The LipoDerm Method for Regeneration and Reconstruction in Plastic Surgery: A Technical Experimental Ex Vivo Note
Abstract
1. Introduction
2. Materials and Methods
2.1. Centrifugation of Lipoaspirate Obtained by Liposuction
2.2. Extraction of SVF/ASCs from Lipoaspirate
2.3. Assessment of Cellular Adherence of ASCs onto Collagen–Elastin Matrices
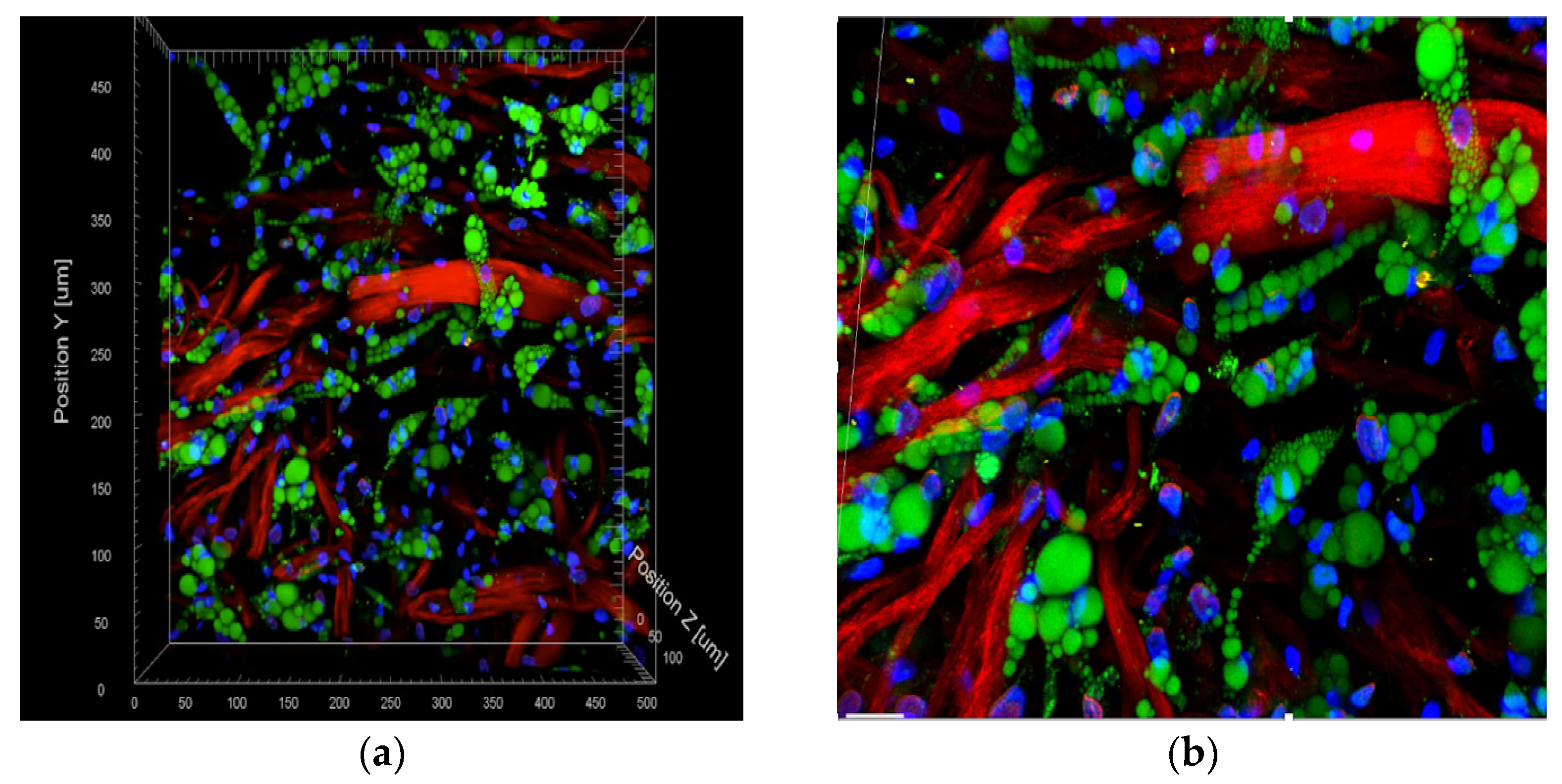
3. Results
Utilizing Two Photon Microscopy to Analyze Cellular Distribution following Incubation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alharbi, Z.; Almakadi, S.; Opländer, C.; Vogt, M.; Rennekampff, H.-O.; Pallua, N. Intraoperative use of enriched collagen and elastin matrices with freshly isolated adipose-derived stem/stromal cells: A potential clinical approach for soft tissue reconstruction. BMC Surg. 2014, 14, 10. [Google Scholar] [CrossRef]
- Min, J.H.; Yun, I.S.; Lew, D.H.; Roh, T.S.; Lee, W.J. The Use of Matriderm and Autologous Skin Graft in the Treatment of Full Thickness Skin Defects. Arch. Plast. Surg. 2014, 41, 330–336. [Google Scholar] [CrossRef]
- Picard, F.; Hersant, B.; la Padula, S.; Meningaud, J.-P. Platelet-rich plasma-enriched autologous fat graft in regenerative and aesthetic facial surgery: Technical note. J. Stomatol. Oral Maxillofac. Surg. 2017, 118, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Biedermann, T.; Widmer, D.; Montano, I.; Meuli, M.; Reichmann, E.; Schiestl, C. Matriderm® versus Integra®: A comparative experimental study. Burns 2009, 35, 51–57. [Google Scholar] [CrossRef]
- Nakhi, M.B.; Al Saqqa, B. The use of MatriDerm® and skin graft for reconstruction of complex wounds. Wounds Middle East 2020, 7, 6–11. Available online: https://www.woundsinternational.com/resources/details/use-matriderm-and-skin-graft-reconstruction-complex-wounds (accessed on 20 January 2023).
- Böttcher-Haberzeth, S.; Biedermann, T.; Schiestl, C.; Hartmann-Fritsch, F.; Schneider, J.; Reichmann, E.; Meuli, M. Matriderm® 1 mm versus Integra® Single Layer 1.3 mm for one-step closure of full thickness skin defects: A comparative experimental study in rats. Pediatr. Surg. Int. 2012, 28, 171–177. [Google Scholar] [CrossRef]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of Angiogenic and Antiapoptotic Factors by Human Adipose Stromal Cells. Circulation 2004, 109, 1292–1298. [Google Scholar] [CrossRef]
- Hassan, W.U.; Greiser, U.; Wang, W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014, 22, 313–325. [Google Scholar] [CrossRef]
- Cherubino, M.; Rubin, J.P.; Miljkovic, N.; Kelmendi-Doko, A.; Marra, K.G. Adipose-Derived Stem Cells for Wound Healing Applications. Ann. Plast. Surg. 2011, 66, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimian, T.G.; Pouzoulet, F.; Squiban, C.; Buard, V.; André, M.; Cousin, B.; Gourmelon, P.; Benderitter, M.; Casteilla, L.; Tamarat, R. Cell Therapy Based on Adipose Tissue-Derived Stromal Cells Promotes Physiological and Pathological Wound Healing. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.K.; Zhu, M.; Wulur, I.; Alfonso, Z. Adipose-Derived Stem Cells. In Mesenchymal Stem Cells; Clifton, N.J., Ed.; Humana Press: Totowa, NJ, USA, 2008; pp. 59–67. [Google Scholar] [CrossRef]
- Alharbi, Z.; Opländer, C.; Almakadi, S.; Fritz, A.; Vogt, M.; Pallua, N. Conventional vs. micro-fat harvesting: How fat harvesting technique affects tissue-engineering approaches using adipose tissue-derived stem/stromal cells. J. Plast. Reconstr. Aesthetic Surg. 2013, 66, 1271–1278. [Google Scholar] [CrossRef]
- Tonnard, P.; Verpaele, A.; Peeters, G.; Hamdi, M.; Cornelissen, M.; Declercq, H. Nanofat Grafting. Plast. Reconstr. Surg. 2013, 132, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Ding, P.; Lu, E.; Li, G.; Sun, Y.; Yang, W.; Zhao, Z. Research Progress on Preparation, Mechanism, and Clinical Application of Nanofat. J. Burn. Care Res. 2022, 43, 1140–1144. [Google Scholar] [CrossRef] [PubMed]
- Simonacci, F.; Bertozzi, N.; Grieco, M.P.; Grignaffini, E.; Raposio, E. Procedure, applications, and outcomes of autologous fat grafting. Ann. Med. Surg. 2017, 20, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Uyulmaz, S.; Sanchez Macedo, N.; Rezaeian, F.; Giovanoli, P.; Lindenblatt, N. Nanofat Grafting for Scar Treatment and Skin Quality Improvement. Aesthetic Surg. J. 2018, 38, 421–428. [Google Scholar] [CrossRef]
- Arena, A.; Committeri, U.; Maglitto, F.; Salzano, G.; Dell’Aversana Orabona, G.; Vaira, L.A.; Piombino, P.; Apolito, M.; de Fazio, G.R.; Califano, L. Three Different Types of Fat Grafting for Facial Systemic Sclerosis: A Case Series. J. Clin. Med. 2022, 11, 5489. [Google Scholar] [CrossRef]
- Pallua, N.; Kim, B.-S. Microfat and Lipoconcentrate for the Treatment of Facial Scars. Clin. Plast. Surg. 2020, 47, 139–145. [Google Scholar] [CrossRef]
- Kim, B.-S.; Ott, V.; Boecker, A.H.; Stromps, J.-P.; Paul, N.E.; Alharbi, Z.; Cakmak, E.; Bernhagen, J.; Bucala, R.; Pallua, N. The Effect of Antiseptics on Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2017, 139, 625–637. [Google Scholar] [CrossRef]
- Choi, J.H.; Gimble, J.M.; Lee, K.; Marra, K.G.; Rubin, J.P.; Yoo, J.J.; Vunjak-Novakovic, G.; Kaplan, D.L. Adipose Tissue Engineering for Soft Tissue Regeneration. Tissue Eng. Part B Rev. 2010, 16, 413–426. [Google Scholar] [CrossRef]
- Doornaert, M.; Depypere, B.; Creytens, D.; Declercq, H.; Taminau, J.; Lemeire, K.; Monstrey, S.; Berx, G.; Blondeel, P. Human decellularized dermal matrix seeded with adipose-derived stem cells enhances wound healing in a murine model: Experimental study. Ann. Med. Surg. 2019, 46, 4–11. [Google Scholar] [CrossRef]
- Wang, J.; Hao, H.; Huang, H.; Chen, D.; Han, Y.; Han, W. The Effect of Adipose-Derived Stem Cells on Full-Thickness Skin Grafts. BioMed Res. Int. 2016, 2016, 1464725. [Google Scholar] [CrossRef]
- Maitz, J.; Wang, Y.; Fathi, A.; Ximena Escobar, F.; Parungao, R.; Zuijlen, P.; Maitz, P.; Li, Z. The effects of cross-linking a collagen-elastin dermal template on scaffold bio-stability and degradation. J. Tissue Eng. Regen. Med. 2020, 14, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Khan, I.M.; Nguyen, P.P.; Lo, E.; Chahadeh, H.; Cerniglia, M.; Noriega, J.A. Skin Graft Techniques. Clin. Podiatr. Med. Surg. 2020, 37, 821–835. [Google Scholar] [CrossRef]
- Kim, W.-S.; Park, B.-S.; Sung, J.-H. The wound-healing and antioxidant effects of adipose-derived stem cells. Expert Opin. Biol. Ther. 2009, 9, 879–887. [Google Scholar] [CrossRef]
- Nie, C.; Yang, D.; Xu, J.; Si, Z.; Jin, X.; Zhang, J. Locally Administered Adipose-Derived Stem Cells Accelerate Wound Healing through Differentiation and Vasculogenesis. Cell Transplant. 2011, 20, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Kemp Bohan, P.M.; Cooper, L.E.; Fletcher, J.L.; Corkins, C.J.; Natesan, S.; Aden, J.K.; Carlsson, A.; Chan, R.K. Impact of dermal matrix thickness on split-thickness skin graft survival and wound contraction in a single-stage procedure. Int. Wound J. 2022, 19, 370–379. [Google Scholar] [CrossRef]
- Cervelli, V.; Brinci, L.; Spallone, D.; Tati, E.; Palla, L.; Lucarini, L.; de Angelis, B. The use of MatriDerm® and skin grafting in post-traumatic wounds. Int. Wound J. 2011, 8, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Gümbel, D.; Ackerl, M.; Napp, M.; Daeschlein, G.; Spranger, N.; Stope, M.B.; Ekkernkamp, A.; Matthes, G. Retrospective analysis of 56 soft tissue defects treated with one-stage reconstruction using dermal skin substitutes. JDDG J. Dtsch. Dermatol. Ges. 2016, 14, 595–601. [Google Scholar] [CrossRef]
- Frueh, F.S.; Menger, M.D.; Lindenblatt, N.; Giovanoli, P.; Laschke, M.W. Current and emerging vascularization strategies in skin tissue engineering. Crit. Rev. Biotechnol. 2017, 37, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Güven, S.; Zimoch, J.; Zapiórkowska, N.A.; Biedermann, T.; Böttcher-Haberzeth, S.; Meuli-Simmen, C.; Martin, I.; Scherberich, A.; Reichmann, E.; et al. Characterization of vasculogenic potential of human adipose-derived endothelial cells in a three-dimensional vascularized skin substitute. Pediatr. Surg. Int. 2016, 32, 17–27. [Google Scholar] [CrossRef]
- Foubert, P.; Barillas, S.; Gonzalez, A.D.; Alfonso, Z.; Zhao, S.; Hakim, I.; Meschter, C.; Tenenhaus, M.; Fraser, J.K. Uncultured adipose-derived regenerative cells (ADRCs) seeded in collagen scaffold improves dermal regeneration, enhancing early vascularization and structural organization following thermal burns. Burns 2015, 41, 1504–1516. [Google Scholar] [CrossRef] [PubMed]
- Später, T.; Frueh, F.S.; Nickels, R.M.; Menger, M.D.; Laschke, M.W. Prevascularization of collagen-glycosaminoglycan scaffolds: Stromal vascular fraction versus adipose tissue-derived microvascular fragments. J. Biol. Eng. 2018, 12, 24. [Google Scholar] [CrossRef] [PubMed]
- Mandili, A.; SAlsadi, H.H.; Khamis, S.; Alharbi, Z. Nanofat, Microfat, Platelet Rich Plasma (PRP), Microneedling and Fractional CO2 Laser: A Regenerative Multimodality Approach and Protocol in Scar Management. Arch. Clin. Med. Case Rep. 2022, 6, 511–516. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alharbi, Z.; Qari, S.; Bader, M.; Khamis, S.; Almarzouqi, F.; Vogt, M.; Opländer, C. The LipoDerm Method for Regeneration and Reconstruction in Plastic Surgery: A Technical Experimental Ex Vivo Note. Med. Sci. 2023, 11, 16. https://doi.org/10.3390/medsci11010016
Alharbi Z, Qari S, Bader M, Khamis S, Almarzouqi F, Vogt M, Opländer C. The LipoDerm Method for Regeneration and Reconstruction in Plastic Surgery: A Technical Experimental Ex Vivo Note. Medical Sciences. 2023; 11(1):16. https://doi.org/10.3390/medsci11010016
Chicago/Turabian StyleAlharbi, Ziyad, Sarah Qari, Maryam Bader, Sherif Khamis, Faris Almarzouqi, Michael Vogt, and Christian Opländer. 2023. "The LipoDerm Method for Regeneration and Reconstruction in Plastic Surgery: A Technical Experimental Ex Vivo Note" Medical Sciences 11, no. 1: 16. https://doi.org/10.3390/medsci11010016
APA StyleAlharbi, Z., Qari, S., Bader, M., Khamis, S., Almarzouqi, F., Vogt, M., & Opländer, C. (2023). The LipoDerm Method for Regeneration and Reconstruction in Plastic Surgery: A Technical Experimental Ex Vivo Note. Medical Sciences, 11(1), 16. https://doi.org/10.3390/medsci11010016






