Evaluating the Effects of In Utero Heat Stress on Piglet Physiology and Behavior Following Weaning and Transport
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Gestation and Farrowing
2.2. Transport
2.3. Post-Transportation
2.4. Blood Analyses
2.5. Post-Transport Behavior
2.6. Statistical Analysis
3. Results
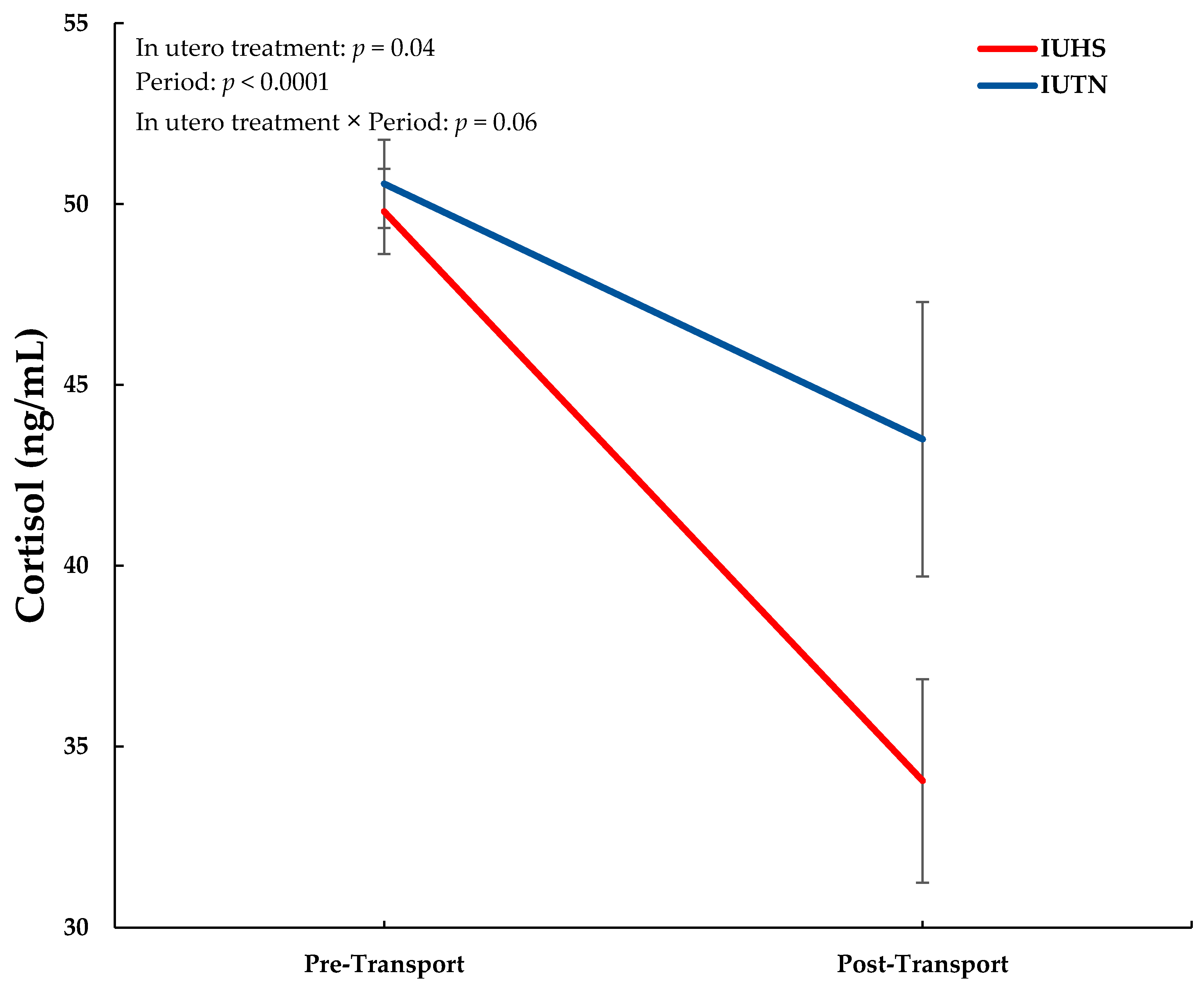
3.1. Blood Analyses
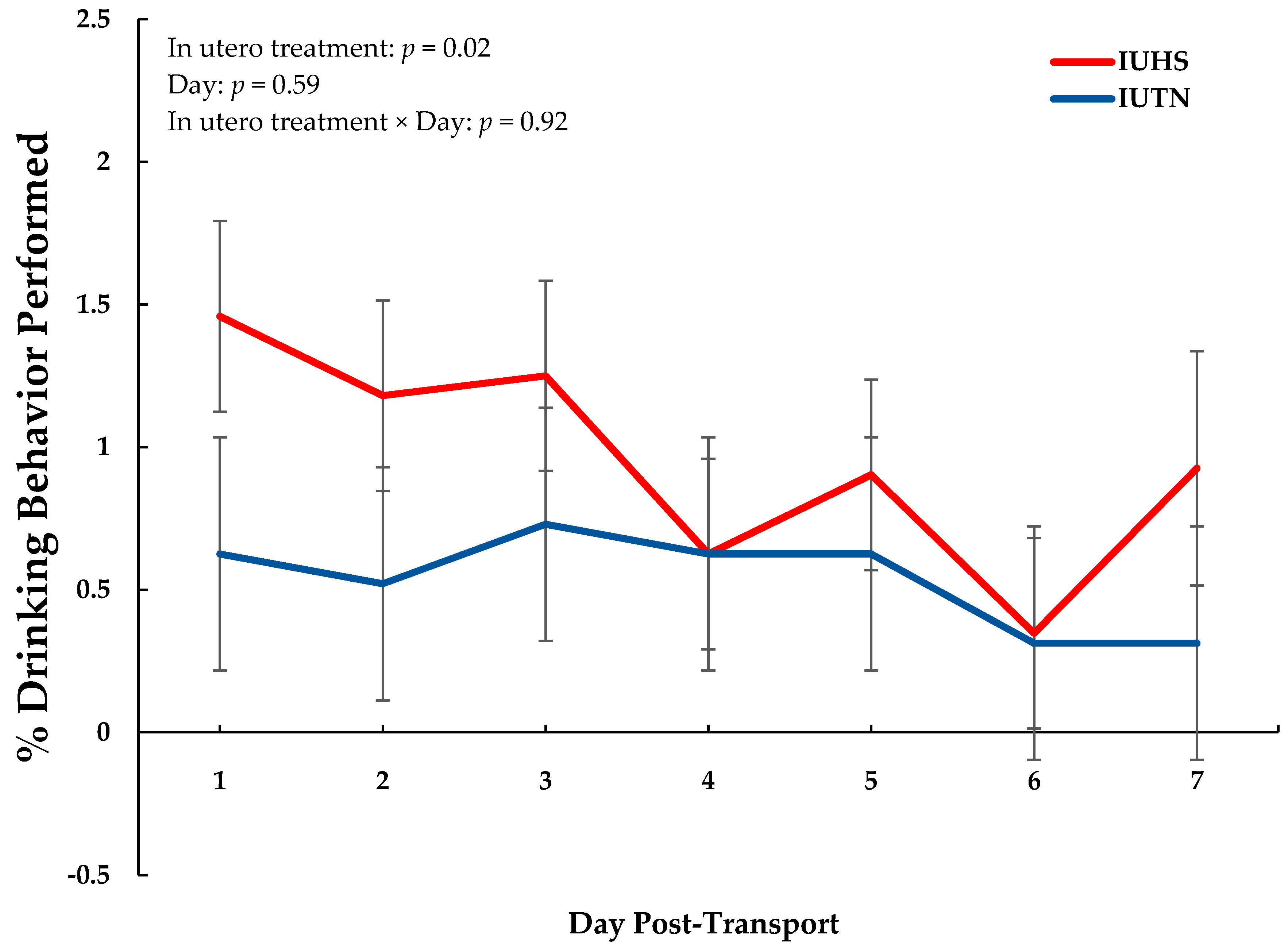
3.2. Behavior
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, N.J. Transport of early weaned piglets. Appl. Anim. Behav. Sci. 2008, 110, 128–135. [Google Scholar] [CrossRef]
- Von Borell, E.; Schäffer, D. Legal requirements and assessment of stress and welfare during transportation and pre-slaughter handling of pigs. Livest. Prod. Sci. 2005, 97, 81–87. [Google Scholar] [CrossRef]
- Berry, R.J.; Lewis, N.J. The effect of duration and temperature of simulated transport on the performance of early-weaned piglets. Can. J. Anim. Sci. 2001, 81, 199–204. [Google Scholar] [CrossRef]
- Wamnes, S.; Lewis, N.J.; Berry, R.J. The behaviour of early-weaned piglets following transport: Effect of season and weaning weight. Can. J. Anim. Sci. 2008, 88, 357–367. [Google Scholar] [CrossRef]
- Colson, V.; Martin, E.; Orgeur, P.; Prunier, A. Influence of housing and social changes on growth, behaviour and cortisol in piglets at weaning. Physiol. Behav. 2012, 107, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Averós, X.; Herranz, A.; Sánchez, R.; Comella, J.X.; Gosalvez, L.F. Serum stress parameters in pigs transported to slaughter under commercial conditions in different seasons. Vet. Med. 2007, 52, 333–342. [Google Scholar] [CrossRef]
- Lewis, N.J.; Berry, R.J. Effects of season on the behaviour of early-weaned piglets during and immediately following transport. Appl. Anim. Behav. Sci. 2006, 100, 182–192. [Google Scholar] [CrossRef]
- Fride, E.; Dan, Y.; Feldon, J.; Halevy, G.; Weinstock, M. Effects of prenatal stress on vulnerability to stress in prepubertal and adult rats. Phys. Behav. 1986, 37, 681–687. [Google Scholar] [CrossRef]
- Fride, E.; Weinstock, M. Prenatal stress increases anxiety related behavior and alters cerebral lateralization of dopamine activity. Life Sci. 1988, 42, 1059–1065. [Google Scholar] [CrossRef]
- Kanitz, E.; Otten, W.; Tuchscherer, M.; Manteuffel, G. Effects of prenatal stress on corticosteroid receptors and monoamine concentrations in limbic areas of suckling pigs (Sus scrofa) at different ages. J. Vet. Med. 2003, 50, 132–139. [Google Scholar] [CrossRef]
- Haussmann, M.F.; Carroll, J.A.; Weesner, G.D.; Daniels, M.J.; Matteri, R.L.; Lay, D.C., Jr. Administration of ACTH to restrained, pregnant sows alters their pigs’ hypothalamic-pituitary-adrenal (HPA) axis. J. Anim. Sci. 2000, 78, 2399–2411. [Google Scholar] [CrossRef]
- Otten, W.; Kanitz, E.; Tuchscherer, M. Prenatal stress in pigs: Effects on growth, physiological stress reactions and immune function. Arch. Tierz. 2000, 43, 159–164. [Google Scholar]
- Lay, D.C., Jr.; Kattesh, H.G.; Cunnick, J.E.; Daniels, M.J.; McMunn, K.A.; Toscano, M.J.; Roberts, M.P. Prenatal stress effects on pig development and response to weaning. J. Anim. Sci. 2008, 86, 1316–1324. [Google Scholar] [CrossRef]
- Kranendonk, G.; Hopster, H.; Fillerup, M.; Ekkel, E.D.; Mulder, E.J.H.; Taverne, M.A.M. Cortisol administration to pregnant sows affects novelty-induced locomotion, aggressive behaviour, and blunts gender differences in their offspring. Horm. Behav. 2006, 49, 663–672. [Google Scholar] [CrossRef]
- Otten, W.; Kanitz, E.; Tuchscherer, M.; Puppe, B.; Nürnberg, G. Repeated administrations of adrenocorticotropic hormone during gestation in gilts: Effects of growth, behaviour, and immune responses of their piglets. Livest. Sci. 2007, 106, 261–270. [Google Scholar] [CrossRef]
- Brajon, S.; Ringgenberg, N.; Torrey, S.; Bergeron, R.; Devilliers, N. Impact of prenatal stress and environmental enrichment prior to weaning on activity and social behaviour of piglets (Sus scrofa). Appl. Anim. Behav. Sci. 2017, 197, 15–23. [Google Scholar] [CrossRef]
- Jarvis, S.; Moinard, C.; Robson, S.K.; Baxter, E.; Ormandy, E.; Douglas, A.J.; Seckl, J.R.; Russell, J.A.; Lawrence, A.B. Programming the offspring of the pig by prenatal social stress: Neuroendocrine activity and behaviour. Horm. Behav. 2006, 49, 68–80. [Google Scholar] [CrossRef]
- Nienaber, J.A.; Hahn, G.L. Livestock production system management responses to thermal challenges. Int. J. Biometeorol. 2007, 52, 149–157. [Google Scholar] [CrossRef]
- Collins, M.; Knutti, R.; Arblaster, J.; Dufresne, J.-L.; Fichefet, T.; Friedlingstein, P.; Gao, X.; Gutowski, W.J.; Johns, T.; Krinner, G.; et al. Long-Term Climate Change: Projections, Commitments and Irreversibility. In Climate Change 2013: The Physical Science Basis; Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Johnson, J.S.; Baumgard, L.H. Postnatal consequences of prenatal heat stress in pigs. J. Anim. Sci. 2018, 97, 962–971. [Google Scholar] [CrossRef]
- Johnson, J.S.; Sanz-Fernandez, M.V.; Seibert, J.T.; Ross, J.W.; Lucy, M.C.; Safranski, T.J.; Elasser, T.H.; Kahl, S.; Rhoads, R.P.; Baumgard, L.H. In utero heat stress increases postnatal core body temperature in pigs. J. Anim. Sci. 2015, 93, 4312–4322. [Google Scholar] [CrossRef]
- Cruzen, S.M.; Boddicker, R.L.; Graves, K.L.; Johnson, T.P.; Arkfeld, E.K.; Baumgard, L.H.; Ross, J.W.; Safranski, T.J.; Lucy, M.C.; Lonergan, S.M. Carcass composition of market weight pigs subjected to heat stress in utero and during finishing. J. Anim. Sci. 2015, 93, 2587–8347. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.S.; Sanz-Fernandez, M.V.; Patience, J.F.; Ross, J.W.; Gabler, N.K.; Lucy, M.C.; Safranski, T.J.; Rhoads, R.P.; Baumgard, L.H. Effects of in utero heat stress on postnatal body composition in pigs: II. Finishing phase. J. Anim. Sci. 2015, 93, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Chapel, N.M.; Byrd, C.J.; Lugar, D.W.; Morello, G.M.; Baumgard, L.H.; Ross, J.W.; Safranski, T.J.; Lucy, M.C.; Johnson, J.S. Determining the effects of early gestation in utero heat stress on postnatal fasting heat production and circulating biomarkers associated with metabolism in growing pigs. J. Anim. Sci. 2017, 95, 3914–3921. [Google Scholar] [CrossRef] [PubMed]
- Boddicker, R.L.; Seibert, J.T.; Johnson, J.S.; Pearce, S.C.; Selsby, J.T.; Gabler, N.K.; Lucy, M.C.; Safranski, T.J.; Rhoads, R.P.; Baumgard, L.H.; et al. Gestational heat stress alters postnatal offspring body composition indices and metabolic parameters in pigs. PLoS ONE 2014, 9, e110859. [Google Scholar] [CrossRef] [PubMed]
- Lugar, D.W.; Proctor, J.A.; Safranski, T.J.; Lucy, M.C.; Stewart, K.R. In utero heat stress causes reduced testicular area at puberty, reduced total sperm production, and increased sperm abnormalities in boars. Anim. Reprod. Sci. 2018, 192, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Bernhard, C.J.; Safranski, T.J.; Lucy, M.C.; Lamberson, W.R.; Moore, S.G.; Mayo, L.M.; Molina-Coto, R. Development of the fetus and fetal reproductive tract in gilts subjected to heat stress from week 4 to 8 of gestation. J. Anim. Sci. 2016, 94 (Suppl. 5), 557. [Google Scholar] [CrossRef]
- Federation of Animal Science Societies. Guide for the Care and Use of Agricultural Animals in Research and Teaching; Federation of Animal Science Societies: Champaign, IL, USA, 2010; ISBN 978-1-884706-11-0. [Google Scholar]
- National Research Council (NRC). Nutrient Requirements of Swine, 11th rev. ed.; Natl. Acad. Press: Washington, DC, USA, 2012. [Google Scholar] [CrossRef]
- Mina, T.H.; Reynolds, R.M. Mechanisms linking in utero stress to altered offspring behaviour. Curr. Top. Behav. Neurosci. 2014, 18, 93–122. [Google Scholar] [CrossRef]
- Otten, W.; Kanitz, E.; Tuchscherer, M.; Brüssow, K.-P.; Nürnberg, G. Repeated administration of adrenocorticotropic hormone during late gestation in pigs: Maternal cortisol response and effects on fetal HPA axis and brain neurotransmitter systems. Theriogenology 2008, 69, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Collier, R.J.; Doelger, S.G.; Head, H.H.; Thatcher, W.W.; Wilcox, C.J. Effects of heat stress during pregnancy on maternal hormone concentrations, calf birth weight and postpartum milk yield of Holstein cows. J. Anim. Sci. 1982, 54, 309–319. [Google Scholar] [CrossRef]
- Baumgard, L.H.; Rhoads, R.P. Effects of heat stress on post-absorptive metabolism and energetics. Annu. Rev. Anim.Biosci. 2013, 1, 311–337. [Google Scholar] [CrossRef]
- Rhoads, R.P.; Baumgard, R.M.; Suagee, J.K.; Sanders, S.R. Nutritional interventions to alleviate the negative consequences of heat stress. Adv. Nutr. 2013, 4, 267–276. [Google Scholar] [CrossRef]
- Kanitz, E.; Otten, W.; Tuchscherer, M. Changes in endocrine and neurochemical profiles in neonatal pigs prenatally exposed to increased maternal cortisol. J. Endocrinol. 2006, 191, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Collier, C.T.; Williams, P.N.; Carroll, J.A.; Welsh, T.H., Jr.; Laurenz, J.C. Effect of maternal restraint-stress during gestation on temporal lipopolysaccharide-induced neuroendocrine and immune responses of progeny. Domest. Anim. Endocrinol. 2011, 40, 40–50. [Google Scholar] [CrossRef]
- Marchant-Forde, J.N.; Matthews, D.L.; Poletto, R.; McCain, R.R.; Mann, D.D.; DeGraw, R.T.; Hampsch, J.M.; Peters, S.; Knipp, G.T.; Kissinger, C.B. Plasma cortisol and noradrenalin concentrations in pigs: Automated sampling of freely moving pigs housed in the PigTurn® versus manually sampled and restrained pigs. Anim. Welf. 2012, 21, 197–205. [Google Scholar] [CrossRef]
- Parrott, R.; Bradshaw, R.H.; Lloyd, D.M.; Goode, J.A. Effects of transport and indomethacin on telemetered body temperature release of cortisol and prolactin in pre-pubertal pigs. Res. Vet. Sci. 1998, 64, 51–55. [Google Scholar] [CrossRef]
- Sutherland, M.; Bryer, P.J.; Davis, B.L.; McGlone, J.J. Space requirements of weaned pigs during a sixty-minute transport in summer. J. Anim. Sci. 2009, 87, 363–370. [Google Scholar] [CrossRef]
- Sutherland, M.; Bryer, P.J.; Davis, B.L.; Smith, J.F.; McGlone, J.J. The combined effects of transport and foot and water deprivation on the physiology of breeding age gilts. Livest. Sci. 2012, 144, 124–131. [Google Scholar] [CrossRef]
- Averós, X.; Herranz, A.; Sánchez, R.; Gosálvez, L.F. Effect of the duration of commercial journeys between rearing farms and growing-finishing farms on the physiological stress response of weaned piglets. Livest. Sci. 2009, 122, 339–344. [Google Scholar] [CrossRef]
- Bradshaw, R.H.; Parrott, R.F.; Goode, J.A.; Lloyd, D.M.; Rodway, R.G.; Broom, D.M. Behavioural and hormonal responses of pigs during transport: Effect of mixing and duration of journey. J. Anim. Sci. 1996, 62, 547–554. [Google Scholar] [CrossRef]
- McGrane, M.M. Carbohydrate metabolism: Synthesis and oxidation. In Biochemical, Physiological, and Molecular Aspects of Human Nutrition; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Inc.: St. Louis, MO, USA, 2013; pp. 209–255. ISBN 0323441815. [Google Scholar]
- Lawrence, A.B.; Terlouw, E.M. A review of behavioral factors involved in the development and continued performance of stereotypic behaviors in pigs. J. Anim. Sci. 1993, 71, 2815–2825. [Google Scholar] [CrossRef]
- Dantzer, R. Stress, stereotypies, and welfare. Behav. Process. 1991, 25, 95–102. [Google Scholar] [CrossRef]
- Horback, K. Nosing around: Play in pigs. ABC 2014, 1, 186–196. [Google Scholar] [CrossRef]
- Bergeron, R.; Badnell-Waters, A.J.; Lambton, S.; Mason, G. Stereotypic oral behavior in captive ungulates: Foraging, diet and gastrointestinal function. In Stereotypic Animal Behavior Fundamentals and Applications to Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CABI: Oxfordshire, UK, 2006; pp. 19–57. ISBN 1-84593-042-8. [Google Scholar]
- Johnson, J.S.; Lay, D.C., Jr. Evaluating the behavior, growth performance, immune parameters, and intestinal morphology of weaned piglets after simulated transport and heat stress when antibiotics are eliminated from the diet or replaced with L-glutamine. J. Anim. Sci. 2017, 95, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Rushen, J.; De Passille, A.M.B.; Schouten, W. Stereotypic behavior, endogenous opoids, and postfeeding hypoalgesia in pigs. Physiol. Behav. 1990, 48, 91–96. [Google Scholar] [CrossRef]
- Rushen, J. Stereotyped behaviour, adjunctive drinking and the feeding periods of tethered sows. Anim Behav. 1984, 32, 1059–1067. [Google Scholar] [CrossRef]
- Otten, W.; Kanitz, E.; Tuchscherer, M. The impact of pre-natal stress on offspring development in pigs. J. Agric. Sci. 2015, 153, 907–919. [Google Scholar] [CrossRef]
- Gotlib, I.H.; Joorman, J.; Minor, K.L.; Hallmayer, J. HPA axis reactivity: A mechanism underlying the associations among serotonin TLPR stress, and depression. Biol. Psychiatry. 2008, 63, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Ison, S.H.; D’Eath, R.B.; Robson, S.K.; Baxter, E.M.; Ormandy, E.; Douglas, A.J.; Russell, J.A.; Lawrence, A.B.; Jarvis, S. ‘Subordination style’ in pigs? The response of pregnant sows to mixing stress affects their offspring’s behaviour and stress reactivity. Appl. Anim. Behav. Sci. 2010, 124, 16–27. [Google Scholar] [CrossRef]
- Lay, D.C., Jr.; Kattesh, H.G.; Cunnick, J.E.; Daniels, M.J.; Kranendonk, G.; McMunn, K.A.; Toscano, M.J.; Roberts, M.P. Effect of prenatal stress on subsequent response to mixing stress and a lipopolysaccharide challenge in pigs. J. Anim. Sci. 2011, 89, 1787–1794. [Google Scholar] [CrossRef]
- D’Souza, D.N.; Dunshea, F.R.; Leury, B.J.; Warner, R.D. Effect of mixing boars during lairage and pre-slaughter handling on pork quality. Aust. J. Agric. Res. 1999, 50, 109–113. [Google Scholar] [CrossRef]
- Allrich, R.D.; Christenson, R.K.; Ford, J.J.; Zimmerman, D.R. Pubertal development of the boar: Testosterone, estradiol-17β, cortisol and LH concentrations before and after castration at various ages. J. Anim. Sci. 1982, 55, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Giersing, M.; Ladewig, J.; Forkman, B. Animal welfare aspects of preventing boar taint. Acta. Vet. Scand. Suppl. 2006, 48, S3. [Google Scholar] [CrossRef]
- Quiniou, N.; Courboulay, V.; Salaün, Y.; Chevillon, P. Impact of the Non Castration of Male Pigs on Growth Performance and Behaviour-Comparison with Barrows and Gilts. In Proceedings of the 61st Annual Meeting of the European Association for Animal Production, Heraklion, Greece, 23–27 August 2010; pp. 1–5. [Google Scholar]
- Moya, S.L.; Boyle, L.A.; Lynch, P.B.; Arkins, S. Effects of surgical castration on the behavioural and physiological responses of weaned pigs. Acta Vet. Scand. Suppl. 2006, 48, P4. [Google Scholar] [CrossRef]
| Behavior | Description |
|---|---|
| Standing | Piglet is upright with all legs extended perpendicular to the ground. All feet are in contact with the ground. |
| Sitting | Piglet is upright with front legs extended perpendicular to the ground. Rear legs are not extended. All four feet are in contact with the ground. |
| Lying | Piglet is sternal or lateral on the ground. Legs may be extended but are parallel to the ground. Feet may be in contact with the ground but do not support the weight of the piglet. |
| Huddling | Both piglets are lying approximately parallel with at least ¾ body length in contact with one another. |
| Eating | Piglet is standing at feeder, ingesting feed. |
| Drinking | Piglet is standing at nipple drinker, ingesting water. |
| Aggression | Classified as one of three criteria, all resulting in retreat of the other pen mate:
|
| Interaction with enrichment (straw) | Piglet is using one or two feet to scratch, move, or manipulate straw 1. |
| Parameter | Pre-Transport | SE | Post-Transport | SE | p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| IUTN | IUHS | IUTN | IUHS | Trt 1 | Period 2 | Trt × Period 3 | |||
| NEFA (mEq/L) | 777.4 | 665.7 | 232.7 | 1058.7 | 788.4 | 231.9 | 0.08 | 0.009 | 0.25 |
| Insulin (ng/mL) | 0.030 a | 0.023 | 0.003 | 0.022 b | 0.023 | 0.003 | 0.48 | 0.002 | 0.003 |
| Glucose (mg/dL) | 144.3 | 130.5 | 6.3 | 112.7 | 102.0 | 6.6 | 0.17 | <0.001 | 0.70 |
| Behavior | IUTN 1 | IUHS 2 | SE 3 | P-Value |
|---|---|---|---|---|
| Standing | 33.80 | 29.70 | 5.20 | 0.59 |
| Lying | 65.80 | 69.70 | 5.10 | 0.60 |
| Huddling | 46.20 | 52.10 | 5.60 | 0.48 |
| Eating 1 | 5.12 | 4.31 | 2.60 | 0.83 |
| Drinking | 0.54 | 0.96 | 0.09 | 0.02 |
| Aggression | 0.48 | 0.80 | 0.1 | 0.07 |
| Interaction with straw | 1.06 | 0.71 | 0.17 | 0.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byrd, C.J.; Anderson, N.C.; Lugar, D.W.; Safranski, T.J.; Lucy, M.C.; Johnson, J.S. Evaluating the Effects of In Utero Heat Stress on Piglet Physiology and Behavior Following Weaning and Transport. Animals 2019, 9, 191. https://doi.org/10.3390/ani9040191
Byrd CJ, Anderson NC, Lugar DW, Safranski TJ, Lucy MC, Johnson JS. Evaluating the Effects of In Utero Heat Stress on Piglet Physiology and Behavior Following Weaning and Transport. Animals. 2019; 9(4):191. https://doi.org/10.3390/ani9040191
Chicago/Turabian StyleByrd, Christopher J., Nichole Chapel Anderson, Drew W. Lugar, Timothy J. Safranski, Matthew C. Lucy, and Jay S. Johnson. 2019. "Evaluating the Effects of In Utero Heat Stress on Piglet Physiology and Behavior Following Weaning and Transport" Animals 9, no. 4: 191. https://doi.org/10.3390/ani9040191
APA StyleByrd, C. J., Anderson, N. C., Lugar, D. W., Safranski, T. J., Lucy, M. C., & Johnson, J. S. (2019). Evaluating the Effects of In Utero Heat Stress on Piglet Physiology and Behavior Following Weaning and Transport. Animals, 9(4), 191. https://doi.org/10.3390/ani9040191





