Influence of Pasture on Stearoyl-CoA Desaturase and miRNA 103 Expression in Goat Milk: Preliminary Results
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Diets and Management
2.2. Milk Analysis
2.3. Statistical Analysis
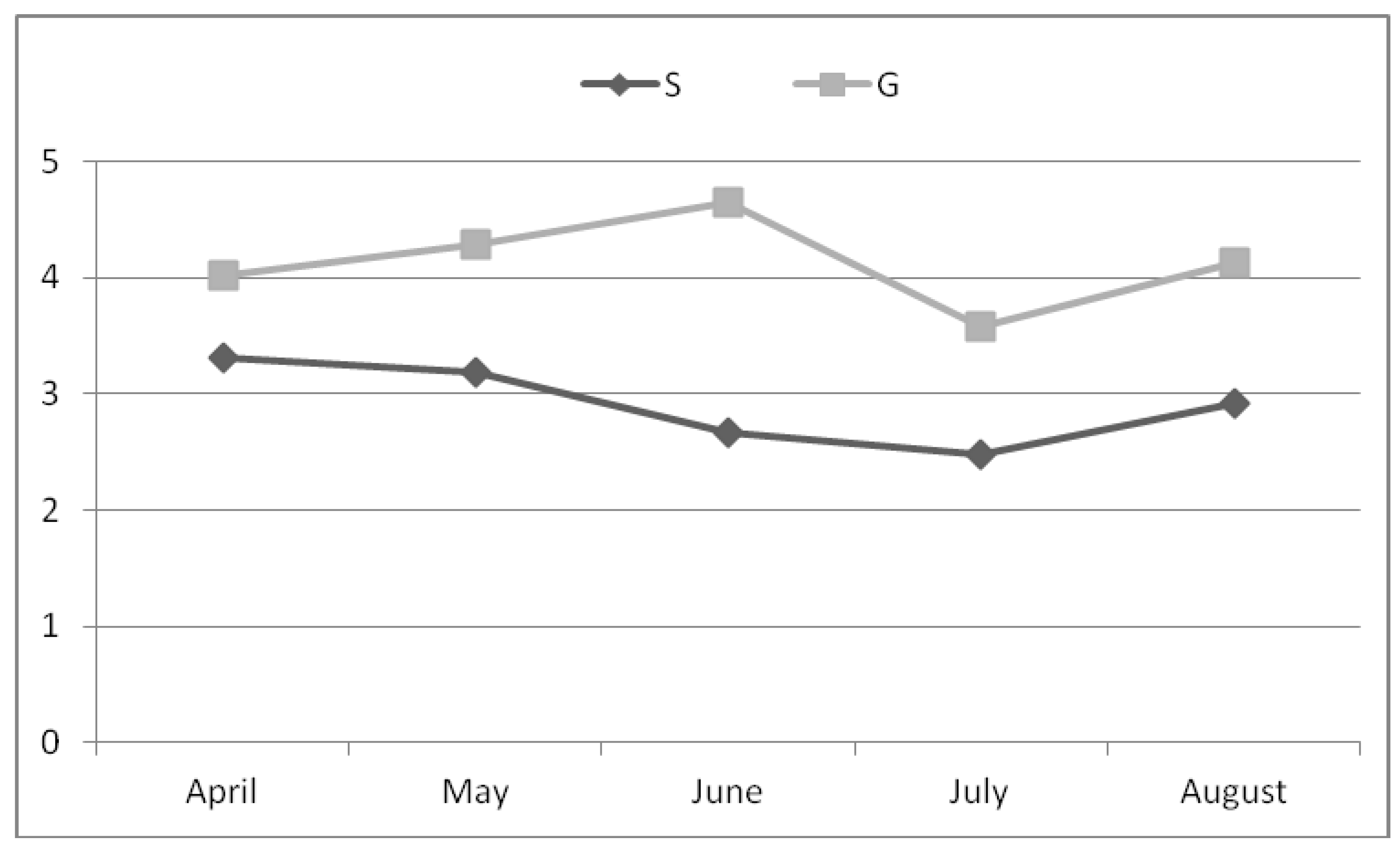
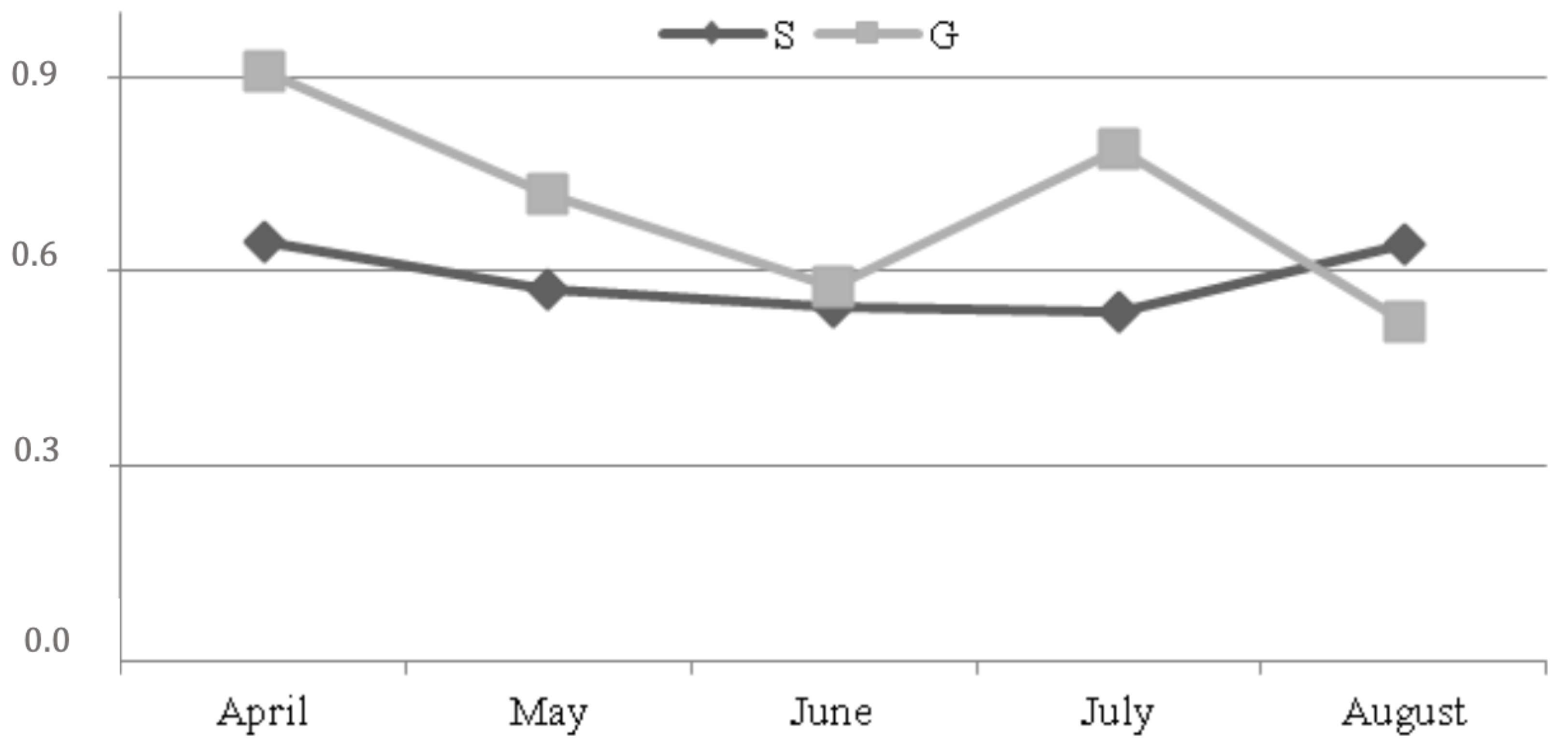
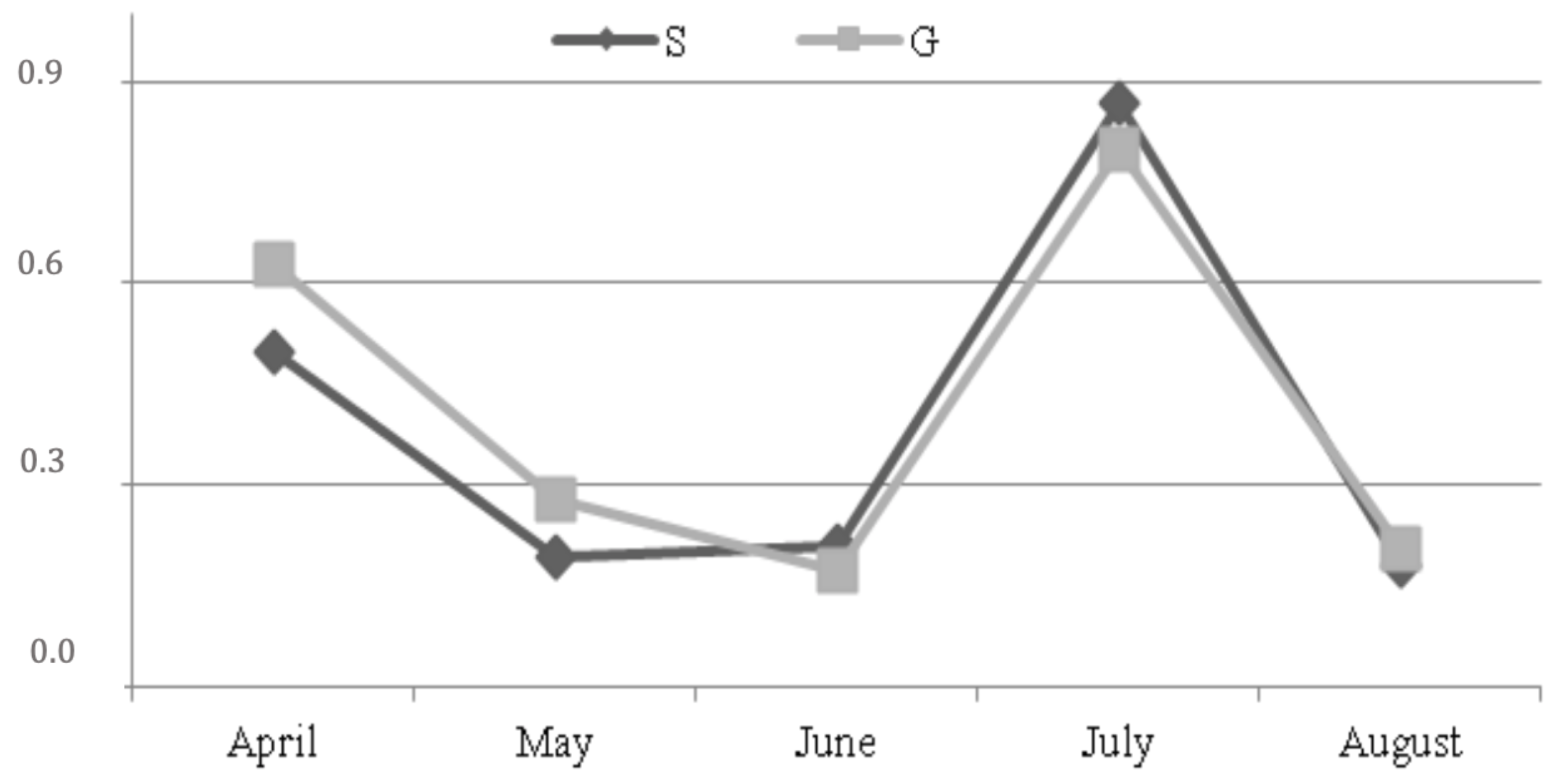
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chilliard, Y.; Ferlay, A.; Mansbridge, R.M.; Doreau, M. Ruminant milk fat plasticity: Nutritional control of saturated, polyunsaturated, trans and conjugated fatty acids. Ann. Zootech. 2000, 49, 181–205. [Google Scholar] [CrossRef]
- Pastuschenko, V.; Matthes, H.D.; Hein, T.; Holzer, Z. Impact of cattle grazing on meat fatty acid composition in relation to human nutrition. In Proceedings of the 13th International IFOAM Scientific Conference, Basel, Switzerland, 28–31 August 2000; Alföldi, T., Lockeretz, W., Niggli, U., Eds.; pp. 293–296. [Google Scholar]
- Whigham, L.; Cook, M.E.; Atkinson, R.L. Conjugated linoleic acid: Implications for human health. Pharmacol. Res. 2000, 42, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, T.A.; Lippolis, J.D.; Nonnecke, B.J.; Sacco, R.E. Bovine milk exosome proteome. J. Proteom. 2012, 75, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- Izumi, H.; Kosaka, N.; Shimizu, T.; Sekine, K.; Ochiya, T.; Takase, M. Bovine milk contains microRNA and messenger RNA that are stable under degradative conditions. J. Dairy Sci. 2012, 95, 4831–4841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, Y.; Li, M.; Wang, T.; Liang, Y.; Zhong, Z.; Wang, X.; Zhou, Q.; Chen, L.; Lang, Q.; He, Z.; et al. Lactation-Related MicroRNA Expression Profiles of Porcine Breast Milk Exosomes. PLoS ONE 2012, 7, e43691. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Luo, J.; Zhang, L.; Zhu, J. MicroRNAs Synergistically Regulate Milk Fat Synthesis in Mammary Gland Epithelial Cells of Dairy Goats. Gene Expr. 2013, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Luo, J.; Zhang, L.; Wang, W.; Gou, D. MiR-103 Controls Milk Fat Accumulation in Goat (Capra hircus) Mammary Gland during Lactation. PLoS ONE 2013, 8, e79258. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.-Z.; Luo, J.; Zhang, L.P.; Wang, W.; Shi, H.B.; Zhu, J.J. miR-27a suppresses triglyceride accumulation and affects gene mRNA expression associated with fat metabolism in dairy goat mammary gland epithelial cells. Gene 2013, 521, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Romao, J.M.; Jin, W.; He, M.; McAllister, T.; Guan, L.L. Altered MicroRNA Expression in Bovine Subcutaneous and Visceral Adipose Tissues from Cattle under Different Diet. PLoS ONE 2012, 7, e40605. [Google Scholar] [CrossRef]
- Circulating profiling reveals the effect of a polyunsaturated fatty acid-enriched diet on common microRNAs. J. Nutr. Biochem. 2015, 26, 1095–1101. [CrossRef]
- Davidson, L.A.; Wang, N.; Shah, M.S.; Lupton, J.R.; Ivanov, I.; Chapkin, R.S. n-3 Polyunsaturated fatty acids modulate carcinogen-directed non-coding microRNA signatures in rat colon. Carcinogenesis 2009, 30, 2077–2084. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Ge, Y.; Zhang, J.; Xue, M.; Li, Q.; Lin, D.; Ma, W. PUFA diets alter the microRNA expression profiles in an inflammation rat model. Mol. Med. Rep. 2015, 11, 4149–4157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Urso, S.; Cutrignelli, M.I.; Calabrò, S.; Bovera, F.; Tudisco, R.; Piccolo, V.; Infascelli, F. Influence of pasture on fatty acid profile of goat milk. J. Anim. Physiol. Anim. Nutr. 2008, 92, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Tudisco, R.; Calabrò, S.; Cutrignelli, M.I.; Moniello, G.; Grossi, M.; Gonzalez, O.J.; Piccolo, V.; Infascelli, F. Influence of organic systems on Stearoyl-CoA desaturase in goat milk. Small Rumin. Res. 2012, 106, 37–42. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3598. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 19th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 2012. [Google Scholar]
- INRA. Alimentation Des Ruminants; INRA: Paris, France, 1978. [Google Scholar]
- Folch, J.; Lees, M.; Sloane, G.H. A simple method for isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Christie, W.W. A simple procedure of rapid transmethylation of glycerolipids and cholesteryl esters. J. Lipid Res. 1982, 23, 1072–1075. [Google Scholar] [PubMed]
- Chouinard, P.Y.; Corneau, L.; Barbano, D.M.; Metzger, L.E.; Bauman, D.E. Conjugated linoleic acids alter milk fatty acid compo-sition and inhibit milk fat secretion in dairy cows. J. Nutr. 1999, 129, 1579–1584. [Google Scholar] [CrossRef]
- Tudisco, R.; Cutrignelli, M.I.; Calabrò, S.; Piccolo, G.; Bovera, F.; Guglielmelli, A.; Infascelli, F. Influence of organic systems on milk fatty acid profile and CLA in goats. Small Rumin. Res. 2010, 88, 151–155. [Google Scholar] [CrossRef]
- Hara, A.; Radin, N.S. Lipid extraction of tissues with a low-toxicity solvent. Anal. Biochem. 1978, 90, 420–426. [Google Scholar] [CrossRef] [Green Version]
- Lock, A.L.; Garnsworthy, P.C. Seasonal variation in milk conjugated linoleic acid and 9-desaturase activity in dairy cows. Livest. Prod. Sci. 2003, 79, 47–59. [Google Scholar] [CrossRef]
- Tudisco, R.; Chiofalo, B.; Addi, L.; Lo Presti, V.; Rao, R.; Calabro’, S.; Musco, N.; Grossi, M.; Cutrignelli, M.I.; Mastellone, V.; et al. Effect of hydrogenated palm oil dietary supplementation on milk yield and composition, fatty acids profile and Stearoyl-CoA desaturase expression in goat milk. Small Rumin. Res. 2015, 132, 72–78. [Google Scholar] [CrossRef]
- SAS. SAS/STAT Software: Changes and Enhancements Through Release 8.1, SAS Institute Inc.: Cary, NC, USA, 2000.
- Loewenstein, M.; Frank, J.L.; Speck, S.J. Goat cheese. In Extension Goat Handbook; USDA: Washington, DC, USA, 1984; p. 10. [Google Scholar]
- Schroeder, A.; Mueller, O.; Stocker, S.; Salowsky, R.; Leiber, M.; Gassmann, M.; Lightfoot, S.; Menzel, W.; Granzow, M.; Ragg, T. The RIN: An RNA integrity number for assigning integrity values to RNA measurements. BMC Mol. Biol. 2006, 7, 3–12. [Google Scholar] [CrossRef]
- Rubino, R. L’allevamento Caprino; ASSONAPA: Roma, Italy, 1996. [Google Scholar]
- Schroeder, G.F.; Gagliostro, G.A.; Bargo, F.; Delahoy, L.D. MullerEffect of fat supplementation on milk production and composition by dairy cows on pasture: A review. Livest. Prod. Sci. 2004, 86, 1–18. [Google Scholar] [CrossRef]
- Dhiman, T.R.; Satter, L.D.; Shaver, R.D. Milk Production and Blood Phosphorus Concentrations of Cows Fed Low and High Dietary Phosphorus; U.S. Dairy Forage Research Center 1995 Res. Summaries: Madison, WI, USA, 2006; p. 105. [Google Scholar]
- Nudda, A.; Mele, M.; Battacone, G.; Usai, M.G.; Macciotta, N.P.P. Comparison of conjugated linoleic acid (CLA) content in milk of ewes and goats with the same dietary regimen. Ital. J. Anim. Sci. 2003, 2, 515–517. [Google Scholar]
- Tudisco, R.; Grossi, M.; Calabrò, S.; Cutrignelli, M.I.; Musco, N.; Addi, A.; Infascelli, F. Influence of pasture on goat milk fatty acids and Stearoyl-CoA desaturase expression in milk somatic cells. Small Rumin. Res. 2014, 122, 38–43. [Google Scholar] [CrossRef]
- Zicarelli, F.; Addi, L.; Tudisco, R.; Calabrò, S.; Lombardi, P.; Cutrignelli, M.I.; Moniello, G.; Grossi, M.; Tozzi, B.; Musco, N.; et al. The influence of diet supplementation with Saccharomyces cerevisiae or Saccharomyces cerevisiae plus Aspergillus oryzae on milk yield of Cilentana grazing dairy goats. Small Rumin. Res. 2016, 135, 90–94. [Google Scholar] [CrossRef]
- Bergamo, P.; Fedele, E.; Iannibelli, L.; Marzillo, G. Fat-soluble vitamin contents and fatty acid composition in organic and conventional Italian dairy products. Food Chem. 2003, 82, 625–631. [Google Scholar] [CrossRef]
- Secchiari, P.; Campanile, G.; Mele, M. Fatty acid composition and CLA content of milk fat from Italian buffalo. In Indicators of Milk and Beff Quality; Hocquette, Gili, Ed.; EAAP Scientific Series; Wageningen Academic Publishers: Wageningen, Holland, The Netherlands, 2005; Volume 112, pp. 339–343. [Google Scholar]
- Jahreis, G.; Fritsche, J.; Möckel, P.; Schöne, F.; Möller, U.; Steinhart, H. The potential anticarcinogenic conjugated linoleic acid, cis-9, trans-11 C18:2, in milk of different species: Cow, goat, ewe, sow, mare, woman. Nutr. Res. 1999, 19, 1541–1549. [Google Scholar] [CrossRef]
- Tsiplakou, E.; Mountzouris, K.C.; Zervas, G. Concentration of conjugated linoleic acid in grazing sheep and goat milk fat. Livest. Sci. 2006, 103, 74–84. [Google Scholar] [CrossRef]
- Kemp, P.; Lander, D.J. Hydrogenation in vitro of α-linolenic acid to stearic acid by mixed cultures of pure strains of rumen bacteria. J. Gen. Microbiol. 1984, 130, 527–533. [Google Scholar] [CrossRef]
- Kim, Y.J.; Liu, R.H.; Bond, D.R.; Russell, J.B. Effect of linoleic acid concentration on conjugated linoleic acid by Butyrivibrio fibrisolvens A38. Appl. Environ. Microbiol. 2000, 66, 5226–5230. [Google Scholar] [CrossRef]
- Bernard, L.; Rouel, J.; Leroux, C.; Ferlay, A.; Faulconnier, Y.; Legrand, P.; Chilliard, Y. Mammary lipid metabolism and milk fatty acid secretion in alpine goats fed vegetable lipids. J. Dairy Sci. 2005, 88, 1478–1489. [Google Scholar] [CrossRef]
- Bernard, L.; Bonnet, M.; Leroux, C.; Shingfield, K.J.; Chilliard, Y. Effect of sunflower-seed oil and linseed oil on tissue lipid metabolism, gene expression, and milk fatty acid secretion in alpine goats fed maizesilage-based diets. J. Dairy Sci. 2009, 92, 6083–6094. [Google Scholar] [CrossRef]
- Bernard, L.; Leroux, C.; Faulconnier, Y.; Durand, D.; Shingfield, K.J.; Chilliard, Y. Effect of sunflower-seed oil or linseed oil on milk fatty acid secretion and lipogenic gene expression in goats fed hay-based diets. J. Dairy Sci. 2009, 76, 241–248. [Google Scholar] [CrossRef]
- Bernard, L.; Leroux, C.; Bonnet, M.; Rouel, J.; Martin, P.; Chilliard, Y. Expression and nutritional regulation of lipogenic genes in mammary gland and adipose of lactating goats. J. Dairy Sci. 2005, 72, 250–255. [Google Scholar] [CrossRef]
- Bernard, L.; Leroux, C.; Chilliard, Y. Expression and nutritional regulation of Stearoyl-CoA desaturase genes in the ruminant mammary gland: Relationship with milk fatty acid composition. In Stearoyl-CoA Desaturase Genes in Lipid Metabolism; Ntambi, J.M., Ed.; SpringerScience+Business Media: New York, NY, USA, 2013; pp. 161–193. [Google Scholar]
- Avril-Sassen, S.; Goldstein, L.D.; Stingl, J.; Blenkiron, C.; Quesne, J.L.; Spiteri, I.; Karagavriilidou, K.; Watson, C.J.; Tavaré, S.; Miska, E.A.; et al. Characterisation of microRNA expression in post-natal mouse mammary gland development. BMC Genom. 2009, 10, 548–563. [Google Scholar] [CrossRef]
- Chen, X.; Gao, C.; Li, H.; Huang, L.; Sun, Q.; Dong, Y.; Tian, C.; Gao, S.; Dong, H.; Guan, D.; et al. Identification and characterization of microRNAs in raw milk during different periods of lactation, commercial fluid, and powdered milk products. Cell Res. 2010, 20, 1128–1137. [Google Scholar] [CrossRef]
- Wang, M.; Moisá, S.; Khan, M.J.; Wang, J.; Bu, D.; Loor, J.J. MicroRNA expression patterns in the bovine mammary gland are affected by stage of lactation. J. Dairy Sci. 2012, 95, 6529–6535. [Google Scholar] [CrossRef] [Green Version]
- Dong, F.; Ji, Z.B.; Chen, C.X.; Wang, G.Z.; Wang, J.M. Target gene and function prediction of differentially expressed microRNAs in lactating mammary glands of dairy goats. Int. J. Genom. 2013, 917342. [Google Scholar] [CrossRef]
- Hansen, J.; Lacis, A.; Rind, D.; Russell, G.; Stone, P.; Fung, I.; Ruedy, R.; Lerner, J. Climate Sensitivity: Analysis of Feedback Mechanisms; Geophy Mono. Series 29; American Geophysical Union: Washington, DC, USA, 1984; pp. 130–163. [Google Scholar]
- Bionaz, M.; Loor, J.J. Gene networks driving bovine mammary protein synthesis during the lactation cycle. Bioinform. Biol. Insights 2011, 5, 83–89. [Google Scholar] [CrossRef]
- Mura, M.C.; Daga, C.; Bodano, S.; Paludo, M.; Luridiana, S.; Pazzola, M.; Det-tori, M.L.; Vacca, G.M.; Carcangiu, V. Development of a RNA extraction method from milk for gene expression study in the mammary gland of sheep. Mol. Biol. Reprod. 2013, 40, 2169–2173. [Google Scholar] [CrossRef]
| Chemical Composition (g/kg DM) | Hay | Concentrate * | Pasture |
|---|---|---|---|
| Crude protein | 164.2 | 180.0 | 167.0 |
| Ether extract | 16.3 | 30.0 | 19.0 |
| NDF | 434.1 | 270.0 | 495.0 |
| ADF | 311.4 | 115.0 | 345.0 |
| ADL | 54.3 | 30.0 | 50.0 |
| UFL (kg DM) | 0.75 | 1.03 | 0.76 |
| Fatty acid profile (% of total FA) | |||
| SFA | 23.0 | 24.6 | 17.6 |
| MUFA | 8.4 | 16.0 | 6.4 |
| PUFA | 68.6 | 59.4 | 76.0 |
| C18:2 | 18.1 | 46.0 | 26.0 |
| C18:3 | 38.8 | 9.3 | 42.6 |
| Milk | Yield | Fat | Protein | Lactose | ||||
|---|---|---|---|---|---|---|---|---|
| Group | S | G | S | G | S | G | S | G |
| 1798.1 | 1793.0 | 2.94 | 4.10 | 3.25 | 3.43 | 4.02 | 3.99 | |
| Group effect | NS | ** | ** | NS | ||||
| Sampling effect | ** | ** | NS | ** | ||||
| G × S | NS | NS | NS | NS | ||||
| SEM | 167.5 | 0.536 | 0.336 | 0.220 | ||||
| Milk Fatty Acids Profile | S | G | Group Effect | Sampling Effect | G × S | SEM |
|---|---|---|---|---|---|---|
| C4:0 | 0.0002 | 0.0002 | NS | NS | NS | 0.001 |
| C6:0 | 0.039 | 0.02 | NS | ** | NS | 0.073 |
| C8:0 | 0.949 | 0.883 | * | ** | * | 0.577 |
| C10:0 | 8.96 | 10.08 | ** | ** | * | 2.046 |
| C12:0 | 5.41 | 3.99 | ** | * | ** | 0.901 |
| C14:0 | 10.56 | 13.00 | ** | ** | NS | 1.025 |
| C14:1 cis9 | 0.199 | 0.119 | NS | ** | * | 0.111 |
| C16:0 | 34.43 | 30.22 | ** | ** | ** | 2.350 |
| C16:1 | 0.82 | 0.50 | ** | ** | ** | 0.190 |
| C17:0 | 0.742 | 0.761 | NS | NS | NS | 0.236 |
| C17:1 | 0.233 | 0.213 | NS | ** | NS | 0.093 |
| C18:0 | 13.39 | 12.01 | ** | ** | ** | 1.606 |
| Total C18:1 trans | 0.678 | 2.007 | ** | NS | ** | 0.374 |
| Total C18:1 cis | 19.68 | 21.48 | NS | NS | NS | 2.344 |
| C18:2 omega-6 | 2.048 | 1.906 | NS | ** | NS | 0.374 |
| C18:3 omega-3 | 0.797 | 1.351 | ** | * | ** | 0.251 |
| C20:0 | 0.187 | 0.213 | NS | NS | * | 0.11 |
| C20:1 | 0.126 | 0.043 | NS | NS | NS | 0.238 |
| C21:0 | 0.046 | 0.054 | NS | NS | ** | 0.021 |
| C20:2 | 0.008 | 0.014 | ** | * | NS | 0.011 |
| C22:0 | 0.076 | 0.108 | * | ** | ** | 0.060 |
| C20:3 omega-6 | 0.018 | 0.051 | * | NS | NS | 0.048 |
| C22:1 | 0.016 | 0.020 | NS | * | NS | 0.011 |
| C20:3 omega-3 | 0.032 | 0.050 | NS | NS | NS | 0.056 |
| C20:4 omega-6 | 0.079 | 0.072 | NS | NS | NS | 0.064 |
| C23:0 | 0.035 | 0.026 | NS | ** | * | 0.036 |
| C22:2 | 0.022 | 0.018 | NS | ** | NS | 0.016 |
| C24:0 | 0.036 | 0.056 | ** | * | ** | 0.025 |
| C20:5 omega-3 | 0.040 | 0.053 | ** | NS | NS | 0.022 |
| C24:1 | 0.006 | 0.009 | NS | ** | NS | 0.011 |
| C22:6 omega-3 | 0.024 | 0.031 | NS | ** | * | 0.018 |
| cis-9 trans-11 CLA | 0.304 | 0.623 | ** | ** | * | 0.145 |
| trans-10 cis-12 CLA | 0.008 | 0.023 | ** | NS | NS | 0.018 |
| SFA | 74.860 | 71.420 | ** | ** | ** | 3.008 |
| MUFA | 21.760 | 24.388 | ** | ** | ** | 2.64 |
| PUFA | 3.380 | 4.192 | ** | NS | * | 1.008 |
| ∑ CLA | 0.312 | 0.646 | ** | NS | ** | 0.153 |
| ∑ omega-3 | 0.893 | 1.485 | ** | * | ** | 0.160 |
| ∑ omega-6 | 2.145 | 2.029 | NS | ** | * | 1.032 |
| C14:1 cis9/C14:0 | 0.018 | 0.009 | * | * | NS | <0.01 |
| Group | April | May | June | July | August | Group Effect | Sampling Effect | G × S | SEM | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S | G | S | G | S | G | S | G | S | G | |||||
| C14:0 | 10.7 | 11.1 | 10.2 | 11.7 | 10.3 | 14.5 | 11.1 | 14.5 | 10.5 | 13.3 | ** | ** | NS | 1.025 |
| C14:1 | 0.18 | 0.07 | 0.12 | 0.17 | 0.33 | 0.21 | 0.16 | 0.08 | 0.21 | 0.07 | NS | ** | * | 0.111 |
| C18:2 | 2.35 | 2.52 | 2.29 | 2.64 | 2.04 | 1.03 | 1.68 | 1.67 | 1.88 | 1.67 | NS | ** | NS | 0.374 |
| C18:3 | 1.16 | 1.09 | 0.89 | 1.30 | 0.58 | 1.24 | 0.74 | 1.68 | 0.61 | 1.45 | ** | * | ** | 0.251 |
| c9 t11 CLA | 0.48 | 0.61 | 0.30 | 0.55 | 0.29 | 0.52 | 0.21 | 0.68 | 0.24 | 0.75 | ** | ** | * | 0.145 |
| C14:1/C14 | 0.015 | 0.006 | 0.010 | 0.016 | 0.022 | 0.020 | 0.011 | 0.007 | 0.015 | 0.006 | * | * | NS | < 0.01 |
| Pasture Fatty Acid Profile | April | May | June | July | August |
|---|---|---|---|---|---|
| SFA | 22.8 | 16.2 | 16.2 | 20.9 | 11.8 |
| MUFA | 8.9 | 5.0 | 5.0 | 5.9 | 7.4 |
| PUFA | 68.3 | 78.8 | 78.8 | 73.2 | 80.8 |
| C18:2 | 20.0 | 28.2 | 34.2 | 19.2 | 28.2 |
| C18.3 | 32.6 | 42.6 | 44.4 | 40.6 | 48.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tudisco, R.; Morittu, V.M.; Addi, L.; Moniello, G.; Grossi, M.; Musco, N.; Grazioli, R.; Mastellone, V.; Pero, M.E.; Lombardi, P.; et al. Influence of Pasture on Stearoyl-CoA Desaturase and miRNA 103 Expression in Goat Milk: Preliminary Results. Animals 2019, 9, 606. https://doi.org/10.3390/ani9090606
Tudisco R, Morittu VM, Addi L, Moniello G, Grossi M, Musco N, Grazioli R, Mastellone V, Pero ME, Lombardi P, et al. Influence of Pasture on Stearoyl-CoA Desaturase and miRNA 103 Expression in Goat Milk: Preliminary Results. Animals. 2019; 9(9):606. https://doi.org/10.3390/ani9090606
Chicago/Turabian StyleTudisco, Raffaella, Valeria Maria Morittu, Laura Addi, Giuseppe Moniello, Micaela Grossi, Nadia Musco, Raffaella Grazioli, Vincenzo Mastellone, Maria Elena Pero, Pietro Lombardi, and et al. 2019. "Influence of Pasture on Stearoyl-CoA Desaturase and miRNA 103 Expression in Goat Milk: Preliminary Results" Animals 9, no. 9: 606. https://doi.org/10.3390/ani9090606







