3.1. Growth Performance
The feeding trials were conducted over a period of ten days. Larvae were sampled and weighed at the start of the test (Day 0) and on Days 2, 3, 4, 5, 6, 7, 8, 9, and 10, respectively. On Day 10, the larval and residue yield were also calculated.
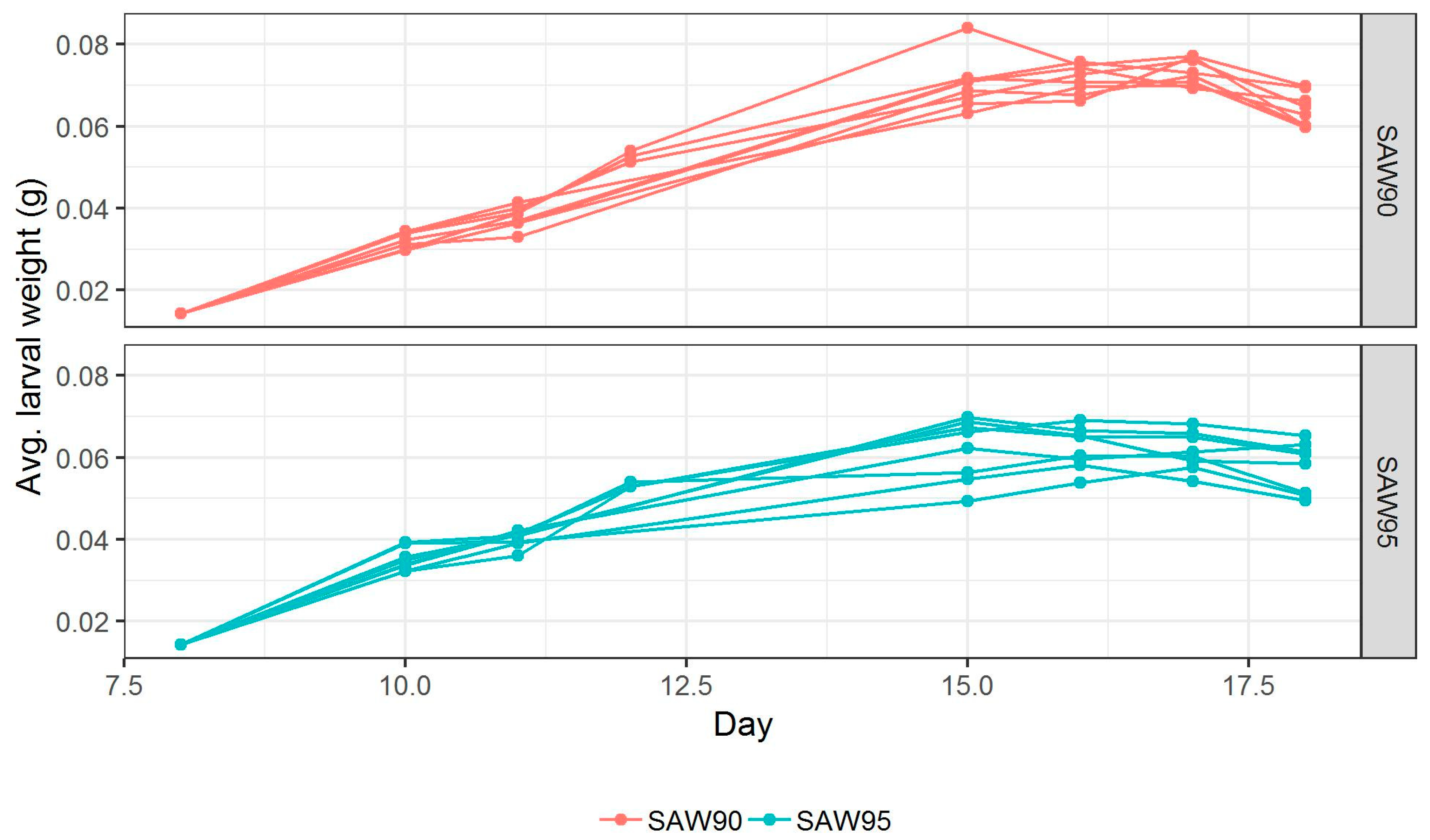
Figure 3 shows the growth curves for 16 crates studied in the present work. Growth of larvae fed with two SAW mixes was more or less the same before day 7. On Days 7, 8, and 9, the biomass of larvae fed on SAW 90 mix reached higher values than that of the larvae fed SAW 95 mix. From
Figure 3, it is also visible that the average larval weights fed with two recipes diverge the most on Day 9. The difference in average weight on Day 9 was estimated using linear regression, showing that SAW 90 mix fed larvae were heavier than SAW 95 mix fed larvae by approximately 0.012 g (
p-value < 0.001). On Day 8, the difference was also significant (
p-value < 0.001), but smaller at 0.009 g. These results might be due to the crude protein content or if the protein conversion factor is inaccurate then the nitrogen present in the feeding media, which was higher in SAW 90 (21.4% of DM) than SAW 95 (12.9% of DM). Between Day 9 and 10, the larvae are not fed, which is responsible for general weight loss. This phenomenon also results in the re-convergence of the weights of larvae from two SAW mixes. This weight loss is due to dehydration and starvation. Pupation had begun, at which point the animals stop eating.
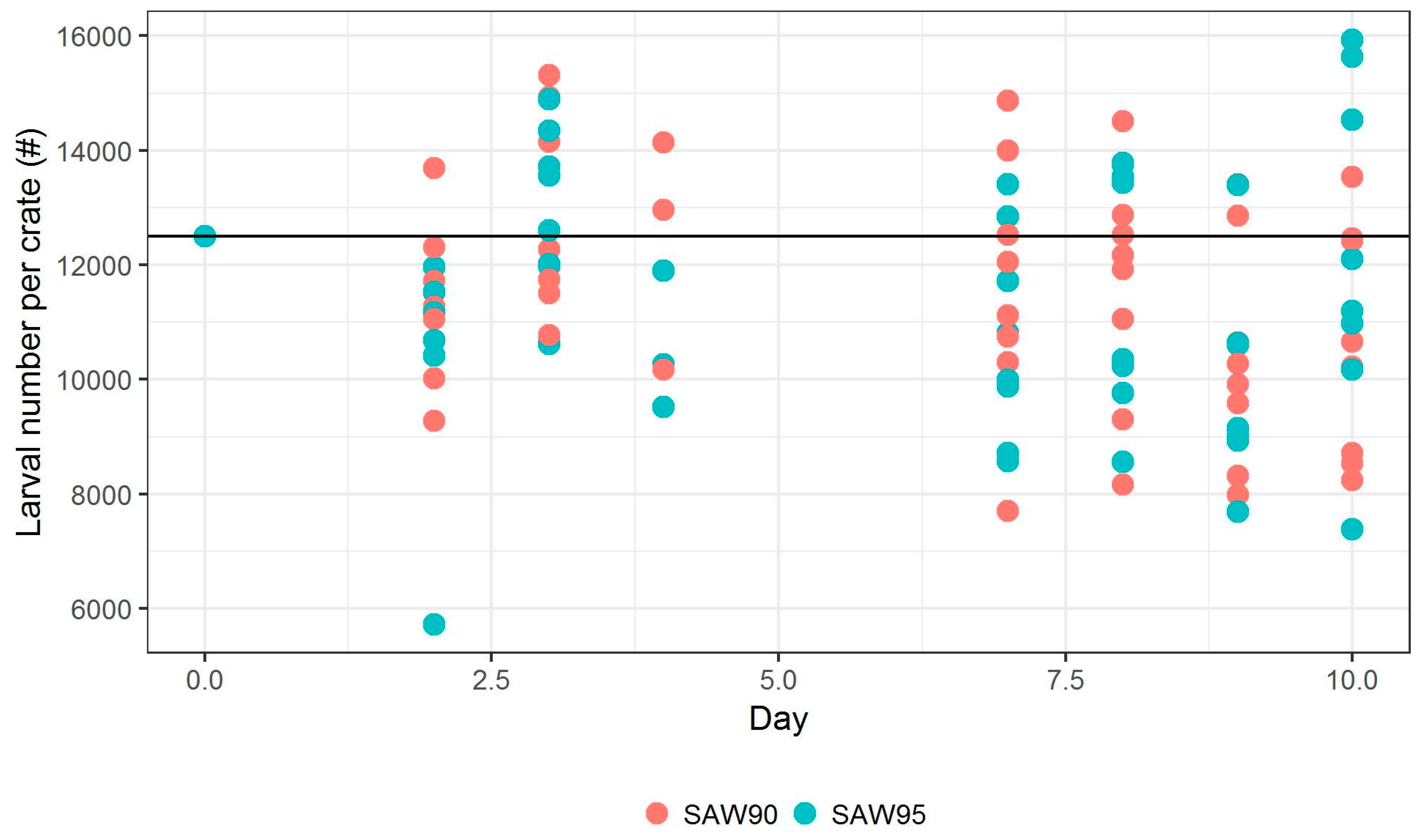
Number of larvae in each container was also estimated on multiple days.
Figure 4 shows the estimation of these larvae numbers, showing their number per container. The black horizontal line shows the targeted population size of each container. At the first in-test measurements on Day 2, 3, and 4, we see a slight increase in population from under target line to above target line. Obviously, the population could not have increased; this increase is observed due to the increase in weight of larvae, which improves the ease of measurement. Approximately 10% and 20% decreases in population were observed on Day 8 and 9, respectively. The reason behind this is not known. On Day 10, the average mortality rate that was observed for SAW90 was 9.1% (SD 10.79%), while the mortality rate for SAW 95 was 15.4% (SD 16.28%). According to the Wilcoxon rank-sum test, we do not reject the null hypothesis that these mortality rates differ (
p-value = 0.6454).
The crates that were fed SAW95 had an average larval yield of 0.57kg (SD 0.13kg) and the crates fed SAW90 had an average larval yield of 0.62kg (SD 0.1kg). Dry matter larval yields were measured at the end of the test. Consistent with the average larval weight measurement, the containers in which larvae were fed with SAW 90 mix achieved higher yields. Dry matter conversion has been calculated as the difference between the end and start weight (kg) of the larvae (fresh larval biomass at the start of rearing), divided by the kilograms of dry matter fed throughout the experiment. The average dry matter conversion rate is 11.9 (SD = 3.1) and 14.8 (SD = 5.7) for the SAW 90 and SAW 95 fed containers, respectively. The crates fed SAW95 had an average residue yield of 2.36kg (SD 0.23kg) and the crates fed SAW90 had an average residue yield of 2.15kg (SD 0.07kg). Thus, in both cases residue yield was approximately 3.7 times more than the wet larval yield.
3.2. Composition Analyses
Table 2 shows the dry matter, crude protein, lipid, ash, and carbohydrate contents of the larvae. The statistical difference between the composition of larvae obtained using two feed mixes was analysed using the Wilcoxon rank-sum test, with only the dry matter showing a statistically significant difference (
p-value = 0.003). In the current study, the crude protein content of the larvae fed with two SAW mixes (~ 40% of DM) were similar to previously published data of BSFL [
6], but lipid content was significantly lower (~ 7% of DM) as compared to the commonly reported lipid composition of BSFL (~ 25–30% of DM) [
6]. Lipid accumulation in insects varies with exact life stage, diet, and environment, and these larvae may have lower lipid amounts due to the lack of available lipids and carbohydrates in the diet, which also likely remains a reason for their low growth [
29]. It has been already indicated in
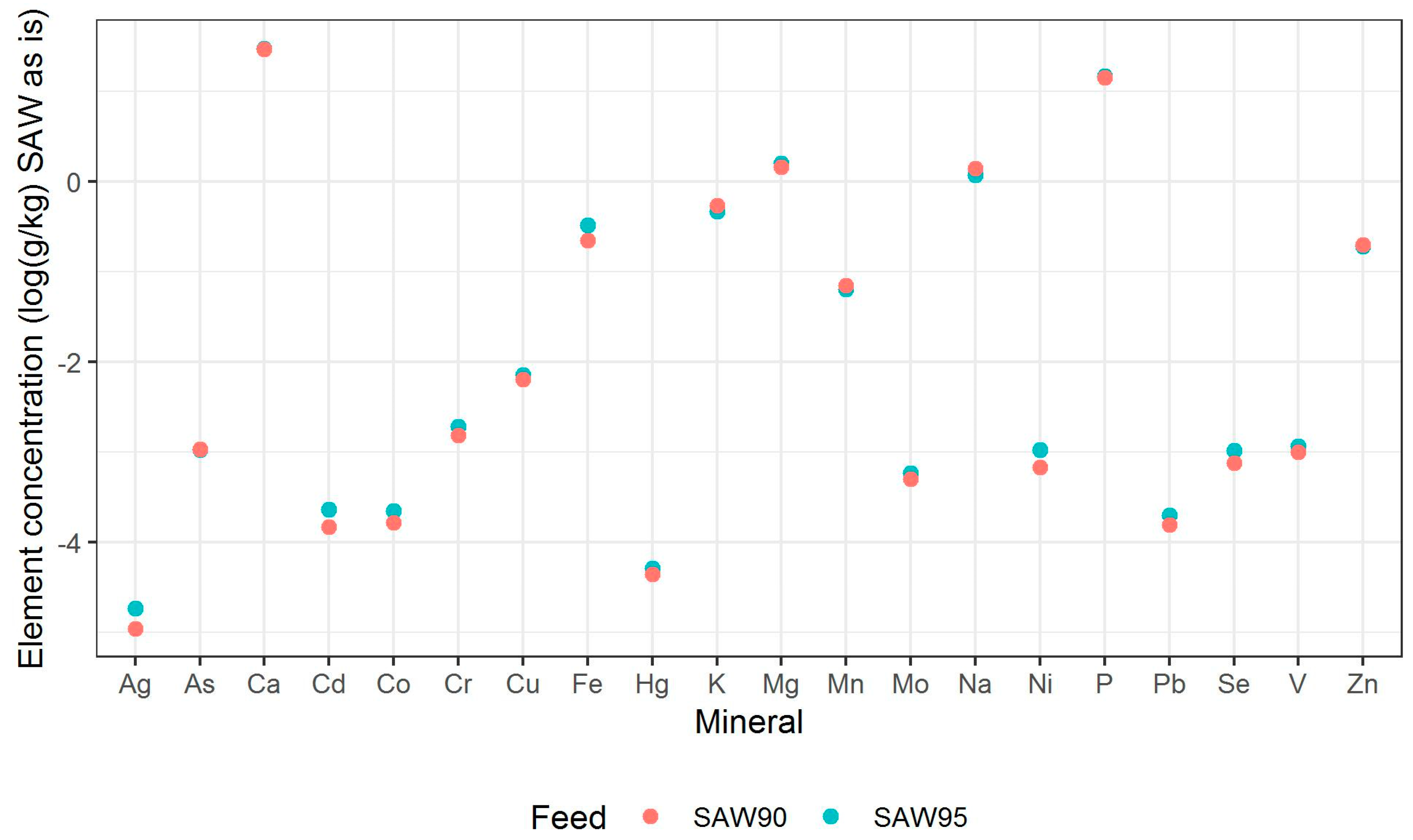
Figure 2 that both SAW ingredients have substantial amounts of cadmium, copper, and zinc. Lindqvist et al. (2001) and Ortel et al. (1995) describe how high levels of dietary metals, including cadmium, copper, and zinc are linked to reduced fat accumulation in insects [
30,
33]. It could be that the low availability of nutrients, such as lipids and carbohydrates, increases the accumulation of heavy metals and other elements, but additional research is needed to confirm this hypothesis.
The mineral composition of the two SAW mixes and larvae that were fed with two SAW mixes as well as a control feed are mentioned in
Table 3 and
Table 4, respectively. Trace metals, such as vanadium, chromium, nickel, arsenic, and mercury have previously been analyzed in BSFL [
34,
35,
36,
37]. However, to the best of our knowledge, this is the first report indicating the presence (or accumulation) of silver in BSFL, though silver was present at concentrations of just a few micrograms per kilogram. Calcium was the macro-mineral present in highest concentrations in larvae fed with both SAW mixes, while manganese was the micro-mineral that was present in a highest concentration. Both calcium and manganese were also the major macro-mineral and trace mineral present in the two SAW materials, respectively (see
Figure 2).
When evaluating mineral accumulation with regards to regulations, three types of limits (or levels) have been taken into account: (1) Rejection limits; (2) Action limits; and, (3) Maximum tolerance levels. The rejection limits or maximum content could be defined as ‘maximum levels in mg/kg of the feed material or compound feeds, based on a moisture content of 12%, unless mentioned differently. If this limit is exceeded, then the product is not suitable for use as feed material or animal feed’ [
38,
39,
40]. Action limit is a term that is commonly used in GMP+ (most popular animal feed and pet food quality management systems) documents. This limit is defined in agreement with the sector, supplier, or customer. It could be defined as ‘maximum levels in mg/kg of feed materials or compound feeds, based on a moisture content of 12%, unless differently mentioned. If this limit is exceeded, then an investigation into the cause should be undertaken and corrective measures should be taken to remove or control the cause’ [
40]. Maximum tolerance levels are more strict in comparison to the rejection limits or actions limits and could be defined as ‘dietary levels that, when fed for a defined period of time, will not impair animal health or performance’ [
41].
Table 5 provides the maximum tolerance levels (based on chronic consumption), action limits, and rejection limits of minerals that were considered in this study. The maximum tolerance levels that were used in this study were according to the values estimated for poultry [
38], because live larvae are already approved for poultry consumption in Europe [
42]. When required, the data was converted into dry matter basis, so that all measurements are in the same units.
Table 6 indicates the comparison of rejection limits with the amounts detected in larvae. The comparison is most effectively made using one-sided t-tests, the outcomes of which are provided, to test whether the concentration of an element in the larvae exceeded this limit. The one-sided t-test is efficient in this case, because we are concerned with whether the concentrations are too high. The action limits were not compared with the obtained results, because it was clearly visible that larvae were below the action limits. Both larvae fed with SAW 90 and 95 mixes were detected to contain arsenic (As) in concentrations that were above rejection limits proposed by the European Commission [
39]. The inorganic form of arsenic, particularly the trivalent arsenic (arsenite), is known to be extremely toxic during chronic exposure [
43]. Chronic dietary exposure to arsenic may result in the development of peripheral neuropathy, modifications in normal heme metabolism, impairment of renal function, and other serious disorders in animals [
44]. Larvae with such levels of arsenic cannot be used for direct feeding to poultry animals in Europe (
Table 5). However, it may be possible to process them into insect protein meal, which could be included in compound animal feeds, such that the total amount of arsenic in compound feed is below the European Union (EU) rejection limits, since the insect meal might be a low enough portion of the total mix that it is not likely that an animal consuming it will intake a large dose.
Table 7 shows the comparison of maximum tolerance levels of minerals with the amounts detected in larvae. To our surprise, larvae were found to contain calcium, potassium, magnesium, and phosphorus in amounts exceeding the maximum tolerance levels. In normal concentrations, these minerals have an important role in animal nutrition [
45]. However, repeated dietary exposure to these minerals in such concentrations could negatively affect animal health. In pets animals, like dogs, a high level of dietary calcium (hypercalcemia) is responsible for neoplasia, primary perparathyroidism, chronic kidney disease, and other serious health issues [
46]. On the other hand, high levels of potassium, magnesium, and phosphorus in animals diets is associated with issues, such as chronic kidney disease, blood electrolyte abnormalities, and increased risk of cardiovascular diseases, respectively [
41,
42,
44].
3.3. Bioaccumulation
Table 8 displays the bioaccumulation factors of the elements in the feed and their standard deviation. The bioaccumulation of the heavy metals As, Cd, and Pb was previously studied in [
28]. The results that are reported here are not entirely consistent with those reported in [
28]. Both the present work and [
28] concur that Cd is highly accumulative. Divalent Cd cation has ionic radii that are similar to the divalent Ca cation, which makes the entry of cadmium in animal cells easy via calcium channels without any dependency on endocytosis or ATP [
47].
As is shown to accumulate when larvae fed on SAW90 but not on SAW95 and the factors are higher than that reported previously for BSF (0.49–0.58 [
28]). Studies on another dipteran larva (
Chironomus riparius) have shown dose-dependent accumulation of As with feed. Moreover, it was suggested that
C. riparius larvae could biotransform the arsenate form into the arsenite form as a part of detoxification metabolism. In the same study, it was also found that pupae contain lower amounts of As in comparison to larvae, indicating that larvae excrete substantial amounts of As before transforming into pupae [
48]. Pb does not appear to accumulate, whereas bioaccumulation factors that are higher than 1 have been reported (1.1–1.8 [
28]) for this metal.
Additionally, Hg is also found to be bioaccumulative in larvae that were fed on the SAW diets. Dipteran larvae are also known to bioaccumulate Hg. A study indicated that aquatic dipteran larvae can accumulate more Hg in comparison to their adults [
49], suggesting that bioaccumulation of Hg is linked to the life cycle of these insects. The results of the present study show how the bioaccumulation of these three heavy metals might manifest in a heavy metal rich, real-world feed. In addition to heavy metals,
Table 8 reports the bioaccumulation of other elements. The accumulation of many of these elements have been studied in Liland et al. (2017), where larvae were also fed on a sea-sourced diet anda number of these are reported to be bioaccumulative [
24]. This is also the case in the present study, in which K shows particularly high accumulation.
This paper studies the performance and safety of SAW materials. However, it is crucial to expand studies in order to provide general guidelines for assessing the safety of different feeds and BSFL produced on them also for other feeds. In this research, we show that, in addition to well-known contaminants of BSFL, such as heavy metals, micronutrients can also reach levels that exceed regulatory norms. It may not be obvious to companies and practitioners which metals and micronutrients are likely to pose problems, so it is recommended that a broad screening of metals and micronutrients is performed on feeds for the BSFL that do not have known characteristics, especially if the feed material is likely to contain concentrated micronutrients. The paper provides a list of regulatory limits that can be used to evaluate the concentration of metals and micronutrients in the larvae. However, this is potentially incomplete and it will become out of date. Readers are encouraged to look to the most recent regulatory updates for current best practices.