Effect of Dietary Hemp Seed on Oxidative Status in Sows during Late Gestation and Lactation and Their Offspring
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Treatments
2.2. Blood Sample Collection
2.3. Determination of Bioactive Compounds in Hemp Seed and Diets
2.3.1. Determination of total Polyphenol Content and Antiradical Activity
2.3.2. Determination of Polyphenol and Cannabinoid Profile (LC–MS)
2.3.3. Determination of Polyunsaturated Fatty Acid (PUFA) Profiles
2.4. Measurement of Antioxidant Enzyme Activities (CAT, SOD, Gpx) in Plasma
2.5. Measurement of Plasma Lipid Peroxidation (TBARS)
2.6. Measurement of Plasma Total Antioxidant Capacity (TAC)
2.7. Measurement of Plasma Nitric Oxide Production (NO)
2.8. Measurement of Plasma Reactive Oxygen Species (ROS)
2.9. Statistical Analysis
3. Results
3.1. Bioactive Compounds
3.1.1. Total Polyphenol Content and Antiradical Activity
3.1.2. Polyunsaturated Fatty Acid Profiles
3.1.3. Cannabinoid Profile
3.2. Animal Performance
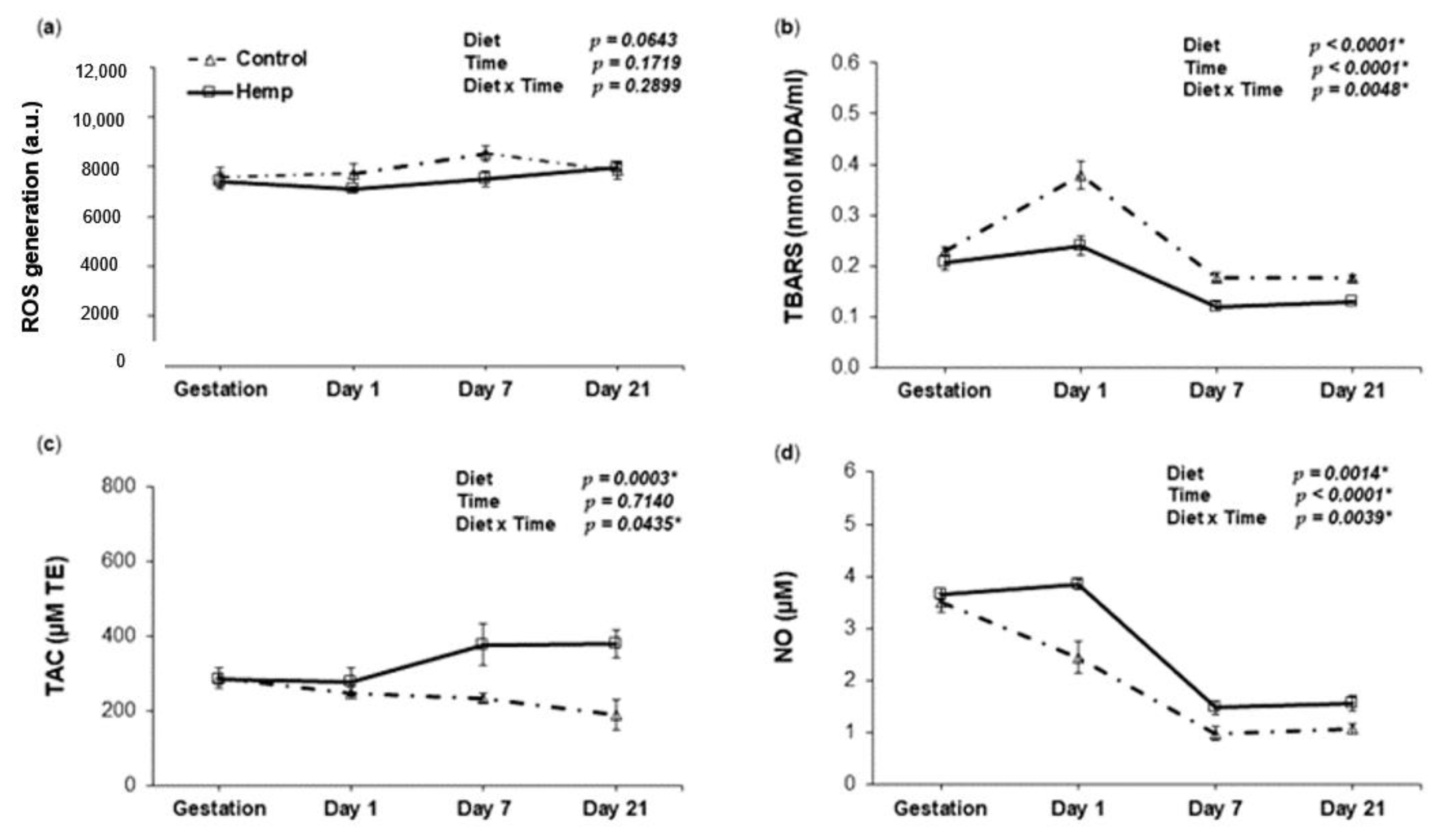
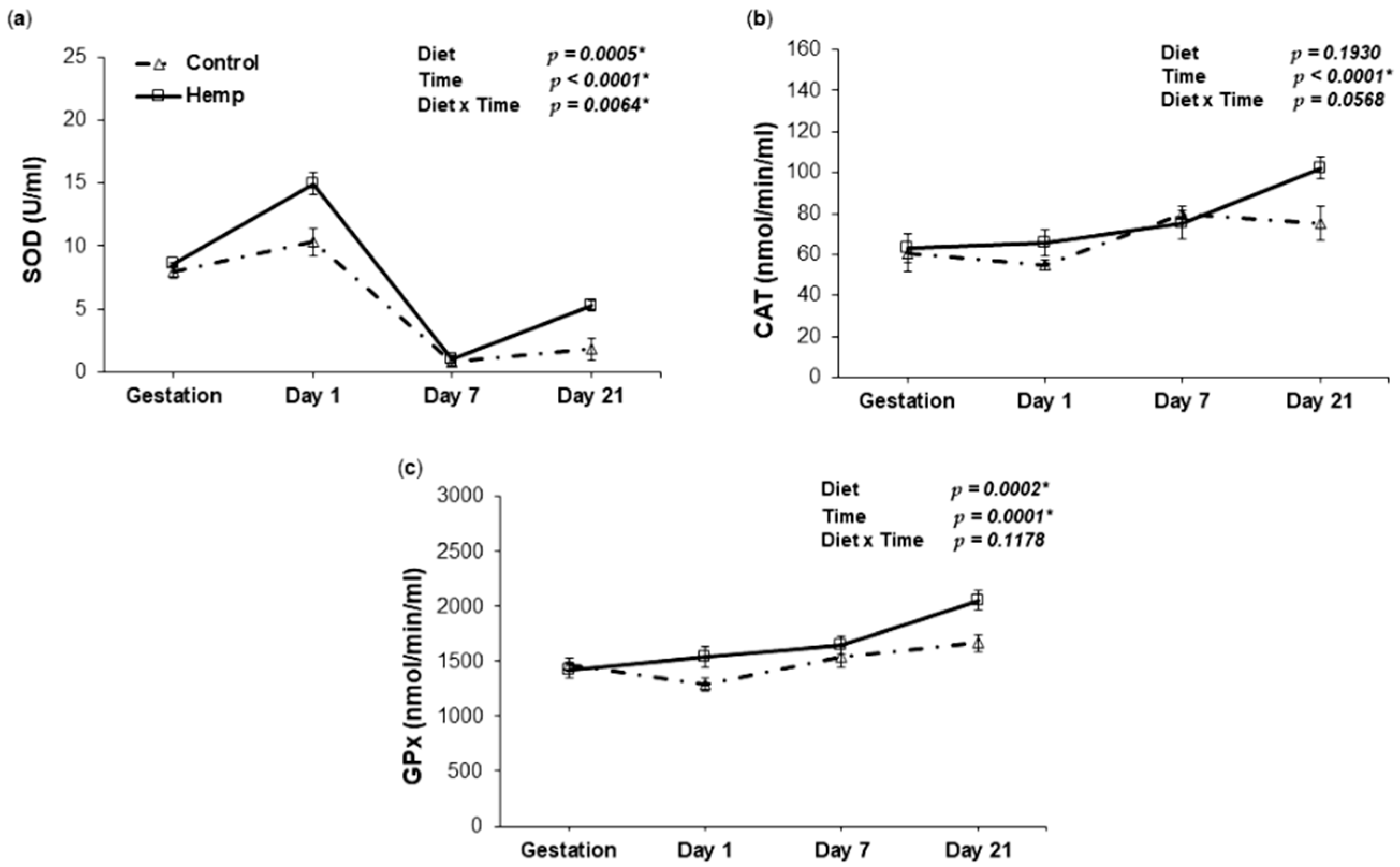
3.3. Effect of Hemp Dietary Intervention on Oxidative Status in Sows
3.3.1. Reactive Oxygen Species, Lipid Peroxidation, Total Antioxidant Capacity and Nitric Oxide
3.3.2. Antioxidant Enzyme Activities
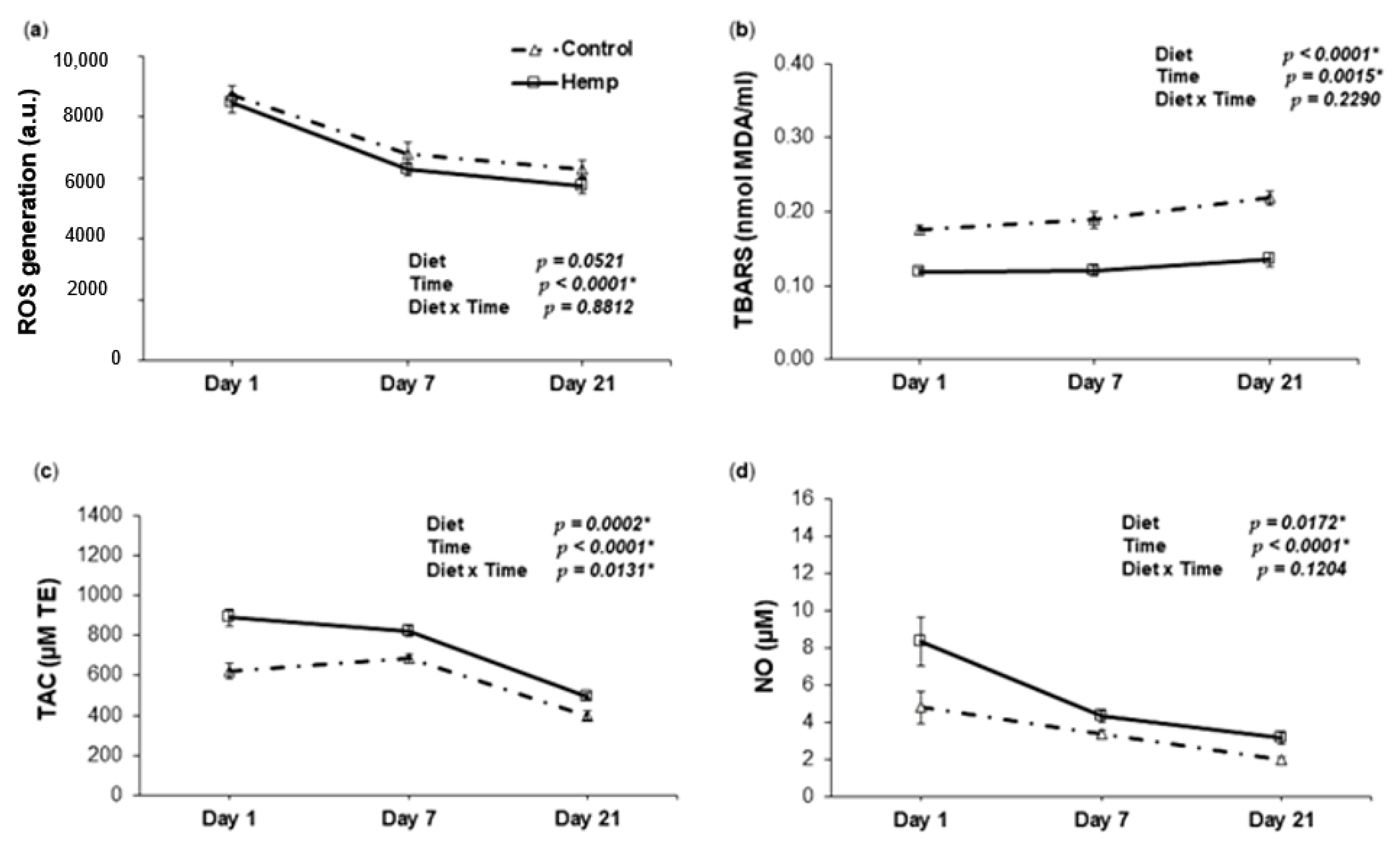
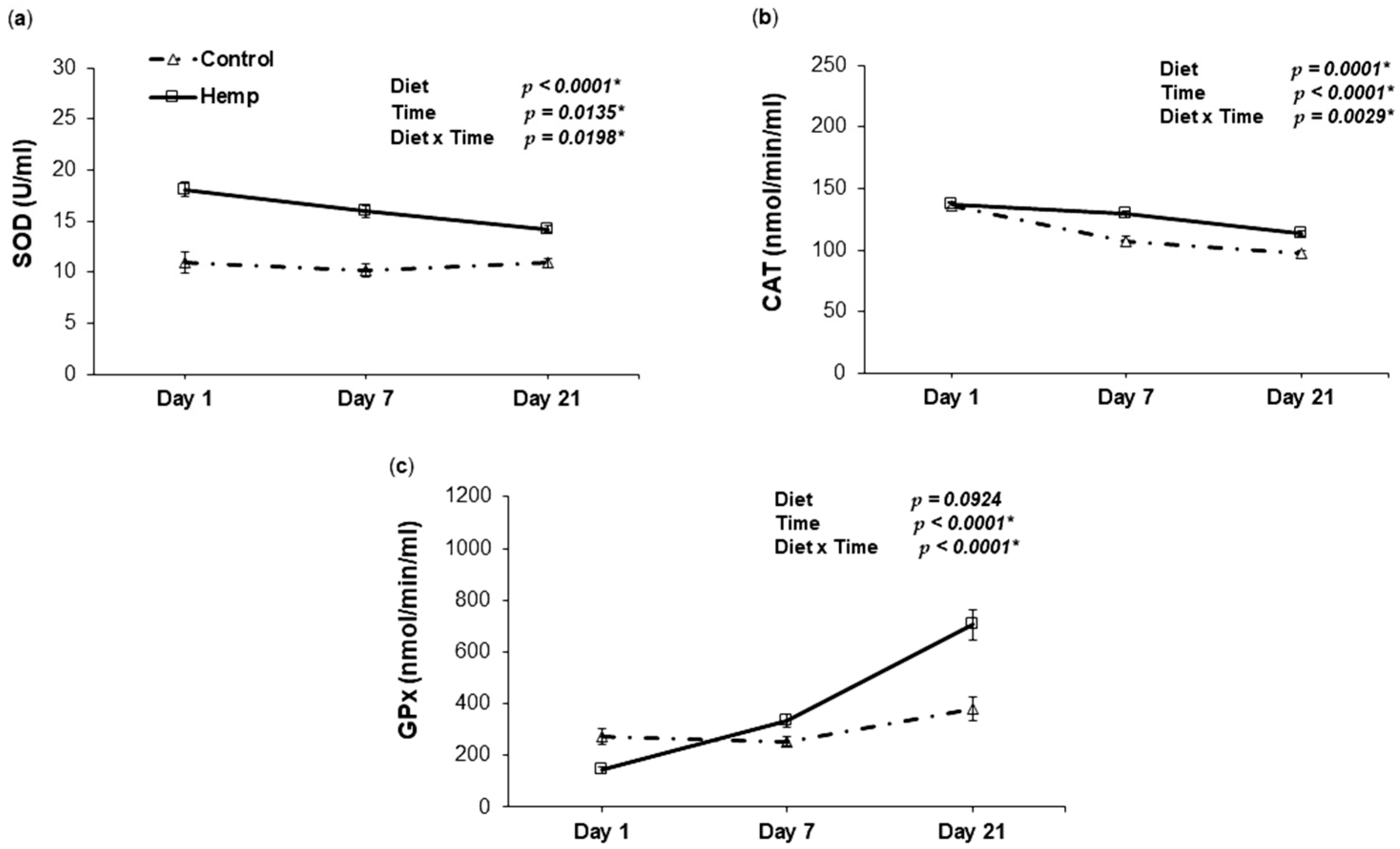
3.4. Effect of Hemp Dietary Intervention on Oxidative Status In Piglets
3.4.1. Reactive Oxygen Species, Lipid Peroxidation, Total Antioxidant Capacity and Nitric Oxide
3.4.2. Antioxidant Enzyme Activities
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Pié, S.; Lallès, J.P.; Blazy, F.; Laffitte, J.; Sève, B.; Oswald, I.P. Weaning is associated with an upregulation of expression of inflammatory cytokines in the intestine of piglets. J. Nutr. 2004, 134, 641–647. [Google Scholar] [CrossRef]
- Buchet, A.; Belloc, C.; Leblanc-Maridor, M.M.; Merlot, E. Effects of age and weaning conditions on blood indicators of oxidative status in pigs. PLoS ONE 2017, 12, e0178487. [Google Scholar] [CrossRef]
- Pluske, J.R. Feed- and feed additives-related aspects of gut health and development in weanling pigs. J. Anim. Sci. Biotechnol. 2013, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Brunton, P.J. Effects of maternal exposure to social stress during pregnancy: Consequences for mother and offspring. Reproduction 2013, 146. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Song, P.; Huang, C.; Rezaei, A.; Farrar, S.; Brown, M.A.; Ma, X. Dietary grape seed proanthocyanidins (GSPs) improve weaned intestinal microbiota and mucosal barrier using a piglet model. Oncotarget 2016, 7, 80313–80326. [Google Scholar] [CrossRef] [PubMed]
- Horn, N.; Miller, G.; Ajuwon, K.M.; Adeola, O. Ability of garlic-derived diallyl disulfide and diallyl trisulfide supplemented by oral gavage to mitigate effects of an acute postweaning feed and water deprivation event in nursery pigs. J. Anim. Sci. 2017, 95, 3579–3590. [Google Scholar] [CrossRef] [PubMed]
- Amagase, H. Clarifying the real bioactive constituents of garlic. J. Nutr. 2006, 136, 716S–725S. [Google Scholar] [CrossRef]
- Lee, H.-S.; Barraza-Villarreal, A.; Hernandez-Vargas, H.; Sly, P.D.; Biessy, C.; Ramakrishnan, U.; Romieu, I.; Herceg, Z. Modulation of DNA methylation states and infant immune system by dietary supplementation with omega-3 PUFA during pregnancy in an intervention study. Am. J. Clin. Nutr. 2013, 98, 480–487. [Google Scholar] [CrossRef]
- Farmer, C.; Giguère, A.; Lessard, M. Dietary supplementation with different forms of flax in late gestation and lactation: Effects on sow and litter performances, endocrinology, and immune response. J. Anim. Sci. 2010, 88, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Hanson, A.R.; Wang, L.; Johnston, L.J.; Baidoo, S.K.; Torrison, J.L.; Chen, C.; Shurson, G.C. Effect of feeding peroxidized dried distillers grains with solubles to sows and progeny on growth performance and metabolic oxidative status of nursery pigs. J. Anim. Sci. 2015, 93, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Chavatte-Palmer, P.; Tarrade, A.; Rousseau-Ralliard, D. Diet before and during pregnancy and offspring health: The importance of animal models and what can be learned from them. Int. J. Environ. Res. Public Health 2016, 13, 586. [Google Scholar] [CrossRef] [PubMed]
- Petrujkic, B.T.; Beier, R.C.; He, H.; Genovese, K.J.; Swaggerty, C.L.; Hume, M.E.; Crippen, T.L.; Harvey, R.B.; Anderson, R.C.; Nisbet, D.J. Nigella sativa L. as an alternative antibiotic feed supplement and effect on growth performance in weanling pigs. J. Sci. Food Agric. 2018, 98, 3175–3181. [Google Scholar] [CrossRef] [PubMed]
- Kantas, D.; Papatsiros, V.G.; Tassis, P.D.; Athanasiou, L.V.; Tzika, E.D. Effect of a natural feed additive (Macleaya cordata), containing sanguinarine, on the performance and health status of weaning pigs. Anim. Sci. J. 2015, 86, 92–98. [Google Scholar] [CrossRef]
- Liu, G.; Guan, G.; Fang, J.; Martínez, Y.; Chen, S.; Bin, P.; Duraipandiyan, V.; Gong, T.; Tossou, M.C.B.; Al-Dhabi, N.A.; et al. Macleaya cordata Extract Decreased Diarrhea Score and Enhanced Intestinal Barrier Function in Growing Piglets. BioMed Res. Int. 2016, 2016, 1069585. [Google Scholar] [CrossRef] [PubMed]
- Devi, S.M.; Lee, S.I.; Kim, I.H. Effect of phytogenics on growth performance, fecal score, blood profiles, fecal noxious gas emission, digestibility, and intestinal morphology of weanling pigs challenged with Escherichia coli K88. Pol. J. Vet. Sci. 2015, 18, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Namkung, H.; Li, J.; Gong, M.; Yu, H.; Cottrill, M.; de Lange, C.F.M. Impact of feeding blends of organic acids and herbal extracts on growth performance, gut microbiota and digestive function in newly weaned pigs. Can. J. Anim. Sci. 2004, 84, 697–704. [Google Scholar] [CrossRef]
- Newton, S.M.; Lau, C.; Gurcha, S.S.; Besra, G.S.; Wright, C.W. The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis. J. Ethnopharmacol. 2002, 79, 57–67. [Google Scholar] [CrossRef]
- Boudry, G.; Douard, V.; Mourot, J.; Lallès, J.-P.; Le Huërou-Luron, I. Linseed oil in the maternal diet during gestation and lactation modifies fatty acid composition, mucosal architecture, and mast cell regulation of the ileal barrier in piglets. J. Nutr. 2009, 139, 1110–1117. [Google Scholar] [CrossRef]
- Tanghe, S.; Millet, S.; De Smet, S. Echium oil and linseed oil as alternatives for fish oil in the maternal diet: Blood fatty acid profiles and oxidative status of sows and piglets. J. Anim. Sci. 2013, 91, 3253–3264. [Google Scholar] [CrossRef]
- Meng, Q.; Guo, T.; Li, G.; Sun, S.; He, S.; Cheng, B.; Shi, B.; Shan, A. Dietary resveratrol improves antioxidant status of sows and piglets and regulates antioxidant gene expression in placenta by Keap1-Nrf2 pathway and Sirt1. J. Anim. Sci. Biotechnol. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Xie, C.; Wu, X.; Long, C.; Wang, Q.; Fan, Z.; Li, S.; Yin, Y. Chitosan oligosaccharide affects antioxidant defense capacity and placental amino acids transport of sows. BMC Vet. Res. 2016, 12, 1–8. [Google Scholar] [CrossRef]
- Hu, Y.J.; Gao, K.G.; Zheng, C.T.; Wu, Z.J.; Yang, X.F.; Wang, L.; Ma, X.Y.; Zhou, A.G.; Jiang, Z.J. Effect of dietary supplementation with glycitein during late pregnancy and lactation on antioxidative indices and performance of primiparous sows. J. Anim. Sci. 2015, 93, 2246–2254. [Google Scholar] [CrossRef]
- Bai, X.M.; Ma, Q.G.; Zhao, L.H.; Xi, L.; Ji, C. Effects of alpha-lipoic acid supplementation on antioxidative ability and performance of sows and nursing piglets. J. Anim. Physiol. Anim. Nutr. (Berl.) 2012, 96, 955–961. [Google Scholar] [CrossRef]
- Lin, S.; Zhang, Y.; Long, Y.; Wan, H.; Che, L.; Lin, Y.; Xu, S.; Feng, B.; Li, J.; Wu, D.; et al. Mammary inflammatory gene expression was associated with reproductive stage and regulated by docosahexenoic acid: In Vitro and in vivo studies. Lipids Health Dis. 2016, 15, 1–8. [Google Scholar] [CrossRef]
- Russo, E.B.; Jiang, H.E.; Li, X.; Sutton, A.; Carboni, A.; Del Bianco, F.; Mandolino, G.; Potter, D.J.; Zhao, Y.X.; Bera, S.; et al. Sen Phytochemical and genetic analyses of ancient cannabis from Central Asia. J. Exp. Bot. 2008, 59, 4171–4182. [Google Scholar] [CrossRef]
- Rodriguez-Leyva, D.; Pierce, G.N. The cardiac and haemostatic effects of dietary hempseed. Nutr. Metab. (Lond.) 2010, 7, 32. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef]
- Galasso, I.; Russo, R.; Mapelli, S.; Ponzoni, E.; Brambilla, I.M.; Battelli, G.; Reggiani, R. Variability in Seed Traits in a Collection of Cannabis sativa L. Genotypes. Front. Plant Sci. 2016, 7, 688. [Google Scholar] [CrossRef]
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar] [CrossRef]
- Jeong, M.; Cho, J.; Shin, J.I.L.; Jeon, Y.J.; Kim, J.H.; Lee, S.J.; Kim, E.S.; Lee, K. Hempseed oil induces reactive oxygen species- and C/EBP homologous protein-mediated apoptosis in MH7A human rheumatoid arthritis fibroblast-like synovial cells. J. Ethnopharmacol. 2014, 154, 745–752. [Google Scholar] [CrossRef]
- Teh, S.-S.; Bekhit, A.; Birch, J. Antioxidative Polyphenols from Defatted Oilseed Cakes: Effect of Solvents. Antioxidants 2014, 3, 67–80. [Google Scholar] [CrossRef]
- Smeds, A.I.; Eklund, P.C.; Willfor, S.M. Content, composition, and stereochemical characterisation of lignans in berries and seeds. Food Chem. 2012, 134, 1991–1998. [Google Scholar] [CrossRef]
- Girgih, A.T.; Alashi, A.; He, R.; Malomo, S.; Aluko, R.E. Preventive and treatment effects of a hemp seed (Cannabis sativa L.) meal protein hydrolysate against high blood pressure in spontaneously hypertensive rats. Eur. J. Nutr. 2014, 53, 1237–1246. [Google Scholar] [CrossRef]
- Berquin, I.M.; Edwards, I.J.; Chen, Y.Q. Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett. 2008, 269, 363–377. [Google Scholar] [CrossRef]
- Rossi, R.; Pastorelli, G.; Cannata, S.; Corino, C. Recent advances in the use of fatty acids as supplements in pig diets: A review. Anim. Feed Sci. Technol. 2010, 162, 1–11. [Google Scholar] [CrossRef]
- Duan, Y.; Li, F.; Li, L.; Fan, J.; Sun, X.; Yin, Y. N-6:n-3 PUFA ratio is involved in regulating lipid metabolism and inflammation in pigs. Br. J. Nutr. 2014, 111, 445–451. [Google Scholar] [CrossRef]
- Goldberg, E.M.; Gakhar, N.; Ryland, D.; Aliani, M.; Gibson, R.A.; House, J.D. Fatty Acid Profile and Sensory Characteristics of Table Eggs from Laying Hens Fed Hempseed and Hempseed Oil. J. Food Sci. 2012, 77, S153–S160. [Google Scholar] [CrossRef]
- Gakhar, N.; Goldberg, E.; Jing, M.; Gibson, R.; House, J.D. Effect of feeding hemp seed and hemp seed oil on laying hen performance and egg yolk fatty acid content: Evidence of their safety and efficacy for laying hen diets. Poult. Sci. 2012, 91, 701–711. [Google Scholar] [CrossRef]
- Montserrat-de la Paz, S.; Marin-Aguilar, F.; Garcia-Gimenez, M.D.; Fernandez-Arche, M.A. Hemp ( Cannabis sativa L.) seed oil: Analytical and phytochemical characterization of the unsaponifiable fraction. J. Agric. Food Chem. 2014, 62, 1105–1110. [Google Scholar] [CrossRef]
- Prociuk, M.A.; Adel, A.L.; Gavel, N.; Deniset, J.; Ganguly, R.; Austria, J.A.; Ander, B.P.; Lukas, A.; Pierce, G.N. The effects of dietary hempseed on cardiac ischemia/reperfusion injury in hypercholesterolemic rabbits. Exp. Clin. Cardiol. 2006, 11, 198–205. [Google Scholar]
- Lefer, D.J. Pharmacology of selectin inhibitors in ischemia/reperfusion states. Annu. Rev. Pharmacol. Toxicol. 2000, 40, 283–294. [Google Scholar] [CrossRef]
- Pini, A.; Mannaioni, G.; Pellegrini-Giampietro, D.; Passani, M.B.; Mastroianni, R.; Bani, D.; Masini, E. The Role of Cannabinoids in Inflammatory Modulation of Allergic Respiratory Disorders, Inflammatory Pain and Ischemic Stroke. Curr. Drug Targets 2012, 13, 984–993. [Google Scholar] [CrossRef]
- Pagano, E.; Capasso, R.; Piscitelli, F.; Romano, B.; Parisi, O.A.; Finizio, S.; Lauritano, A.; Di Marzo, V.; Izzo, A.A.; Borrelli, F. An Orally Active Cannabis Extract with High Content in Cannabidiol attenuates Chemically-induced Intestinal Inflammation and Hypermotility in the Mouse. Front. Pharmacol. 2016, 7, 341. [Google Scholar] [CrossRef]
- Izzo, A.A.; Capasso, R.; Aviello, G.; Borrelli, F.; Romano, B.; Piscitelli, F.; Gallo, L.; Capasso, F.; Orlando, P.; Di Marzo, V. Inhibitory effect of cannabichromene, a major non-psychotropic cannabinoid extracted from Cannabis sativa, on inflammation-induced hypermotility in mice. Br. J. Pharmacol. 2012, 166, 1444–1460. [Google Scholar] [CrossRef]
- Romano, B.; Pagano, E.; Orlando, P.; Capasso, R.; Cascio, M.G.; Pertwee, R.; Di Marzo, V.; Izzo, A.A.; Borrelli, F. Pure Δ9-tetrahydrocannabivarin and a Cannabis sativa extract with high content in Δ9-tetrahydrocannabivarin inhibit nitrite production in murine peritoneal macrophages. Pharmacol. Res. 2016, 113, 199–208. [Google Scholar] [CrossRef]
- Hong, S.; Sowndhararajan, K.; Joo, T.; Lim, C.; Cho, H.; Kim, S.; Kim, G.Y.; Jhoo, J.W. Ethanol and supercritical fluid extracts of hemp seed (Cannabis sativa L.) increase gene expression of antioxidant enzymes in HepG2 cells. Asian Pac. J. Reprod. 2015, 4, 147–152. [Google Scholar] [CrossRef]
- Tan, C.; Wei, H.; Sun, H.; Ao, J.; Long, G.; Jiang, S.; Peng, J. Effects of Dietary Supplementation of Oregano Essential Oil to Sows on Oxidative Stress Status, Lactation Feed Intake of Sows, and Piglet Performance. BioMed Res. Int. 2015, 2015, 9. [Google Scholar] [CrossRef]
- Kim, S.W.; Weaver, A.C.; Shen, Y.B.; Zhao, Y. Improving efficiency of sow productivity: Nutrition and health. J. Anim. Sci. Biotechnol. 2013, 4, 2–9. [Google Scholar] [CrossRef]
- Arikan, S.; Konukoglu, D.; Arikan, C.; Akcay, T.; Davas, I. Lipid peroxidation and antioxidant status in maternal and cord blood. Gynecol. Obstet. Invest. 2001, 51, 145–149. [Google Scholar] [CrossRef]
- Inanc, F.; Kilinc, M.; Kiran, G.; Guven, A.; Kurutas, E.B.; Cikim, I.G.; Akyol, O. Relationship between oxidative stress in cord blood and route of delivery. Fetal Diagn. Ther. 2005, 20, 450–453. [Google Scholar] [CrossRef]
- Girgih, A.T.; Alashi, A.M.; He, R.; Malomo, S.A.; Raj, P.; Netticadan, T.; Aluko, R.E. A novel hemp seed meal protein hydrolysate reduces oxidative stress factors in spontaneously hypertensive rats. Nutrients 2014, 6, 5652–5666. [Google Scholar] [CrossRef] [PubMed]
- Berchieri-Ronchi, C.B.; Kim, S.W.; Zhao, Y.; Correa, C.R.; Yeum, K.-J.; Ferreira, A.L.A. Oxidative stress status of highly prolific sows during gestation and lactation. Animal 2011, 5, 1774–1779. [Google Scholar] [CrossRef] [PubMed]
- Su, G.; Zhao, J.; Luo, G.; Xuan, Y.; Fang, Z.; Lin, Y.; Xu, S.; Wu, D.; He, J.; Che, L. Effects of oil quality and antioxidant supplementation on sow performance, milk composition and oxidative status in serum and placenta. Lipids Health Dis. 2017, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Mahan, D.C.; Peters, J.C. Long-term effects of dietary organic and inorganic selenium sources and levels on reproducing sows and their progeny. J. Anim. Sci. 2004, 82, 1343–1358. [Google Scholar] [CrossRef]
- Umesiobi, D.O. Vitamin e supplementation to sows and effects on fertility rate and subsequent body development of their weanling piglets. J. Agric. Rural Dev. Trop. Subtrop. 2009, 110, 155–168. [Google Scholar]
- Zhan, X.; Qie, Y.; Wang, M.; Li, X.; Zhao, R. Selenomethionine: An effective selenium source for sow to improve se distribution, antioxidant status, and growth performance of pig offspring. Biol. Trace Elem. Res. 2011, 142, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, H.; Jiang, Z.; Zhang, T.; Wang, Y.; Li, Z.; Wu, Y.; Ji, S.; Xiao, S.; Ryffel, B.; et al. Interleukin-33 Increases Antibacterial Defense by Activation of Inducible Nitric Oxide Synthase in Skin. PLoS Pathog. 2014, 10. [Google Scholar] [CrossRef]
- Bondonno, C.P.; Croft, K.D.; Ward, N.; Considine, M.J.; Hodgson, J.M. Dietary flavonoids and nitrate: Effects on nitric oxide and vascular function. Nutr. Rev. 2015, 73, 216–235. [Google Scholar] [CrossRef]
- Baradaran, A.; Nasri, H.; Rafieian-Kopaei, M. Oxidative stress and hypertension: Possibility of hypertension therapy with antioxidants. J. Res. Med. Sci. 2014, 19, 358–367. [Google Scholar]
- MacMicking, J.; Xie, Q.; Nathan, C. Nitric oxide and macrophage function. Annu. Rev. Immunol. 1997, 15, 323–350. [Google Scholar] [CrossRef]
- Beckman, J.S.; Crow, J.P. Pathological implications of nitric oxide, superoxide and peroxynitrite formation. Biochem. Soc. Trans. 1993, 21, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Guo, X.; Long, C.; Fan, Z.; Xiao, D.; Ruan, Z.; Deng, Z.; Wu, X.; Yin, Y. Supplementation of the sow diet with chitosan oligosaccharide during late gestation and lactation affects hepatic gluconeogenesis of suckling piglets. Anim. Reprod. Sci. 2015, 159, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Li, J.; Wang, J.J.; Zhou, W.; Wang, Q.; Zhu, R.; Wang, F.; Thacker, P. Effects of dietary ratio of n-6 to n-3 polyunsaturated fatty acids on immunoglobulins, cytokines, fatty acid composition, and performance of lactating sows and suckling piglets. J. Anim. Sci. Biotechnol. 2012, 3, 43. [Google Scholar] [CrossRef] [PubMed]
- Hăbeanu, M.; Gheorghe, A.; Surdu, I.; Chedea, V.S.; Lefter, N.A.; Stoian, G.; Panait, A.-A.-D.; Beia, I. N-3 PUFA-Enriched Hemp Seed Diet Modifies Beneficially Sow Milk Composition and Piglets’ Performances. Sci. Pap. Ser. Manag. Econ. Eng. Agric. Rural Dev. 2018, 18, 181–190. [Google Scholar]
- TOPIGS Feeding Manual TEMPO 2012. Available online: https://topigsnorsvin.be/wp-content/uploads/2017/12/Manual-Topigs-20-ENG (accessed on 26 June 2017).
- Council, N.R. Nutrient Requirements of Swine: Eleventh Revised Edition; The National Academies Press: Washington, DC, USA, 2012; ISBN 978-0-309-22423-9. [Google Scholar]
- EFSA Panel on Additives and Products or Substances used in Animal Feed, (FEEDAP) Scientific Opinion on the safety of hemp (Cannabis genus) for use as animal feed. EFSA J. 2011, 9, 41. [CrossRef]
- ASRO Romanian Standardization Association. Available online: https://www.asro.ro (accessed on 27 April 2018).
- Arnous, A.; Makris, D.P.; Kefalas, P. Correlation of Pigment and Flavanol Content with Antioxidant Properties in Selected Aged Regional Wines from Greece. J. Food Compos. Anal. 2002, 15, 655–665. [Google Scholar] [CrossRef]
- Habeanu, M.; Lefter, N.; Gheorghe, A.; Nagy, A.; Marin, D.; Ropota, M. Effects of dietary flaxseed oil on the muscle fatty acid composition in Mangalitsa pigs in an extensive rearing system. S. Afr. J. Anim. Sci. 2014, 44, 240–244. [Google Scholar] [CrossRef]
- Amigo-Benavent, M.; Clemente, A.; Caira, S.; Stiuso, P.; Ferranti, P.; del Castillo, M.D. Use of phytochemomics to evaluate the bioavailability and bioactivity of antioxidant peptides of soybean β-conglycinin. Electrophoresis 2014, 35, 1582–1589. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Taranu, I.; Gras, M.; Pistol, G.C.; Motiu, M.; Marin, D.E.; Lefter, N.; Ropota, M.; Habeanu, M. ω-3 PUFA rich Camelina oil by-products improve the systemic metabolism and spleen cell functions in fattening pigs. PLoS ONE 2014, 9, e110186. [Google Scholar] [CrossRef] [PubMed]
- Marin, D.E.; Pistol, G.C.; Neagoe, I.V.; Calin, L.; Taranu, I. Effects of zearalenone on oxidative stress and inflammation in weanling piglets. Food Chem. Toxicol. 2013, 58, 408–415. [Google Scholar] [CrossRef]
- Westhoek, H.; Rood, T.; van den Berg, M.; Janse, J.; Nijdam, D.; Reudink, M.; Stehfest, E. The Protein Puzzle. The Consumption and Production of Meat, Dairy and Fish in the European Union; Wageningen University: Wageningen, The Netherlands, 2011. [Google Scholar]
- Taranu, I.; Habeanu, M.; Gras, M.A.; Pistol, G.C.; Lefter, N.; Palade, M.; Ropota, M.; Sanda Chedea, V.; Marin, D.E. Assessment of the effect of grape seed cake inclusion in the diet of healthy fattening-finishing pigs. J. Anim. Physiol. Anim. Nutr. (Berl.) 2018, 1–13. [Google Scholar] [CrossRef]
- Pastorelli, G.; Rossi, R.; Corino, C. Influence of Lippia citriodora verbascosideon growth performance antioxidant status, and serum immunoglobulins content in piglets. Czech J. Anim. Sci 2012, 57, 312–322. [Google Scholar] [CrossRef]
- Papadopoulos, G.A.; Maes, D.G.D.; Van Weyenberg, S.; van Kempen, T.A.T.G.; Buyse, J.; Janssens, G.P.J. Peripartal feeding strategy with different n-6:n-3 ratios in sows: Effects on sows’ performance, inflammatory and periparturient metabolic parameters. Br. J. Nutr. 2009, 101, 348–357. [Google Scholar] [CrossRef]
- Gessner, D.K.; Gröne, B.; Couturier, A.; Rosenbaum, S.; Hillen, S.; Becker, S.; Erhardt, G.; Reiner, G.; Ringseis, R.; Eder, K. Dietary fish oil inhibits pro-inflammatory and ER stress signalling pathways in the liver of sows during lactation. PLoS ONE 2015, 10, e137684. [Google Scholar] [CrossRef]
- Welling, M.T.; King, G.J.; Liu, L.; Shapter, T.; Raymond, C.A. Characterisation of cannabinoid composition in a diverse Cannabis sativa L. germplasm collection. Euphytica 2015, 208, 463–475. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 31. [Google Scholar] [CrossRef]
- Repetto, M.; Semprine, J.; Boveris, A. Lipid Peroxidation: Chemical Mechanism, Biological Implications and Analytical Determination. J. Free Radic. Biol. Med. 2012, 1, 3–30. [Google Scholar]
- La Marca, M.; Beffy, P.; Pugliese, A.; Longo, V. Fermented wheat powder induces the antioxidant and detoxifying system in primary rat hepatocytes. PLoS ONE 2013, 8, e83538. [Google Scholar] [CrossRef]
- Shen, Y.; Wan, H.; Zhu, J.; Fang, Z.; Che, L.; Xu, S.; Lin, Y.; Li, J.; Wu, D. Fish Oil and Olive Oil Supplementation in Late Pregnancy and Lactation Differentially Affect Oxidative Stress and Inflammation in Sows and Piglets. Lipids 2015, 50, 647–658. [Google Scholar] [CrossRef]
- Lluís, L.; Taltavull, N.; Muñoz-Cortés, M.; Sánchez-Martos, V.; Romeu, M.; Giralt, M.; Molinar-Toribio, E.; Torres, J.L.; Pérez-Jiménez, J.; Pazos, M.; et al. Protective effect of the omega-3 polyunsaturated fatty acids: Eicosapentaenoic acid/Docosahexaenoic acid 1:1 ratio on cardiovascular disease risk markers in rats. Lipids Health Dis. 2013, 12, 1–8. [Google Scholar] [CrossRef]
- Das, U.N. Nutritional factors in the prevention and management of coronary artery disease and heart failure. Nutrition 2015, 31, 283–291. [Google Scholar] [CrossRef]
- Krogh, U.; Oksbjerg, N.; Storm, A.C.; Feyera, T.; Theil, P.K. Mammary nutrient uptake in multiparous sows fed supplementary arginine during gestation and lactation. J. Anim. Sci. 2017, 95, 2517–2532. [Google Scholar] [CrossRef]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef]
- Gyires, K.; Zádori, Z.S. Role of Cannabinoids in Gastrointestinal Mucosal Defense and Inflammation. Curr. Neuropharmacol. 2016, 14, 935–951. [Google Scholar] [CrossRef]
- Giacoppo, S.; Gugliandolo, A.; Trubiani, O.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. Cannabinoid CB2 receptors are involved in the protection of RAW264.7 macrophages against the oxidative stress: An in vitro study. Eur. J. Histochem. 2017, 61, 2749. [Google Scholar] [CrossRef]
- Bekenev, V.; Garcia, A.; Hasnulin, V. Adaptation of piglets using different methods of stress prevention. Animals 2015, 5, 349–360. [Google Scholar] [CrossRef]
- Balakumar, P.; Taneja, G. Fish oil and vascular endothelial protection: Bench to bedside. Free Radic. Biol. Med. 2012, 53, 271–279. [Google Scholar] [CrossRef]
- Lu, H.; Cui, W.; Klaassen, C.D. Nrf2 protects against 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-induced oxidative injury and steatohepatitis. Toxicol. Appl. Pharmacol. 2011, 256, 122–135. [Google Scholar] [CrossRef]
- Li, X.; Han, D.; Tian, Z.; Gao, B.; Fan, M.; Li, C.; Li, X.; Wang, Y.; Ma, S.; Cao, F. Activation of Cannabinoid Receptor Type II by AM1241 Ameliorates Myocardial Fibrosis via Nrf2-Mediated Inhibition of TGF-β1/Smad3 Pathway in Myocardial Infarction Mice. Cell. Physiol. Biochem. 2016, 39, 1521–1536. [Google Scholar] [CrossRef]
- Hochhauser, E.; Lahat, E.; Sultan, M.; Pappo, O.; Waldman, M.; Sarne, Y.; Shainberg, A.; Gutman, M.; Safran, M.; Ben Ari, Z. Ultra Low Dose Delta 9-Tetrahydrocannabinol Protects Mouse Liver from Ischemia Reperfusion Injury. Cell. Physiol. Biochem. 2015, 36, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Rajesh, M.; Pan, H.; Patel, V.; Mukhopadhyay, B.; Batkai, S.; Gao, B.; Hasko, G.; Pacher, P. Cannabinoid-2 receptor limits inflammation, oxidative/nitrosative stress, and cell death in nephropathy. Free Radic. Biol. Med. 2010, 48, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Avramovic, N.; Dragutinovic, V.; Krstic, D.; Colovic, M.; Trbovic, A.; de Luka, S.; Milovanovic, I.; Popovic, T. The effects of omega 3 fatty acid supplementation on brain tissue oxidative status in aged wistar rats. Hippokratia 2012, 16, 241–245. [Google Scholar]
- Lionetti, L.; Cavaliere, G.; Bergamo, P.; Trinchese, G.; De Filippo, C.; Gifuni, G.; Gaita, M.; Pignalosa, A.; Donizzetti, I.; Putti, R.; et al. Diet supplementation with donkey milk upregulates liver mitochondrial uncoupling, reduces energy efficiency and improves antioxidant and antiinflammatory defences in rats. Mol. Nutr. Food Res. 2012, 56, 1596–1600. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, J.; Hakkarainen, J.; Lundeheim, N. Variation in Vitamin E, Glutathione Peroxidase and Retinol Concentrations in Blood Plasma of Primiparous Sows and their Piglets, and in Vitamin E, Selenium and Retinol Contents in Sows’ Milk. Acta Agric. Scand. Sect. A Anim. Sci. 2001, 51, 224–234. [Google Scholar] [CrossRef]
| Sample | Analysis | |||
|---|---|---|---|---|
| TPC (mg GAE/100 g Raw Material) | AAR (µM TE) | |||
| Mean | SEM | Mean | SEM | |
| HS | 1000.05 b | 7.5 | 467.63 b | 3.4 |
| CD | 1178.04 a | 4.1 | 517.17 a | 4.9 |
| HD (5%) | 1146.66 a | 5.5 | 513.83 a | 10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palade, L.M.; Habeanu, M.; Marin, D.E.; Chedea, V.S.; Pistol, G.C.; Grosu, I.A.; Gheorghe, A.; Ropota, M.; Taranu, I. Effect of Dietary Hemp Seed on Oxidative Status in Sows during Late Gestation and Lactation and Their Offspring. Animals 2019, 9, 194. https://doi.org/10.3390/ani9040194
Palade LM, Habeanu M, Marin DE, Chedea VS, Pistol GC, Grosu IA, Gheorghe A, Ropota M, Taranu I. Effect of Dietary Hemp Seed on Oxidative Status in Sows during Late Gestation and Lactation and Their Offspring. Animals. 2019; 9(4):194. https://doi.org/10.3390/ani9040194
Chicago/Turabian StylePalade, Laurentiu Mihai, Mihaela Habeanu, Daniela Eliza Marin, Veronica Sanda Chedea, Gina Cecilia Pistol, Iulian Alexandru Grosu, Anca Gheorghe, Mariana Ropota, and Ionelia Taranu. 2019. "Effect of Dietary Hemp Seed on Oxidative Status in Sows during Late Gestation and Lactation and Their Offspring" Animals 9, no. 4: 194. https://doi.org/10.3390/ani9040194
APA StylePalade, L. M., Habeanu, M., Marin, D. E., Chedea, V. S., Pistol, G. C., Grosu, I. A., Gheorghe, A., Ropota, M., & Taranu, I. (2019). Effect of Dietary Hemp Seed on Oxidative Status in Sows during Late Gestation and Lactation and Their Offspring. Animals, 9(4), 194. https://doi.org/10.3390/ani9040194








