Intrusion of Saline Water into a Coastal Aquifer Containing Palaeogroundwater in the Viimsi Peninsula in Estonia
Abstract
1. Introduction
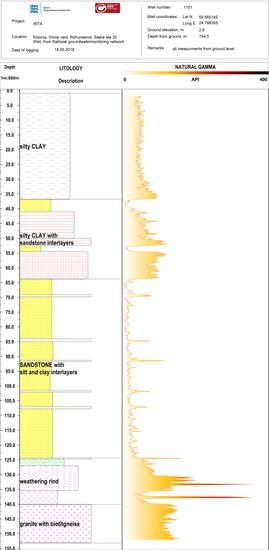
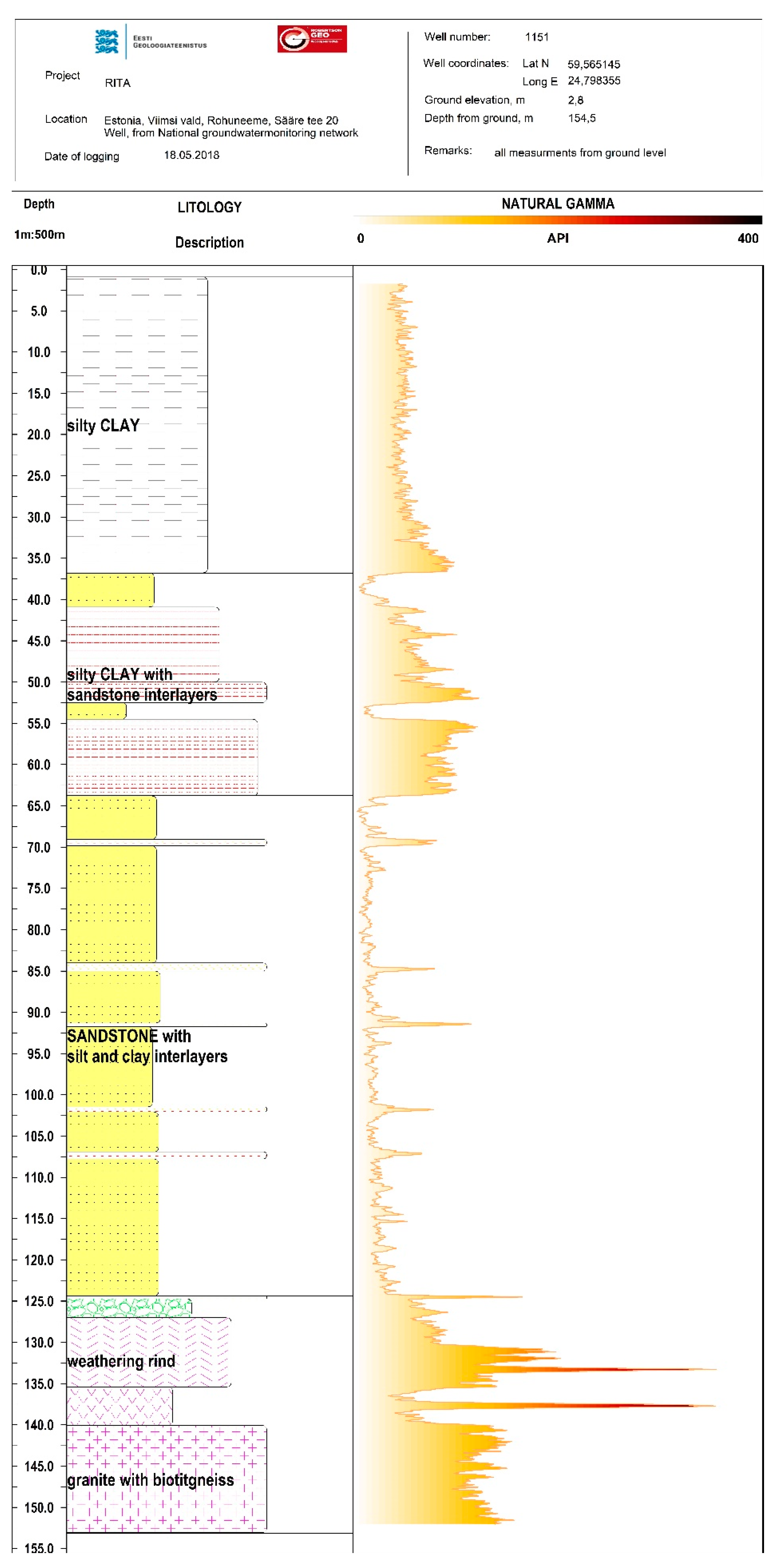
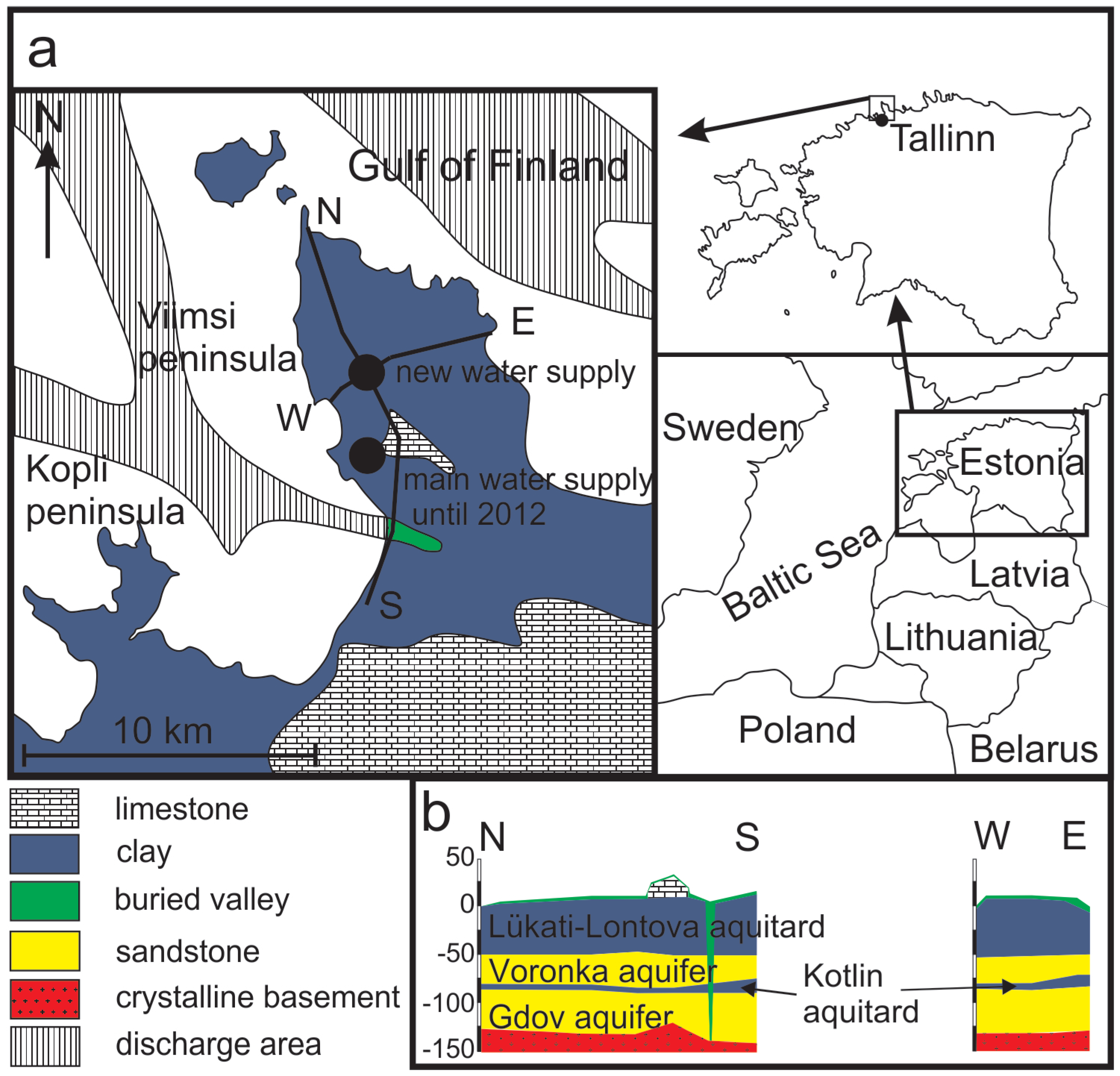
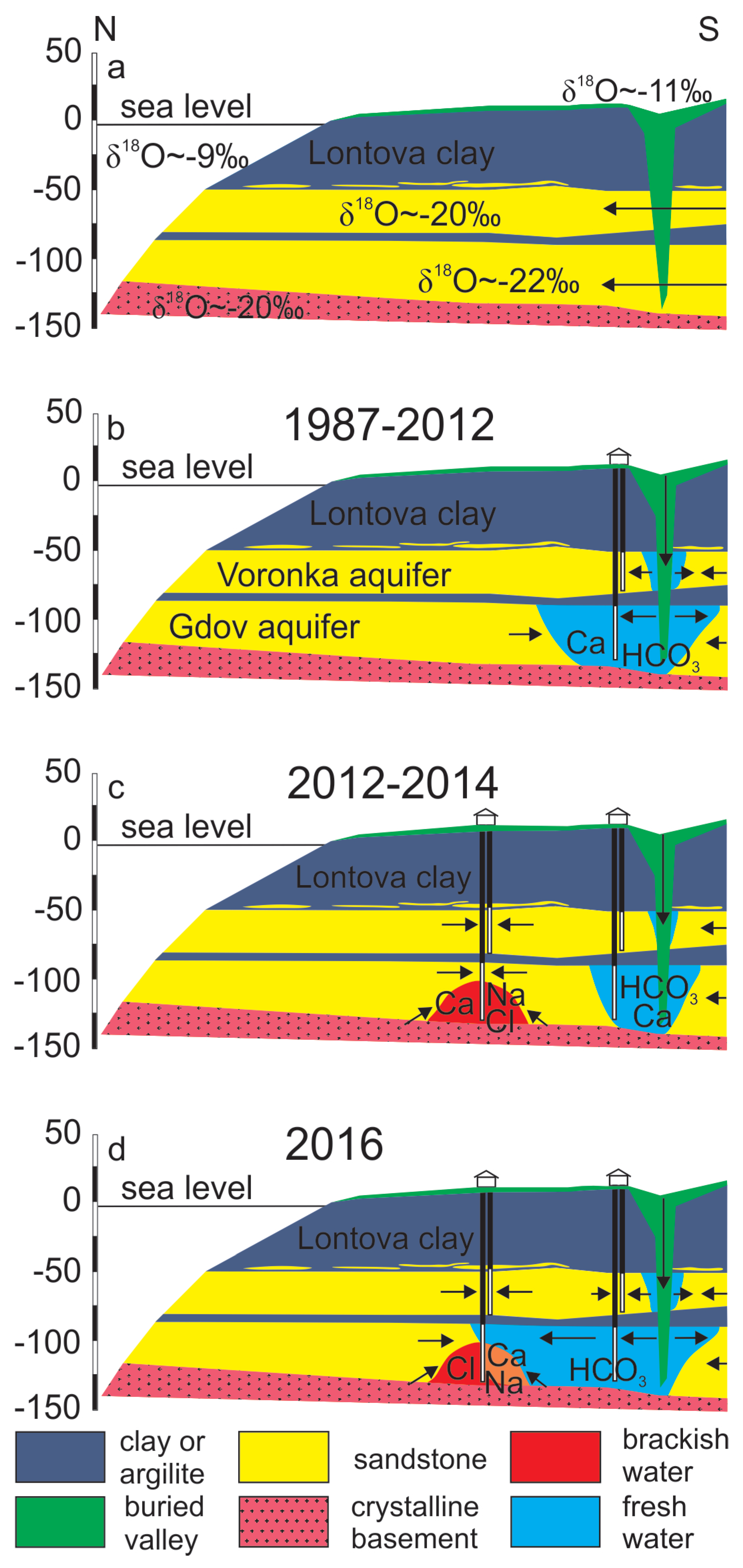
2. Geology and Hydrogeological Setting
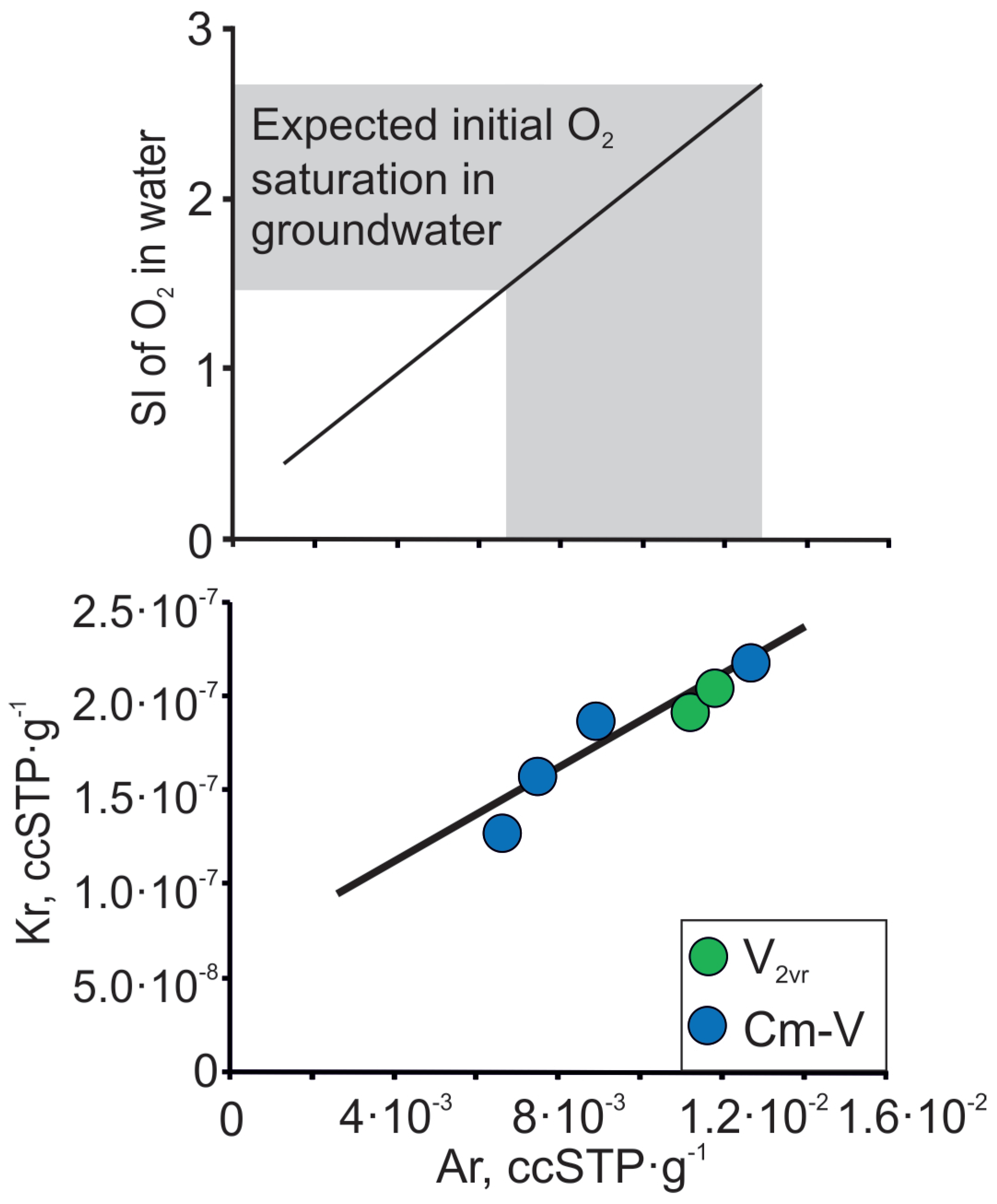
3. Material and Methods
4. Results and Discussion
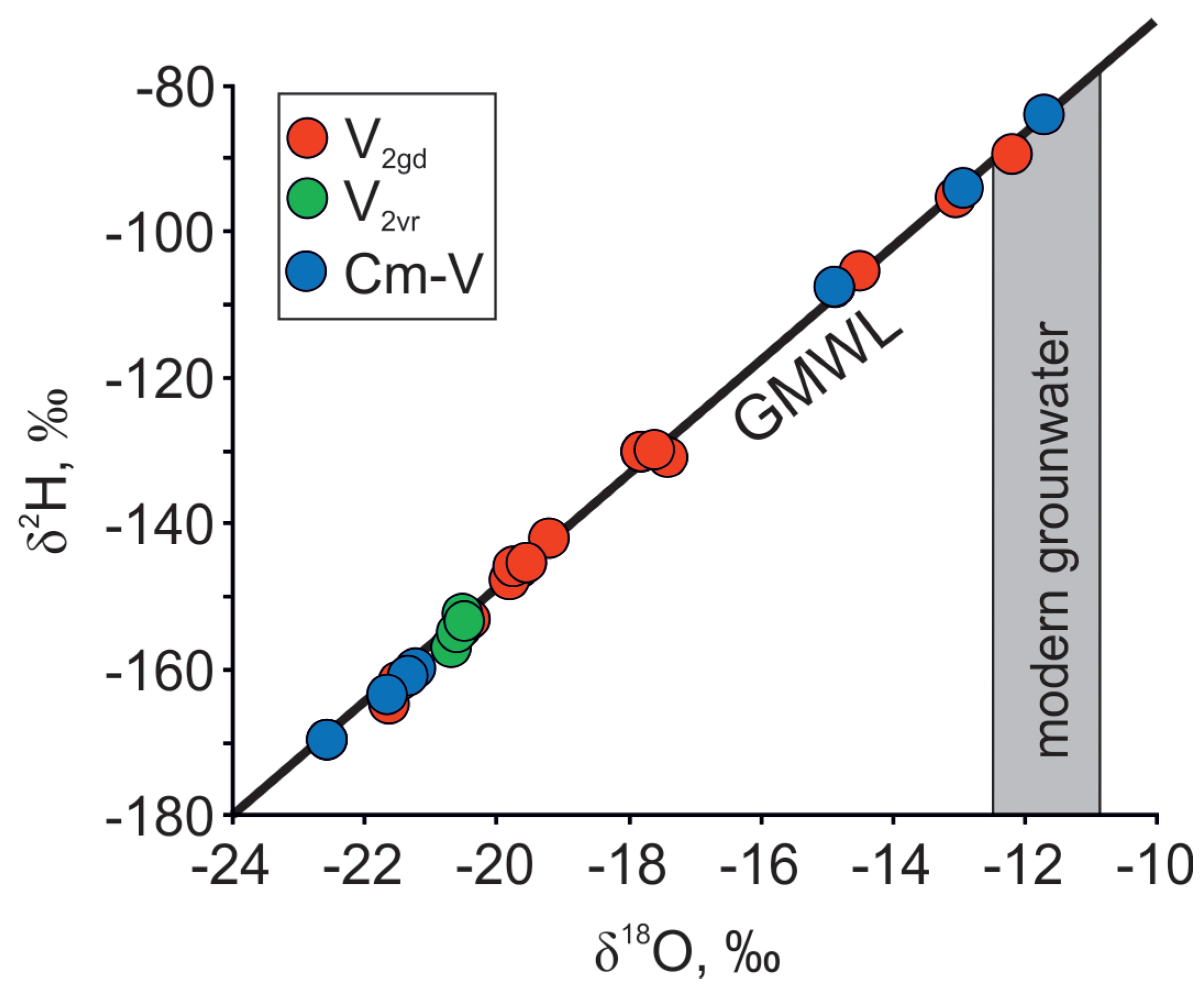
4.1. Mixing
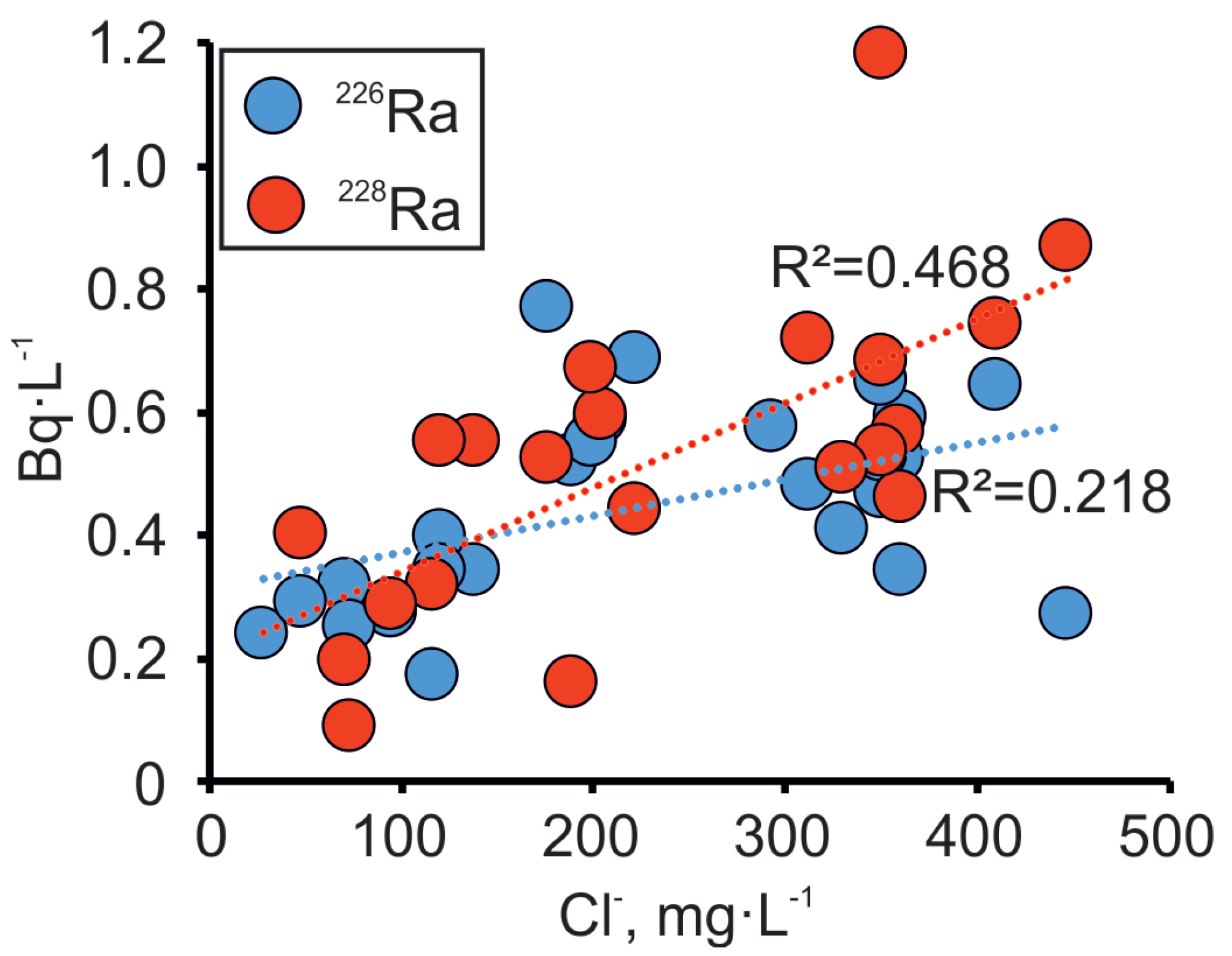
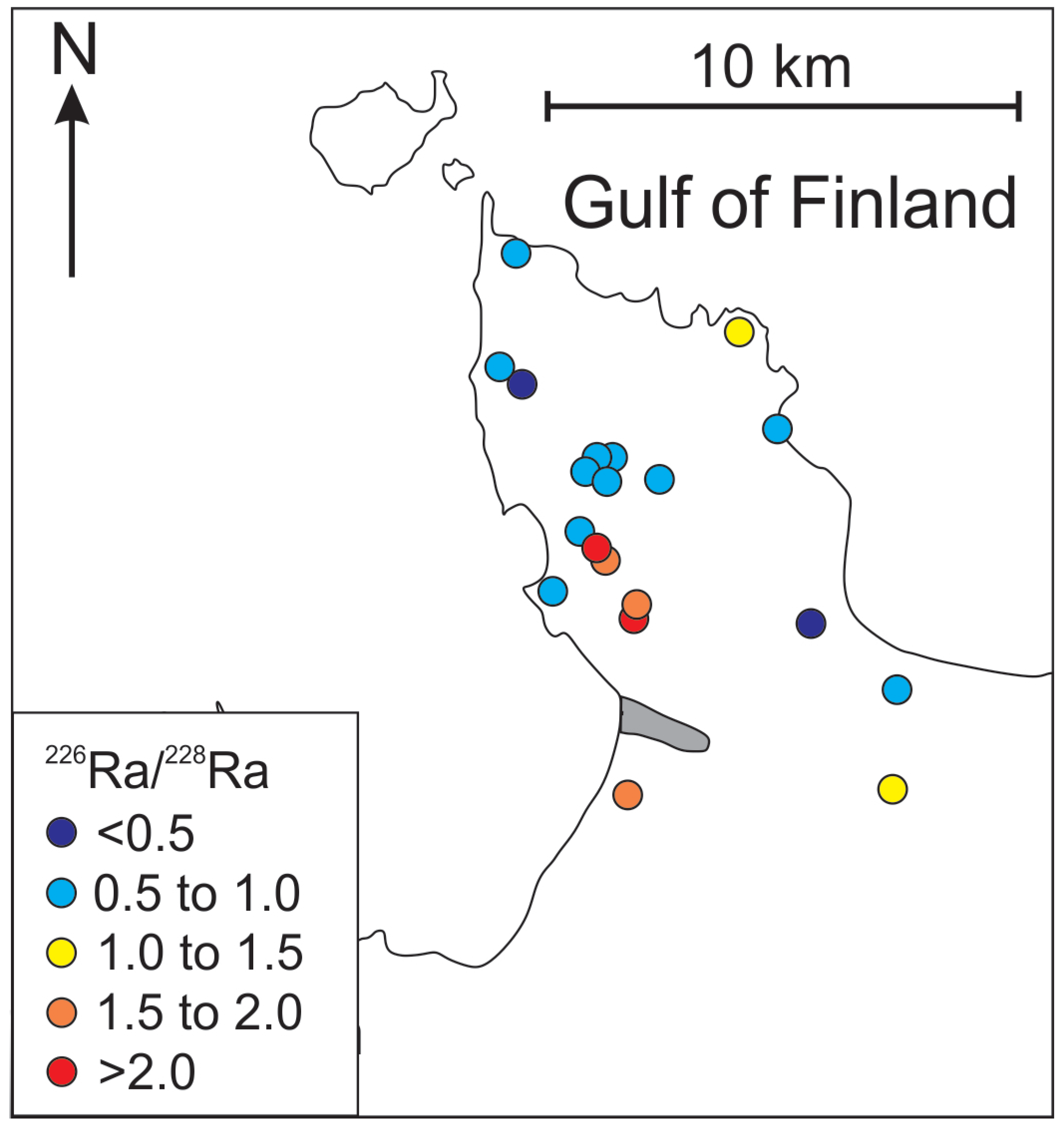
4.2. Spatial Distribution and the Origin of Radium in Groundwater
4.3. Secondary U Deposits as the Source of 226Ra
4.4. Future Prospects
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Appendix A
References
- Ferguson, G.; Gleeson, T. Vulnerability of coastal aquifers to groundwater use and climate change. Nat. Clim. Chang. 2012, 2, 342–345. [Google Scholar] [CrossRef]
- Werner, A.D.; Bakker, M.; Post, V.E.A.; van den Bohede, A.; Lu, C.; Ataie-Ashtiani, B.; Simmons, C.T.; Barry, D.A. Seawater intrusion processes, investigation and management: Recent advances and future challenges. Adv. Water Resour. 2013, 5, 3–26. [Google Scholar] [CrossRef]
- Wetzel, M.; Kühn, M. Salinization of freshwater aquifers due to subsurface fluid injection quantified by species transport simulations. Energy Procedia 2016, 97, 411–418. [Google Scholar] [CrossRef]
- Colombani, N.; Osti, A.; Volta, G.; Mastrocicco, M. Impact of climate change on salinization of coastal water resources. Water Resour. Manag. 2016, 30, 2483–2496. [Google Scholar] [CrossRef]
- Delfs, J.-O.; Nordbeck, J.; Bauer, S. Upward brine migration resulting from pressure increases in a layered subsurface system. Env. Earth Sci. 2016, 75, 1441. [Google Scholar] [CrossRef]
- Vallejos, A.; Sola, F.; Yechieli, Y.; Pulido-Bosch, A. Influence of the paleogeographic evolution on the groundwater salinity in a coastal aquifer. Cabo De Gata Aquiferse Spain J. Hydrol. 2018, 557, 55–66. [Google Scholar]
- Edmunds, W.M. Paleowater in European coastal aquifers—The goals and main conclusions of the PALAEAUX project. In Paleowaters in Coastal Europe: Evolution of Groundwater Since the Late Pleistocene; Edmunds, W.M., Milne, C.J., Eds.; Geological Society London Special Publications: London, UK, 2001; pp. 1–16. [Google Scholar]
- Cary, L.; Petelet-Giraud, E.; Bertrand, G.; Kloppmann, W.; Aquilina, L.; Martins, V.; Hirata, R.; Montenegro, S.; Pauwels, H.; Chatton, E.; et al. Origins and processes of groundwater salinization in the urban coastal aquifers of Recife (Pernambuco, Brazil): A multi-isotope approach. Sci. Total Environ. 2015, 530–531, 411–429. [Google Scholar] [CrossRef] [PubMed]
- Daniele, L.; Vallejos, A.; Corbellá, M.; Molina, L.; Pulido-Bosch, A. Hydrogeochemistry and geochemical simulations to assess water–rock interactions in complex carbonate aquifers: The case of Aguadulce (SE Spain). Appl. Geochem. 2013, 29, 43–54. [Google Scholar] [CrossRef]
- Mollema, P.; Antonellini, M.; Dinelli, E.; Gabbianelli, G.; Greggio, N.; Stuyfzand, P. Hydrochemical and physical processes influencing salinization and freshening in mediterranean low-lying coastal environments. Appl. Geochem. 2013, 34, 207–221. [Google Scholar] [CrossRef]
- Carreira, P.M.; Marques, J.M.; Nunes, D. Source of groundwater salinity in coastline aquifers based on environmental isotopes (Portugal): Natural vs. human interference—A review and reinterpretation. Appl. Geochem. 2014, 41, 163–175. [Google Scholar] [CrossRef]
- Valocchi, A.J.; Street, R.L.; Roberts, P.V. Transport of ion-exchanging solutes in groundwater: Chromatographic theory and field simulation. Water Resour. Res. 1981, 17, 1517–1527. [Google Scholar] [CrossRef]
- Appelo, C.A.J. Cation and proton exchange, pH variations, and carbonate reactions in a freshening aquifer. Water Resour. Res. 1994, 30, 2793–2805. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Willemsen, A. Geochemical calculations and observations on salt water intrusions. I: A combined geochemical/mixing cell model. J. Hydrol. 1987, 94, 313–330. [Google Scholar] [CrossRef]
- Van der Kemp, W.; Appelo, C.A.J.; Walraevens, K. Inverse chemical modeling and radiocarbon dating of palaeogroundwaters: The Tertiary Ledo-Paniselian aquifer in Flanders, Belgium. Water Resour. Res. 2000, 36, 1277–1287. [Google Scholar] [CrossRef]
- Martinez, D.E.; Bocanegra, E.M. Hydrochemistry and cation-exchange processes in the coastal aquifer of Mar Del Plata, Argentina. Hydrogeol. J. 2002, 10, 393–408. [Google Scholar] [CrossRef]
- Giménez, F.E. Dynamic of sea water interface using hydrochemical facies evolution diagram. Ground Water 2010, 48, 212–216. [Google Scholar] [CrossRef]
- Raidla, V.; Kirsimäe, K.; Vaikmäe, R.; Jõeleht, A.; Karro, E.; Marandi, A.; Savitskaja, L. Geochemical evolution of groundwater in the Cambrian–Vendian aquifer system of the Baltic Basin. Chem. Geol. 2009, 258, 219–231. [Google Scholar] [CrossRef]
- Raidla, V.; Kern, Z.; Pärn, J.; Babre, A.; Erg, K.; Ivask, J.; Kalvāns, A.; Kohán, B.; Lelgus, M.; Martma, T.; et al. A δ18O isoscape for the shallow groundwater in the Baltic Artesian Basin. J. Hydrol. 2016, 542, 254–267. [Google Scholar] [CrossRef]
- Vaikmäe, R.; Vallner, L.; Loosli, H.H.; Blaser, P.C.; Juillard-Tardent, M. Palaeogroundwater of glacial origin in the Cambrian-Vendian aquifer of northern Estonia. In Palaeowaters of Coastal Europe: Evolution of Groundwater since the late Pleistocene; Edmunds, W.M., Milne, C.J., Eds.; Geological Society: London, UK, 2001; Volume 189, pp. 17–27. [Google Scholar]
- Raidla, V.; Kirsimäe, K.; Vaikmäe, R.; Kaup, E.; Martma, T. Carbon isotope systematics of the Cambrian–Vendian aquifer system in the northern Baltic Basin: Implications to the age and evolution of groundwater. Appl. Geochem. 2012, 27, 2042–2052. [Google Scholar] [CrossRef]
- Perens, R.; Savitski, L.; Savva, V.; Jaštšuk, S.; Häelm, M. Delineation of Groundwater Bodies and Description of Their Boundaries and Hydrogeological Conceptual Models; Geological Survey of Estonia: Tallinn, Estonia, 2012. (In Estonian) [Google Scholar]
- Erg, K.; Truu, M.; Kebbinau, K.; Lelgus, M.; Tarros, S. Report of Estonian Environmental Monitoring. Monitoring of Groundwater Bodies in 2016 of State Environmental Programme; Geological Survey of Estonia: Tallinn, Estonia, 2017. (In Estonia) [Google Scholar]
- Suursoo, S.; Hill, L.; Raidla, V.; Kiisk, M.; Jantsikene, A.; Nilb, N.; Czuppon, G.; Putk, K.; Munter, R.; Koch, R.; et al. Temporal changes in radiological and chemical composition of Cambrian-Vendian groundwater in conditions of intensive water consumption. Sci. Total Environ. 2017, 601–602, 679–690. [Google Scholar] [CrossRef]
- Savitskaja, L.; Viigand, A. Report of Microcomponent and Isotope Composition Research in Cm–V Aquifer Groundwater for Estimating Drinking Water Quality in North Estonia; Geological Survey of Estonia: Tallinn, Estonia, 1994. (In Estonian) [Google Scholar]
- Mokrik, R.; Karro, E.; Savitskaja, L.; Drevaliene, G. The origin of barium in the Cambrian-Vendian aquifer system, North Estonia. Est. J. Earth Sci. 2009, 58, 193–208. [Google Scholar] [CrossRef]
- Forte, M.; Bagnato, L.; Caldognetto, E.; Risica, S.; Trotti, F.; Rusconi, R. Radium isotopes in Estonian groundwater: Measurements, analytical correlations, population dose and a proposal for a monitoring stradegy. J. Radiat. Prot. 2010, 30, 761–780. [Google Scholar] [CrossRef] [PubMed]
- Vinson, D.S.; Vengosh, A.; Hirschfeld, D.; Dwyer, G.S. Relationships between radium and radon occurrence and hydrochemistry in fresh groundwater from fractured crystalline rocks, North Carolina (USA). Chem. Geol. 2009, 260, 159–171. [Google Scholar] [CrossRef]
- Vengosh, A.; Hirschfeld, D.; Vinson, D.; Dwyer, G.; Raanan, H.; Rimawi, O.; Al-Zoubi, A.; Akkawi, E.; Marie, A.; Haquin, G.; et al. High naturally occurring radioactivity in fossil groundwater from the Middle East. Environ. Sci. Technol. 2009, 43, 1769–1775. [Google Scholar] [CrossRef] [PubMed]
- Szabo, Z.; DePaul, V.T.; Fischer, J.M.; Kraemer, T.F.; Jacobsen, E. Occurrence and geochemistry of radium in water from principal drinking-water aquifer systems of the United States. Appl. Geochem. 2012, 27, 729–752. [Google Scholar] [CrossRef]
- Langmuir, D.; Riese, A.C. The thermodynamic properties of radium. Geochim. Cosmochim. Acta 1985, 49, 1593–1601. [Google Scholar] [CrossRef]
- Kraemer, T.F.; Reid, D.F. The occurrence and behavior of radium in saline formation water of the U.S. Gulf Coast region. Chem. Geol. 1984, 2, 153–174. [Google Scholar] [CrossRef]
- Sturchio, N.C.; Banner, J.L.; Binz, C.M.; Heraty, L.B.; Musgrove, M. Radium geochemistry of ground waters in Paleozoic carbonate aquifers, midcontinent, USA. Appl. Geochem. 2001, 16, 109–122. [Google Scholar] [CrossRef]
- Swarzenski, P.W.; Baskaran, M.; Rosenbauer, R.J.; Edwards, B.D.; Land, M. A combined radio- and stable-isotopic study of a California coastal aquifer system. Water 2013, 5, 480–504. [Google Scholar] [CrossRef]
- Sherif, M.I.; Linb, J.; Poghosyan, A.; Abouelmagd, A.; Sultan, M.I.; Sturchio, N.C. Geological and hydrogeochemical controls on radium isotopes in groundwater of the Sinai Peninsula, Egypt. Sci. Total Environ. 2018, 613–614, 877–885. [Google Scholar] [CrossRef]
- Porcelli, D.; Swarzenski, P.W. The behavior of U- and Th-series nuclides in groundwater. Rev. Mineral. Geochem. 2003, 52, 317–361. [Google Scholar] [CrossRef]
- Vaaramaa, K.; Lehto, J.; Ervanne, H. Soluble and Particle-Bound 234,238U, 226Ra and 210Po in ground waters. Radiochim. Acta 2003, 91, 21–27. [Google Scholar] [CrossRef]
- Vinson, D.S.; Tagma, T.; Bouchaou, L.; Dwyer, G.; Warner, N.R. Occurrence and mobilization of radium in fresh to saline coastal groundwater inferred from geochemical and isotopic tracers. Appl. Geochem. 2013, 38, 161–175. [Google Scholar] [CrossRef]
- Perens, R.; Vallner, L. Water-bearing formation. In Geology and Mineral Resources of Estonia; Raukas, A., Teedumäe, A., Eds.; Estonian Academy Publishers: Tallinn, Estonia, 1997; pp. 137–145. [Google Scholar]
- Puura, V.; Vaher, R.; Klein, V.; Koppelmaa, H.; Niin, M.; Vanamb, V.; Kirs, J. Zoning and stratification of the rock complexes of the cristaline basement. In The Crystalline Basement of Estonian Territory; Viiding, H., Ed.; Nauka: Moscow, Russia, 1983; pp. 15–45. (In Russian) [Google Scholar]
- Kirs, J.; Puura, V.; Soesoo, A.; Klein, V.; Konsa, M.; Koppelmaa, H.; Niin, M.; Urtson, K. The crystalline basement of Estonia: Rock complexes of the Palaeoproterozoic Orosirian and Statherian and Mesoproterozoic Calymmian periods, and regional correlations. Est. J. Earth Sci. 2009, 58, 219–228. [Google Scholar] [CrossRef]
- Karro, E.; Marandi, A.; Vaikmäe, R. The origin of increased salinity in the Cambrian-Vendian aquifer system on the Kopli Peninsula, northern Estonia. Hydrogeol. J. 2004, 12, 424–435. [Google Scholar] [CrossRef]
- Savitskaja, L.; Jaštšuk, S. Determination of Radionuclides Concentrations in Groundwater from Cambrian-Vendian Aquifer System and Assessment of Its Conformity to the Norms set by EU Drinking Water Directive 98/83/EC; Geological Survey of Estonia: Tallinn, Estonia, 2001. (In Estonian) [Google Scholar]
- Suursoo, S.; Kiisk, M.; Al-Malahmeh, A.; Jantsikene, A.; Lumiste, L. 226Ra measurement by lsc as a tool to assess the efficiency of a water treatment technology for removing radionuclides from groundwater. Appl. Radiat. Isot. 2014, 93, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Aeschbach-Hertig, W.; Solomon, D.K. Noble gas thermometry in groundwater hydrology. In The Noble Gases as Geochemical Tracers; Burnard, P., Ed.; Springer: Berlin, Germany, 2013; pp. 81–122. [Google Scholar]
- Yezhova, M.; Polyakov, V.; Tkachenko, A.; Savitski, L.; Belkina, V. Palaeowaters of North Estonia and their influence on changes of resources and the quality of fresh groundwaters of large coastal water supplies. Geologija 1996, 19, 37–40. [Google Scholar]
- Savitski, L.; Viigand, A.; Belkina, V.; Jashtshuk, S. The Estimation of Groundwater Resources in Tallinn. Seeking Groundwater Supplies for Tallinn and Its Vicinity; Geological Survey of Estonia: Tallinn, Estonia, 1993. (In Estonian) [Google Scholar]
- Raidla, V.; Kirsimäe, K.; Ivask, J.; Kaup, E.; Knöller, K.; Marandi, A.; Martma, T.; Vaikmäe, R. Sulphur isotope composition of dissolved sulphate in the Cambrian-Vendian aquifer system in the northern part of the Baltic Artesian Basin. Chem. Geol. 2014, 383, 147–154. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Feistel, R.; Weinreben, S.; Wolf, H.; Seitz, S.; Spitzer, P.; Adel, B.; Nausch, G.; Schneider, B.; Wright, D.G. Density and Absolute Salinity of the Baltic Sea 2006–2009. Ocean Sci. 2010, 6, 3–24. [Google Scholar] [CrossRef]
- Arhangelski, B.N.; Fedorova, A.M. Cambrian–Vendian aquifer system. In Hydrogeology in U.S.S.R. XXX; Arhangelski, B.N., Ed.; Nedra: Moscow, Russia, 1966; pp. 170–192. (In Russian) [Google Scholar]
- Petersell, V.; Kivisilla, J.; Pukkonen, E.; Põldvere, A.; Täht, K. Evaluation of Ore Events and Mineralization Points in Estonian Bedrock and Crystalline Basement; Geological Survey of Estonia: Tallinn, Estonia, 1991. (In Russian) [Google Scholar]
- Raudsep, R. Mineral resources, mineral occurrences. In Geology and Mineral Resources of Estonia; Raukas, A., Teedumäe, A., Eds.; Estonian Academy Publishers: Tallinn, Estonia, 1997; pp. 369–372. [Google Scholar]
- Raidla, V.; Kirsimäe, K.; Bityukova, L.; Jõeleht, A.; Shogenova, A.; Šliaupa, S. Lithology and diagenesis of the poorly consolidated Cambrian siliciclastic sediments in the northern Baltic Sedimentary Basin. Geol. Q. 2006, 50, 11–22. [Google Scholar]
- Liivamägi, S.; Somelar, P.; Mahaney, W.C.; Kirs, J.; Vircava, I.; Kirsimäe, K. Late Neoproterozoic Baltic paleosol: Intense weathering at high latitude? Geology 2014, 42, 323–326. [Google Scholar] [CrossRef]
- Liivamägi, S.; Somelar, P.; Vircava, I.; Mahaney, W.C.; Kirs, J.; Kirsimäe, K. Petrology, mineralogy and geochemical climofunctions of the Neoproterozoic Baltic paleosol. Precambrian Res. 2015, 256, 170–188. [Google Scholar] [CrossRef]
- Stackelberg, P.E.; Szabo, Z.; Jurgens, B.C. Radium mobility and the age of groundwater in public-drinking-water supplies from the Cambrian-Ordovician aquifer system, north-central USA. Appl. Geochem. 2018, 89, 34–48. [Google Scholar] [CrossRef]
- Swarzenski, P.W. U/Th series radionuclides as tracers of coastal groundwater. Chem. Rev. 2007, 107, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, D. Aqueous Environmental Geochemistry, 1st ed.; Prentice Hall: New Jersey, NJ, USA, 1997. [Google Scholar]
- King, P.T.; Michel, J.; Moore, W.S. Ground water geochemistry of 228Ra, 226Ra and 222Rn. Geochim. Cosmochim. Acta 1982, 46, 1173–1182. [Google Scholar] [CrossRef]
- Chau, N.D.; Lucyna, R.; Jakub, N.; Paweł, J. Radium isotopes in the Polish Outer Carpathian mineral waters of various chemical composition. J. Environ. Radioact. 2012, 112, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Rosholt, J.N. Isotopic composition of uranium and thorium in crystalline rocks. J. Geophys. Res. Solid Earth 1983, 88, 7315–7330. [Google Scholar] [CrossRef]
- Langmuir, D. Uranium solution–mineral equilibria at low temperatures with applications to sedimentary ore deposits. Geochim. Cosmochim. Acta 1978, 42, 547–569. [Google Scholar] [CrossRef]
- Herczeg, A.L.; Simpson, H.J.; Anderson, R.F.; Tfjer, R.M.; Mathieu, G.G.; Deck, B.L. Uranium and radium mobility in groundwaters and brines within the Delaware basin, southeastern New Mexico, U.S.A. Chem. Geol. 1988, 72, 181–196. [Google Scholar]
- Guerrero, J.L.; Vallejos, A.; Ceron, J.C.; Sanchez-Martos, F.; Pulido-Bosch, A.; Bolívar, J.P. U-isotopes and 226Ra as tracers of hydrogeochemical processes in carbonated karst aquifers from arid areas. J. Environ. Radioact. 2016, 158–159, 9–20. [Google Scholar] [CrossRef]
- Kipfer, R.; Aeschbach-Hertig, W.; Peters, F.; Stute, M. Noble Gases in Lakes and Groundwaters; Porcelli, D., Ballentine, C.J., Wieler, R., Eds.; Mineralogical Society of America: Washington, DC, USA, 2002; pp. 615–690. [Google Scholar]
- Grundl, T.; Magnusson, N.; Brennwald, M.S.; Kipfer, R. Mechanisms of subglacial groundwater recharge as derived from noble gas, 14C, and stable isotopic data. Earth Planet. Sci. Lett. 2013, 369–370, 78–85. [Google Scholar] [CrossRef]
- Severinghaus, J.P.; Sowers, T.; Brook, E.J.; Alley, R.B.; Michael, L.; Bender, M.L. Timing of abrupt climate change at the end of the Younger Dryas interval from thermally fractionated gases in polar ice. Nature 1998, 391, 141–146. [Google Scholar] [CrossRef]
- Gilkeson, R.H.; Cartwright, K.; Cowart, J.B.; Holtzman, R.B. Hydrogeologic and Geochemical Studies of Selected Natural Radioisotopes and Barium in Groundwater in Illinois; Final Technical Completion Report to U.S. Bureau of Reclamation, Project B-108-ILL; University of Illinois at Urbana-Champaign, Water Resources Center: St Paul, MN, USA, 1983. [Google Scholar]
- Drake, H.; Suksi, J.; Tullborg, E.-L.; Lahaye, Y. Quaternary redox transitions in deep crystalline rock fractures at the western margin of the Greenland ice sheet. Appl. Geochem. 2017, 76, 196–209. [Google Scholar] [CrossRef]
- FinEst Link. 2018. Available online: http://www.finestlink.fi/en/ (accessed on 14 January 2018).
- Pärn, J. Origin and Geochemical Evolution of Palaeogroundwater in the Northern Part of the Baltic Artesian Basin. Doctoral Thesis, Tallinn University of Technology, Tallinn, Estonia, 2018; p. 226. [Google Scholar]
- Clayton, R.N.; Friedman, I.; Graf, D.L.; Mayeda, T.K.; Meents, W.F.; Shimp, N.F. The origin of saline formation waters. Isotopic composition. J. Geophys. Res. 1966, 71, 3869–3882. [Google Scholar] [CrossRef]
- Grasby, S.E.; Osadetz, K.; Betcher, R.; Render, F. Reversal of the regional-scale flowsystem of the Williston Basin in response to Pleistocene glaciation. Geology 2000, 29, 635–638. [Google Scholar] [CrossRef]
- McIntosh, J.C.; Walter, L.M. Paleowaters in Silurian–Devonian carbonate aquifers: Geochemical evolution of groundwater in the Great Lakes region since the late Pleistocene. Geochim. Cosmochim. Acta 2006, 70, 2454–2479. [Google Scholar] [CrossRef]
- Person, M.; McIntosh, J.C.; Remenda, V.; Bense, V. Pleistocene hydrology of North America: The role of ice sheets in reorganizing groundwater systems. Rev. Geophys. 2007, 45, RG3007. [Google Scholar] [CrossRef]
- Siegel, D.I.; Mandle, R.J. Isotopic evidence for glacial meltwater recharge to the Cambrian-Ordovician aquifer, north-central United States. Quat. Res. 1984, 22, 328–335. [Google Scholar] [CrossRef]
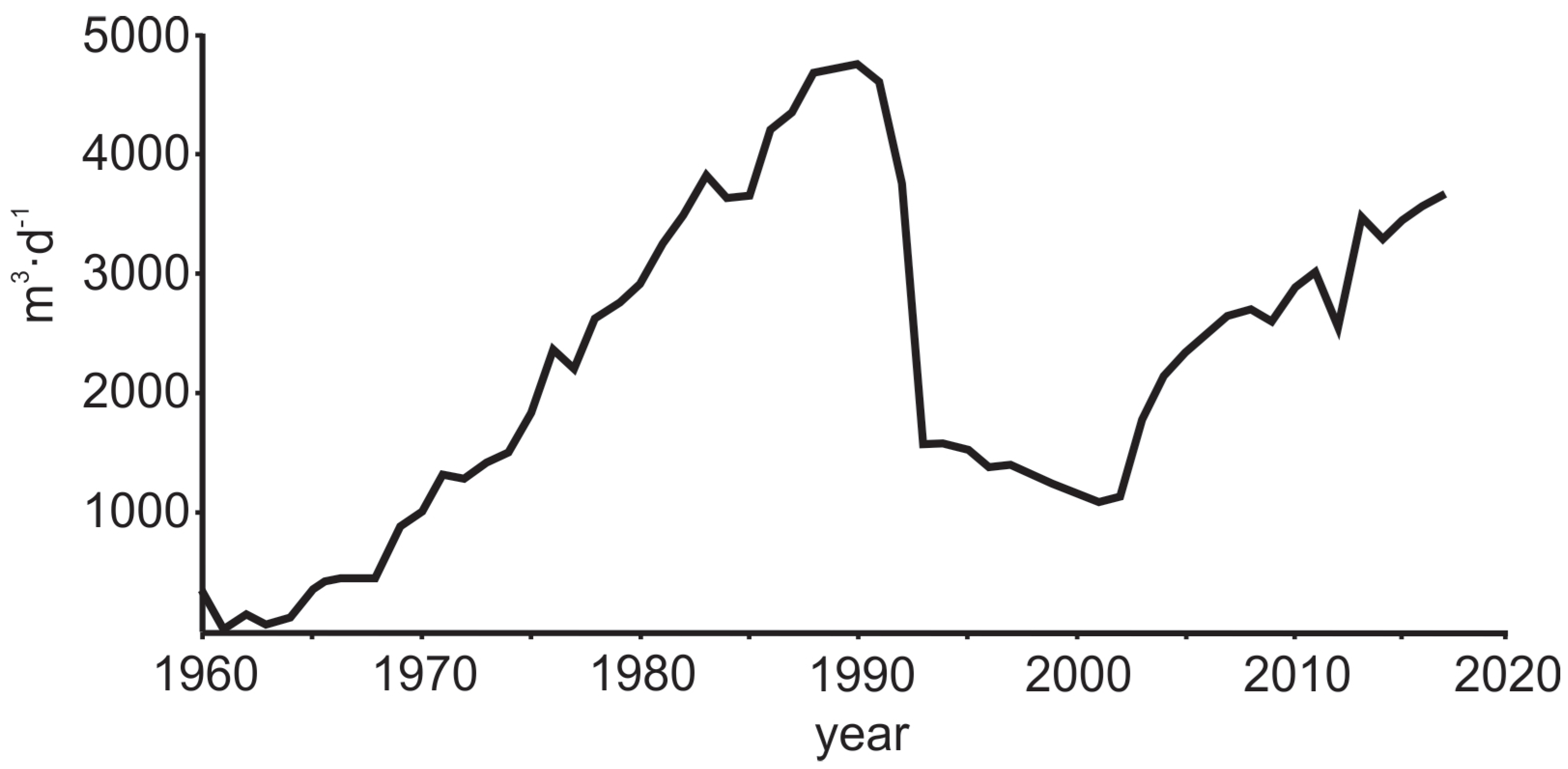
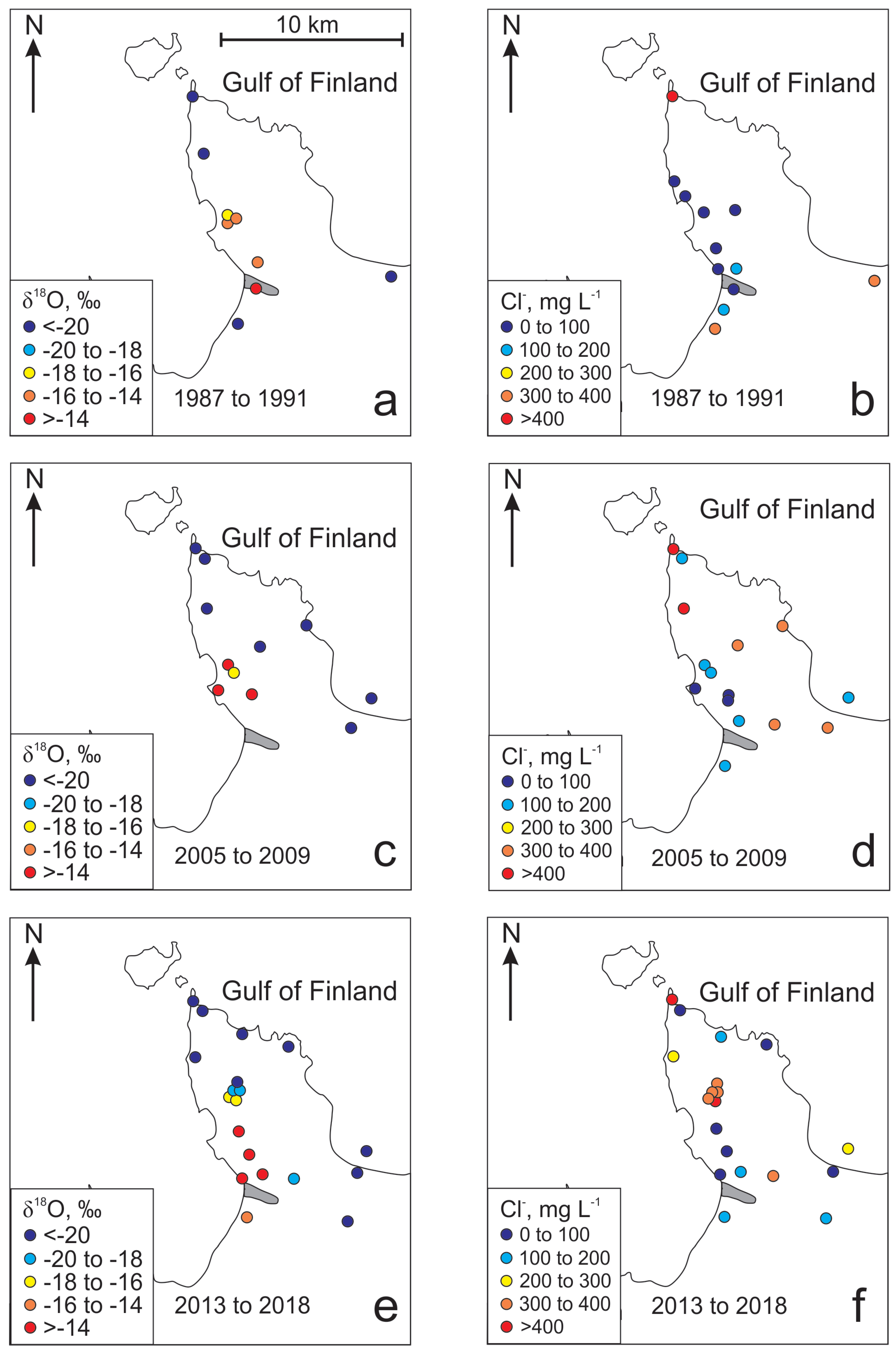
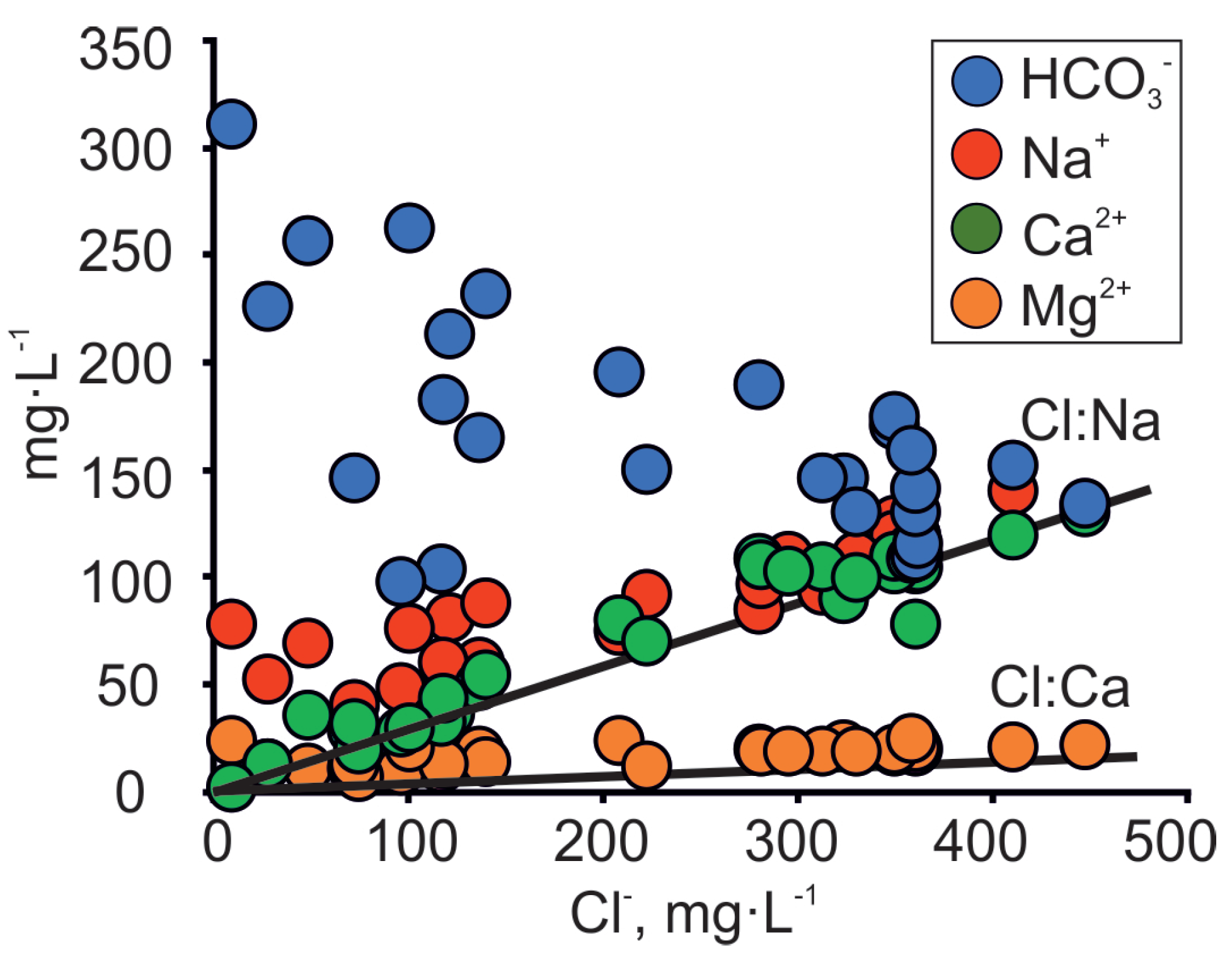
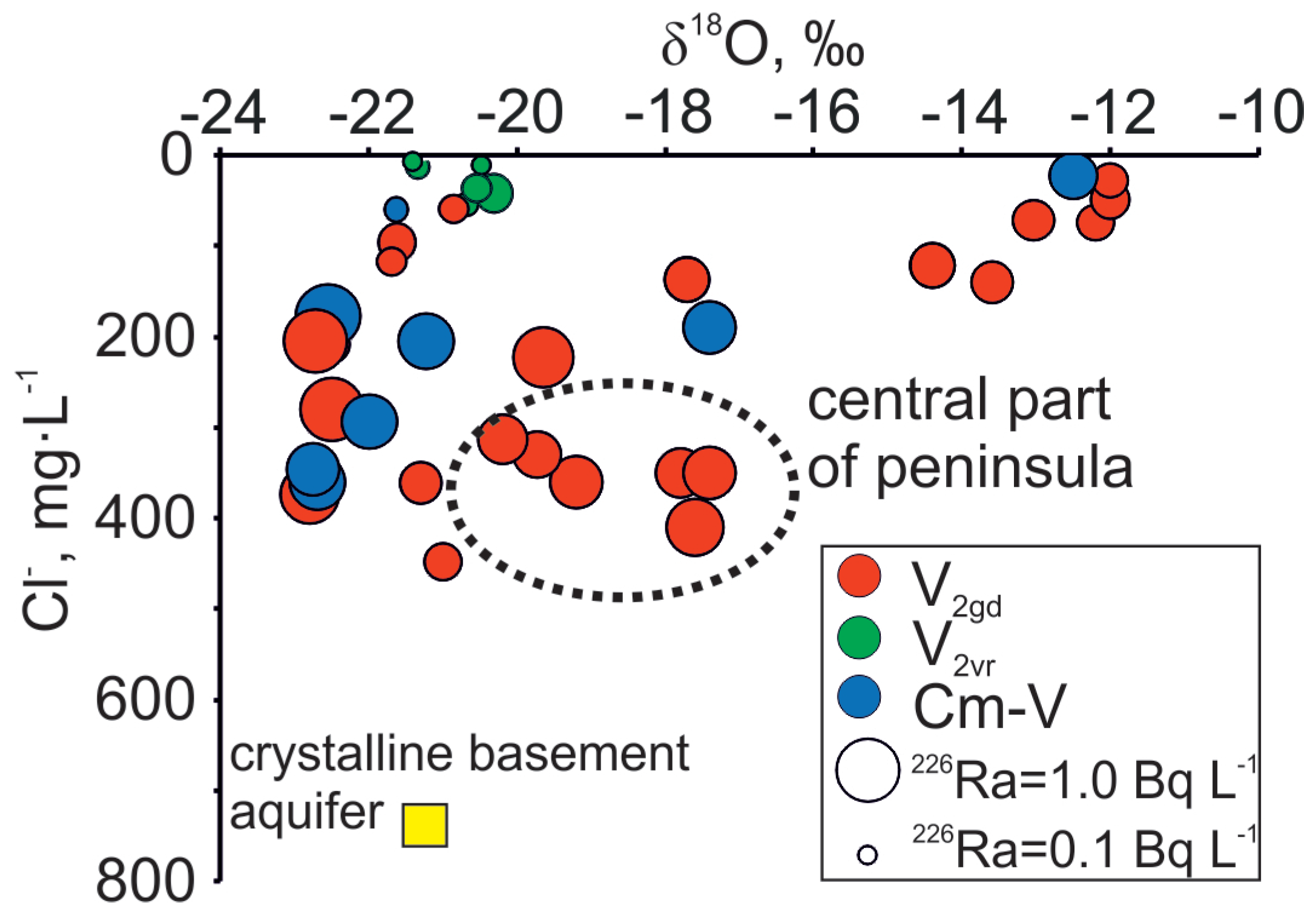
| No | Well ID | Date | Depth | pH | EC | T | δ18O | δ2H | 226Ra | 228Ra | Ca2+ | Mg2+ | Na+ | K+ | HCO3− | Cl− | SO42− | Br− | Ba2+ | Mn2+ | NH4+ | NO3− | Sr2+ | Fe2+ | F− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m | μS·cm−1 | °C | ‰ VSMOW | Bq·L−1 | mg·L−1 | ||||||||||||||||||||
| Voronka aquifer (V2vr) | |||||||||||||||||||||||||
| 12 | 124 | 01.06.1988 | 101.5 | −21.5 | |||||||||||||||||||||
| 25 | 124 | 13.04.2005 | 101.5 | 8.0 | 411 | 8.4 | −20.8 | 0.16 | 0.10 | 34.5 | 10.1 | 34.7 | 7.3 | 146.4 | 56.7 | bdl | 0.2 | bdl | 0.07 | 0.18 | |||||
| 3 | 158 | 26.06.1990 | 110 | 7.6 | 102.1 | 21.3 | 79.1 | 1.0 | 311.2 | 12.8 | 33.3 | 0.03 | bdl | 12.10 | |||||||||||
| 4 | 158 | 28.10.2014 | 110 | −14.9 | −107.9 | ||||||||||||||||||||
| 5 | 171 | 08.10.2014 | 95 | 8.2 | 461 | 8.4 | −20.7 | −157.2 | 0.11 | 0.30 | 44.7 | 13.5 | 29.6 | 8.8 | 170.8 | 53.3 | 0.7 | bdl | bdl | 0.41 | 0.47 | ||||
| 65 | 179 | 12.04.2005 | 90 | 7.8 | 364 | 8.2 | −20.3 | 0.40 | 0.16 | 30.9 | 13.6 | 28.0 | 8.7 | 183.1 | 41.1 | bdl | 0.7 | bdl | 0.05 | 0.13 | |||||
| 7 | 179 | 09.06.2014 | 90 | 8.1 | 405 | 8.1 | −20.5 | −154.6 | 0.07 | 0.20 | 35.9 | 13.2 | 28.1 | 8.5 | 36.5 | 0.1 | bdl | 0.37 | 0.27 | 0.17 | 0.63 | 0.49 | |||
| 82 | 185 | 11.08.1991 | 95 | −20.4 | |||||||||||||||||||||
| 95 | 187 | 13.04.2005 | 70 | 7.8 | 254 | 8.3 | −21.3 | 0.13 | 0.10 | 22.6 | 8.4 | 15.0 | 6.0 | 152.5 | 11.0 | bdl | 0.6 | bdl | 0.05 | 0.08 | |||||
| 10 | 187 | 13.11.2013 | 70 | 8.0 | 136 | 8.5 | −21.3 | −161.1 | 0.11 | 0.25 | 21.9 | 3.7 | 11.7 | 4.2 | 155.6 | 6.7 | 0.2 | 1.2 | 0.08 | 0.10 | 0.01 | 0.23 | 0.17 | 0.29 | |
| 111 | 509 | 05.12.1989 | 44.1 | 21.9 | 42.6 | 220.0 | 80.0 | 1.2 | 0.40 | ||||||||||||||||
| 122 | 1153 | 04.07.1991 | 77.5 | −21.8 | |||||||||||||||||||||
| 133 | 1153 | 05.07.1994 | 77.5 | 7.4 | −21.4 | 23.2 | 10.0 | 18.2 | 7.0 | 158.7 | 20.6 | 5.9 | |||||||||||||
| 144 | 1153 | 30.08.1995 | 77.5 | −21.5 | 36.1 | 32.7 | 128.1 | 81.0 | 13.0 | ||||||||||||||||
| 155 | 1153 | 13.04.2005 | 77.5 | 7.8 | 253 | 8.5 | −21.5 | bdl | 0.11 | 24.6 | 7.2 | 16.7 | 7.0 | 140.3 | 8.5 | bdl | 0.7 | bdl | 0.07 | 0.14 | |||||
| 164 | 8914 | 30.08.1995 | 75 | −20.0 | |||||||||||||||||||||
| 176 | 8914 | 12.08.1996 | 75 | −20.3 | |||||||||||||||||||||
| 18 | 8914 | 06.09.2013 | 75 | 8.1 | 262 | 9.5 | 0.24 | 0.36 | 30.7 | 11.7 | 25.1 | 6.4 | 170.8 | 36.7 | bdl | bdl | 0.39 | 0.17 | 0.04 | 0.40 | bdl | 0.31 | |||
| 19 | 8914 | 02.09.2016 | 75 | 8.0 | 386 | 9.1 | −20.6 | −153.7 | 33.0 | 12.0 | 25.0 | 8.2 | 163.5 | 38.0 | bdl | bdl | bdl | bdl | 0.22 | 0.49 | 0.65 | ||||
| 20 | 11570 | 01.07.2016 | 100 | 8.1 | 649 | 9.1 | −21.4 | −161.0 | 37.0 | 8.3 | 20.0 | 5.6 | 109.0 | 68.0 | 14.0 | 0.7 | bdl | 0.60 | 0.12 | 0.14 | 0.45 | ||||
| 215 | 19570 | 13.04.2005 | 109 | 8.0 | 700 | 8.4 | −17.7 | 0.39 | 0.31 | 59.9 | 13.7 | 64.0 | 8.8 | 195.2 | 134.0 | bdl | 0.4 | bdl | 0.07 | 0.07 | |||||
| 228 | 23886 | 09.09.2016 | 124.5 | 8.2 | 1366 | 8.8 | −20.6 | −152.1 | 28.0 | 9.9 | 18.0 | 7.6 | 141.7 | 14.0 | bdl | bdl | bdl | bdl | 0.20 | 0.45 | 0.51 | ||||
| 258 | 25687 | 20.11.2012 | 82 | 7.8 | 294 | 8.8 | −20.5 | 28.0 | 10.0 | 22.0 | 7.3 | 23.2 | 4.6 | bdl | |||||||||||
| 26 | 25687 | 09.09.2016 | 82 | 8.0 | 359 | 8.8 | −20.5 | −152.4 | 32.0 | 12.0 | 21.0 | 8.2 | 174.4 | 27.0 | bdl | bdl | bdl | 0.41 | 0.19 | 0.49 | 0.47 | ||||
| 23 | 25689 | 13.11.2013 | 85 | 8.1 | 158.6 | 8.0 | 0.08 | 0.22 | 25.0 | 4.9 | 13.9 | 4.9 | 124.0 | 11.3 | 0.2 | bdl | 0.15 | 0.10 | 0.04 | 0.32 | 0.00 | 0.33 | |||
| 248 | 25689 | 05.08.2014 | 85 | 7.7 | 297 | 8.3 | −20.6 | −155.1 | 28.7 | 11.0 | 19.5 | 7.5 | 14.0 | bdl | 0.3 | bdl | 0.12 | 0.13 | 0.47 | 0.42 | |||||
| Gdov aquifer (V2gd) | |||||||||||||||||||||||||
| 27 | 122 | 14.09.2018 | 170 | 7.4 | 1022 | −19.5 | −145.6 | 113.8 | 25.0 | 108.7 | 11.0 | 158.6 | 358.1 | 16.5 | 0.3 | 0.56 | |||||||||
| 282 | 154 | 10.07.1991 | 134 | −20.3 | |||||||||||||||||||||
| 295 | 154 | 12.04.2005 | 130 | 7.8 | 1649 | 8.5 | −21.0 | 0.27 | 0.87 | 133.3 | 21.6 | 130.0 | 10.7 | 134.2 | 447.1 | bdl | 0.3 | bdl | 0.18 | 0.48 | |||||
| 302 | 160 | 01.06.1990 | 142 | −15.0 | |||||||||||||||||||||
| 311 | 160 | 30.07.1991 | 142 | −14.4 | 0.44 | ||||||||||||||||||||
| 32 | 160 | 08.10.2014 | 142 | 409 | 8.9 | −13.0 | −95.3 | 0.32 | 0.20 | 71.0 | bdl | bdl | 0.49 | 0.38 | |||||||||||
| 311 | 161 | 26.06.1990 | 140 | 7.7 | 78.0 | 23.6 | 2.4 | 1.0 | 311.2 | 9.2 | 30.5 | 0.05 | bdl | 6.10 | |||||||||||
| 343 | 161 | 13.04.2005 | 140 | 7.9 | 788 | 8.8 | −13.6 | 0.34 | 0.55 | 88.4 | 13.7 | 54.3 | 10.6 | 231.8 | 139.3 | bdl | 0.8 | bdl | 0.10 | 0.28 | |||||
| 35 | 170 | 01.04.2004 | 0.59 | 0.82 | |||||||||||||||||||||
| 365 | 172 | 12.04.2005 | 136 | 7.9 | 1226 | 8.5 | −20.2 | 0.48 | 0.72 | 93.8 | 19.3 | 105.0 | 11.4 | 146.4 | 313.0 | bdl | 0.3 | bdl | 0.15 | 0.36 | |||||
| 372 | 174 | 14.06.1988 | 145.4 | 7.7 | −12.0 | 0.24 | 52.7 | 13.5 | 13.0 | 4.4 | 225.8 | 27.7 | 0.0 | bdl | bdl | ||||||||||
| 385 | 183 | 12.04.2005 | 140 | 7.9 | 548 | 9.1 | −21.3 | 0.34 | 0.46 | 110.6 | 20.4 | 105.0 | 10.7 | 115.9 | 361.3 | bdl | 0.5 | bdl | 0.18 | 0.34 | |||||
| 395 | 188 | 12.04.2005 | 90 | 7.7 | 555 | 8.9 | −21.7 | 0.17 | 0.32 | 49.7 | 11.4 | 33.0 | 7.3 | 103.7 | 117.0 | 10.3 | 0.3 | bdl | 0.09 | 0.32 | |||||
| 40 | 188 | 08.10.2014 | 90 | 8.2 | 513 | 8.7 | −21.6 | −164.9 | 0.28 | 0.29 | 48.7 | 9.8 | 28.2 | 6.7 | 97.6 | 95.9 | 9.0 | bdl | bdl | 0.68 | 0.48 | ||||
| 411 | 296 | 26.10.1989 | 148 | 7.7 | 60.3 | 19.2 | 50.0 | 8.0 | 164.8 | 136.9 | 10.0 | 0.10 | 0.46 | bdl | |||||||||||
| 42 | 296 | 09.04.2007 | 148 | 8.0 | 0.45 | 0.25 | |||||||||||||||||||
| 43 | 296 | 14.09.2018 | 148 | 7.5 | 467 | −14.5 | −105.6 | 60.9 | 13.1 | 44.0 | 7.8 | 183.1 | 117.7 | bdl | 0.2 | 0.22 | |||||||||
| 442 | 300 | 14.06.1988 | 165 | 7.6 | −14.4 | −105.0 | 0.40 | 81.0 | 15.9 | 36.7 | 5.2 | 213.6 | 120.9 | 12.7 | bdl | 0.9 | |||||||||
| 46 | 300 | 16.02.2009 | 165 | 7.6 | 0.34 | 0.55 | 81.0 | 15.9 | 36.7 | 5.2 | 213.6 | 120.9 | 12.7 | bdl | 0.9 | ||||||||||
| 47 | 300 | 14.09.2018 | 160 | 7.4 | 496 | −12.9 | −94.0 | 76.6 | 21.4 | 30.0 | 5.4 | 262.4 | 100.7 | bdl | 0.3 | 0.37 | |||||||||
| 48 | 375 | 05.07.1994 | 160 | 7.8 | −22.5 | 85.6 | 20.1 | 108.6 | 11.0 | 189.2 | 280.1 | 5.9 | |||||||||||||
| 492 | 386 | 05.07.1988 | 150 | 7.7 | −22.7 | −176.0 | 0.59 | 104.1 | 22.3 | 78.5 | 10 | 109.8 | 360.5 | 16.9 | bdl | bdl | |||||||||
| 50 | 412 | 08.10.2014 | 156 | 349 | 9.2 | −12.2 | −89.4 | 0.25 | 0.09 | 74.3 | bdl | bdl | 0.50 | 0.35 | |||||||||||
| 51 | 424 | 08.10.2014 | 150 | 8.3 | 490 | 9.4 | −20.2 | −153.0 | 40.6 | 13.5 | 32.1 | 8.5 | 146.4 | 72.1 | 2.2 | bdl | 0.57 | 0.45 | 0.65 | ||||||
| 521 | 4460 | 26.10.1989 | 145 | 7.6 | 99.2 | 22.4 | 90.0 | 10.0 | 146.4 | 323.0 | 20.6 | 0.25 | 1.20 | bdl | |||||||||||
| 535 54 | 14798 23887 | 13.04.2005 09.09.2016 | 159.5 88 | 7.9 8.0 | 526 399 | 100 8.3 | −12.0 −20.4 | −153.1 | 0.29 | 0.40 | 69.7 120.0 | 11.3 19.0 | 35.8 110.0 | 7.7 11.0 | 256.2 130.8 | 48.2 360.0 | bdl 17.0 | 0.6 bdl | bdl bdl | 4.70 | 0.18 | 0.09 | 0.94 | 0.26 | 1.10 |
| 55 | 25686 | 09.09.2016 | 122 | 8.1 | 1391 | 9.0 | −19.2 | −142.1 | 0.52 | 0.57 | 120.0 | 21.0 | 110.0 | 12.0 | 141.7 | 360.0 | 17.0 | bdl | bdl | 4.60 | 0.19 | 0.84 | 1.20 | ||
| 568 | 25688 | 20.11.2012 | 118 | 7.9 | 1003 | 8.9 | −17.7 | 97.0 | 19.0 | 106.0 | 11.0 | 281.0 | 6.0 | bdl | |||||||||||
| 57 | 25688 | 09.09.2016 | 118 | 8.0 | 1380 | 9.2 | −17.8 | −130.4 | 0.47 | 0.54 | 120.0 | 20.0 | 110.0 | 12.0 | 174.4 | 350.0 | 17.0 | bdl | bdl | 4.80 | 0.18 | 0.94 | 1.20 | ||
| 58 | 25690 | 13.11.2013 | 122 | 8.2 | 577 | 8.5 | 0.69 | 0.44 | 92.0 | 12.1 | 70.4 | 7.9 | 150.0 | 222.1 | 2.4 | bdl | 2.48 | 0.09 | 0.16 | 0.55 | 0.01 | 0.30 | |||
| 598 | 25690 | 05.08.2014 | 122 | 7.9 | 1204 | 8.5 | −19.8 | −147.7 | 109.4 | 19.2 | 102.6 | 10.7 | 295.7 | 2.5 | 1.1 | bdl | 2.02 | bdl | 1.04 | 0.36 | |||||
| 60 | 25690 | 09.09.2016 | 122 | 8.1 | 1285 | 9.0 | −19.7 | −146.1 | 0.41 | 0.51 | 110.0 | 19.0 | 100.0 | 11.0 | 130.8 | 330.0 | 16.0 | bdl | bdl | 4.70 | 0.18 | 0.81 | bdl | ||
| 618 | 25692 | 08.10.2014 | 120 | 8.3 | 1381 | 8.6 | −17.4 | −131.9 | 0.53 | 0.68 | 126.7 | 19.3 | 103.5 | 11.2 | 170.8 | 350.0 | 2.8 | bdl | bdl | 2.18 | 0.39 | ||||
| 62 | 25692 | 09.09.2016 | 120 | 8.1 | 1558 | 9.2 | −17.6 | −130.1 | 0.64 | 0.74 | 140.0 | 21.0 | 120.0 | 13.0 | 152.6 | 410.0 | 17.0 | bdl | bdl | 5.70 | 0.21 | 1.00 | 1.20 | ||
| Voronka + Gdov aquifers | |||||||||||||||||||||||||
| 631 | 153 | 26.06.1990 | 8.1 | 101.3 | 20.9 | 79.3 | 8.4 | 231.9 | 96.8 | 59.3 | 0.05 | bdl | 0.20 | ||||||||||||
| 641 | 156 | 26.06.1990 | 110 | 7.4 | 101.6 | 21.0 | 79.2 | 21.0 | 427.1 | 34.7 | 1.5 | 0.03 | bdl | 0.20 | |||||||||||
| 65 | 157 | 09.06.2014 | 120 | 8.0 | 1274 | 9.3 | −21.2 | −159.9 | 0.59 | 0.60 | 136.6 | 20.5 | 99.5 | 9.9 | 204.8 | 5.3 | 1.2 | bdl | 0.87 | 1.11 | bdl | ||||
| 661 | 162 | 01.08.1990 | 140 | −16.3 | −111.0 | 0.32 | |||||||||||||||||||
| 675 | 162 | 13.04.2005 | 140 | 7.9 | 856 | 8.7 | −17.4 | 0.52 | 0.16 | 87.4 | 16.0 | 58.6 | 9.0 | 183.0 | 189.3 | bdl | 0.5 | bdl | 0.10 | 0.26 | |||||
| 681 | 163 | 30.07.1991 | 138 | −14.7 | 0.31 | 100.1 | 20.3 | 79.7 | |||||||||||||||||
| 692 | 163 | 01.06.1990 | 138 | −15.1 | −102.0 | ||||||||||||||||||||
| 701 | 169 | 26.06.1990 | 7.6 | 102.8 | 21.7 | 78.8 | 1.0 | 311.2 | 12.8 | 49.4 | 0.03 | bdl | 17.40 | ||||||||||||
| 712 | 298 | 14.06.1988 | 145 | 7.3 | −22.0 | −164.0 | 0.58 | 103.1 | 21.8 | 78.8 | 9.6 | 134.2 | 293.6 | 10.0 | bdl | 0.7 | |||||||||
| 721 | 299 | 26.10.1989 | 144 | 7.6 | 103.3 | 21.9 | 78.7 | 10.0 | 146.4 | 291.4 | 22.6 | 0.20 | 1.80 | bdl | |||||||||||
| 735 | 477 | 12.04.2005 | 90 | 7.7 | 1380 | 9.2 | −22.7 | 0.65 | 1.18 | 108.8 | 20.4 | 130.0 | 12.9 | 158.6 | 350.6 | 26.5 | bdl | bdl | 0.14 | 0.62 | |||||
| 741 | 485 | 26.10.1989 | 145 | 8 | 104.3 | 22.4 | 78.4 | 4.0 | 238.0 | 39.0 | 0.0 | 0.22 | 0.56 | bdl | |||||||||||
| 75 | 485 | 14.09.2018 | 145 | 7.6 | 285 | −11.7 | −84.1 | 50.1 | 12.5 | 14.2 | 4.6 | 250.2 | 8.5 | bdl | 0.4 | 0.20 | |||||||||
| 762 | 115 | 07.07.1991 | 120 | 8.3 | −21.9 | 159.5 | 15.3 | 116.0 | 8.0 | 73.2 | 453.4 | 21.4 | 0.08 | ||||||||||||
| 773 | 1152 | 05.07.1994 | 122 | 7.9 | −21.4 | 150.7 | 14.7 | 122.0 | 8.3 | 85.4 | 455.6 | 18.9 | |||||||||||||
| 784 | 1152 | 28.09.1995 | 122 | −21.3 | |||||||||||||||||||||
| 79 804 | 1152 4734 | 28.10.2014 12.08.1996 | 122 100 | −21.5 −22.0 | −161.8 | 146.9 63.1 | 13.1 21.3 | 150.0 43.4 | 7.3 7.3 | 85.4 146.4 | 482.2 185.8 | 7.0 6.6 | |||||||||||||
| 817 | 4734 | 27.03.2007 | 100 | 8.0 | 728 | 9.3 | −22.0 | ||||||||||||||||||
| 82 | 4734 | 02.09.2016 | 100 | 7.9 | 1069 | 9.4 | −22.5 | −168.4 | 82.0 | 17.0 | 95.0 | 11.0 | 152.6 | 240.0 | 19.0 | bdl | bdl | 3.40 | 0.19 | 0.55 | 1.00 | ||||
| 835 | 5157 | 12.04.2005 | 125 | 7.8 | 445 | 9.1 | −12.5 | 0.43 | 0.23 | 52.5 | 15.9 | 20.0 | 9.3 | 280.7 | 22.3 | bdl | 0.3 | bdl | 0.08 | 0.29 | |||||
| 84 | 11569 | 01.07.2016 | 100 | 8.0 | 409 | 8.5 | −21.5 | −161.7 | 61.0 | 9.8 | 35.0 | 6.3 | 150.0 | 15.0 | 0.6 | 3.3 | 1.40 | bdl | 0.24 | 0.34 | |||||
| 85 | 15831 | 13.11.2013 | 123.3 | 8.3 | 527 | 8.2 | −22.6 | −169.7 | 0.77 | 0.52 | 71.8 | 10.7 | 63.3 | 7.5 | 149.5 | 176.9 | 17.2 | bdl | 0.75 | 0.18 | 0.09 | 0.46 | 0.01 | 0.28 | |
| 86 | 15841 | 14.04.2005 | 117 | 7.9 | 933 | 9.0 | −22.7 | 0.55 | 0.67 | 64.7 | 16.5 | 82.7 | 10.0 | 170.8 | 199.6 | bdl | 0.2 | bdl | 0.08 | 0.25 | |||||
| 87 | 16557 | 09.06.2014 | 110 | 8.1 | 393 | 9.2 | −21.6 | −163.4 | 0.12 | 0.10 | 45.1 | 8.6 | 24.6 | 6.7 | 59.8 | 10.2 | bdl | bdl | bdl | 0.08 | 0.36 | 0.54 | |||
| 88 | 16557 | 28.10.2014 | 110 | −21.6 | −162.2 | ||||||||||||||||||||
| 89 | 485 | 14.09.2018 | 145 | 7.6 | 285 | −11.7 | −84.1 | 0.18 | 0.30 | 50.1 | 12.5 | 14.2 | 4.6 | 250.2 | 8.5 | bdl | 0.4 | 0.20 | |||||||
| Crystalline basement aquifer | |||||||||||||||||||||||||
| 902 | 1151 | 06.09.1994 | 160 | 7.7 | −21.2 | 206.0 | 32.6 | 211.0 | 12.5 | 79.3 | 735.4 | 27.6 | |||||||||||||
| 915 | 1151 | 25.04.2001 | 160 | 8.5 | 1540 | 8.8 | −21.3 | 188.0 | 25.2 | 210.0 | 11.7 | 64.1 | 629.0 | 14.8 | |||||||||||
| No | Well ID | SI-calcite | SI-dolomte | SI-dolomite (disordered) | SI-CO2(g) |
|---|---|---|---|---|---|
| 2 | 124 | 0.05 | −0.32 | −0.93 | −2.94 |
| 5 | 171 | 0.41 | 0.43 | −0.18 | −3.07 |
| 6 | 179 | −0.11 | −0.45 | −1.07 | −2.65 |
| 9 | 187 | −0.30 | −0.90 | −1.51 | −2.72 |
| 10 | 187 | −0.09 | −0.82 | −1.44 | −2.90 |
| 15 | 1153 | −0.29 | −1.00 | −1.61 | −2.76 |
| 18 | 8914 | 0.17 | 0.04 | −0.58 | −2.97 |
| 21 | 19570 | 0.37 | 0.21 | −0.40 | −2.82 |
| 23 | 25689 | −0.03 | −0.64 | −1.25 | −3.10 |
| 29 | 154 | 0.28 | −0.12 | −0.73 | −2.81 |
| 34 | 161 | 0.49 | 0.30 | −0.32 | −2.66 |
| 36 | 172 | 0.29 | 0.02 | −0.59 | −2.86 |
| 38 | 183 | 0.25 | −0.10 | −0.72 | −2.97 |
| 39 | 188 | −0.27 | −1.06 | −1.68 | −2.80 |
| 40 | 188 | 0.21 | −0.15 | −0.77 | −3.32 |
| 46 | 300 | 0.11 | −0.36 | −0.97 | −2.40 |
| 53 | 14798 | 0.46 | 0.24 | −0.37 | −2.61 |
| 55 | 25686 | 0.57 | 0.50 | −0.11 | −3.08 |
| 57 | 25688 | 0.55 | 0.45 | −0.16 | −2.89 |
| 58 | 25690 | 0.62 | 0.48 | −0.14 | −3.14 |
| 60 | 25690 | 0.50 | 0.37 | −0.24 | −3.11 |
| 61 | 25692 | 0.87 | 1.05 | 0.43 | −3.20 |
| 62 | 25692 | 0.65 | 0.60 | −0.01 | −3.05 |
| 67 | 162 | 0.38 | 0.15 | −0.47 | −2.76 |
| 73 | 477 | 0.16 | −0.28 | −0.89 | −2.64 |
| 83 | 5157 | 0.28 | 0.16 | −0.45 | −2.47 |
| 85 | 15831 | 0.62 | 0.53 | −0.08 | −3.24 |
| 86 | 15841 | 0.23 | −0.02 | −0.63 | −2.79 |
| 89 | 485 | 0.02 | −0.45 | −1.06 | −2.32 |
| No | Well ID | Date | Aquifer | Well Depth | He | He (Error) | Ne | Ne Error | Ar | Ar (Error) | Kr | Kr (Error) | Xe | Xe (Error) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m | cm3·STP·g−1 | |||||||||||||
| 10 | 187 | 13-Nov-13 | Cm-V | 70 | 1.46·10−5 | 1.09·10−7 | 3.77·10−7 | 8.28·10−10 | 7.53·10−4 | 2.05·10−6 | 1.57·10−7 | 1.05·10−9 | 2.12·10−8 | 1.97·10−10 |
| 85 | 15831 | 13-Nov-13 | Cm-V | 123.3 | 2.50·10−5 | 1.88·10−7 | 3.81·10−7 | 8.39·10−10 | 1.27·10−3 | 3.41·10−6 | 2.18·10−7 | 1.16·10−9 | 2.34·10−8 | 2.15·10−10 |
| 84 | 11569 | 1-Jul-16 | Cm-V | 100 | 7.09·10−5 | 7.09·10−7 | 5.95·10−7 | 4.90·10−9 | 8.93·10−4 | 7.14·10−6 | 1.86·10−7 | 2.40·10−9 | 2.24·10−8 | 4.06·10−10 |
| 19 | 8914 | 2-Sep-16 | V2vr | 75 | 3.66·10−5 | 3.66·10−7 | 5.79·10−7 | 4.90·10−9 | 1.12·10−3 | 8.97·10−6 | 1.92·10−7 | 2.21·10−9 | 2.31·10−8 | 4.42·10−10 |
| 82 | 4734 | 2-Sep-16 | Cm-V | 100 | 1.68·10−5 | 1.69·10−7 | 1.77·10−7 | 1.50·10−9 | 6.67·10−4 | 5.34·10−6 | 1.27·10−7 | 1.38·10−9 | 1.57·10−8 | 3.13·10−10 |
| 26 | 25687 | 9-Sep-16 | V2vr | 82 | 7.88·10−5 | 7.88·10−7 | 8.85·10−7 | 7.32·10−9 | 1.18·10−3 | 9.46·10−6 | 2.04·10−7 | 2.28·10−9 | 2.38·10−8 | 4.34·10−10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raidla, V.; Pärn, J.; Aeschbach, W.; Czuppon, G.; Ivask, J.; Kiisk, M.; Mokrik, R.; Samalavičius, V.; Suursoo, S.; Tarros, S.; et al. Intrusion of Saline Water into a Coastal Aquifer Containing Palaeogroundwater in the Viimsi Peninsula in Estonia. Geosciences 2019, 9, 47. https://doi.org/10.3390/geosciences9010047
Raidla V, Pärn J, Aeschbach W, Czuppon G, Ivask J, Kiisk M, Mokrik R, Samalavičius V, Suursoo S, Tarros S, et al. Intrusion of Saline Water into a Coastal Aquifer Containing Palaeogroundwater in the Viimsi Peninsula in Estonia. Geosciences. 2019; 9(1):47. https://doi.org/10.3390/geosciences9010047
Chicago/Turabian StyleRaidla, Valle, Joonas Pärn, Werner Aeschbach, György Czuppon, Jüri Ivask, Madis Kiisk, Robert Mokrik, Vytautas Samalavičius, Siiri Suursoo, Siim Tarros, and et al. 2019. "Intrusion of Saline Water into a Coastal Aquifer Containing Palaeogroundwater in the Viimsi Peninsula in Estonia" Geosciences 9, no. 1: 47. https://doi.org/10.3390/geosciences9010047
APA StyleRaidla, V., Pärn, J., Aeschbach, W., Czuppon, G., Ivask, J., Kiisk, M., Mokrik, R., Samalavičius, V., Suursoo, S., Tarros, S., & Weissbach, T. (2019). Intrusion of Saline Water into a Coastal Aquifer Containing Palaeogroundwater in the Viimsi Peninsula in Estonia. Geosciences, 9(1), 47. https://doi.org/10.3390/geosciences9010047