Healthcare Cost and Utilization before and after Diagnosis of Pseudomonas aeruginosa among Patients with Non-Cystic Fibrosis Bronchiectasis in the U.S.
Abstract
:1. Introduction
2. Materials and Methods
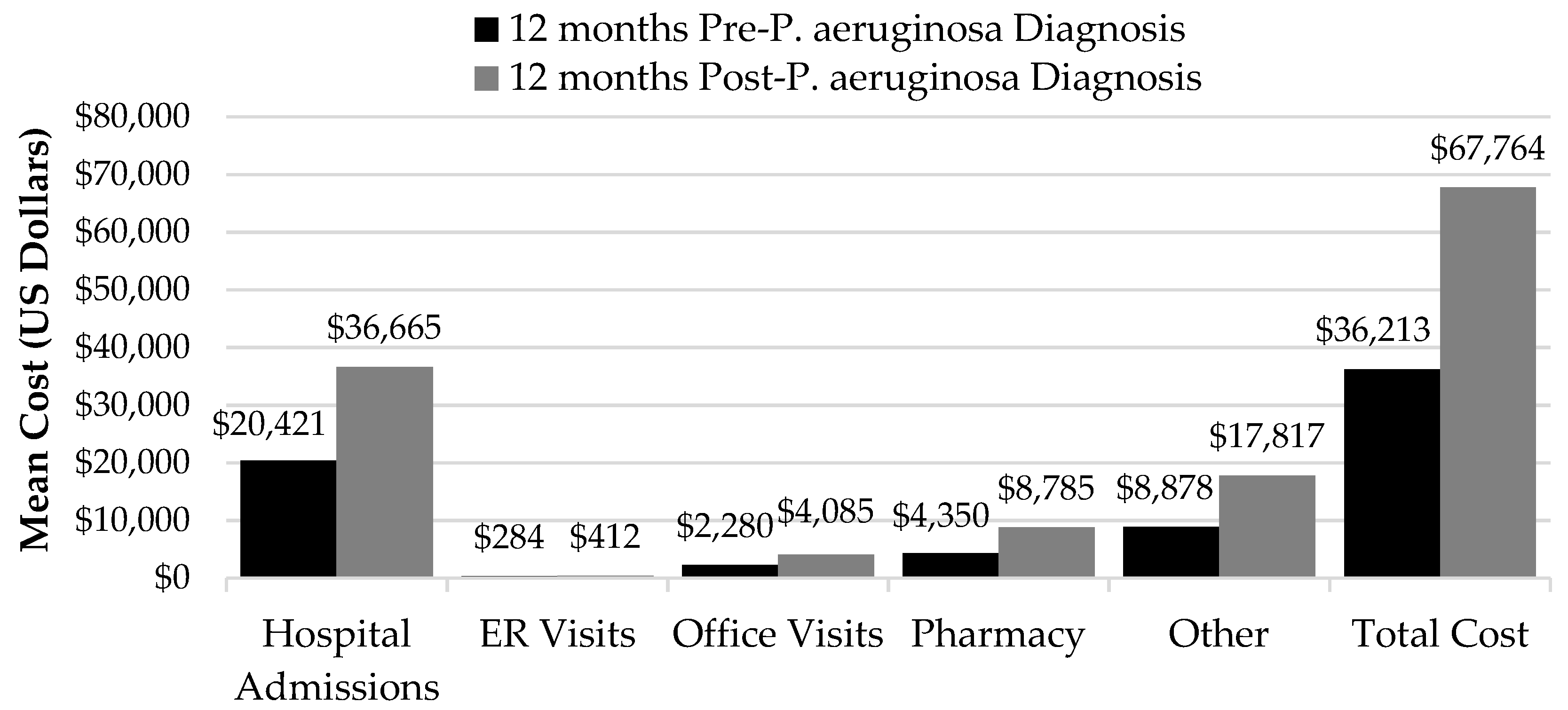
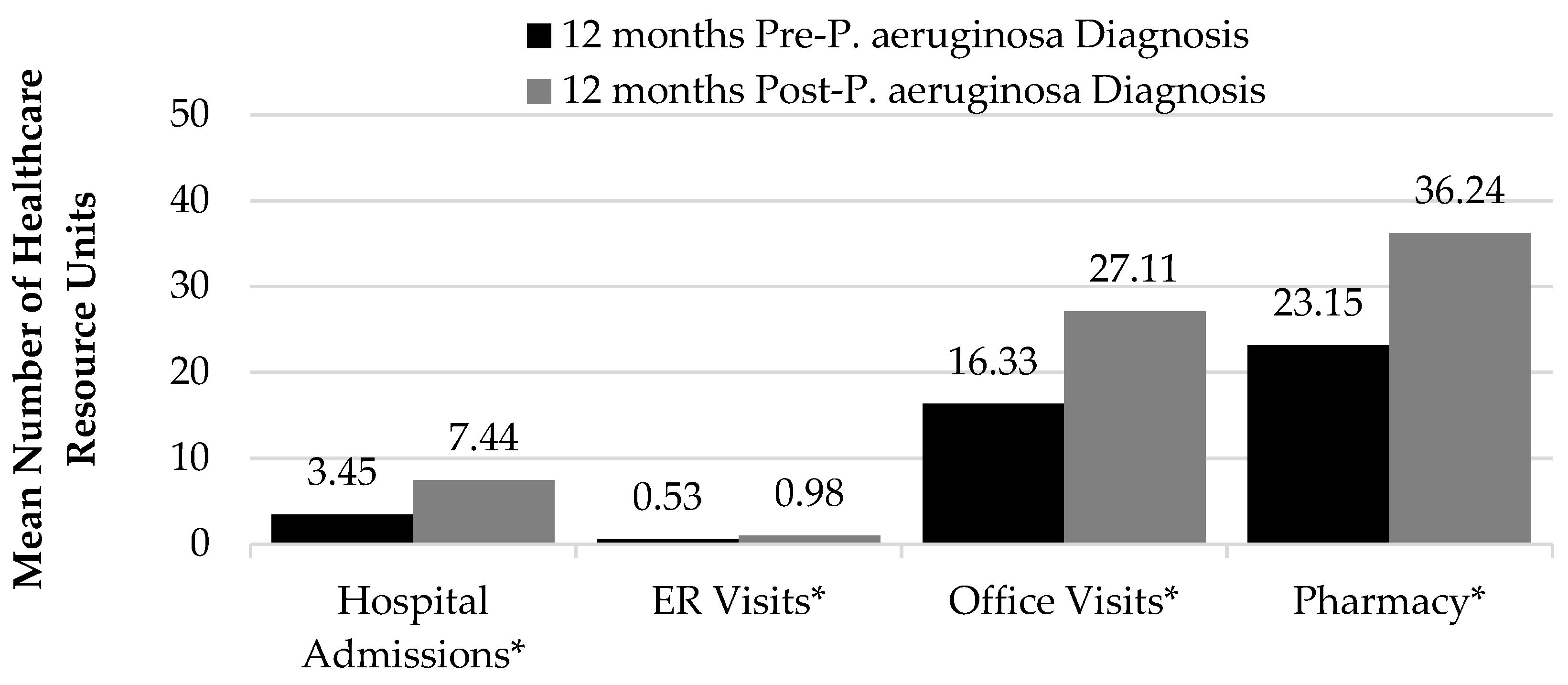
3. Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weycker, D.; Hansen, G.L.; Seifer, F.D. Prevalence and incidence of noncystic fibrosis bronchiectasis among US adults in 2013. Chronic Respir. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, M.J.; Jary, H.R.; Perry, A.; MacFarlane, J.G.; Hester, K.L.; Small, T.; Molyneux, C.; Perry, J.D.; Walton, K.E.; De Soyza, A. Non cystic fibrosis bronchiectasis: A longitudinal retrospective observational cohort study of pseudomonas persistence and resistance. Respir. Med. 2015, 109, 716–726. [Google Scholar] [CrossRef] [PubMed]
- Weycker, D.; Edelsberg, J.; Oster, G.; Tino, G. Prevalence and economic burden of bronchiectasis. Clin. Pulm. Med. 2005, 12, 205–209. [Google Scholar] [CrossRef]
- Joish, V.N.; Spilsbury-Cantalupo, M.; Operschall, E.; Luong, B.; Boklage, S. Economic burden of non-cystic fibrosis bronchiectasis in the first year after diagnosis from a US health plan perspective. Appl. Health Econ. Health Policy 2013, 11, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.J.; Bara, A.I.; Greenstone, M. Prolonged antibiotics for purulent bronchiectasis. Cochrane Database Syst. Rev. 2003. [Google Scholar] [CrossRef]
- O'Donnell, A.E. Bronchiectasis. Chest 2008, 134, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Felix, L.M.; Grundy, S.; Milan, S.J.; Armstrong, R.; Harrison, H.; Lynes, D.; Spencer, S. Dual antibiotics for non-cystic fibrosis bronchiectasis (protocol). Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
- Cole, P.J. Inflammation: A two-edged sword—the model of bronchiectasis. Eur. J. Respir. Dis. Suppl. 1986, 147, 6–15. [Google Scholar] [PubMed]
- Wilson, R.; Aksamit, T.; Aliberti, S.; De Soyza, A.; Elborn, J.S.; Goeminne, P.; Hill, A.T.; Menendez, R.; Polverino, E. Challenges in managing Pseudomonas aeruginosa in non-cystic fibrosis bronchiectasis. Respir. Med. 2016, 117, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.B.; Jones, P.W.; O'Leary, C.J.; Hansell, D.M.; Cole, P.J.; Wilson, R. Effect of sputum bacteriology on the quality of life of patients with bronchiectasis. Eur. Respir. J. 1997, 10, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Goeminne, P.; Aliberti, S.; McDonnell, M.J.; Lonni, S.; Davidson, J.; Poppelwell, L.; Salih, W.; Pesci, A.; Dupont, L.J.; et al. The bronchiectasis severity index. An international derivation and validation study. Am. J. Respir. Crit. Care Med. 2014, 189, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Goeminne, P.C.; Scheers, H.; Decraene, A.; Seys, S.; Dupont, L.J. Risk factors for morbidity and death in non-cystic fibrosis bronchiectasis: A retrospective cross-sectional analysis of CT diagnosed bronchiectatic patients. Respir. Res. 2012, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Davies, G.; Wells, A.U.; Doffman, S.; Watanabe, S.; Wilson, R. The effect of Pseudomonas aeruginosa on pulmonary function in patients with bronchiectasis. Eur. Respir. J. 2006, 28, 974–979. [Google Scholar] [CrossRef] [PubMed]
- McShane, P.J.; Naureckas, E.T.; Strek, M.E. Bronchiectasis in a diverse US population: Effects of ethnicity on etiology and sputum culture. Chest 2012, 142, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Goeminne, P.C.; Nawrot, T.S.; Ruttens, D.; Seys, S.; Dupont, L.J. Mortality in non-cystic fibrosis bronchiectasis: A prospective cohort analysis. Respir. Med. 2014, 108, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.A.; Turner, S.M.; Bosch, B.J.; Hardy, C.C.; Woodhead, M.A. Lung function in bronchiectasis: The influence of Pseudomonas aeruginosa. Eur. Respir. J. 1996, 9, 1601–1604. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Garcia, M.A.; Soler-Cataluna, J.J.; Perpina-Tordera, M.; Roman-Sanchez, P.; Soriano, J. Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest 2007, 132, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Gao, Y.H.; Xu, G.; Lin, Z.Y.; Tang, Y.; Li, H.M.; Lin, Z.M.; Zheng, J.P.; Chen, R.C.; Zhong, N.S. Characterization of lung function impairment in adults with bronchiectasis. PLoS ONE 2014, 9, e113373. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.B.; van der Gast, C.J.; Cuthbertson, L.; Thomson, S.K.; Bruce, K.D.; Martin, M.L.; Serisier, D.J. Clinical measures of disease in adult non-CF bronchiectasis correlate with airway microbiota composition. Thorax 2013, 68, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Smith, M.P.; McHugh, B.J.; Doherty, C.; Govan, J.R.; Hill, A.T. Short- and long-term antibiotic treatment reduces airway and systemic inflammation in non-cystic fibrosis bronchiectasis. Am. J. Respir. Crit. Care Med. 2012, 186, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.B.; Zain, N.M.; Bruce, K.D.; Burr, L.D.; Chen, A.C.; Rivett, D.W.; McGuckin, M.A.; Serisier, D.J. A novel microbiota stratification system predicts future exacerbations in bronchiectasis. Ann. Am. Thorac. Soc. 2014, 11, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Loebinger, M.R.; Wells, A.U.; Hansell, D.M.; Chinyanganya, N.; Devaraj, A.; Meister, M.; Wilson, R. Mortality in bronchiectasis: A long-term study assessing the factors influencing survival. Eur. Respir. J. 2009, 34, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.T.; Routh, C.; Welham, S. National BTS bronchiectasis audit 2012: Is the quality standard being adhered to in adult secondary care? Thorax 2014, 69, 292–294. [Google Scholar] [CrossRef] [PubMed]
- McShane, P.J.; Naureckas, E.T.; Tino, G.; Strek, M.E. Non-cystic fibrosis bronchiectasis. Am. J. Respir. Crit. Care Med. 2013, 188, 647–656. [Google Scholar] [CrossRef] [PubMed]
- King, P.T.; Holdsworth, S.R.; Freezer, N.J.; Villanueva, E.; Holmes, P.W. Microbiologic follow-up study in adult bronchiectasis. Respir. Med. 2007, 101, 1633–1638. [Google Scholar] [CrossRef] [PubMed]
- Yayan, J.; Ghebremedhin, B.; Rasche, K. Antibiotic resistance of Pseudomonas aeruginosa in pneumonia at a single university hospital center in Germany over a 10-year period. PLoS ONE 2015, 10, e0139836. [Google Scholar] [CrossRef] [PubMed]
- Tunney, M.M.; Einarsson, G.G.; Wei, L.; Drain, M.; Klem, E.R.; Cardwell, C.; Ennis, M.; Boucher, R.C.; Wolfgang, M.C.; Elborn, J.S. Lung microbiota and bacterial abundance in patients with bronchiectasis when clinically stable and during exacerbation. Am. J. Respir. Crit. Care Med. 2013, 187, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Langton Hewer, S.C.; Smyth, A.R. Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis. Cochrane Database Syst. Rev. 2017, 4, CD004197. [Google Scholar] [CrossRef] [PubMed]
- Quintiles IMS. Available online: http://www.imshealth.com/en (accessed on 20 September 2017).
- Elixhauser, A.; Steiner, C.; Harris, D.R.; Coffey, R.M. Comorbidity measures for use with administrative data. Med. Care 1998, 36, 8–27. [Google Scholar] [CrossRef] [PubMed]
- Stuart, B.; Lin, J.H.; Mogayzel, P.J., Jr. Early eradication of Pseudomonas aeruginosa in patients with cystic fibrosis. Paediatr. Respir. Rev. 2010, 11, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, M.C.; Bilton, D.; Hill, A.T.; British Thoracic Society Bronchiectasis non-CF Guideline Group. British thoracic society guideline for non-CF bronchiectasis. Thorax 2010, 65, i1–i58. [Google Scholar] [CrossRef] [PubMed]
- Vendrell, M.; de Gracia, J.; Olveira, C.; Martinez, M.A.; Giron, R.; Maiz, L.; Canton, R.; Coll, R.; Escribano, A.; Sole, A. Diagnosis and treatment of bronchiectasis. Spanish society of pneumology and thoracic surgery. Archivos de Bronconeumología 2008, 44, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Pressler, T.; Bohmova, C.; Conway, S.; Dumcius, S.; Hjelte, L.; Hoiby, N.; Kollberg, H.; Tummler, B.; Vavrova, V. Chronic Pseudomonas aeruginosa infection definition: EuroCareCF working group report. J Cyst. Fibros. 2011, 10, S75–S78. [Google Scholar] [CrossRef]
| N | % | |
|---|---|---|
| Total | 716 | 100 |
| Mean | ||
| 0–17 years | 22 | 3.07 |
| 18-49 years | 54 | 7.54 |
| 50+ years | 636 | 88.83 |
| Unknown | 4 | 0.56 |
| Sex | ||
| Female | 433 | 60.47 |
| Male | 282 | 39.39 |
| Unknown | 1 | 0.14 |
| Comorbidities | ||
| Chronic obstructive pulmonary disease | 619 | 86.45 |
| Arterial hypertension, uncontrolled | 355 | 49.58 |
| Cardiac arrhythmias | 205 | 28.63 |
| Fluid and electrolyte disorders | 149 | 20.81 |
| Diabetes, uncontrolled | 148 | 20.67 |
| Congestive heart failure | 118 | 16.48 |
| Valvular disease | 108 | 15.08 |
| Depression | 100 | 13.97 |
| Hypothyroidism | 99 | 13.83 |
| Weight loss | 90 | 12.57 |
| Rheumatoid arthritis | 86 | 12.01 |
| Peripheral ulcer disease | 77 | 10.75 |
| Tumor | 76 | 10.61 |
| Other neurological disease | 73 | 10.2 |
| Pulmonary circulation disease | 69 | 9.64 |
| Deficiency anemia | 64 | 8.94 |
| Arterial hypertension, controlled | 62 | 8.66 |
| Renal failure | 57 | 7.96 |
| Coagulopathies | 42 | 5.87 |
| Liver disease | 41 | 5.73 |
| Diabetes, controlled | 38 | 5.31 |
| Obesity | 35 | 4.89 |
| Paralysis | 29 | 4.05 |
| Lymphoma | 26 | 3.63 |
| Blood loss anemia | 14 | 1.96 |
| Psychosis | 12 | 1.68 |
| Alcohol abuse | 11 | 1.54 |
| Drug abuse | 10 | 1.4 |
| Peptic ulcer disease | 10 | 1.4 |
| Human immunodeficiency syndrome/acquired immunodeficiency syndrome | 1 | 0.14 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blanchette, C.M.; Noone, J.M.; Stone, G.; Zacherle, E.; Patel, R.P.; Howden, R.; Mapel, D. Healthcare Cost and Utilization before and after Diagnosis of Pseudomonas aeruginosa among Patients with Non-Cystic Fibrosis Bronchiectasis in the U.S. Med. Sci. 2017, 5, 20. https://doi.org/10.3390/medsci5040020
Blanchette CM, Noone JM, Stone G, Zacherle E, Patel RP, Howden R, Mapel D. Healthcare Cost and Utilization before and after Diagnosis of Pseudomonas aeruginosa among Patients with Non-Cystic Fibrosis Bronchiectasis in the U.S. Medical Sciences. 2017; 5(4):20. https://doi.org/10.3390/medsci5040020
Chicago/Turabian StyleBlanchette, Christopher M., Joshua M. Noone, Glenda Stone, Emily Zacherle, Ripsi P. Patel, Reuben Howden, and Douglas Mapel. 2017. "Healthcare Cost and Utilization before and after Diagnosis of Pseudomonas aeruginosa among Patients with Non-Cystic Fibrosis Bronchiectasis in the U.S." Medical Sciences 5, no. 4: 20. https://doi.org/10.3390/medsci5040020
APA StyleBlanchette, C. M., Noone, J. M., Stone, G., Zacherle, E., Patel, R. P., Howden, R., & Mapel, D. (2017). Healthcare Cost and Utilization before and after Diagnosis of Pseudomonas aeruginosa among Patients with Non-Cystic Fibrosis Bronchiectasis in the U.S. Medical Sciences, 5(4), 20. https://doi.org/10.3390/medsci5040020





