Risk Stratification in Acute Coronary Syndromes: The Systemic Immune-Inflammation Index as Prognostic Marker
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Statistical Analysis
2.3. Ethical Approval
3. Results
3.1. SII as a Predictor of Short-Term Mortality in ACS Patients
- Patients who did not undergo CAG; this included older, frail individuals with severe comorbidities, those who refused the procedure, and those who died before it could be performed.
- Patients with extensive CAD who could not be treated with PCI and were instead referred for CABG.
- Patients without significant coronary artery stenosis (defined as less than 50–75% narrowing).
3.2. SII as a Predictor of MACCE in ACS Patients
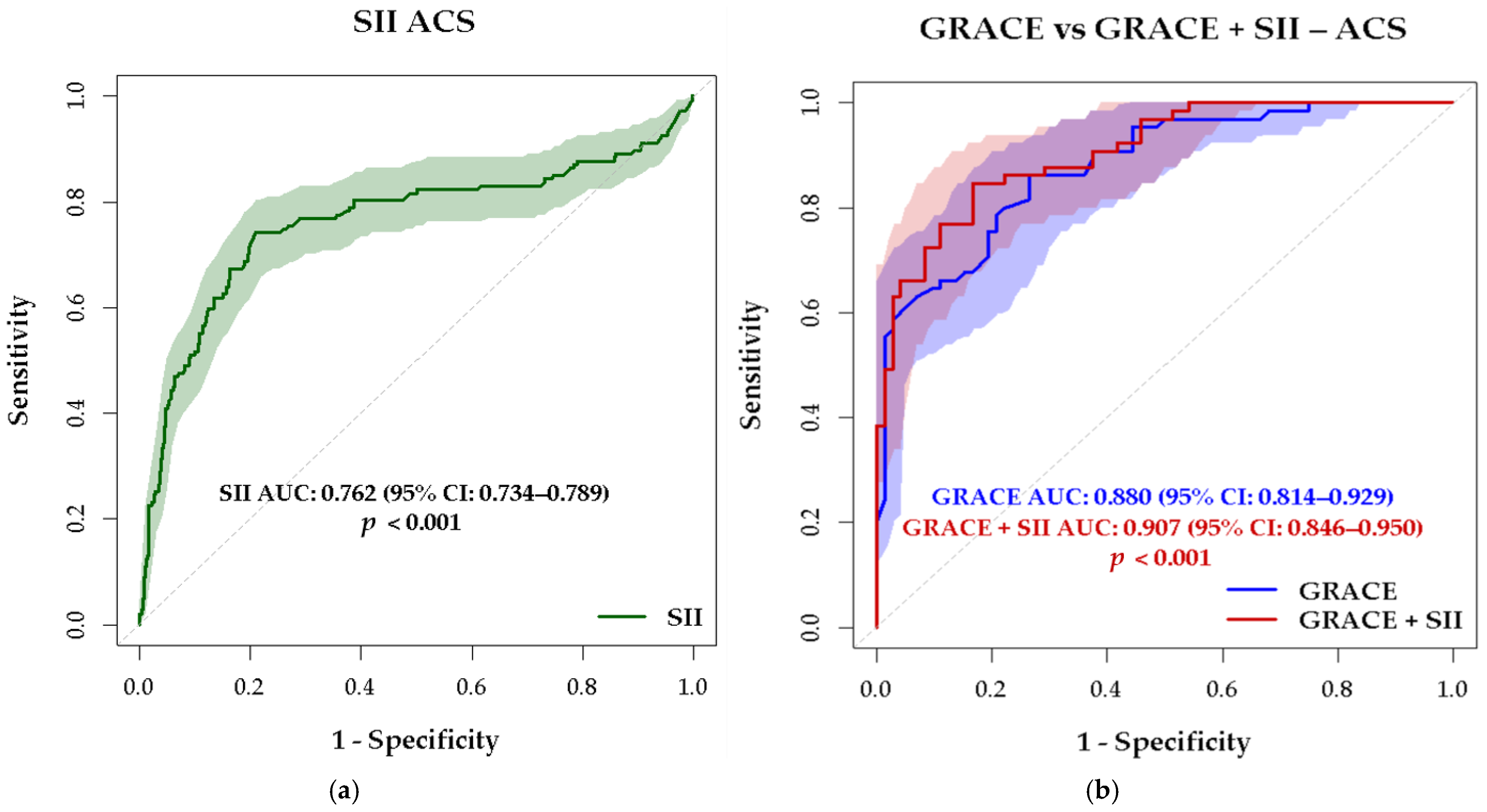
3.3. The Discriminative Value of SII as a Predictor of MACCE in ACS Patients. Correlation with GRACE 2 Risk Score
3.4. SII as a Predictor of MACCE in STEMI Patients
3.5. Comparison of Inflammatory Markers in Predicting MACCE in ACS Patients
3.6. Serial SII Assessment in Patients with STEMI
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACS | Acute Coronary Syndromes |
| ANOVA | Analysis of Variance |
| ARB | Angiotensin Receptor Blocker |
| AUC | Area Under the Curve |
| BMI | Body Mass Index |
| CAD | Coronary Artery Disease |
| CBC | Complete Blood Count |
| CCS | Chronic Coronary Syndromes |
| CI | Confidence Interval |
| CKD | Chronic Kidney Disease |
| CRP | C-Reactive Protein |
| CT | Computed Tomography |
| CVD | Cardiovascular Disease |
| ECG | Electrocardiogram |
| ESC | European Society of Cardiology |
| GRACE | Global Registry of Acute Coronary Events |
| Hs-Tni | High-Sensitivity Troponin i |
| LVEF | Left Ventricular Ejection Fraction |
| MACCE | Major Adverse Cardiac and Cerebrovascular Events |
| MACE | Major Adverse Cardiac Events |
| MI | Myocardial Infarction |
| MLR | Monocyte to Lymphocyte Ratio |
| MRI | Magnetic Resonance Imaging |
| NLR | Neutrophil to Lymphocyte Ratio |
| NSTEMI | Non-ST-Elevation Myocardial Infarction |
| OR | Odds Ratio |
| PCI | Percutaneous Coronary Intervention |
| PPCI | Primary Percutaneous Coronary Intervention |
| PLR | Platelet to Lymphocyte Ratio |
| SD | Standard Deviation |
| SII | Systemic Immune-Inflammation Index |
| SPSS | Statistical Package for the Social Sciences |
| STEMI | ST-Elevation Myocardial Infarction |
| UA | Unstable Angina |
| WBC | White Blood Cells |
References
- Yang, Y.L.; Wu, C.H.; Hsu, P.F.; Chen, S.C.; Huang, S.S.; Chan, W.L.; Lin, S.J.; Chou, C.Y.; Chen, J.W.; Pan, J.P.; et al. Systemic Immune-Inflammation Index (SII) Predicted Clinical Outcome in Patients with Coronary Artery Disease. Eur. J. Clin. Investig. 2020, 50, e13230. [Google Scholar] [CrossRef]
- Ralapanawa, U.; Sivakanesan, R. Epidemiology and the Magnitude of Coronary Artery Disease and Acute Coronary Syndrome: A Narrative Review. J. Epidemiol. Glob. Health 2021, 11, 169–177. [Google Scholar] [CrossRef]
- Piironen, M.; Ukkola, O.; Huikuri, H.; Havulinna, A.S.; Koukkunen, H.; Mustonen, J.; Ketonen, M.; Lehto, S.; Airaksinen, J.; Antero Kesäniemi, Y.; et al. Trends in Long-Term Prognosis after Acute Coronary Syndrome. Eur. J. Prev. Cardiol. 2017, 24, 274–280. [Google Scholar] [CrossRef]
- Gur, D.O.; Efe, M.M.; Alpsoy, S.; Akyüz, A.; Uslu, N.; Çelikkol, A.; Gur, O. Systemic Immune-Inflammatory Index as a Determinant of Atherosclerotic Burden and High-Risk Patients with Acute Coronary Syndromes. Arq. Bras. Cardiol. 2022, 119, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Pasterkamp, G.; Crea, F.; Jang, I.K. Reassessing the Mechanisms of Acute Coronary Syndromes: The “Vulnerable Plaque” and Superficial Erosion. Circ. Res. 2019, 124, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Dziedzic, E.A.; Gąsior, J.S.; Tuzimek, A.; Paleczny, J.; Junka, A.; Dąbrowski, M.; Jankowski, P. Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence. Int. J. Mol. Sci. 2022, 23, 9553. [Google Scholar] [CrossRef] [PubMed]
- Akkaya, S.; Cakmak, U. The Association Between Systemic Immune Inflammation Index and Intermediate-Term Mortality in Patients with Acute Coronary Syndromes. J. Harran Univ. Med. Fac. 2024, 21, 88–94. [Google Scholar] [CrossRef]
- Kim, J.H.; Lim, S.; Park, K.S.; Jang, H.C.; Choi, S.H. Total and Differential WBC Counts Are Related with Coronary Artery Atherosclerosis and Increase the Risk for Cardiovascular Disease in Koreans. PLoS ONE 2017, 12, e0180332. [Google Scholar] [CrossRef]
- Wu, T.H.; Chien, K.L.; Lin, H.J.; Hsu, H.C.; Su, T.C.; Chen, M.F.; Lee, Y.T. Total White Blood Cell Count or Neutrophil Count Predict Ischemic Stroke Events among Adult Taiwanese: Report from a Community-Based Cohort Study. BMC Neurol. 2013, 13, 7. [Google Scholar] [CrossRef]
- Wheeler, J.G.; Mussolino, M.E.; Gillum, R.F.; Danesh, J. Associations between Differential Leucocyte Count and Incident Coronary Heart Disease: 1764 Incident Cases from Seven Prospective Studies of 30,374 Individuals. Eur. Heart J. 2004, 25, 1287–1292. [Google Scholar] [CrossRef]
- Wang, T.; Jiang, C.Q.; Xu, L.; Zhang, W.S.; Zhu, F.; Jin, Y.L.; Thomas, G.N.; Cheng, K.K.; Lam, T.H. White Blood Cell Count and All-Cause and Cause-Specific Mortality in the Guangzhou Biobank Cohort Study. BMC Public Health 2018, 18, 1232. [Google Scholar] [CrossRef]
- Abete, I.; Lu, Y.; Lassale, C.; Verschuren, M.; Van Der Schouw, Y.; Bueno-De-Mesquita, B. White Cell Counts in Relation to Mortality in a General Population of Cohort Study in the Netherlands: A Mediating Effect or Not? BMJ Open 2019, 9, e030949. [Google Scholar] [CrossRef] [PubMed]
- Distelmaier, K.; Winter, M.P.; Dragschitz, F.; Redwan, B.; Mangold, A.; Gleiss, A.; Perkmann, T.; Maurer, G.; Adlbrecht, C.; Lang, I.M. Prognostic Value of Culprit Site Neutrophils in Acute Coronary Syndrome. Eur. J. Clin. Investig. 2014, 44, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Karabinos, I.; Koulouris, S.; Kranidis, A.; Pastromas, S.; Exadaktylos, N.; Kalofoutis, A. Neutrophil Count on Admission Predicts Major In-Hospital Events in Patients with a Non-ST-Segment Elevation Acute Coronary Syndrome. Clin. Cardiol. 2009, 32, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Tabas, I.; Fredman, G.; Fisher, E.A. Inflammation and Its Resolution as Determinants of Acute Coronary Syndromes. Circ. Res. 2014, 114, 1867–1879. [Google Scholar] [CrossRef]
- Hedrick, C.C. Lymphocytes in Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 253–257. [Google Scholar] [CrossRef]
- Hoffman, M.; Blum, A.; Baruch, R.; Kaplan, E.; Benjamin, M. Leukocytes and Coronary Heart Disease. Atherosclerosis 2004, 172, 1–6. [Google Scholar] [CrossRef]
- Fan, Z.; Ji, H.; Li, Y.; Jian, X.; Li, L.; Liu, T. Relationship between Monocyte-to-Lymphocyte Ratio and Coronary Plaque Vulnerability in Patients with Stable Angina. Biomark. Med. 2017, 11, 979–990. [Google Scholar] [CrossRef]
- Ateş, A.H.; Aytemir, K.; Koçyiğit, D.; Yalcin, M.U.; Gürses, K.M.; Yorgun, H.; Canpolat, U.; Hazırolan, T.; Özer, N. Association of Neutrophil-to-Lymphocyte Ratio with the Severity and Morphology of Coronary Atherosclerotic Plaques Detected by Multidetector Computerized Tomography. Acta Cardiol. Sin. 2016, 32, 676–683. [Google Scholar] [CrossRef]
- Balta, S.; Ozturk, C. The Platelet-Lymphocyte Ratio: A Simple, Inexpensive and Rapid Prognostic Marker for Cardiovascular Events. Platelets 2015, 26, 680–681. [Google Scholar] [CrossRef]
- Babes, E.E.; Radu, A.-F.; Vlad Babe¸sbabe¸s, V.; Tunduc, P.I.; Radu, A.; Bungau, G.; Bustea, C. The Prognostic Role of Hematological Markers in Acute Pulmonary Embolism: Enhancing Risk Stratification. Medicina 2025, 61, 1095. [Google Scholar] [CrossRef]
- Budzianowski, J.; Pieszko, K.; Burchardt, P.; Rzeźniczak, J.; Hiczkiewicz, J. The Role of Hematological Indices in Patients with Acute Coronary Syndrome. Dis. Markers 2017, 2017, 3041565. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.H.; Kobayashi, Y.; Nishi, T.; Kim, H.K.; Ki, Y.J.; Kim, S.S.; Park, K.H.; Song, H.; Fearon, W.F. Combination of Mean Platelet Volume and Neutrophil to Lymphocyte Ratio Predicts Long-Term Major Adverse Cardiovascular Events After Percutaneous Coronary Intervention. Angiology 2019, 70, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Çiçek, G.; Açikgoz, S.K.; Bozbay, M.; Altay, S.; Uğur, M.; Uluganyan, M.; Uyarel, H. Neutrophil-Lymphocyte Ratio and Platelet-Lymphocyte Ratio Combination Can Predict Prognosis in Patients With ST-Segment Elevation Myocardial Infarction Undergoing Primary Percutaneous Coronary Intervention. Angiology 2015, 66, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, Q.; Wang, R.; Ji, H.; Chen, Y.; Quan, X.; Zhang, C. Systemic Immune-Inflammatory Index Predicts Clinical Outcomes for Elderly Patients with Acute Myocardial Infarction Receiving Percutaneous Coronary Intervention. Med. Sci. Monit. 2019, 25, 9690. [Google Scholar] [CrossRef]
- Erdogan, M.; Erdöl, M.A.; Öztürk, S.; Durmaz, T. Systemic Immune-Inflammation Index Is a Novel Marker to Predict Functionally Significant Coronary Artery Stenosis. Biomark. Med. 2020, 14, 1553–1561. [Google Scholar] [CrossRef]
- Liu, Y.; Ye, T.; Chen, L.; Jin, T.; Sheng, Y.; Wu, G.; Zong, G. Systemic Immune-Inflammation Index Predicts the Severity of Coronary Stenosis in Patients with Coronary Heart Disease. Coron. Artery Dis. 2021, 32, 715–720. [Google Scholar] [CrossRef]
- Candemir, M.; Kiziltunç, E.; Nurkoç, S.; Şahinarslan, A. Relationship Between Systemic Immune-Inflammation Index (SII) and the Severity of Stable Coronary Artery Disease. Angiology 2021, 72, 575–581. [Google Scholar] [CrossRef]
- Kelesoglu, S.; Yilmaz, Y.; Elcık, D.; Kalay, N. Systemic Immune Inflammation Index: A Novel Predictor for Coronary Collateral Circulation. Perfusion 2022, 37, 605–612. [Google Scholar] [CrossRef]
- Li, Q.; Ma, X.; Shao, Q.; Yang, Z.; Wang, Y.; Gao, F.; Zhou, Y.; Yang, L.; Wang, Z. Prognostic Impact of Multiple Lymphocyte-Based Inflammatory Indices in Acute Coronary Syndrome Patients. Front. Cardiovasc. Med. 2022, 9, 811790. [Google Scholar] [CrossRef]
- Han, K.; Shi, D.; Yang, L.; Wang, Z.; Li, Y.; Gao, F.; Liu, Y.; Ma, X.; Zhou, Y. Prognostic Value of Systemic Inflammatory Response Index in Patients with Acute Coronary Syndrome Undergoing Percutaneous Coronary Intervention. Ann. Med. 2022, 54, 1667–1677. [Google Scholar] [CrossRef] [PubMed]
- Urbanowicz, T.; Michalak, M.; Al-Imam, A.; Olasińska-Wiśniewska, A.; Rodzki, M.; Witkowska, A.; Haneya, A.; Buczkowski, P.; Perek, B.; Jemielity, M. The Significance of Systemic Immune-Inflammatory Index for Mortality Prediction in Diabetic Patients Treated with Off-Pump Coronary Artery Bypass Surgery. Diagnostics 2022, 12, 634. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Jung, J.; Ahn, Y.; Oh, J. Systemic Immune-inflammation Index Predicted Short-term Outcomes in Patients Undergoing Isolated Tricuspid Valve Surgery. J. Clin. Med. 2021, 10, 4147. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Zhang, J.; Liu, T.; Yin, Z.; Jin, Y.; Han, J.; Guo, Z.; Wang, H. The Systemic-Immune-Inflammation Index Predicts the Recurrence of Atrial Fibrillation after Cryomaze Concomitant with Mitral Valve Surgery. BMC Cardiovasc. Disord. 2022, 22, 45. [Google Scholar] [CrossRef]
- Selcuk, M.; Cinar, T.; Saylik, F.; Dogan, S.; Selcuk, I.; Orhan, A.L. Predictive Value of Systemic Immune Inflammation Index for Postoperative Atrial Fibrillation in Patients Undergoing Isolated Coronary Artery Bypass Grafting. Medeni. Med. J. 2021, 36, 318–324. [Google Scholar] [CrossRef]
- Keykhaei, M.; Ashraf, H.; Rashedi, S.; Farrokhpour, H.; Heidari, B.; Zokaei, S.; Bagheri, S.; Foroumadi, R.; Asgarian, S.; Amirian, A.; et al. Differences in the 2020 ESC Versus 2015 ESC and 2014 ACC/AHA Guidelines on the Management of Acute Coronary Syndromes in Patients Presenting Without Persistent ST-Segment Elevation. Curr. Atheroscler. Rep. 2021, 23, 77. [Google Scholar] [CrossRef]
- Bursi, F.; Weston, S.A.; Killian, J.M.; Gabriel, S.E.; Jacobsen, S.J.; Roger, V.L. C-Reactive Protein and Heart Failure after Myocardial Infarction in the Community. Am. J. Med. 2007, 120, 616–622. [Google Scholar] [CrossRef]
- Hennessy, T.; Soh, L.; Bowman, M.; Kurup, R.; Schultz, C.; Patel, S.; Hillis, G.S. The Low Dose Colchicine after Myocardial Infarction (LoDoCo-MI) Study: A Pilot Randomized Placebo Controlled Trial of Colchicine Following Acute Myocardial Infarction. Am. Heart J. 2019, 215, 62–69. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Zheng, P.G.; Chen, P.; Wang, L.J.; Zhang, N. The Association of the Systemic Immune-Inflammation Index and Stent Thrombosis in Myocardial Infarction Patients after Coronary Stent Implantation—A Retrospectively Study. J. Thorac. Dis. 2023, 15, 1726–1733. [Google Scholar] [CrossRef]
- Kim, T.; Sim, J.; Hong, S.Y.; Kim, B.W. Systemic Immune-Inflammatory Marker of High Meld Patients Is Associated With Early Mortality After Liver Transplantation. Transplant. Proc. 2021, 53, 2945–2952. [Google Scholar] [CrossRef]
- Byrne, R.A.; Rossello, X.; Coughlan, J.J.; Barbato, E.; Berry, C.; Chieffo, A.; Claeys, M.J.; Dan, G.A.; Dweck, M.R.; Galbraith, M.; et al. 2023 ESC Guidelines for the Management of Acute Coronary Syndromes. Eur. Heart J. 2023, 44, 3720–3826. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D. Fourth Universal Definition of Myocardial Infarction (2018). Circulation 2018, 138, e618–e651. [Google Scholar] [CrossRef]
- Das, D.; McGrath, B.M. Sgarbossa Criteria for Acute Myocardial Infarction. CMAJ 2016, 188, E395. [Google Scholar] [CrossRef] [PubMed]
- Global Registry of Acute Coronary Events (GRACE) 2.0 Score. Available online: https://www.mdcalc.com/calc/1304/pulmonary-embolism-severity-index-pesi (accessed on 20 June 2025).
- Pinto, A.S.; Aroor, A.R.; Pereira, P. Systemic Immune-Inflammation Index in Acute Coronary Syndrome and Its Role in Predicting Disease Severity: A Cohort Study. J. Clin. Diagn. Res. 2022, 16, OC05–OC10. [Google Scholar] [CrossRef]
- Alinity Hq System Operations Manual. Available online: http://190.71.26.186:84/biomedica/storage/app/public/documents/1601/manual-alinity-h-seriespdf.pdf (accessed on 24 July 2025).
- Babes, E.E.; Zaha, D.C.; Tit, D.M.; Nechifor, A.C.; Bungau, S.; Andronie-Cioara, F.L.; Behl, T.; Stoicescu, M.; Munteanu, M.A.; Rus, M.; et al. Value of Hematological and Coagulation Parameters as Prognostic Factors in Acute Coronary Syndromes. Diagnostics 2021, 11, 850. [Google Scholar] [CrossRef]
- Stolker, J.M.; Cohen, D.J.; Kennedy, K.F.; Pencina, M.J.; Lindsey, J.B.; Mauri, L.; Cutlip, D.E.; Kleiman, N.S. Repeat Revascularization after Contemporary Percutaneous Coronary Intervention: An Evaluation of Staged, Target Lesion, and Other Unplanned Revascularization Procedures during the First Year. Circ. Cardiovasc. Interv. 2012, 5, 772–782. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- Su, G.; Zhang, Y.; Xiao, R.; Zhang, T.; Gong, B. Systemic Immune-Inflammation Index as a Promising Predictor of Mortality in Patients with Acute Coronary Syndrome: A Real-World Study. J. Int. Med. Res. 2021, 49, 03000605211016274. [Google Scholar] [CrossRef]
- Gao, Y.; Li, Y.; Chen, X.; Wu, C.; Guo, Z.; Bai, G.; Liu, T.; Li, G. The Systemic Inflammation Index Predicts Poor Clinical Prognosis in Patients with Initially Diagnosed Acute Coronary Syndrome Undergoing Primary Coronary Angiography. J. Inflamm. Res. 2023, 16, 5205–5219. [Google Scholar] [CrossRef]
- Shi, S.; Kong, S.; Ni, W.; Lu, Y.; Li, J.; Huang, Y.; Chen, J.; Lin, K.; Li, Y.; Ke, J.; et al. Association of the Systemic Immune-Inflammation Index with Outcomes in Acute Coronary Syndrome Patients with Chronic Kidney Disease. J. Inflamm. Res. 2023, 16, 1343–1356. [Google Scholar] [CrossRef]
- Orhan, A.L.; Şaylık, F.; Çiçek, V.; Akbulut, T.; Selçuk, M.; Çınar, T. Evaluating the Systemic Immune-Inflammation Index for in-Hospital and Long-Term Mortality in Elderly Non-ST-Elevation Myocardial Infarction Patients. Aging Clin. Exp. Res. 2022, 34, 1687–1695. [Google Scholar] [CrossRef] [PubMed]
- Öcal, L.; Keskin, M.; Cersit, S.; Eren, H.; Özgün Cakmak, E.; Karagöz, A.; Cakir, H.; Gürsoy, M.O.; Doǧan, S.; Zhalilov, M.; et al. Systemic Immune-Inflammation Index Predicts in-Hospital and Long-Term Outcomes in Patients with ST-Segment Elevation Myocardial Infarction. Coron. Artery Dis. 2022, 33, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Qin, X.; Zhang, X.; Zhang, Y.; Yuan, F.; Shi, W.; Liu, B.; Wei, Y. Prognostic Implications of Systemic Immune-Inflammation Index in Myocardial Infarction Patients with and without Diabetes: Insights from the NOAFCAMI-SH Registry. Cardiovasc. Diabetol. 2024, 23, 41. [Google Scholar] [CrossRef] [PubMed]
- Poznyak, A.; Grechko, A.V.; Poggio, P.; Myasoedova, V.A.; Alfieri, V.; Orekhov, A.N. The Diabetes Mellitus–Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. Int. J. Mol. Sci. 2020, 21, 1835. [Google Scholar] [CrossRef]
- Tuzimek, A.; Dziedzic, E.A.; Beck, J.; Kochman, W. Correlations Between Acute Coronary Syndrome and Novel Inflammatory Markers (Systemic Immune-Inflammation Index, Systemic Inflammation Response Index, and Aggregate Index of Systemic Inflammation) in Patients with and without Diabetes or Prediabetes. J. Inflamm. Res. 2024, 17, 2623–2632. [Google Scholar] [CrossRef]
- Spadafora, L.; Pastena, P.; Cacciatore, S.; Betti, M.; Biondi-Zoccai, G.; D’Ascenzo, F.; De Ferrari, G.M.; De Filippo, O.; Versaci, F.; Sciarretta, S.; et al. One-Year Prognostic Differences and Management Strategies between ST-Elevation and Non-ST-Elevation Myocardial Infarction: Insights from the PRAISE Registry. Am. J. Cardiovasc. Drugs 2025, 1–11. [Google Scholar] [CrossRef]
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, J.A.; Halvorsen, S.; et al. 2017 ESC Guidelines for the Management of Acute Myocardial Infarction in Patients Presenting with ST-Segment Elevation. Eur. Heart J. 2018, 39, 119–177. [Google Scholar] [CrossRef]
- Fox, K.A.A.; Dabbous, O.H.; Goldberg, R.J.; Pieper, K.S.; Eagle, K.A.; Van De Werf, F.; Avezum, Á.; Goodman, S.G.; Flather, M.D.; Anderson, F.A.; et al. Prediction of Risk of Death and Myocardial Infarction in the Six Months after Presentation with Acute Coronary Syndrome: Prospective Multinational Observational Study (GRACE). Br. Med. J. 2006, 333, 1091–1094. [Google Scholar] [CrossRef]
- Libby, P.; Ridker, P.M.; Hansson, G.K. Inflammation in Atherosclerosis. From Pathophysiology to Practice. J. Am. Coll. Cardiol. 2009, 54, 2129–2138. [Google Scholar] [CrossRef]
- Koganti, S.; Karanasos, A.; Regar, E.; Rakhit, R.D. Association of Systemic Inflammatory Biomarkers with Morphological Characteristics of Coronary Atherosclerotic Plaque by Intravascular Optical Coherence Tomography. Hell. J. Cardiol. 2021, 62, 101–106. [Google Scholar] [CrossRef]
- Li, L.; Ma, Y.; Geng, X.B.; Tan, Z.; Wang, J.H.; Cui, C.; Wang, H.L.; Shang, X.M. Platelet-to-Lymphocyte Ratio Relates to Poor Prognosis in Elderly Patients with Acute Myocardial Infarction. Aging Clin. Exp. Res. 2021, 33, 619–624. [Google Scholar] [CrossRef]
- Zhang, S.; Wan, Z.; Zhang, Y.; Fan, Y.; Gu, W.; Li, F.; Meng, L.; Zeng, X.; Han, D.; Li, X. Neutrophil Count Improves the GRACE Risk Score Prediction of Clinical Outcomes in Patients with ST-Elevation Myocardial Infarction. Atherosclerosis 2015, 241, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Adamstein, N.H.; MacFadyen, J.G.; Rose, L.M.; Glynn, R.J.; Dey, A.K.; Libby, P.; Tabas, I.A.; Mehta, N.N.; Ridker, P.M. The Neutrophil-Lymphocyte Ratio and Incident Atherosclerotic Events: Analyses from Five Contemporary Randomized Trials. Eur. Heart J. 2021, 42, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Saylik, F.; Akbulut, T. Systemic Immune-Inflammation Index Predicts Major Cardiovascular Adverse Events in Patients with ST-Segment Elevated Myocardial Infarction. Arq. Bras. Cardiol. 2022, 119, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Esenboğa, K.; Kurtul, A.; Yamantürk, Y.Y.; Tan, T.S.; Tutar, D.E. Systemic Immune-Inflammation Index Predicts No-Reflow Phenomenon after Primary Percutaneous Coronary Intervention. Acta Cardiol. 2022, 77, 59–65. [Google Scholar] [CrossRef]
- Pantea-Roșan, L.R.; Bungau, S.G.; Radu, A.-F.; Pantea, V.A.; Moisi, M.I.; Vesa, C.M.; Behl, T.; Nechifor, A.C.; Babes, E.E.; Stoicescu, M.; et al. A Narrative Review of the Classical and Modern Diagnostic Methods of the No-Reflow Phenomenon. Diagnostics 2022, 12, 932. [Google Scholar] [CrossRef]
- Omar, T.; Akdağ, B.; Karakayalı, M.; Artaç, İ.; Karabağ, Y.; Dündar, C.; Arslan, A.; Rencüzoğulları, İ.; Omar, T.; Akdağ, B.; et al. The Prognostic Value of the Systemic Immune-Inflammation Index in ST-Segment Elevation Myocardial Infarction Patients and Its Correlation with Syntax II Score and TIMI Risk Score. Int. J. Cardiovasc. Acad. 2023, 9, 36–41. [Google Scholar] [CrossRef]
- Liu, A.; Sun, N.; Gao, F.; Wang, X.; Zhu, H.; Pan, D. The Prognostic Value of Dynamic Changes in SII for the Patients with STEMI Undergoing PPCI. BMC Cardiovasc. Disord. 2024, 24, 67. [Google Scholar] [CrossRef]
- Bustea, C.; Tit, D.M.; Bungau, A.F.; Bungau, S.G.; Pantea, V.A.; Babes, E.E.; Pantea-Roșan, L.R. Predictors of Readmission after the First Acute Coronary Syndrome and the Risk of Recurrent Cardiovascular Events—Seven Years of Patient Follow-Up. Life 2023, 13, 950. [Google Scholar] [CrossRef]
- Byoun, J.T.; Yun, K.H.; Jo, S.; Joo, D.; Cho, J.Y. Prognostic Role of Pan-Immune-Inflammatory Value in Patients with Non-ST-Segment Elevation Acute Coronary Syndrome. J. Cardiovasc. Dev. Dis. 2025, 12, 79. [Google Scholar] [CrossRef]





| Aspect | Details |
|---|---|
| Software used | SPSS v. 25 (IBM Corp.), MedCalc v. 19.4 (MedCalc Software), R Core Team, (Vienna, Austria) (2025) |
| Data presentation | Continuous variables: mean ± SD; categorical variables: frequencies and percentages |
| Test for continuous variables | Independent samples t-test |
| Test for categorical variables | Chi-square test |
| Statistical significance | p value < 0.05 |
| Predictive accuracy assessment | ROC curve analysis with AUC calculation |
| Cutoff determination | Cutoffs based on Youden index maximizing sensitivity and specificity |
| Multivariate modeling | Binary logistic regression for independent predictors |
| Mortality analysis | Multivariable Cox proportional hazards regression |
| Temporal SII variation | Repeated measures ANOVA (General Linear Model) with Bonferroni correction |
| Power analysis tool | Post hoc using G*Power version 3.1 with α = 0.05, OR ≥ 1.5 [50] |
| Overall cohort power (n = 946) | 99%, event rate: 15.25% |
| STEMI subgroup power (n = 380) | 87%, event rate: 25.79% |
| NSTEMI subgroup power (n = 283) | 77%, event rate: 12.23% |
| UA subgroup power (n = 301) | 77%, event rate: 4.31% |
| Limitations in subgroups | Power below 80% limits multivariate modeling reliability |
| Subgroup analysis decision | Detailed analysis restricted to STEMI subgroup for robustness and power |
| Parameters | Total (n = 964) | Deceased (n = 76) | Survivors (n = 888) | p |
|---|---|---|---|---|
| Age (years) | 65.59 ± 11.758 | 73.39 ± 11.775 | 64.91 ± 11.51 | <0.001 * |
| Sex (M) | 621/964 (64.42%) | 36/76 (47.37%) | 585/888 (65.88%) | 0.002 * |
| Smoking | 232/964 (24.06%) | 8/76 (10.52%) | 224/888 (25.22%) | 0.004 * |
| Hypertension | 650/964 (67.43%) | 23/76 (30.26%) | 627/888 (70.60%) | <0.001 * |
| DM | 308/964 (31.95%) | 29/76 (38.15%) | 279/888 (31.41%) | 0.22 |
| BMI (kg/m2) | 30.95 ± 5.31 | 29.053 ± 4.65 | 31.116 ± 5.33 | 0.09 |
| LVEF (%) | 45.96 ± 9.45 | 34.25 ± 9.47 | 46.97 ± 8.75 | 0.001 * |
| Creatinine (mg/dL) | 1.09 ± 0.64 | 1.79 ± 1.11 | 1.03 ± 0.55 | |
| CRP (mg/L) | 3.99 ± 6.70 | 12.35 ± 9.4 | 3.57 ± 6.26 | |
| WBC (×103/μL) | 10.83 ± 4.59 | 14.75 ± 7.06 | 10.46 ± 4.10 | |
| NLR | 4.57 ± 4.52 | 10.63 ± 6.01 | 4.05 ± 3.97 | |
| SII | 825.24 ± 983.85 | 2003.79 ± 1601.17 | 722.04 ± 837.25 | |
| hs-Tni (pg/mL) | 1627.92 ± 2849.621 | 4623.42 ± 5884.92 | 1367.89 ± 2237.818 | <0.001 * |
| GRACE 2 | 139.12 ± 41.89 | 175.28 ± 32.43 | 118.34 ± 31.29 | |
| STEMI | 380/964 (39.41%) | 61/76 (80.26%) | 319/888 (35.92%) | |
| NSTEMI | 283/964 (29.36%) | 12/76 (15.79%) | 271/888 (30.52%) | 0.006 * |
| UA | 301/964 (31.22%) | 4/76 (5.26%) | 297/888 (33.44%) | <0.001 * |
| Cholesterol (mg/dL) | 176.98 ± 47.56 | 164.83 ± 44.64 | 178.04 ± 47.69 | 0.88 |
| PCI | 509/964 (52.80%) | 32/76 (42.11%) | 477/888 (53.72%) | 0.05 * |
| One vessel | 211/509 (41.45%) | 8/32 (25%) | 203/477 (42.56%) | 0.02 * |
| Two vessels | 153/509 (30.5%) | 4/32 (12.5%) | 149/477 (31.24%) | 0.012 * |
| Three vessels | 145/509 (28.48%) | 20/32 (62.5%) | 125/477 (26.21%) | 0.005 * |
| No PCI | 455/964 (47.19%) | 44/76 (57.90%) | 411/888 (46.28%) | 0.02 * |
| No CAG | 239/455 (52.52%) | 29/44 (65.91%) | 210/411 (51.09%) | 0.03 * |
| Not amenable to PCI | 93/455 (20.44%) | 15/44 (34.09%) | 78/411 (18.98%) | 0.018 * |
| No significant lesions | 123/455 (27.03%) | 0 | 123/411 (29.93%) | <0.001 * |
| Parameters | Total (n = 964) | MACCE (n = 147) | Non-MACCE (n = 817) | p |
|---|---|---|---|---|
| Age (years) | 65.59 ± 11.758 | 70.81 ± 11.572 | 64.65 ± 11.550 | <0.001 * |
| Sex (M) | 621/964 (64.4%) | 82/147 (55.78%) | 539/817 (65.97%) | <0.01 * |
| Smoking | 232/964 (24.06%) | 25/147 (17%) | 207/817 (25.33%) | 0.13 |
| Hypertension | 650/964 (67.42%) | 69/147 (46.93%) | 581/817 (71.11%) | <0.001 * |
| DM | 308/964 (31.95%) | 50/147 (34.01%) | 258/817 (31.57%) | 0.27 |
| BMI (kg/m2) | 30.95 ± 5.31 | 29.782 ± 5.07 | 31.164 ± 5.33 | |
| LVEF (%) | 45.96 ± 9.45 | 36.73 ± 10.75 | 47.82 ± 8.17 | <0.001 * |
| Creatinine (mg/dL) | 1.09 ± 0.64 | 1.54 ± 1.03 | 1.01 ± 0.50 | |
| CRP (mg/L) | 3.99 ± 6.70 | 8.84 ± 11.59 | 3.23 ± 5.11 | |
| WBC (×103/μL) | 10.83 ± 4.59 | 13.52 ± 6.3 | 10.32 ± 3.99 | 0.001 * |
| NLR | 4.57 ± 4.52 | 8.76 ± 5.79 | 3.81 ± 3.79 | <0.001 * |
| SII | 825.24 ± 983.85 | 1717 ± 1611.32 | 664.68 ± 713.11 | |
| Cholesterol (mg/dL) | 176.98 ± 47.56 | 173.41 ± 45.815 | 177.62 ± 47.87 | 0.34 |
| GRACE | 139.12 ± 41.89 | 167.446 ± 34.26 | 113.55 ± 30.10 | <0.001 * |
| Hs-troponin (pg/mL) | 1627.92 ± 2849.62 | 3620.86 ± 5186.601 | 1269.34 ± 1985.997 | |
| STEMI | 380/964 (39.42%) | 98/147 (66.66%) | 282/817 (34.52%) | |
| NSTEMI | 283/964 (29.36%) | 36/147 (24.49%) | 247/817 (30.23%) | 0.16 |
| UA | 301/964 (31.22%) | 13/147 (8.84%) | 288/817 (35.25%) | <0.001 * |
| PCI | 509/964 (52.80%) | 60/147 (40.82) | 449/817 (54.96%) | 0.001 * |
| One vessel | 211/509 (41.45%) | 21/55 (38.18%) | 190/454 (41.85%) | 0.66 |
| Two vessels | 153/509 (30.5%) | 17/55 (30.9%) | 136/454 (29.95%) | 0.75 |
| Three vessels | 145/509 (28.48%) | 22/55(40%) | 123/454 (27.09%) | 0.04 * |
| No PCI | 455/964 (47.19%) | 92/147 (62.58%) | 363/817 (44.43%) | 0.002 * |
| No CAG | 239/455 (52.52%) | 54/92 (58.69%) | 185/363 (50.96%) | 0.23 |
| Not amenable to PCI | 93/455 (20.44%) | 37/92 (40.21%) | 56/363 (15.42%) | <0.001 * |
| No significant lesions | 123/455 (27.03%) | 1/92 (1.08%) | 122/363 (33.60%) |
| Parameters | Total (n = 380) | MACCE (n = 98) | Non-MACCE (n = 282) | p |
|---|---|---|---|---|
| Age (years) | 64.49 ± 12.84 | 70.64 ± 12.42 | 62.35 ± 12.31 | <0.001 * |
| Sex (M) | 248/380 (65.3%) | 45/98 | 203/282 | |
| Smoking | 104/380 (27.36%) | 18/98 | 86/282 | 0.02 * |
| Hypertension | 210/380 (55.26%) | 39/98 (39.79%) | 171/282 (60.63%) | <0.001 * |
| DM | 115 (30.26%) | 38/98 (38.77%) | 77/282 (27.30%) | 0.04 * |
| BMI (kg/m2) | 29.87 ± 4.63 | 29.265 ± 4.60 | 30.082 ± 4.63 | 0.134 |
| LVEF (%) | 43.72% ± 10.25 | 34.56 ± 10.50 | 46.91 ± 8.01 | <0.001 * |
| Creatinine (mg/dL) | 1.11 ± 0.61 | 1.51 ± 0.97 | 0.97 ± 0.31 | |
| CRP (mg/L) | 6.05 ± 8.28 | 12.63 ± 13.23 | 4.72 ± 6.10 | 0.001 * |
| WBC (×103/μL) | 12.68 ± 11.95 | 14.08 ± 6.19 | 12.22 ± 4.42 | 0.01 * |
| NLR | 6.20 ± 5.25 | 9.31 ± 5.87 | 5.12 ± 4.56 | <0.001 * |
| SII | 1059.02 ± 1171.16 | 2042.16 ± 1630.77 | 717.37 ± 691.17 | |
| Cholesterol (mg/dL) | 178.52 ± 41.92 | 177.33 ± 44.97 | 178.94 ± 40.89 | 0.74 |
| GRACE 2 | 165.21 ± 35.22 | 170.38 ± 34.47 | 133.66 ± 21.04 | 0.003 * |
| hs-Tni (pg/mL) | 2700.12 ± 3714.24 | 4601.34 ± 5923.52 | 2039.41 ± 2189.87 | <0.001 * |
| PCI | 275 (72.36%) | 43 (43.87%) | 232 (82.26%) | <0.001 |
| One-vessel CAD | 126/275 (45.81%) | 19/43 (44.18%) | 107/232 (46.12%) | 0.8 |
| Two-vessel CAD | 83/275 (30.18%) | 8/43(18.60%) | 75/232 (32.33%) | 0.06 |
| Three-vessel CAD | 66/275 (24%) | 16/43 (37.20%) | 50/232 (21.55%) | 0.07 |
| No PCI | 105 (27.63%) | 55 (56.12%) | 50 (17.73%) | <0.001 |
| No CAG | 32 (30.47%) | 29 (52.82%) | 3 (6%) | |
| Not amenable to PCI | 31 (29.52%) | 26 (47.37%) | 5 (10%) | <0.001 |
| No significant lesions | 42 (40%) | 0 | 42 (84%) | <0.001 |
| Time (h) | SII Mean | Std. Error | 95% CI Lower Bound | Upper Bound |
|---|---|---|---|---|
| With STEMI | ||||
| Baseline | 1416.13 | 68.15 | 1282.022 | 1550.25 |
| 24 | 1868.02 | 79.47 | 1711.63 | 2024.42 |
| 48 | 995.485 | 65.23 | 867.11 | 1123.86 |
| With STEMI-PPCI | ||||
| Baseline | 1437.03 | 86.30 | 1266.85 | 1607.22 |
| 24 | 1808.04 | 91.26 | 1628.08 | 1987.99 |
| 48 | 1126.61 | 73.82 | 981.05 | 1272.19 |
| Time (I) | Time (J) | Mean Difference (I-J) | St. Error | 95% CI Lower Bound | 95% CI Upper Bound |
|---|---|---|---|---|---|
| STEMI | |||||
| Baseline | 24 h | −451.889 * | 54.995 | −584.295 | −319.483 |
| 48 h | 420.649 * | 66.325 | 260.964 | 580.334 | |
| 24 h | baseline | 451.889 * | 54.995 | 319.483 | 584.295 |
| 48 h | 872.538 * | 67.935 | 708.977 | 1036.099 | |
| 48 h | baseline | −420.649 * | 66.325 | −580.334 | −260.964 |
| 24 h | −872.538 * | 67.935 | −1036.099 | −708.977 | |
| STEMI PPCI | |||||
| Baseline | 24 h | −370.999 * | 51.900 | −496.307 | −245.692 |
| 48 h | 310.420 * | 56.862 | 173.134 | 447.706 | |
| 24 h | baseline | 370.999 * | 51.900 | 245.692 | 496.307 |
| 48 h | 681.419 * | 56.808 | 544.262 | 818.576 | |
| 48 h | baseline | −310.420 * | 56.862 | −447.706 | −173.134 |
| 24 h | −681.419 * | 56.808 | −818.576 | −544.262 | |
| MACCE | Time (h) | SII Mean | St. Error | 95% CI Lower Bound | 95% Upper Bound |
|---|---|---|---|---|---|
| STEMI | |||||
| No | Baseline | 720.881 | 81.913 | 559.677 | 882.084 |
| 24 | 1031.277 | 95.525 | 843.287 | 1219.268 | |
| 48 | 515.257 | 78.407 | 360.952 | 669.561 | |
| Yes | Baseline | 2111.387 | 108.932 | 1897.010 | 2325.764 |
| 24 | 2704.769 | 127.034 | 2454.768 | 2954.770 | |
| 48 | 1475.713 | 104.270 | 1270.511 | 1680.915 | |
| STEMI PPCI | |||||
| No | Baseline | 730.476 | 80.756 | 571.228 | 889.723 |
| 24 | 1052.369 | 85.394 | 883.976 | 1220.763 | |
| 48 | 523.554 | 69.076 | 387.340 | 659.769 | |
| Yes | Baseline | 2143.599 | 152.545 | 1842.787 | 2444.412 |
| 24 | 2563.705 | 161.307 | 2245.615 | 2881.794 | |
| 48 | 1729.682 | 130.482 | 1472.378 | 1986.986 | |
| Parameter | AUC | 95% Lower Bound | 95% Upper Bound | Sensitivity % | Specificity % | Cutoff (Youden Index) | p |
|---|---|---|---|---|---|---|---|
| SII baseline | 0.874 | 0.836 | 0.906 | 88.78 | 80.14 | >866 | <0.001 |
| SII 24 h | 0.866 | 0.821 | 0.903 | 86.73 | 81.15 | >1022 | |
| SII 48 h | 0.787 | 0.742 | 0.827 | 81.44 | 67.73 | >542 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babes, E.E.; Radu, A.-F.; Cretu, N.A.; Bungau, G.; Diaconu, C.C.; Tit, D.M.; Babes, V.V. Risk Stratification in Acute Coronary Syndromes: The Systemic Immune-Inflammation Index as Prognostic Marker. Med. Sci. 2025, 13, 116. https://doi.org/10.3390/medsci13030116
Babes EE, Radu A-F, Cretu NA, Bungau G, Diaconu CC, Tit DM, Babes VV. Risk Stratification in Acute Coronary Syndromes: The Systemic Immune-Inflammation Index as Prognostic Marker. Medical Sciences. 2025; 13(3):116. https://doi.org/10.3390/medsci13030116
Chicago/Turabian StyleBabes, Elena Emilia, Andrei-Flavius Radu, Noemi Adaus Cretu, Gabriela Bungau, Camelia Cristina Diaconu, Delia Mirela Tit, and Victor Vlad Babes. 2025. "Risk Stratification in Acute Coronary Syndromes: The Systemic Immune-Inflammation Index as Prognostic Marker" Medical Sciences 13, no. 3: 116. https://doi.org/10.3390/medsci13030116
APA StyleBabes, E. E., Radu, A.-F., Cretu, N. A., Bungau, G., Diaconu, C. C., Tit, D. M., & Babes, V. V. (2025). Risk Stratification in Acute Coronary Syndromes: The Systemic Immune-Inflammation Index as Prognostic Marker. Medical Sciences, 13(3), 116. https://doi.org/10.3390/medsci13030116









