Hair Cortisol in Sheltered Cows and Its Association with Other Welfare Indicators
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Welfare Measurement
2.1.1. Cleanliness, Lesions and Disease Measures
2.1.2. Cow Behaviour Measures
2.1.3. Shelter-Based Measures
2.2. Hair Cortisol
2.2.1. Sampling
2.2.2. Extraction of Cortisol from Hair
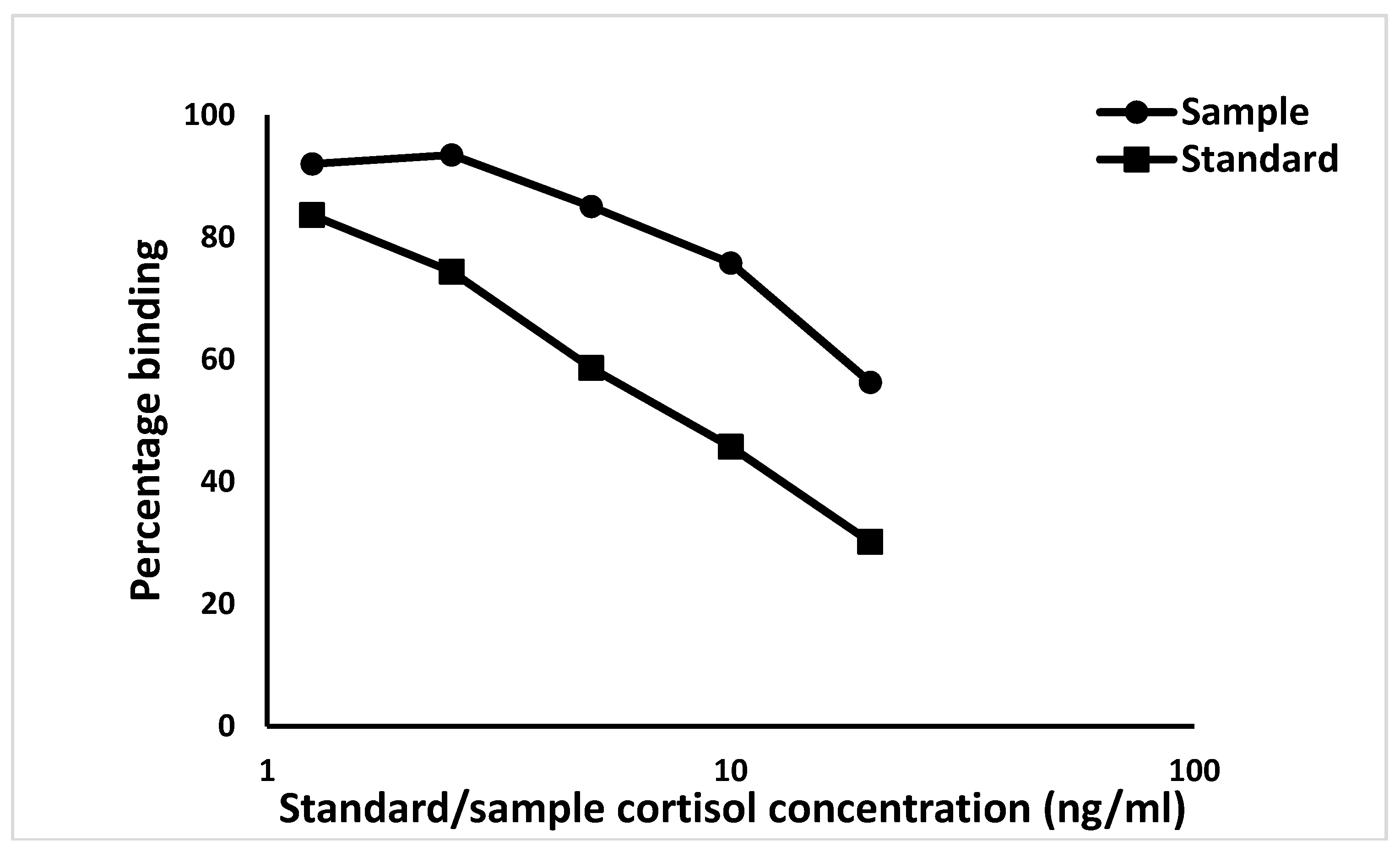
2.2.3. Cortisol Enzyme Immunoassay (EIA) for Determination of Hair Cortisol Concentration
2.3. Statistical Analyses
3. Results
3.1. Animal and Shelter Based Measures
3.2. Correlations between Hair Cortisol and Animal and Shelter Based Measures
4. Discussion
4.1. Hair Cortisol Concentrations
4.2. Hair Cortisol and Animal-Based Measures
4.3. Hair Cortisol and Shelter-Based Measures
4.4. Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | Description | Scales and Scores |
|---|---|---|
| Lactation | 0—Non-lactating; 1—Lactating | |
| Body Condition Score (BCS) [30,31] | A cow with a score of ≤1.25 was considered emaciated, 1.5–2 thin, 2.25–3.75 normal and 4 or more obese Visual examination | 1 to 5 with increments of 0.25. |
| General temperament [32] | Visual examination | 0—docile; 1—aggressive |
| Dirtiness of the hind limbs, udder and flanks [33] | By visual inspection of the cows from both sides (left and right) and from behind | 1—no dirtiness; 2—mildly dirty (small soiled areas of dirtiness with no thick scabs); 3—medium dirtiness (large soiled areas but with <1 cm thick scabs of dung); 4—severely dirty (large soiled areas with >1 cm thick dung scabs); |
| Body hair loss [33] | Visual inspection | 0—absence of hair loss;1—mild; 2—medium; 3—severe |
| Hock joint swellings [33,34] | Visual examination | 0—no swollen joint; 1—mild swollen joint; 2—medium swollen joint; 3—severely swollen joint |
| Hock joint hair loss and ulceration [33,34] | Visual examination | 0—no hair loss or ulceration; 1—mild hair loss or ulceration <2 cm2; 2—medium hair loss or ulceration (approximately 2.5 cm2); 3—severe hair loss or ulceration >2.5 cm2 |
| Carpal joint injuries [34] | Visual examination | 0—no skin change; 1—hairless; 2—swollen; 3—wound(s) |
| Lesions from shelter furniture [35] | Visual examination | 0—normal (no lesions present); 1—small area of hair loss; 2—moderate area of hair loss and/or thickening of the skin; 3—severe (a large area of hair loss and/or breakage of the skin |
| Neck lesions [36] | Visual examination | 1—no observable skin change; 2—hair loss; 3—swollen; 4—closed wounds (haematomas or closed abscesses); 5—open wounds |
| Ocular lesions [37] | Visual examination | 0—absent; 1—present |
| Nasal discharge [37] | Visual examination | 0—absent; 1—present |
| Hampered respiration [37] | Visual examination | 0—absent; 1—present |
| Diarrhoea [37] | Visual examination | 0—absent; 1—present |
| Vulvar discharge [37] | Visual examination | 0—absent; 1—present |
| Rumen Fill Score [38] | Visually by standing behind the cow on the left side and observing the left paralumbar fossa between the last rib, the lumbar transverse processes and the hip bone | 1—the paralumbar fossa is empty, presenting a rectangular cavity that is more than a hand’s width behind the last rib and a hand’s width under the lumbar transversal processes; 2—the paralumbar fossa forms a triangular cavity with a width about the size of a hand behind the last rib, but less than this under the lumbar transverse processes; 3—the paralumbar fossa forms a cavity less than a hand’s width behind the last rib and about a hand’s width vertically downwards from the lumbar transverse processes and then bulges out; 4—the paralumbar fossa skin covers the area behind the last rib and arches immediately outside below the lumbar transverse processes due to a bloated rumen; 5—the rumen is distended and almost fills up the paralumbar fossa; the last rib and the lumbar transverse processes are not visible |
| Faecal consistency [38] | Visual inspection | 1—thin and watery and not truly recognizable as faeces; 2—thin custard-like consistency, structurally recognizable as faeces, splashing out wide upon falling on the floor; 3—thick custard-like consistency, making a plopping sound while falling on the floor and a well-circumscribed pad which spreads out and is about 2 cm thick; 4—stiff with a heavy plopping sound while falling on the floor and a proper circumscribed pad with visible rings and minimal spreading out; 5—hard faecal balls like horse faeces |
| Lameness Score [39] | 1 to 5 scale Visual examination | 1—not lame (smooth and fluid movement) 2—mildly lame but not observable easily (an imperfect gait but able to freely move with a mildly arched back) 3—moderately lame (able to move but not freely, with an arched back) 4—lame, with inability to move freely with and asymmetrical gait and abnormal head movement 5—severely lame (severely restricted in movement, requiring considerable encouragement to move, and a severely arched back) |
| Claw overgrowth [35] | Visual examination | 0—Normal claws; 1—Mild claw overgrowth; 2—Moderate claw overgrowth; 3—Severe claw overgrowth |
| Skin lesions/Integument alterations [40] | Visual examination | 0—normal (no apparent lesions); 1—mild hair loss (<2 cm2); 2—moderate (>2 cm2 hair loss and inflamed skin); 3—severe (a large >4 cm2 area of hair loss with extensive skin inflammation and breakage) |
| Oral lesions | Visual examination | 0—absent; 1—present |
| Teat and/or udder condition | Visual inspection | 1—Lactating udder and teats; 2—Non-lactating udder and teats; 3—Teat cracks; 4—Warts on teats and udder; 5—Acute lesions on the teats and udder; 6—Chronic lesions on teats and udder |
| Skin tenting time [69] | Visual examination by skin pinch of the cervical region of neck | 1—≤2 s; 2—>2 s; 3—≥6 s |
| Ectoparasitism [41] | Visual examination | 1—Absence of ectoparasites; 2—Mild infestation—no lesions (not easily visible by naked eye but on tactile perception in the neck region; 3—Moderate—mild infestation visually observable ectoparasites or immature forms or eggs in the neck, groin, peri rectal, tail root and switch regions; 4—Severe—observation of mature ectoparasites all over the body especially regions mentioned in score 3 |
| Avoidance Distance (AD) [42] | Cows that were standing at the feeding manger were approached at the front at a rate of one step per second, starting at 2 m from the manger. The distance between the assessor’s hand and the cow’s head was estimated at the moment the cow moved away and turned its head. | 0—touched; 1—0 to 50 cm; 2—51 to 100 cm; 3—>100 cm |
| Rising difficulty [43,44] | All cows lying in the shelter were coaxed to get up with use of a minimum amount of force. If the presence of the assessor did not evoke rising they were given one or two gentle slaps on the back, followed by a break of 5 s, then more slaps with slightly more force if required, up to a maximum of 30 s. | 1—Normal (smooth and a normal sequence of rising behaviour; 2—Easy but slightly interfered (smooth movement with slight twisting of the head but with normal sequence of rising process; 3—Uneasy with effort (sudden movement and difficulty in rising with awkward twisting of the head and neck but following a normal sequential rising process); 4—Abnormal (uncharacteristic sequence of a rising event); 5—refused to get up |
| Cortisol | Lactation | BCS | Temperament | Dirty Hind Limbs | Dirty Udder | Dirty Flanks | |
|---|---|---|---|---|---|---|---|
| Lactation | −0.090 | ||||||
| 0.041 | |||||||
| BCS | −0.173 | 0.150 | |||||
| 0.000 | 0.001 | ||||||
| Temperament | −0.029 | 0.072 | 0.279 | ||||
| 0.511 | 0.104 | 0.000 | |||||
| Dirty Hind Limbs | 0.232 | 0.049 | −0.166 | −0.067 | |||
| 0.000 | 0.269 | 0.000 | 0.132 | ||||
| Dirty Udder | 0.270 | 0.027 | −0.255 | −0.057 | 0.765 | ||
| 0.000 | 0.544 | 0.000 | 0.195 | 0.000 | |||
| Dirty Flanks | 0.297 | 0.047 | −0.236 | −0.061 | 0.747 | 0.906 | |
| 0.000 | 0.291 | 0.000 | 0.170 | 0.000 | 0.000 | ||
| Body Hair Loss | 0.083 | −0.068 | −0.453 | −0.112 | 0.263 | 0.423 | 0.421 |
| 0.060 | 0.124 | 0.000 | 0.011 | 0.000 | 0.000 | 0.000 | |
| Hock Joint Swell | 0.066 | −0.058 | 0.038 | −0.004 | 0.146 | 0.155 | 0.143 |
| 0.137 | 0.188 | 0.396 | 0.920 | 0.001 | 0.000 | 0.001 | |
| Hock Joint Hair Loss | 0.155 | −0.077 | −0.207 | −0.022 | 0.040 | 0.101 | 0.093 |
| 0.000 | 0.083 | 0.000 | 0.618 | 0.365 | 0.022 | 0.035 | |
| Hock Joint Ulcer | 0.213 | −0.018 | −0.035 | 0.032 | 0.043 | 0.106 | 0.125 |
| 0.000 | 0.677 | 0.432 | 0.469 | 0.329 | 0.016 | 0.005 | |
| Carpal Joint Injuries | 0.276 | 0.009 | −0.060 | 0.053 | 0.200 | 0.323 | 0.335 |
| 0.000 | 0.847 | 0.174 | 0.233 | 0.000 | 0.000 | 0.000 | |
| Neck Lesions | 0.012 | −0.007 | −0.150 | −0.038 | 0.059 | 0.049 | 0.046 |
| 0.788 | 0.871 | 0.001 | 0.395 | 0.186 | 0.267 | 0.296 | |
| Ocular Lesions | 0.100 | −0.035 | −0.067 | −0.019 | 0.007 | 0.069 | 0.065 |
| 0.023 | 0.435 | 0.131 | 0.666 | 0.872 | 0.121 | 0.144 | |
| Coughing | 0.060 | −0.017 | −0.071 | −0.027 | 0.074 | 0.076 | 0.076 |
| 0.175 | 0.698 | 0.110 | 0.538 | 0.097 | 0.086 | 0.085 | |
| Nasal Discharge | 0.149 | −0.080 | −0.114 | −0.048 | 0.113 | 0.114 | 0.084 |
| 0.001 | 0.069 | 0.010 | 0.279 | 0.010 | 0.010 | 0.057 | |
| Hampered Respiration | −0.066 | −0.017 | −0.010 | −0.027 | −0.043 | −0.011 | −0.012 |
| 0.136 | 0.698 | 0.817 | 0.538 | 0.328 | 0.796 | 0.786 | |
| Diarrhoea | 0.040 | 0.001 | −0.028 | 0.074 | 0.115 | 0.126 | 0.140 |
| 0.366 | 0.977 | 0.520 | 0.093 | 0.009 | 0.004 | 0.002 | |
| Vulvar Discharge | 0.056 | 0.006 | −0.006 | −0.050 | 0.052 | 0.069 | 0.091 |
| 0.209 | 0.893 | 0.887 | 0.261 | 0.236 | 0.120 | 0.040 | |
| Rumen Fill Score | −0.224 | 0.016 | 0.182 | 0.016 | −0.054 | −0.168 | −0.224 |
| 0.000 | 0.714 | 0.000 | 0.725 | 0.225 | 0.000 | 0.000 | |
| Lameness | 0.177 | −0.010 | −0.105 | −0.065 | 0.205 | 0.257 | 0.255 |
| 0.000 | 0.820 | 0.017 | 0.144 | 0.000 | 0.000 | 0.000 | |
| Lesions on the Body | 0.176 | −0.048 | −0.375 | −0.108 | 0.166 | 0.313 | 0.334 |
| 0.000 | 0.276 | 0.000 | 0.015 | 0.000 | 0.000 | 0.000 | |
| Claw Overgrowth | 0.157 | 0.052 | −0.087 | 0.005 | 0.175 | 0.273 | 0.277 |
| 0.000 | 0.238 | 0.049 | 0.914 | 0.000 | 0.000 | 0.000 | |
| Faecal Consistency | −0.042 | −0.136 | −0.135 | −0.124 | −0.209 | −0.175 | −0.194 |
| 0.344 | 0.002 | 0.002 | 0.005 | 0.000 | 0.000 | 0.000 | |
| Coat Condition | −0.049 | 0.129 | 0.617 | 0.232 | −0.265 | −0.367 | −0.354 |
| 0.265 | 0.004 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | |
| Teat Score | 0.169 | −0.596 | −0.228 | −0.100 | 0.013 | 0.041 | 0.037 |
| 0.000 | 0.000 | 0.000 | 0.024 | 0.770 | 0.359 | 0.404 | |
| Ectoparasitism | 0.021 | −0.149 | −0.203 | 0.033 | 0.149 | 0.249 | 0.220 |
| 0.635 | 0.001 | 0.000 | 0.454 | 0.001 | 0.000 | 0.000 | |
| Skin Tenting Time | 0.178 | −0.053 | −0.206 | −0.057 | 0.173 | 0.245 | 0.259 |
| 0.000 | 0.234 | 0.000 | 0.196 | 0.000 | 0.000 | 0.000 | |
| Age | 0.111 | −0.132 | −0.117 | −0.061 | −0.012 | 0.029 | −0.000 |
| 0.012 | 0.003 | 0.008 | 0.166 | 0.783 | 0.511 | 1.000 | |
| Body Hair Loss | Hock Joint Swell | Hock Joint Hair Loss | Hock Joint Ulcer | Carpal Joint Injuries | Neck Lesions | Ocular Lesions | |
| Hock Joint Swell | 0.056 | ||||||
| 0.207 | |||||||
| Hock Joint Hair Loss | 0.162 | 0.381 | |||||
| 0.000 | 0.000 | ||||||
| Hock Joint Ulcer | 0.051 | 0.267 | 0.611 | ||||
| 0.251 | 0.000 | 0.000 | |||||
| Carpal Joint Injuries | 0.112 | 0.198 | 0.172 | 0.247 | |||
| 0.012 | 0.000 | 0.000 | 0.000 | ||||
| Neck Lesions | 0.185 | −0.106 | −0.006 | −0.031 | 0.048 | ||
| 0.000 | 0.016 | 0.893 | 0.487 | 0.278 | |||
| Ocular lesions | −0.005 | 0.048 | 0.073 | 0.079 | 0.039 | −0.034 | |
| 0.904 | 0.278 | 0.101 | 0.075 | 0.383 | 0.436 | ||
| Coughing | 0.080 | 0.026 | 0.057 | 0.073 | 0.019 | −0.012 | −0.015 |
| 0.071 | 0.562 | 0.200 | 0.098 | 0.676 | 0.793 | 0.737 | |
| Nasal Discharge | 0.091 | 0.029 | 0.131 | 0.092 | 0.129 | 0.009 | 0.048 |
| 0.039 | 0.506 | 0.003 | 0.037 | 0.003 | 0.847 | 0.279 | |
| Hampered respiration | −0.044 | −0.055 | −0.064 | −0.041 | −0.045 | −0.012 | 0.132 |
| 0.317 | 0.210 | 0.151 | 0.350 | 0.307 | 0.793 | 0.003 | |
| Diarrhoea | −0.003 | −0.061 | −0.018 | 0.002 | 0.072 | −0.046 | 0.095 |
| 0.949 | 0.166 | 0.679 | 0.960 | 0.106 | 0.303 | 0.032 | |
| Vulvar Discharge | 0.049 | −0.013 | −0.034 | −0.024 | 0.025 | 0.052 | 0.102 |
| 0.273 | 0.778 | 0.438 | 0.590 | 0.565 | 0.239 | 0.021 | |
| Rumen Fill Score | −0.197 | 0.047 | −0.000 | −0.159 | −0.147 | −0.039 | −0.183 |
| 0.000 | 0.287 | 0.994 | 0.000 | 0.001 | 0.373 | 0.000 | |
| Lameness | 0.091 | 0.080 | 0.054 | 0.095 | 0.201 | 0.034 | 0.051 |
| 0.039 | 0.070 | 0.225 | 0.032 | 0.000 | 0.439 | 0.253 | |
| Lesions on the Body | 0.708 | 0.021 | 0.165 | 0.150 | 0.110 | 0.173 | 0.067 |
| 0.000 | 0.631 | 0.000 | 0.001 | 0.013 | 0.000 | 0.129 | |
| Claw Overgrowth | 0.163 | 0.098 | −0.004 | 0.081 | 0.252 | 0.054 | 0.131 |
| 0.000 | 0.027 | 0.925 | 0.068 | 0.000 | 0.223 | 0.003 | |
| Faecal Consistency | 0.023 | 0.003 | 0.087 | 0.061 | −0.130 | 0.023 | 0.011 |
| 0.609 | 0.938 | 0.050 | 0.168 | 0.003 | 0.608 | 0.797 | |
| Coat Condition | −0.494 | −0.063 | −0.138 | −0.007 | −0.098 | −0.144 | −0.001 |
| 0.000 | 0.155 | 0.002 | 0.870 | 0.027 | 0.001 | 0.984 | |
| Teat Score | 0.078 | 0.038 | 0.089 | 0.090 | 0.056 | 0.018 | 0.070 |
| 0.076 | 0.397 | 0.045 | 0.041 | 0.204 | 0.684 | 0.113 | |
| Ectoparasitism | 0.245 | 0.050 | 0.105 | 0.097 | 0.147 | 0.043 | 0.053 |
| 0.000 | 0.257 | 0.017 | 0.028 | 0.001 | 0.337 | 0.229 | |
| Skin Tenting Time | 0.177 | −0.014 | 0.065 | 0.106 | 0.145 | 0.067 | 0.091 |
| 0.000 | 0.748 | 0.143 | 0.017 | 0.001 | 0.131 | 0.040 | |
| Age | 0.155 | 0.135 | 0.150 | 0.112 | 0.143 | 0.046 | 0.100 |
| 0.000 | 0.002 | 0.001 | 0.011 | 0.001 | 0.297 | 0.023 | |
| Coughing | Nasal Discharge | Hampered Respiration | Diarrhoea | Vulvar Discharge | Rumen Fill Score | Lameness | |
| Nasal Discharge | 0.126 | ||||||
| 0.004 | |||||||
| Hampered Respiration | −0.002 | −0.016 | |||||
| 0.965 | 0.726 | ||||||
| Diarrhoea | −0.008 | 0.088 | 0.255 | ||||
| 0.862 | 0.048 | 0.000 | |||||
| Vulvar Discharge | 0.264 | 0.095 | 0.264 | 0.042 | |||
| 0.000 | 0.032 | 0.000 | 0.344 | ||||
| Rumen Fill Score | 0.036 | −0.022 | 0.036 | −0.121 | −0.068 | ||
| 0.412 | 0.625 | 0.412 | 0.006 | 0.122 | |||
| Lameness | 0.105 | 0.094 | −0.018 | −0.005 | 0.070 | −0.121 | |
| 0.018 | 0.033 | 0.692 | 0.910 | 0.114 | 0.006 | ||
| Lesions on the Body | 0.058 | 0.090 | 0.012 | 0.032 | 0.090 | −0.323 | 0.125 |
| 0.190 | 0.041 | 0.787 | 0.464 | 0.042 | 0.000 | 0.004 | |
| Claw Overgrowth | 0.037 | 0.063 | 0.037 | 0.105 | 0.048 | −0.187 | 0.346 |
| 0.401 | 0.152 | 0.401 | 0.017 | 0.283 | 0.000 | 0.000 | |
| Faecal Consistency | 0.034 | −0.068 | −0.050 | −0.280 | 0.036 | −0.000 | −0.019 |
| 0.449 | 0.126 | 0.258 | 0.000 | 0.410 | 0.996 | 0.665 | |
| Coat Condition | −0.046 | −0.139 | 0.042 | −0.019 | 0.017 | 0.099 | −0.098 |
| 0.299 | 0.002 | 0.339 | 0.673 | 0.701 | 0.026 | 0.027 | |
| Teat Score | 0.014 | 0.074 | 0.014 | −0.034 | 0.019 | −0.069 | 0.040 |
| 0.749 | 0.092 | 0.749 | 0.444 | 0.663 | 0.118 | 0.369 | |
| Ectoparasitism | 0.076 | −0.001 | −0.037 | 0.055 | 0.097 | 0.004 | 0.028 |
| 0.086 | 0.990 | 0.409 | 0.217 | 0.028 | 0.936 | 0.534 | |
| Skin Tenting Time | 0.160 | 0.105 | −0.013 | 0.122 | 0.173 | −0.164 | 0.139 |
| 0.000 | 0.017 | 0.765 | 0.006 | 0.000 | 0.000 | 0.002 | |
| Age | 0.048 | 0.072 | 0.048 | −0.009 | 0.058 | −0.070 | 0.024 |
| 0.281 | 0.103 | 0.281 | 0.839 | 0.189 | 0.113 | 0.590 | |
| Lesions on the Body | Claw Overgrowth | Faecal Consistency | Coat Condition | Teat Score | Ectoparasitism | Skin Tenting Time | |
| Claw Overgrowth | 0.277 | ||||||
| 0.000 | |||||||
| Faecal Consistency | 0.015 | −0.069 | |||||
| 0.733 | 0.121 | ||||||
| Coat Condition | −0.401 | −0.092 | −0.074 | ||||
| 0.000 | 0.037 | 0.095 | |||||
| Teat Score | 0.107 | −0.010 | 0.155 | −0.213 | |||
| 0.015 | 0.820 | 0.000 | 0.000 | ||||
| Ectoparasitism | 0.225 | 0.132 | 0.061 | −0.328 | 0.196 | ||
| 0.000 | 0.003 | 0.171 | 0.000 | 0.000 | |||
| Skin Tenting Time | 0.252 | 0.193 | 0.085 | −0.199 | 0.142 | 0.249 | |
| 0.000 | 0.000 | 0.055 | 0.000 | 0.001 | 0.000 | ||
| Age | 0.118 | 0.079 | 0.089 | −0.095 | 0.118 | 0.025 | −0.022 |
| 0.007 | 0.075 | 0.045 | 0.031 | 0.007 | 0.579 | 0.616 |
| Cortisol | Shed Flooring | Shed Bed Type | Shed %Dung LyA | Shed %Dung Pa | Shed Urine LA | Shed Urine in Pa | |
|---|---|---|---|---|---|---|---|
| Shed flooring | −0.007 | ||||||
| 0.879 | |||||||
| Shed bed type | 0.044 | −0.284 | |||||
| 0.319 | 0.000 | ||||||
| Shed %Dung LyA | −0.082 | −0.198 | 0.044 | ||||
| 0.062 | 0.000 | 0.315 | |||||
| Shed %Dung Pa | 0.003 | −0.193 | 0.072 | 0.914 | |||
| 0.947 | 0.000 | 0.103 | 0.000 | ||||
| Shed urine LA | 0.105 | 0.179 | −0.089 | 0.115 | 0.101 | ||
| 0.017 | 0.000 | 0.045 | 0.009 | 0.022 | |||
| Shed urine in Pa | 0.059 | 0.183 | −0.089 | 0.102 | 0.123 | 0.578 | |
| 0.182 | 0.000 | 0.043 | 0.021 | 0.005 | 0.000 | ||
| Shed cleaned /cl | −0.095 | 0.169 | −0.046 | −0.622 | −0.594 | 0.144 | 0.039 |
| 0.032 | 0.000 | 0.298 | 0.000 | 0.000 | 0.001 | 0.373 | |
| Shed Bed Thickness | 0.044 | −0.284 | 1.000 | 0.050 | 0.077 | −0.089 | −0.089 |
| 0.316 | 0.000 | 0.000 | 0.262 | 0.082 | 0.045 | 0.043 | |
| Shed run off Ly | 0.196 | 0.206 | −0.129 | 0.018 | −0.015 | −0.048 | 0.064 |
| 0.000 | 0.000 | 0.003 | 0.687 | 0.731 | 0.274 | 0.145 | |
| Shed average gradient LyA | −0.109 | −0.284 | −0.010 | 0.057 | 0.058 | −0.030 | −0.124 |
| 0.014 | 0.000 | 0.829 | 0.196 | 0.193 | 0.498 | 0.005 | |
| Shed Average gradient Pa | 0.007 | −0.320 | 0.221 | 0.139 | 0.132 | −0.263 | −0.087 |
| 0.871 | 0.000 | 0.000 | 0.002 | 0.003 | 0.000 | 0.051 | |
| Yard flooring | 0.061 | 0.264 | −0.105 | −0.021 | 0.057 | 0.016 | 0.081 |
| 0.166 | 0.000 | 0.018 | 0.628 | 0.198 | 0.718 | 0.065 | |
| Yard %Dung | 0.076 | −0.360 | 0.218 | 0.285 | 0.384 | −0.120 | −0.293 |
| 0.109 | 0.000 | 0.000 | 0.000 | 0.000 | 0.012 | 0.000 | |
| Yard urine | 0.103 | 0.062 | −0.072 | −0.089 | −0.064 | 0.478 | 0.324 |
| 0.019 | 0.158 | 0.103 | 0.045 | 0.151 | 0.000 | 0.000 | |
| Yard area/cow | −0.035 | −0.189 | −0.029 | 0.018 | 0.019 | −0.128 | −0.315 |
| 0.466 | 0.000 | 0.543 | 0.706 | 0.697 | 0.007 | 0.000 | |
| Yard avg gradient | −0.059 | 0.065 | −0.094 | 0.008 | −0.011 | 0.108 | 0.095 |
| 0.231 | 0.188 | 0.055 | 0.874 | 0.828 | 0.027 | 0.054 | |
| Scraping Frequency | −0.014 | 0.466 | −0.167 | −0.405 | −0.431 | 0.075 | 0.117 |
| 0.757 | 0.000 | 0.000 | 0.000 | 0.000 | 0.092 | 0.008 | |
| Scraping method | −0.024 | −0.018 | −0.073 | 0.172 | 0.121 | 0.157 | 0.329 |
| 0.594 | 0.685 | 0.101 | 0.000 | 0.006 | 0.000 | 0.000 | |
| Avoidance Distance | 0.087 | 0.015 | −0.125 | −0.103 | −0.074 | 0.231 | 0.024 |
| 0.048 | 0.734 | 0.005 | 0.019 | 0.092 | 0.000 | 0.593 | |
| Rising up difficulty | 0.227 | 0.091 | −0.054 | −0.005 | −0.060 | 0.179 | 0.177 |
| 0.000 | 0.040 | 0.225 | 0.914 | 0.172 | 0.000 | 0.000 | |
| Shed area/cow | −0.056 | −0.040 | −0.038 | −0.032 | −0.097 | −0.097 | 0.044 |
| 0.207 | 0.366 | 0.388 | 0.473 | 0.028 | 0.028 | 0.319 | |
| Shed Cleaned/cl | Shed Bed Thickness | Shed Run off LyA | Shed Average Gradient LyA | Shed Average Gradient Pa | Yard Flooring | Yard %Dung | |
| Shed Bed Thickness | −0.050 | ||||||
| 0.258 | |||||||
| Shed run off Ly | −0.107 | −0.129 | |||||
| 0.016 | 0.003 | ||||||
| Shed average gradient | −0.061 | −0.009 | −0.036 | ||||
| 0.166 | 0.832 | 0.422 | |||||
| Shed Average gradient | −0.186 | 0.223 | 0.066 | 0.287 | |||
| 0.000 | 0.000 | 0.142 | 0.000 | ||||
| Yard flooring | 0.112 | −0.105 | −0.140 | −0.072 | −0.067 | ||
| 0.011 | 0.018 | 0.001 | 0.103 | 0.135 | |||
| Yard %Dung | −0.424 | 0.220 | 0.012 | 0.219 | 0.252 | −0.301 | |
| 0.000 | 0.000 | 0.809 | 0.000 | 0.000 | 0.000 | ||
| Yard urine | 0.171 | −0.072 | −0.093 | 0.148 | −0.075 | 0.305 | −0.112 |
| 0.000 | 0.103 | 0.035 | 0.001 | 0.094 | 0.000 | 0.018 | |
| Yard area/cow | 0.071 | −0.023 | 0.265 | 0.165 | 0.152 | −0.420 | 0.318 |
| 0.134 | 0.634 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | |
| Yard avg gradient | 0.092 | −0.092 | −0.234 | 0.298 | 0.030 | −0.059 | −0.112 |
| 0.061 | 0.062 | 0.000 | 0.000 | 0.548 | 0.233 | 0.022 | |
| Scraping Frequency | 0.407 | −0.167 | 0.051 | −0.311 | −0.355 | 0.072 | −0.530 |
| 0.000 | 0.000 | 0.251 | 0.000 | 0.000 | 0.102 | 0.000 | |
| Scraping method | −0.187 | −0.073 | 0.038 | −0.193 | 0.090 | 0.023 | −0.169 |
| 0.000 | 0.101 | 0.394 | 0.000 | 0.044 | 0.598 | 0.000 | |
| Avoidance Distance | −0.047 | −0.125 | 0.246 | 0.063 | −0.035 | −0.166 | −0.070 |
| 0.286 | 0.005 | 0.000 | 0.156 | 0.442 | 0.000 | 0.140 | |
| Rising up difficulty | −0.095 | −0.054 | 0.176 | −0.029 | −0.128 | −0.132 | −0.103 |
| 0.032 | 0.225 | 0.000 | 0.520 | 0.004 | 0.003 | 0.031 | |
| Shed area/cow | 0.068 | −0.034 | 0.137 | −0.213 | −0.035 | −0.169 | −0.038 |
| 0.125 | 0.440 | 0.002 | 0.000 | 0.436 | 0.000 | 0.423 | |
| Yard Urine | Yard Area/Cow | Yard Avg Gradient | Scraping Frequency | Scraping Method | Avoidance Distance | Rising up Difficulty | |
| Yard area/cow | −0.117 | ||||||
| 0.014 | |||||||
| Yard avg gradient | 0.064 | −0.097 | |||||
| 0.191 | 0.049 | ||||||
| Scraping Frequency | −0.074 | −0.036 | −0.020 | ||||
| 0.097 | 0.453 | 0.689 | |||||
| Scraping method | 0.238 | −0.126 | −0.032 | −0.249 | |||
| 0.000 | 0.008 | 0.513 | 0.000 | ||||
| Avoidance Distance | 0.302 | 0.111 | 0.029 | −0.165 | 0.192 | ||
| 0.000 | 0.019 | 0.553 | 0.000 | 0.000 | |||
| Rising up difficulty | 0.121 | −0.167 | −0.024 | 0.026 | 0.106 | 0.297 | |
| 0.006 | 0.000 | 0.632 | 0.557 | 0.017 | 0.000 | ||
| Shed area/cow | −0.149 | 0.065 | −0.094 | 0.167 | 0.069 | −0.311 | −0.009 |
| 0.001 | 0.174 | 0.056 | 0.000 | 0.120 | 0.000 | 0.844 |
References
- Heimbürge, S.; Kanitz, E.; Otten, W. The use of hair cortisol for the assessment of stress in animals. Gen. Comp. Endocr. 2018, 270, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Stalder, T.; Kirschbaum, C. Analysis of cortisol in hair–state of the art and future directions. Brain Behav. Immun. 2012, 26, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Macbeth, B.J.; Cattet, M.R.; Obbard, M.E.; Middel, K.; Janz, D.M. Evaluation of hair cortisol concentration as a biomarker of long-term stress in free-ranging polar bears. Wildl. Soc. Bull. 2012, 36, 747–758. [Google Scholar] [CrossRef]
- Koren, L.; Mokady, O.; Karaskov, T.; Klein, J.; Koren, G.; Geffen, E. A novel method using hair for determining hormonal levels in wildlife. Anim. Behav. 2002, 63, 403–406. [Google Scholar] [CrossRef]
- Yang, H.Z.; Lan, J.; Meng, Y.J.; Wan, X.J.; Han, D.W. A preliminary study of steroid reproductive hormones in human hair. J. Steroid Biochem. Mol. Biol. 1998, 67, 447–450. [Google Scholar] [CrossRef]
- Touma, C.; Palme, R. Measuring fecal glucocorticoid metabolites in mammals and birds: The importance of validation. Ann. N. Y. Acad. Sci. 2005, 1046, 54–74. [Google Scholar] [CrossRef]
- Sheriff, M.J.; Dantzer, B.; Delehanty, B.; Palme, R.; Boonstra, R. Measuring stress in wildlife: Techniques for quantifying glucocorticoids. Oecologia 2011, 166, 869–887. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, C.E.; Thierfelder, T.; Svennersten-Sjaunja, K.; Berg, C.; Orihuela, A.; Lidfors, L. Time lag between peak concentrations of plasma and salivary cortisol following a stressful procedure in dairy cattle. Acta Vet. Scand. 2014, 56, 61. [Google Scholar] [CrossRef] [PubMed]
- Davenport, M.D.; Tiefenbacher, S.; Lutz, C.K.; Novak, M.A.; Meyer, J.S. Analysis of endogenous cortisol concentrations in the hair of rhesus macaques. Gen. Comp. Endocr. 2006, 147, 255–261. [Google Scholar] [CrossRef]
- D’Anna-Hernandez, K.L.; Ross, R.G.; Natvig, C.L.; Laudenslager, M.L. Hair cortisol levels as a retrospective marker of hypothalamic–pituitary axis activity throughout pregnancy: Comparison to salivary cortisol. Physiol. Behav. 2011, 104, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Bévalot, F.; Gaillard, Y.; Lhermitte, M.A.; Pépin, G. Analysis of corticosteroids in hair by liquid chromatography–electrospray ionization mass spectrometry. J. Chromatogr. B 2000, 740, 227–236. [Google Scholar] [CrossRef]
- Tallo-Parra, O.; Manteca, X.; Sabes-Alsina, M.; Carbajal, A.; Lopez-Bejar, M. Hair cortisol detection in dairy cattle by using EIA: Protocol validation and correlation with faecal cortisol metabolites. Animal 2015, 9, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Probst, J.; Neff, A.S.; Hillmann, E.; Kreuzer, M.; Koch-Mathis, M.; Leiber, F. Relationship between stress-related exsanguination blood variables, vocalisation, and stressors imposed on cattle between lairage and stunning box under conventional abattoir conditions. Livest. Sci. 2014, 164, 154–158. [Google Scholar] [CrossRef]
- Comin, A.; Peric, T.; Corazzin, M.; Veronesi, M.; Meloni, T.; Zufferli, V.; Cornacchia, G.; Prandi, A. Hair cortisol as a marker of hypothalamic-pituitary-adrenal axis activation in Friesian dairy cows clinically or physiologically compromised. Livest. Sci. 2013, 152, 36–41. [Google Scholar] [CrossRef]
- Comin, A.; Prandi, A.; Peric, T.; Corazzin, M.; Dovier, S.; Bovolenta, S. Hair cortisol levels in dairy cows from winter housing to summer highland grazing. Livest. Sci. 2011, 138, 69–73. [Google Scholar] [CrossRef]
- Del Rosario, G.D.L.V.; Valdez, R.A.; Lemus-Ramirez, V.; Vázquez-Chagoyán, J.C.; Villa-Godoy, A.; Romano, M.C. Effects of adrenocorticotropic hormone challenge and age on hair cortisol concentrations in dairy cattle. Can. J. Vet. Res. 2011, 75, 216–221. [Google Scholar]
- Burnett, T.A.; Madureira, A.M.; Silper, B.F.; Nadalin, A.; Tahmasbi, A.; Veira, D.M.; Cerri, R.L. Factors affecting hair cortisol concentrations in lactating dairy cows. J. Dairy Sci. 2014, 97, 7685–7690. [Google Scholar] [CrossRef] [PubMed]
- Cerri, R.; Tabmasbi, A.; Veira, D. Hair cortisol concentrations–influence of color and location in Holstein cows. J. Dairy Sci. 2012, 95, 574. [Google Scholar]
- Bennett, A.; Hayssen, V. Measuring cortisol in hair and saliva from dogs: Coat color and pigment differences. Domest. Anim. Endocrinol. 2010, 39, 171–180. [Google Scholar] [CrossRef]
- Duran, M.C.; Janz, D.M.; Waldner, C.L.; Campbell, J.R.; Marques, F.J. Hair Cortisol Concentration as a Stress Biomarker in Horses: Associations with Body Location and Surgical Castration. J. Equine Vet. Sci. 2017, 55, 27–33. [Google Scholar] [CrossRef]
- Casal, N.; Manteca, X.; Peña, L.R.; Bassols, A.; Fàbrega, E. Analysis of cortisol in hair samples as an indicator of stress in pigs. J. Vet. Behav. 2017, 19, 1–6. [Google Scholar] [CrossRef]
- Scorrano, F.; Carrasco, J.; Pastor-Ciurana, J.; Belda, X.; Rami-Bastante, A.; Bacci, M.L.; Armario, A. Validation of the long-term assessment of hypothalamic–pituitary–adrenal activity in rats using hair corticosterone as a biomarker. FASEB J. 2014, 29, 859–867. [Google Scholar] [CrossRef] [PubMed]
- Schell, C.J.; Young, J.K.; Lonsdorf, E.V.; Mateo, J.M.; Santymire, R.M. Investigation of techniques to measure cortisol and testosterone concentrations in coyote hair. Zoo Biol. 2017, 36, 220–225. [Google Scholar] [CrossRef]
- Sotohira, Y.; Suzuki, K.; Sano, T.; Arai, C.; Asakawa, M.; Hayashi, H. Stress assessment using hair cortisol of kangaroos affected by the lumpy jaw disease. J. Vet. Med. Sci. 2017, 79, 852–854. [Google Scholar] [CrossRef] [PubMed]
- Rigalma, K.; Duvaux-Ponter, C.; Barrier, A.; Charles, C.; Ponter, A.; Deschamps, F.; Roussel, S. Medium-term effects of repeated exposure to stray voltage on activity, stress physiology, and milk production and composition in dairy cows. J. Dairy Sci. 2010, 93, 3542–3552. [Google Scholar] [CrossRef] [PubMed]
- Moya, D.; Schwartzkopf-Genswein, K.; Veira, D. Standardization of a non-invasive methodology to measure cortisol in hair of beef cattle. Livest. Sci. 2013, 158, 138–144. [Google Scholar] [CrossRef]
- Creative Research Systems. Available online: www.surveysystem.com/sscalc.htm (accessed on 28 November 2018).
- Peric, T.; Comin, A.; Corazzin, M.; Montillo, M.; Cappa, A.; Campanile, G.; Prandi, A. Hair cortisol concentrations in Holstein-Friesian and crossbreed F1 heifers. J. Dairy Sci. 2013, 96, 3023–3027. [Google Scholar] [CrossRef] [PubMed]
- Edmonson, A.; Lean, I.; Weaver, L.; Farver, T.; Webster, G. A body condition scoring chart for Holstein dairy cows. J. Dairy Sci. 1989, 72, 68–78. [Google Scholar] [CrossRef]
- Thomsen, P.T.; Baadsgaard, N.P. Intra-and inter-observer agreement of a protocol for clinical examination of dairy cows. Prev. Vet. Med. 2006, 75, 133–139. [Google Scholar] [CrossRef]
- Cafe, L.M.; Robinson, D.L.; Ferguson, D.M.; McIntyre, B.L.; Geesink, G.H.; Greenwood, P.L. Cattle temperament: Persistence of assessments and associations with productivity, efficiency, carcass and meat quality traits. J. Anim. Sci. 2011, 89, 1452–1465. [Google Scholar] [CrossRef] [PubMed]
- Whay, H.; Main, D.; Green, L.; Webster, A. Assessment of the welfare of dairy cattle using animal-based measurements: Direct observations and investigation of farm records. Vet. Rec. 2003, 153, 197–202. [Google Scholar] [CrossRef]
- Wechsler, B.; Schaub, J.; Friedli, K.; Hauser, R. Behaviour and leg injuries in dairy cows kept in cubicle systems with straw bedding or soft lying mats. Appl. Anim. Behav. Sci. 2000, 69, 189–197. [Google Scholar] [CrossRef]
- Huxley, J.; Whay, H.R. Welfare: Cow based assessments Part 3: Locomotion scoring, claw overgrowth and injuries associated with farm furniture. UK Vet Livest. 2006, 11, 51–56. [Google Scholar] [CrossRef]
- Kielland, C.; Boe, K.E.; Zanella, A.J.; Osteras, O. Risk factors for skin lesions on the necks of Norwegian dairy cows. J. Dairy Sci. 2010, 93, 3979–3989. [Google Scholar] [CrossRef] [PubMed]
- Coignard, M.; Guatteo, R.; Veissier, I.; des Roches, A.d.B.; Mounier, L.; Lehébel, A.; Bareille, N. Description and factors of variation of the overall health score in French dairy cattle herds using the Welfare Quality® assessment protocol. Prev. Vet. Med. 2013, 112, 296–308. [Google Scholar] [CrossRef]
- Zaaijer, D.; Noordhuizen, J. A novel scoring system for monitoring the relationship between nutritional efficiency and fertility in dairy cows. Ir. Vet. J. 2003, 56, 145–152. [Google Scholar]
- Flower, F.; Weary, D. Effect of Hoof Pathologies on Subjective Assessments of Dairy Cow Gait. J. Dairy Sci. 2006, 89, 139–146. [Google Scholar] [CrossRef]
- Leeb, C.; Main, D.; Whay, H.; Webster, A. Bristol Welfare Assurance Programme–Cattle Assessment. University of Bristol, Bristol, UK. Available online: http://www. vetschool. bris. ac. uk/animal welfare 2004 (accessed on 24 July 2016).
- Popescu, S.; Borda, C.; Sandru, C.D.; Stefan, R.; Lazar, E. The welfare assessment of tied dairy cows in 52 farms in Transylvania using animal-based measurements. Slov. Vet. Res. 2010, 47, 77–82. [Google Scholar]
- Welfare Quality®. Welfare Quality Assessment Protocol for Cattle; Welfare Quality Consortium: Lelystad, The Netherlands, 2009; Available online: http://www.welfarequalitynetwork.net/media/1088/cattle_protocol_without_veal_calves.pdf (accessed on 27 December 2018).
- Chaplin, S.; Munksgaard, L. Evaluation of a simple method for assessment of rising behaviour in tethered dairy cows. Anim. Sci. 2016, 72, 191–197. [Google Scholar] [CrossRef]
- Rousing, T.; Bonde, M.; Badsberg, J.H.; Sørensen, J.T. Stepping and kicking behaviour during milking in relation to response in human–animal interaction test and clinical health in loose housed dairy cows. Livest. Prod. Sci. 2004, 88, 1–8. [Google Scholar] [CrossRef]
- Von Keyserlingk, M.; Barrientos, A.; Ito, K.; Galo, E.; Weary, D. Benchmarking cow comfort on North American freestall dairies: Lameness, leg injuries, lying time, facility design, and management for high-producing Holstein dairy cows. J. Dairy Sci. 2012, 95, 7399–7408. [Google Scholar] [CrossRef] [PubMed]
- Otten, N.; Rousing, T.; Houe, H.; Thomsen, P.T.; Sørensen, J.T. Comparison of animal welfare indices in dairy herds based on different sources of data. Anim. Welf. 2016, 25, 207–215. [Google Scholar] [CrossRef]
- DecibelX Android Phone Application. Available online: https://play.google.com/store/apps/details?id=com.skypaw.decibel&hl+en AU (accessed on 23 August 2018).
- Phillips, C.; Morris, I. The locomotion of dairy cows on floor surfaces with different frictional properties. J. Dairy Sci. 2001, 84, 623–628. [Google Scholar] [CrossRef]
- Phillips, C.J.C. Principles of Cattle Production, 3rd ed.; CABI: Cambridge, MA, USA, 2018; p. 261. [Google Scholar]
- Sharma, A.; Kennedy, U.; Phillips, C. A Novel Method of Assessing Floor Friction in Cowsheds and Its Association with Cow Health. Animals 2019, 9, 120. [Google Scholar] [CrossRef]
- Bartussek, H.; Leeb, C.; Held, S. Animal Needs Index for Cattle (Ani 35 L/2000-cattle); Federal Research Institute for Agriculture: Irdning, Austria, 2000. [Google Scholar]
- Costa, J.; Hötzel, M.; Longo, C.; Balcão, L. A survey of management practices that influence production and welfare of dairy cattle on family farms in southern Brazil. J. Dairy Sci. 2013, 96, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.B. The influence of barn design on dairy cow hygiene, lameness and udder health. In Proceedings of the 35th Annual Convention of American Association of Bovine Practitioners, Madison, WI, USA, 26–28 September 2002. [Google Scholar]
- Regula, G.; Danuser, J.; Spycher, B.; Wechsler, B. Health and welfare of dairy cows in different husbandry systems in Switzerland. Prev. Vet. Med. 2004, 66, 247–264. [Google Scholar] [CrossRef]
- Budithi, N.R.B.; Kumar, V.; Yalla, S.K.; Rai, U.; Umapathy, G. Non-invasive monitoring of reproductive and stress hormones in the endangered red panda (Ailurus fulgens fulgens). Anim. Reprod. Sci. 2016, 172, 173–181. [Google Scholar] [CrossRef]
- Umapathy, G.; Deepak, V.; Kumar, V.; Chandrasekhar, M.; Vasudevan, K. Endocrine Profiling of Endangered Tropical Chelonians Using Noninvasive Fecal Steroid Analyses. Chelonian Conserv. Biol. 2015, 14, 108–115. [Google Scholar] [CrossRef]
- Kumar, V.; Reddy, V.P.; Kokkiligadda, A.; Shivaji, S.; Umapathy, G. Non-invasive assessment of reproductive status and stress in captive Asian elephants in three south Indian zoos. Gen. Comp. Endocr. 2014, 201, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.A.; Hamel, A.F.; Coleman, K.; Lutz, C.K.; Worlein, J.; Menard, M.; Ryan, A.; Rosenberg, K.; Meyer, J.S. Hair loss and hypothalamic–pituitary–adrenocortical axis activity in captive rhesus macaques (Macaca mulatta). J. Am. Assoc. Lab. Anim. Sci. 2014, 53, 261–266. [Google Scholar]
- Kirschbaum, C.; Tietze, A.; Skoluda, N.; Dettenborn, L. Hair as a retrospective calendar of cortisol production—Increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology 2009, 34, 32–37. [Google Scholar] [CrossRef]
- Wester, V.L.; van der Wulp, N.R.P.; Koper, J.W.; de Rijke, Y.B.; van Rossum, E.F.C. Hair cortisol and cortisone are decreased by natural sunlight. Psychoneuroendocrinology 2016, 72, 94–96. [Google Scholar] [CrossRef]
- Acker, M.; Mastromonaco, G.; Schulte-Hostedde, A.I. The effects of body region, season and external arsenic application on hair cortisol concentration. Conserv. Physiol. 2018, 6, coy037. [Google Scholar] [CrossRef]
- Ito, T. Recent advances in the pathogenesis of autoimmune hair loss disease alopecia areata. Clin. Dev. Immunol. 2013, 2013, 348546. [Google Scholar] [CrossRef]
- Ito, T. Hair follicle is a target of stress hormone and autoimmune reactions. J. Dermatol. Sci. 2010, 60, 67–73. [Google Scholar] [CrossRef]
- Cadegiani, F.A.; Kater, C.E. Adrenal fatigue does not exist: A systematic review. BMC Endocr. Disord. 2016, 16, 48. [Google Scholar] [CrossRef]
- Busato, A.; Trachsel, P.; Blum, J. Frequency of traumatic cow injuries in relation to housing systems in Swiss organic dairy herds. J. Vet. Med. A 2000, 47, 221–229. [Google Scholar] [CrossRef]
- Munoz, M.; Bennett, G.; Ahlström, C.; Griffiths, H.; Schukken, Y.; Zadoks, R. Cleanliness scores as indicator of Klebsiella exposure in dairy cows. J. Dairy Sci. 2008, 91, 3908–3916. [Google Scholar] [CrossRef]
- Schreiner, D.; Ruegg, P. Relationship between udder and leg hygiene scores and subclinical mastitis. J. Dairy Sci. 2003, 86, 3460–3465. [Google Scholar] [CrossRef]
- Schubach, K.M.; Cooke, R.F.; Brandão, A.P.; Lippolis, K.D.; Silva, L.G.T.; Marques, R.S.; Bohnert, D.W. Impacts of stocking density on development and puberty attainment of replacement beef heifers. Animal 2017, 11, 2260–2267. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.G.G.; Cockcroft, P.D. (Eds.) Clinical Examination of the skin. In Clinical Examination of Farm; Blackwell Science: Ames, IA, USA, 2008; pp. 16–28. [Google Scholar]
- Broom, D.M.; Fraser, A.F. Domestic Animal Behaviour and Wwelfare, 5th ed.; CAB International: Wallingford, UK, 2015. [Google Scholar]
- Leaver, J.D. Dairy Cattle. In Management and Welfare of Farm Animals—The UFAW Handbook, 4th ed.; Ewbank, R., Kim-Madslien, F., Hart, C.B., Eds.; Universities Federation for Animal Welfare: Wheathampstead, UK, 1999; pp. 495–509. [Google Scholar]
- Ellis, K.A.; Innocent, G.T.; Mihm, M.; Cripps, P.; McLean, W.G.; Howard, C.V.; Grove-White, D. Dairy cow cleanliness and milk quality on organic and conventional farms in the UK. J. Dairy Res. 2007, 74, 302–310. [Google Scholar] [CrossRef]
- Norring, M.; Manninen, E.; de Passillé, A.M.; Rushen, J.; Saloniemi, H. Preferences of dairy cows for three stall surface materials with small amounts of bedding. J. Dairy Sci. 2010, 93, 70–74. [Google Scholar] [CrossRef]
- Aalseth, E. Fresh cow management: What is important, what does it cost, and what does it return? In Proceedings of the 7th Western Dairy Management Conference, Reno, NV, USA, 9–11 March 2005; pp. 1–12. [Google Scholar]
- Hartnell, G.F.; Satter, L.D. Determination of rumen fill, retention time and ruminal turnover rates of ingesta at different stages of lactation in dairy cows. J. Anim. Sci. 1979, 48, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Aitchison, E.; Gill, M.; Dhanoa, M.; Osbourn, D. The effect of digestibility and forage species on the removal of digesta from the rumen and the voluntary intake of hay by sheep. Br. J. Nutr. 1986, 56, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Llamas-Lamas, G.; Combs, D. Effect of forage to concentrate ratio and intake level on utilization of early vegetative alfalfa silage by dairy cows. J. Dairy Sci. 1991, 74, 526–536. [Google Scholar] [CrossRef]
- Oetzel, G.R. Monitoring and testing dairy herds for metabolic disease. Vet. Clin. N. Am. Food Anim. Pract. 2004, 20, 651–674. [Google Scholar] [CrossRef]
- Olmos, G.; Mee, J.F.; Hanlon, A.; Patton, J.; Murphy, J.J.; Boyle, L. Peripartum health and welfare of Holstein-Friesian cows in a confinement-TMR system compared to a pasture-based system. Anim. Welf. 2009, 18, 467–476. [Google Scholar]
- Burfeind, O.; Sepúlveda, P.; von Keyserlingk, M.A.G.; Weary, D.M.; Veira, D.M.; Heuwieser, W. Technical note: Evaluation of a scoring system for rumen fill in dairy cows. J. Dairy Sci. 2010, 93, 3635–3640. [Google Scholar] [CrossRef]
- Esposito, G.; Irons, P.C.; Webb, E.C.; Chapwanya, A. Interactions between negative energy balance, metabolic diseases, uterine health and immune response in transition dairy cows. Anim. Reprod. Sci. 2014, 144, 60–71. [Google Scholar] [CrossRef]
- Dobson, H.; Esslemont, R. Stress and its effects on fertility in dairy cows. Adv. Dairy Technol. 2002, 14, 193–206. [Google Scholar]
- Bergsten, C.; Telezhenko, E.; Ventorp, M. Influence of Soft or Hard Floors before and after First Calving on Dairy Heifer Locomotion, Claw and Leg Health. Animals 2015, 5, 662–686. [Google Scholar] [CrossRef]
- Relun, A.; Lehebel, A.; Chesnin, A.; Guatteo, R.; Bareille, N. Association between digital dermatitis lesions and test-day milk yield of Holstein cows from 41 French dairy farms. J. Dairy Sci. 2013, 96, 2190–2200. [Google Scholar] [CrossRef]
- Nash, C.G.R.; Kelton, D.F.; DeVries, T.J.; Vasseur, E.; Coe, J.; Heyerhoff, J.C.Z.; Bouffard, V.; Pellerin, D.; Rushen, J.; de Passillé, A.M.; et al. Prevalence of and risk factors for hock and knee injuries on dairy cows in tiestall housing in Canada. J. Dairy Sci. 2016, 99, 6494–6506. [Google Scholar] [CrossRef]
- McDowell, R.E. Improvement of Livestock Production in Warm Climates; CABI Publishing: San Francisco, CA, USA, 1972; p. 711. [Google Scholar]
- Satterlee, D.; Roussel, J.; Gomila, L.; Segura, E. Effect of exogenous corticotropin and climatic conditions on bovine adrenal cortical function. J. Dairy Sci. 1977, 60, 1612–1616. [Google Scholar] [CrossRef]
- Wise, M.; Rodriguez, R.; Armstrong, D.; Huber, J.; Wiersma, F.; Hunter, R. Fertility and hormonal responses to temporary relief of heat stress in lactating dairy cows. Theriogenology 1988, 29, 1027–1035. [Google Scholar] [CrossRef]
- Elvinger, F.; Natzke, R.P.; Hansen, P.J. Interactions of Heat Stress and Bovine Somatotropin Affecting Physiology and Immunology of Lactating Cows. J. Dairy Sci. 1992, 75, 449–462. [Google Scholar] [CrossRef]
- Collier, R.; Doelger, S.; Head, H.; Thatcher, W.; Wilcox, C. Effects of Heat Stress during Pregnancy on Maternal Hormone Concentrations, Calf Birth Weight and Postpartum Milk Yield of Holstein Cows. J. Anim. Sci. 1982, 54, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Correa-Calderon, A.; Armstrong, D.; Ray, D.; DeNise, S.; Enns, M.; Howison, C. Thermoregulatory responses of Holstein and Brown Swiss heat-stressed dairy cows to two different cooling systems. Int. J. Biometeorol. 2004, 48, 142–148. [Google Scholar] [CrossRef]
- El-Nouty, F.; Elbanna, I.; Davis, T.P.; Johnson, H. Aldosterone and ADH response to heat and dehydration in cattle. J. Appl. Physiol. 1980, 48, 249–255. [Google Scholar] [CrossRef]
- Wise, M.; Armstrong, D.; Huber, J.; Hunter, R.; Wiersma, F. Hormonal Alterations in the Lactating Dairy Cow in Response to Thermal Stress1. J. Dairy Sci. 1988, 71, 2480–2485. [Google Scholar] [CrossRef]
- Johnson, H.; Li, R.; Manalu, W.; Spencer-Johnson, K.; Becker, B.A.; Collier, R.; Baile, C. Effects of somatotropin on milk yield and physiological responses during summer farm and hot laboratory conditions. J. Dairy Sci. 1991, 74, 1250–1262. [Google Scholar] [CrossRef]
- Arnott, G.; Ferris, C.; O’Connell, N. welfare of dairy cows in continuously housed and pasture-based production systems. Animal 2017, 11, 261–273. [Google Scholar] [CrossRef]
- Chen, J.M.; Stull, C.L.; Ledgerwood, D.N.; Tucker, C.B. Muddy conditions reduce hygiene and lying time in dairy cattle and increase time spent on concrete. J. Dairy Sci. 2017, 100, 2090–2103. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi Nejad, J.; Kim, B.W.; Lee, B.H.; Sung, K.I. Coat and hair color: Hair cortisol and serotonin levels in lactating Holstein cows under heat stress conditions. Anim. Sci. J. 2017, 88, 190–194. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Median | First Quartile (Q1) | Third Quartile (Q3) | Interquartile Range (IQR) |
|---|---|---|---|---|
| Cows/shed | 70 | 47.8 | 137.3 | 89.5 |
| Shed area/cow (m2) | 2.73 | 1.56 | 3.62 | 2.06 |
| % dung in the lying area of the shed | 15.0 | 5.0 | 40.0 | 35.0 |
| % dung in the passages of shed | 10.0 | 5.0 | 42.5 | 37.5 |
| Height of shed eaves (m) | 3.80 | 2.99 | 5.34 | 2.35 |
| Gradient of shed lying area | 1.46 | 0.96 | 2.2 | 1.23 |
| Gradient of shed passages | 2.36 | 1.27 | 3.52 | 2.24 |
| Coefficient of friction of shed flooring (CoF) | 0.43 | 0.27 | 0.65 | 0.37 |
| Shed Luminosity level (lux) | 582 | 89 | 1036 | 946 |
| Shed noise level (decibels) | 27.7 | 21.3 | 37.2 | 15.83 |
| Shed dry bulb reading (°C) | 29.5 | 27.2 | 32.8 | 5.6 |
| Shed wet bulb reading (%) | 34.0 | 24.7 | 45.2 | 20.50 |
| Number of water points in the shelter | 1.0 | 1.0 | 2.0 | 1.0 |
| Percent dung in the yard | 20 | 10 | 40 | 30 |
| Yard area/cow (m2) | 5.9 | 3.6 | 21.5 | 17.9 |
| Coefficient of friction of yard flooring (COFyards) | 0.64 | 0.34 | 0.68 | 0.34 |
| Gradient of the yard flooring (degrees) | 1.51 | 1.13 | 2.43 | 1.30 |
| Nose levels in the yard (decibel) | 25.3 | 20.3 | 33.0 | 12.7 |
| Number of trees in the yard | 2.0 | 0.0 | 6.0 | 6.0 |
| Provision of ad lib water in the yard | 10 | 0.0 | 1.0 | 1.0 |
| Availability of access to yards (h) | 8.0 | 4.0 | 24.0 | 20.0 |
| Frequency of feeding to the cows (times/day) | 3.0 | 2.0 | 3.0 | 1.0 |
| Quantity of fodder provided (kg) | 17.5 | 13.0 | 20.0 | 7.0 |
| Parameter | % Score | |||||
|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | |
| Dirty hind limbs score (Scale 0–3) | 2.41 | 43.3 | 41.8 | 12.4 | - | - |
| Dirty udder score (Scale 0–3) | 21.6 | 42.7 | 28.3 | 7.2 | - | - |
| Dirty flanks score (Scale 0–3) | 22.2 | 39.6 | 31.8 | 6.3 | - | - |
| Body hair loss score (Scale 0–3) | 46.6 | 27.5 | 23.1 | 2.5 | - | - |
| Hock joint swelling score (Scale 0–3) | 38.3 | 33.5 | 26.7 | 1.5 | - | - |
| Hock joint hair loss score (Scale 0–3) | 71.8 | 22.4 | 5.1 | 0.5 | - | - |
| Hock joint ulceration score (Scale 0–3) | 83.7 | 13.1 | 2.9 | 0.1 | - | - |
| Carpal joint injuries score (Scale 0–3) | 44.0 | 32.7 | 22.5 | 0.5 | - | - |
| Neck lesions score (Scale 1–4) | - | 93.5 | 5 | 0.5 | 0.9 | |
| Ocular lesions score (Scale 0–1) | 90 | 10 | - | - | - | - |
| Lesions on the body score (Scale 0–3) | 41.2 | 32.2 | 24.2 | 2.3 | - | - |
| Nasal discharge score (Scale 0–1) | 88.1 | 11.8 | - | - | - | - |
| Diarrhoea score (Scale 0–1) | 96.3 | 3.7 | - | - | - | - |
| Faecal consistency score (Scale 1–5) | - | 0.5 | 4.63 | 35.9 | 57.4 | 1.4 |
| Rumen Fill Score (Scale 1–5) | - | 0.0 | 4.4 | 38.5 | 56.8 | 0.1 |
| Lameness score (Scale 1–5) | - | 85.7 | 9.0 | 3.3 | 1.8 | - |
| Claw overgrowth score (Scale 0–3) | 54.0 | 34.6 | 9.2 | 2.0 | - | - |
| Teat score (Scale 0–5) | 14.6 | 82.4 | 0.9 | 0.3 | 0.0 | 1.6 |
| Ectoparasitism score (Scale 0–4) | 0.1 | 56.3 | 29.8 | 13.5 | 0.1 | - |
| Skin tenting time score (Scale 0–4) | 90.9 | 5.7 | 2.5 | 0.7 | - | - |
| Rising up difficulty score (Scale 1–5) | - | 93.3 | 3.8 | 2.9 | - | - |
| Avoidance Distance score (Scale 0–3) | 72.0 | 20.3 | 5.8 | 1.9 | - | - |
| Animal-Based Parameter | Correlation Coefficient | p-Value |
|---|---|---|
| Dirty hind limbs score | 0.232 | <0.001 |
| Dirty udder score | 0.270 | <0.001 |
| Dirty flanks score | 0.297 | <0.001 |
| Hock joint hair loss score | 0.086 | 0.046 |
| Hock joint ulceration score | 0.213 | <0.001 |
| Carpal joint injuries score | 0.276 | <0.001 |
| Diarrhoea score | 0.152 | <0.001 |
| Rumen fill score | −0.224 | <0.001 |
| Claw overgrowth score | 0.157 | <0.001 |
| Lameness score | 0.177 | <0.001 |
| Lesions on the body score | 0.176 | <0.001 |
| Avoidance distance score | 0.222 | <0.001 |
| Age | 0.111 | 0.012 |
| Rising up difficulty score | 0.270 | <0.001 |
| Lactation | −0.090 | 0.041 |
| Body Condition Score (BCS) | −0.173 | <0.001 |
| Ocular lesions score | 0.100 | 0.023 |
| Nasal discharge score | 0.149 | 0.001 |
| Teat and udder score | 0.169 | <0.001 |
| Resource-Based Parameter | Correlation Coefficient | p-Value |
|---|---|---|
| Shed runoff in the lying area | 0.298 | 0.028 |
| Availability of access to yards | −0.370 | 0.006 |
| Cleaning of the areas in addition to sheds and yards | −0.317 | 0.019 |
| Parameter | Correlation Coefficient | p-Value |
|---|---|---|
| Temperament score | −0.029 | 0.511 |
| Hock joint swelling score | 0.066 | 0.137 |
| Neck lesions score | 0.012 | 0.788 |
| Hampered respiration score | −0.066 | 0.136 |
| Diarrhoea score | 0.040 | 0.366 |
| Vulvar discharge score | 0.056 | 0.209 |
| Faecal consistency score | −0.042 | 0.344 |
| Ectoparasitism score | 0.021 | 0.635 |
| Shed flooring | −0.007 | 0.879 |
| Shed bedding type | 0.044 | 0.319 |
| % dung in the lying area | −0.082 | 0.062 |
| % dung in the passages | 0.003 | 0.947 |
| Presence of urine in shed passages | 0.059 | 0.182 |
| Thickness of bedding | 0.044 | 0.316 |
| Type of yard flooring | 0.061 | 0.166 |
| % dung in the yard | 0.076 | 0.109 |
| Area/cow in the shed | −0.056 | 0.207 |
| Area/cow in the yard | −0.035 | 0.466 |
| Frequency of scrapping the floors | −0.014 | 0.757 |
| Method of floor scrapping | −0.024 | 0.594 |
| Parameter | Coefficient | SE of Coefficent | p-Value | VIF |
|---|---|---|---|---|
| Constant | 0.20 | 0.084 | 0.017 | |
| Dirty flanks | 0.07 | 0.014 | ≤0.001 | 1.46 |
| Body hair loss | −0.06 | 0.018 | 0.001 | 2.47 |
| Hock joint ulceration | 0.03 | 0.015 | 0.04 | 1.12 |
| Carpal joint injuries | 0.04 | 0.013 | 0.002 | 1.21 |
| Rumen fill score | −0.06 | 0.019 | 0.002 | 1.17 |
| Lesions on the body | 0.03 | 0.018 | 0.04 | 2.39 |
| Skin tenting time (s) | 0.08 | 0.025 | ≤0.001 | 1.15 |
| Age of cows (years) | 0.005 | 0.002 | 0.03 | 1.09 |
| Parameter | Coefficient | SE of Coefficent | p-Value | VIF |
|---|---|---|---|---|
| Constant | 6.15 | 0.881 | ≤0.001 | |
| Dung in the lying area of shed (%) | 0.01 | 0.005 | 0.02 | 1.83 |
| Dry bulb temperature in the shed (°C) | −0.15 | 0.029 | 0.001 | 2.00 |
| Duration of access to yards (h/day) | −0.07 | 0.024 | 0.015 | 1.16 |
| Reference | Hair Cortisol Concentration (pg/mg) | Sample Size |
|---|---|---|
| Burnett et al. [18] | 5.7 ± 1.7 | 18 |
| del Rosario et al. [17] | 12.15 ± 1.85 | 5 |
| Moya et al. [27] | 2.35 ± 0.176 | 12 |
| Comin et al. [15] | 2.1 ± 0.10–2.9 ± 0.17 | 83 |
| Comin et al. [16] | 3.29 (0.76–20.41) 5.12 (1.62–28.95) | 257 218 |
| Peric et al. [29] | Holsteins: 5.38 (1.91–27.95) Crossbreds: 4.40 (2.11–41.74) | 142 148 |
| Tallo-Parra et al. [13] | White hair: 2.1 ± 1.10 Black hair: 3.9 ± 1.44 | 17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, A.; Umapathy, G.; Kumar, V.; Phillips, C.J.C. Hair Cortisol in Sheltered Cows and Its Association with Other Welfare Indicators. Animals 2019, 9, 248. https://doi.org/10.3390/ani9050248
Sharma A, Umapathy G, Kumar V, Phillips CJC. Hair Cortisol in Sheltered Cows and Its Association with Other Welfare Indicators. Animals. 2019; 9(5):248. https://doi.org/10.3390/ani9050248
Chicago/Turabian StyleSharma, Arvind, Govindhaswamy Umapathy, Vinod Kumar, and Clive J. C. Phillips. 2019. "Hair Cortisol in Sheltered Cows and Its Association with Other Welfare Indicators" Animals 9, no. 5: 248. https://doi.org/10.3390/ani9050248
APA StyleSharma, A., Umapathy, G., Kumar, V., & Phillips, C. J. C. (2019). Hair Cortisol in Sheltered Cows and Its Association with Other Welfare Indicators. Animals, 9(5), 248. https://doi.org/10.3390/ani9050248







