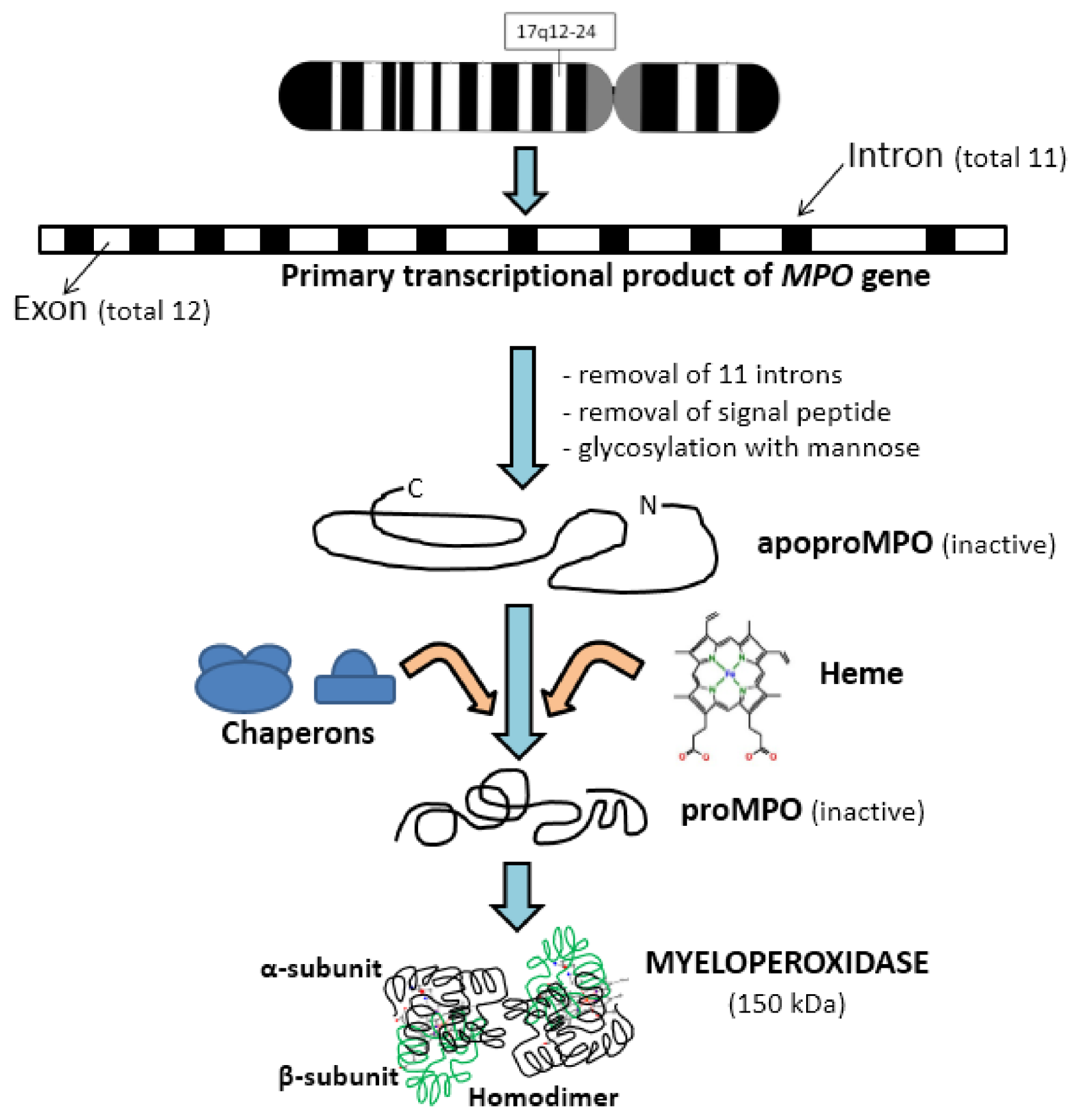
The MPO gene is located on the long arm segment q12–24 of chromosome 17 and the primary transcriptional product of this gene consists of 11 introns and 12 exons [12,13]. After some modifications like signal peptide removal and glycosylation with mannose-rich side chains, it produces apoproMPO [14]. This protein product is enzymatically inactive and further forms complexes with some chaperons like calreticulin and calnexin in the endoplasmic reticulum [15,16]. Enzymatically inactive, proMPO is formed from apoproMPO by the insertion of a heme moiety [17]. Furthermore, the removal of some N-terminal amino acids results in the production of 72–75 kDa protein, which undergoes further cleavage to produce α and β subunits. The α-subunit is heavy, 57 kDa, and consists of 467 amino acids, while the β-subunit is light, 12 kDa, and consists of 112 amino acids (Figure 1).
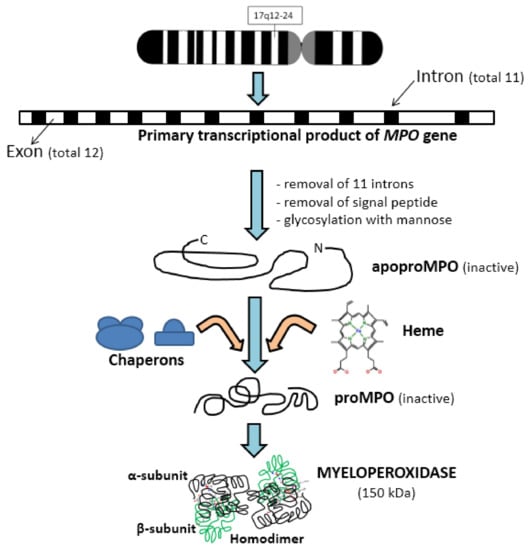
Figure 1.
Sequential steps involved in the synthesis of myeloperoxidase. MPO: myeloperoxidase.
Mature MPO consists of cationic homodimer heavy-light chain protomers and is about 150 kDa by weight. Each heavy subunit of mature MPO is covalently linked with a heme group and a mannose-rich moiety [18,19]. On the basis of the size of heavy chains in MPO, three isoforms have been observed: MPO I, MPO II, and MPO III [20]. A calcium-binding site is also present in MPO, which is very important for active site structure and function [21].
2.1. Activation and Release of Myeloperoxidase by Neutrophils
Although the coordination of MPO release by the degranulation of neutrophils is not fully understood, evidence supports that increased levels of oxidative stress by reactive oxygen species (ROS) and the activation of Src and p38 mitogen activated protein (MAP) kinase signaling pathways performs prominent roles in the process [22,23].
A fine coordination is necessary between different biochemical pathways, such as neutrophil activation, the production of ROS by superoxide generating NADPH oxidase, and MPO release by exocytosis. All of these organized reactions lead to the elimination of the bacterial invasion. Invading bacteria initiate enhanced production of H2O2 by superoxide dismutase (SOD), which is utilized by MPO for the production of chloramine and hypochlorite. Both of these products are highly toxic for the invading bacteria [8,24]. This biochemical phenomenon is also called respiratory burst. A clear illustration of the role of MPO is also observed in MPO knockout mice, which are highly susceptible to infections by Klebsiella and Candida and show persistent inflammation [25,26].
During bacterial infection, various other pro-inflammatory factors also trigger the release of MPO and ROS from neutrophils. During bacterial infection, one of the important mediators for this cascade is formylated peptide, which also works as a chemoattractant. Neutrophils are activated by this chemoattractant via formyl peptide receptor (fPR), a G protein-coupled receptor [27]. Some more proteins involved in antibacterial activities include phospholipases and protein kinases, such as mitogen-activated protein kinases (MAPK) and protein kinase C (PKC) [28,29,30]. During different pathological situations, or by the influence of several drugs, this signaling cascade gets impaired and finally leads to neutrophil dysfunction. These aberrations can be detrimental to host defense against several diseases or disease-causing microorganisms [27,31,32].
2.2. Reaction Mechanism and Functions of Myeloperoxidase
Activated neutrophils, monocytes, and some tissue macrophages release MPO at the sites of inflammation, using H2O2 to oxidize several substrates, such as halides (Cl−, Br−, and pseudohalides like thiocyanate (SCN−). This reaction leads to the formation of hypohalous acid, hypochlorous acid (HOCl−), hypobromous acid (HOBr−), and hypothiocyanous acid (HOSCN) [33]. MPO is able to interact with diverse ionic, atomic, and molecular entities via the interface with H2O2, including HOCl−, hydroxyl radicals, singlet oxygen, ozone, chloramines, and aldehydes [34,35,36]. These species are potent oxidants, which under normal and controlled circumstances are toxic to several microorganisms and play an important role in the immune system [37,38,39]. However, any excessive or unregulated production of theses oxidants can lead to damage of host cells and result in several diseases.
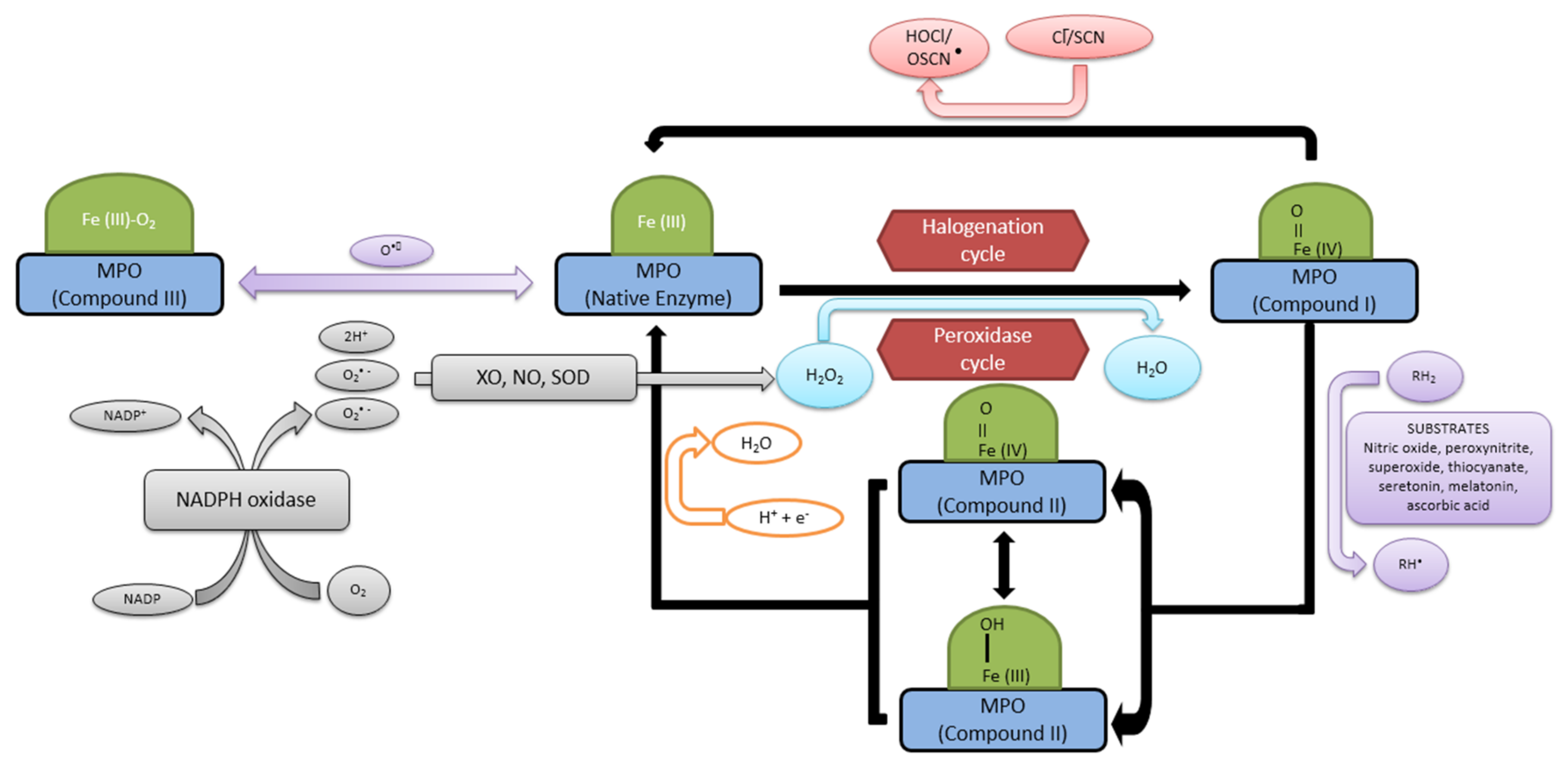
Myeloperoxidase produces reactive oxidants and other free radicals either through its peroxidase cycle or through a halogenation cycle, depending up on the substrate availability [40]. It is the only type of peroxidase that facilitates the oxidation of chloride to HOCl− by consuming H2O2. During its reaction cycles, MPO is converted to many transitional forms with different half-lives. Myeloperoxidase contains ferric heme (MPO-Fe(III)) in its resting state. During the peroxidase cycle, compound I is formed by the reaction with H2O2 [41]. In the absence of Cl−, the intermediate (MPO-Fe(IV)=O) in the presence of peroxide is reduced back to the ferric state in two sequential steps. The first step leads to the formation of compound II. This compound is reduced to compound III by second equivalent AH2 [42,43]. During the halogenation cycle, MPO-compound I exclusively oxidizes Cl− to HOCl− and no further intermediates are formed in this reaction, as compound I gets converted directly to its native form (Figure 2).
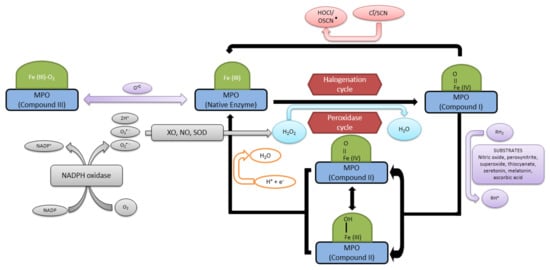
Figure 2.
Reaction mechanism of myeloperoxidase in the presence of different substrates. XO: xanthine oxidase; NO: nitric oxide; SOD: superoxide dismutase; Cl−/SCN: chloride/thiocyanate.
The fate of H2O2 as a substrate of MPO via peroxidation or chlorination reaction depends upon the concentration of chloride and the reducing substrates. A number of sources such as NADPH oxidase, xanthine oxidase, and nitric oxide synthase (NOS) are the sources of H2O2 for the MPO reaction, which also enhance the oxidative potential of H2O2 [44].
A strong antimicrobial cascade of reactions (respiratory burst), takes place in the presence of NADPH oxidase [45]. The initial products of this reaction are superoxide anions (O2−), produced as NADPH + O2 → O2− + NADP+ + H+.
During normal conditions, the antibacterial activities of MPO involve the production of different reactive oxygen and nitrogen species (ROS and RNS, respectively). These ROS and RNS also cause lipid peroxidation, protein nitration, and protein carbomylations. Myeloperoxidase plays a role in the oxidation and chemical modifications of different lipoproteins as well. In addition to this, MPO also mediates protein nitrosylation, dityrosine crosslinking, and 3-chlorotyrosine formation [46,47].
With the help of H2O2, this enzyme also oxidizes tyrosine to the tyrosyl radical. Neutrophils use HOCl− and the MPO-derived tyrosyl radical as cytotoxic agents against different types of bacteria and other pathogens [48]. As a signaling molecule, HOCl can activate several pathways, which later induce cellular senescence or apoptosis [49].
The polycationic protein nature of MPO helps it to bind several negatively charged surfaces of pathogens and causes cell membrane destruction, which ultimately leads to cell death [50]. In addition to pathogens, this enzyme can bind other cell surfaces like epithelial cells, fibroblasts, endothelial cells, macrophages, platelets, neutrophils, and low-density lipoproteins (LDLs) and very low-density lipoproteins (VLDLs) [51,52,53,54,55,56,57,58,59]. The binding of this enzyme to these cell surface alters some functional properties; for example, interaction with neutrophil integrins causes enhanced tyrosine phosphorylation of some proteins. This activates protein tyrosine kinase, which results in degranulation and leads to respiratory burst [58]. The binding of MPO to platelets causes the reorganization of the platelet cytoskeleton, and thus alters the aggregation properties [60]. MPO also oxidizes a variety of aromatic compounds by a one-electron mechanism to produce substrate radicals (R˙) [61,62].
In contrast to activation, different types of cytoplasmic enzymes are indirectly inactivated by myeloperoxidase-derived HOCl, due to its high chemical reactivity. Some of these enzymes include creatine kinase, lactate dehydrogenase, hexokinase, glyceraldehyde-3-phosphate dehydrogenase, etc. [63].
In addition to the many positive roles of myeloperoxidase discussed above, this enzyme has some drawbacks as well. Carbon nanotubes are used as drug delivery vehicles by some clinicians, but myeloperoxidase remains a significant hurdle as this enzyme suddenly breaks down these vehicles, thus limiting its applications [64].
2.3. Measurement of Myeloperoxidase Activity
Myeloperoxidase assays have been widely reported in the literature. However, no proper unanimity for most standard assays has been established. The complications are due to the fact that the substrates of MPO are the same as general peroxidase substrates. In addition to this, myoglobin and hemoglobin also show some peroxidase activity, thus interfering with the actual results. Unfortunately, no comparisons have been made between different myeloperoxidase assays, so standardization and validation are the first priorities in confirming the results from various studies [65]. Myeloperoxidase can be detected by flow cytometry, immunohistochemistry, or cytochemical staining. Some of the common assays of MPO are briefly described below.
Myeloperoxidase assays have been commonly performed by using different substrates, such as tetramethylbenzidine (TMB), 10-acetyl-3,7-dihydroxyphenoxazine (ADHP), and o-dianisidine dihydrochloride. These substrates react in the presence of H2O2 and form different colored products, which are estimated colorimetrically [66,67,68].
Myeloperoxidase assays have also been checked through chlorination activity evaluated with 39-(p-aminophenyl) fluorescein (APF) and 39-(p-hydroxyphenyl) fluorescein in the presence of H2O2. The fluorescence by this assay is checked by spectrofluorometry [65]. Myeloperoxidase activity has also been assayed by bromide-dependent chemoluminescence using luminol in the presence of H2O2 [69].
Nowadays, the most common method of MPO measurement is via commercially-available enzyme-linked immunosorbent assay (ELISA) kits. The MPO level is measured by sandwich ELISA with a monoclonal antibody [70]. Human plasma diluted samples, a control, and a standard are pipetted in wells coated with biotin-conjugated mouse anti-human MPO monoclonal antibodies bound to streptavidin–horseradish peroxidase (HRP) and incubated on a vibrating shaker at room temperature. After some time, the contents of the wells are washed and replaced with TMB as a substrate, and then incubated for a specific time. This reaction mixture is followed by a stop solution. Absorbance is determined by an ELISA reader and the quantity of MPO is interpreted from the calibration curve of the standard. The assay is sensitive (the lower limit of detection may be 0.026 ng/mL) [71].
2.4. Inhibitors and Activators of Myeloperoxidase
Even though a strong correlation has been found between atherosclerosis, inflammatory diseases, and MPO release, little work has been done to inhibit MPO to suppress these diseases. Several naturally occurring compounds possess inhibitory activities against MPO, including polyphenols, melatonin, flavonoids, etc. [72].
The MPO reaction is inhibited by general peroxidase inhibitors azide and benzoic acid hydrazide-containing compounds, but the proper mechanism of its inhibition is still unknown [73]. There are some specific inhibitors of MPO, such as 4-amino benzoic acid hydrazide (4-ABH) [74]. Ceruloplasmin, an acute phase plasma protein produced from hepatocytes and activated macrophages, is a physiologic inhibitor of MPO [75]. Niacin can also inhibit cellular ROS production and MPO release through some complex signaling mechanisms [76].
In addition to the above compounds, some naturally occurring anti-inflammatory, antioxidant, and antihistaminic compounds possess inhibitory activities against MPO. These compounds include nonsteroidal anti-inflammatory drugs (NSAIDs), e.g., diclofenac, ferulic acid, caffeic acid, resveratrol, indomethacin, flufenamic acid, chalcones, and gallic acid [77,78,79].
As compared to inhibitors, little is known about the activators of MPO. This type of peroxidase is present as an inactive or partially active form in resting granulocytes and its activation is instigated by different factors. Some of the activators discovered include granulocyte macrophage colony stimulating factor (GM-CSF), N-formyl-methionyl-leucyl-phenylalanine (fMLP), and phorbol mysristate acetate [80,81].
3. Role of Myeloperoxidase in Different Diseases
In addition to the antipathogenic or bactericidal role of MPO-derived HOCl during normal conditions, under some pathological circumstances the overproduction of these oxidizing agents also causes oxidative damage of proteins and DNA in host cells. Several types of tissue injuries and the pathogenesis of various chronic diseases such as atherosclerosis, cancer, renal disease, lung injury, and multiple sclerosis. Additionally, Alzheimer’s and Parkinson’s diseases have been reported to be directly/indirectly linked with MPO-derived oxidants [8] (Table 1). Thus, the enhanced level of MPO is one of the best inflammatory and oxidative stress markers among these commonly occurring diseases [82,83].

Table 1.
Brief etiology and the direct/indirect involvement of MPO in different types of diseases.
3.1. Inflammation
Some of the common examples of diseases and conditions with chronic inflammation are: tuberculosis, asthma, rheumatoid arthritis, chronic sinusitis, chronic hepatitis, peptic ulcer, ulcerative colitis, and chronic periodontitis. Myeloperoxidase is now considered as a new biomarker of inflammation in these diseases as well as other ailments like ischemic heart disease and acute coronary syndrome. This enzyme is released into the extracellular medium during different inflammatory processes [111]. During inflammation, vascular permeability is increased by the activation of various inflammatory mediators, which results in the influx of immunoglobulins and serum proteins at the site of inflammation [112,113]. This cascade of inflammatory process also motivates the migration of polymorphonuclear neutrophils, which result in the release of MPO [114,115].
Inflammatory processes are also triggered by lipid peroxidation and the synthesis of eicosanoids. Cytochrome P450, lipoxygenase, and cyclooxygenase also play a prominent role in these events. Myeloperoxidase generates reactive intermediates that stimulate lipid peroxidation. This oxidoreductase can oxidize tyrosine and nitrite to form tyrosyl radical and nitrogen dioxide (˙NO2), respectively. These reactive intermediates can oxidize lipids in plasma and the cell membrane [116]. The lipoprotein phospholipid peroxidation of the membrane is linked to their interference, leading to cellular dysfunctions. Lipid peroxidation can be a normal physiological activity or a potential contributor to the pathophysiological consequence of acute and chronic inflammatory diseases [117,118].
Myeloperoxidase also leads to the formation of tyrosyl radical, formed when MPO initiates lipid peroxidation, also leading to the formation of phenolic cross-links on proteins. A typical molecular fingerprint, protein-bound dityrosine, is enhanced during atheroma and other sites of inflammation [119]. In addition to this, lipid peroxidation also occurs by nitrogen dioxide (˙NO2), which is formed by MPO enzymatic action. Myeloperoxidase is also indirectly involved in the post-translational modification of some proteins, resulting in the formation of nitrotyrosine, which can also occur in the presence of ˙NO2 [120,121].
3.2. Rheumatoid Arthritis
Elevated MPO levels have been observed in several inflammatory diseases including rheumatoid arthritis (RA) [122]. Rheumatoid arthritis is well characterized by chronically inflamed synovial joints with some destruction of cartilage and bones [123]. Several factors have been proposed for this disease, among which oxidative stress is a leading hypothesis [124,125]. Inflamed synovium is often intervened by B and T lymphocytes, macrophages, and neutrophils. The intrusion of these cells in the synovium during RA leads to the release of multiple pro-inflammatory mediators. The degranulation of neutrophils leads to the release of enzymes and peptides, leading to respiratory burst and oxidative stress [126,127,128,129]. The overproduction of ROS is a potential tissue-damaging agent that is further formed by the cascades of reactions by HOCl− produced by the activated neutrophils present in synovial fluid [101,130]. This was verified in the inflamed cartilage of patients suffering from RA [102]. Currently, a firm hypothesis is that the enhanced levels of MPO in inflamed cartilage of RA are causally associated with the lifelong disease progression.
3.3. Cardiovascular Diseases and Atherosclerosis
Myeloperoxidase gained special importance in 2001, due to its association with coronary artery diseases (CAD). Since that time, MPO has been considered a circulating marker of related diseases such as acute coronary syndrome, CAD, and chronic heart failure [131,132,133,134,135]. Elevated levels of circulating MPO are observed in patients with coronary artery diseases, unstable angina, and acute myocardial infarction [8,136,137,138]. Plasma MPO concentration was reported to be higher in myocardial infarction (MI) patients (55 ng/mL) as compared to control subjects (39 mg/mL) [139].
Circulating MPO also shows a link with red blood cells (RBCs)’ rigidity index in several patients with combined ischemic heart disease. This enzyme induces some changes in RBCs’ cellular morphology and biophysical properties like plasma membrane fluidity, transmembrane potential, cell size, hemolysis sensitivity, and cellular deformability. This enzyme also alters the intracellular Ca2+ level in addition to causing some changes in band 3 proteins and glycophorins of the RBC membrane [60].
Atherosclerosis is the major cause of cardiovascular diseases (CVD). Neutrophils and monocytes play a key role in atherosclerosis, leading to chronic inflammatory problems. Different events and sequences occur during CVD, which include endothelial dysfunction besides the formation and rupture of atherosclerotic plaque [140]. In the arterial wall subendothelial region, all of these stages occur during inflammation, which ultimately leads to the accumulation and deposition of altered lipids [141].
Atherosclerosis leads to the accumulation of cholesterol and cholesteryl esters on arterial walls, which are derived from LDL. In addition to this, LDL retention on these walls triggers an immune response, resulting in a cascade of production of oxidants and inflammation [142,143]. Plasma LDL interacts with circulating MPO, which has been reported to be higher in patients suffering from atherosclerosis [144]. It has been reported that in some patients undergoing hemodialysis, HOCl reacts with LDL, which promotes atherogenesis [91,145]. Macrophage exposure to HOCl−-LDL results in an accumulation of cholesterol and its esters as well as the production of lipid-rich foam cells [146].
3.4. Obesity
The infiltration of neutrophils in adipose tissue is an initial and persistent step for the advancement of diet-induced obesity [84,147]. This infiltration of neutrophils leads to increased MPO expression, as recent reports have also revealed a higher level of MPO in obese adults [148,149,150]. Thus, with this concern of obesity, prolonged low-grade inflammation combined with cardiovascular risk factors probably occur in parallel. A proper mechanism to justify the regulation of MPO activity in cases of obesity requires further research [151,152].
To investigate the possible role of MPO in obesity, Wang et al. [148], during his research on animal models, found that MPO-deficient mice showed resistance to progression towards diet-induced obesity. In another study, isolated neutrophils were treated with an inhibitor against MPO action, and this prevented insulin resistance in obese mice. All of these observations indicate the probability that MPO targets white and brown fat and is a chief contributor to the progression of inflammation-induced obesity, but this assumption also requires further investigation [148].
In humans, a strong positive correlation has been found between obesity and diabetes with leukocytosis [153,154,155], as the plasma MPO level was found to be raised in prepubertal obese children [85]. A study of MPO as an early biomarker of inflammation and obesity in prepubertal obese children indicated an MPO level of about 22 µg/L, as compared to normal weight children with an MPO level of about 14 µg/L. Thus, these studies support the hypothesis that serum myeloperoxidase is a suitable and potential biomarker for assessing various cardiovascular risks among obese patients [156].
3.5. Neurodegenerative Diseases
Oxidative stress is also proposed to be responsible for the release of neurotoxic mediators commanded by MPO derived from cells like neurons, astrocytes, and activated microglia, as well as peripheral inflammatory cells [11]. In the brain, of the different neurotoxic oxidants, HOCl is a stable, highly reactive, and predominant one. This acid is involved in a number of neurodegenerative diseases, including stroke, epilepsy, multiple sclerosis, Parkinson’s and Alzheimer’s diseases, and multiple sclerosis, etc. [157].
In addition to inflammation and oxidative stress, MPO is related to depression, which is an emotional disorder affecting a vast majority of the world’s population [158]. Furthermore, patients suffering from bipolar disorders are prescribed anti-inflammatory drugs, such as lithium and valproate, indicating some links with MPO [159]. Still, the complete mechanism of the role of myeloperoxidase biochemistry in neuronal diseases remains unknown.
3.6. Diabetes/Diabetic Retinopathy
Diabetes mellitus and its complication, diabetic retinopathy (DR), are also known as diseases with subclinical inflammation. During diabetic retinopathy, retinal structural and biochemical alterations cause the activation of neutrophils [160]. The increased expression of various types of growth factors and cytokines including TNF-α occurs due to biochemical modifications during DR. Inflammatory mediator priming causes MPO translocation and interaction with anti-MPO antibodies.
In the vessels of diabetic retina, the upregulation of leukocyte adhesion molecules occurs, and leukocytes are also observed in the lumen of human microaneurysm. Furthermore, the vitreous samples of patients with DR show elevated levels of CD4/CD8 and T lymphocytes [90].
Chronic inflammation during diabetic retinopathy is sustained by cytokine-producing B-lymphocytes. There is a correlation between the activity of proliferative DR (PDR) and increased lymphocyte infiltration [161]. Increased vascular permeability due to leukocytosis leads to retinal abnormalities, endothelial injury, and capillary occlusion [162,163,164]. Neutrophils and monocytes can be activated by proteinase-3 antineutrophilic cytoplasmic antibody (PR3-ANCA) and MPO antinutrophilic cytoplasmic antibody (MPO-ANCA) to release acute inflammatory mediators, which causes endothelial cell injuries [165,166]. Priming by proinflammatory factors, such as cytokines, TNF-α, and microbial products like bacterial formyl peptides, etc., triggers circulating neutrophils to express more ANCA antigens.
3.7. Liver Diseases
Among several types of liver disease, such as fibrosis, necrosis, inflammation, and steatosis, alcoholic cirrhosis denotes a major cause of mortality with an estimated 3.8% of all worldwide deaths [167,168]. Cirrhosis is closely related to immune dysfunctions, and thus to the inability of the host to protect against various infections [169]. In several types of liver injuries, for example alcoholic steatohepatitis in human beings or in animal models, neutrophils contribute to the pathogenesis of cirrhosis [170]. The infiltration of neutrophils in the liver is good for predicting disease [93], as these cells increase the intracellular oxidative stress during liver injury [171]. In addition to this, the stellate macrophages or Kupffer cells located in the liver also synthesize MPO. The activation of these cells results in hepatic fibrosis, which is proposed to be developed by the local release of oxidants and cytokines [92,172].
Neutrophils employ their favorable effects through different factors such as granulopoiesis [144,173], the production of hepatocyte growth factor [174], and collagen degradation. Granulocyte-colony stimulating factor (G-CSF) therapy has been observed to be beneficial in cases with severe alcoholic hepatitis [175]. Patients with cirrhosis have impaired neutrophilic ROS production, as well as phagocytotic and microbicidal activities [176,177,178]. Post-hepatic cirrhosis has also been observed to be closely related to diminished ROS production in some liver transplant recipients [179]. The mechanism of impaired signaling events of neutrophils in relation to alcoholic cirrhosis is not fully understood. Several researchers have observed an erroneous MAPK-dependent phosphorylation of p47phox, an important component of NADPH oxidase [30].
3.8. Cancer
The knowledge of the precise biochemical relationship between the inflammatory response and specific malignancy is a vast field to be understood, although growing evidence points to links between the relationships of MPO, inflammation, and cancer [88,180,181]. Cancer progression advances by the biochemical alterations of different biomolecules and genes by various oxidative species, ultimately produced through MPO.
DNA damage can be caused by oxidants directly or indirectly produced by MPO, which can lead to mutagenesis [86]. An abnormal MPO expression and greater risks of different forms of cancers are directly associated with MPO gene polymorphism [182]. In the promoter region of this peroxidase gene, single nucleotide polymorphisms (SNPs) can possibly affect transcription and protein levels [87,183]. In addition to this, the substitution of thymidine for cytosine in codon 569 causes the substitution of an amino acid from arginine to tryptophan, which may also cause some genetic defects of MPO [184].
In addition to gene polymorphism, MPO induces cancer through the activation of genotoxic intermediates and procarcinogens through an indirect implication of MPO [185,186]. The metabolism of unsaturated fats and some amino acids like serine and threonine can form byproducts, like acrolein, which in turn form acrolein-protein adducts [187]. In humans, these new protein adducts can transform colon tumors from benign to malignant states [188]. Still, little information is available about such proteins that form adducts with acrolein or their role in tumor progression.
Several reports are available about the relationship between breast cancer and increased serum MPO level as compared to control groups. The promotion of this cancer is also enhanced by inflammatory leukocytes, which produce ROS, chemo- and cytokines, proteases, histamine, and other mediators [189]. Various types of DNA damages and genomic instability are instigated by MPO-synthesized ROS [180,190]. Thus, in premenopausal women suffering from breast cancer, MPO acts as an efficient marker [191]. Furthermore, risks of the development of cancer are directly linked to the endogenous production of high MPO levels [192].
Myeloperoxidase is a hallmark enzyme of acute myeloid lineage and the clinical relevance of the circulating MPO level in acute myeloid leukemia (AML) patients showed higher plasma MPO levels (range 1.0–9514 ng/mL) as compared to control subjects (range 3.5–20.6 ng/mL) [193].
3.9. Cystic Fibrosis
Cystic fibrosis (CF), a disease of the respiratory tract, is characterized by severe bacterial infections, especially Pseudomonas aeruginosa, as well as very large numbers of infiltrating neutrophils [194]. Neutrophils are also thought to contribute to lung damage instead of eliminating bacteria from the respiratory tract [94]. Cystic fibrosis patients’ sputum contains high concentrations of MPO and human neutrophil elastase (HNE), and these levels correlate with the severity of the lung disease [195,196]. The clear mechanism for the release of inflammatory mediators like HNE, extracellular DNA, and MPO from neutrophils during CF is not known. However, neutrophil extracellular traps reveal a potential mechanism for the release of these mediators [197].
4. Myeloperoxidase Deficiency
Several studies have shown that, in the USA and Europe, partial or complete MPO deficiency is relatively common among the human population (affecting 1 in 2000 to 1 in 4000 people) [198,199]. However, there is a geographic heterogeneity between the frequencies of hereditary MPO deficiency in different populations. For example, these findings are compared to the reported 1 in 55,000 in Japan [200,201,202]. Generally, MPO deficiency results in a modest increase of either inflammatory problems or infectious complications [203]. MPO-deficient neutrophils exhibit impaired bactericidal and candidacidal activities against Staphylococcus aureus and many species of Candida [204,205].
The deficiency of MPO is a hereditary problem that may also lead to immune deficiency [206]. In addition, several different types of autoantibodies have been observed to be raised against MPO in various types of vasculitis. The three most clinically prominent vasculitis forms of this type are granulomatosis with polyangitis, and eosinophilic granulomatosis with polyangitis (EGPA) and microscopic polyangitis. Autoantibodies against neutrophils, also known as ANCAs, have also been detected in the perinuclear region staining [207].
5. Conclusions
Myeloperoxidase enzyme is the most abundant pro-inflammatory biomarker present in neutrophilic granulocytes. It is released from these cells by proinflammatory factors and during oxidative stress at the site of infection to combat different types of microbial activities. The antibacterial activities of MPO involve the production of different reactive oxygen and nitrogen species. Myeloperoxidase also plays a role in the chemical modifications of different lipoproteins, protein nitrosylation, tyrosyl radical formation, and dityrosine crosslinking, etc. Myeloperoxidase gained special importance as a well-known biomarker due to its role in a number of inflammatory diseases including rheumatoid arthritis, cardiovascular diseases, neurodegenerative diseases, diabetic retinopathy, liver diseases, cancer, and transplant rejection. As the activation of macrophages and neutrophils may occur in any type of inflammation, significant future research is required to precisely understand the role of MPO in these diseases. Different assays have been conducted to check the level of MPO in several diseases due to the lack of a specific substrate, but no comparisons have yet been made between these assays. Thus, it is very important to standardize the assays of MPO with some specific substrate with the aim of understanding the reference range of MPO in different diseases.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Khan, A.A.; Rahmani, A.H.; Aldebasi, Y.H.; Aly, S.H. Biochemical and pathological studies on peroxidases –An updated review. Glob. J. Health Sci. 2014, 6, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-Q.; Zhang, Y.-Z.; Wu, Y.; Zhang, J.-J.; Li, T.-B.; Jiang, T.; Xiong, X.-M.; Luo, X.-J.; Ma, Q.-L.; Peng, J. Myeloperoxidase-derived hypochlorous acid promotes ox-LDL induced senescence of endothelial cells through a mechanism involving β-catenin signaling in hyperlipidemia. Biochem. Biophys. Res. Commun. 2015, 467, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, S.J.; Hazen, S.L. Myeloperoxidase and cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.J. Tissue destruction by neutrophils. N. Engl. J. Med. 1989, 320, 365–376. [Google Scholar] [PubMed]
- Chen, Y.; Hashiguchi, N.; Yip, L.; Junger, W.G. Hypertonic saline enhances neutrophil elastase release through activation of P2 and A3 receptors. Am. J. Physiol. 2006, 290, C1051–C1059. [Google Scholar] [CrossRef] [PubMed]
- Lacy, P. Mechanism of degranulation in neutrophils. Allergy Asthma Clin. Immunol. 2006, 2, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Naegelen, N.; Beaume, N.; Plançon, S.; Schenten, V.; Tschirhart, E.J.; Bréchard, S. Regulation of neutrophil degranulation and cytokine secretion: A novel model approach based on linear fitting. J. Immunol. Res. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, S.J. Myeloperoxidase: Friend and foe. J. Leukoc. Biol. 2005, 77, 598–625. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, R.; Kawata, J.; Yamamoto, T.; Ishimaru, Y.; Sakamoto, A.; Ono, T.; Narahara, S.; Sugiuchi, H.; Hirose, E.; Yamaguchi, Y. Mechanism of interferon-γ production by monocytes stimulated with myeloperoxidase and neutrophil extracellular traps. Blood Cells Mol. Dis. 2015, 55, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Segal, A.W. How neutrophils kill microbes. Annu. Rev. Immunol. 2005, 23, 197–223. [Google Scholar] [CrossRef] [PubMed]
- Panasenko, O.M.; Gorudko, I.V.; Sokolov, A.V. Hypochlorous acid as a precursor of free radicals in living systems. Biochemistry 2013, 78, 1466–1489. [Google Scholar] [CrossRef] [PubMed]
- Inazawa, J.; Inoue, K.; Nishigaki, H.; Tsuda, S.; Taniwaki, M.; Misawa, S.; Abe, T. Assignment of the human myeloperoxidase gene (MPO) to bands q21.3 → q23 of chromosome 17. Cytogenet. Cell Genet. 1989, 50, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Zaki, S.R.; Austin, G.E.; Chan, W.C.; Conaty, A.L.; Trusler, S.; Trappier, S.; Lindsey, R.B.; Swan, D.C. Chromosomal localization of the human myeloperoxidase gene by in situ hybridization using oligonucleotide probes. Genes Chromosomes Cancer 1990, 2, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.R.; Nauseef, W.M. Molecular biology of MPO. In Peroxidases in Chemistry and Biology; Everse, J., Everse, K.E., Grisham, M.B., Eds.; CRC: Boca Raton, FL, USA, 1991; Volume I, pp. 63–81. [Google Scholar]
- Nauseef, W.M.; McCormick, S.J.; Clark, R.A. Calreticulin functions as a molecular chaperone in the biosynthesis of myeloperoxidase. J. Biol. Chem. 1995, 270, 4741–4747. [Google Scholar] [CrossRef] [PubMed]
- Nauseef, W.M.; McCormick, S.J.; Goedken, M. Coordinated participation of calreticulin and calnexin in the biosynthesis of myeloperoxidase. J. Biol. Chem. 1998, 273, 7107–7111. [Google Scholar] [CrossRef] [PubMed]
- Pinnix, I.B.; Guzman, G.S.; Bonkovsky, H.L.; Zaki, S.R.; Kinkade, J.M., Jr. The post-translational processing of myeloperoxidase is regulated by the availability of heme. Arch. Biochem. Biophys. 1994, 312, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Andrews, P.C.; Krinsky, N.I. The reductive cleavage of myeloperoxidase in half, producing enzymatically active hemi-myeloperoxidase. J. Biol. Chem. 1981, 256, 4211–4218. [Google Scholar] [PubMed]
- Olsen, R.L.; Little, C. Studies on the subunits of human myeloperoxidase. Biochem. J. 1984, 222, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Miyasaki, K.T.; Nemirovskiy, E. Myeloperoxidase isoform activities released by human neutrophils in response to dental and periodontal bacteria. Oral Microbiol. Immunol. 1997, 12, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.; Havasawa, H.; Lonnerdal, B. Mutations affecting the calcium-binding site of myeloperoxidase and lactoperoxidases. Biochem. Biophys. Res. Commun. 2001, 281, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Ganji, S.H.; Kamanna, V.S.; Kashyap, M.L. Niacin decreases leukocyte myeloperoxidase: Mechanistic role of redox agents and Src/p38MAP kinase. Atherosclerosis 2014, 235, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Anatoliotakis, N.; Deftereos, S.; Bouras, G.; Giannopoulos, G.; Tsounis, D.; Angelidis, C.; Kaoukis, A.; Stefanadis, C. Myeloperoxidase: Expressing inflammation and oxidative stress in cardiovascular disease. Curr. Top. Med. Chem. 2013, 13, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Nauseef, W. Myeloperoxidase in human neutrophil host defense. Cell Microbiol. 2014, 16, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Aratani, Y.; Kura, F.; Watanabe, H.; Akagawa, H.; Takano, Y.; Suzuki, K.; Dinauer, M.C.; Maeda, N.; Koyama, H. In vivo role of myeloperoxidase for the host defense. Jpn. J. Infect. Dis. 2004, 57, S15. [Google Scholar] [PubMed]
- El Kebir, D.; Jozsef, L.; Pan, W.; Filep, J.G. Myeloperoxidase delays neutrophil apoptosis through CD11b/CD18 integrins and prolongs inflammation. Circ. Res. 2008, 103, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Dorward, D.A.; Lucas, C.D.; Chapman, G.B.; Haslett, C.; Dhaliwal, K.; Rossi, A.G. The role of formylated peptides and formyl peptide receptor-1 in governing neutrophil function during acute inflammation. Am. J. Pathol. 2015, 185, 1172–1184. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Germena, G.; Hirsch, E. Dissection of the interplay between class I PI3Ks and Rac13 signaling in phagocytic functions. Sci. World J. 2010, 10, 1826–1839. [Google Scholar] [CrossRef] [PubMed]
- Futosi, K.; Fodor, S.; Mócsai, A. Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int. Immunopharmacol. 2013, 17, 638–650. [Google Scholar] [CrossRef] [PubMed]
- Rolas, L.; Makhezer, N.; Hadjoudj, S.; El-Benna, J.; Djerdjouri, B.; Elkrief, L.; Moreau, R.; Perianin, A. Inhibition of mammalian target of rapamycin aggravates the respiratory burst defect of neutrophils from decompensated patients with cirrhosis. Hepatology 2013, 57, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Dinauer, M.C. Disorders of neutrophil function: An overview. Methods Mol. Biol. 2014, 1124, 501–515. [Google Scholar] [PubMed]
- Kruger, P.; Saffarzadeh, M.; Weber, A.N.; Rieber, N.; Radsak, M.; von Bernuth, H.; Benarafa, C.; Roos, D.; Skokowa, J.; Hartl, D. Neutrophils: Between host defence, immune modulation, and tissue injury. PLoS Pathog. 2015, 11, e1004651. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J. Myeloperoxidase-derived oxidation: Mechanisms of biological damage and its prevention. J. Clin. Biochem. Nutr. 2011, 48, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Deimann, W. Endogenous peroxidase activity in mononuclear phagocytes. Prog. Histochem. Cytochem. 1984, 15, 1–56. [Google Scholar] [CrossRef]
- Davies, M.J.; Hawkins, C.L.; Pattison, D.I.; Rees, M.D. Mammalian heme peroxidases: From molecular mechanisms to health implications. Antioxid. Redox Signal. 2008, 10, 1199–1234. [Google Scholar] [CrossRef] [PubMed]
- Chapman, A.L.; Skaff, O.; Senthilmohan, R.; Kettle, A.J.; Davies, M.J. Hypobromous acid and bromamine production by neutrophils and modulation by superoxide. Biochem. J. 2009, 417, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Britigan, B.E.; Ratcliffe, H.R.; Buettner, G.R.; Rosen, G.M. Binding of myeloperoxidase to bacteria: Effect on hydroxyl radical formation and susceptibility to oxidant-mediated killing. Biochim. Biophys. Acta 1996, 1290, 231–240. [Google Scholar] [CrossRef]
- Hampton, M.B.; Kettle, A.J.; Winterbourn, C.C. Involvement of superoxide and myeloperoxidase in oxygen-dependent killing of Staphylococcus aureus by neutrophils. Infect. Immun. 1996, 64, 3512–3517. [Google Scholar] [PubMed]
- Hampton, M.B.; Kettle, A.J.; Winterbourn, C.C. Inside the neutrophil phagosome: Oxidants, myeloperoxidase, and bacterial killing. Blood 1998, 92, 3007–3017. [Google Scholar] [PubMed]
- Flemming, J.; Remmler, J.; Rohring, F.; Arnold, J. (−)-Epicatechin regenerates the chlorinating activity of myeloperoxidase in vitro and in neutrophil granulocytes. J. Inorg. Biochem. 2014, 130, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.E.; Araiso, T.; Palcic, M.M.; Dunford, H.B. Compound I of myeloperoxidase. Biochem. Biophys. Res. Commun. 1980, 94, 34–40. [Google Scholar] [CrossRef]
- Andrews, P.C.; Krinsky, N.I. Quantitative determination of myeloperoxidase using tetramethylbenadine as substrate. Anal. Biochem. 1982, 127, 346–350. [Google Scholar] [CrossRef]
- Andrews, P.C.; Krinsky, N.I. A kinetic analysis of the interaction of human myeloperoxidase with hydrogen peroxide, chloride ions, and protons. J. Biol. Chem. 1982, 257, 13240–13245. [Google Scholar] [PubMed]
- Cai, H.; Griendling, K.K.; Harrison, D.G. The vascular NAD(P)H oxidases as therapeutic targets in cardiovascular diseases. Trends Pharmacol. Sci. 2003, 24, 471–478. [Google Scholar] [CrossRef]
- Nathan, C.; Xie, Q. Nitric oxide synthases: Roles, tolls, and controls. Cell 1994, 78, 915–918. [Google Scholar] [CrossRef]
- Rutkowski, R.; Pancewicz, S.A.; Rutkowski, K.; Rutkowska, J. Reactive oxygen and nitrogen species in inflammatory process. Pol. Merkur. Lekarski 2007, 134, 131–136. [Google Scholar]
- Freeman, T.A.; Parvizi, J.; Della-Valle, C.J.; Steinbeck, M.J. Reactive oxygen and nitrogen species induced protein and DNA modifications driving arthrofibrosis following total knee arthroplasty. Fibrogenesis Tissue Repair 2009, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Weis, V.M.; Small, A.L.; Mc-Fall-Ngai, M.J. A peroxidase related to the mammalian antimicrobial protein myeloperoxidase in the Euprymna–Vibrio mutualism. Proc. Natl. Acad. Sci. USA 1996, 93, 13683–13688. [Google Scholar] [CrossRef] [PubMed]
- Goud, A.P.; Goud, P.T.; Diamond, M.P.; Gonik, B.; Abu-Soud, H.M. Reactive oxygen species and oocyte aging: Role of superoxide, hydrogen peroxide, and hypochlorous acid. Free Radic. Biol. Med. 2008, 44, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Miyasaki, K.T.; Zambon, J.J.; Jones, C.A.; Wilson, M.E. Role of high avidity binding of human neutrophil myeloperoxidase in the killing of Actinobacillus actinomycetemcomatins. Infect. Immun. 1987, 55, 1029–1036. [Google Scholar] [PubMed]
- Haegens, A.; Vernooy, J.H.J.; Heeringa, P.; Mossman, B.T. Wouter, E.F. Myeloperoxidase modulates lung epithelial responses to pro-inflammatory agents. Eur. Respir. J. 2008, 31, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Zabucchi, G.; Soranzo, M.R.; Menegazzi, R.; Bertoncin, P.; Nardon, E.; Patriarca, P. Uptake of human eosinophil peroxidase and myeloperoxidase by cells involved in the inflammatory process. J. Histochem. Cytochem. 1989, 37, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Klinke, A.; Nussbaum, C.; Kubala, L.; Friedrichs, K.; Rudolph, T.K.; Rudolph, V.; Paust, H.J.; Schröder, C.; Benten, D.; Lau, D.; et al. Myeloperoxidase attracts neutrophils by physical factors. Blood 2011, 117, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Vargunam, M.; Adu, D.; Taylor, C.M.; Michael, J.; Richards, N.; Neuberger, J.; Thompson, R.A. Endothelium myeloperoxidase–antimyeloperoxidase interaction in vasculitis. Nephrol. Dial. Transplant. 1992, 7, 1077–1081. [Google Scholar] [PubMed]
- Lefkowitz, D.L.; Mills, K.; Morgan, D.; Lefkowitz, S.S. Macrophage activation and immunomodulation by myeloperoxidase. Proc. Soc. Exp. Biol. Med. 1992, 199, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Gorudko, I.V.; Sokolov, A.V.; Shamova, E.V.; Grudinina, N.A.; Drozd, E.S.; Shishlo, L.M.; Grigorieva, D.V.; Bushuk, S.B.; Bushuk, B.A.; Chizhik, S.A.; et al. Myeloperoxidase modulates human platelet aggregation via actin cytoskeleton reorganization and store-operated calcium entry. Biol. Open 2013, 2, 916–923. [Google Scholar] [CrossRef] [PubMed]
- Kolarova, H.; Klinke, A.; Kremserova, S.; Adam, M.; Pekarova, M.; Baldus, S.; Eiserich, J.P.; Kubala, L. Myeloperoxidase induces the priming of platelets. Free Radic. Biol. Med. 2013, 61, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Lau, D.; Mollnau, H.; Eiserich, J.P.; Freeman, B.A.; Daiber, A.; Gehling, U.M.; Brümmer, J.; Rudolph, V.; Münzel, T.; Heitzer, T.; et al. Myeloperoxidase mediates neutrophil activation by association with CD 11b/CD 18 integrins. Proc. Natl. Acad. Sci. USA 2005, 102, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, A.V.; Ageeva, K.V.; Cherkalina, O.S.; Pulina, M.O.; Zakharova, E.T.; Prozorovskii, V.N.; Aksenov, D.V.; Vasilyev, V.B.; Panasenko, O.M. Identification and properties of complexes formed by myeloperoxidase with lipoproteins and ceruloplasmin. Chem. Phys. Lipids 2010, 163, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Gorudko, I.V.; Sokolov, A.V.; Shamova, E.V.; Grigorieva, D.V.; Mironova, E.V.; Kudryavtsev, I.V.; Gusev, S.A.; Gusev, A.A.; Chekanov, A.V.; Vasilyev, V.B.; et al. Binding of human myeloperoxidase to red blood cells: Molecular targets and biophysical consequences at the plasma membrane level. Arch. Biochem. Biophy. 2016, 591, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Heinecke, J.W.; Li, W.; Daehnke, H.L.; Goldstein, J.A. Dityrosine, a specific marker of oxidation, is synthesized by the myeloperoxidase hydrogen peroxide system of human neutrophils and macrophages. J. Biol. Chem. 1993, 268, 4069–4077. [Google Scholar] [PubMed]
- Marquez, L.A.; Dunford, H.B. Kinetics of oxidation of tyrosine and dityrosine by myeloperoxidase compounds I and II. J. Biol. Chem. 1996, 270, 30434–30440. [Google Scholar] [CrossRef]
- Schraufstatter, I.U.; Browne, K.; Harris, A.; Hyslop, P.A.; Jackson, J.H.; Quehenberger, O.; Cochrane, C.G. Mechanisms of hypochlorite injury of target cells. J. Clin. Investig. 1990, 85, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Kagan, V.E.; Konduru, N.V.; Feng, W.; Allen, B.L.; Conroy, J.; Volkov, Y.; Vlasova, I.I.; Belikova, N.A.; Yanamala, N.; Kapralov, A.; et al. Carbon nanotubes degraded by neutrophil myeloperoxidase induce less pulmonary inflammation. Nat. Nanotechnol. 2010, 5, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Pulli, B.; Ali, M.; Forghani, R.; Schob, S.; Hsieh, K.L.; Wojtkiewicz, G.; Linnoila, J.J.; Chen, J.W. Measuring myeloperoxidase activity in biological samples. PLoS ONE 2013, 8, e67976. [Google Scholar] [CrossRef] [PubMed]
- Bozeman, P.M.; Learn, D.B.; Thomas, E.L. Assay of human leukocyte enzymes myeloperoxidase and eosinophil peroxidase. J. Immunol. Methods 1990, 126, 125–133. [Google Scholar] [CrossRef]
- Suzuki, K.; Ota, H.; Sasagawa, S.; Sakatani, T.; Fujikura, T. Assay method for myeloperoxidase in human polymorphonuclear leukocytes. Anal. Biochem. 1983, 132, 345–352. [Google Scholar] [CrossRef]
- Dhiman, M.; Estrada-Franco, J.G.; Pando, J.M.; Ramirez-Aguilar, F.J.; Spratt, H.; Vazquez-Corzo, S.; Perez-Molina, G.; Gallegos-Sandoval, R.; Moreno, R.; Garg, N.J. Increased myeloperoxidase activity and protein nitration are indicators of inflammation in patients with Chagas’ disease. Clin. Vaccine Immunol. 2009, 16, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Haqqani, A.S.; Sandhu, J.K.; Birnboim, H.C. A myeloperoxidase-specific assay based upon bromide dependent chemiluminiscence of luminol. Anal. Biochem. 1999, 273, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Cooray, R.; Petersson, C.G.B.; Gronvik, K.-O. Preparation and characterization of monoclonal antibodies against bovine myeloperoxidase. Vet. Immunol. Immunopathol. 1995, 46, 211–221. [Google Scholar] [CrossRef]
- Mayyas, F.A.; Al-Jarrah, M.I.; Ibrahim, K.S.; Alzoubi, K.H. Level and significance of plasma myeloperoxidase and the neutrophil to lymphocyte ratio in patients with coronary artery disease. Exp. Ther. Med. 2014, 8, 1951–1957. [Google Scholar] [CrossRef] [PubMed]
- Bensalem, S.; Soubhye, J.; Aldib, I.; Bournine, L.; Nguyen, A.T.; Vanhaeverbeek, M.; Rousseau, A.; Boudjeltia, K.Z.; Sarakbi, A.; Kauffmann, J.M.; et al. Inhibition of myeloperoxidase activity by the alkaloids of Peganumharmala L. (Zygophyllaceae). J. Ethnopharmacol. 2014, 154, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Smith, F.; Panizzi, J.R.; Goodwin, D.C.; Panizzi, P. Inactivation of myeloperoxidase by benzoic acid hydrazide. Arch. Biochem. Biophys. 2015, 570, 14–22. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kettle, A.J.; Gedye, C.A.; Winterbourn, C.C. Mechanism of inactivation of myeloperoxidase by 4-aminobenzoic acid hydrazide. Biochem. J. 1997, 321, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Segelmark, M.; Persson, B.; Hellmark, T.; Wieslander, J. Binding and inhibition of myeloperoxidase (MPO): A major function of ceruloplasmin? Clin. Exp. Immunol. 1997, 108, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Ganji, S.H.; Qin, S.; Zhang, L.; Kamanna, V.S.; Kashyap, M.L. Niacin inhibits vascular oxidative stress, redox-sensitive genes, and monocyte adhesion to human aortic endothelial cells. Atherosclerosis 2009, 202, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Van Antwerpen, P.; Dufrasne, F.; Lequeux, M.; Boudjeltia, K.Z.; Lessgyer, I.; Babar, S.; Moreau, P.; Moguilevsky, N.; Vanhaeverbeek, M.; Ducobu, J.; et al. Inhibition of the myeloperoxidase chlorinating activity by non-steroidal anti-inflammatory drugs: Flufenamic acid and its 5-chloro-derivative directly interact with a recombinant human myeloperoxidase to inhibit the synthesis of hypochlorous acid. Eur. J. Pharmacol. 2007, 570, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Kohnen, S.; Franck, T.; Van Antwerpen, P.; Boudjeltia, K.Z.; Mouithys-Mickalad, A.; Deby, C.; Moguilevsky, N.; Deby-Dupont, G.; Lamy, M.; Serteyn, D. Resveratrol inhibits the activity of equine neutrophil myeloperoxidase by a direct interaction with the enzyme. J. Agric. Food Chem. 2007, 55, 8080–8087. [Google Scholar] [CrossRef] [PubMed]
- Zeraik, M.L.; Ximenes, V.F.; Regasini, L.O.; Dutra, L.A.; Silva, D.H.; Fonseca, L.M.; Coelho, D.; Machado, S.A.; Bolzani, V.S. 4′-Aminochalcones as novel inhibitors of the chlorinating activity of myeloperoxidase. Curr. Med. Chem. 2012, 19, 5405–5413. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, M.; Carmine, T.C.; Gerber, C.; Niethammer, D.; Bruchelt, G. Evidence for the activation of myeloperoxidase by f-Meth-Leu-Phe prior to its release from neutrophil granulocytes. Biochem. Biphy. Res. Commun. 1997, 232, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Franck, T.; Kohnen, S.; de la Rebiere, G.; Deby-Dupont, G.; Deby, C.; Niesten, A.; Serteyn, D. Activation of equine neutrophils by phorbol myristate acetate or N-formyl-methionyl-leucyl-phenylalanine induces a different response in reactive oxygen species production and release of active myeloperoxidase. Vet. Immunol. Immunopathol. 2009, 130, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Faith, M.; Sukumaran, A.; Pulimood, A.B.; Jacob, M. How reliable an indicator of inflammation is myeloperoxidase activity? Clin. Chim. Acta 2008, 396, 23–25. [Google Scholar] [CrossRef] [PubMed]
- Lefkowitz, D.L.; Lefkowitz, S.S. Microglia and myeloperoxidase: A deadly partnership in neurodegenerative disease. Free Radic. Biol. Med. 2008, 45, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Elgazar-Carmon, V.; Rudich, A.; Hadad, N.; Levy, R. Neutrophils transiently infiltrate intra-abdominal fat early in the course of high-fat feeding. J. Lipid Res. 2008, 49, 1894–1903. [Google Scholar] [CrossRef] [PubMed]
- Olza, J.; Aguilera, C.M.; Gil-Campos, M.; Leis, R.; Bueno, G.; Martinez-Jimenez, M.D.; Valle, M.; Canete, R.; Tojo, R.; Moreno, L.A.; et al. Myeloperoxidase is an early biomarker of inflammation and cardiovascular risk in prepubertal obese children. Diabetes Care 2012, 35, 2373–2376. [Google Scholar] [CrossRef] [PubMed]
- VanderVeen, L.A.; Hisham, M.F.; Nechev, L.V.; Harris, T.M.; Harris, C.M.; Mamett, L.D. Evaluation of the mutagenic potential of the principal DNA adduct of acrolein. J. Biol. Chem. 2001, 276, 9066–9070. [Google Scholar] [CrossRef] [PubMed]
- Feyler, A.; Voho, A.; Bouchardy, C.; Kuokkanen, K.; Dayer, P.; Hirvonen, A.; Benhamou, S. Point: myeloperoxidase-463G3A polymorphism and lung cancer risk. Cancer Epidemiol. Biomarkers Prev. 2002, 11, 1550–1554. [Google Scholar] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, H.; Al-Shabrawey, M.; Caldwell, R.W.; Caldwell, R.B. Inflammation and diabetic retinal microvascular complications. J. Cardiovasc. Dis. Res. 2011, 2, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Urbancic, M.; Prevodnik, K.V.; Petrovic, D.; Petrovic, G.M. A flow cytometric analysis of vitreous inflammatory cells in patients with proliferative diabetic retinopathy. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Malle, E.; Marsche, G.; Arnhold, J.; Davies, M.J. Modification of low density lipoprotein by myeloperoxidase-derived oxidants and reagent hypochlorous acid. Biochim. Biophys. Acta 2006, 1761, 392–415. [Google Scholar] [CrossRef] [PubMed]
- Beard, M.R.; Jones, B.E. Hepatitis C virus and oxidative stress: A dangerous liaison. Future Virol. 2006, 1, 223–232. [Google Scholar] [CrossRef]
- Altamirano, J.; Miquel, R.; Katoonizadeh, A.; Abraldes, J.G.; Duarte-Rojo, A.; Louvet, A.; Augustin, S.; Mookerjee, R.P.; Michelena, J.; Smyrk, T.C.; et al. A histologic scoring system for prognosis of patients with alcoholic hepatitis. Gastroenterology 2014, 146, 1231–1239. [Google Scholar] [CrossRef] [PubMed]
- Cohen, T.S.; Prince, A. Cystic fibrosis: A mucosal immunodeficiency syndrome. Nat. Med. 2012, 18, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Gray, E.; Thomas, T.L.; Betmouni, S.; Scolding, N.; Love, S. Elevated myeloperoxidase activity in white matter in multiple sclerosis. Neurosci. Lett. 2008, 444, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Green, P.S.; Mendez, A.J.; Jacob, J.S.; Crowley, J.R.; Growdon, W.; Hyman, B.T.; Heinecke, J.W. Neuronal expression of myeloperoxidase is increased in Alzheimer’s disease. J. Neurochem. 2004, 90, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Teismann, P. Myeloperoxidase in the neurodegenerative process of Parkinson’s disease. Dtsch. Med. Wochenschr. 2014, 139, 99–102. [Google Scholar] [PubMed]
- Koziol-Montewka, M.; Kolodziejek, A.; Oles, J. Study of myeloperoxidase role in antituberculosis defense in the context of cytokine activation. Inflammation 2004, 28, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Monteseirín, J.; Bonilla, I.; Camacho, J.; Conde, J.; Sobrino, F. Elevated secretion of myeloperoxidase by neutrophils from asthmatic patients: The effect of immunotherapy. J. Allergy Clin. Immunol. 2001, 107, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Schiller, J.; Fuchs, B.; Arnhold, J.; Arnhold, K. Contribution of reactive oxygen species to cartilage degradation in rheumatic diseases: Molecular pathways, diagnosis and potential therapeutic strategies. Curr. Med. Chem. 2003, 10, 2123–2145. [Google Scholar] [CrossRef] [PubMed]
- Prokopowicz, Z.; Marcinkiewicz, J.; Katz, D.R.; Chain, B.M. Neutrophil myeloperoxidase: Soldier and statesman. Arch. Immunol. Ther. Exp. 2012, 60, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Demoly, P.; Crampette, L.; Mondain, M.; Enander, I.; Jones, I.; Bousquet, J. Myeloperoxidase and interleukin-8 levels in chronic sinusitis. Clin. Exp. Allergy 1997, 27, 672–675. [Google Scholar] [CrossRef] [PubMed]
- Panneerselvam, S.; Arumugam, G. A biochemical study on the gastroprotective effect of hydroalcoholic extract of Andrographis paniculata in rats. Indian J. Pharmacol. 2011, 43, 402–408. [Google Scholar] [PubMed]
- Nishida, K.; Ohta, Y.; Ishiguro, I. Contribution of NO synthase to neutrophil infiltration in the gastric mucosal lesions in rats with water immersion restraint stress. FEBS Lett. 1998, 425, 243–248. [Google Scholar] [CrossRef]
- Warzecha, Z.; Ceranowicz, D.; Dembinski, A.; Ceranowicz, P.; Cieszkowski, J.; Kuwahara, A.; Kato, I.; Dembinski, M.; Konturek, P.C. Ghrelin accelerates the healing of cysteamine-induced duodenal ulcers in rats. Med. Sci. Monit. 2012, 18, 181–187. [Google Scholar] [CrossRef]
- Ceranowicz, P.; Warzecha, Z.; Cieszkowski, J.; Ceranowicz, D.; Kusnierz-Cabala, B.; Bonior, J.; Jaworek, J.; Ambrozy, T.; Gil, K.; Olszanecki, R.; et al. Essential role of growth hormone and IGF-1 in therapeutic effect of ghrelin in the course of acetic acid-induced colitis. Int. J. Mol. Sci. 2017, 18, 1118. [Google Scholar] [CrossRef] [PubMed]
- Matuszyk, A.; Ceranowicz, P.; Warzecha, Z.; Cieszkowski, J.; Ceranowicz, D.; Galazka, K.; Bonior, J.; Jaworek, J.; Bartus, K.; Gil, K.; et al. Exogenous ghrelin accelerates the healing of acetic acid-induced colitis in rats. Int. J. Mol. Sci. 2016, 17, 1455. [Google Scholar] [CrossRef] [PubMed]
- Dembinski, A.; Warzecha, Z.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Gosiewski, T.; Bulanda, M.; Kusnierz-Cabala, B.; Galazka, K.; Konturek, P.C. Synergic interaction of Rifaximinand and Mutaflor (Escherichia coli Nissle 1917) in the treatment of acetic acid-induced colitis in rats. Gastroenterol. Res. Pract. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Chooklin, S.; Pereyaslov, A.; Bihalskyy, I. Pathogenic role of myeloperoxidase in acute pancreatitis. Hepatobiliary Pancreat. Dis. Int. 2009, 8, 627–631. [Google Scholar] [PubMed]
- Klangprapan, S.; Chaiyarit, P.; Hormdee, D.; Kampichai, A.; Khampitak, T.; Daduang, J.; Tavichakorntrakool, R.; Panijpan, B.; Boonsiri, P. Salivary myeloperoxidase, assessed by 3,3′-diaminobenzidine colorimetry, can differentiate periodontal patients from nonperiodontal subjects. Enzyme Res. 2016, 2016, 7517928. [Google Scholar] [CrossRef] [PubMed]
- Loria, V.; Dato, I.; Graziani, F.; Biasucci, L. Myeloperoxidase: A new biomarker of inflammation in ischemic heart disease and acute coronary syndromes. Mediat. Inflamm. 2008, 2008. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, D.L. Mechanism responsible for increased vascular permeability in acute inflammation. Inflamm. Res. 1973, 3, 297–306. [Google Scholar] [CrossRef]
- Phillipson, M.; Kubes, P. The neutrophil in vascular inflammation. Nat. Med. 2011, 17, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Selders, G.S.; Fetz, A.E.; Radic, M.Z.; Bowlin, G.L. An overview of the role of neutrophils in innate immunity, inflammation and host-biomaterial integration. Regen. Biomater. 2017, 4, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, T.A.; Best, T.M.; Merrick, M.A. The dual roles of neutrophils and macrophages in inflammation: A critical balance between tissue damage and repair. J. Athl. Train. 2006, 41, 457–465. [Google Scholar] [PubMed]
- Zhang, R.; Brennan, M.-L.; Shen, Z.; MacPherson, J.C.; Schmitt, D.; Molenda, C.E.; Hazen, S.L. Myeloperoxidase functions as a major enzymatic catalyst for initiation of lipid peroxidation at sites of inflammation. J. Biol. Chem. 2002, 277, 46116–46122. [Google Scholar] [CrossRef] [PubMed]
- Funk, C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science 2001, 294, 1871–1875. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.S.; Pontsler, A.V.; Marathe, G.K.; Harrison, K.A.; Murphy, R.C.; Hinshaw, J.C.; Prestwich, G.D.; Hilaire, A.S.; Prescott, S.M.; Zimmerman, G.A.; et al. oxidized alkyl phospholipids are specific, high affinity peroxisome proliferator-activated receptor Y ligands and agonists. J. Biol. Chem. 2001, 276, 16015–16023. [Google Scholar] [CrossRef] [PubMed]
- Hazen, S.L.; Hsu, F.F.; Gaut, J.P.; Crowley, J.R.; Heinecke, J.W. Modification of proteins and lipids by myeloperoxidase. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; pp. 88–105. [Google Scholar]
- Van Dalen, C.J.; Winterbourn, C.C.; Senthilmohan, R.; Kettle, A.J. Nitrate as a substrate and inhibitor of myeloperoxidase, implication for nitration and hypochlorous acid production at sites of inflammation. J. Biol. Chem. 2000, 275, 11638–11644. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.L.; Wu, W.; Fu, X.; Shen, Z.; Song, W.; Frost, H.; Vadseth, C.; Narine, L.; Lenkiewicz, E.; Borchers, M.T.; et al. A tale of two controversies, defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J. Biol. Chem. 2002, 277, 17415–17427. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.M.S.N.; da Silva, N.P.; Sato, E.I. Increased myeloperoxidase plasma levels in rheumatoid arthritis. Rheumatol. Int. 2012, 32, 1606–1609. [Google Scholar] [CrossRef] [PubMed]
- Stamp, L.K.; Khalilova, I.; Tarr, J.M.; Senthilmohan, R.; Turner, R.; Haigh, R.C.; Winyard, P.G.; Kettle, A.J. Myeloperoxidase and oxidative stress in rheumatoid arthritis. Rheumatology 2012, 51, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Gowder, S.J.G. Glutathione peroxidase: A potential marker for the most common diseases and disorders. Recent Patents Biomarkers 2014, 4, 43–52. [Google Scholar] [CrossRef]
- Morgan, P.E.; Sturgess, A.D.; Davies, M.J. Increased levels of serum protein oxidation and correlation with disease activity in systemic lupus erythematosus. Arthritis Rheumatol. 2005, 52, 2069–2079. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.W.; Hallett, M.B. Seeing the wood for the trees: The forgotten role of neutrophils in rheumatoid arthritis. Immunol. Today 1997, 18, 320–324. [Google Scholar] [CrossRef]
- Wright, H.L.; Moots, R.J.; Bucknall, R.C.; Edward, S.W. Neutrophil function in inflammation and inflammatory diseases. Rheumatology 2010, 49, 1618–1631. [Google Scholar] [CrossRef] [PubMed]
- Kettle, A.J.; Winterbourn, C.C. Myeloperoxidase: A key regulator of neutrophil oxidant production. Redox Rep. 1997, 3, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Ece, A.; Kelekçi, S.; Hekimoglu, A.; Kocamaz, H.; Balik, H.; Yolbas, I.; Erel, O. Neutrophil activation, protein oxidation and ceruloplasmin levels in children with Henoch–Schönlein purpura. Pediat. Nephrol. 2007, 22, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Kassim, S.Y.; Parks, W.C.; Heinecke, J.W. Hypochlorous acid generated by myeloperoxidase modifies adjacent tryptophan and glycine residues in the catalytic domain of matrix metalloproteinase-7 (matrilysin): An oxidative mechanism for restraining proteolytic activity during inflammation. J. Biol. Chem. 2003, 278, 28403–28409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Brennan, M.L.; Fu, X.; Aviles, R.J.; Pearce, G.L.; Penn, M.S.; Topol, E.J.; Sprecher, D.L.; Hazen, S.L. Association between myeloperoxidase levels and risk of coronary artery disease. JAMA 2001, 286, 2136–2142. [Google Scholar] [CrossRef] [PubMed]
- Cavusoglu, E.; Ruwende, C.; Eng, C.; Marmur, J.D. Usefulness of baseline plasma myeloperoxidase levels as an independent predictor of myocardial infarction at two years in patients presenting with acute coronary syndrome. Am. J. Cardiol. 2007, 99, 1364–1368. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Brennan, M.L.; Philip, K.; Tong, W.; Mann, S.; van Lente, F.; Hazen, S.L. Plasma myeloperoxidase levels in patients with chronic heart failure. Am. J. Cardiol. 2006, 98, 796–799. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Tong, W.; Troughton, R.W.; Martin, M.G.; Shrestha, K.; Borowski, A.; Jasper, S.; Hazen, S.L.; Klein, A.L. Prognostic value and echocardiographic determinants of plasma myeloperoxidase levels in chronic heart failure. J. Am. Coll. Cardiol. 2007, 49, 2364–2370. [Google Scholar] [CrossRef] [PubMed]
- Ndrepepa, G.; Braun, S.; Mehilli, J.; von Beckerath, N.; Schomig, A.; Kastrati, A. Myeloperoxidase level in patients with stable coronary artery disease and acute coronary syndromes. Eur. J. Clin. Investig. 2008, 38, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Roman, R.M.; Camargo, P.V.; Borges, F.K.; Rossini, A.P.; Polanczyk, C.A. Prognostic value of myeloperoxidase in coronary artery disease: Comparison of unstable and stable angina patients. Coron. Atery Dis. 2010, 21, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.L.; Penn, M.S.; Van Lente, F.; Nambi, V.; Shishehbor, M.H.; Aviles, R.J.; Goormastic, M.; Pepoy, M.L.; McErlean, E.S.; Topol, E.J.; et al. Prognostic value of myeloperoxidase in patients with chest pain. N. Engl. J. Med. 2003, 349, 1595–1604. [Google Scholar] [CrossRef] [PubMed]
- Baldus, S.; Heeschen, C.; Meinertz, T.; Zeiher, A.M.; Eiserich, J.P.; Münze, L.T.; Simoons, M.L.; Hamm, C.W. Myeloperoxidase serum levels predict risk in patients with acute coronary syndromes. Circulation 2003, 108, 1440–1445. [Google Scholar] [CrossRef] [PubMed]
- Mocatta, T.J.; Pilbrow, A.P.; Cameron, V.A.; Senthilmohan, R.; Frampton, C.M.; Richards, A.M.; Winterbourn, C.C. Plasma concentrations of myeloperoxidase predict mortality after myocardial infarction. J. Am. Coll. Cardiol. 2007, 49, 1993–2000. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Galkina, E.; Ley, K. Immune and inflammatory mechanisms of atherosclerosis. Annu. Rev. Immunol. 2009, 27, 165–197. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Changing concepts of atherogenesis. J. Intern. Med. 2000, 247, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Stocker, R.; Keaney, J.F., Jr. Role of oxidative modification in atherosclerosis. Physiol. Rev. 2004, 84, 1381–1478. [Google Scholar] [CrossRef] [PubMed]
- Traub-Dargatz, J.L.; Salman, M.D.; Voss, J.L. Medical problems of adult horses, as ranked by equine practitioners. J. Am. Vet. Med. Assoc. 1991, 198, 1745–1747. [Google Scholar] [PubMed]
- Delporte, C.; Boudjeltia, K.Z.; Noyon, C.; Furtmuller, P.G.; Nuyens, V.; Slomianny, M.-C.; Madhoun, P.; Desmet, J.-M.; Raynal, P.; Dufour, D.; et al. Impact of myeloperoxidase–LDL interactions on enzyme activity and subsequent posttranslational oxidative modifications of apoB-100. J. Lipid Res. 2014, 55, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Hazell, L.J.; Stocker, R. Oxidation of low-density lipoprotein with hypochlorite causes transformation of the lipoprotein into a high-uptake form for macrophages. Biochem. J. 1993, 290, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, S.; Oh, Y.; Bandyopadhyay, G.; Li, D.; Xu, J.; McNelis, J.; Lu, M.; Li, P.; Yan, Q.; Zhu, Y.; et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat. Med. 2012, 18, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Xie, Z.; Zhang, W.; Zhou, J.; Wu, Y.; Zhang, M.; Zhu, H.; Zou, M.H. Myeloperoxidase deletion prevents high-fat diet-induced obesity and insulin resistance. Diabetes 2014, 63, 4172–4185. [Google Scholar] [CrossRef] [PubMed]
- Zur, B.; Look, M.; Holdenrieder, S.; Stoffel-Wagner, B. Elevated plasma myeloperoxidase concentration in adults with obesity. Clin. Chim. Acta 2011, 412, 1891–1892. [Google Scholar] [CrossRef] [PubMed]
- Andrade, V.L.; Petruceli, E.; Belo, V.A.; Andrade-Fernandes, C.M.; Caetano Russi, C.V.; Bosco, A.A.; Tanus-Santos, J.E.; Sandrim, V.C. Evaluation of plasmatic MMP-8, MMP-9, TIMP-1 and MPO levels in obese and lean women. Clin. Biochem. 2012, 45, 412–415. [Google Scholar] [CrossRef] [PubMed]
- Heinecke, J.W.; Goldberg, I.J. Myeloperoxidase: A therapeutic target for preventing insulin resistance and the metabolic sequelae of obesity? Diabetes 2014, 63, 4001–4003. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, W.; Lu, J.; Weng, J.; Jia, W.; Ji, L.; Xiao, J.; Shan, Z.; Liu, J.; Tian, H.; Ji, Q.; et al. Prevalence of diabetes among men and women in China. N. Engl. J. Med. 2010, 362, 1090–1101. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.I.; Duncan, B.B.; Sharrett, A.R.; Lindberg, G.; Savage, P.J.; Offenbacher, S.; Azambuja, M.I.; Tracy, R.P.; Heiss, G. Markers of inflammation and prediction of diabetes mellitus in adults (Atherosclerosis Risk in Communities study): A cohort study. Lancet 1999, 353, 1649–1652. [Google Scholar] [CrossRef]
- Ford, E.S. Leukocyte count, erythrocyte sedimentation rate, and diabetes incidence in a national sample of U.S. adults. Am. J. Epidemiol. 2002, 155, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Ohshita, K.; Yamane, K.; Hanafusa, M.; Mori, H.; Mito, K.; Okubo, M.; Hara, H.; Kohno, N. Elevated white blood cell count in subjects with impaired glucose tolerance. Diabetes Care 2004, 27, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Borato, D.C.; Parabocz, G.C.; Riba, J.T.; Netto, H.P.; Erdmann, F.C.; Wiecheteck, L.D.; Manente, F.A.; Mello, L.R.; Bello, C.; dos Santos, F.A. Biomarkers in obesity: Serum myeloperoxidase and traditional cardiac risk parameters. Exp. Clin. Endocrinol. Diabetes 2016, 124, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Ray, R.S.; Katyal, A. Myeloperoxidase: Bridging the gap in neurodegeneration. Neurosci. Biobehav. Rev. 2016, 68, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Vaccarino, V.; Johnson, B.D.; Sheps, D.S.; Reis, S.E.; Kelsey, S.F.; Bittner, V.; Rutledge, T.; Shaw, L.J.; Sopko, G.; Bairey Merz, C.N. Depression, inflammation, and incident cardiovascular disease in women with suspected coronary ischemia. J. Am. Coll. Cardiol. 2007, 50, 2044–2050. [Google Scholar] [CrossRef] [PubMed]
- Selek, S.; Altindag, A.; Saracoglu, G.; Aksoy, N. Oxidative markers of myeloperoxidase and catalase and their diagnostic performance in bipolar disorder. J. Affect. Dis. 2015, 181, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Kaji, Y.; Usui, T.; Ishida, S.; Yamashiro, K.; Moore, T.C.; Moore, J.; Yamamoto, Y.; Yamamoto, H.; Adamis, A.P. Inhibition of diabetic leukostasis and bloodretinal barrier breakdown with a soluble form of a receptor for advanced glycation end products. Investig. Ophthalmol. Vis. Sci. 2007, 48, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Urbancic, M.; Stunf, S.; Milutinovic, A.Z.; Petrovic, D.; Petrovic, M.G. Epiretinal membrane inflammatory cell density might reflect the activity of proliferative diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8576–8582. [Google Scholar]
- Miyamoto, K.; Ogura, Y. Pathogenetic potential of leukocytes in diabetic retinopathy. Semin. Ophthalmol. 1999, 14, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Khosrof, S.; Bursell, S.E.; Rohan, R.; Murata, T.; Clermont, A.C.; Aiello, L.P.; Ogura, Y.; Adamis, A.P. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc. Natl. Acad. Sci. USA 1999, 96, 10836–10841. [Google Scholar] [CrossRef] [PubMed]
- Joussen, A.M.; Murata, T.; Tsujikawa, A.; Kirchhof, B.; Bursell, S.E.; Adamis, A.P. Leukocyte-mediated endothelial cell injury and death in the diabetic retina. Am. J. Pathol. 2001, 158, 147–152. [Google Scholar] [CrossRef]
- Rarok, A.A.; Limburg, P.C.; Kallenberg, C.G. Neutrophil-activating potential of antineutrophil cytoplasm autoantibodies. J. Leukoc. Biol. 2003, 74, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.M.; Kamesh, L.; Savage, C.O. Translating basic science into patient therapy for ANCA-associated small vessel vasculitis. Clin. Sci. 2005, 108, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Rehm, J.; Mathers, C.; Popova, S.; Thavorncharoensap, M.; Teerawattananon, Y.; Patra, J. Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009, 373, 2223–2233. [Google Scholar] [CrossRef]
- Ceni, E.; Mello, T.; Galli, A. Pathogenesis of alcoholic liver disease: Role of oxidative metabolism. World J. Gastroenterol. 2014, 20, 17756–17772. [Google Scholar] [CrossRef] [PubMed]
- Albillos, A.; Lario, M.; Álvarez-Mon, M. Cirrhosis-associated immune dysfunction: Distinctive features and clinical relevance. J. Hepatol. 2014, 61, 1385–1396. [Google Scholar] [CrossRef] [PubMed]
- Ramaiah, S.K.; Jaeschke, H. Role of neutrophils in the pathogenesis of acute inflammatory liver injury. Toxicol. Pathol. 2007, 35, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Jaeschke, H.; Ho, Y.S.; Fisher, M.A.; Lawson, J.A.; Farhood, A. Glutathione peroxidase deficient mice are more susceptible to neutrophil-mediated hepatic parenchymal cell injury during endotoxemia: Importance of an intracellular oxidant stress. Hepatology 1999, 29, 443–445. [Google Scholar] [CrossRef] [PubMed]
- Poli, G. Pathogenesis of liver fibrosis: Role of oxidative stress. Mol. Aspects Med. 2000, 21, 49–98. [Google Scholar] [CrossRef]
- Eash, K.J.; Means, J.M.; White, D.W.; Link, D.C. CXCR4 is a key regulator of neutrophil release from the bone marrow under basal and stress granulopoiesis conditions. Blood 2009, 113, 4711–4719. [Google Scholar] [CrossRef] [PubMed]
- Taieb, J.; Delarche, C.; Paradis, V.; Mathurin, P.; Grenier, A.; Crestani, B.; Dehoux, M.; Thabut, D.; Gougerot-Pocidalo, M.-A.; Thierry Poynard, T.; et al. Polymorphonuclear neutrophils are a source of hepatocyte growth factor in patients with severe alcoholic hepatitis. J. Hepatol. 2002, 36, 342–348. [Google Scholar] [CrossRef]
- Singh, V.; Sharma, A.K.; Narasimhan, R.L.; Bhalla, A.; Sharma, N.; Sharma, R. Granulocyte colony stimulating factor in severe alcoholic hepatitis: A randomized pilot study. Am. J. Gastroenterol. 2014, 109, 1417–1423. [Google Scholar] [CrossRef] [PubMed]
- Stadlbauer, V.; Mookerjee, R.P.; Hodges, S.; Wright, G.A.; Davies, N.A.; Jalan, R. Effect of probiotic treatment on deranged neutrophil function and cytokine responses in patients with compensated alcoholic cirrhosis. J. Hepatol. 2008, 48, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.A. Role of C5 activation products in sepsis. Sci. World J. 2010, 10, 2395–2402. [Google Scholar] [CrossRef] [PubMed]
- Markwick, L.J.; Riva, A.; Ryan, J.M.; Cooksley, H.; Palma, E.; Tranah, T.H.; Manakkat Vijay, G.K.; Vergis, N.; Thursz, M.; Evans, A.; et al. Blockade of PD1 and TIM3 restores innate and adaptive immunity in patients with acute alcoholic hepatitis. Gastroenterology 2015, 148, 590–602. [Google Scholar] [CrossRef] [PubMed]
- Juttner, B.; Younes, A.; Weissig, A.; Ahrens, J.; Becker, T.; Scheinichen, D. Reduced post-operative neutrophil activation in liver transplant recipients suffering from post-hepatitic cirrhosis. Clin. Transplant. 2009, 23, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Al-Salihi, M.; Reichert, E.; Fitzpatrick, F.A. Influence of myeloperoxidase on colon tumor occurrence in inflamed versus non-inflamed colons of ApcMin/+ mice. Redox Biol. 2015, 6, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; Liu, C.; Feng, J.; Xu, Q. Ding Association between the myeloperoxidase gene polymorphisms and the susceptibility to prostate cancer: A case-control study in a Chinese population. Actas Urol. Esp. 2013, 37, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Hoy, A.; Tregouet, D.; Leininger-Muller, B.; Poirier, O.; Maurice, M.; Sass, C.; Siest, G.; Tiret, L.; Visvikis, S. Serum myeloperoxidase concentration in a healthy population: Biological variations, familial resemblance and new genetic polymorphisms. Eur. J. Hum. Genet. 2001, 9, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Nauseef, W.M.; Brigham, S.; Cogley, M. Hereditary myeloperoxidase deficiency due to a missense mutation of arginine 569 to trypophan. J. Biol. Chem. 1994, 269, 1212–1216. [Google Scholar] [PubMed]
- Trush, M.A.; Seed, J.L.; Kensler, T.W. Oxidant-dependent metabolic activation of polycyclic aromatic hydrocarbons by phorbol ester stimulated human polymorphonuclear leukocytes: Possible link between inflammation and cancer. Proc. Natl. Acad. Sci. USA 1985, 82, 5194–5198. [Google Scholar] [CrossRef] [PubMed]
- Eastmond, D.A.; French, R.C.; Ross, D.; Smith, M.T. Metabolic activation of diethylstilbestrol by stimulated human leukocytes. Cancer Lett. 1987, 35, 79–86. [Google Scholar] [CrossRef]
- Stevens, J.F.; Maier, C.S. Acrolein: Sources, metabolism, and biomolecular interactions relevant to human health and disease. Mol. Nutr. Food Res. 2008, 52, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Zarkovic, K.; Uchida, K.; Kolenc, D.; Hlupic, L.; Zarkovic, N. Tissue distribution of lipid peroxidation product acrolein in human colon carcinogenesis. Free Radic. Res. 2006, 40, 543–552. [Google Scholar] [CrossRef] [PubMed]
- DeNardo, D.G.; Coussens, L.M. Inflammation and breast cancer. Balancing immune response: Crosstalk between adaptive and innate immune cells during breast cancer progression. Breast Cancer Res. 2007, 9, 212. [Google Scholar] [CrossRef] [PubMed]
- Van der Veen, B.S.; de Winther, M.P.J.; Heeringa, P. Myeloperoxidase: Molecular mechanisms of action and their relevance to human health and disease. Antioxid. Redox Signal. 2009, 11, 2899–2937. [Google Scholar] [CrossRef] [PubMed]
- Jesneck, J.L.; Mukherjee, S.; Yurkovetsky, Z.; Clyde, M.; Marks, J.R.; Lokshin, A.E.; Lo, J.Y. Do serum biomarkers really measure breast cancer? BMC Cancer 2009, 9, 164. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Tamimi, R.M.; Hankinson, S.E.; Hunter, D.J.; Han, J. A prospective study of genetic polymorphism in MPO, antioxidant status, and breast cancer risk. Breast Cancer Res. Treat. 2009, 113, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Jilani, M.; Vincent, T.; Faraji, H.; Giles, F.J.; Estey, E.; Kantarjian, H.P.; Albitar, M. Clinical relevance of circulating myeloperoxidase (MPO) in acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS). Blood 2004, 104, 1073. [Google Scholar]
- Lynch, S.V.; Bruce, K.D. The cystic fibrosis airway microbiome. Cold Spring Harb. Perspect. Med. 2013, 3, a009738. [Google Scholar] [CrossRef] [PubMed]
- Sagel, S.D.; Wagner, B.D.; Anthony, M.M.; Emmett, P.; Zemanick, E.T. Sputum biomarkers of inflammation and lung function decline in children with cystic fibrosis. Am. J. Respir. Crit. Care Med. 2012, 186, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Sly, P.D.; Gangell, C.L.; Chen, L.; Ware, R.S.; Ranganathan, S.; Mott, L.S.; Conor, P.; Murray, C.P.; Stick, S.M. Risk factors for bronchiectasis in children with cystic fibrosis. N. Engl. J. Med. 2013, 368, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinraunch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, C.; Patriarca, P.; Solero, G.P.; Baralle, F.E.; Romano, M. Genetic studies on myeloperoxidase deficiency in Italy. Jpn. J. Infect. Dis. 2004, 57, S10e2. [Google Scholar]
- Rudolph, T.K.; Wipper, S.; Reiter, B.; Rudolph, V.; Coym, A.; Detter, C.; Lau, D.; Klinke, A.; Friedrichs, K.; Rau, T.; et al. Myeloperoxidase deficiency preserves vasomotor function in humans. Eur. Heart J. 2012, 33, 1625–1634. [Google Scholar] [CrossRef] [PubMed]
- Nunoi, H.; Kohi, F.; Kajiwara, H.; Suzuki, K. Prevalence of inherited myeloperoxidase deficiency in Japan. Microbiol. Immunol. 2003, 47, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, Y.Y.; Kameoka, Y.; Persad, A.S.; Koi, F.; Yamagoe, S.; Hashimoto, K.; Suzuki, K. Novel missense mutation found in a Japanese patient with myeloperoxidase deficiency. Gene 2004, 327, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Persad, A.S.; Kameoka, Y.; Kanda, S.; Niho, Y.; Suzuki, K. Arginine to cysteine mutation (R499C) found in a Japanese patient with complete myeloperoxidase deficiency. Gene Expr. 2006, 13, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, E.; Huitema, M.G.; Mulder, A.H.; Heeringa, P.; van Goor, H.; Tervaert, J.W.; Weening, J.J.; Kallenberg, C.G. Neutrophil activation in vitro and in vivo in Wegener’s granulomatosis. Kidney Int. 1994, 45, 1120–1131. [Google Scholar] [CrossRef] [PubMed]
- Wibke, B.T.; Allen, P.M. Essential role of neutrophils in the initiation and progression of a murine model of rheumatoid arthritis. J. Immunol. 2001, 167, 1601–1608. [Google Scholar] [CrossRef]
- Singbartl, K.; Bockhorn, S.G.; Zarbock, A.; Schmolke, M.; Van Aken, H. T cell modulates neutrophils-dependent acute renal failure during endotoxemia: Critical role of CD28. J. Am. Soc. Nephrol. 2005, 16, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Lanza, F. Clinical manifestation of myeloperoxidase deficiency. J. Mol. Med. 1998, 76, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Flint, S.M.; McKinney, E.F.; Smith, K.G. Emerging concepts in the pathogenesis of antineutrophil cytoplasmic antibody-associated vasculitis. Curr. Opin. Rheumatol. 2015, 27, 197–203. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).