A Case of Letting the Cat out of The Bag—Why Trap-Neuter-Return Is Not an Ethical Solution for Stray Cat (Felis catus) Management
Simple Summary
Abstract
1. Introduction
2. Population Control Under TNR Programs—Fundamental Issues
2.1. Do TNR Programs Successfully Deliver on Reduction in Stray Cat Numbers?
2.2. How Many Cats Would Be Saved from Euthanasia?
2.3. How Much Would TNR Programs Cost?
- Hughes and Slater [64] estimated that neutering 158 colony cats at a university veterinary teaching hospital in 1998 cost approximately US$9800 (discounted to US$4900). Blood tests, vaccines and sub-dermal microchips were donated (including surgical labor at 35 hours/student; number of students unspecified). The labor to trap cats was also donated (25–35 hours/person/week; trapping regime: 4–6 nights with 20 traps set/night every 4–6 weeks over two years). Traps were bought outright (cost US$900) and more traps were borrowed to meet the trapping effort required. Food was donated, as was the time for feeding/monitoring (approximately 15 h/person/week; number of people unspecified).
- Nutter et al. [88] estimated that the mean number of trap-nights required to trap 90% of adult cats (or until only one cat remained untrapped) in nine colonies was 8.9 ± S.D. 3.9 trap nights/cat.
- Webb [89] estimated that implementing TNR programs for approximately 150,000 stray cats across Melbourne, Australia, would initially cost AU$3 million to trap and neuter ($20/cat) and AU$39 million per year to feed ($5/cat/week).
- Lohr et al. [90] modelled the costs and benefits of TNR vs. trap-euthanasia in Hawaii and found that TNR was approximately twice as expensive to implement. Even TNR programs that used volunteers were more expensive than trap-euthanasia programs that paid professionals.
2.4. Who Would Be Responsible for TNR Cats?
3. Can TNR Programs Improve Stray Cat Welfare in Australia?
- Where would cats under TNR management live?
- Is the TNR process itself stressful?
- Would TNR cats be more vulnerable to injury?
- Are stray cats vulnerable to high parasite loads and diseases?
- Can parasites and diseases be treated in TNR cats? And at what cost?
- Are TNR cats in poor health and body condition?
- What would TNR cats eat?
- Would TNR cat management impact people in urban areas?
- Would TNR cat management impact pet cats?
- Would TNR cat management impact urban wildlife?
3.1. Challenge 1. Where Would Cats Under TNR Management Live?
3.2. Challenge 2. Is the TNR Process Itself Stressful?
3.3. Challenge 3. Would TNR Cats Be Vulnerable to Injury?
3.4. Challenge 4. Are Stray Cats Vulnerable to High Parasite Loads and Diseases?
3.4.1. Ectoparasites
3.4.2. Gastrointestinal Parasites
3.4.3. Haemoparasites
3.4.4. Viruses
3.5. Challenge 5. Can Parasites and Diseases Be Treated in TNR Cats? And at What Cost?
3.6. Challenge 6. Are TNR Cats in Poor Health and Body Condition?
3.7. Challenge 7. What Would TNR Cats Eat?
3.8. Challenge 8. Would TNR Cat Management Impact People in Urban Areas?
3.8.1. Zoonotic Parasites and Disease
3.8.2. Public Nuisance
3.8.3. Physical Injury, Waste and Allergies
3.9. Challenge 9. Would TNR Cat Management Impact Pet Cats?
3.10. Challenge 10. Would TNR Cat Management Impact Urban Wildlife?
3.11. Summary
- Where would cats under TNR management live? Where TNR cats live has many potential consequences for whole communities. Many TNR cats are maintained in private backyards and in public spaces (e.g., schools), but many more such spaces would be needed in urban environments to save the cats euthanized annually in Australia and overseas. Maintenance of colonies in these areas does not prevent cats from roaming across whole neighborhoods.
- Is the TNR process itself stressful? The TNR process is stressful in the short term and possibly in the long term, though studies are lacking. The stress of TNR is overlooked in favor of the potential benefits of neutering. Placing stray cats in shelters after neutering to assess temperament and implement rehabilitation procedures may increase adoptions and decrease euthanasia of strays without re-releasing cats back onto urban streets.
- Would TNR cats be vulnerable to injury? There is overwhelming evidence for injuries to cats in urban environments. TNR cats are just as likely as roaming pets to encounter dangers, including vehicle collisions, exposure to poisons, fighting with cats and other species, and human cruelty but less likely to receive veterinary care. Increasing or maintaining the number or density of cat colonies may increase nuisance complaints and could increase acts of cruelty to cats.
- Are stray cats vulnerable to high parasite loads and diseases? Globally, stray cats can carry high parasite loads and diseases that compromise their health. Maintaining or establishing more cat colonies increases the likelihood of pathogen transmission. TNR cats are therefore potential vectors of these to other strays, pet cats, wildlife and humans.
- Can parasites and diseases be treated in TNR cats? And at what cost? Parasites and some diseases are difficult to effectively treat in stray cats, even when they live in colonies. It is difficult to administer effective dosages and regularity of treatments, particularly for cryptic or aggressive cats. The usefulness of carrying out incomplete regimes of worming and vaccination is questionable and a costly exercise. Without being able to administer prophylactic health care, TNR cannot guarantee the prevention of parasites and disease in colonies.
- Are TNR cats in poor health and body condition? TNR cats can be in poor body condition without obvious physical symptoms, which compromises their short- and long-term health and welfare. Visual assessments are not adequate for assessing stray cat health. Therefore, TNR programs need to assess health using multiple methods and will require regular funds and labor.
- What would TNR cats eat? Provision of regular food by colony caretakers does not prevent TNR cats from scavenging anthropogenic refuse that does not always provide cats with nutrition and can compromise short- and long-term health. To prevent scavenging, caretakers need to be diligent in removing refuse from colony environments, which requires extra labor.
- Would TNR cat management impact people in urban areas? Although literature on the frequency of the transmission of some zoonotic pathogens from stray cats to humans is limited, the potential for cats to rapidly spread diseases necessitates active risk management. Stray cats are also a significant cause of public nuisance complaints, that neutering alone will not prevent. Aggressive cats can also injure people and cause allergies. These issues could preclude approval of TNR management.
- Would TNR cat management impact pet cats? TNR cats can compromise the health and welfare of pet cats through fighting, disease and intimidation, and prioritizing TNR cats over pet cats can lead to community conflict.
- Would TNR cat management impact urban wildlife? TNR compromises the welfare and persistence of urban wildlife and prioritizes stray cats over wildlife. It is irresponsible to introduce or maintain colonies where threatened wildlife occurs. More local research is needed on this issue.
4. Prevention Is Better Than Cure—Improving Responsible Pet Ownership in Australia
4.1. Legislation and Community Initiatives
4.2. Increasing Adoption
4.3. Limiting Unplanned Breeding
4.4. Future Investment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Glossary
| TNR | Trap–Neuter–Return (TNR) includes synonymous terminology in the literature: Trap-Neuter-Release, Trap-Neuter-Vaccinate-Return, Trap-Castrate-Return, Trap-Vasectomize-Return, and Trap-Spay-Return. |
| Neuter | Terminology synonymous with ‘castrate, spay, sterilize, desex’, etc. |
| Pet cats | Also ‘owned cats’. Cats that receive food, shelter and medical care from an owner(s). |
| Stray cats | Sensu [4]. Also ‘semi-feral, street/alley cats’, also ‘feral’ in some countries including Italy, USA, UK. Cats that are born ‘on the streets’ of urban environments and cats that were owned by people at some point but have been abandoned or become separated from their owners. Stray cats move freely within urban environments and can choose their level of association with people by adjusting their temporal or spatial activity patterns. Stray cats can be dispersed broadly across urban environments and/or be locally concentrated around specific resources such as dockyards/quays, refuse tips and bush reserves (i.e., ‘colony’ cats). |
| Colony cats | Also ‘fed cats’. Stray cats that are deliberately provided with food at one or more locations are classified as belonging to a ‘colony’, whether they permanently or temporarily congregate around these resources. |
| Feral cats | In literature from the USA and UK, stray cats in urban areas are often termed ‘feral’ (sensu [4]). However, in Australian literature, feral cats are totally wild animals that live and breed beyond the periphery of human settlements, surviving without human interactions or resources. Animal shelters (including some in Australia) sometimes also use the term ‘feral’ to describe cats with ‘wild’ aggressive temperaments, regardless of ownership history [40]. For consistency with Australian literature, we distinguish between stray cats in urban areas and feral cats in rural environments. |
| Caretakers | People who deliberately feed stray cats and may provide treatment for injuries, illness or parasites. Maintain ≥1 cat colonies that may or may not be part of organized TNR programs. Caretakers are normally female with a median age of 45–58 [59,63]. Also termed ‘semi-owners’ or ‘casual owners’. |
References
- Bradshaw, J.W.S.; Casey, R.A.; Brown, S.L. The Behaviour of the Domestic Cat, 2nd ed.; CABI Publishers: Oxfordshire, UK, 2012; p. 264. [Google Scholar]
- Hornok, S.; Baneth, G.; Grima, A.; Takacs, N.; Kontschan, J.; Meli, M.L.; Suter, V.; Salant, H.; Farkas, R.; Hofmann-Lehmann, R. Molecular investigations of cat fleas (Ctenocephalides felis) provide the first evidence of Rickettsia felis in Malta and Candidatus Rickettsia senegalensis in Israel. New Microb. New Infect. 2018, 25, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.C. A review of over three decades of research on cat-human and human-cat interactions and relationships. Behav. Process. 2017, 141, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, J.W.S.; Horsfield, G.F.; Allen, J.A.; Robinson, I.H. Feral cats: Their role in the population dynamics of Felis catus. Appl. Anim. Behav. Sci. 1999, 65, 273–283. [Google Scholar] [CrossRef]
- Jessup, D.A. The welfare of feral cats and wildlife. J. Am. Vet. Med. Assoc. 2004, 225, 1377–1383. [Google Scholar] [CrossRef] [PubMed]
- Winter, L.; Wallace, G.E. Impacts of Feral and Free-Ranging Cats on Bird Species of Conservation Concern: A Five-State Review of New York, New Jersey, Florida, California, and Hawaii; American Bird Conservancy: The Plains, VA, USA, 2006; p. 29. [Google Scholar]
- Marra, P.P.; Santella, C. Cat Wars: The Devastating Consequences of a Cuddly Killer; Princeton University Press: Princeton, NJ, USA, 2016; p. 212. [Google Scholar]
- Birhane, M.G.; Cleaton, J.M.; Monroe, B.P.; Wadhwa, A.; Orciari, L.A.; Yager, P.; Blanton, J.; Velasco-Villa, A.; Petersen, B.W.; Wallace, R.M. Rabies surveillance in the United States during 2015. J. Am. Vet. Med. Assoc. 2017, 250, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Jessup, D.; Stone, E. More on feral cats. J. Am. Vet. Med. Assoc. 2010, 235, 7. [Google Scholar]
- Dabritz, H.A.; Atwill, R.; Gardner, I.A.; Miller, M.A.; Conrad, P.A. Outdoor fecal deposition by free-roaming cats and attitudes of cat owners and nonowners toward stray pets, wildlife, and water pollution. J. Am. Vet. Med. Assoc. 2006, 229, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Eymann, J.; Herbert, C.A.; Cooper, D.W.; Dubey, J.P. Serologic survey for Toxoplasma gondii and Neospora caninum in the common brushtail possum (Trichosurus vulpecula) from urban Sydney, Australia. J. Parasitol. 2006, 92, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.L.; Akstein, R.B.; Hlavsa, M.C.; Lopez, A.S.; Wilson, M.; Holland, G.N. Follow-up of the 1977 Georgia outbreak of toxoplasmosis. Am. J. Trop. Med. Hyg. 2016, 94, 1299–1300. [Google Scholar] [CrossRef]
- Stanek, J.F.; Stich, R.W.; Dubey, J.P.; Reed, S.M.; Njoku, C.J.; Lindsay, D.S.; Schmall, L.M.; Johnson, G.K.; LaFave, B.M.; Saville, W.J.A. Epidemiology of Sarcocystis neurona infections in domestic cats (Felis domesticus) and its association with equine protozoal myeloencephalitis (EPM) case farms and feral cats from a mobile spay and neuter clinic. Vet. Parasitol. 2003, 117, 239–249. [Google Scholar] [CrossRef]
- Gerhold, R.W.; Jessup, D.A. Zoonotic diseases associated with free-roaming cats. Zoonoses Pub. Health 2013, 60, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Carver, S.; Bevins, S.N.; Lappin, M.R.; Boydston, E.E.; Lyren, L.M.; Alldredge, M.; Logan, K.A.; Sweanor, L.L.; Riley, S.P.D.; Serieys, L.E.K.; et al. Pathogen exposure varies widely among sympatric populations of wild and domestic felids across the United States. Ecol. Appl. 2016, 26, 367–381. [Google Scholar] [CrossRef]
- Hwang, J.; Gottdenker, N.L.; Oh, D.-H.; Nam, H.-W.; Hang Lee, A.; Myung-Sun, C. Disentangling the link between supplemental feeding, population density, and the prevalence of pathogens in urban stray cats. Peer J. 2018, 6, e4988. [Google Scholar] [CrossRef] [PubMed]
- Norris, J.M.; Bell, E.T.; Hales, L.; Toribio, J.A.L.M.L.; White, J.D.; Wigney, D.I.; Baral, R.M.; Malik, R. Prevalence of feline immunodeficiency virus infection in domesticated and feral cats in eastern Australia. J. Feline Med. Surg. 2007, 9, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Rees, P.A. The Ecology and Management of Feral Cat Colonies. A Survey of Feral Cat Colonies in Great Britain and an Experimental Field Study of the Effect of Neutering on the Ecology, Behaviour and Social Organisation of a Single Colony. Ph.D. Thesis, University of Bradford, Bradford, UK, 1982. [Google Scholar]
- Yamane, A.; Ono, Y.; Doi, T. Home range size and spacing pattern of a feral cat population on a small island. J. Mammal. Soc. Jpn. 1994, 19, 9–20. [Google Scholar] [CrossRef]
- Mirmovitch, V. Spatial organisation of urban feral cats (Felis catus) in Jerusalem. Wildl. Res. 1995, 22, 299–310. [Google Scholar] [CrossRef]
- Gunther, I.; Raz, T.; Berke, O.; Klement, E. Nuisances and welfare of free-roaming cats in urban settings and their association with cat reproduction. Prev. Vet. Med. 2015, 119, 203–210. [Google Scholar] [CrossRef]
- Uetake, K.; Yamada, S.; Yano, M.; Tanaka, T. A survey of attitudes of local citizens of a residential area toward urban stray cats in Japan. J. Appl. Anim. Welf. Sci. 2014, 17, 172–177. [Google Scholar] [CrossRef]
- Scarlett, J.; Johnston, N. Impact of a subsidized spay neuter clinic on impoundments and euthanasia in a community shelter and on service and complaint calls to animal control. J. Appl. Anim. Welf. Sci. 2012, 15, 53–69. [Google Scholar] [CrossRef]
- Blancher, P. Estimated number of birds killed by house cats (Felis catus) in Canada. Avian Conserv. Ecol. 2013, 8, 3–22. [Google Scholar] [CrossRef]
- Loss, S.R.; Will, T.; Marra, P.P. The impact of free-ranging domestic cats on wildlife of the United States. Nat. Commun. 2013, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Barratt, E.M.; Gottelli, D.; Kitchener, A.C.; Daniels, M.J.; Pritchard, J.K.; Bruford, M.W. Genetic diversity and introgression in the Scottish wildcat. Mol. Ecol. 2001, 10, 319–336. [Google Scholar] [CrossRef] [PubMed]
- Rochlitz, I. Clinical study of cats injured and killed in road traffic accidents in Cambridgeshire. J. Small Anim. Pract. 2004, 45, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Childs, J.E.; Ross, L. Urban cats: Characteristics and estimation of mortality due to motor vehicles. Am. J. Vet. Res. 1986, 47, 1643–1648. [Google Scholar] [PubMed]
- Natoli, E.; Say, L.; Cafazzo, S.; Bonanni, R.; Schmid, M.; Pontier, D. Bold attitude makes male urban feral domestic cats more vulnerable to Feline Immunodeficiency Virus. Neurosci. Biobehav. Rev. 2005, 29, 151–157. [Google Scholar] [CrossRef]
- Xavier, F.G.; Kogika, M.M.; de Souza Spinosa, H. Common causes of poisoning in dogs and cats in a Brazilian veterinary teaching hospital from 1998 to 2000. Vet. Hum. Toxicol. 2002, 44, 115–116. [Google Scholar] [PubMed]
- Baker, P.J.; Soulsbury, C.D.; Iossa, G.; Harris, S. Domestic Cat (Felis catus) and Domestic Dog (Canis familiaris). In Urban Carnivores: Ecology, Conflict, and Conservation; Gehrt, S.D., Riley, S.P.D., Cypher, B.L., Eds.; The John Hopkins University Press: Baltimore, MD, USA, 2010; pp. 157–171. [Google Scholar]
- Vnuk, D.; Capak, H.; Gusak, V.; Maticic, D.; Popovic, M.; Bottegaro, N.B. Metal projectile injuries in cats: Review of 65 cases (2012–2014). J. Feline Med. Surg. 2016, 18, 626–631. [Google Scholar] [CrossRef]
- Slater, M.R. Chapter 6. The Welfare of Feral Cats. In The Welfare of Cats; Rochlitz, I., Ed.; Springer: Dordrecht, The Netherlands, 2007; pp. 141–175. [Google Scholar]
- Robertson, S.A. A review of feral cat control. J. Feline Med. Surg. 2008, 10, 366–375. [Google Scholar] [CrossRef]
- Marston, L.C.; Bennett, P.C. Admissions of cats to animal welfare shelters in Melbourne, Australia. J. Appl. Anim. Welf. Sci. 2009, 12, 189–213. [Google Scholar] [CrossRef]
- Alberthsen, C.; Rand, J.S.; Bennett, P.C.; Paterson, M.; Lawrie, M.; Morton, J.M. Cat admissions to RSPCA shelters in Queensland, Australia: Description of cats and risk factors for euthanasia after entry. Aust. Vet. J. 2013, 91, 35–42. [Google Scholar] [CrossRef]
- Arbe Montoya, A.I.; Rand, J.S.; Greer, R.M.; Alberthsen, C.; Vankan, D. Relationship between sources of pet acquisition and euthanasia of cats and dogs in an animal shelter: A pilot study. Aust. Vet. J. 2017, 95, 194–200. [Google Scholar] [CrossRef]
- Tanaka, A.; Wagner, D.C.; Kass, P.H.; Hurley, K.F. Associations among weight loss, stress, and upper respiratory tract infection in shelter cats. J. Am. Vet. Med. Assoc. 2012, 240, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Maubach, B. Benchmarking Best Practices in Decreasing Animal Shelter Euthanasia Rates Across the West Coast; The University of Arizona: Tucson, AZ, USA, 2014. [Google Scholar]
- Alberthsen, C.; Rand, J.; Morton, J.; Bennett, P.; Paterson, M.; Vankan, D. Numbers and characteristics of cats admitted to Royal Society for the Prevention of Cruelty to Animals (RSPCA) Shelters in Australia and reasons for surrender. Animals 2016, 6, 23. [Google Scholar] [CrossRef]
- Kerr, C.; Rand, J.; Morton, J.; Reid, R.; Paterson, M. Changes associated with improved outcomes for cats entering RSPCA Queensland shelters from 2011 to 2016. Animals 2018, 8, 95. [Google Scholar] [CrossRef]
- Tan, K.; Rand, J.; Morton, J. Trap-Neuter-Return activities in urban stray cat colonies in Australia. Animals 2017, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Swarbrick, H.; Rand, J. Application of a protocol based on Trap-Neuter-Return (TNR) to manage unowned urban cats on an Australian university campus. Animals 2018, 8, 77. [Google Scholar] [CrossRef]
- Gotsis, T. Feral Cats: Do Trap-Neuter-Return Programs Work? New South Wales Parliament: Sydney, Australia, 2014; p. 19. [Google Scholar]
- Australian Veterinary Association. Policy 13.2: Management of Cats in Australia. Available online: https://www.ava.com.au/policy/132-management-cats-australia (accessed on 25 January 2019).
- Levy, J.K.; Isaza, N.M.; Scott, K.C. Effect of high-impact targeted trap-neuter-return and adoption of community cats on cat intake to a shelter. Vet. J. 2014, 201, 269–274. [Google Scholar] [CrossRef]
- Scott, K.C.; Levy, J.K.; Gorman, S.P. Body condition of feral cats and the effect of neutering. J. Appl. Anim. Welf. Sci. 2002, 5, 203–213. [Google Scholar] [CrossRef]
- Hosie, M.J.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Lutz, H.; Marsilio, F.; et al. Feline Immunodeficiency: ABCD Guidelines on Prevention and Management. J. Feline Med. Surg. 2009, 11, 575–584. [Google Scholar] [CrossRef]
- Horn, J.A.; Mateus-Pinilla, N.; Warner, R.E.; Heske, E.J. Home range, habitat use, and activity patterns of free-roaming domestic cats. J. Wildl. Manag. 2011, 75, 1177–1185. [Google Scholar] [CrossRef]
- Johnson, K.L.; Cicirelli, J. Study of the effect on shelter cat intakes and euthanasia from a shelter neuter return project of 10,080 cats from March 2010 to June 2014. Peer J. 2014, 2, e646. [Google Scholar] [CrossRef]
- Zito, S.; Aguilar, G.; Vigeant, S.; Dale, A. Assessment of a targeted trap-neuter-return pilot study in Auckland, New Zealand. Animals 2018, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Cat Alliance of Australia Inc. Why Trap-Neuter-Return Feral Cats?: The Case for TNR. Available online: http://www.catallianceaustralia.org/main/page_why_trap_neuter_and_return.html (accessed on 25 January 2019).
- Sparkes, A.H.; Bessant, C.; Cope, K.; Ellis, S.L.H.; Finka, L.; Halls, V.; Hiestand, K.; Horsford, K.; Laurence, C.; MacFarlaine, I.; et al. ISFM Guidelines on population management and welfare of unowned domestic cats (Felis catus). J. Feline Med. Surg. 2013, 15, 811–817. [Google Scholar] [CrossRef]
- PetSmart Charities. Attitudes on Pet Homelessness Are Shifting: 2014 U.S. Shelter Pet Report; PetSmart Charities: Phoenix, AZ, USA, 2014; p. 40. [Google Scholar]
- Longcore, T.; Rich, C.; Sullivan, L.M. Critical assessment of claims regarding management of feral cats by trap–neuter–return. Conserv. Biol. 2009, 23, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Barrows, P.L. Professional, ethical, and legal dilemmas of trap-neuter-release. J. Am. Vet. Med. Assoc. 2004, 225, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Roebling, A.D.; Johnson, D.; Blanton, J.D.; Levin, M.; Slate, D.; Fenwick, G.; Rupprecht, C.E. Rabies prevention and management of cats in the context of Trap–Neuter–Vaccinate–Release programmes. Zoonoses Pub. Health 2014, 61, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Royal Society for the Prevention of Cruelty to Animals. Response to Public Consultation on the Discussion Paper: Identifying Best Practice Domestic Cat Management in Australia; RSPCA Australia: Deakin West, Australia, 2018. [Google Scholar]
- Centonze, L.A.; Levy, J.K. Characteristics of free-roaming cats and their caretakers. J. Am. Vet. Med. Assoc. 2002, 220, 1627–1633. [Google Scholar] [CrossRef] [PubMed]
- Loyd, K.T.; Miller, C.A. Factors related to preferences for trap–neuter–release management of feral cats among Illinois homeowners. J. Wildl. Manag. 2010, 74, 160–165. [Google Scholar] [CrossRef]
- Lohr, C.A.; Lepczyk, C.A. Desires and management preferences of stakeholders regarding feral cats in the Hawaiian Islands. Conserv. Biol. 2014, 28, 392–403. [Google Scholar] [CrossRef]
- Lohr, C.A.; Lepczyk, C.A.; Cox, L.J. Identifying people’s most preferred management technique for feral cats in Hawaii. Hum. Wildl. Interact. 2014, 8, 35–45. [Google Scholar]
- Gunther, I.; Raz, T.; Even Zor, Y.; Bachowski, Y.; Klement, E. Feeders of free-roaming cats: Personal characteristics, feeding practices and data on cat health and welfare in an urban setting of Israel. Front. Vet. Sci. 2016, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.L.; Slater, M.R. Implementation of a feral cat management program on a university campus. J. Appl. Anim. Welf. Sci. 2002, 5, 15–28. [Google Scholar] [CrossRef]
- Wallace, J.L.; Levy, J.K. Population characteristics of feral cats admitted to seven trap-neuter-return programs in the United States. J. Feline Med. Surg. 2006, 8, 279–284. [Google Scholar] [CrossRef]
- Ireland, T.; Miller Neilan, R. A spatial agent-based model of feral cats and analysis of population and nuisance controls. Ecol. Model. 2016, 337, 123–136. [Google Scholar] [CrossRef]
- Schmidt, P.M.; Swannack, T.M.; Lopez, R.R.; Slater, M.R. Evaluation of euthanasia and trap-neuter-return (TNR) programs in managing free-roaming cat populations. Wildl. Res. 2009, 36, 117–125. [Google Scholar] [CrossRef]
- Guttilla, D.A.; Stapp, P. Effects of sterilization on movements of feral cats at a wildland-urban interface. J. Mammal. 2010, 91, 482–489. [Google Scholar] [CrossRef]
- Cove, M.V.; Gardner, B.; Simons, T.R.; Kays, R.; O’Connell, A.F. Free-ranging domestic cats (Felis catus) on public lands: Estimating density, activity, and diet in the Florida Keys. Biol. Invasions 2018, 20, 333–344. [Google Scholar] [CrossRef]
- Hernandez, S.M.; Loyd, K.A.T.; Newton, A.N.; Gallagher, M.; Carswell, B.L.; Abernathy, K.J. Activity patterns and interspecific interactions of free-roaming, domestic cats in managed Trap-Neuter-Return colonies. Appl. Anim. Behav. Sci. 2018, 202, 63–68. [Google Scholar] [CrossRef]
- Hernandez, S.M.; Loyd, K.A.T.; Newton, A.N.; Carswell, B.L.; Abernathy, K.J. The use of point-of-view cameras (Kittycams) to quantify predation by colony cats (Felis catus) on wildlife. Wildl. Res. 2018, 45, 357–365. [Google Scholar] [CrossRef]
- Loyd, K.A.T.; DeVore, J.L. An evaluation of feral cat management options using a decision analysis network. Ecol. Soc. 2010, 15, 10. [Google Scholar] [CrossRef]
- Andersen, M.C.; Martin, B.J.; Roemer, G.W. Use of matrix population models to estimate the efficacy of euthanasia versus trap-neuter-return for management of free-roaming cats. J. Am. Vet. Med. Assoc. 2004, 225, 1871–1876. [Google Scholar] [CrossRef]
- Levy, J.K.; Crawford, P.C. Humane strategies for controlling feral cat populations. J. Am. Vet. Med. Assoc. 2004, 9, 1354–1360. [Google Scholar] [CrossRef]
- Levy, J.K.; Gale, D.W.; Gale, L.A. Evaluation of the effect of a long-term trap-neuter-return and adoption program on a free-roaming cat population. J. Am. Vet. Med. Assoc. 2003, 222, 42–46. [Google Scholar] [CrossRef]
- Spehar, D.; Wolf, P. A case study in citizen science: The effectiveness of a trap-neuter-return program in a Chicago neighborhood. Animals 2018, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Zaunbrecher, K.I.; Smith, R.E. Neutering of feral cats as an alternative to eradication programs. J. Am. Vet. Med. Assoc. 1993, 203, 449–452. [Google Scholar]
- Winter, L. Trap-neuter-release programs: The reality and the impacts. J. Am. Vet. Med. Assoc. 2004, 225, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Bissonnette, V.; Lussier, B.; Doizé, B.; Arsenault, J. Impact of a trap-neuter-return event on the size of free-roaming cat colonies around barns and stables in Quebec: A randomized controlled trial. Can. J. Vet. Res. 2018, 82, 192–197. [Google Scholar]
- Natoli, E.; Maragliano, L.; Cariola, G.; Faini, A.; Bonanni, R.; Cafazzo, S.; Fantini, C. Management of feral domestic cats in the urban environment of Rome (Italy). Prev. Vet. Med. 2006, 77, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Kilgour, R.J.; Magle, S.B.; Slater, M.; Christian, A.; Weiss, E.; DiTullio, M. Estimating free-roaming cat populations and the effects of one year Trap-Neuter-Return management effort in a highly urban area. Urban Ecosyst. 2017, 20, 207–216. [Google Scholar] [CrossRef]
- Castillo, D.; Clarke, A.L. Trap/Neuter/Release methods ineffective in controlling domestic cat “colonies” on pubic lands. Nat. Areas J. 2003, 23, 247–253. [Google Scholar]
- Neville, P.F.; Remfry, J. Effect of neutering on two groups of feral cats. Vet. Rec. 1984, 114, 447–450. [Google Scholar] [CrossRef]
- Lord, L.K.; Wittum, T.E.; Ferketich, A.K.; Funk, J.A.; Rajala-Schultz, P.; Kauffman, R.M. Demographic trends for animal care and control agencies in Ohio from 1996 to 2004. J. Am. Vet. Med. Assoc. 2006, 229, 48–54. [Google Scholar] [CrossRef]
- Royal Society for the Prevention of Cruelty to Animals. Available online: http://www.rspca.org.au/facts/annual-statistics/published-statistics (accessed on 1 April 2019).
- Legge, S.; Murphy, B.P.; McGregor, H.; Woinarski, J.C.Z.; Augusteyn, J.; Ballard, G.; Baseler, M.; Buckmaster, T.; Dickman, C.R.; Doherty, T.; et al. Enumerating a continental-scale threat: How many feral cats are in Australia? Biol. Conserv. 2017, 206, 293–303. [Google Scholar] [CrossRef]
- Nutter, F.B. Evaluation of a Trap-Neuter-Return Management Program for Feral Cat Colonies: Population Dynamics, Home Ranges, and Potentially Zoonotic Diseases; North Carolina State University: Ann Arbor, MI, USA, 2006. [Google Scholar]
- Nutter, F.B.; Stoskopf, M.K.; Levine, J.F. Time and financial costs of programs for live trapping feral cats. J. Am. Vet. Med. Assoc. 2004, 225, 1403–1405. [Google Scholar] [CrossRef] [PubMed]
- Webb, C. Trap, neuter, release: A cat welfare organisation’s perspective. In Proceedings of the 15th National Conference on Urban Animal Management in Australia, Hobart, Australia, 30 Auguest–1 September 2006. [Google Scholar]
- Lohr, C.A.; Cox, L.J.; Lepczyk, C.A. Costs and benefits of trap-neuter-release and euthanasia for removal of urban cats in Oahu, Hawaii. Conserv. Biol. 2013, 27, 64–73. [Google Scholar] [CrossRef]
- Zito, S.; Vankan, D.; Bennett, P.; Paterson, M.; Phillips, C. Cat ownership perception and caretaking explored in an internet survey of people associated with cats. PLoS ONE 2015, 10, e0133293. [Google Scholar] [CrossRef] [PubMed]
- Finkler, H.; Terkel, J. Dichotomy in the emotional approaches of caretakers of free-roaming cats in urban feeding groups: Findings from in-depth interviews. Anthrozoos 2011, 24, 203–218. [Google Scholar] [CrossRef]
- Toukhsati, S.R.; Bennett, P.C.; Coleman, G.J. Behaviors and attitudes towards semi-owned cats. Anthrozoos 2007, 20, 131–142. [Google Scholar] [CrossRef]
- Sharp, A.; Hartnett, N. Semi-Owned Cat Attitudes and Behaviours in South Australia; Dog and Cat Management Board: Adelaide, Australia, 2009; p. 44. [Google Scholar]
- Finkler, H.; Hatna, E.; Terkel, J. The impact of anthropogenic factors on the behavior, reproduction, management and welfare of urban, free-roaming cat populations. Anthrozoos 2011, 24, 31–49. [Google Scholar] [CrossRef]
- Gilhofer, E.M.; Windschnurer, I.; Troxler, J.; Heizmann, V. Welfare of feral cats and potential influencing factors. J. Vet. Behav. 2019, 30, 114–123. [Google Scholar] [CrossRef]
- Berry, C.; Patronek, G.; Lockwood, R. Long-term outcomes in animal hoarding cases. Anim. Law 2005, 11, 167–194. [Google Scholar]
- Halls, V. Understanding animal hoarding. Feline Focus 2017, 3, 243–247. [Google Scholar]
- Taetzsch, S.J.; Bertke, A.S.; Gruszynski, K.R. Zoonotic disease transmission associated with feral cats in a metropolitan area: A geospatial analysis. Zoonoses Pub. Health 2018, 65, 412–419. [Google Scholar] [CrossRef]
- Gilardi, K.; Gillin, C.; Greenacre, C.B.; Groskin, R.; Jessup, D.A.; Karesh, W.B.; Mader, D.; Souza, M.J.; Wolff, P.L.; Ziccardi, M.; et al. Feral cat policy is about more than cats. J. Am. Vet. Med. Assoc. 2015, 246, 402. [Google Scholar] [PubMed]
- Rodan, I. Understanding feline behavior and application for appropriate handling and management. Top. Companion Anim. Med. 2010, 25, 178–188. [Google Scholar] [CrossRef]
- Anseeuw, E.; Apker, C.; Ayscue, C.; Barker, L.; Blair, D.; Brennan, J.; Brooks, S.; Case-Pall, D.; Caspersen, H.; Clark, J.; et al. Handling cats humanely in the veterinary hospital. J. Vet. Behav. Clin. Appl. Res. 2006, 1, 84–88. [Google Scholar] [CrossRef]
- Anaesthesia of Cats; Hall, L.W., Taylor, P.M., Eds.; Baillière-Tindall: Avon, UK, 1994; p. 362. [Google Scholar]
- Teixeira, C.P.; de Azevedo, C.S.; Mendl, M.; Cipreste, C.F.; Young, R.J. Revisiting translocation and reintroduction programmes: The importance of considering stress. Anim. Behav. 2007, 73, 1–13. [Google Scholar] [CrossRef]
- Gourkow, N.; Hamon, S.C.; Phillips, C.J.C. Effect of gentle stroking and vocalization on behaviour, mucosal immunity and upper respiratory disease in anxious shelter cats. Prev. Vet. Med. 2014, 117, 266–275. [Google Scholar] [CrossRef]
- Gourkow, N.; Fraser, D. The effect of housing and handling practices on the welfare, behaviour and selection of domestic cats (Felis sylvestris catus) by adopters in an animal shelter. Anim. Welf. 2006, 15, 371–377. [Google Scholar]
- National Centre for the Replacement, Refinement and Reduction of Animals in Research (NC3Rs). Available online: https://www.nc3rs.org.uk/3rs-resources/housing-and-husbandry/housing-and-husbandry-cats (accessed on 1 April 2019).
- Mozes, R.; Pearl, D.L.; Niel, L.; Weese, J.S. Epidemiological investigation of euthanasia in an Ontario animal shelter. J. Feline Med. Surg. 2018, 20, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Berny, P.; Caloni, F.; Croubels, S.; Sachana, M.; Vandenbroucke, V.; Davanzo, F.; Guitart, R. Animal poisoning in Europe. Part 2: Companion animals. Vet. J. 2010, 183, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Milewski, L.M.; Khan, S.A. An overview of potentially life-threatening poisonous plants in dogs and cats. J. Vet. Emerg. Crit. Care 2006, 16, 25–33. [Google Scholar] [CrossRef]
- Giuliano Albo, A.; Nebbia, C. Incidence of poisonings in domestic carnivores in Italy. Vet. Res. Commun. 2004, 28, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Loyd, K.A.T.; Hernandez, S.M.; Abernathy, K.J.; Shock, B.C.; Marshall, G.J. Risk behaviours exhibited by free-roaming cats in a suburban US town. Vet. Rec. 2013, 173, 295–300. [Google Scholar] [CrossRef] [PubMed]
- DeClementi, C.; Sobczak, B.R. Common rodenticide toxicoses in small animals. Vet. Clin. N. Am. Small Anim. Pract. 2012, 42, 349–360. [Google Scholar] [CrossRef]
- Calver, M.C.; Adams, G.; Clark, W.; Pollock, K.H. Assessing the safety of collars used to attach predation deterrent devices and ID tags to pet cats. Anim. Welf. 2013, 22, 95–105. [Google Scholar] [CrossRef]
- McDonald, J.L.; Cleasby, I.R.; Brodbelt, D.C.; Church, D.B.; O’Neill, D.G. Mortality due to trauma in cats attending veterinary practices in central and south-east England. J. Small Anim. Pract. 2017, 58, 570–576. [Google Scholar] [CrossRef]
- Wilson, J.L.; Gruffydd-Jones, T.; Murray, J.K. Risk factors for road traffic accidents in cats up to age 12 months that were registered between 2010 and 2013 with the UK Pet Cat Cohort (“Bristol Cats”). Vet. Rec. 2017, 180, 195. [Google Scholar] [CrossRef]
- Warner, R.E. Demography and movements of free-ranging domestic cats in rural Illinois. J. Wildl. Manag. 1985, 49, 340–346. [Google Scholar] [CrossRef]
- Grubbs, S.E.; Krausman, P.R. Observations of coyote-cat interactions. J. Wildl. Manag. 2009, 73, 683–685. [Google Scholar] [CrossRef]
- Shamir, M.H.; Leisner, S.; Klement, E.; Gonen, E.; Johnston, D.E. Dog bite wounds in dogs and cats: A retrospective study of 196 cases. J. Vet. Med. Ser. 2002, 49, 107–112. [Google Scholar] [CrossRef]
- Finkler, H.; Gunther, I.; Terkel, J. Behavioral differences between urban feeding groups of neutered and sexually intact free-roaming cats following a trap-neuter-return procedure. J. Am. Vet. Med. Assoc. 2011, 238, 1141–1149. [Google Scholar] [CrossRef]
- Gunther, I.; Finkler, H.; Terkel, J. Demographic differences between urban feeding groups of neutered and sexually intact free-roaming cats following a trap-neuter-return procedure. J. Am. Vet. Med. Assoc. 2011, 238, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, R. Cruelty Toward Cats: Changing Perspectives. In The State of the Animals III; Salem, D.J., Rowan, A.N., Eds.; Humane Society Press: Washington, DC, USA, 2005; pp. 15–26. [Google Scholar]
- Pedersen, N.C. Feline Infectious Diseases; American Veterinary Publications, Inc.: Berkeley, CA, USA, 1988; p. 404. [Google Scholar]
- Arbabi, M.; Hooshyar, H. Gastrointestinal parasites of stray cats in Kashan, Iran. Trop. Biomed. 2009, 26, 16–22. [Google Scholar]
- Takeuchi-Storm, N.; Mejer, H.; Al-Sabi, M.N.S.; Olsen, C.S.; Thamsborg, S.M.; Enemark, H.L. Gastrointestinal parasites of cats in Denmark assessed by necropsy and concentration McMaster technique. Vet. Parasitol. 2015, 214, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Abu-Madi, M.A.; Behnke, J.M.; Prabhaker, K.S.; Al-Ibrahim, R.; Lewis, J.W. Intestinal helminths of feral cat populations from urban and suburban districts of Qatar. Vet. Parasitol. 2010, 168, 284–292. [Google Scholar] [CrossRef]
- Waap, H.; Gomes, J.; Nunes, T. Parasite communities in stray cat populations from Lisbon, Portugal. J. Helminthol. 2014, 88, 389–395. [Google Scholar] [CrossRef]
- Zito, S.; Walker, J.; Gates, M.C.; Dale, A. A preliminary description of companion cat, managed stray cat, and unmanaged stray cat welfare in Auckland, New Zealand using a 5-component assessment scale. Front. Vet. Sci. 2019, 6, 1–10. [Google Scholar] [CrossRef]
- Wilson, P.M.; Tidemann, C.R.; Meischke, H.R.C. Are Cats on Rubbish Dumps a Problem? Australian Veterinary: Sydney, Australia, 1994; pp. 163–174. [Google Scholar]
- Palmer, C.S.; Thompson, R.C.A.; Traub, R.J.; Rees, R.; Robertson, I.D. National study of the gastrointestinal parasites of dogs and cats in Australia. Vet. Parasitol. 2008, 151, 181–190. [Google Scholar] [CrossRef]
- Canto, G.J.; Guerrero, R.I.; Olvera-Ramírez, A.M.; Milian, F.; Mosqueda, J.; Aguilar-Tipacamu, G. Prevalence of fleas and gastrointestinal parasites in free-roaming cats in central Mexico. PLoS ONE 2013, 8, e60744. [Google Scholar] [CrossRef]
- Duarte, A.; Castro, I.; da Fonseca, I.M.P.; Almeida, V.; de Carvalho, L.M.M.; Meireles, J.; Fazendeiro, M.I.; Tavares, L.; Vaz, Y. Survey of infectious and parasitic diseases in stray cats at the Lisbon Metropolitan Area, Portugal. J. Feline Med. Surg. 2010, 12, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Gracenea, M.; Gómez, M.S.; Torres, J. Prevalence of intestinal parasites in shelter dogs and cats in the metropolitan area of Barcelona (Spain). Acta Parasitol. 2009, 54, 73–77. [Google Scholar] [CrossRef]
- Rodríguez-Ponce, E.; González, J.F.; de Felipe, M.C.; Hernández, J.N.; Jaber, J.R. Epidemiological survey of zoonotic helminths in feral cats in Gran Canaria island (Macaronesian archipelago—Spain). Acta Parasitol. 2016, 61, 443–450. [Google Scholar] [CrossRef]
- Zanzani, S.A.; Gazzonis, A.; Magistrelli, S.; Manfredi, M.T. Pulmonary and intestinal parasites in colony cats as markers for biodiversity in an urban area. Urban Ecosyst. 2015, 18, 1415–1425. [Google Scholar] [CrossRef]
- Lefkaditis, M.A.; Paştiu, A.I.; Rodi-Buriel, A.; Sossidou, A.V.; Panorias, A.H.; Eleftheriadis, T.G.; Cozma, V.; Mihalca, A.D. Helminth burden in stray cats from Thessaloniki, Greece. Helminthologia 2014, 51, 73–76. [Google Scholar] [CrossRef]
- Becker, A.C.; Rohen, M.; Epe, C.; Schnieder, T. Prevalence of endoparasites in stray and fostered dogs and cats in Northern Germany. Parasitol. Res. 2012, 111, 849–857. [Google Scholar] [CrossRef]
- El-Seify, M.A.; Aggour, M.G.; Sultan, K.; Marey, N.M. Gastrointestinal helminths of stray cats in Alexandria, Egypt: A fecal examination survey study. Vet. Parasitol. Reg. Stud. Rep. 2017, 8, 104–106. [Google Scholar] [CrossRef]
- Khalafalla, R.E. A survey study on gastrointestinal parasites of stray cats in northern region of Nile Delta, Egypt. PLoS ONE 2011, 6, e20283. [Google Scholar] [CrossRef]
- Khademvatan, S.; Rahman Abdizadeh, F.R.; Hashemitabar, M.; Ghasemi, M.; Tavalla, M. Stray cats gastrointestinal parasites and its association with public health in Ahvaz City, South Western of Iran. Jundishapur J. Microbiol. 2014, 7, e11079. [Google Scholar] [CrossRef]
- Borji, H.; Razmi, G.; Ahmadi, A.; Karami, H.; Yaghfoori, S.; Abedi, V. A survey on endoparasites and ectoparasites of stray cats from Mashhad (Iran) and association with risk factors. J. Parasit. Dis. 2011, 35, 202–206. [Google Scholar] [CrossRef]
- Jamshidi, S.; Meshki, B.; Meshki, M. A study of helminthic infection of gastrointestinal tract in stray cats at urban areas in Isfahan. J. Vet. Res. 2002, 57, 25–27. [Google Scholar]
- Schuster, R.K.; Thomas, K.; Sivakumar, S.; O’Donovan, D. The parasite fauna of stray domestic cats (Felis catus) in Dubai, United Arab Emirates. Parasitol. Res. 2009, 105, 125–134. [Google Scholar] [CrossRef]
- Borthakur, S.K.; Mukharjee, S.N. Gastrointestinal helminthes in stray cats (Felis catus) from Aizawl, Mizoram, India. Southeast Asian J. Trop. Med. Pub. Health 2011, 42, 255. [Google Scholar]
- Mohd Zain, S.N.; Sahimin, N.; Pal, P.; Lewis, J.W. Macroparasite communities in stray cat populations from urban cities in Peninsular Malaysia. Vet. Parasitol. 2013, 196, 469–477. [Google Scholar] [CrossRef]
- Lefkaditis, M.A.; Sossidou, A.V.; Panorias, A.H.; Koukeri, S.E.; Paştiu, A.I.; Athanasiou, L.V. Urban stray cats infested by ectoparasites with zoonotic potential in Greece. Parasitol. Res. 2015, 114, 3931–3934. [Google Scholar] [CrossRef]
- Salant, H.; Mumcuoglu, K.Y.; Baneth, G. Ectoparasites in urban stray cats in Jerusalem, Israel: Differences in infestation patterns of fleas, ticks and permanent ectoparasites. Med. Vet. Entomol. 2013, 28, 314–318. [Google Scholar] [CrossRef]
- Thomas, J.E.; Staubus, L.; Goolsby, J.L.; Reichard, M.V. Ectoparasites of free-roaming domestic cats in the central United States. Vet. Parasitol. 2016, 228, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Shearer, D. Veterinary Ectoparasites: Biology, Pathology and Control, 2nd ed.; Blackwell Science: Oxford, UK, 2001; p. 275. [Google Scholar]
- Barrs, V.R.; Beatty, J.A.; Wilson, B.J.; Evans, N.; Gowan, R.; Baral, R.M.; Lingard, A.E.; Perkovic, G.; Hawley, J.R.; Lappin, M.R. Prevalence of Bartonella species, Rickettsia felis, haemoplasmas and the Ehrlichia group in the blood of cats and fleas in eastern Australia. Aust. Vet. J. 2010, 88, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Schloderer, D.; Owen, H.; Clark, P.; Stenos, J.; Fenwick, S.G. Rickettsia felis in fleas, Western Australia. Emerg. Infect. Dis. 2006, 12, 841–843. [Google Scholar] [CrossRef]
- Shaw, S.E.; Kenny, M.J.; Tasker, S.; Birtles, R.J. Pathogen carriage by the cat flea Ctenocephalides felis (Bouché) in the United Kingdom. Vet. Microbiol. 2004, 102, 183–188. [Google Scholar] [CrossRef]
- Akucewich, L.H.; Philman, K.; Clark, A.; Gillespie, J.; Kunkle, G.; Nicklin, C.F.; Greiner, E.C. Prevalence of ectoparasites in a population of feral cats from north central Florida during the summer. Vet. Parasitol. 2002, 109, 129–139. [Google Scholar] [CrossRef]
- Sommerfelt, I.E.; Cardillo, N.; López, C.; Ribicich, M.; Gallo, C.; Franco, A. Prevalence of Toxocara cati and other parasites in cats’ faeces collected from the open spaces of public institutions: Buenos Aires, Argentina. Vet. Parasitol. 2006, 140, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P. Toxoplasmosis of Animals and Humans, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2009; p. 336. [Google Scholar] [CrossRef]
- Van Bree, F.P.J.; Bokken, G.C.A.M.; Mineur, R.; Franssen, F.; Opsteegh, M.; van der Giessen, J.W.B.; Lipman, L.J.A.; Overgaauw, P.A.M. Zoonotic bacteria and parasites found in raw meat-based diets for cats and dogs. Vet. Rec. 2018, 182, 50. [Google Scholar] [CrossRef]
- Romich, J.A. Understanding Zoonotic Diseases; Thomson Delmar Learning: Ontario, ON, Canada, 2008; p. 701. [Google Scholar]
- Tiao, N.; Darrington, C.; Molla, B.; Saville, W.J.A.; Tilahun, G.; Kwok, O.C.H.; Gebreyes, W.A.; Lappin, M.R.; Jones, J.L.; Dubey, J.P. An investigation into the seroprevalence of Toxoplasma gondii, Bartonella spp., feline immunodeficiency virus (FIV), and feline leukaemia virus (FeLV) in cats in Addis Ababa, Ethiopia. Epidemiol. Infect. 2013, 141, 1029–1033. [Google Scholar] [CrossRef]
- Haddadzadeh, H.R.; Khazraiinia, P.; Aslani, M.; Rezaeian, M.; Jamshidi, S.; Taheri, M.; Bahonar, A. Seroprevalence of Toxoplasma gondii infection in stray and household cats in Tehran. Vet. Parasitol. 2006, 138, 211–216. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, D.H.; Zhou, P.; Lun, Z.R.; Chen, X.G.; Lin, R.Q.; Yuan, Z.G.; Zhu, X.Q. Seroprevalence of Toxoplasma gondii infection in stray and household cats in Guangzhou, China. Zoonoses Pub. Health 2009, 56, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Miró, G.; Montoya, A.; Jiménez, S.; Frisuelos, C.; Mateo, M.; Fuentes, I. Prevalence of antibodies to Toxoplasma gondii and intestinal parasites in stray, farm and household cats in Spain. Vet. Parasitol. 2004, 126, 249–255. [Google Scholar] [CrossRef]
- Fancourt, B.A.; Jackson, R.B. Regional seroprevalence of Toxoplasma gondii antibodies in feral and stray cats (Felis catus) from Tasmania. Aust. J. Zool. 2014, 62, 272–283. [Google Scholar] [CrossRef]
- Foster, S.F.; Martin, P.; Allan, G.S.; Barrs, V.R.; Malik, R. Lower respiratory tract infections in cats: 21 cases (1995–2000). J. Feline Med. Surg. 2004, 6, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Sumner, B.; Ackland, M.L. Toxoplasma gondii antibody in domestic cats in Melbourne. Aust. Vet. J. 1999, 77, 447–449. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.; Thompson, R.C.A.; Grigg, M.E.; Sundar, N.; Smith, A.; Lymbery, A.J. Western Australian marsupials are multiply infected with genetically diverse strains of Toxoplasma gondii. PLoS ONE 2012, 7, e45147. [Google Scholar] [CrossRef] [PubMed]
- Infectious Diseases of the Dog and Cat, 4th ed.; Greene, C.E., Ed.; Elsevier Health Sciences: St. Louis, MO, USA, 2012; p. 1354. [Google Scholar]
- Yamamoto, J.K.; Hansen, H.; Ho, E.W.; Morishita, T.Y.; Okuda, T.; Sawa, T.R.; Nakamura, R.M.; Pedersen, N.C. Epidemiologic and clinical aspects of feline immunodeficiency virus infection in cats from the continental United States and Canada and possible mode of transmission. J. Am. Vet. Med. Assoc. 1989, 194, 213–220. [Google Scholar] [PubMed]
- Pontier, D.; Fromont, E.; Courchamp, F.; Artois, M.; Yoccoz, N.G. Retroviruses and sexual size dimorphism in domestic cats (Felis catus L.). Proc. R. Soc. Biol. Sci. 1998, 265, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Akhtardanesh, B.; Ziaali, N.; Sharifi, H.; Rezaei, S. Feline immunodeficiency virus, feline leukemia virus and Toxoplasma gondii in stray and household cats in Kerman–Iran: Seroprevalence and correlation with clinical and laboratory findings. Res. Vet. Sci. 2010, 89, 306–310. [Google Scholar] [CrossRef]
- Luria, B.J.; Levy, J.K.; Lappin, M.R.; Breitschwerdt, E.B.; Legendre, A.M.; Hernandez, J.A.; Gorman, S.P.; Lee, I.T. Prevalence of infectious diseases in feral cats in Northern Florida. J. Feline Med. Surg. 2004, 6, 287–296. [Google Scholar] [CrossRef]
- Lutz, H.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Hosie, M.J.; Lloret, A.; et al. Feline leukaemia: ABCD guidelines on prevention and management. J. Feline Med. Surg. 2009, 11, 565–574. [Google Scholar] [CrossRef]
- Truyen, U.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Hosie, M.J.; Lloret, A.; et al. Feline panleukopenia: ABCD guidelines on prevention and management. J. Feline Med. Surg. 2009, 11, 538–546. [Google Scholar] [CrossRef]
- Lopes, P.C. When is it socially acceptable to feel sick? Proc. R. Soc. Biol. Sci. 2014, 281, 20140218. [Google Scholar] [CrossRef] [PubMed]
- Seo, A.; Tanida, H. Three-year route census study on welfare status of free-roaming cats in old-town Onomichi, Japan. J. Appl. Anim. Welf. Sci. 2018, 21, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Castro-Prieto, J.; Andrade-Nunez, M.J. Health and ecological aspects of stray cats in Old San Juan, Puerto Rico: Baseline information to develop an effective control program. P. R. Health Sci. J. 2018, 37, 110–114. [Google Scholar]
- Charleston, L.J. Sydney’s Escalating Cat Crisis. Available online: https://www.news.com.au/technology/science/animals/sydneys-escalating-cat-crisis/news-story/67733dbe685e28f3afdcda64435b75fa (accessed on 12 January 2019).
- Campos, C.B.; Esteves, C.F.; Ferraz, K.M.P.M.B.; Crawshaw, P.G.; Verdade, L.M. Diet of free-ranging cats and dogs in a suburban and rural environment, south-eastern Brazil. J. Zool. Proc. Zool. Soc. Lond. 2007, 273, 14–20. [Google Scholar] [CrossRef]
- Brickner-Braun, I.; Geffen, E.; Yom-Tov, Y. The domestic cat as a predator of Israeli wildlife. Isr. J. Ecol. Evol. 2007, 53, 129–142. [Google Scholar] [CrossRef]
- Hutchings, S. The diet of feral house cats (Felis catus) at a regional rubbish tip, Victoria. Wildl. Res. 2003, 30, 103–110. [Google Scholar] [CrossRef]
- Morris, J.G.; Rogers, Q.R.; Pacioretty, L.M. Taurine: An essential nutrient for cats. J. Small Anim. Pract. 1990, 31, 502–509. [Google Scholar] [CrossRef]
- Lepczyk, C.A.; Lohr, C.A.; Duffy, D.C. A review of cat behavior in relation to disease risk and management options. Appl. Anim. Behav. Sci. 2015, 173, 29–39. [Google Scholar] [CrossRef]
- Alonso, J.M.; Stein, M.; Chamorro, M.C.; Bojanich, M.V. Contamination of soils with eggs of Toxocara in a subtropical city in Argentina. J. Helminthol. 2001, 75, 165–168. [Google Scholar] [CrossRef]
- Uga, S.; Minami, T.; Nagata, K. Defecation habits of cats and dogs and contamination by Toxocara eggs in public park sandpits. Am. J. Trop. Med. Hyg. 1996, 54, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Dubná, S.; Langrová, I.; Jankovská, I.; Vadlejch, J.; Pekár, S.; Nápravník, J.; Fechtner, J. Contamination of soil with Toxocara eggs in urban (Prague) and rural areas in the Czech Republic. Vet. Parasitol. 2007, 144, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.A.; Sischo, W.M.; Hunter, A.; Miles, T. Animal bite epidemiology and surveillance for rabies postexposure prophylaxis. J. Am. Vet. Med. Assoc. 2000, 217, 190–194. [Google Scholar] [CrossRef]
- Sparkes, J.; Fleming, P.J.; Ballard, G.; Scott-Orr, H.; Durr, S.; Ward, M.P. Canine rabies in Australia: A review of preparedness and research needs. Zoonoses Pub. Health 2015, 62, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Gage, K.L.; Dennis, D.T.; Orloski, K.A.; Ettestad, P.; Brown, T.L.; Reynolds, P.J.; Pape, W.J.; Fritz, C.L.; Carter, L.G.; Stein, J.D. Cases of cat-associated human plague in the Western US, 1977–1998. Clin. Infect. Dis. 2000, 30, 893–900. [Google Scholar] [CrossRef]
- Rimmelzwaan, G.F.; van Riel, D.; Baars, M.; Bestebroer, T.M.; van Amerongen, G.; Fouchier, R.A.; Osterhaus, A.D.; Kuiken, T. Influenza A virus (H5N1) infection in cats causes systemic disease with potential novel routes of virus spread within and between hosts. Am. J. Pathol. 2006, 168, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Moore, S. Cat confinement—Does it work? In Proceedings of the 10th National Conference on Urban Animal Management in Australia, Melbourne, Australia; pp. 57–61. Available online: http://aiam.org.au/resources/Documents/2001%20UAM/PUB_Pro01_StevenMoore.pdf (accessed on 16 April 2019).
- Crawford, H.M.; Fontaine, J.B.; Calver, M.C. Ultrasonic deterrents reduce nuisance cat (Felis catus) activity on suburban properties. Glob. Ecol. Conserv. 2018, 15, e00444. [Google Scholar] [CrossRef]
- Government of Western Australia. Cat Act 2011. Available online: www.legislation.wa.gov.au (accessed on 1 April 2019).
- Barratt, D.G. Home range size, habitat utilisation and movement patterns of suburban and farm cats Felis catus. Ecography 1997, 20, 271–280. [Google Scholar] [CrossRef]
- Hall, C.M.; Bryant, K.A.; Haskard, K.; Major, T.; Bruce, S.J.; Calver, M.C. Factors determining the home ranges of pet cats: A meta-analysis. Biol. Conserv. 2016, 203, 313–320. [Google Scholar] [CrossRef]
- Hanmer, H.J.; Thomas, R.L.; Fellowes, M.D.E. Urbanisation influences range size of the domestic cat (Felis catus): Consequences for conservation. J. Urban Ecol. 2017, 3, 1–11. [Google Scholar] [CrossRef]
- Hart, B.L.; Barrett, R.E. Effects of castration on fighting, roaming, and urine spraying in adult male cats. J. Am. Vet. Med. Assoc. 1973, 163, 290–292. [Google Scholar]
- Hart, B.L.; Cooper, L. Factors relating to urine spraying and fighting in prepubertally gonadectomized cats. J. Am. Vet. Med. Assoc. 1984, 184, 1255–1258. [Google Scholar] [PubMed]
- McCune, S. The impact of paternity and early socialisation on the development of cats’ behaviour to people and novel objects. Appl. Anim. Behav. Sci. 1995, 45, 109–124. [Google Scholar] [CrossRef]
- Oehler, R.L.; Velez, A.P.; Mizrachi, M.; Lamarche, J.; Gompf, S. Bite-related and septic syndromes caused by cats and dogs. Lancet Infect. Dis. 2009, 9, 439–447. [Google Scholar] [CrossRef]
- Dog and Cat Management Board. Dog and Cat Management Board: 2016–17 Year In Review; Government of South Australia: South Australia, Australia, 2017.
- Scarlett, J.M.; Salman, M.D.; New, J.G.; Kass, P.H. Reasons for relinquishment of companion animals in U.S. animal shelters: Selected health and personal issues. J. Appl. Anim. Welf. Sci. 1999, 2, 41–57. [Google Scholar] [CrossRef]
- Casey, R.A.; Vandenbussche, S.; Bradshaw, J.W.S.; Roberts, M.A. Reasons for relinquishment and return of domestic cats (Felis silvestris catus) to rescue shelters in the UK. Anthrozoos 2009, 22, 347–358. [Google Scholar] [CrossRef]
- Love, D.N.; Malik, R.; Norris, J.M. Bacteriological warfare amongst cats: What have we learned about cat bite infections? Vet. Microbiol. 2000, 74, 179–193. [Google Scholar] [CrossRef]
- Costanzo, C.; Garosi, L.S.; Glass, E.N.; Rusbridge, C.; Stalin, C.E.; Volk, H.A. Brain abscess in seven cats due to a bite wound: MRI findings, surgical management and outcome. J. Feline Med. Surg. 2011, 13, 672–680. [Google Scholar] [CrossRef]
- McGlade, T.R.; Robertson, I.D.; Elliot, A.D.; Read, C.; Thompson, R.C.A. Gastrointestinal parasites of domestic cats in Perth, Western Australia. Vet. Parasitol. 2003, 117, 251–262. [Google Scholar] [CrossRef]
- Loyd, K.A.T.; Hernandez, S.M.; Carroll, J.P.; Abernathy, K.J.; Marshall, G.J. Quantifying free-roaming domestic cat predation using animal-borne video cameras. Biol. Conserv. 2013, 160, 183–189. [Google Scholar] [CrossRef]
- Adamec, R.E. The interaction of hunger and preying in the domestic cat (Felis catus): An adaptive hierarchy? Behav. Biol. 1976, 18, 263–272. [Google Scholar] [CrossRef]
- McRuer, D.L.; Gray, L.C.; Horne, L.; Clark, E.E. Free-roaming cat interactions with wildlife admitted to a wildlife hospital. J. Wildl. Manag. 2017, 81, 163–173. [Google Scholar] [CrossRef]
- Bonnington, C.; Gaston, K.J.; Evans, K.L. Fearing the feline: Domestic cats reduce avian fecundity through trait-mediated indirect effects that increase nest predation by other species. J. Appl. Ecol. 2013, 50, 15–24. [Google Scholar] [CrossRef]
- van Heezik, Y.M.; Smyth, A.; Adams, A.; Gordon, J.K. Do domestic cats impose an unsustainable harvest on urban bird populations? Biol. Conserv. 2010, 143, 121–130. [Google Scholar] [CrossRef]
- Australian Government. Delma impar—Striped Legless Lizard. Available online: http://www.environment.gov.au/cgi-bin/sprat/public/publicspecies.pl?taxon_id=1649 (accessed on 20 January 2019).
- Osborne, W.; Williams, K. Conservation problems with cats in the Australian Capital Territory. In Proceedings of the Impact of Cats on Native Wildlife, Canberra, Australia, 8–9 May 1991. [Google Scholar]
- Australian Government. Perameles gunnii Victorian Subspecies—Eastern Barred Bandicoot (Mainland). Available online: http://www.environment.gov.au/cgi-bin/sprat/public/publicspecies.pl?taxon_id=88020 (accessed on 20 January 2019).
- Dufty, A.C. Population demography of the eastern barred bandicoot (Perameles gunnii) at Hamilton, Victoria. Wildl. Res. 1994, 21, 445–457. [Google Scholar] [CrossRef]
- Barratt, D.G. Predation by house cats, Felis catus (L.), in Canberra, Australia. I. Prey composition and preference. Wildl. Res. 1997, 24, 263–277. [Google Scholar] [CrossRef]
- Brunner, H.; Moro, D.; Wallis, L.R.; Andrasel, A. Comparison of the diets of foxes, dogs and cats in an urban park. Vic. Nat. 1991, 108, 35–37. [Google Scholar]
- Calver, M.; Thomas, S.; Bradley, S.; McCutcheon, H. Reducing the rate of predation on wildlife by pet cats: The efficacy and practicability of collar-mounted pounce protectors. Biol. Conserv. 2007, 137, 341–348. [Google Scholar] [CrossRef]
- Dickman, C.R. Overview of the Impacts of Feral Cats on Australian Native Fauna; Australian Nature Conservation Agency: Canberra, Australia, 1996; p. 92. [Google Scholar]
- Hall, C.M.; Fontaine, J.B.; Bryant, K.A.; Calver, M.C. Assessing the effectiveness of the Birdsbesafe® anti-predation collar cover in reducing predation on wildlife by pet cats in Western Australia. Appl. Anim. Behav. Sci. 2015, 173, 40–51. [Google Scholar] [CrossRef]
- Paton, D.C. Loss of wildlife to domestic cats. In Proceedings of the Impact of Cats on Native Wildlife, Canberra, Australia, 8–9 May 1991. [Google Scholar]
- Ives, C.D.; Lentini, P.E.; Threlfall, C.G.; Ikin, K.; Shanahan, D.F.; Garrard, G.E.; Bekessy, S.A.; Fuller, R.A.; Mumaw, L.; Rayner, L.; et al. Cities are hotspots for threatened species. Glob. Ecol. Biogeogr. 2016, 25, 117–126. [Google Scholar] [CrossRef]
- Bryant, G.L.; Kobryn, H.T.; Hardy, G.E.S.J.; Fleming, P.A. Habitat islands in a sea of urbanisation. Urban For. Urban Green. 2017, 28, 131–137. [Google Scholar] [CrossRef]
- Bamford, M.J.; Calver, M.C. Cat predation and suburban lizards: A 22 year study at a suburban Australian property. Open Conserv. Biol. J. 2012, 6, 25–29. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Thomas, S.; Wright, J.W. Bells reduce predation of wildlife by domestic cats (Felis catus). J. Zool. Proc. Zool. Soc. Lond. 2002, 256, 81–83. [Google Scholar] [CrossRef]
- Nelson, S.H.; Evans, A.D.; Bradbury, R.B. The efficacy of collar-mounted devices in reducing the rate of predation of wildlife by domestic cats. Appl. Anim. Behav. Sci. 2005, 94, 273–285. [Google Scholar] [CrossRef]
- Mills, D.S.; Bailey, S.L.; Thurstans, R.E. Evaluation of the welfare implications and efficacy of an ultrasonic “deterrent” for cats. Vet. Rec. 2000, 147, 678–680. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.H.; Evans, A.D.; Bradbury, R.B. The efficacy of an ultrasonic cat deterrent. Appl. Anim. Behav. Sci. 2006, 96, 83–91. [Google Scholar] [CrossRef]
- Baker, D. Cat curfew—Casey city council. In Proceedings of the 10th National Conference on Urban Animal Management in Australia, Melbourne, Australia; pp. 63–64. Available online: http://aiam.org.au/resources/Documents/2001%20UAM/PUB_Pro01_DaveBaker.pdf (accessed on 1 April 2019).
- Australian Capital Territory Government. Responsible Cat Ownership Community Research, Planning, Conservation and Land (PCL); Nature Conservation Division, Environment and Sustainable Development Directorate: Canberra, Australia, 2011; p. 46.
- Lilith, M.; Calver, M.; Garkaklis, M.J. Roaming habits of pet cats on the suburban fringe in Perth, Western Australia: What size buffer zone is needed to protect wildlife in reserves? In Too Close for Comfort: Contentious Issues in Human-Wildlife Encounters; Lunney, D., Munn, A., Meikle, W., Eds.; Royal Zoological Society of New South Wales: Mosman, Australia, 2008; pp. 65–72. [Google Scholar]
- Feng, A.Y.T.; Himsworth, C.G. The secret life of the city rat: A review of the ecology of urban Norway and black rats (Rattus norvegicus and Rattus rattus). Urban Ecosyst. 2014, 17, 149–162. [Google Scholar] [CrossRef]
- Leyhausen, P. Cat Behavior: The Predatory and Social Behavior of Domestic and Wild Cats; Garland STPM Press: New York, NY, USA, 1979. [Google Scholar]
- Childs, J.E. Size-dependent predation on rats (Rattus norvegicus) by house cats (Felis catus) in an urban setting. J. Mammal. 1986, 67, 196–199. [Google Scholar] [CrossRef]
- Glass, G.E.; Gardner-Santana, L.C.; Holt, R.D.; Chen, J.; Shields, T.M.; Roy, M.; Schachterle, S.; Klein, S.L. Trophic garnishes: Cat-rat interactions in an urban environment. PLoS ONE 2009, 4, e5794. [Google Scholar] [CrossRef]
- Plantinga, E.A.; Bosch, G.; Hendriks, W.H. Estimation of the dietary nutrient profile of free-roaming feral cats: Possible implications for nutrition of domestic cats. Br. J. Nutr. 2011, 106, S35–S48. [Google Scholar] [CrossRef]
- Bradshaw, J.W.S. The evolutionary basis for the feeding behavior of domestic dogs (Canis familiaris) and cats (Felis catus). J. Nutr. 2006, 136, 1927S–1931S. [Google Scholar] [CrossRef] [PubMed]
- Read, J.L.; Bengsen, A.J.; Meek, P.D.; Moseby, K.E. How to snap your cat: Optimum lures and their placement for attracting mammalian predators in arid Australia. Wildl. Res. 2015, 42, 1–12. [Google Scholar] [CrossRef]
- Catling, P.C. Similarities and contrasts in the diets of foxes, Vulpes vulpes, and cats, Felis catus, relative to fluctuating prey populations and drought. Wildl. Res. 1988, 15, 307–317. [Google Scholar] [CrossRef]
- Bradshaw, J.W.S.; Goodwin, D.; Legrand-Defrétin, V.; Nott, H.M.R. Food selection by the domestic cat, an obligate carnivore. Compa. Biochem. Physiol. Part Physiol. 1996, 114, 205–209. [Google Scholar] [CrossRef]
- Bradshaw, J.W.S.; Healey, L.M.; Thorne, C.J.; Macdonald, D.W.; Arden-Clark, C. Differences in food preferences between individuals and populations of domestic cats Felis silvestris catus. Appl. Anim. Behav. Sci. 2000, 68, 257–268. [Google Scholar] [CrossRef]
- Moro, D.; Byrne, M.; Kennedy, M.; Campbell, S.; Tizard, M. Identifying knowledge gaps for gene drive research to control invasive animal species: The next CRISPR step. Glob. Ecol. Conserv. 2018, 13, e00363. [Google Scholar] [CrossRef]
- Zito, S.; Paterson, M.; Morton, J.; Vankan, D.; Bennett, P.; Rand, J.; Phillips, C.J.C. Surrenderers’ relationships with cats admitted to four Australian animal shelters. Animals 2018, 8, 23. [Google Scholar] [CrossRef]
- Grayson, J.; Calver, M.; Styles, I. Attitudes of suburban Western Australians to proposed cat control legislation. Aust. Vet. J. 2002, 80, 536–543. [Google Scholar] [CrossRef]
- Cat Protection Society of New South Wales. The Good Neighbour Project. Available online: https://catprotection.org.au/responsible-cat-ownership/ (accessed on 12 January 2019).
- Animal Welfare League Queensland. National Desexing Network. Available online: https://ndn.org.au/ (accessed on 12 January 2019).
- Zoos Victoria. Safe Cat, Safe Wildlife. Available online: http://www.safecat.org.au/ (accessed on 10 January 2019).
- Webb, C. Australia Asks “Who’s For Cats?”; Victorian Bureau of Animal Welfare: Melbourne, Australia, 2007; p. 26.
- Royal Society for the Prevention of Cruelty to Animals. RSPCA Victoria Annual Report 2010–2011. Available online: http://rspcavic.org/documents/About%20us/Annual%20Report/RSPCA-Annual-Report-2011.pdf (accessed on 1 April 2019).
- Animal Health Alliance (Australia). Pet Ownership in Australia Report 2013. Available online: www.animalhealthalliance.org.au (accessed on 1 April 2019).
- Animal Medicines Australia. Pet Ownership in Australia 2016. Available online: http://animalmedicinesaustralia.org.au/pet-report/ (accessed on 1 April 2019).
- Royal Society for the Prevention of Cruelty to Animals. Australia National Statistics 2007–2017. Available online: https://www.rspca.org.au/facts/annual-statistics-2015-16/published-statistics (accessed on 1 April 2019).
- Crawford, H.M.; Fontaine, J.B.; Calver, M.C. Using free adoptions to reduce crowding and euthanasia at cat shelters: An Australian case study. Animals 2017, 7, 92. [Google Scholar] [CrossRef] [PubMed]
- Zito, S.; Paterson, M.; Vankan, D.; Morton, J.; Bennett, P.; Phillips, C. Determinants of cat choice and outcomes for adult cats and kittens adopted from an Australian animal shelter. Animals 2015, 5, 276–314. [Google Scholar] [CrossRef] [PubMed]
- Root Kustritz, M.V. Determining the optimal age for gonadectomy of dogs and cats. J. Am. Vet. Med. Assoc. 2007, 231, 1665–1675. [Google Scholar] [CrossRef]
- Root Kustritz, M.V. Clinical Canine and Feline Reproduction: Evidence-Based Answers; Wiley & Blackwell: Ames, IA, USA, 2009; p. 334. [Google Scholar]
- Welsh, C.P.; Gruffydd-Jones, T.; Roberts, M.A.; Murray, J.K. Poor owner knowledge of feline reproduction contributes to the high proportion of accidental litters born to UK pet cats. Vet. Rec. 2014, 174, 118–123. [Google Scholar] [CrossRef]
- Crawford, H.M.; Calver, M.C. Attitudes and practices of Australian veterinary professionals and students towards Early Age Desexing of cats. Animals 2019, 9, 2. [Google Scholar] [CrossRef]
- Makin, M.R.; Webb, C.; Studdert, V.P.; Goerke, M. Early Age Desexing of Puppies and Kittens. Australian Veterinary Association and the Minister for Agriculture’s Animal Welfare Advisory Committee. Available online: http://www.gungahlinvet.com.au/petcare-info/publications/early-age-desexing-victorian-seminar.html (accessed on 1 April 2019).
- Root Kustritz, M.V. Early spay-neuter: Clinical considerations. Clin. Tech. Small Anim. Pract. 2002, 17, 124–128. [Google Scholar] [CrossRef]
- Spain, C.V.; Scarlett, J.M.; Houpt, K.A. Long-term risks and benefits of early-age gonadectomy in cats. J. Am. Vet. Med. Assoc. 2004, 224, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Calver, M.C. Prevalence of desexed cats in relation to age in a convenience sample of Western Australian cats. Aust. Vet. J. 2014, 92, 226–227. [Google Scholar] [CrossRef] [PubMed]
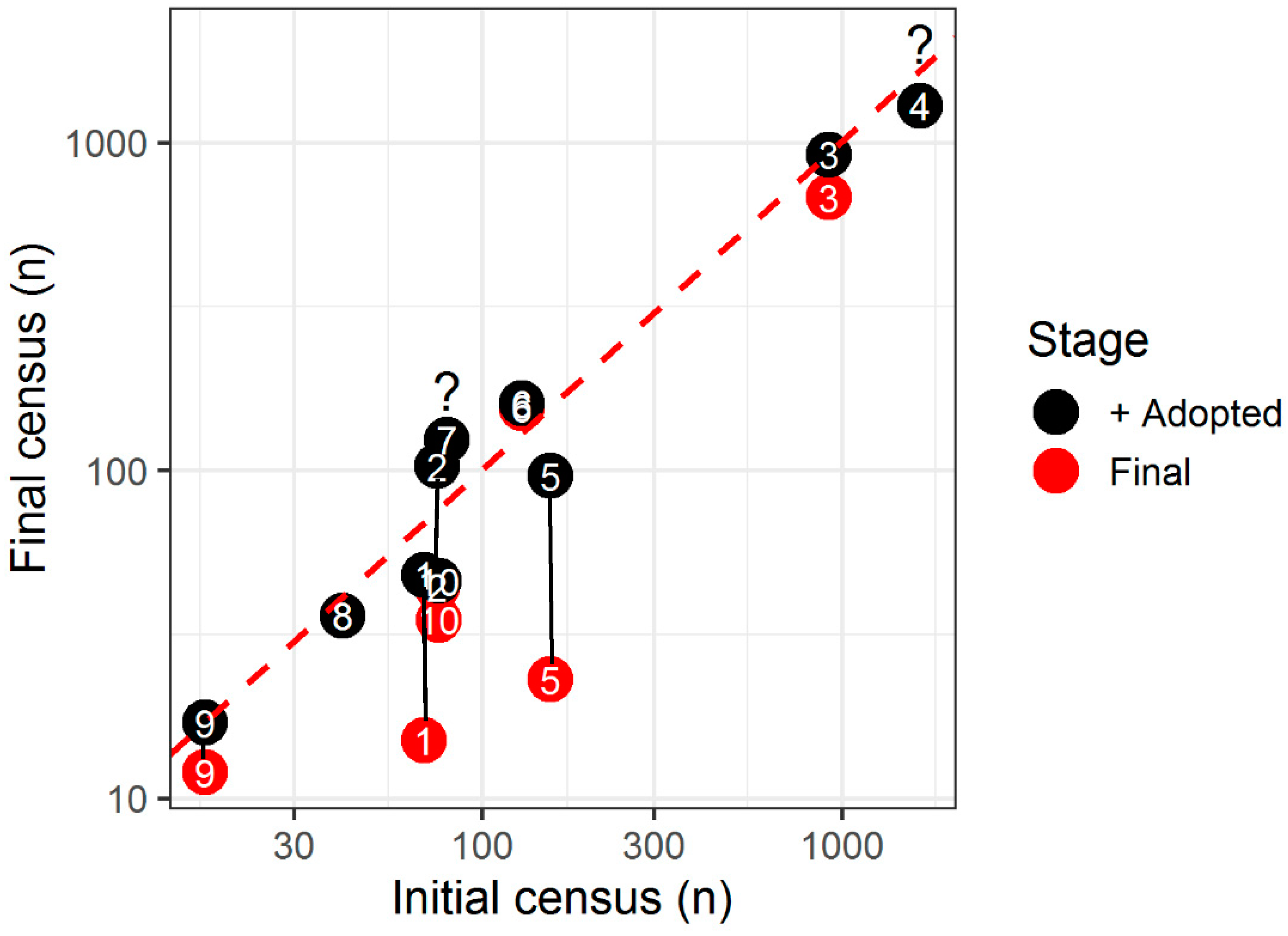



| Location | University of NSW, Australia | Chicago, USA | 12 Counties, Florida, USA | Rome, Italy | University of Central Florida Campus, USA | Brooklyn, New York, USA | Park Marina, Florida, USA | Hospital, Carville, Louisiana, USA | Regent’s Park, London, UK | Hospital, Cheshire, UK | Rural Cat Colonies, Quebec, Canada | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reference | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | |
| Study Length (y) | 9 | 4–10 | 1 | 2–6 | 11 | 2 | 1 | 3 | 1 | 2 | 1 | |
| Colony number | 1 | 20 | 132 | 103 | 11 | 2 | 2 | 1 | 2 | 1 | 10 | |
| Number of individual cats (n) | Initial census † | 69 | 75 | 920 | 1655 | 155 | 129 | 80 | 41 | 17 | 76 A | 143 B, median 13.5/colony |
| Neutered | 55 | 180 | 643 | 1424 | 155 | 185 | - | 38 | 13 | 42 | 132 B | |
| Adopted | 33 | 59 | 238 | - | 73 | 5 | - | 0 | 5 C | 11 | - | |
| Euthanized | 21 | 6 | - | - | 17 | 1 | 47 | 1 | 12 | - | ||
| Disappeared | 35 | 67 | 149 | - | 23 D | 0 | - | 5 | 1 E | 12 | - | |
| Dead | 15 | 13 | 151 | - | 10 | 0 | - | 8 | 0 | 7 | - | |
| Other | 3 | 0 | 0 | - | 9 | 0 | - | 0 | 1 | 0 | - | |
| Joined | 53 | - | 601 | 441 | - | 26 | 86 | 6 | 2 | - | - | |
| Final census † | 15 | 44 | 678 | 1293 | 23 | 155 | 124 | 36 | 12 | 35 | –, median 13.0/colony | |
| Overall num. response (%) | –78 | –41 | –26 | –22 | −79 | +20 | +55 | –12 | –29 | –54 | No change | |
| Excluding adoptions (%) | –71 | +175 | –1 | - | –72 | +25 | - | –12 | - | –46 | - | |
| Study Area | n | Roundworms (Nematoda) | Tapeworms (Cestoda) | Protozoa | Reference | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Necropsy/Scats † | Ancylostoma braziliense * | Ancylostoma tubaeformae * | Hookworm spp. * | Ollulanus tricuspis | Physaloptera spp. | Strongyloides spp. * | Toxascaris leonina * | Toxocara cati * | Toxocara spp. * | Uncinaria stenocephala | Diplopylidium nolleri | Dipylidium caninum * | Mesocestoides spp. | Joyeuxiella spp. | Spirometra erinacei * | Taenia taeniaeformis | Taenia hydatigena | Cystoisospora spp. * | Cryptosporidium spp. * | Giardia spp. * | Isospora spp. | Sarcocycstis spp. | |||
| Australia, national refuges | 491 | S | 3% | <1% | 5% | <1% | 3% | 3% | 3% | 10% | [130] | ||||||||||||||
| Mexico, Queretaro | 273 | N | 1% | 3% | <1% | 2% | 29% | 3% | [131] | ||||||||||||||||
| Portugal, Lisbon | 162 | N | 19% | 31% | 38% | 4% | 53% | 15% | 3% | 46% | 1% | [127] | |||||||||||||
| Portugal, Lisbon | 231 | S | 1% | 1% | 11% | 3% | 1% | 5% | [132] | ||||||||||||||||
| Spain, Barcelona | 50 | S | 4% | 22% | 4% | 6% | [133] | ||||||||||||||||||
| Spain, Canary Islands | 48 | N | 19% | 21% | 65% | 31% | [134] | ||||||||||||||||||
| Italy, Milan | 103 | S | 2% | 1% | 26% | 1% | 1% | 12% | [135] | ||||||||||||||||
| Greece, Thessaloniki | 215 | S | 12% | 18% | 40% | 8% | [136] | ||||||||||||||||||
| Denmark, Zealand region | 92 | N | 13% | 1% | 1% | 85% | 1% | 3% | 36% | [125] | |||||||||||||||
| Germany, Lower Saxony | 837 | S | 1% | 27% | 1% | 8% | [137] | ||||||||||||||||||
| Egypt, Alexandria | 170 | S | 1% | 1% | 8% | 8% | 19% | 1% | [138] | ||||||||||||||||
| Egypt, Kafrelsheikh | 113 | S | 4% | 5% | 9% | 5% | 22% | 2% | 2% | 1% | [139] | ||||||||||||||
| Iran, Ahvaz | 140 | S | 45% | 11% | 21% | 17% | [140] | ||||||||||||||||||
| Iran, Ahvaz | 52 | N | 4% | 8% | 29% | 23% | 13% | 8% | 10% | 2% | 24% | [141] | |||||||||||||
| Iran, Isfahan | 131 | N | 37% | 13% | 1% | 8% | 76% | 9% | [142] | ||||||||||||||||
| Iran, Kashan | 113 | N | 40% | 13% | 2% | 65% | 68% | 7% | 85% | 15% | 1% | 5% | 8% | [124] | |||||||||||
| Qatar, Doha | 568 | N | 15% | 5% | 0% | 1% | 74% | [126] | |||||||||||||||||
| UAE, Dubai | 240 | N | 9% | 1% | 1% | 3% | 37% | 66% | 17% | 0% | [143] | ||||||||||||||
| India, Aizawl Mizoram | 27 | N | 7% | 44% | 59% | 41% | 70% | [144] | |||||||||||||||||
| Malaysia, Kuala Lumpur | 241 | N | 31% | 35% | 0% | 3% | 8% | 24% | 12% | 5% | [145] | ||||||||||||||
| Malaysia, Georgetown | 102 | N | 23% | 20% | 1% | 0% | 14% | 37% | 2% | 3% | [145] | ||||||||||||||
| Malaysia, Kuantan | 100 | N | 39% | 33% | 1% | 0% | 17% | 42% | 5% | 7% | [145] | ||||||||||||||
| Malaysia, Malacca | 100 | N | 31% | 33% | 9% | 0% | 5% | 10% | 1% | 16% | [145] | ||||||||||||||
| Examples | Treatment | Requires Capture | Frequency of Treatment | Annual Cost Per Cat A | |
|---|---|---|---|---|---|
| Ectoparasites B | Fleas, Ticks C | Body spray | Y | Every 2 months D | $30/250ml |
| Fleas | Flea collar | Y | Each lasts 8 months | $40/collar | |
| Tablet | N * | Daily or Weekly E | $184 | ||
| Chew | N * | Monthly | $150 | ||
| Spot-on liquid | Y | Monthly | $129–182 | ||
| Fleas, Lice | Spot-on liquid | Y | Monthly | $146 | |
| G.I. Parasites B | Tapeworm | Tablet | N * | Every 3 months | $6 |
| Roundworms, hookworms, tapeworms | Tablet | N * | Every 3 months | $21 | |
| Roundworms, hookworms, tapeworms, lungworms | Spot-on liquid | Y | Monthly | $150 | |
| Parasite Combinations | Fleas, ear mites, hookworms, roundworms, lungworm, heartworm F | Spot-on liquid | Y | Monthly | $150 |
| Fleas, ear mites, mites, hookworms, roundworms, lice, heartworms F | Spot-on liquid | Y | Monthly | $150 | |
| Viruses G | 3-in-1 vaccine: Feline Enteritis, Feline Viral Rhinotracheitis, Feline Calicivirus H | Injection | Y | Annually | $14 |
| 4-in-1 vaccine: Feline Calicivirus, Feline Herpes Virus, FPV, Chlamydophillia felis H | Injection | Y | Annually | $16 | |
| 5-in-1 vaccine: Feline Enteritis, Feline Viral Rhinotracheitis, Feline Calicivirus, Chlamydophillia felis, FeLV H | Injection | Y | Annually | $21 | |
| FIV I | Injection | Y | Annually | $18 | |
| FeLV J | Injection | Y | Annually | $13 | |
| Rabies Virus K | – | – | – | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crawford, H.M.; Calver, M.C.; Fleming, P.A. A Case of Letting the Cat out of The Bag—Why Trap-Neuter-Return Is Not an Ethical Solution for Stray Cat (Felis catus) Management. Animals 2019, 9, 171. https://doi.org/10.3390/ani9040171
Crawford HM, Calver MC, Fleming PA. A Case of Letting the Cat out of The Bag—Why Trap-Neuter-Return Is Not an Ethical Solution for Stray Cat (Felis catus) Management. Animals. 2019; 9(4):171. https://doi.org/10.3390/ani9040171
Chicago/Turabian StyleCrawford, Heather M., Michael C. Calver, and Patricia A. Fleming. 2019. "A Case of Letting the Cat out of The Bag—Why Trap-Neuter-Return Is Not an Ethical Solution for Stray Cat (Felis catus) Management" Animals 9, no. 4: 171. https://doi.org/10.3390/ani9040171
APA StyleCrawford, H. M., Calver, M. C., & Fleming, P. A. (2019). A Case of Letting the Cat out of The Bag—Why Trap-Neuter-Return Is Not an Ethical Solution for Stray Cat (Felis catus) Management. Animals, 9(4), 171. https://doi.org/10.3390/ani9040171





