Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
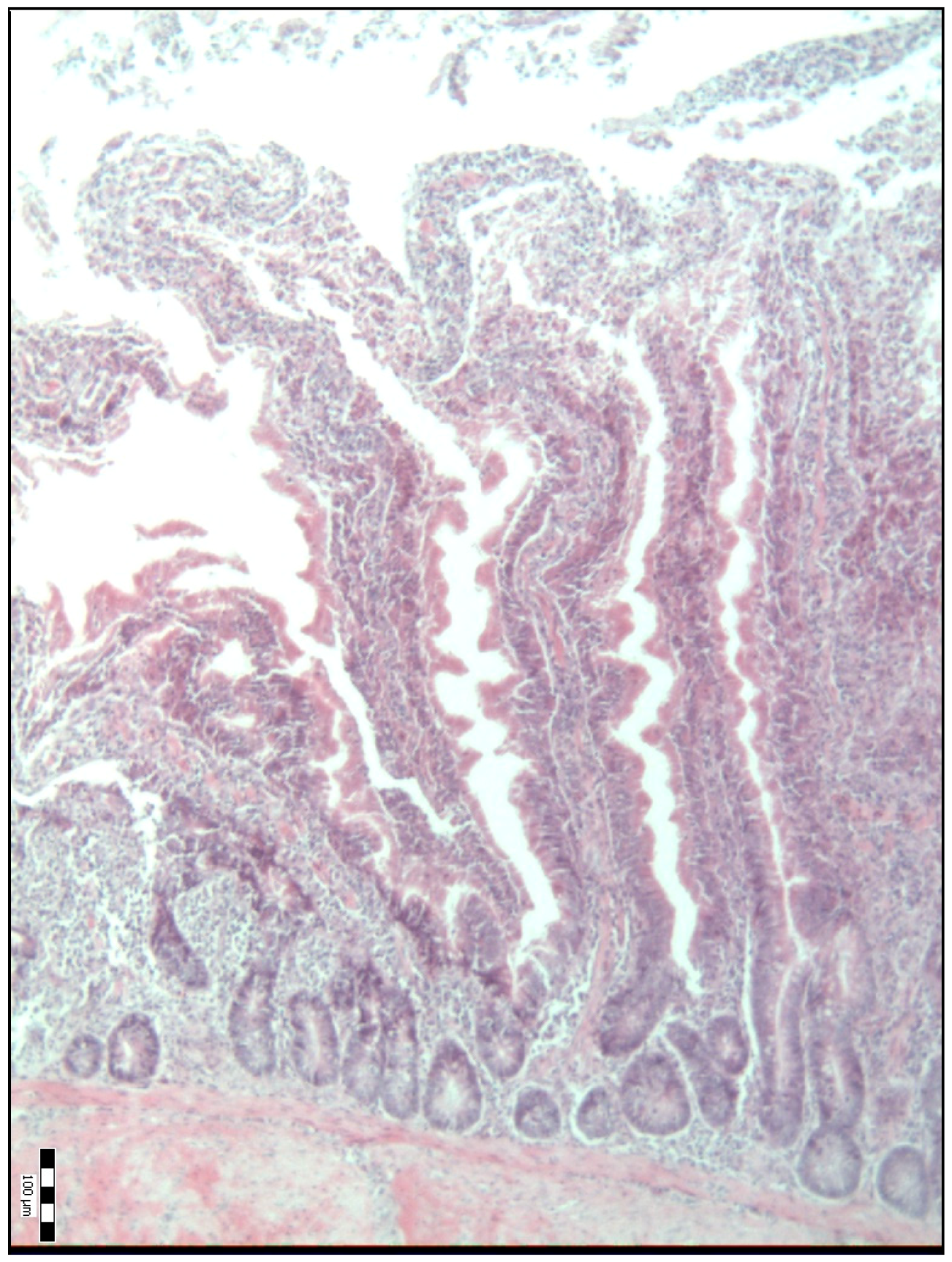
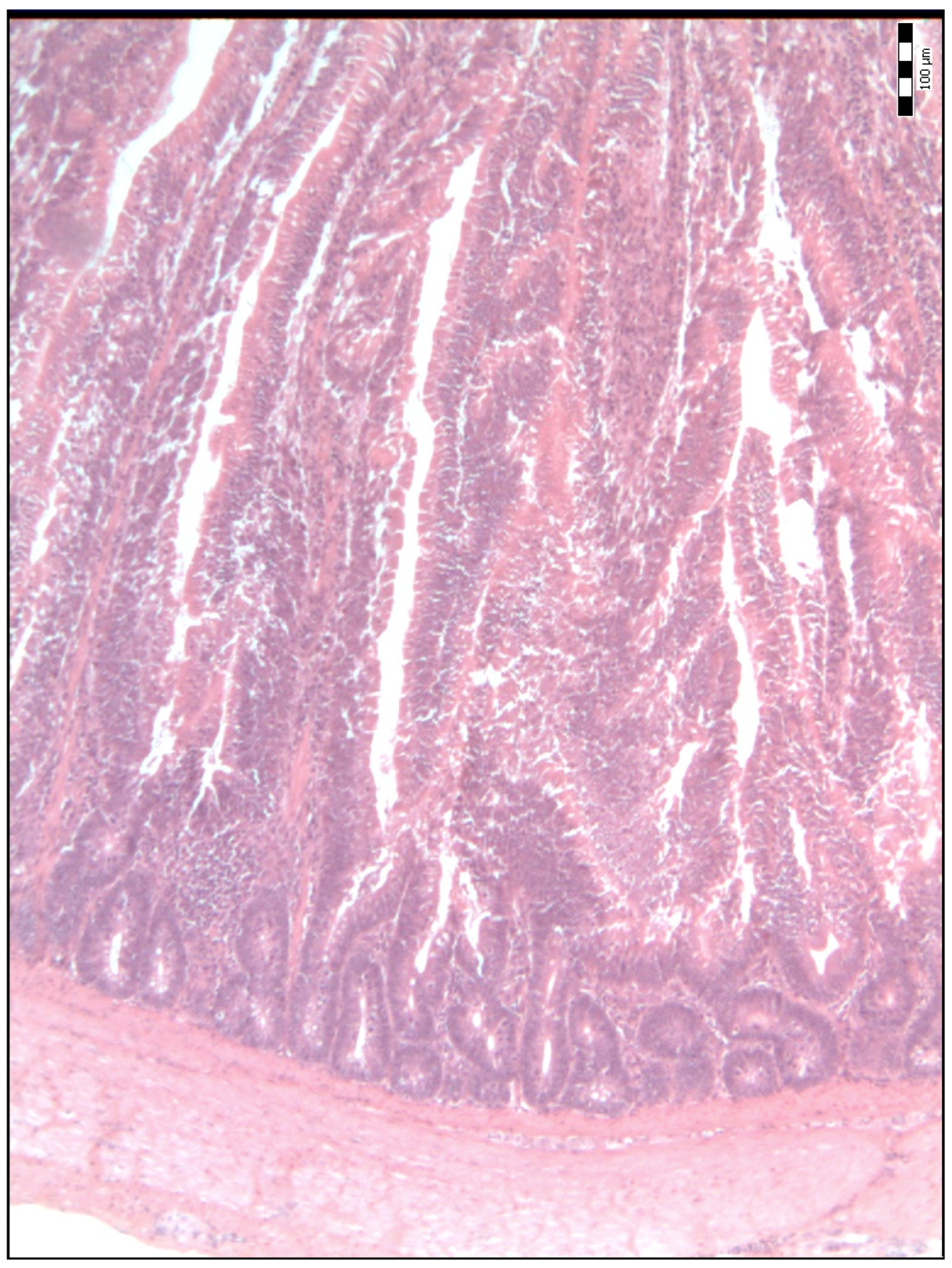
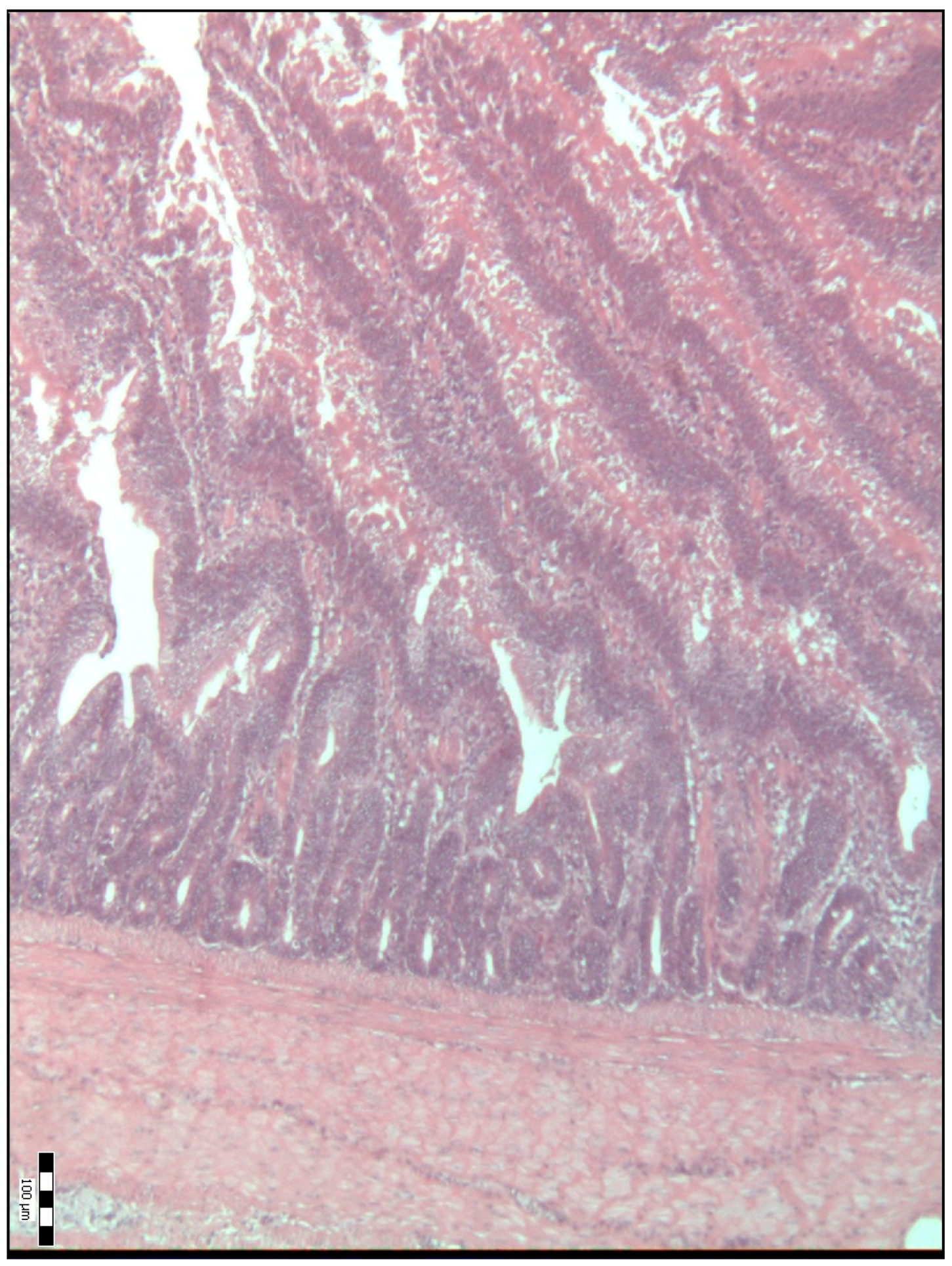
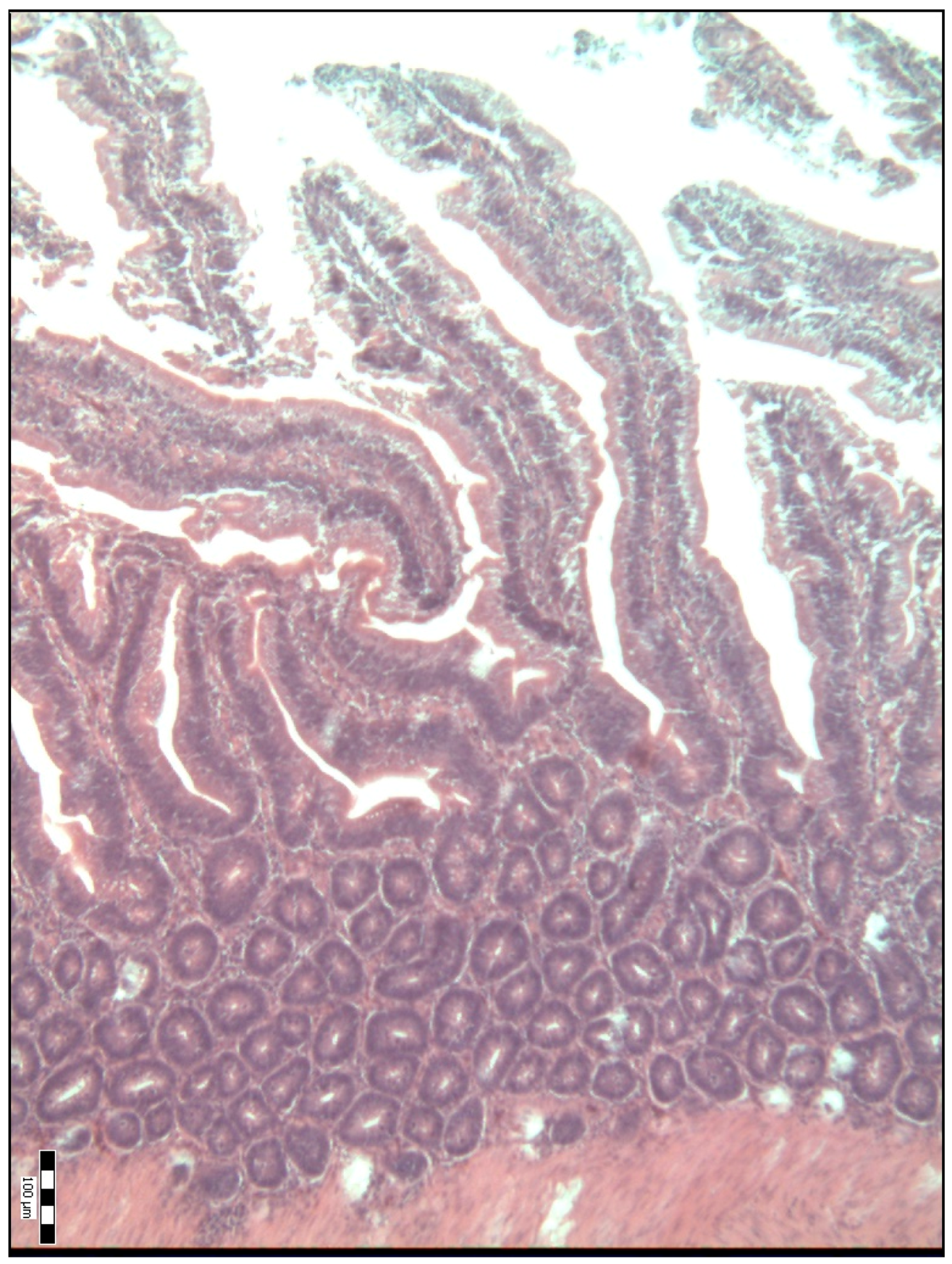
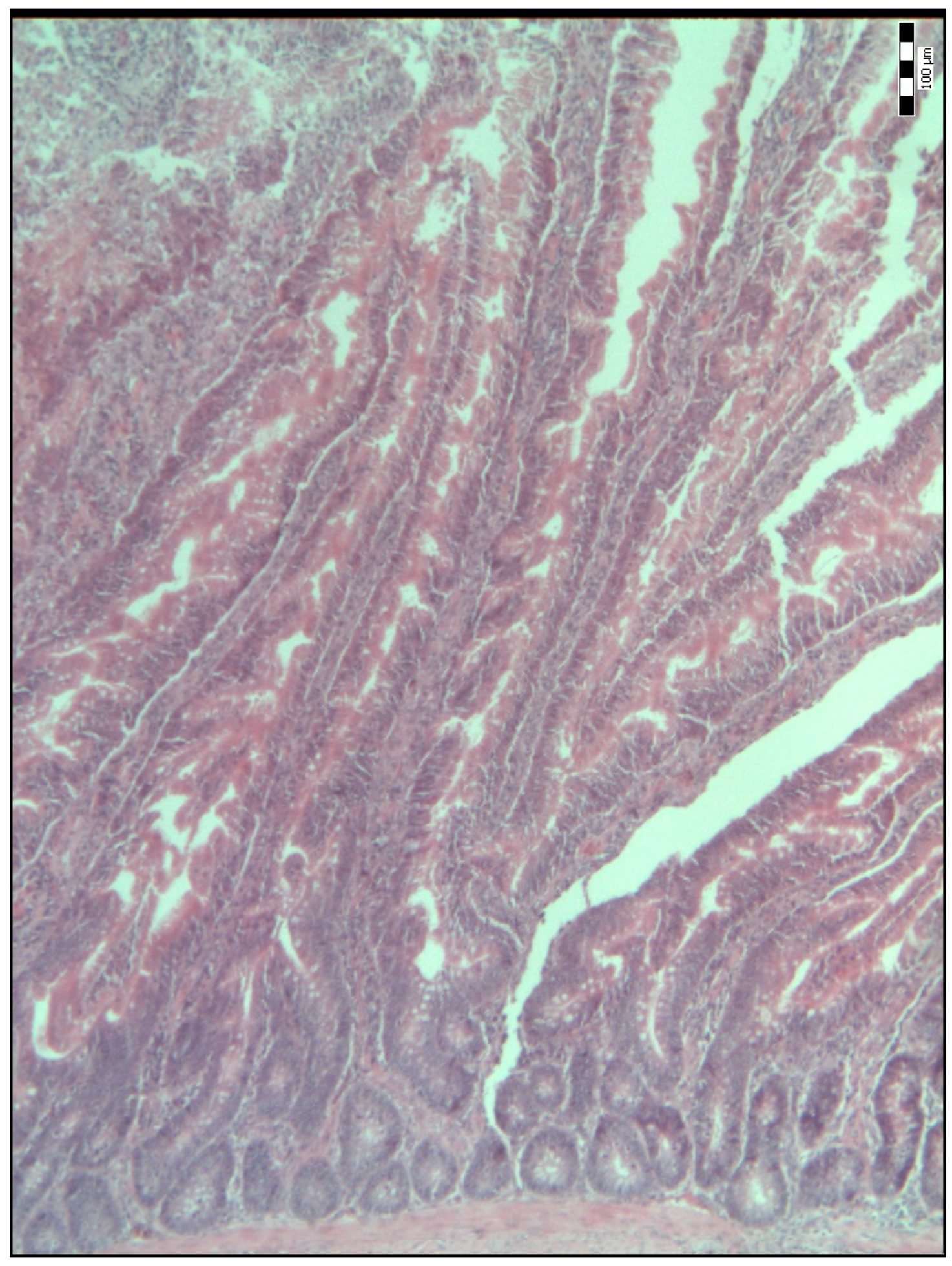
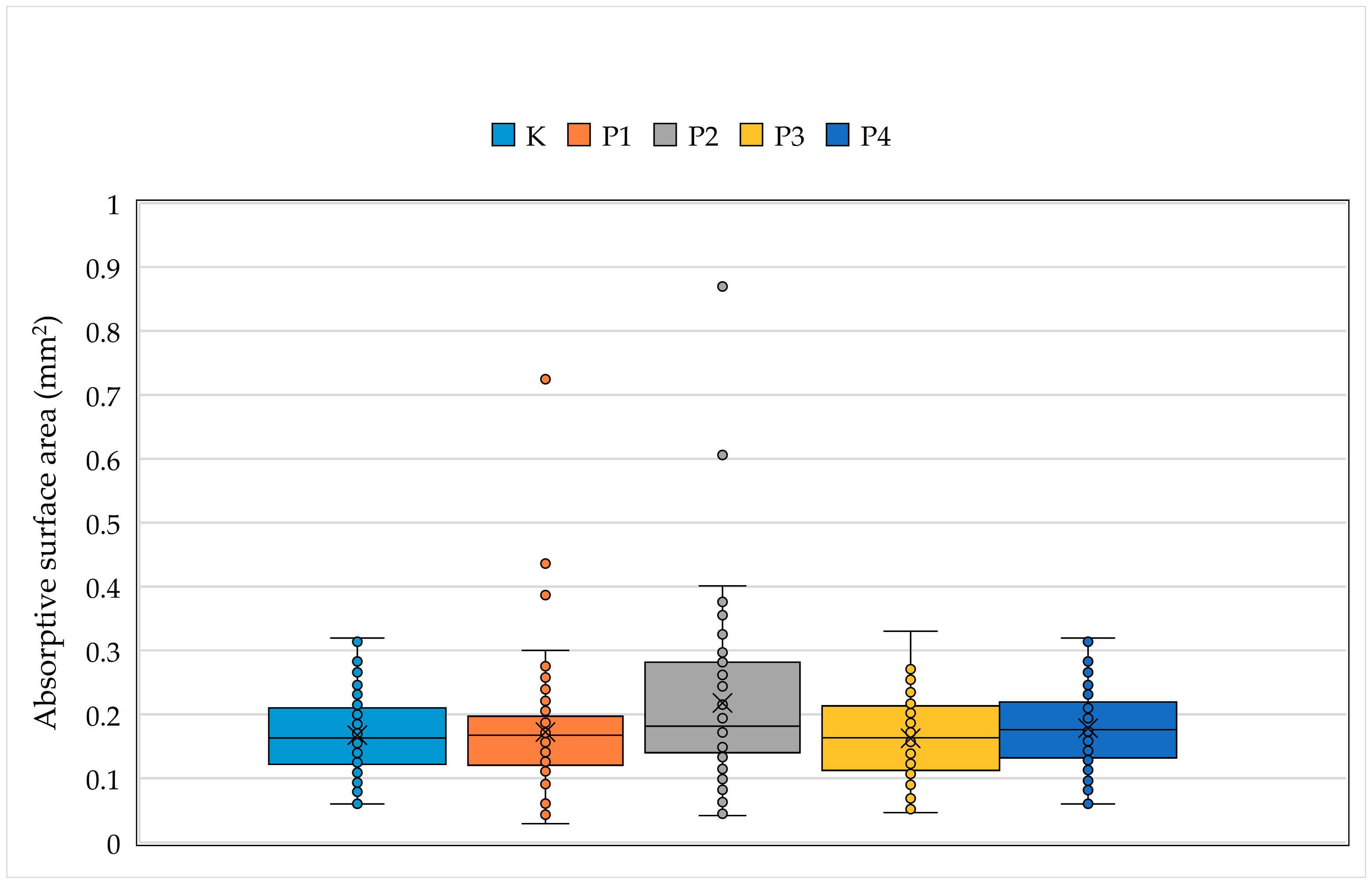
2.2. Sample Collection, Measurements and Analysis
2.3. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Krocko, M.; Canigova, M.; Bezekova, J.; Lavova, M.; Hascik, P.; Duckova, V. Effect of nutrition with propolis and bee pollen supplements on bacteria colonization pattern in gastrointestinal tract of broiler chickens. Sci. Pap. Anim. Sci. Biotechnol. 2012, 45, 63–67. [Google Scholar]
- Babinska, I.; Kleczek, K.; Szarek, J.; Makowski, W. Modulating effect of propolis and bee pollen on chicken breeding parameters and pathomorphology of liver and kidneys in the course of natural infection with Salmonella enteritidis. Bull. Vet. Inst. Pulawy 2012, 56, 3–8. [Google Scholar] [CrossRef]
- Klaric, I.; Pavic, M.; Miskulin, I.; Blazicevic, V.; Dumic, A.; Miskulin, M. Influence of Dietary Supplementation of Propolis and Bee Pollen on Liver Pathology in Broiler Chickens. Animals 2018, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Tekeli, A.; Kutlu, H.R.; Celik, L. Effects of Z. officinale and propolis extracts on the performance, carcass and some blood parameters of broiler chicks. Curr. Res. Poult. Sci. 2011, 1, 12–23. [Google Scholar] [CrossRef]
- Hascik, P.; Elimam, I.; Garlik, J.; Kacaniova, M.; Cubon, J.; Bobko, M.; Abdulla, H. Impact of bee pollen as feed supplements on the body weight of broiler Ross 308. Afr. J. Biotechnol. 2012, 11, 15596–15599. [Google Scholar]
- Attia, A.; Abd Al-Hamid, A.E.; Ibrahim, M.S.; Al-Harthi, M.A.; El-Naggar, A.S. Effects of propolis and/or bee pollen as alternatives to mannan oligosaccharides when given continuously or intermittently on production, and physiological performance of broilers. Poult. Sci. 2013, 92, 61–62. [Google Scholar]
- Attia, Y.A.; Abd Al-Hamid, A.E.; Ibrahim, M.S.; Al-Harthi, M.A.; Bovera, F.; El-Naggar, A.S. Productive performance, biochemical and hematological traits of broiler chickens supplemented with propolis, bee pollen, and mannan oligosaccharides continuously or intermittently. Livest. Sci. 2014, 164, 87–95. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Vakili Azghandi, M.; Ahani, S.; Nourmohammadi, R. Effect of bee pollen and propolis (bee glue) on growth performance and biomarkers of heat stress in broiler chickens reared under high ambient temperature. J. Anim. Feed Sci. 2016, 25, 45–51. [Google Scholar] [CrossRef]
- Zafarnejad, K.; Afzali, N.; Rajabzadeh, M. Effect of bee glue on growth performance and immune response of broiler chickens. J. App. Anim. Res. 2017, 45, 280–284. [Google Scholar] [CrossRef]
- Kacaniova, M.; Rovna, K.; Arpasova, H.; Cubon, J.; Hleba, L.; Pochop, J.; Kunova, S.; Hascik, P. In vitro and in vivo antimicrobial activity of propolis on the microbiota from gastrointestinal tract of chickens. J. Environ. Sci. Health A 2012, 47, 1665–1671. [Google Scholar] [CrossRef]
- Ao, Z.; Choct, M. Oligosaccharides Affect Performance and Gut Development of Broiler Chickens. Asian-Australas. J. Anim. Sci. 2013, 26, 116–121. [Google Scholar] [CrossRef]
- Duarte, C.R.A.; Eyng, C.; Murakami, A.E.; Santos, T.C. Intestinal morphology and activity of digestive enzymes in broilers fed crude propolis. Can. J. Anim. Sci. 2014, 94, 105–114. [Google Scholar] [CrossRef]
- Abou-Zeid, A.E.; El-Damarawy, S.Z.; Mariey, Y.A.; El-Kasass, S.M. Effect of dietary propolis supplementation on performance and activity of antioxidant enzymes in broiler chickens. Egypt. J. Nutr. Feeds 2015, 18, 391–400. [Google Scholar]
- Shaddel-Tili, A.; Eshratkhah, B.; Kouzehgari, H.; Ghasemi-Sadabadi, M. The effect of different levels of propolis in diets on performance, gastrointestinal morphology and some blood parameters in broiler chickens. Bulg. J. Vet. Med. 2017, 20, 215–224. [Google Scholar] [CrossRef]
- Toman, R.; Hajkova, Z.; Hluchy, S. Changes in Intestinal Morphology of Rats Fed with Different Levels of Bee Pollen. Pharmacogn. Commn. 2015, 5, 261–264. [Google Scholar] [CrossRef]
- Wang, J.; Li, S.; Wang, Q.; Xin, B.; Wang, H. Trophic Effect of Bee Pollen on Small Intestine in Broiler Chickens. J. Med. Food 2007, 10, 276–280. [Google Scholar] [CrossRef]
- Eyng, C.; Murakami, A.E.; Duarte, C.R.A.; Santos, T.C. Effect of dietary supplementation with an ethanolic extract of propolis on broiler intestinal morphology and digestive enzyme activity. J. Anim. Physiol. Anim. Nutr. 2014, 98, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Fazayeli-Rad, A.R.; Afzali, N.; Farhangfar, H.; Asghari, M.R. Effect of bee pollen on growth performance, intestinal morphometry and immune status of broiler chicks. Eur. Poult. Sci. 2015, 79, 1–9. [Google Scholar]
- Chegini, S.; Kiani, A.; Rokni, H. Alleviation of thermal and overcrowding stress in finishing broilers by dietary propolis supplementation. Ital. J. Anim. Sci. 2018, 17, 377–385. [Google Scholar] [CrossRef]
- Aviagen. Ross 308 Broiler: Performance Objectives; Aviagen Group: Huntsville, AL, USA, 2014. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of Total Flavonoid Content in Propolis by Two Complementary Colorimetric Methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Brudnicki, A.; Brudnicki, W.; Szymeczko, R.; Bednarczyk, M.; Pietruszynska, D.; Kirkillo-Stacewicz, K. Histo-morphometric adaptation in the small intestine of broiler chicken, after embryonic exposure to a—Galactosides. J. Anim. Plant. Sci. 2017, 27, 1075–1082. [Google Scholar]
- Nain, S.; Renema, R.A.; Zuidhof, M.J.; Korver, D.R. Effect of metabolic efficiency and intestinal morphology on variability in n-3 polyunsatureated fatty acid enrichment of eggs. Poult. Sci. 2012, 91, 888–898. [Google Scholar] [CrossRef] [PubMed]
- Sohail, M.U.; Hume, M.E.; Byrd, J.A.; Nisbet, D.J.; Ijaz, A.; Sohail, A.; Shabbir, M.Z.; Rehman, H. Effect of supplementation of prebiotic mannan-oligosaccharides and probiotic mixture on growth performance of broilers subjected to chronic heat stress. Poult. Sci. 2012, 91, 2235–2240. [Google Scholar] [CrossRef] [PubMed]
- Tekeli, A.; Kutlu, H.R.; Çelik, L.; Doran, F. Determination of the effects of Z. officinale and propolis extracts on intestinal microbiology and histological characteristics in broilers. Int. J. Poult. Sci. 2010, 9, 898–906. [Google Scholar] [CrossRef]
- Sayrafi, R.; Shahrooz, R.; Soltanalinejad, F.; Rahimi, S. Histomorphometrical study of the prebiotic effects on intestine morphology and growth performance of broiler chickens. Vet. Res. Forum 2011, 2, 45–51. [Google Scholar]
- Hamedi, S.; Rezaian, M.; Shomali, T. Histological changes of small intestinal mucosa of cocks due to sunflower meal single feeding. Am. J. Anim. Vet. Sci. 2011, 6, 171–175. [Google Scholar]
- Izadi, H.; Arshami, J.; Golian, A.; Reza Raji, M. Effects of chicory root powder on growth performance and histomorphometry of jejunum in broiler chicks. Vet. Res. Forum 2013, 4, 169–174. [Google Scholar] [PubMed]
- Caspary, W.F. Physiology and pathophysiology of intestinal absorption. Am. J. Clin. Nutr. 1992, 55, 2995–3085. [Google Scholar] [CrossRef]
- Awad, W.A.; Bohm, J.; Razzazi-Fazeli, E.; Ghareeband, K.; Zentek, J. Effect of addition of a probiotic microorganism to broiler diets contaminated with deoxynivalenol on performance and histological alterations of intestinal villi of broiler chickens. Poult. Sci. 2006, 85, 974–979. [Google Scholar] [CrossRef]
- Samanya, M.; Yamauchi, K. Histological alterations of intestinal villi in chickens fed dried Bacillus subtilis var. natto. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2002, 133, 95–104. [Google Scholar] [CrossRef]
- Onderci, M.; Sahin, N.; Sahin, K.; Cikim, G.; Aydin, A.; Ozercan, I. Efficacy of supplementation of alpha-amylase-producing bacterial culture on the performance, nutrient use and gut morphology of broiler chickens fed a corn-based diet. Poult. Sci. 2006, 85, 505–510. [Google Scholar] [CrossRef]
- Saeid, J.M.; Mohamed, A.B.; Al-Baddy, M.A. Effect of garlic powder (Allium sativum) and black seed (Nigella sativa) on broiler growth performance and intestinal morphology. Iran. J. Appl. Anim. Sci. 2013, 3, 185–188. [Google Scholar]
- Silva, M.A.; Pessotti, B.M.S.; Zanini, S.F.; Colnago, G.L.; Rodrigues, M.R.A.; Nunes, L.C.; Zanini, M.S.; Martins, I.V.F. Intestinal mucosa structure of broiler chickens infected experimentally with Eimeria tenella and treated with essential oil of oregano. Cienc. Rural 2009, 39, 1471–1477. [Google Scholar] [CrossRef]
- Langhout, D.J.; Schutte, J.B.; Vanleeuwen, P.; Wiebengaand, J.; Tamminga, S. Effect of dietary high-and low-methylated citrus pectin on the activity of the ileal microflora and morphology of the small intestinal wall of broiler chicks. Br. Poult. Sci. 1999, 40, 340–347. [Google Scholar] [CrossRef]
- Abdullah, A.Y.; Mahmoud, K.Z.; Nusairat, B.M.; Qudsieh, R.I. Small intestinal histology, production parameters, and meat quality as influenced by dietary supplementation of garlic (Allium sativum) in broiler chicks. Ital. J. Anim. Sci. 2010, 9, e80. [Google Scholar] [CrossRef]
- Oladele, O.A.; Emikpe, B.O.; Bakare, H. Effects of dietary garlic (Allium sativum Linn.) supplementation on body weight and gut morphometry of commercial broilers. Int. J. Morphol. 2012, 30, 238–240. [Google Scholar] [CrossRef]
- Akbarian, A.; Golian, A.; Kermanshahi, H.; Farhoosh, R.; Raji, A.R.; De Smet, S.; Michiels, J. Growth performance and gut health parameters of finishing broilers supplemented with plant extracts and exposed to daily increased temperature. Span. J. Agric. Res. 2013, 11, 109–119. [Google Scholar] [CrossRef]
- Rhodes, M.J. Physiologically-active compounds in plant foods: An overview. Proc. Nutr. Soc. 1996, 55, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Si, W.; Gong, J.; Tsao, R.; Zhou, T.; Yu, H.; Poppe, C.; Johnson, R.; Du, Z. Antimicrobial activity of essential oils and structurally related synthetic food additives towards selected pathogenic and beneficial gut bacteria. J. Appl. Microbiol. 2006, 100, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Klaric, I. Production and Health Effects of Propolis and Bee Pollen as Food Additives in Broilers Feeding. Ph.D. Thesis, Faculty of Agriculture in Osijek, Josip Juraj Strossmayer University of Osijek, Osijek, Croatia, 2014. [Google Scholar]
| Ingredients, % | Starter | Finisher |
|---|---|---|
| Day 1–21 | Day 22–42 | |
| Corn grain | 45.00 | 46.10 |
| Flour middling | 2.80 | 3.00 |
| Dehydrated alfalfa | 2.80 | 4.00 |
| Soybean meal | 20.20 | 10.00 |
| Sunflower meal | 4.00 | 4.00 |
| Yeast | 4.00 | 3.00 |
| Full fat soybean | 12.40 | 20.00 |
| Vegetable oil | 3.70 | 5.00 |
| Monocalcium phosphate | 1.20 | 1.20 |
| Limestone | 1.60 | 1.40 |
| Salt | 0.30 | 0.30 |
| Premix * | 1.00 | 1.00 |
| Pigozen 801 | 1.00 | 1.00 |
| Total | 100.00 | 100.00 |
| Calculated Analysis | ||
| Crude protein, % | 21.02 | 19.15 |
| Crude fat, % | 8.36 | 10.96 |
| Crude fiber, % | 4.96 | 5.05 |
| Lysine, % | 1.11 | 0.96 |
| Methionine, % | 0.66 | 0.61 |
| Tryptophan, % | 0.26 | 0.23 |
| Calcium, % | 1.04 | 0.98 |
| Phosphorous, % | 0.70 | 0.67 |
| ME, MJ/kg | 12.30 | 13.10 |
| The Amount of Total Flavonoids (mg/g), Expressed as Equivalents of Quercetin | Propolis | Bee Pollen |
| 248.24 | 31.80 |
| Parameter | Group of Chickens Median (Q1–Q3) | * p | ||||
|---|---|---|---|---|---|---|
| K | P1 | P2 | P3 | P4 | ||
| Duodenal villi height (μm) | 718.50 a (584.50–841.50) | 834.00 b (695.00–990.00) | 992.00 c (814.50–1111.50) | 886.00 bcd (697.00–1134.50) | 798.50 bde (658.75–1088.00) | <0.001 |
| Base width of the duodenal villi (μm) | 48.00 a (38.00–68.00) | 59.00 b (52.00–69.50) | 67.00 bc (51.00–77.25) | 54.00 bd (45.50–67.00) | 65.00 ce (58.75–75.25) | <0.001 |
| Duodenal villi crypt depth (μm) | 78.00 a (66.00–93.00) | 85.00 a (75.50–93.50) | 71.50 b (63.75–80.25) | 64.00 c (54.00–74.50) | 78.00 ad (70.00–85.25) | <0.001 |
| Parameter | Group of Chickens Median (Q1–Q3) | * p | ||||
|---|---|---|---|---|---|---|
| K | P1 | P2 | P3 | P4 | ||
| The Villus Height-to-Crypt Depth Ratio | 8.86 a (7.16–10.60) | 9.81 b (8.51–12.09) | 14.24 c (11.64–16.36) | 13.61 cd (10.54–16.70) | 10.89 be (8.70–12.73) | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prakatur, I.; Miskulin, M.; Pavic, M.; Marjanovic, K.; Blazicevic, V.; Miskulin, I.; Domacinovic, M. Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen. Animals 2019, 9, 301. https://doi.org/10.3390/ani9060301
Prakatur I, Miskulin M, Pavic M, Marjanovic K, Blazicevic V, Miskulin I, Domacinovic M. Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen. Animals. 2019; 9(6):301. https://doi.org/10.3390/ani9060301
Chicago/Turabian StylePrakatur, Ivana, Maja Miskulin, Mirela Pavic, Ksenija Marjanovic, Valerija Blazicevic, Ivan Miskulin, and Matija Domacinovic. 2019. "Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen" Animals 9, no. 6: 301. https://doi.org/10.3390/ani9060301
APA StylePrakatur, I., Miskulin, M., Pavic, M., Marjanovic, K., Blazicevic, V., Miskulin, I., & Domacinovic, M. (2019). Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen. Animals, 9(6), 301. https://doi.org/10.3390/ani9060301







